Abstract
Introduction
Bone morphogenetic protein-2 (BMP-2) has high potential for bone formation, but its in vivo effects are unpredictable due to the short life time. This study was designed to evaluate the effects of recombinant human (rh) BMP-2 suspended in fibrin on bone formation during distraction osteogenesis (DO) in rabbits.
Material and methods
The in vitro release kinetics of rhBMP-2 suspended in fibrin was tested using an enzyme-linked immunosorbent assay. Unilateral tibial lengthening for 10 mm was achieved in 48 rabbits. At the completion of osteodistraction, vehicle, fibrin, rhBMP-2 or rhBMP-2 suspended in fibrin (rhBMP-2 + fibrin) was injected into the center of the lengthened gap, with 12 animals in each group. Eight weeks later, the distracted callus was examined by histology, micro-CT and biomechanical testing. Radiographs of the distracted tibiae were taken at both 4 and 8 weeks after drug treatment.
Results
It was found that fibrin prolonged the life span of rhBMP-2 in vitro with sustained release during 17 days. The rhBMP-2 + fibrin treated animals showed the best results in bone mineral density, bone volume fraction, cortical bone thickness by micro-CT evaluation and mechanical properties by the three-point bending test when compared to the other groups (p < 0.05). In histological images, rhBMP-2 + fibrin treatment showed increased callus formation and better gap bridging compared to the other groups.
Conclusions
The results of this study suggest that fibrin holds promise to be a good carrier of rhBMP-2, and rhBMP-2 suspended in fibrin showed a stronger promoting effect on bone formation during DO in rabbits.
Keywords: bone morphogenic protein-2, distraction osteogenesis, fibrin, bone formation, release kinetics
Introduction
Distraction osteogenesis (DO), also called callus distraction, callotasis, or osteodistraction, is a surgical process used to reconstruct skeletal deformities and lengthen the long bones of the body. A corticotomy is used to fracture the bone into two segments, and the two ends of the bone are gradually moved apart during the distraction phase, allowing new bone to form in the gap [1, 2]. Distraction osteogenesis has the benefit of simultaneously increasing bone length and the volume of surrounding soft tissues; thus this technique is widely used in the field of orthopedics and correction of craniofacial deformities [3, 4]. However, the overall treatment time of bone lengthening is still long and can be a cause of considerable morbidity for the patients.
Various approaches have been tested to promote bone formation during DO, such as mechanical compression, electrical and electromagnetic stimulation, low velocity ultrasound, and certain growth factors or cytokines [5–8]. Although some of these studies have yielded promising results, few of these approaches have been shown to be efficient in humans. Bone morphogenic proteins (BMPs) are among the most potent of the osteoinductive factors that play a crucial role in the process of embryological bone formation, fracture repair, and bone regeneration during DO [9–11]. Local injection of recombinant human (rh) BMP-2 has been shown to accelerate bone formation in a number of distraction models [12–14]. However, optimal bone formation appears to require a suitable carrier matrix because the BMP-2 administered in solution does not retain significant activity in vivo due to its short half-life.
Although numerous carriers have been shown to be compatible with the osteoinductive activity of rhBMPs [15–18], the use of natural biomaterials may be an attractive concept. Fibrin glue, a composite of fibrinogen and thrombin, is a physiologically relevant matrix. Its principal component, fibrin, is a natural wound healing matrix that allows cells to invade and interact in a wound and might provide a suitable matrix for bone healing [19, 20]. Previous studies have reported that fibrin glue is a potentially suitable biological vehicle for the transfer of cells and for the application of growth factors because of its biocompatibility, biodegradability and binding capacity to cells [21–23].
Here, we report a novel controlled-release system of rhBMP-2 using fibrin to accelerate bone formation in rabbit tibia lengthening. The in vitro release kinetics of rhBMP-2 was tested using the enzyme-linked immunosorbent assay (ELISA), and the in vivo effects of this delivery system were examined by radiology, histology, micro-computed tomography (micro-CT), and biomechanical testing.
Material and methods
Preparation of fibrin glue
Fibrin glue was prepared using a Tissucols kit (Baxter AG, Vienna, Austria) according to the manufacturer's instructions, by ejecting equal volumes of a fibrinogen solution (25 µl) and two respective thrombin solutions (50 or 500 IU) through a two-channel syringe. Previous studies indicated that 100 µg of rhBMP-2 significantly increased callus formation. Thus, the dose of 100 µg of rhBMP-2 (R&D Systems, Minneapolis, USA) for each animal was used in this study. The rhBMP-2 was added to the thrombin component, mixed and then pipetted into a sterile syringe before addition of the fibrinogen component.
