Abstract
Objective
In HCV infected individuals graft infection occurs shortly after orthotopic liver transplantation (OLT). We aimed to describe the composition of the inflammatory response at this time, how it affects the HCV replication cycle and identify novel proviral and antiviral factors.
Design
We used a Luminex assay to quantify 50 inflammatory mediators in sera before and shortly after OLT. In vitro grown HCV based on the JFH-1 isolate were used to characterise the effects of patient sera and individual mediators on HCV.
Results
Although the mediator composition is highly variable between individuals, sera drawn immediately post-OLT significantly enhance HCV infectivity compared with control sera from before OLT in about half of the cases. Among 27 non-interferon inflammatory mediators fibroblast growth factor (FGF)-2 stood out as it enhanced HCV RNA replication and release of infectious particles. The effect was concentration-dependent and detectable in dividing and non-dividing cells. Moreover, pharmacological inhibition of FGF-2 receptor signalling abrogated the enhancing effect of FGF-2 and inhibited HCV replication in the absence of serum FGF-2 suggesting that HCV replication is dependent on basal activation of the FGF-2 triggered signalling pathway. Finally, in individuals with chronic HCV infection with high viral load, serum FGF-2 was significantly higher compared with those with low viral load.
Conclusions
Although no single mediator may account for this effect, serum shortly post-OLT enhances HCV infection. FGF-2 is a novel endogenous driver of HCV replication and a potential therapeutic target.
Keywords: HCV, HEPATITIS C, LIVER TRANSPLANTATION
Significance of this study.
What is already known on this subject?
Graft reinfection is near ubiquitous in HCV-positive orthotopic liver transplantation (OLT)-recipients and results in poor outcomes.
An intense inflammatory response driven by multiple simultaneous biological events forms the backdrop of graft reinfection by viral particles from the blood happening within hours to days after OLT.
Extracellular mediators such as epidermal growth factor (EGF) or transforming growth factor (TGF) that stimulate cellular signalling cascades thereby modulate the HCV replication cycle.
What are the new findings?
The mediator composition in acute phase sera from the immediate post-OLT period is highly variable between individuals, but in about half of cases acute phase sera stimulates HCV replication. In this study all except one serum with HCV replication enhancing properties were from individuals with viral hepatitis.
Extracellular fibroblast growth factor-2 (FGF-2) and the cellular signalling cascade triggered by it are novel host factors that promote HCV replication and new particle production.
The proviral effect of FGF-2 on HCV replication is independent of cellular proliferation and likely mediated through FGF receptor 3.
FGF-2 serum levels are increased in individuals with chronic HCV infection with high viral load compared with those with low viral load.
How might it impact on clinical practice in the foreseeable future?
Measures to counteract the proviral effect of post-OLT sera may help to prevent or attenuate graft reinfection in HCV-positive recipients. Moreover, our work identifies FGF-2 signalling as a potential antiviral target for the treatment of HCV infection.
Introduction
Worldwide 160 million people are chronically infected with HCV and therefore are at risk to develop liver cirrhosis or hepatocellular carcinoma (HCC).1 Accordingly, end-stage liver disease due to chronic HCV infection is the major indication for orthotopic liver transplantation (OLT) worldwide.2
The liver is thought to be the major or even the only site of HCV replication in vivo. Extrahepatic reservoirs in lymphatic or neuronal cells have been proposed but their significance remains unclear. Conversely, transplantation of human hepatocytes into immunodeficient mice is sufficient to render them susceptible to infection.3 OLT generally does not cure HCV infection and nearly all patients develop graft reinfection.2 This may change for some if highly potent new direct acting antivirals are confirmed to be safe for use in the transplant setting, but treatment of certain patient groups such as those with impaired renal function is likely to remain problematic for some time to come. In patients with viraemia, viral load drops shortly after transplantation because with the explanted liver the major production site of viral particles has been removed.4 Yet, the liver graft is quickly infected by virions from the blood and viral load rises again to somewhat above pre-OLT levels within a few weeks.4 The consequences of graft reinfection are dire: 28% of patients develop graft cirrhosis within 5 years after OLT and decompensation tends to occur early.5 Therefore, the outcome of HCV-infected OLT recipients is worse in comparison with patients transplanted for end-stage liver disease of other causes.6 In some cases reinfection takes a severe cholestatic course with very high viral load, progressive jaundice and a high risk for rapid graft failure.7
The HCV replication cycle has been linked to cellular signalling networks that are triggered by mediators present in the extracellular space such as epidermal growth factor and transforming growth factors α and β.8 9 During the peritransplant period, an intense inflammatory response occurs driven by a number of stimuli that include surgical trauma, graft reperfusion, antigraft immune response, immunosuppression and concomitant infections. There is limited data describing the exact composition of the inflammatory reaction following liver transplantation.10 11 12 Moreover, how this ‘cytokine storm’ type milieu affects the ability of the virus to reinfect the graft and whether it contributes to more severe forms of graft hepatitis is unknown.
This study was conducted to investigate how the inflammatory response that occurs in the peritransplantation period modulates the HCV replication cycle and whether it may contribute to the poor outcome of HCV-positive individuals after OLT.
Materials and methods
Compounds
All soluble mediators used in this study (see online supplementary table S1) were purchased from Biomol (Hamburg, Germany). AZD4547, SU5402, Masitinib and PD173074 were purchased from Toronto Research Chemicals (Ontario, Canada).
