Abstract
ATPase inhibitory factor 1 (IF1), an inhibitor of the mitochondrial H+-adenosine triphosphate (ATP) synthase, is putatively involved in tumor progression. This study aimed to evaluate the expression levels of IF1 in non-small cell lung cancer (NSCLC) and the prognostic value for the patients. IF1 protein expression levels were detected in 149 cases of NSCLC by using immunohistochemistry. Kaplan-Meier analysis showed that NSCLC patients with high expression of IF1 possessed poorer outcome than those with low expression of IF1 (P=0.007). Moreover, IF1 was also prognostic in the patients with early stages (stage I/II) (P=0.042) and low grade (grade I/II) (P=0.002). Multivariable Cox-regression analysis showed that high expression of IF1 (HR=1.67, P=0.034), tumor size (HR=1.79, P=0.001), and lymph node metastasis (HR=2.66, P=0.000) were independent indicators for NSCLC patients. In conclusion, our study demonstrated that elevated expression of IF1 may associated with lymph node metastasis of NSCLC and served as an independent prognostic and recurrent indicator for the patients.
Keywords: IF1, non-small cell lung cancer, lymph node metastasis, prognosis
Introduction
Lung cancer continues to be the most common causes of cancer-related deaths worldwide [1]. Patients with non-small cell lung cancer (NSCLC), which accounts for more than 80% of lung cancer, carry a poor clinical outcome with 5-years survival rate of 10%-15% [2]. According to the origin, NSCLC can be further divided into squamous cell carcinoma (SCC), lung adenocarcinoma (LAC), and large-cell carcinoma (LCC). Despite various traditional and novel therapies regimes were improved and developed, the overall survival of patients remained poor. Most patients were diagnosed in advanced stages and therefore missed the best opportunity to be cured. An integrated understanding of the biochemical signaling molecules and genetic factors of NSCLC could provide potential prognostic parameter and appropriate therapeutic targets [3-5].
In animals and plants, the mitochondrial F1F0-ATPsynthase is molecularly regulated by an endogenous, nuclear encoded polypeptide, the ATPase inhibitory factor 1 (IF1) [6]. IF1 is a small, basic, heat-stable protein composed of 80-84 amino acids (~10 kDa) in mammals and predominantly compartmentalized inside the mitochondrial matrix. Without affecting the ATP synthesis during oxidative phosphorylation, IF1 has the unique capacity to inhibit the adenosine triphosphate (ATP)-hydrolyzing activity of the F1F0-ATPsynthase. Recently, IF1 was found to be overexpressed in several human cancers, including breast cancer, colon cancer and lung cancer [7]. However, the biological relevance of IF1 in the NSCLC remains unknown. Therefore, we examined the expression of IF1 and evaluated its prognostic value in 149 cases of NSCLC in this study. The results obtained from systematically analyzed NSCLC cohort indicated that the expression of IF1 was increased in the cancerous tissues, and positively correlated to tumor stages and lymph node metastasis. Elevated expression of IF1 also served as an independent prognostic factor in NSCLC patients, especially in those with early tumor-node-metastasis (TNM) stages (stage I/II), which may developed as a potential marker to stratify therapeutic decisions.
Material and methods
Patients and tissue specimens
One hundred and forty-nine patients were included in this study, which carried out in accordance with the principles of the Helsinki Declaration and approved by the Ethical Committee of the Southwest Hospital, Third Military Medical University. All patients were present with NSCLC and underwent surgical resection without preoperative treatment between January 2006 and December 2009. Specimens were provided by the Department of Pathology, Southwest Hospital, Third Military Medical University, Chongqing, China. The specimens were evaluated separately by two pathologists according to the seventh edition of TNM classification of the International Union Against Cancer [8]. Additionally, 10 samples of normal lung tissues were enrolled as normal controls.
Tumor tissue microarrays (TMAs) construction
Formalin-fixed, paraffin-embedded tissues were applied to construct tissue microarrays (TMAs) as previously described [9]. Briefly, after verification with hematoxylin and eosin (H&E) staining, representative tumor areas with 1.0 mm-diameter were selected and deposited into a recipient paraffin block with tissue-arraying instrument (Beecher Instruments®, Silver Spring, MD, USA). Consecutive 3 μm-thick TMAs sections were mounted on coated glass sliders for immunohistochemistry analysis.