In vitro release test of rhBMP-2
Fibrin glue scaffolds with suspended rhBMP-2 (100 µg) were separately placed into microcentrifuge tubes with 1 ml of PBS and incubated at 37°C. After 24 h, the release medium was replaced with 1 ml of fresh PBS to maintain the continuous release of rhBMP-2 from the scaffold. The amount of rhBMP-2 released was quantified at room temperature using ELISA (BMP-2 Quantikine enzyme-linked immunosorbent assay kit, R&D Systems, Minneapolis, USA) according to the manufacturer's instructions. Such procedures were performed every 24 h in triplicate for over 21 days, and the mean value was used to graph the total cumulative release of rhBMP-2.
Animals and study design
Forty-eight New Zealand white rabbits (6 months old, 2.5–3.2 kg) were included. The animal study was approved by the Animal Ethics Committee of Sichuan University in accordance with Guidelines for the Care and Use of Laboratory Animals. The animals were kept in separate cages with food and water under standardized environmental conditions with 12-h light/dark cycles, and allowed to move freely throughout the experimental period. Seven days after osteotomy surgery, the distraction procedure was started at a rate of 0.5 mm every 12 h (1.0 mm/day) for 10 days, and the actual lengthening of 10 mm was achieved. At the time of completion of distraction, all rabbits were randomly divided into 4 groups with 12 animals in each group according to the agents injected at the center of the lengthened gap: (a) control group with vehicle; (b) fibrin group with 50 µl of fibrin glue; (c) rhBMP-2 group with 100 µg of rhBMP-2; (d): rhBMP-2 + fibrin group with 100 µg of rhBMP-2 suspended in fibrin. All rabbits were sacrificed by injection of sodium pentobarbitone 8 weeks after drug treatment.
Surgery procedure
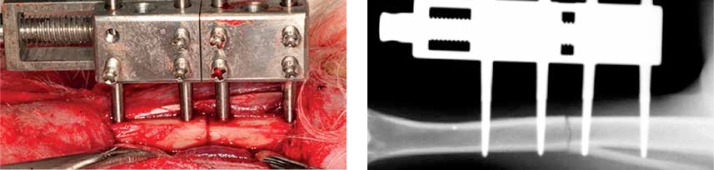
All rabbits were anaesthetized by intravenous injection of sodium pentobarbital and sodium ketamine hydrochloride at doses of 20 mg/kg and 4 mg/kg body weight, respectively. Each animal was placed in the dorsal position, and a 2.0 cm incision was made along the medial side of the right hind leg to expose the tibia. The periosteum was then carefully elevated circumferentially. After a self-constructed external fixator was applied unilaterally to the tibia with four self-tapping screws, subperiosteal osteotomy was performed between the middle two screws using a fine wire saw at a level immediately below the area attached to the fibula (Figure 1). The periosteum, muscle, and skin were repositioned and closed with 3/0 silk sutures. The external fixator remained in place until necropsy.
Figure 1.
The picture taken during surgery and the radiograph immediately after the operation showed that the custom-made distractor was fixated on the rabbit tibia
Radiology
Four and 8 weeks after drug treatment, radiographs of the distracted tibiae in standard antero-posterior position and lateral projections were taken using RADspeed at settings of 40 kilovolts and 1.2 milliamps. Quantitative analysis was performed using the pixel value ratio (PVR) in relation to original bone by a picture archiving communication system (PACS, INFINITT Co. Ltd, Seoul Korea) according to previous studies [24, 25].
Micro-CT examination
Eight weeks after drug treatment, the regenerated bone tissue and the neighboring bone of the distracted tibia (6 specimens per group) were examined morphologically and quantitatively by a micro-CT 80 scanner (Scano Medical AG, Bassersdorf, Switzerland). Specimens were kept in 10% formalin to avoid drying during the scanning process. After three-dimensional (3D) reconstruction, the volume of interest (VOI) was defined as the 10 mm long distracted callus. The threshold was adjusted empirically to visualize the defect site and the mineralization and was then kept constant for every sample (375–1034 Hounsfield units for all reconstructions). Bone mineral density (BMD), bone mineral content (BMC), bone volume fraction (the ratio between bone volume and total volume, BV/TV) and cortical bone thickness (CBT) within the VOI of the regenerated bone were measured.
Histology
After micro-CT examination, specimens (6 specimens per group) were fixed in 10% neutral formalin for 5 days and decalcified in 15% ethylene diamine tetraacetic acid (EDTA) for 6 weeks at 4°C. After dehydration, the specimens were embedded in paraffin and 6-µm coronal sections were stained with hematoxylin and eosin (H + E) for histological analysis.
Biomechanical testing
For analysis of the biomechanical properties of the distracted tibiae, soft tissue was dissected carefully and a three-point bending test was performed immediately using a commercial materials testing system (Instron 4302; Instron, Norwood, MA, USA). The contralateral tibiae in the control group were also tested as a normal control. The load-displacement curve was recorded during the downward compression, and the ultimate load at failure (N; maximum force that the specimen sustained) was used for statistical analysis (6 specimens per group).
