Abstract
Objective
Report on the 7-year experience with bilateral ophthalmic artery chemosurgery (OAC-Tandem therapy) for bilateral retinoblastoma.
Design
Retrospective, single institution study.
Subjects
120 eyes of 60 children with bilateral retinoblastoma treated since March 2008.
Methods
Retrospective review of all children treated at Memorial Sloan Kettering with bilateral ophthalmic artery chemosurgery (Melphalan, Carboplatin, Topotecan, Methotrexate) delivered in the same initial session to both naïve and previously treated eyes.
Main Outcome Measures
Ocular survival, metastatic disease, patient survival from metastases, second cancers, systemic adverse effects, need for transfusion of blood products, electroretinogram before and after treatment.
Results
116 eyes were salvaged (4 eyes were enucleated: 3 because of progressive disease, 1 family choice). Kaplan Meier ocular survival was 99.2% at one year, 96.9% at 2 and 3 years and 94.9% for years 4 through 7. There were no cases of metastatic disease or metastatic deaths with a mean follow-up of 3.01 years. Two children developed second cancers (both pineoblastoma) and one of them died. Transfusion of blood products was required in 3 cases (4 transfusions), 1.9%. Two children developed fever/neutropenia requiring hospitalization (0.95%). ERGs were improved in 21.6% and unchanged after treatment in 52.5% of cases (increase or decrease of less than 25μV).
Conclusions
Bilateral ophthalmic artery chemosurgery is a safe and effective technique for managing bilateral retinoblastoma-even when eyes are advanced bilaterally, and if both eyes have progressed after systemic chemotherapy. Ocular survival was excellent (94.9% at 8 years), there were no cases of of metastatic disease and no deaths from metastatic disease, but children remain at risk for second cancers. In 21.6% of cases ERG function improved. Despite using chemotherapy in both eyes in the same session, systemic toxicity was low.
Introduction
Ophthalmic artery chemosurgery (OAC) was introduced almost 10 years ago for the treatment of retinoblastoma[1] and has been performed in more than 45 countries worldwide for unilateral retinoblastoma.[2] In a recent survey it was the first-line choice for the majority of retinoblastoma centers worldwide for patients with advanced unilateral disease.[3] The technique has been successful for naïve eyes[4,5] and eyes that failed all prior therapies.[6,7] It has enabled the majority of advanced eyes that were previously managed with enucleation to be saved–[4,8–10] sometimes with useful vision–[10,11] without compromising patient survival.[11,12] In addition to saving eyes with advanced intraocular disease[8,9] it has been used for eyes with less advanced disease,[10,11] eyes with vitreous seeding, sub-retinal seeding [13,14] and as salvage therapy in eyes that progressed after a prior course of intrarterial chemotherapy.[14,15] It has also been shown to significantly reduce the development of new intraocular tumor foci in germline cases. [15,16]
OAC has been used for one eye in bilateral cases by many centers after one eye was enucleated or in cases of asymmetric bilateral disease (allowing focal treatments only for one eye and OAC for the fellow eye) but the first report on simultaneous treatment of both eyes (called “tandem therapy”) was published just 5 years ago.[12] Since that initial report there have been more than 200 peer review publications on the indications, results, complications and other issues in unilateral cases but no additional case series employing simultaneous use for bilateral disease. Fewer than 10 such cases have been reported in the literature since our description of the technique.
We now have experience with more than 200 bilateral cases and more than 60 bilateral cases treated simultaneously so we performed a single institution retrospective analysis of all bilateral cases where both eyes were treated in the same session to determine patient survival, systemic complications, ocular survival and ocular complications.
Methods
This is a retrospective chart review, approved by the Memorial Sloan Kettering Cancer Center (MSKCC) Institutional Review Board (IRB), of all bilateral retinoblastoma patients whose initial management was simultaneous bilateral OAC at MSKCC between March 2008 and May 2015. Bilateral patients who received OAC in one eye and non-OAC treatments in the fellow eye were not included. Patients who had previously been treated elsewhere with OAC and then received simultaneous OAC at our institution were not included, but any patient who had prior therapy that did not include OAC was included so long as the first treatment at MSKCC was with tandem therapy. Clinical characteristics including laterality, treatment parameters, sex, age at diagnosis, electroretinography (ERG) before and after treatment, ocular and patient survival and second tumors were collected. Patient information was anonymized and deidentified prior to analysis.