Immunohistochemical (IHC) staining and scoring
IHC staining was performed as previous procedure [10]. Sections were dewaxed and rehydrated in graded alcohols. Endogenous peroxidase was blocked by using a 3% solution of hydrogen peroxide for 30 minutes. For antigen retrieval, sections were boiled in EDTA buffer (pH=8.0) for 15 minutes. After naturally cooling to room temperature and washing with PBS, sections were then incubated with anti-human IF1 (Cell Signaling Technology, Billerica, MA, dilution 1:2000) at 4°C overnight. Following incubation with secondary antibody (DAKO, Denmark), the sections were then visualized using diaminobenzidine solution (DAKO, Denmark), and lightly counterstained with hematoxylin. The sections were then photographed under high magnification (400X). Five random areas of each section were selected to calculate the average optical density (AOD) by the Image-Pro Plus 6.0 software. The data were then normalized by determining a z-score using the following calculation: [(OD value)-(mean of all OD values in cohort)]/(standard deviation of all OD values in cohort) [11,12]. The cutoff value was determined by X-tile [13]. Therefore, low and high expression of IF1 were defined as the z-scores ≤-0.049 and >-0.049, respectively. Additionally, the normal lung tissues were also scored for the normal controls of NSCLC.
Statistical analysis
Statistical analysis was performed using the SPSS statistical software package (standard version 18.0). Two tail Student’s test was used to determine the difference of IF1 between normal and cancerous tissues. Pearson chi-square test was applied to determine the relationship between IF1 expression and the clinicopathological characteristics. For survival analysis, Kaplan-Meier curve was generated with log-rank test. Cox-regression analysis was applied to test for the independence of the prognostic value of IF1 expression. P<0.05 was considered statistically significant.
Results
IF1 was highly expressed in NSCLC
In total, 149 cases of NSCLC patients were enrolled in the study. The median age of the patients was 60.85 years (range from 36 to 81 years old). Among the subjects, 119 (79.87%) cases were males, and 30 (20.13%) cases were females. Seventy-three (49.0%) specimens were lung adenocarcinoma (LAC), and seventy-six (51.0%) specimens were squamous cell carcinoma (SCC). In addition, 69 (46.31%) patients were found to be IF1 highly expressed compared with 80 (53.69%) patients of IF1 lowly expressed. The details were summarized in Table 1. The cancerous tissues showed markedly stronger IHC staining signals than that in the normal lung tissues (Figure 1A). Accordingly, the z-scores were significantly higher in NSCLC than that in the normal tissues (0.042 ± 1.017 vs. -0.621 ± 0.282, P=0.042) (Figure 1B).
Table 1.
Correlation between IF1 expression levels and clinicopathological characters in NSCLC patients
| Clinical characters | NSCLC | |||
|---|---|---|---|---|
|
| ||||
| Cases N=149 | IF1 Expression | P value | ||
|
| ||||
| Low | High | |||
| Age (years) | 0.618 | |||
| ≤60 | 68 | 35 | 33 | |
| >60 | 81 | 45 | 36 | |
| Gender | 0.203 | |||
| Male | 119 | 67 | 52 | |
| Female | 30 | 13 | 17 | |
| Smoking | 0.017 | |||
| No | 50 | 20 | 30 | |
| Yes | 99 | 60 | 39 | |
| Histology | 0.695 | |||
| LAC | 73 | 38 | 35 | |
| SCC | 76 | 42 | 34 | |
| Differentiation | 0.384 | |||
| Well | 31 | 15 | 16 | |
| Moderate | 80 | 41 | 39 | |
| Poor | 38 | 24 | 14 | |
| Stage | 0.032 | |||
| I | 77 | 49 | 28 | |
| II | 50 | 23 | 27 | |
| III/IV | 22 | 8 | 14 | |
| N status | 0.043 | |||
| pN0 | 105 | 62 | 43 | |
| pN1-2 | 44 | 18 | 26 | |
| T status | 0.139 | |||
| pT1 | 45 | 22 | 23 | |
| pT2 | 85 | 51 | 34 | |
| pT3-4 | 19 | 7 | 12 | |
LAC lung adenocarcinoma, SCC squamous cell carcinoma. P value was evaluated by Chi-square test.