Statistical analysis
All data are expressed as mean ± SD (standard deviation). One-way analysis of variance (ANOVA) and the Student-Newman-Keuls (SNK) test were conducted to compare differences between groups using SPSS 12.0 (SPSS, Chicago, IL, USA). Differences were considered to be significant when p < 0.05.
Results
In vitro release of rhBMP-2
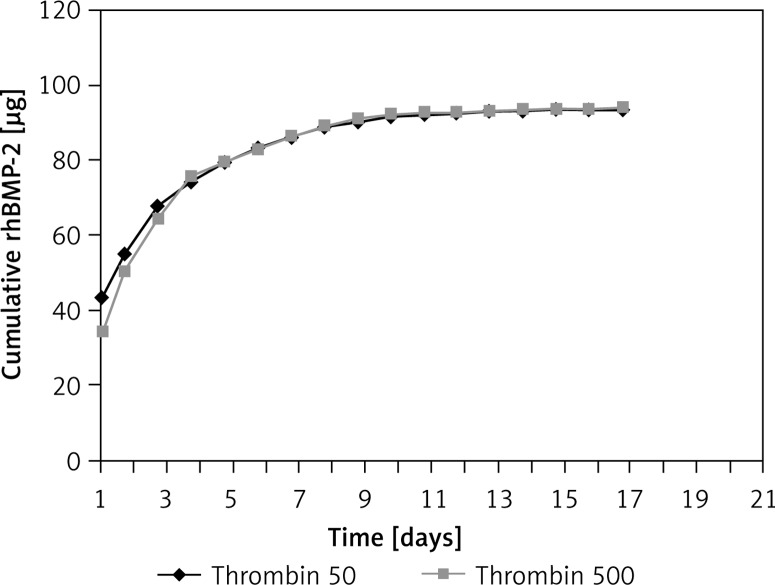
Both thrombin 50 and 500 components were used to evaluate the release kinetics of rhBMP-2 from the fibrin glue. Similarities were observed in both groups and the released rhBMP-2 followed the same pattern (Figure 2). The cumulative level of rhBMP-2 released from the fibrin glue increased gradually over time. An initial burst release of over 70% rhBMP-2 was observed within the first 3 days, followed by sustained release detectable until day 17. Thus the thrombin 50 scaffolds were used for the in vivo experiment.
Figure 2.
In vitro analysis of release kinetics of rhBMP-2 from fibrin scaffolds revealed a sharp initial burst of over 70% total rhBMP-2 released in the first 3 days followed by a sustained release of up to 95% over 17 days. There was no significant difference between thrombin 50 and 500 groups
Radiological findings
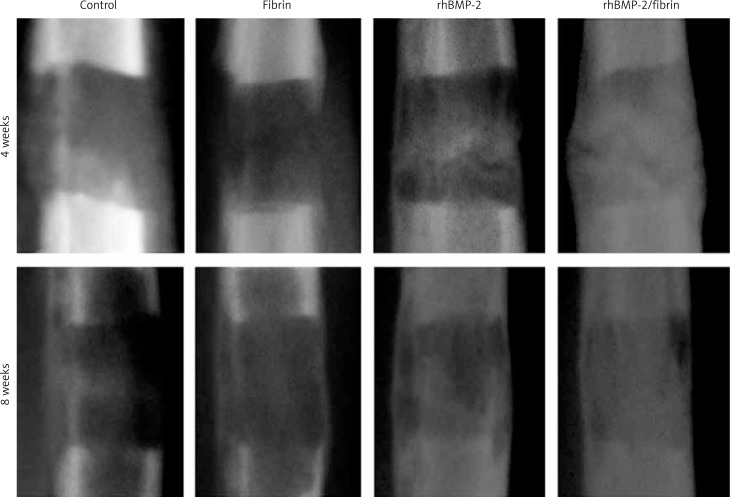
Radiological examination showed consecutive phases of new bone formation and remodeling in the distracted gap in all groups (Figure 3). For group comparison, there was no discernable difference of the radiodensity between the control and fibrin glue group. In the rhBMP-2 group, radiodensity of the distracted gap was higher compared to the control and fibrin glue group, suggesting increased mineralization. The highest radiodensity and best cortical bone bridging of the distracted gap were observed in the rhBMP-2 + fibrin group. Quantitative results of PVR in relation to original bone of the distracted tibiae using the PACS system are summarized in Table I. Compared to rhBMP-2 alone, rhBMP-2 + fibrin significantly increased PVR values by 71.1% at 4 weeks and 37.5% at 8 weeks after drug treatment (p < 0.05).