We have previously described our tandem technique[16,17] though there have been some refinements in technique since our initial report 5 years ago. A microcatheter is inserted into the femoral artery and passed through the abdominal and thoracic aorta into the internal carotid artery on one side. After advancing the catheter to the orifice of the ophthalmic artery, chemotherapy is infused. The catheter is then withdrawn to the aorta and advanced up the contralateral side to the orifice of the fellow ophthalmic artery for the second chemotherapy infusion. At the end of the procedure the microcatheter is removed, manual pressure is applied, and the patients are observed for 4–6 hours before being discharged the same day.
Catheterization of the ophthalmic artery is performed with a microcatheter only- a guide catheter is no longer used. When treating both eyes we deliver all drug(s) within a one hour time period. Nasal vasoconstrictors and topical phenylephrine on the skin in the distribution of the supratrochlear artery were routinely used.
Electroretinograms (ERGs) were performed before and at the follow up visits 3–4 weeks after each treatment. Measurements are taken immediately following anesthetic induction but before the ocular exam; scleral indentations affect recorded potentials.[18]
Electroretinography
As previously reported we adapted the International Society for Clinical Electrophysiology of Vision’s standard ERG protocol to obtain electroretinography (ERG) recordings.[19] The 30-Hz photopic flicker amplitude data were used, which we have previously reported to be an accurate representation of the complete ERG response protocol.[20] The initial ERG was compared to the most recent ERG. 30-Hz response amplitude changes of >25 μV were considered clinically significant. As in our earlier published series ERG amplitudes were capped at 100.1 μV. ERG amplitudes were classified as follows: less than 0.1 μV, undetectable; 0.1–25 μV, poor; 25.1–50 μV, fair; 50.1–75 μV, good; 75.1–100 μV very good; more than 100 μV, excellent.
Results
Patient Characteristics are presented in Table 1. Ocular Characteristics of treated eyes are presented in Table 2. OAC treatment details are presented in Tables 3 & 4. Of the 120 eyes treated 13 subsequently received either an additional course(s) of OAC or radiation (7 cases received additional OAC, 6 received a plaque, and 1 received external beam radiation at another Institution).
Table 1. Patient Demographics.
| Gender | Male: | 26 | |
| Female: | 34 | ||
| Total: | 60 | ||
| Family History | Neg: | 48 | |
| Pos: | 11 | ||
| Unkn: | 1 | ||
| Total: | 60 | ||
| Mean | Median | Range | |
| Age at Presentation (yrs) | 0.91 | 0.71 | 0.16—4.46 |
| Patient Follow up (yrs) | 3.12 | 3.05 | 0.18—7.12 |
Table 2. Eye Classifications & Group Salvage.
| International Classification (ICRb) | Reese Ellsworth (RE) | ||||
|---|---|---|---|---|---|
| Group | Number of Eyes (% of total) | Eyes Salvaged (% salvaged) | Group | Number of Eyes (% of total) | Eyes Salvaged (% salvaged) |
| A: | 2 (1.7) | 2 (100) | I: | 3 (2.5) | 3 (100) |
| B: | 18 (15.0) | 18 (100) | II: | 9 (7.5) | 9 (100) |
| C: | 24 (20.0) | 24 (100) | III: | 19 (15.8) | 19 (100) |
| D: | 56 (46.7) | 54 (100) | IV: | 12 (10.0) | 12 (100) |
| E: | 20 (16.7) | 18 (90.0) | V: | 77 (64.2) | 73 (94.8) |
| Total: | 120 (100) | 116/120 (96.7) | Total: | 120 (100) | 116/120 (96.7) |
Table 3. OAC Treatment Details.
| OAC Treatments | |
| Total (per eye) | 418 |
| Mean (per eye) | 3.48 |
| Range | 1.0–8.0 |
| OAC Sessions* | |
| Total | 256 |
| Mean (per patient) | 4.27 |
| Range | 1.0–11.0 |
| Tandem Therapy | |
| Total | 156.0 |
| Mean (per patient) | 1.3 |
| Range | 1.0–6.0 |
*Session defined as either unilateral or tandem OAC following an anesthetic induction.
Table 4. Drug Combinations Used Per OAC Treatment.
| Chemotherapy | Number of treatments (#) | Percent of total (%) |
|---|---|---|
| M+T | 101 | 24.3 |
| M | 98 | 23.6 |
| T+C | 79 | 19.0 |
| M+T+C | 58 | 14.0 |
| C | 51 | 12.3 |
| M+C | 25 | 6.0 |
| T | 2 | 0.5 |
| Mtx | 1 | 0.2 |
| Total*: | 415 |
M = Melphalan, T = Topotecan, C = Carboplatin, Mtx = Methotrexate
*In 3 cases the OAC procedure was terminated prior to the administration of chemotherapy.