Figure 1.
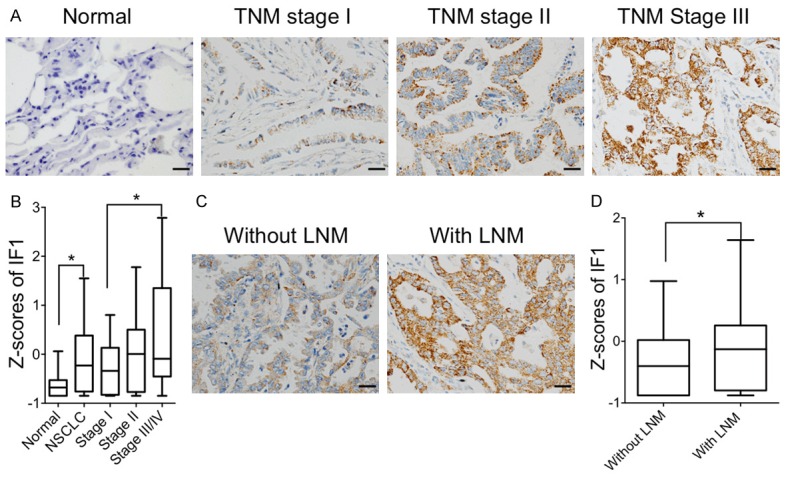
Expression levels of IF1 in NSCLC. A. Representative IHC staining images of IF1 expression levels in normal lung tissues and NSCLC patients with TNM stage I, II and III, respectively. B. Statistical analysis of z-scores in normal lung tissues and patients with different TNM stage. C. Representative IHC staining images showing the expression of IF1 in NSCLC without or with lymph node metastasis (LNM). D. Statistical analysis of z-scores in patients without LNM compared with those with LNM. Original magnification: x400. Scale bar 25 μm. *P<0.05.
Elevated expression of IF1 correlated with advanced tumor stages and lymphatic metastasis
The relationship between IF1 expression and clinicopathological characters was evaluated (Table 1). IHC staining showed the expression levels of IF1 were significantly increased in patients with advanced TNM stages (Figure 1A and 1B, Table 1, P=0.032) or lymphatic metastasis (Figure 1C and 1D, Table 1, P=0.043) as compared with those with early TNM stages or without lymphatic metastasis, respectively. These results suggest that IF1 associated to the aggressiveness of NSCLC patients.
IF1 served as an independent prognostic indicator in NSCLC
The prognostic value of IF1 was evaluated in NSCLC patients by comparing the IFlow and IFhigh group. Kaplan-Meier analysis showed that the patients with high levels of IF1 had significant worse overall survival than those with low levels of IF1 (Figure 2A left panel, P=0.007) in NSCLC. Moreover, IF1 expression status was significantly associated with tumor recurrence. The recurrence rate in patients with IF1 low expression was much lesser than those with IF1 high expression (Figure 2A right panel, P=0.003). Cox-regression analysis was used to determine the hazard ratio of prognostic relevant characters including gender (male vs. female), age (blow vs. above the median age of 60.85 years), smoking (former/current vs. never), TNM stage (stage I vs. stage II vs. stage III/IV), histological subtype (LAC vs. SCC) and IF1 expression level (dichotomized by z-scores -0.049). Multivariable Cox-regression analysis revealed that elevated expression of IF1 could be an independent prognostic factor (HR=1.667, P=0.034) in NSCLC (Table 2). These results demonstrate that elevated expression of IF1 predicted the aggressiveness property and might serve as a considerable independent indicator in NSCLC.
Figure 2.