Figure 3.
Radiographic changes of the distracted gap between groups 4 and 8 weeks after drug treatment. Cortical continuity and bony union were achieved in all experimental groups 8 weeks after the completion of distraction, while bone regeneration within the distracted callus appeared to arise much earlier and faster in the fibrin + rhBMP-2 group compared to others
Table I.
Quantitative results of pixel value ratio (PVR) in relation to original bone of the distracted tibiae using a picture archiving communication system
| Time [weeks] | Control | Fibrin | rhBMP-2 | rhBMP-2/Fibrin |
|---|---|---|---|---|
| 4 | 0.25 ±0.04 | 0.23 ±0.05 | 0.38 ±0.07* | 0.65 ±0.11# |
| 8 | 0.45 ±0.07 | 0.48 ±0.08 | 0.64 ±0.12* | 0.88 ±0.13# |
P-value < 0.05 vs. control and fibrin group
p < 0.05 vs. rhBMP-2 group (by one-way ANOVA and Student-Newman-Keuls test, n = 6 specimens in each group).
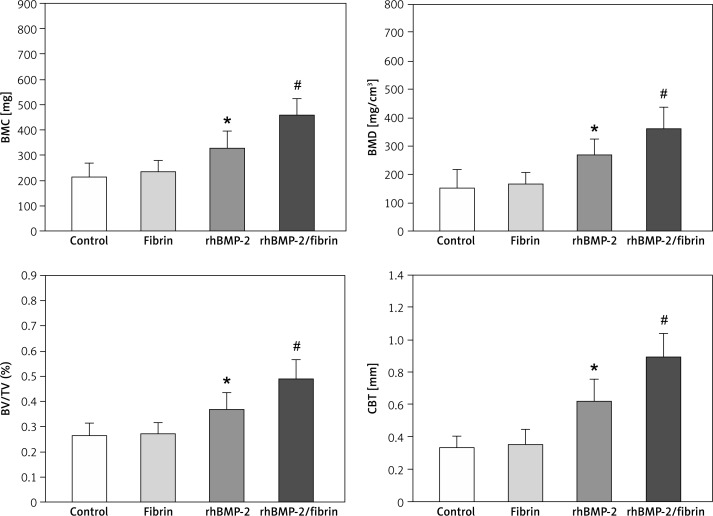
Micro-CT examination
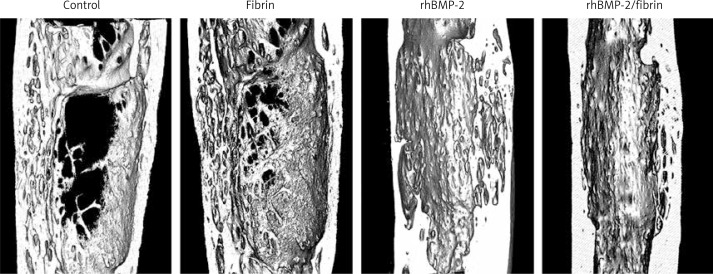
Micro-CT images are presented in Figure 4 and the quantitative results in Figure 5. Consistent with radiological findings, the lowest values of BMC, BMD, BV/TV and CBT were observed in the control and fibrin groups. The rhBMP-2 group showed better results than those of the control and fibrin groups. Images from the rhBMP-2 + fibrin group revealed better cortical bone formation and bony bridging of the distracted gap than others, resulting in the best results of BMC, BMD, BV/TV and CBT between groups in quantitative analysis.
Figure 4.
The 3D micro-CT images 8 weeks after drug treatment revealed a superior quality of ossification of the distracted gap in the rabbits treated with rhBMP-2 + fibrin compared to others. Cross-sectional images at the center of the lengthened segment indicated that the rhBMP-2 + fibrin group showed the thickest cortical bone
Figure 5.
Results of quantitative micro-CT analysis of the distracted callus 8 weeks after drug treatment
Data are expressed as mean ± SD. *p < 0.05 vs. control and fibrin group, and #p < 0.05 vs. rhBMP-2 group (by one-way ANOVA and Student-Newman-Keuls test, n = 6 specimens in each group). BMC – bone mineral content, BMD – bone mineral density, BV/TV (%) – bone volume fraction, CBT – cortical bone thickness.
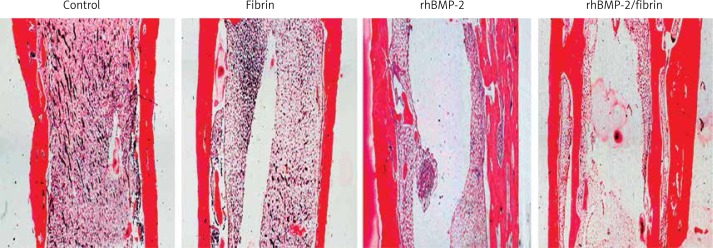
Histology
Histological images of the distracted tibiae are presented in Figure 6. Consistent with the radiological and micro-CT findings, the fibrin group produced no significant effect on callus formation compared to controls. Both rhBMP-2 treated groups exhibited thicker cortical bone than control and fibrin groups, but the rhBMP-2 + fibrin group showed the best results of cortical bone formation and bony bridging of the distracted gap.