Patient characteristics
Sera from four patient cohorts or control groups were examined as part of this study: (1) Thirteen patients undergoing OLT at Hannover Medical School between 2011 and 2013 for decompensated cirrhosis of different aetiologies (see Table 1). From these 13 patients, paired sera were collected before OLT and within 1 day to 3 days after OLT. (2) Seventy non-cirrhotic patients with chronic HCV infection. (3) Fourteen additional patients with decompensated cirrhosis of different aetiologies that had not yet undergone OLT. (4) Ten healthy blood donors that served as controls.
Table 1.
Patient characteristics
| No. | Sex | Age at OLT | Diagnosis |
|---|---|---|---|
| 1 | Male | 60 | HCV |
| 2 | Male | 59 | HCV |
| 3 | Male | 52 | HCV |
| 4 | Male | 44 | HBV/HDV |
| 5 | Female | 61 | HBV/HDV |
| 6 | Female | 36 | HBV/HDV |
| 7 | Female | 58 | HBV/HDV |
| 8 | Male | 57 | HBV/HDV |
| 9 | Male | 58 | Alcoholic liver disease |
| 10 | Male | 56 | Cryptogenic cirrhosis |
| 11 | Male | 61 | Cryptogenic cirrhosis with HCC |
| 12 | Male | 56 | Primary sclerosing cholangitis |
| 13 | Female | 49 | Polycystic liver disease |
Basic clinical features of 13 OLT recipients evaluated in this study.
HCC, hepatocellular carcinoma; HDV, chronic hepatitis D; OLT, orthotopic liver transplantation.
Cell culture and cell lines
Huh-7.5, Huh-6 cells and Lunet cells were maintained in Dulbecco's modified eagle medium (DMEM; Invitrogen, Karlsruhe, Germany) supplemented with 10% fetal bovine serum, L-glutamine, non-essential amino acids, penicillin and streptomycin (Invitrogen). Dimethylsulfoxide (DMSO) differentiation of Huh-7.5 cells was performed according to the method by Sainz and Chisari.13 Isolation and maintenance of primary human hepatocytes (PHHs) has previously been described.14
Plasmids
The genotype 2a reporter virus genome Luc-Jc1 and expression plasmids for HCV E1/E2 proteins of the Con1 isolate (genotype 1b) or H77 (genotype 1a) have been described before.15 A pLenti3-based construct for fibroblast growth factor (FGF) receptor 3 (FGFR3) overexpression was from Origene (Maryland, USA). Short hairpin RNA (shRNA) constructs in the pGIPZ vector were purchased from Dharmacon—GE Healthcare (Freiburg, Germany).
Luminex assay
A total of 50 chemokines and growth factors in sera of patients who underwent liver transplantation were quantified by Luminex-based multiplex assay according to the manufacturer’s instruction (Bio-Rad, USA). The detection range of all cytokines was between 0.1 pg/mL and 40 µg/mL. Only data points where ‘pre-OLT’ and ‘post-OLT’ measurements for any given patient and mediator were technically successful and within the dynamic range of the assay were included in the analysis.
Preparation of lentiviral pseudoparticles and HCVpp infection
Lentiviral pseudoparticles encoding a luciferase reporter were generated as previously described.16
HCV replication and infection assay
Huh-7.5 cells were transfected with HCV genome by electroporation as previously described.17 Five hours post transfection, different concentrations of mediators were added and 48 h post transfection HCV replication was quantified by measuring luciferase activity. To investigate HCV assembly and release of new HCVcc particles, supernatants were collected 48 h post transfection with Luc-Jc1, filtered through 0.45 µm pore size filters, and used to infect target cells. Target cells were inoculated with 500 µL virus-containing supernatant in 12-well format. HCV infection was quantified by measuring luciferase activity. For infection experiments, HCVcc particles (Luc-Jc1) were produced in the absence of any mediators, filtered and used to infect naïve Huh-7.5 cells in the presence of mediators. After 6 h cells were washed with phosphate-buffered saline and medium was replaced. In some experiments patients’ serum was heated up to 56°C for 15 min and diluted to a total concentration of 3%. Serum was added 6 h after infection with HCVcc.
Luciferase assay
Cells were washed with phosphate-buffered saline, lysed directly on the plate with either 1 mL or 350 µL lysate buffer (0.1% Triton X-100, 25 mmol/L glycylglycine, 15 mmol/L MgSO4, 4 mmol/L ethylene glycoltetra-acetic acid (EGTA) and 1 mmol/L dithiothreitol (DTT); pH 7.8) and frozen to −20°C. For quantifying luciferase activity, 100 µL defrosted lysate was mixed with 360 µL assay buffer (25 mmol/L glycylglycine, 15 mmol/L MgSO4, 4 mmol/L EGTA, 1 mmol/L DTT, 2 mmol/L ATP, 15 mmol/L K2PO4, pH 7.8) and combined with 200 µL luciferin solution (200 µmol/L luciferin, 25 mmol/L glycylglycine, pH 8.0) in a luminometer (Lumat LB9508; Berthold, Freiburg, Germany) measuring luciferase activity for 20 s. All measurements were performed in duplicates for HCV replication and in quadruplicates for HCV infection.