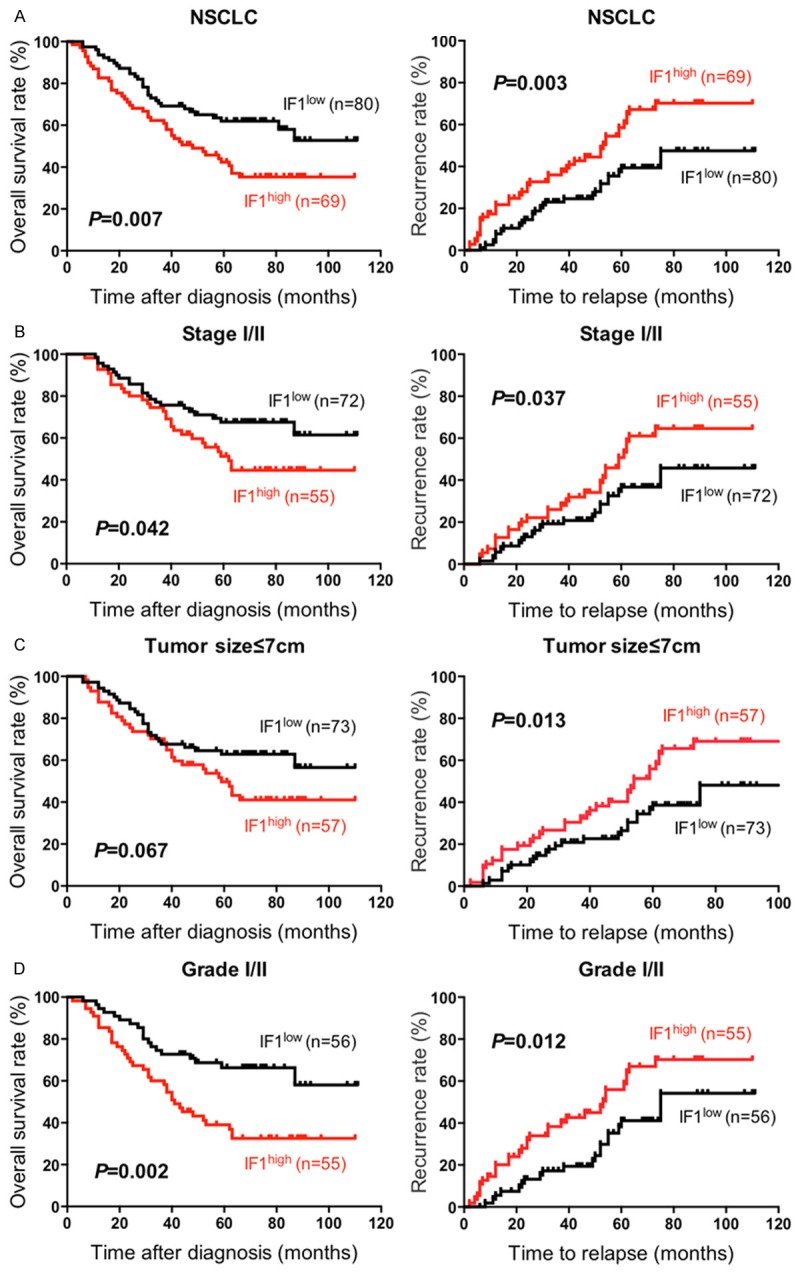
The prognostic value of IF1 in NSCLC patients. A. Kaplan-Meier curves of time after diagnosis and time to relapse in NSCLC patients. B. Kaplan-Meier curves of time after diagnosis and time to relapse in the NSCLC patients with early TNM stage (stage I/II). C. Kaplan-Meier curves of time after diagnosis and time to relapse in the NSCLC patients with tumor size ≤7 cm. D. Kaplan-Meier curves of time after diagnosis and time to relapse in the NSCLC patients with low grade (grade I/II).
Table 2.