Figure 6.
Longitudinal sections of the distracted tibiae prepared at 8 weeks after drug treatment (H + E staining, 40×)
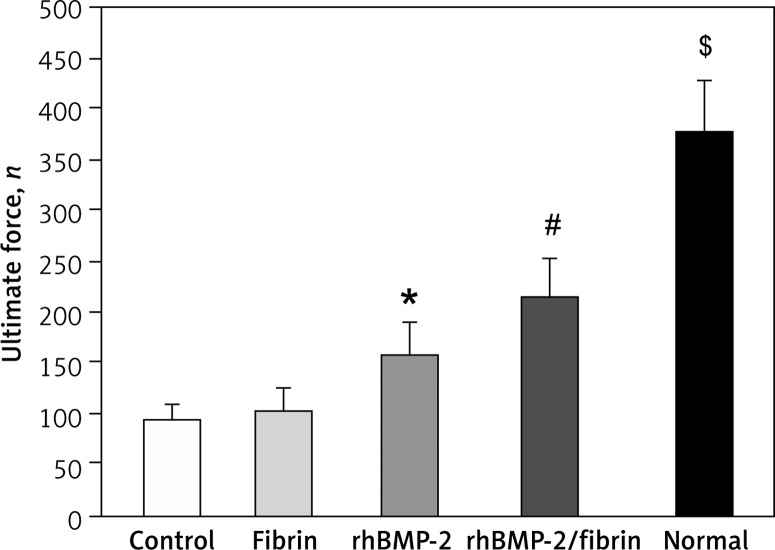
Biomechanical testing
The results of the three-point bending test are shown in Figure 7. There was no significant difference of the ultimate force of the lengthened tibiae between control and fibrin groups. Although the ultimate force of lengthened tibiae was significantly higher in both rhBMP-2 treated groups compared to controls, the rhBMP-2 + fibrin group induced the highest increase in the ultimate force, indicating the efficiency of rhBMP-2 + fibrin in enhancing bone formation at the distracted gap. However, the healing strength of distracted tibiae from the rhBMP-2 + fibrin group was still far from that of the contralateral normal tibiae in the control group.
Figure 7.
Ultimate force of the lengthened segments and the contralateral tibiae in the control group 8 weeks after drug treatment were measured by three-point bending test
Data are expressed as mean ± SD; *p < 0.05 vs. control and fibrin group, #p < 0.05 vs. rhBMP-2 group, and $p < 0.05 vs. rhBMP-2 + fibrin group (by one-way ANOVA and Student-Newman-Keuls test, n = 6 specimens in each group).
Discussion
BMP-2 acts primarily as a differentiation factor for bone and cartilage precursor cells, and has been shown to induce bone formation and healing of bony defects in a variety of animal experiments. The effect of local application of BMP-2 on bone formation during DO has been examined in different models. In all these studies, BMP-2 significantly increased bone formation within the lengthening segment, attributable mostly to the potent osteogenic ability of this mitogen. However, optimal bone formation is not achieved due to the short half-life of the BMP-2 administered in solution. Although previous studies reported that a higher dose or continuous injection of growth factor was more effective in accelerating bone formation [26–28], there are concerns over the risk of infection, long-term safety and associated costs. Incorporation of BMP-2 into suitable carriers can readily solve these problems because the physical binding of BMP-2 to the carrier material delays their rapid diffusion and the degradation by tissue proteinase, thereby considerably increasing its osteoinductive effect.
Several delivery systems have been developed for the sustained release of BMP-2 in bone defect and fracture healing, but only a few experiments have investigated the effects of BMP-2 in combination with carriers on bone healing in DO. Li et al. found that rhBMP-2 had positive effects on bone formation in lengthening the tibia when applied with absorbable collagen sponge (ACS) [14]. However, no difference was found between the rhBMP-2/buffer and rhBMP-2/ACS groups, suggesting that ACS may not enable sustaining a steady rhBMP-2 level in the lengthened segment. Another important problem is that it requires a second surgical procedure for implantation of collagen sponge. In another study it was found that intermittent rhBMP-2-containing chitosan hydrogel infusion by a drug-release distractor improved radiologic and histological parameters of DO when compared with a single-dose BMP-2-chitosan-injected group [28]. Conclusions about their results, however, are also difficult to make because of the lack of a control group treated with rhBMP-2 alone.