Core ELISA
Intracellular and extracellular HCV cores were measured by a commercially available ELISA (Architect HCV Ag Assay; Abbott Laboratories, Abbott Park, Illinois, USA) as previously described.18
Western blot
Cells were lysed in sample buffer as previously described.15 Proteins were resolved by electrophoresis and blotted onto a polyvinylidene difluoride membrane, and detected with commercially available antibodies against FGFRs 1, 2, 3 and 4 (Cell Signaling Technology, Frankfurt am Main, Germany) or anti-ß-actin (Sigma-Aldrich, Germany). The ECL Plus western blotting detection system (GE Healthcare Europe, Freiburg, Germany) was used for final protein detection.
Cell toxicity and proliferation assay
Three complementary approaches were used to assess effects on cell viability and proliferation. First, as previously described, mediators were added to Huh-7.5 cells stably expressing luciferase from a cytomegalovirus (CMV) promoter.16 After 48 h cells were lysed and luciferase activity was measured. This served as an aggregate measure of growth and proliferation. Second, effects on cell proliferation were measured using the 5-ethynyl-2′-deoxyuridine flow cytometry kit (Baseclick GmbH, Tutzing, Germany) as indicated in the manufacturer's manuals. It uses incorporation of the nucleoside analogue 5-ethynyl-2′-deoxyuridine into DNA and binding it to a fluorescent dye that can be detected by flow cytometry. Third, toxic effects were tested for using the Cyto Tox-Glo (Promega GmbH, Mannheim, Germany), as indicated in the manufacturer's manuals. It takes advantage of release of proteases from cells with compromised membrane integrity. These proteases then cleave a luminogenic substrate.
FGF-2 ELISA
Serum FGF-2 was quantified using a commercial ELISA (R&D Systems, Wiesbaden Nordenstadt, Germany), as indicated in the manufacturer's manuals.
Statistical analyses
Unless stated otherwise the mean±SD of the replicates from one representative of several independent experiments is shown. The number of replicates is indicated. Significant differences were identified using the paired or unpaired two-sided Student's t test as appropriate. Differences with p<0.05 were considered statistically significant and indicated by an asterisk. When multiple comparisons were made the p-threshold was adjusted by dividing it through the number of independent hypotheses being tested in parallel (Bonferroni adjustment).
Results
Peritransplant sera enhance HCV infectivity
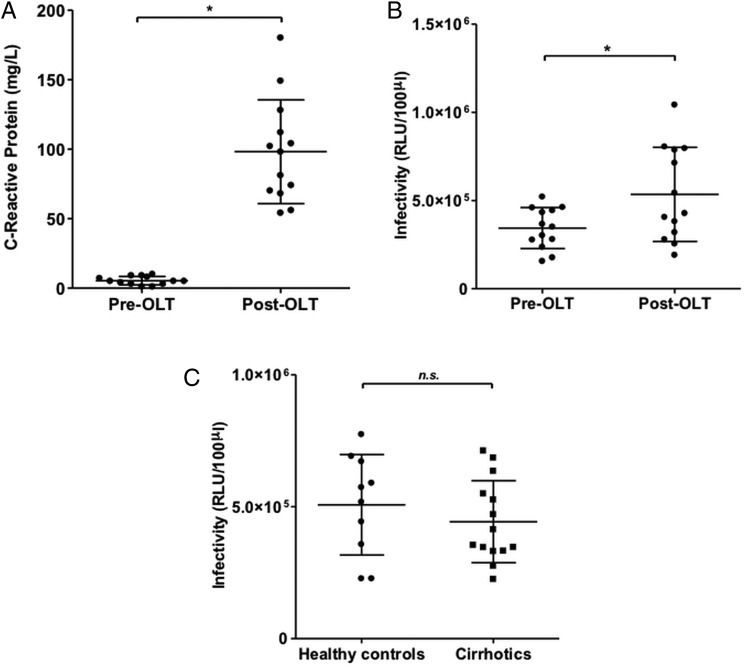
We collected paired sera within 60 days before OLT (‘pre-OLT’) and within 72 h after OLT (‘post-OLT’) from 13 patients who underwent transplantation between 2011 and 2013 at Hannover Medical School because of end-stage chronic liver disease of different aetiologies (table 1). Individuals with severe systemic inflammatory states such as acute liver failure at the time of transplantation were excluded. Accordingly, a marked rise in C reactive protein (CRP) as a global marker of systemic inflammation was seen in all individuals between the pre-OLT and post-OLT time points (figure 1A). At the post-OLT time point all patients had received an interleukin (IL) 2 receptor antibody, a calcineurin inhibitor and glucocorticosteroids for immunosuppression. When sera were added at 3% dilution during infection of naïve Huh-7.5 cells with HCVcc we observed that overall post-OLT sera modestly but significantly increased infectivity (figure 1B). There was however marked variation between individuals: one out of three individuals with hepatitis C (see online supplementary figure S1A) and four out of five individuals with hepatitis B or B/D (see online supplementary figure S1B) showed a greater than 2.5-fold increase in infectivity, while this was only the case in one out of five individuals with non-viral liver disease (see online supplementary figure S1C). To test whether the observed difference between pre-OLT and post-OLT sera might be due to an inhibitory serum factor present in the setting of end-stage liver disease rather than an enhancing factor present shortly after OLT we collected additional sera from 14 individuals with decompensated cirrhosis and from 10 healthy controls. When sera from the two latter groups were added to HCV infections performed in vitro there was no difference in infectivity (figure 1C). These findings suggest that sera from the post-OLT time point contain one or more factor(s) that enhance HCV infectivity. Whether this effect is indeed more common in individuals with viral hepatitis or more specifically hepatitis B/D would need to be confirmed in a larger cohort.