Univariable/Multivariable Cox-regression analysis for overall survival in NSCLC patients
| Factors | Univariable | Multivariable | ||||
|---|---|---|---|---|---|---|
|
|
|
|||||
| HR | (95%) CI | P value | HR | (95%) CI | P value | |
| Gender | 0.982 | 0.571-1.689 | 0.948 | |||
| Age | 1.187 | 0.749-1.881 | 0.466 | 1.686 | 1.036-2.743 | 0.036 |
| Smoking | 0.661 | 0.417-1.047 | 0.078 | |||
| T status | 1.990 | 1.402-2.825 | 0.000 | 1.793 | 1.266-2.539 | 0.001 |
| N status | 2.847 | 2.130-3.806 | 0.000 | 2.661 | 1.962-3.607 | 0.000 |
| Histology | 0.673 | 0.424-1.068 | 0.093 | |||
| IF1 | 1.858 | 1.170-2.949 | 0.000 | 1.667 | 1.040-2.671 | 0.034 |
HR hazard ratio, CI confidence interval.
Moreover, we further detected the prognostic value of IF1 in early TNM stages (stage I/II) patients. Kaplan-Meier analysis showed that the IF1high group suffered significant worse outcome than the IF1low group (Figure 2B left panel, P=0.042). Accordingly, the median overall survival time in the IF1low group (77.2 (95% CI 67.9 to 86.5) months) was longer than in the IF1high group (58.6 (95% CI 48.8 to 68.4) months). The recurrence rate in patients with IF1 high expression was higher than those with IF1 low expression (Figure 2B right panel, P=0.037). We further did a subgroup analysis, stratifying NSCLC patients by both IF1 expression and tumor size. Patients with tumor size ≤7 cm and low IF1 level tended to have longer overall survival (P=0.067) and had the significantly lower recurrent rate (P=0.013) (Figure 2C). Accordingly, the 5-year recurrent rate of patients with tumor size >7 cm was much higher than those with tumor size ≤7 cm (50.88% vs. 31.94%). In addition, analyses of IF1 expression in low-grade (grade I/II) patients revealed that IFlow group possessed markedly promising overall survival rate (P=0.002), as well as lower recurrent rate (P=0.012) than those in IFhigh group (Figure 2D). The results indicate that elevated expression of IF1 predicted the malignancy or the risk of the recurrence of early stage and lower grade NSCLC, and could be recognized as a stratifying marker for adjuvant therapy.
Discussion
Lung cancer is a common malignancy with high mortality rate [1]. Approximately 65% of NSCLC patients were firstly diagnosed at advanced stages (stage III/IV), and led to an unfavorable 5-year overall survival rate of less than 5% [14]. Thus, early detection of lung cancer could significantly improve the therapy response rate and prolonged survival time of patients [15]. In the present study, we found that the IF1 content was low in normal lung tissues, increased in early stages of NSCLC, and reached the highest level in advanced stages. Meanwhile, Kaplan-Meier analysis suggested that the IF1high group possessed poorer prognosis and higher recurrent rate than the IF1low group in patients with early stages. These findings suggested that the expression of IF1 might be developed as a predictive biomarker for early detection and potential indicator for stratify therapeutic decisions.
Downregulation of oxidative phosphorylation and concurrent activation of aerobic glycolysis are hallmark features of human cancers [16-18]. Mitochondria are the primary energy producers of the cell that regulate intracellular energy metabolism. The dysfunctions of mitochondria have been found to participate in tumorgenesis and tumor progression, including proliferation, apoptosis, drug resistance, dedifferentiation and metastasis [19-21]. Laura et al. first reported IF1 in 2010 as a contributor to tumor growth, which promoted the metabolic switch in cancer cells [7]. In the following, more and more studies suggested that high expression of IF1 played a prominent role in tumor progression and was correlated with the regulation of tumor energetic metabolism by controlling the synthase activity of the H+-ATP synthase [22-28]. The increase of IF1 might alter the pattern of energy metabolism and result in the aggressiveness of NSCLC patients. Furthermore, owing to its functional activities in inhibiting the hydrolysis of ATP and preventing mitochondrial remodeling [6,29], it holds strong potential to become a novel target for cancer therapy by reprogramming energy metabolism.
Metastasis is the major cause of NSCLC-related mortality. The epithelial-mesenchymal transition (EMT) is a carcinogenesis-enabling process with pivotal role in the initiation of metastasis and establishment of tumor in the metastatic site [30]. NF-κB/Snail is one of the key signaling pathways in regulating the EMT [31]. Previous study reported that IF1 could activate the Snai1 signaling and mediate metastasis in hepatocellular carcinoma [32]. In the present study, we found that the expression of IF1 was associated with lymph node metastasis in NSCLC patients. Thus, detection of IF1 might help to predict the metastasis event and offer new possibilities in the treatment for patients suffering from NSCLC.