Fibrin glue is an excellent tissue sealant in clinical application and has received increasing interest as a reservoir to deliver growth factors in tissue engineering [19–22]. In this study in vitro release testing revealed sustained release of rhBMP-2 from the fibrin glue at both concentrations of thrombin. The fibrin glue eventually used in animals was composed of a low concentration of thrombin (thrombin 50), as it was difficult to incorporate the rhBMP-2 into the polymerizing glue using higher concentrations (thrombin 500). In a previous study fibrin composites laden with 25 µg IGF-I were observed to release effective concentrations of ligand for as long as 22 days in vitro [29]. Another study suggested that grafted fibrin may last at least 2 weeks in situ [30]. Similarly, in this study it was found that rhBMP-2 was released rapidly in the first 3 days from the fibrin glue with 70% of the total rhBMP-2, followed by sustained release up to 95% until day 17. When applied in vivo, this sharp burst may provide an initial stimulus for osteogenic cells and thus an accelerated onset of bone formation.
Previous studies showed that in the cellular environment fibrin specifically binds to a variety of proteins, including fibronectin, albumin, FGF2, and VEGF, and favors migration and proliferation of undifferentiated mesenchymal cells [31–33]. A previous study found that fibrin glue alone was effective as a scaffold to stimulate bone regeneration in a critical size rat cranial defect [19]. However, the biological mechanism regulating bone regeneration in bone tissue engineering is different from that in DO. In this study no additional benefit for bone formation was observed in the group of fibrin glue alone compared with the empty control, suggesting that the possible role of fibrin glue in DO may be a growth factor carrier to achieve long-term persistence and thus maintain bioactive effects of rhBMP-2, but has no intrinsic effect on acceleration of bone formation by itself.
The consolidation phase is a key period for bone formation during DO, and delivery of exogenous BMP-2 at this stage will target osteogenic cells within the callus and may enhance its mineralization. In agreement with other findings [14, 26, 28], our results clearly demonstrated accelerated bone ossification within the lengthened segment when rhBMP-2 was present. Further comparison between two rhBMP-2-treated groups showed that 8 weeks after the drug treatment, rabbits in the rhBMP-2/fibrin group showed a superior quality of ossification in radiological and histological appearance as well as higher values in micro-CT and mechanical testing when compared with the group of rhBMP-2 alone, indicating that incorporation of rhBMP-2 into fibrin contributed to and enhanced the therapeutic effects of rhBMP-2. Previous studies have shown that the effect of single use of rhBMP-2 on bone healing in DO is dose-dependent [26, 28]. Although the amount of rhBMP-2 injected in both rhBMP-2 treated groups was equal, rhBMP-2 without a carrier tends to diffuse easily and degrade by proteinase, and the effective concentration and exact dose of rhBMP-2 at the injection site may decrease rapidly to below the therapeutic level. The presence of fibrin glue, however, could protect rhBMP-2 from enzymatic degradation in vivo and keep it at a higher level by the sustained release of rhBMP-2 as a reservoir.
In conclusion, the results of this study indicated that treatment with rhBMP-2 suspended in fibrin accelerated bone regeneration within the distracted gap of rabbit tibia after an 8-week observation period, resulting in a stronger effect on callus formation and bony bridging of the distracted gap than rhBMP-2 alone. It suggests that fibrin is a potential carrier of rhBMP-2 to enhance its life-time and anabolic effect on bone formation.
Acknowledgments
Yunfeng Li and Rui Li contributed equally to this work.
This study was supported by a grant from the National Natural Science Foundation of China (No. 81300858).
Conflict of interest
Authors declare no conflict of interest.