The distribution of ICRb in the two eyes was: A&B: 1, A&E: 1, B&B: 2, B&C: 3, B&D: 5, B&E: 5, C&C: 3, C&D: 14, C&E: 1, D&D: 14, D&E: 9, E&E: 2.
Half (30) of the patients were naïve to treatment and half (30) received OAC after having been treated with systemic chemotherapy and/or radiation. No patient developed metastatic disease and no patient died from metastatic disease. Two patients developed second cancers (both pineoblastomas) and one died.
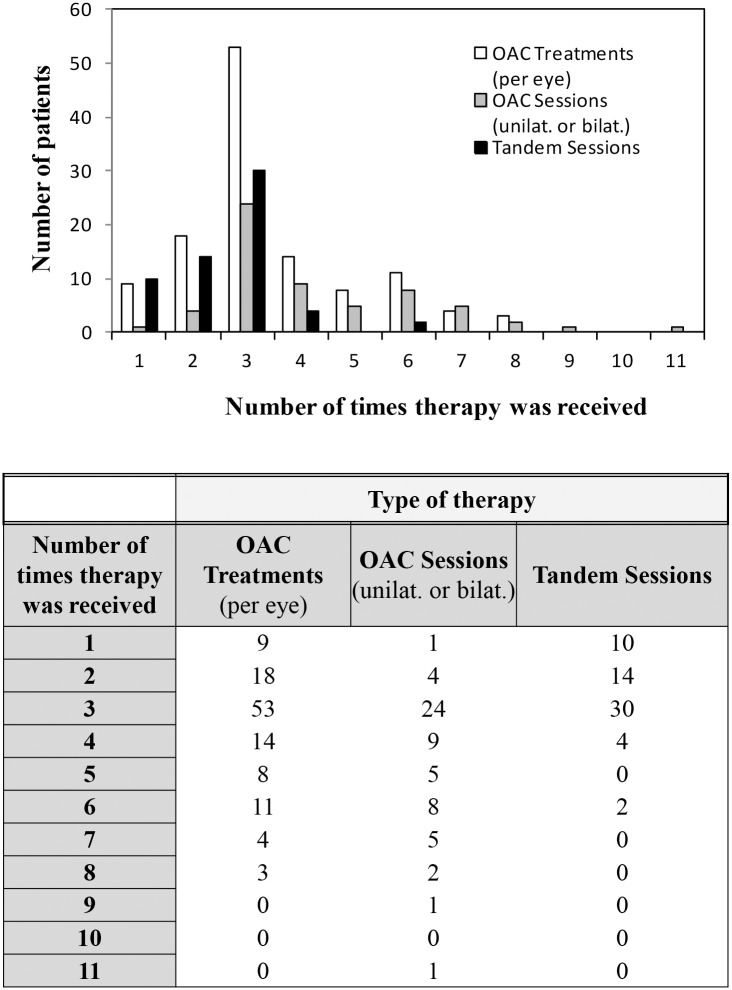
One hundred twenty eyes were treated in a total of 418 sessions, with the total number of treatments per patient ranging from 1 to 11 (mean 3.46 per patient). Tables 3 & 4 and Fig 1 summarize the OAC treatment details.
Fig 1. Type and amount of OAC therapy received per number of patients.
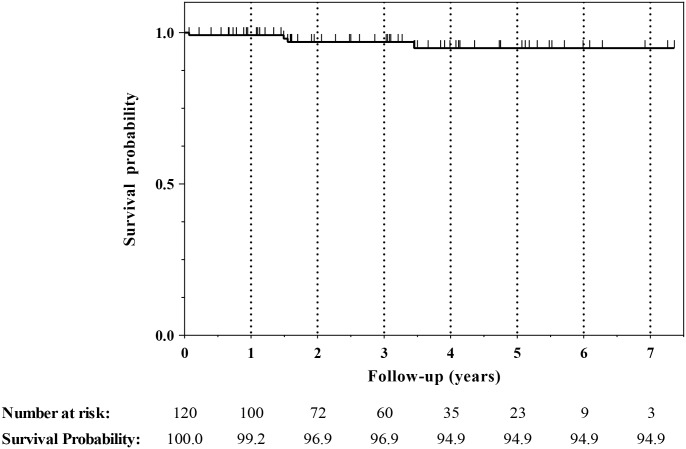
Four of the 120 eyes came to enucleation (3 with progressive disease, 1 family choice), and the Kaplan Meier ocular survival rates are presented in Fig 2. There were no bilateral enucleations. The classification of the enucleated eyes was Va/E:1, Vb/E:1, Vb/D:2. There were no procedure deaths or strokes (1 patient had a TIA). The ERG remained the same before and after treatment in 52.5% of eyes while 21.7% improved and 25.8% decreased.