In conclusion, we demonstrated that IF1 expression levels were significantly higher in NSCLC tissues compared with normal lung tissues. Moreover, our results showed that the elevated expression of IF1 was closely related to lymph node metastasis and served as an independent prognostic and recurrent indicator in NSCLC patients, particularly in those with early TNM stages or lower grade. IF1 may thus be useful for evaluating prognosis and may provide a novel target for the treatment of patients with NSCLC.
Acknowledgements
We thank all the patients who agreed to participate in this study. This work was supported by the National Natural Science Foundation of China (No. 81230062), and the National Basic Research Program of China (973 Program, No. 2010CB529400).
Disclosure of conflict of interest
None.
References
- 1.Siegel RL, Miller KD, Jemal A. Cancer statistics, 2015. CA Cancer J Clin. 2015;65:5–29. doi: 10.3322/caac.21254. [DOI] [PubMed] [Google Scholar]
- 2.Ettinger DS, Wood DE, Akerley W, Bazhenova LA, Borghaei H, Camidge DR, Cheney RT, Chirieac LR, D’Amico TA, Demmy TL, Dilling TJ, Govindan R, Grannis FW Jr, Horn L, Jahan TM, Komaki R, Kris MG, Krug LM, Lackner RP, Lanuti M, Lilenbaum R, Lin J, Loo BW Jr, Martins R, Otterson GA, Patel JD, Pisters KM, Reckamp K, Riely GJ, Rohren E, Schild S, Shapiro TA, Swanson SJ, Tauer K, Yang SC, Gregory K, Hughes M. Non-small cell lung cancer, version 1.2015. J Natl Compr Canc Netw. 2014;12:1738–1761. doi: 10.6004/jnccn.2014.0176. [DOI] [PubMed] [Google Scholar]
- 3.O’Shannessy DJ, Yu G, Smale R, Fu YS, Singhal S, Thiel RP, Somers EB, Vachani A. Folate receptor alpha expression in lung cancer: diagnostic and prognostic significance. Oncotarget. 2012;3:414–425. doi: 10.18632/oncotarget.519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Okamura K, Takayama K, Kawahara K, Harada T, Nishio M, Otsubo K, Ijichi K, Kohno M, Iwama E, Fujii A, Ota K, Koga T, Okamoto T, Suzuki A, Nakanishi Y. PICT1 expression is a poor prognostic factor in non-small cell lung cancer. Oncoscience. 2014;1:375–382. doi: 10.18632/oncoscience.43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Tomida S, Koshikawa K, Yatabe Y, Harano T, Ogura N, Mitsudomi T, Some M, Yanagisawa K, Takahashi T, Osada H, Takahashi T. Gene expression-based, individualized outcome prediction for surgically treated lung cancer patients. Oncogene. 2004;23:5360–5370. doi: 10.1038/sj.onc.1207697. [DOI] [PubMed] [Google Scholar]
- 6.Faccenda D, Campanella M. Molecular Regulation of the Mitochondrial F(1)F(o)-ATPsynthase: Physiological and Pathological Significance of the Inhibitory Factor 1 (IF(1)) Int J Cell Biol. 2012;2012:367934. doi: 10.1155/2012/367934. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Sanchez-Cenizo L, Formentini L, Aldea M, Ortega AD, Garcia-Huerta P, Sanchez-Arago M, Cuezva JM. Up-regulation of the ATPase inhibitory factor 1 (IF1) of the mitochondrial H+-ATP synthase in human tumors mediates the metabolic shift of cancer cells to a Warburg phenotype. J Biol Chem. 2010;285:25308–25313. doi: 10.1074/jbc.M110.146480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Goldstraw P, Crowley J, Chansky K, Giroux DJ, Groome PA, Rami-Porta R, Postmus PE, Rusch V, Sobin L International Association for the Study of Lung Cancer International Staging Committee; Participating Institutions. The IASLC Lung Cancer Staging Project: proposals for the revision of the TNM stage groupings in the forthcoming (seventh) edition of the TNM Classification of malignant tumours. J Thorac Oncol. 2007;2:706–714. doi: 10.1097/JTO.0b013e31812f3c1a. [DOI] [PubMed] [Google Scholar]