References
- 1.McCarthy JG, Katzen JT, Hopper R, Grayson BH. The first decade of mandibular distraction: lessons we have learned. Plast Reconstr Surg. 2002;110:1704–13. doi: 10.1097/01.PRS.0000036260.60746.1B. [DOI] [PubMed] [Google Scholar]
- 2.Takahashi M, Yukata K, Matsui Y, Abbaspour A, Takata S, Yasui N. Bisphosphonate modulates morphological and mechanical properties in distraction osteogenesis through inhibition of bone resorption. Bone. 2006;39:573–81. doi: 10.1016/j.bone.2006.03.010. [DOI] [PubMed] [Google Scholar]
- 3.Saleh M, Royston S. Management of nonunion of fractures by distraction with correction of angulation and shortening. J Bone Joint Surg Br. 1996;78:105–9. [PubMed] [Google Scholar]
- 4.Aronson J. Limb-lengthening, skeletal reconstruction, and bone transport with the Ilizarov method. J Bone Joint Surg Am. 1997;79:1243–58. doi: 10.2106/00004623-199708000-00019. [DOI] [PubMed] [Google Scholar]
- 5.El-Bialy TH, Elgazzar RF, Megahed EE, Royston TJ. Effects of ultrasound modes on mandibular osteodistraction. J Dent Res. 2008;87:953–7. doi: 10.1177/154405910808701018. [DOI] [PubMed] [Google Scholar]
- 6.El-Hakim IE, Azim AM, El-Hassan MF, Maree SM. Preliminary investigation into the effects of electrical stimulation on mandibular distraction osteogenesis in goats. Int J Oral Maxillofac Surg. 2004;33:42–7. doi: 10.1054/ijom.2003.0445. [DOI] [PubMed] [Google Scholar]
- 7.Aronson J. Modulation of distraction osteogenesis in the aged rat by fibroblast growth factor. Clin Orthop Relat Res. 2004;425:264–83. doi: 10.1097/01.blo.0000138186.53426.f9. [DOI] [PubMed] [Google Scholar]
- 8.Li G, Ryaby JT, Carney DH, Wang H. Bone formation is enhanced by thrombin-related peptide TP508 during distraction osteogenesis. J Orthop Res. 2005;23:196–202. doi: 10.1016/j.orthres.2004.05.006. [DOI] [PubMed] [Google Scholar]
- 9.Haque T, Hamade F, Alam N, et al. Characterizing the BMP pathway in a wild type mouse model of distraction osteogenesis. Bone. 2008;42:1144–53. doi: 10.1016/j.bone.2008.01.028. [DOI] [PubMed] [Google Scholar]
- 10.Yang X, Gong P, Lin Y, et al. Cyclic tensile stretch modulates osteogenic differentiation of adipose-derived stem cells via the BMP-2 pathway. Arch Med Sci. 2010;6:152–9. doi: 10.5114/aoms.2010.13886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Rakian A, Yang WC, Gluhak-Heinrich J, et al. Bone morphogenetic protein-2 gene controls tooth root development in coordination with formation of the periodontium. Int J Oral Sci. 2013;5:75–84. doi: 10.1038/ijos.2013.41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Issa JP, do Nascimento C, Lamano T, Iyomasa MM, Sebald W, de Albuquerque RF., Jr Effect of recombinant human bone morphogenetic protein-2 on bone formation in the acute distraction osteogenesis of rat mandibles. Clin Oral Implants Res. 2009;20:1286–92. doi: 10.1111/j.1600-0501.2009.01799.x. [DOI] [PubMed] [Google Scholar]
- 13.Ashinoff RL, Cetrulo CL, Jr, Galiano RD, et al. Bone morphogenic protein-2 gene therapy for mandibular distraction osteogenesis. Ann Plast Surg. 2004;52:585–90. doi: 10.1097/01.sap.0000123023.28874.1e. discussion 591. [DOI] [PubMed] [Google Scholar]
- 14.Li G, Bouxsein ML, Luppen C, et al. Bone consolidation is enhanced by rhBMP-2 in a rabbit model of distraction osteogenesis. J Orthop Res. 2002;20:779–88. doi: 10.1016/S0736-0266(01)00166-8. [DOI] [PubMed] [Google Scholar]
- 15.Miyazaki M, Morishita Y, He W, et al. A porcine collagen-derived matrix as a carrier for recombinant human bone morphogenetic protein-2 enhances spinal fusion in rats. Spine J. 2009;9:22–30. doi: 10.1016/j.spinee.2008.08.009. [DOI] [PubMed] [Google Scholar]
- 16.Chung YI, Ahn KM, Jeon SH, Lee SY, Lee JH, Tae G. Enhanced bone regeneration with BMP-2 loaded functional nanoparticle-hydrogel complex. J Control Release. 2007;121:91–9. doi: 10.1016/j.jconrel.2007.05.029. [DOI] [PubMed] [Google Scholar]
- 17.Hosseinkhani H, Hosseinkhani M, Khademhosseini A, Kobayashi H. Bone regeneration through controlled release of bone morphogenetic protein-2 from 3-D tissue engineered nano-scaffold. J Control Release. 2007;117:380–6. doi: 10.1016/j.jconrel.2006.11.018. [DOI] [PubMed] [Google Scholar]