Figure 1.
Postorthotopic liver transplantation (OLT) sera show increased inflammatory activity and enhance HCV infectivity in vitro. (A) C reactive protein values in patients’ serum before OLT (pre-OLT) and after OLT (post-OLT). (B) Naïve Huh-7.5 cells were inoculated with Fluc-Jc1 HCV cell culture grown particles (HCVcc) in the presence of 3% heat-inactivated patient serum from the sera shown in panel A. Dots represent the mean of n=4 replicates from two independent experiments. Paired Student's t test was performed to test for significance. (C) Effect on HCV infectivity of sera from healthy controls and from 14 additional individuals with decompensated cirrhosis that had not yet undergone OLT (patients with cirrhosis). Dots represent the mean of n=6 replicates from three independent experiments. Unpaired Student's t test was performed to test for significance.
Serum chemokines during the peritransplant period
To characterise the serum chemokine profile in the immediate post-transplant period we performed multiplex analysis (Luminex) of the above paired serum samples from the pre-OLT and post-OLT time points for 50 different non-interferon inflammatory mediators. Different from the CRP, the change in individual mediators between the two time points was highly heterogeneous between patients. Out of 50 mediators we observed a greater than 50% increase of the median concentration for 7 (macrophage migration inhibitor factor (MIF), IL-2Ra, IL-6, vascular endothelial growth factor (VEGF), monocyte chemotactic protein-1 (MCP-1), monokine induced by gamma interferon or CXCL-9 (MIG), platelet-derived growth factor - bb chains or subunit β (PDGF-bb)) and a greater than 50% decrease in 2 (TNF-b, IL-5) (see online supplementary figure S2). However, the change did not reach statistical significance for any of the mediators tested except CRP based on paired t-test with Bonferroni adjustment for 50 parallel comparisons. Also there was no apparent association with the aetiology of liver disease (hepatitis B/D vs hepatitis C vs non-viral aetiologies) nor was there any clear pattern of mediators seen in those individuals whose post-OLT sera enhanced HCV in vitro infectivity (data not shown).
Influence of individual mediators on the HCV replication cycle
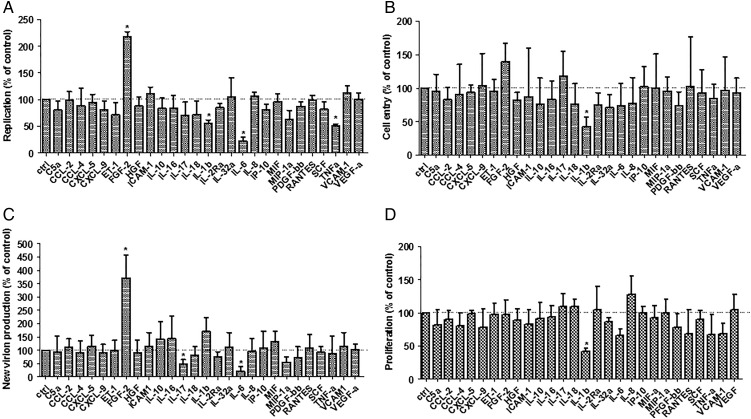
To dissect whether individual mediators have unknown proviral effects we assembled a set of 27 mediators based on our Luminex data and a literature search for mediators that have been reported to be central to the inflammatory response in the immediate post-OLT phase (see online supplementary table S1). These were screened for their effect on HCVcc replication (figure 2A), cell entry (figure 2B) and new particle production (figure 2C) as well as cell growth (figure 2D). While most mediators individually had no or a modest inhibitory effect on the different stages of the HCV replication cycle, FGF-2, also known as FGF-β, was found to markedly enhance viral replication and new particle production. None of the mediators had a significant enhancing or inhibitory effect on HCV pseudoparticles (HCVpp) entry (data not shown). As shown in online supplementary figure S3 the FGF-2 serum concentrations measured in our transplant patient cohort were less than 100 pg/mL in most cases while a minority had values in the 100–500 pg/mL range.
Figure 2.
Screen of 27 non-interferon inflammatory mediators for modulation of HCV replication cycle stages. (A) To evaluate the effects of mediators on HCVcc replication Huh-7.5 cells were electroporated with Fluc-Jc1 RNA and 5 h later exposed to media supplemented with the respective of mediators. Cells were lysed and luciferase measured after 48 h of incubation. Data for the highest concentration tested is shown; the concentration ranges used for each mediator are stated in online supplementary table S1. (B) To evaluate effects on HCV entry Huh-7.5 cells were exposed to Fluc-Jc1 HCVcc in the presence of mediators for 6 h. Then cells were washed and media without mediators was added. Cells were lysed and measured 48 h later. (C) To evaluate effects on new virion production, supernatants from the experiment represented in panel A were collected 48 h after electroporation, filtered and used to infect naïve Huh-7.5 cells. (D) Effects on cell viability and proliferation were assessed by exposing Huh-7.5 cells stably expressing firefly luciferase to mediators for 48 h. Then cells were lysed and luciferase activity was quantified. In each panel, bars represent the mean±SD of a minimum of n=6 replicates from three independent experiments with the mean value obtained in the absence of mediators set to 100%.