- 9.Kononen J, Bubendorf L, Kallioniemi A, Barlund M, Schraml P, Leighton S, Torhorst J, Mihatsch MJ, Sauter G, Kallioniemi OP. Tissue microarrays for high-throughput molecular profiling of tumor specimens. Nat Med. 1998;4:844–847. doi: 10.1038/nm0798-844. [DOI] [PubMed] [Google Scholar]
- 10.Wu F, Zhou Q, Yang J, Duan GJ, Ou JJ, Zhang R, Pan F, Peng QP, Tan H, Ping YF, Cui YH, Qian C, Yan XC, Bian XW. Endogenous axon guiding chemorepulsant semaphorin-3F inhibits the growth and metastasis of colorectal carcinoma. Clin Cancer Res. 2011;17:2702–2711. doi: 10.1158/1078-0432.CCR-10-0839. [DOI] [PubMed] [Google Scholar]
- 11.Shanle EK, Onitilo AA, Huang W, Kim K, Zang C, Engel JM, Xu W, Wisinski KB. Prognostic significance of full-length estrogen receptor beta expression in stage I-III triple negative breast cancer. Am J Transl Res. 2015;7:1246–1259. [PMC free article] [PubMed] [Google Scholar]
- 12.Huang W, Hennrick K, Drew S. A colorful future of quantitative pathology: validation of Vectra technology using chromogenic multiplexed immunohistochemistry and prostate tissue microarrays. Hum Pathol. 2013;44:29–38. doi: 10.1016/j.humpath.2012.05.009. [DOI] [PubMed] [Google Scholar]
- 13.Camp RL, Dolled-Filhart M, Rimm DL. X-tile: a new bio-informatics tool for biomarker assessment and outcome-based cut-point optimization. Clin Cancer Res. 2004;10:7252–7259. doi: 10.1158/1078-0432.CCR-04-0713. [DOI] [PubMed] [Google Scholar]
- 14.Shao Q, Li J, Li F, Wang S, Wang W, Liu S, Zhang Y. Clinical investigation into the initial diagnosis and treatment of 1,168 lung cancer patients. Oncol Lett. 2015;9:563–568. doi: 10.3892/ol.2014.2777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Parkin DM, Bray F, Ferlay J, Pisani P. Global cancer statistics, 2002. CA Cancer J Clin. 2005;55:74–108. doi: 10.3322/canjclin.55.2.74. [DOI] [PubMed] [Google Scholar]
- 16.Cuezva JM, Ortega AD, Willers I, Sanchez-Cenizo L, Aldea M, Sanchez-Arago M. The tumor suppressor function of mitochondria: translation into the clinics. Biochim Biophys Acta. 2009;1792:1145–1158. doi: 10.1016/j.bbadis.2009.01.006. [DOI] [PubMed] [Google Scholar]
- 17.Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144:646–674. doi: 10.1016/j.cell.2011.02.013. [DOI] [PubMed] [Google Scholar]
- 18.Kroemer G, Pouyssegur J. Tumor cell metabolism: cancer’s Achilles’ heel. Cancer Cell. 2008;13:472–482. doi: 10.1016/j.ccr.2008.05.005. [DOI] [PubMed] [Google Scholar]
- 19.Chen EI. Mitochondrial dysfunction and cancer metastasis. J Bioenerg Biomembr. 2012;44:619–622. doi: 10.1007/s10863-012-9465-9. [DOI] [PubMed] [Google Scholar]
- 20.Scatena R. Mitochondria and Cancer: A Growing Role in Apoptosis, Cancer Cell Metabolism and Dedifferentiation. In: Scatena R, Bottoni P, Giardina B, editors. Advances in Mitochondrial Medicine. Dordrecht: Springer Netherlands; 2012. pp. 287–308. [DOI] [PubMed] [Google Scholar]