Fig 2. Kaplan Meier Ocular Survival Curve.
One hundred and twenty eyes of 60 patients who underwent bilateral ophthalmic artery chemosurgery (Tandem Therapy) were assessed for enucleation. Ocular survival was 99.2% at one year, 96.9% at 2 and 3 years, and 94.9% for years 4 through 7.
There were 2 cases of fever/neutropenia (0.96%) and 3 cases (4 transfusions) of blood products required (1.9%). 1 patient was admitted for fever/neutropenia without transfusion. 1 patient was admitted twice for fever/neutropenia following two subsequent OAC sessions, receiving a transfusion during each admission. 2 patients received transfusion without admission.
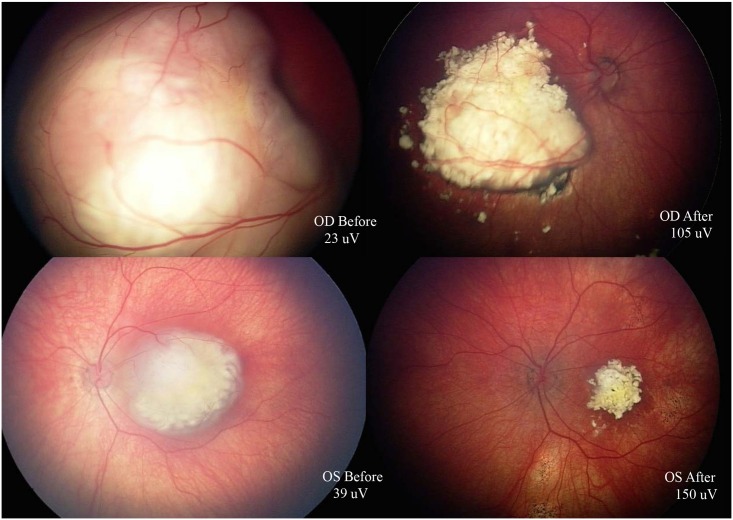
Fig 3 shows both eyes of one patient before and after treatment with tandem OAC.
Fig 3. Fundus images from patient before (left) and after (right) tandem OAC.
Upper row: right eye, lower row: left eye. ERG Right eye: before 23uV, after 105uV. ERG Left eye: before 39uV, after 150uV.
Discussion
Intrarterial chemotherapy has transformed the management of unilateral retinoblastoma worldwide.[2] It has enabled eyes that were previously enucleated worldwide to be salvaged without sacrificing patient survival.[13] Writing in the 1960’s Reese and Duke-Elder emphasized that all unilateral eyes should be enucleated because there was no other therapeutic option available (at the time).[2] In 1982 we published the first large report on the successful treatment of unilateral retinoblastoma without enucleation and demonstrated that survival was not compromised despite trying to save the eye. However, only two of the patients in that series had advanced intraocular disease; they were treated with external beam irradiation.[14]
Bilateral retinoblastoma has been treated with radiation for >80 years. Initially patients were cured but because of radiation damage no eye was salvaged with useful vision. As doses were lowered and techniques improved clinicians would enucleate the “more advanced” eye and irradiate the “less advanced eye” so bilateral simultaneous treatment was unusual.[15] With even more experience and different equipment bilateral simultaneous radiation was successfully done for bilateral retinoblastoma.[16] In cases of bilateral Reese Ellsworth groups I-III success was high. Eighty six per-cent of the eyes were saved and only 5% of the patients died of metastatic retinoblastoma. In select cases bilateral (Reese-Ellsworth groups IV and V) advanced retinoblastoma was also primarily bilaterally radiated. Ocular survival rates were 53% at ten years. Death from metastatic disease was rare but 52.6% of patient developed a second cancer by 18 ¼ years.[17] Largely because of second-cancer development, external beam irradiation was abandoned in favor of multiagent systemic chemotherapy 25 years ago.[21,22] Unfortunately, results with systemic chemotherapy for advanced eyes were disappointing. Virtually all “E” eyes were still enucleated[23] and the majority of “D” eyes were either enucleated or progressed despite systemic chemotherapy.[24] As a result there are few reported successes using systemic chemotherapy for bilateral advanced intraocular disease.