FGF-2 enhances HCV RNA replication and production of new viral particles in a dose-dependent manner
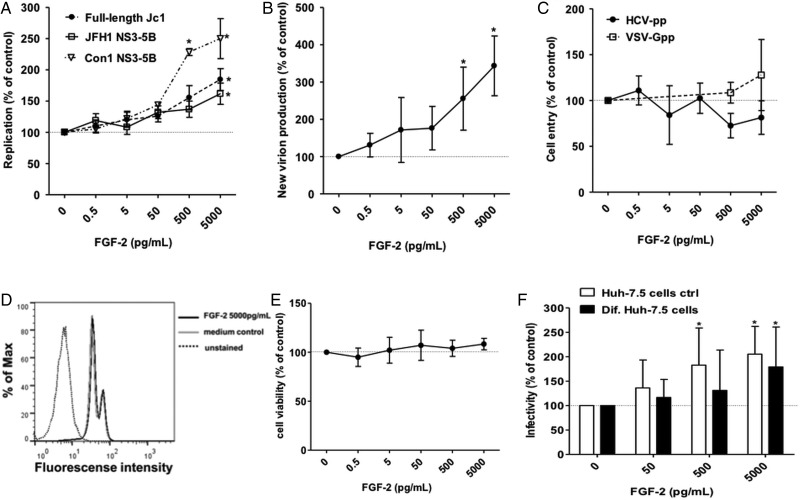
We proceeded to characterise the effect of FGF-2 on the HCV replication cycle in more detail. When Huh-7.5 cells were electroporated with in vitro transcribed HCV genomes and then exposed to FGF-2 concentrations of up to 5 ng/mL there was a concentration-dependent increase of HCV replication using the chimerical genotype 2a Jc1 genome as well as subgenomic NS3-5B genomes of genotypes 1 (Con1 isolate) and 2 (JFH-1 isolate) (figure 3A). Moreover, release of infectious particles from Huh-7.5 cells harbouring a full-length Jc1 genome was increased in the presence of FGF-2 in a concentration-dependent manner (figure 3B). Intracellular and extracellular HCV cores were similarly increased in the presence of FGF-2 suggesting that viral particle production driven by increased replication rather than release of new viral particles may primarily be affected (see online supplementary figure S4). Conversely, the signal obtained with HCV-pseudotyped lentiviral particles was not affected largely ruling out a stimulatory effect of FGF-2 on HCV entry (figure 3C). FGF-2 had no detectable effect on cell proliferation (figure 3D) or cell viability (figure 3E) at the concentrations used. Since FGF-2 is a known stimulator of cell growth we differentiated Huh-7.5 cells by DMSO treatment. Differentiated Huh-7.5 cells stop dividing and have a more ‘hepatocyte-like’ phenotype with production of liver-specific proteins like albumin. FGF-2 treatment increased HCV RNA replication in differentiated and regular Huh-7.5 cells indicating that the effect of FGF-2 is independent of cell proliferation (figure 3F).
Figure 3.
Fibroblast growth factor-2 (FGF-2) enhances HCVcc replication and new particle production in a concentration dependent manner. (A) Huh-7.5 cells were electroporated with the indicated Fluc-reporter subgenomic or full-length HCV genomes RNA. Five hours later media supplemented with the indicated concentration of FGF-2 was added. Cells were lysed and luciferase measured after 48 h of incubation. (B) Supernatants from the full-length Jc1 experiment represented in panel A were collected 48 h after electroporation, filtered and used to infect naïve Huh-7.5 cells. After 6 h media was exchanged and the cells were incubated without FGF-2 for 48 h before lysis and quantification of luciferase activity. (C) Huh-7.5 cells were transduced with HCVpp (clone H77) and VSV-Gpp in the presence of FGF-2 at the indicated concentrations. (D) Huh-7.5 cells were exposed to FGF-2 for 48 h. Then cells were fixed and effect of FGF-2 on cell proliferation was assessed by reading incorporation of 5-ethynyl-2′-deoxyuridine into cellular DNA by flow cytometry. The analysed image is of the highest tested concentration of FGF-2. Medium without FGF-2 was used as control in parallel. (E) Huh-7.5 cells were exposed to FGF-2 for 48 h. Then the effect of FGF-2 on cell viability was assessed by detecting release of intracellular proteases using the CytoTox-Glo Cytotoxicity Assay. (F) Differentiated non-dividing Huh-7.5 cells were infected with Fluc-Jc1 HCVcc. After 6 h cells were washed and treated with FGF-2 at the indicated concentrations. After 48 h of incubation cells were lysed and luciferase expression was quantified. Regular dividing Huh-7.5 cells were used as control in parallel. In each panel, dots or bars represent the mean±SD of minimum n=6 replicates from three independent experiments with the mean value obtained in the absence of FGF-2 set to 100%.