- 21.Wallace DC. Mitochondria and cancer. Nature Reviews Cancer. 2012;12:685–698. doi: 10.1038/nrc3365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Willers IM, Cuezva JM. Post-transcriptional regulation of the mitochondrial H+-ATP synthase: a key regulator of the metabolic phenotype in cancer. Biochim Biophys Acta. 2011;1807:543–551. doi: 10.1016/j.bbabio.2010.10.016. [DOI] [PubMed] [Google Scholar]
- 23.Domenis R, Comelli M, Bisetto E, Mavelli I. Mitochondrial bioenergetic profile and responses to metabolic inhibition in human hepatocarcinoma cell lines with distinct differentiation characteristics. J Bioenerg Biomembr. 2011;43:493–505. doi: 10.1007/s10863-011-9380-5. [DOI] [PubMed] [Google Scholar]
- 24.Domenis R, Bisetto E, Rossi D, Comelli M, Mavelli I. Glucose-modulated mitochondria adaptation in tumor cells: a focus on ATP synthase and inhibitor Factor 1. Int J Mol Sci. 2012;13:1933–50. doi: 10.3390/ijms13021933. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Sánchez-Aragó M, Formentini L, García-Bermúdez J, Cuezva JM. IF1 reprograms energy metabolism and signals the oncogenic phenotype in cancer. Cell Cycle. 2012;11:2963–2964. doi: 10.4161/cc.21387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Barbato S, Sgarbi G, Gorini G, Baracca A, Solaini G. The Inhibitor Protein (IF1) of the F1F0-ATPase Modulates Human Osteosarcoma Cell Bioenergetics. J Biol Chem. 2015;290:6338–6348. doi: 10.1074/jbc.M114.631788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.García-Bermúdez J, Sánchez-Aragó M, Soldevilla B, del Arco A, Nuevo-Tapioles C, Cuezva JM. PKA Phosphorylates the ATPase Inhibitory Factor 1 and Inactivates Its Capacity to Bind and Inhibit the Mitochondrial H+-ATP Synthase. Cell Rep. 2015;12:2143–2155. doi: 10.1016/j.celrep.2015.08.052. [DOI] [PubMed] [Google Scholar]
- 28.Santacatterina F, Sánchez-Cenizo L, Formentini L, Mobasher MA, Casas E, Rueda CB, Martínez-Reyes I, Núñez de Arenas C, García-Bermúdez J, Zapata JM, Sánchez-Aragó M, Satrústegui J, Valverde ÁM, Cuezva JM. Down-regulation of oxidative phosphorylation in the liver by expression of the ATPase inhibitory factor 1 induces a tumor-promoter metabolic state. Oncotarget. 2016;7:490–508. doi: 10.18632/oncotarget.6357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Faccenda D, Tan CH, Seraphim A, Duchen MR, Campanella M. IF1 limits the apoptotic-signalling cascade by preventing mitochondrial remodelling. Cell Death Differ. 2013;20:686–697. doi: 10.1038/cdd.2012.163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Voutsadakis IA. Epithelial-Mesenchymal Transition (EMT) and Regulation of EMT Factors by Steroid Nuclear Receptors in Breast Cancer: A Review and in Silico Investigation. J Clin Med. 2016:5. doi: 10.3390/jcm5010011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Tania M, Khan MA, Fu J. Epithelial to mesenchymal transition inducing transcription factors and metastatic cancer. Tumour Biol. 2014;35:7335–7342. doi: 10.1007/s13277-014-2163-y. [DOI] [PubMed] [Google Scholar]
- 32.Song R, Song H, Liang Y, Yin D, Zhang H, Zheng T, Wang J, Lu Z, Song X, Pei T, Qin Y, Li Y, Xie C, Sun B, Shi H, Li S, Meng X, Yang G, Pan S, Zhu J, Qi S, Jiang H, Zhang Z, Liu L. Reciprocal activation between ATPase inhibitory factor 1 and NF-kappaB drives hepatocellular carcinoma angiogenesis and metastasis. Hepatology. 2014;60:1659–1673. doi: 10.1002/hep.27312. [DOI] [PubMed] [Google Scholar]