In our initial paper on OAC, 100% of the eyes were advanced, scheduled for enucleation and patients had only unilateral disease.[1] Five years ago we were the first to report on simultaneous bilateral OAC treatment and named the technique “tandem therapy”.[12] In those 4 patients (8 eyes) results were encouraging and no patient died. Since then we have had considerable experience in managing bilateral retinoblastoma with OAC. Many of our cases of bilateral disease were managed elsewhere and then referred after failing therapy (including, but not limited to, enucleation of one eye). Often the second, only remaining eye was scheduled elsewhere for enucleation. Similarly, naïve bilateral patients seen and managed initially by us who required either enucleation of one eye (rare nowadays) or only focal treatments in one eye were not included in this series.
There are special considerations for patients treated with “tandem therapy”. Melphalan in both humans[25] and animals[26] causes significant bone marrow suppression when total body dose exceeds 0.4mg/kg. In advanced cases 0.4mg/kg is close to the dose we use to treat one eye, and if we were to give double the dose, the bone marrow suppression would be profound. As a result, in tandem therapy we treat one eye with Melphalan (not exceeding 0.4mg/kg) and the fellow eye with Carboplatin. Carboplatin is well known to be active against retinoblastoma when delivered intravenously,[27–30] periocularly[31–33] or intra-arterially.[34] Topotecan is active against retinoblastoma in pre- clinical studies[35] and used in all three of these routes when treating retinoblastoma.[36,37] Theoretically, the effect of a topoisomerase inhibitor (Topotecan) used in conjunction with an alkylator (Melphalan or Carboplatin) are additive, so we have used Topotecan in conjunction with both Melphalan and Carboplatin in tandem therapy. Head- to-head randomized trials comparing each of these three drugs to each other have not been done. Like the Japanese,[38] our experience has been that in the doses we are using Melphalan is the most potent of the three drugs. We have used this observation to guide the decision about which eye will initially receive Melphalan or Carboplatin. Often Melphalan is given to the eye with more advanced disease, though at times it has initially been given to the eye with better potential vision. The decision about changing drug combinations on subsequent sessions is a clinical one and does not follow a rigid algorithm, but commonly the drugs are switched in the second or third session so that the Melphalan eye then receives Carboplatin based therapy.
Despite using multiple chemotherapeutic agents in the same patient at the same sitting, few systemic complications have developed. Transfusion of blood product was limited to 4 transfusions in three patients (1.9% of the events) and clinically significant fever/neutropenia occurred in 2 cases (0.96%).
Ocular toxicity paralleled our previous reports on unilateral retinoblastoma managed with OAC[39] and in most cases the ERG was unaffected by treatment. It is notable that in more than 20% of the eyes there was measurable improvement in ERG function.
Despite the fact that the majority of eyes treated with tandem therapy had advanced intraocular disease, no patient has developed metastatic disease and no patient has died of metastasis. Simultaneously treating both eyes did not compromise patient survival. Equally striking is the observation that of 120 eyes treated, only four came to enucleation (one due to family preference). The 5-year Kaplan-Meier ocular survival rate was 94.9%, which is noteworthy since more than half of these patients had very advanced intraocular disease (ICRb D or E) and half had previously received systemic chemotherapy.
Tandem therapy was equally effective for patient and ocular survival if the patient was naive or had previously received global therapy.
Among children in the first 5 years of life the most common second cancer to develop in bilateral cases is pineoblastoma. Two of our patients developed this tumor and one patient died as a result. These numbers are consistent with the latest published meta-analysis on the incidence of trilateral retinoblastoma in bilateral patients who do not receive radiation.[40] In the long term follow-up of the Japanese experience with selective ophthalmic artery infusion for retinoblastoma, pineoblastomas also developed but the authors felt the incidence was not increased when they compared the Japanese patients who had and had not received intrarterial chemotherapy.[41]
In a recent paper on the management of retinoblastoma from four leading centers worldwide there was complete agreement that OAC was first line choice for advanced unilateral retinoblastoma.[13] There was also complete agreement in the management of bilateral retinoblastoma when there was less advanced disease but there was disagreement about the management of bilateral advanced retinoblastoma. Two centers preferred tandem therapy as initial choice but two others felt systemic chemotherapy should be tried first and tandem therapy reserved for eyes that failed or progressed following systemic chemotherapy. The present study gives some assurance to clinicians that tandem therapy can be successful without significant ocular or systemic side effects and without compromising patient survival.