The closely related FGF α does not enhance HCV RNA replication
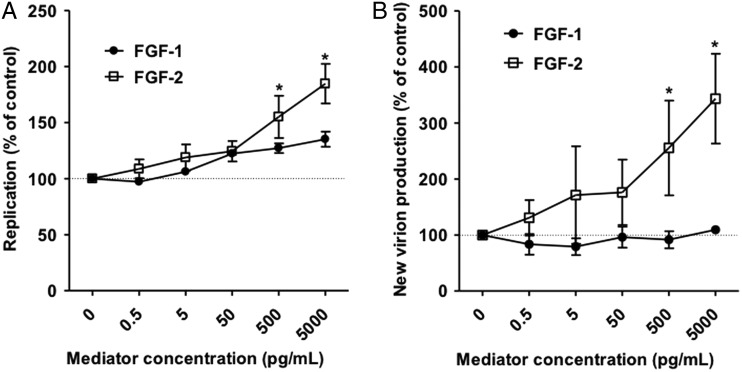
FGF-2 is part of a family of 22 FGFs; all are structurally related. In order to find out how specific the observed proviral effect of FGF-2 is, we compared it with FGF-1 (also known as FGF-α) the closest relative of FGF-2. In contrast to FGF-2, FGF-1 did not significantly increase HCV RNA replication (figure 4A) or release of infectious units (figure 4B).
Figure 4.
The fibroblast growth factor-2 (FGF-2) related mediator FGF-1 does not enhance HCV. (A) Huh-7.5 cells were electroporated with the Fluc-Jc1. Six hours later media supplemented with the indicated concentration of FGF-1 or FGF-2 was added. Cells were lysed and luciferase measured after 48 h of incubation. (B) Supernatants from the experiment represented in panel A were collected 48 h after electroporation, filtered and used to infect naïve Huh-7.5 cells. After 6 h media was exchanged and the cells were incubated without any mediators for 48 h before lysis and quantification of luciferase activity. In all panels dots represent the mean±SD of n=4 replicates from two independent experiments with the mean value obtained without any mediator set to 100%.
FGF-2 stimulation of HCV replication is independent of cell proliferation and may be linked to expression of FGF receptor 3
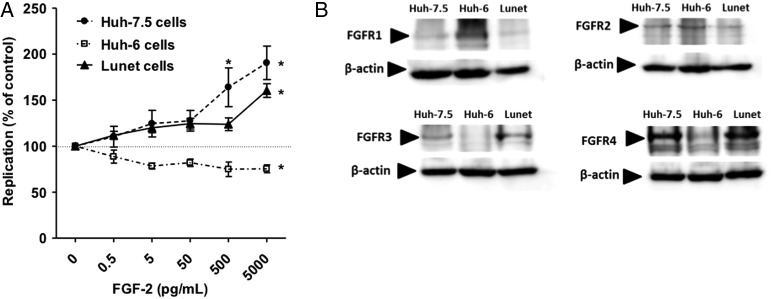
To further explore how FGF-2 stimulates HCV replication, we electroporated three different hepatoma cell lines with a reporter Jc1 genome. While HCV replication was similarly increased by FGF-2 in Lunet and in Huh-7.5 we did not detect this effect in Huh6 cells (figure 5A). FGF-2 binds to four different FGFRs1, 2, 3, 4 on the cell surface. When probing FGFR expression in the above cell lines by immunoblotting we found that Huh6 cells differed from Huh-7.5 and Lunet in that they expressed more FGFR1, less FGFR4 and no detectable FGFR3 (figure 5B). High expression of FGFR3 in Huh-7.5 cells as well as PHHs was also confirmed by microarray (data not shown).
Figure 5.
Fibroblast growth factor-2 (FGF-2) enhancement of HCV replication is dependent on host cell context. (A) Huh-7.5, Lunet and Huh-6 cells were electroporated with Fluc-Jc1 RNA. Five hours after transfection medium containing FGF-2 was added. Dots represent the mean±SD of n=6 independent replicates from three independent experiments. (B) Expression of FGF receptors (FGFRs) 1, 2, 3 and 4 in Huh-7.5, Lunet and Huh6 cells were assessed by immunoblotting. Actin served as loading control.