- 18.Wildemann B, Kandziora F, Krummrey G, et al. Local and controlled release of growth factors (combination of IGF-I and TGF-beta I, and BMP-2 alone) from a polylactide coating of titanium implants does not lead to ectopic bone formation in sheep muscle. J Control Release. 2004;95:249–56. doi: 10.1016/j.jconrel.2003.11.014. [DOI] [PubMed] [Google Scholar]
- 19.Woodruff MA, Rath SN, Susanto E, et al. Sustained release and osteogenic potential of heparan sulfate-doped fibrin glue scaffolds within a rat cranial model. J Mol Histol. 2007;38:425–33. doi: 10.1007/s10735-007-9137-y. [DOI] [PubMed] [Google Scholar]
- 20.Chudek J, Kowalczyk A, Kowalczyk AK, Kwiatkowska J, Raczak G, Kozłowski D. Quality of life (QOL) evaluation after acute coronary syndrome with simultaneous clopidogrel treatment. Arch Med Sci. 2014;10:33–8. doi: 10.5114/aoms.2013.38708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Pandit AS, Wilson DJ, Feldman DS. Fibrin scaffold as an effective vehicle for the delivery of acidic fibroblast growth factor (FGF-1) J Biomater Appl. 2000;14:229–42. doi: 10.1177/088532820001400303. [DOI] [PubMed] [Google Scholar]
- 22.Taylor SJ, Sakiyama-Elbert SE. Effect of controlled delivery of neurotrophin-3 from fibrin on spinal cord injury in a long term model. J Control Release. 2006;116:204–10. doi: 10.1016/j.jconrel.2006.07.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Gorodetsky R, Peylan-Ramu N, Reshef A, Gaberman E, Levdansky L, Marx G. Interactions of carboplatin with fibrin(ogen), implications for local slow release chemotherapy. J Control Release. 2005;102:235–45. doi: 10.1016/j.jconrel.2004.08.029. [DOI] [PubMed] [Google Scholar]
- 24.Singh S, Song HR, Venkatesh KP, et al. Analysis of callus pattern of tibia lengthening in achondroplasia and a novel method of regeneration assessment using pixel values. Skeletal Radiol. 2010;39:261–6. doi: 10.1007/s00256-009-0703-0. [DOI] [PubMed] [Google Scholar]
- 25.Song SH, Sinha S, Kim TY, Park YE, Kim SJ, Song HR. Analysis of corticalization using the pixel value ratio for fixator removal in tibial lengthening. J Orthop Sci. 2011;16:177–83. doi: 10.1007/s00776-011-0036-4. [DOI] [PubMed] [Google Scholar]
- 26.Zheng LW, Wong MC, Rabie AB, Cheung LK. Evaluation of recombinant human bone morphogenetic protein-2 in mandibular distraction osteogenesis in rabbits: effect of dosage and number of doses on formation of bone. Br J Oral Maxillofac Surg. 2006;44:487–94. doi: 10.1016/j.bjoms.2005.09.005. [DOI] [PubMed] [Google Scholar]
- 27.Abbaspour A, Takata S, Sairyo K, Katoh S, Yukata K, Yasui N. Continuous local infusion of fibroblast growth factor-2 enhances consolidation of the bone segment lengthened by distraction osteogenesis in rabbit experiment. Bone. 2008;42:98–106. doi: 10.1016/j.bone.2007.08.031. [DOI] [PubMed] [Google Scholar]
- 28.Konaş E, Emin Mavili M, Korkusuz P, Demir D, Oner F, Canter HI. Acceleration of distraction osteogenesis with drug-releasing distractor. J Craniofac Surg. 2009;20:2041–8. doi: 10.1097/SCS.0b013e3181be8754. [DOI] [PubMed] [Google Scholar]
- 29.Foley RL, Nixon AJ. Insulin-like growth factor I peptide elution profiles from fibrin polymers determined by use of high-performance liquid chromatography. Am J Vet Res. 1997;58:1431–5. [PubMed] [Google Scholar]
- 30.Brittberg M, Sjögren-Jansson E, Lindahl A, Peterson L. Influence of fibrin sealant (Tisseel) on osteochondral defect repair in the rabbit knee. Biomaterials. 1997;18:235–42. doi: 10.1016/s0142-9612(96)00117-2. [DOI] [PubMed] [Google Scholar]
- 31.Fakhry A, Ratisoontorn C, Vedhachalam C, et al. Effects of FGF-2/-9 in calvarial bone cell cultures: differentiation stage-dependent mitogenic effect, inverse regulation of BMP-2 and noggin, and enhancement of osteogenic potential. Bone. 2005;36:254–66. doi: 10.1016/j.bone.2004.10.003. [DOI] [PubMed] [Google Scholar]
- 32.Otto F, Thornell AP, Crompton T, et al. Cbfa1, a candidate gene for cleidocranial dysplasia syndrome, is essential for osteoblast differentiation and bone development. Cell. 1997;89:765–71. doi: 10.1016/s0092-8674(00)80259-7. [DOI] [PubMed] [Google Scholar]
- 33.Choi KY, Kim HJ, Lee MH, et al. Runx2 regulates FGF2-induced Bmp2 expression during cranial bone development. Dev Dyn. 2005;233:115–21. doi: 10.1002/dvdy.20323. [DOI] [PubMed] [Google Scholar]