Data Availability
The data contained within this study has been deidentified and will be shared by either the corresponding author or designated personnel upon written request and in accordance with MSK policies on privacy and data sharing. Please send all requests for data to the following address: David H. Abramson MD Memorial Sloan Kettering Cancer Center Pediatric Ophthalmic Oncology Suite A-330 1275 York Ave New York, NY 10065
Funding Statement
This work was supported by the Fund for Ophthalmic Knowledge, Inc, New York (No grant number; philanthropic fund) and funded in part through the NIH/NCI Cancer Center Support Grant P30 CA008748. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1.Abramson DH, Dunkel IJ, Brodie SE, Kim JW, Gobin YP. A Phase I/II Study of Direct Intraarterial (Ophthalmic Artery) Chemotherapy with Melphalan for Intraocular Retinoblastoma. Ophthalmol. 2008;115: 1398–1404. e1 10.1016/j.ophtha.2007.12.014 [DOI] [PubMed] [Google Scholar]
- 2.Abramson DH. Retinoblastoma: saving life with vision. Annu Rev Med. 2014;65: 171–184. 10.1146/annurev-med-061312-123455 [DOI] [PubMed] [Google Scholar]
- 3.Grigorovski N, Lucena E, Mattosinho C, Parareda A, Ferman S, Catalá J, et al. Use of intra-arterial chemotherapy for retinoblastoma: results of a survey. Int J Ophthalmol. 2014;7: 726–730. 10.3980/j.issn.2222-3959.2014.04.26 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Abramson DH. Chemosurgery for retinoblastoma: what we know after 5 years. Arch Ophthalmol. 2011;129: 1492–1494. 10.1001/archophthalmol.2011.354 [DOI] [PubMed] [Google Scholar]
- 5.Shields CL, Manjandavida FP, Lally SE, Pieretti G, Arepalli SA, Caywood EH, et al. Intra-arterial chemotherapy for retinoblastoma in 70 eyes: outcomes based on the international classification of retinoblastoma. Ophthalmol. 2014;121: 1453–1460. 10.1016/j.ophtha.2014.01.026 [DOI] [PubMed] [Google Scholar]
- 6.Schaiquevich P, Ceciliano A, Millan N, Taich P, Villasante F, Fandino AC, et al. Intra-arterial chemotherapy is more effective than sequential periocular and intravenous chemotherapy as salvage treatment for relapsed retinoblastoma. Pediatr Blood Cancer. 2013;60: 766–770. 10.1002/pbc.24356 [DOI] [PubMed] [Google Scholar]
- 7.Abramson DH. Super selective ophthalmic artery delivery of chemotherapy for intraocular retinoblastoma: “chemosurgery” the first Stallard lecture. Br J Ophthalmol. 2010;94: 396–399. 10.1136/bjo.2009.174268 [DOI] [PubMed] [Google Scholar]
- 8.Shields CL, Fulco EM, Arias JD, Alarcon C, Pelligrini M, Rishi P, et al. Retinoblastoma frontiers with intravenous, intra-arterial, periocular, and intravitreal chemotherapy. Eye (Lond). 2012. 10.1038/eye.2012.175 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Muen WJ, Kingston JE, Robertson F, Brew S, Sagoo MS, Reddy MA. Efficacy and Complications of Super-selective Intra-ophthalmic Artery Melphalan for the Treatment of Refractory Retinoblastoma. Ophthalmol. Elsevier Inc; 2012;119: 611–616. 10.1016/j.ophtha.2011.08.045 [DOI] [PubMed] [Google Scholar]
- 10.Abramson DH, Marr BP, Brodie S, Dunkel IJ, Gobin PY. Intraarterial chemotherapy for kissing macula tumors in retinoblastoma. Retin Cases Brief Rep. 2012;6: 209–211. 10.1097/ICB.0b013e318234ccf4 [DOI] [PubMed] [Google Scholar]
- 11.Abramson DH, Marr BP, Brodie SE, Dunkel I, Palioura S, Gobin YP. Ophthalmic artery chemosurgery for less advanced intraocular retinoblastoma: five year review. Abramson DH, Marr BP, Brodie SE, Dunkel I, Palioura S, Gobin YP, editors. PLoS ONE. 2012;7: e34120 10.1371/journal.pone.0034120.t003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Abramson DH, Dunkel IJ, Brodie SE, Marr B, Gobin YP. Bilateral superselective ophthalmic artery chemotherapy for bilateral retinoblastoma: tandem therapy. Arch Ophthalmol. 2010;128: 370–372. 10.1001/archophthalmol.2010.7 [DOI] [PubMed] [Google Scholar]