Stimulation of HCV replication by FGF-2 is dependent on FGFR signalling
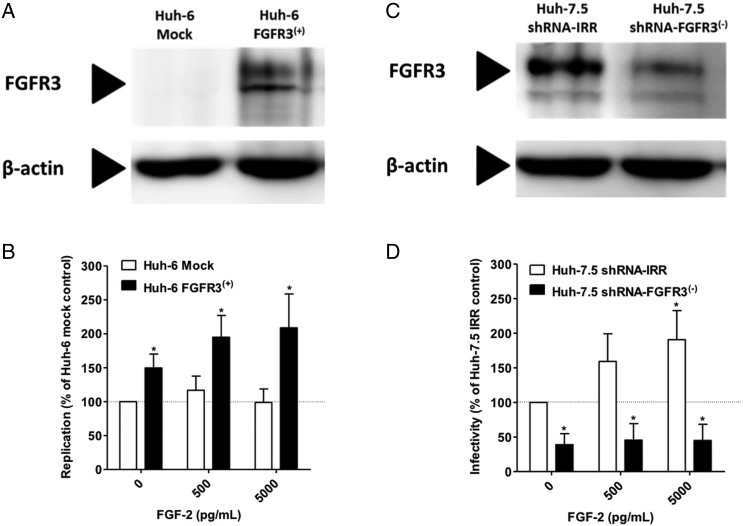
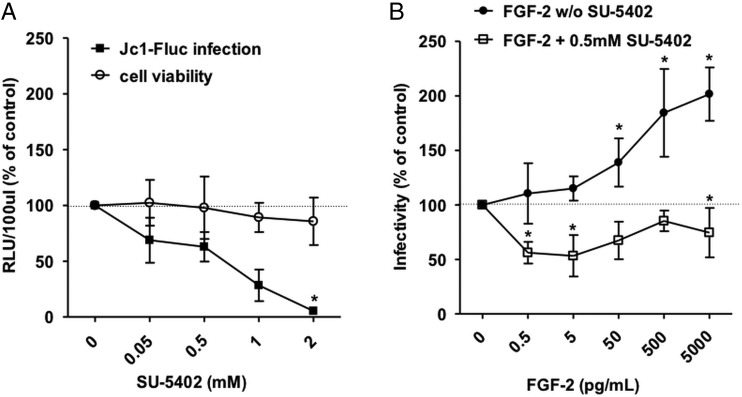
To test the hypothesis that the observed effect is dependent on signalling through FGFR3 we transduced Huh6 cells to express FGFR3 (figure 6A). When electroporated with a full length HCV genome (Jc1) Huh6/FGFR3 cells showed modestly but significantly higher luciferase counts compared with Huh6/mock controls and replication could be further stimulated by addition of FGF-2 (figure 6B). Conversely, when FGFR3 levels were reduced by introduction of a specific shRNA into Huh-7.5 cells (figure 6C) HCV infectivity was decreased compared with Huh-7.5 cells harbouring an irrelevant control shRNA and the stimulatory effect of FGF-2 on HCV replication was abrogated (figure 6D). We then infected Huh-7.5 cells with HCVcc and then treated them with the FGFR inhibitor SU-5402. There was a concentration-dependent inhibition of HCV replication while cell viability was largely unaffected (figure 7A). When infected cells were exposed to increasing concentrations of FGF-2 in the presence of a subinhibitory concentration of SU-5402 the enhancing effect of FGF-2 was abolished (figure 7B). Collectively, these data support that FGF-2 stimulates HCV replication by signalling through FGFR3.
Figure 6.
Fibroblast growth factor-2 (FGF-2) stimulation of HCV replication is dependent on expression of FGF receptor 3 (FGFR3). (A) Normally FGFR3-negative Huh6 cells were transduced with a lentiviral vector encoding FGFR3 or mock. FGFR3 expression was then assessed by immunoblotting. (B) Huh-6/mock and Huh6/FGFR3 cells were electroporated with the Fluc-Jc1. Five hours later media supplemented with the indicated concentration of FGF-2 was added. Cells were lysed and luciferase measured after 48 h of incubation. (C) Normally FGFR3-positive Huh-7.5 cells were transduced to express a short hairpin RNA (shRNA) against FGFR3 or an irrelevant control shRNA. FGFR3 expression was then assessed by immunoblotting. (D) The cells shown in panel C were infected with HCVcc and exposed to the indicated concentrations of FGF-2. Cells were lysed and luciferase measured after 48 h of incubation. In all panels bars represent the mean±SD of n=6 replicates from three independent experiments with the mean value obtained without FGF-2 on control cell lines set to 100%.
Figure 7.
Pharmacological inhibition of fibroblast growth factor receptor (FGFR) signalling decreases HCV replication. (A) Huh-7.5 cells were infected with Fluc-Jc1 HCVcc. After 6 hours the indicated concentrations of SU-5402 were added. After 48 h, cells were lysed and effect on HCV infectivity was quantified by luciferase assay. In parallel naïve Huh-7.5 cells were exposed to the same concentrations of drug for the same time and toxicity was assessed using the CytoTox-Glo Cytotoxicity Assay. (B) HCVcc infected cells were exposed to increasing concentrations of FGF-2 in the presence or absence of 0.5 mM SU-5402. In all panels dots represent the mean±SD of n=6 replicates from three independent experiments with the mean value obtained without any mediator set to 100%.
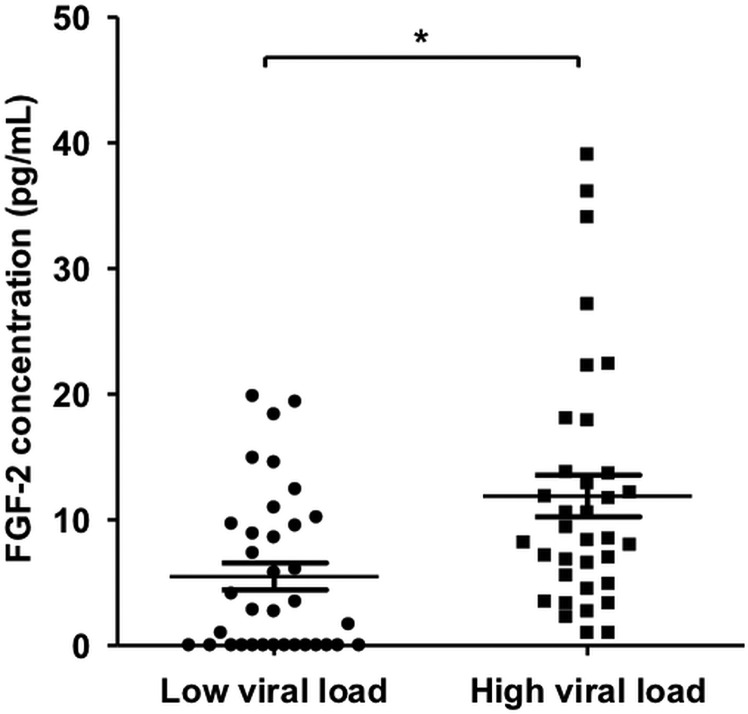
Serum FGF-2 is increased in HCV-infected individuals with high viral load
Finally, we were interested to see if there is an association between serum FGF-2 and viral replication in vivo in individuals with chronic HCV infection. For this purpose, we measured serum FGF-2 in 70 individuals with chronic HCV infection each with low (<100 000 IU/mL; n=35) or high (>1 000 000 IU/mL; n=35) viral load. Serum FGF-2 levels were found to be significantly higher in individuals with high viral load (figure 8).