- 13.Abramson DH, Shields CL, Munier FL, Chantada GL. Treatment of Retinoblastoma in 2015: Agreement and Disagreement. JAMA Ophthalmol. 2015: 1–7. 10.1001/jamaophthalmol.2015.3108 [DOI] [PubMed] [Google Scholar]
- 14.Abramson DH, Marks RF, Ellsworth RM, Tretter P, Kitchin FD. The management of unilateral retinoblastoma without primary enucleation. Arch Ophthalmol. 1982;100: 1249–1252. 10.1001/archopht.1982.01030040227002 [DOI] [PubMed] [Google Scholar]
- 15.Reese AB, Hyman GA, Tapley ND, Forrest AW. The treatment of retinoblastoma by x-ray and triethylene melamine. AMA Arch Ophthalmol. 1958;60: 897–906. [DOI] [PubMed] [Google Scholar]
- 16.Abramson DH, Ellsworth RM, Tretter P, Javitt J, Kitchin FD. Treatment of bilateral groups I through III retinoblastoma with bilateral radiation. Arch Ophthalmol. 1981;99: 1761–1762. [DOI] [PubMed] [Google Scholar]
- 17.Abramson DH, Beaverson KL, Chang ST, Dunkel IJ, McCormick B. Outcome following initial external beam radiotherapy in patients with Reese-Ellsworth group Vb retinoblastoma. Arch Ophthalmol. 2004;122: 1316–1323. 10.1001/archopht.122.9.1316 [DOI] [PubMed] [Google Scholar]
- 18.Francis JH, Abramson DH, Marr BP, Brodie SE. Ocular manipulation reduces both ipsilateral and contralateral electroretinograms. Doc Ophthalmol. 2013. 10.1007/s10633-013-9391-0 [DOI] [PubMed] [Google Scholar]
- 19.Brodie SE, Paulus YM, Patel M, Gobin YP, Dunkel IJ, Marr BP, et al. ERG monitoring of retinal function during systemic chemotherapy for retinoblastoma. Br J Ophthalmol. 2012;96: 877–880. 10.1136/bjophthalmol-2011-301248 [DOI] [PubMed] [Google Scholar]
- 20.Liu CY, Jonna G, Francis JH, Marr BP, Abramson DH, Brodie SE. Non-selectivity of ERG reductions in eyes treated for retinoblastoma. Doc Ophthalmol. 2013. 10.1007/s10633-013-9416-8 [DOI] [PubMed] [Google Scholar]
- 21.Abramson DH. Retinoblastoma in the 20th century: past success and future challenges the Weisenfeld lecture. Investigative ophthalmology & visual science. 2005. pp. 2683–2691. 10.1167/iovs.04-1462 [DOI] [PubMed] [Google Scholar]
- 22.Abramson DH, Ellsworth RM, Zimmerman LE. Nonocular cancer in retinoblastoma survivors. Trans Sect Ophthalmol Am Acad Ophthalmol Otolaryngol. 1976;81: 454–457. [PubMed] [Google Scholar]
- 23.Gallie B. Canadian guidelines for retinoblastoma care. Can J Ophthalmol. 2009;44: 639–642. 10.1139/i09-229 [DOI] [PubMed] [Google Scholar]
- 24.Schefler AC, Abramson DH. Retinoblastoma: what is new in 2007–2008. Curr Opin Ophthalmology. 2008. [DOI] [PubMed] [Google Scholar]
- 25.Dunkel IJ, Shi W, Salvaggio K, Marr BP, Brodie SE, Gobin YP, et al. Risk factors for severe neutropenia following intra-arterial chemotherapy for intra-ocular retinoblastoma. PLoS ONE. 2014;9: e108692 10.1371/journal.pone.0108692 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Taich P, Ceciliano A, Buitrago E, Sampor C, Fandiño A, Villasante F, et al. Clinical pharmacokinetics of intra-arterial melphalan and topotecan combination in patients with retinoblastoma. Ophthalmol. 2014;121: 889–897. 10.1016/j.ophtha.2013.10.045 [DOI] [PubMed] [Google Scholar]
- 27.Murphree AL, Villablanca JG, Deegan WF, Sato JK, Malogolowkin M, Fisher A, et al. Chemotherapy plus local treatment in the management of intraocular retinoblastoma. Arch Ophthalmol. 1996;114: 1348–1356. [DOI] [PubMed] [Google Scholar]
- 28.Allen S, Wilson MW, Watkins A, Billups C, Qaddoumi I, Haik BH, et al. Comparison of two methods for carboplatin dosing in children with retinoblastoma. Pediatr Blood Cancer. 2010;55: 47–54. 10.1002/pbc.22467 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Shields CL, De Potter P, Himelstein BP, Shields JA, Meadows AT, Maris JM. Chemoreduction in the initial management of intraocular retinoblastoma. Arch Ophthalmol. 1996;114: 1330–1338. [DOI] [PubMed] [Google Scholar]
- 30.Dunkel IJ, Lee TC, Shi W, Beaverson KL, Novetsky D, Lyden D, et al. A phase II trial of carboplatin for intraocular retinoblastoma. Pediatr Blood Cancer. 2007;49: 643–648. 10.1002/pbc.21163 [DOI] [PubMed] [Google Scholar]
- 31.Abramson DH, Frank CM, Dunkel IJ. A phase I/II study of subconjunctival carboplatin for intraocular retinoblastoma. Ophthalmol. 1999;106: 1947–1950. 10.1016/S0161-6420(99)90406-2 [DOI] [PubMed] [Google Scholar]
- 32.Vanquill K, Dioguardi P, Tong C, Gilbert J, Aabergjr T, Grossniklaus H, et al. Subconjunctival Carboplatin in Fibrin Sealant in the Treatment of Transgenic Murine Retinoblastoma. Ophthalmol. 2005;112: 1151–1158. 10.1016/j.ophtha.2004.11.060 [DOI] [PubMed] [Google Scholar]
- 33.Marr BP, Brodie SE, Dunkel IJ, Gobin YP, Abramson DH. Three-drug intra-arterial chemotherapy using simultaneous carboplatin, topotecan and melphalan for intraocular retinoblastoma: preliminary results. Br J Ophthalmol. 2012;96: 1300–1303. 10.1136/bjophthalmol-2012-301925 [DOI] [PubMed] [Google Scholar]
- 34.Francis JH, Gobin YP, Dunkel IJ, Marr BP, Brodie SE, Jonna G, et al. Carboplatin +/- topotecan ophthalmic artery chemosurgery for intraocular retinoblastoma. PLoS ONE. 2013;8: e72441 10.1371/journal.pone.0072441 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Chantada GL, Fandino AC, Carcaboso AM, Lagomarsino E, de Davila MTG, Guitter MR, et al. A phase I study of periocular topotecan in children with intraocular retinoblastoma. Invest Ophthalmol Vis Sci. 2009;50: 1492–1496. 10.1167/iovs.08-2737 [DOI] [PubMed] [Google Scholar]
- 36.Carcaboso AM, Chiappetta DA, Opezzo JAW, Höcht C, Fandino AC, Croxatto JO, et al. Episcleral implants for topotecan delivery to the posterior segment of the eye. Invest Ophthalmol Vis Sci. 2010;51: 2126–2134. 10.1167/iovs.09-4050 [DOI] [PubMed] [Google Scholar]
- 37.Schaiquevich P, Carcaboso AM, Buitrago E, Taich P, Opezzo J, Bramuglia G, et al. Ocular pharmacology of topotecan and its activity in retinoblastoma. Retina (Philadelphia, Pa). 2014;34: 1719–1727. 10.1097/IAE.0000000000000253 [DOI] [PubMed] [Google Scholar]
- 38.Inomata M, Kaneko A. Chemosensitivity profiles of primary and cultured human retinoblastoma cells in a human tumor clonogenic assay. Jpn J Cancer Res. 1987;78: 858–868. [PubMed] [Google Scholar]
- 39.Brodie SE, Pierre Gobin Y, Dunkel IJ, Kim JW, Abramson DH. Persistence of retinal function after selective ophthalmic artery chemotherapy infusion for retinoblastoma. Doc Ophthalmol. 2009;119: 13–22. 10.1007/s10633-008-9164-3 [DOI] [PubMed] [Google Scholar]
- 40.de Jong MC, Kors WA, de Graaf P, Castelijns JA, Moll AC, Kivelä T. The Incidence of Trilateral Retinoblastoma: A Systematic Review and Meta-Analysis. American Journal of Ophthalmol. 2015;160: 1116–1126. e5 10.1016/j.ajo.2015.09.009 [DOI] [PubMed] [Google Scholar]
- 41.Suzuki S, Yamane T, Mohri M, Kaneko A. Selective ophthalmic arterial injection therapy for intraocular retinoblastoma: the long-term prognosis. Ophthalmol. 2011;118(10):2081–2087. 10.1016/j.ophtha.2011.03.013 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data contained within this study has been deidentified and will be shared by either the corresponding author or designated personnel upon written request and in accordance with MSK policies on privacy and data sharing. Please send all requests for data to the following address: David H. Abramson MD Memorial Sloan Kettering Cancer Center Pediatric Ophthalmic Oncology Suite A-330 1275 York Ave New York, NY 10065