Figure 8.
Fibroblast growth factor-2 (FGF-2) level is correlated with HCV RNA level in chronic infection. FGF-2 levels were determined by ELISA in sera from individuals with chronic HCV infection with high (>1.000.000 IU/mL; n=35) or low (<100.000 IU/mL; n=35) viral load. Patients were not currently under treatment and no orthotopic liver transplantation recipients were included.
Discussion
In this work we used paired sera from pre-OLT and immediately post-OLT time points to characterise the serum mediator composition at the time of HCV graft reinfection and its impact on the viral replication cycle. We found that despite strong interindividual variability in about half of individuals the post-OLT cytokine milieu enhances HCV infectivity. Moreover, we identified FGF-2 as a novel soluble endogenous enhancer of HCV RNA replication and production of new viral particles. Finally, we show that the proviral effect of FGF-2 is dependent on signalling through FGFR3.
While all individuals in our cohort showed a clinically expected rise in serum CRP as evidence of a systemic inflammatory response in the post-OLT phase, our Luminex data show that at the same time the mediator composition in the serum varies widely between individuals. Clearly, our cohort is small and contains individuals with different forms of liver disease including viral and non-viral aetiologies. A larger and more homogeneous cohort might reveal a clearer inflammatory response pattern. On the other hand, the heterogeneity we observed may simply reflect that the immediately post-OLT phase is a highly complex clinical situation where several biologically significant events occur simultaneously and no two patients may ever be exactly alike. Thus it may be difficult to define a prototypical post-OLT inflammatory pattern of serum markers. A recent study using Luminex to quantify 25 mediators in 73 OLT recipients in order to identify markers of early allograft dysfunction also showed a significant degree of variability.10 Yet, despite this heterogeneity we observed that in more than half of individuals the post-OLT serum enhanced HCV infectivity compared with the matched pre-OLT sample. Although the sample is too small to draw definitive conclusions it is interesting to note that the majority of sera with enhancing activity were from individuals with viral hepatitis, most notably hepatitis B/D. Again, a larger cohort would need to be examined to determine whether this effect is truly more common in individuals with viral hepatitis or hepatitis B/D compared with individuals with end-stage liver disease of non-viral aetiology. In our data set no single mediator was able to predict the enhancement of HCV infectivity observed in some patients. This may be because the enhancement is due either to a combination of factors or a serum factor that is not included in our assay that measures 50 key inflammatory mediators. Thus, the ‘cytokine storm’ at the time of graft reinfection by HCV is complex and heterogeneous between individuals, but it does appear to enhance HCV infectivity in about half of individuals and may thus promote reinfection including severe forms such as cholestatic hepatitis.
Among the mediators we screened directly for their effect on HCV replication cycle, FGF-2 stood out as the only proviral mediator. While all other mediators had either no effect or inhibited HCV replication, FGF-2 enhanced genome replication and release of new particles. To our knowledge FGF family members or the signalling cascades they trigger have not previously been linked to HCV replication. The concentrations we used to elicit this effect were higher than those detected in sera from individuals with chronic HCV infection. However, tissue levels in the liver graft tissue shortly after OLT may well be higher given that FGF-2 has been proposed to be released in response to tissue injury.19 More importantly, we were also able to show that pharmacological blockade of FGFR-mediated signalling inhibits HCV in the absence of exogenous FGF in the supernatant suggesting that viral replication is indeed dependent on a basal degree of activation of FGFR-mediated signalling pathways. In addition we were able to show that serum FGF-2 levels are higher in individuals with chronic HCV infection with high viral load compared with those with low viral load. Although this association does not prove causality it does support the concept of a link between FGF-2 signalling and HCV replication in vivo.
First identified in the 1980s, FGF-2 is the prototypical member of the FGF family.20 21 Its low molecular weight (17–18 kDa) form that was used in this study is—differently from the nuclear high molecular weight forms—physiologically located in the cytosol and can be released from the cell into the extracellular space and thus be found in the serum.22 FGF-2 has been reported to mediate diverse biological processes including angiogenesis, vascular remodelling, cardiac hypertrophy, fracture healing, tumour growth and long-term memory.22–25 However, to our knowledge it has not previously been linked to viral replication. In the liver, FGF-2 has been found to be a driver of HCC development and vascularisation26 27 and elevated serum FGF-2 seems to predict recurrence and poor prognosis in patients with HCC.28 Brivanib, an inhibitor of vascular endothelial growth factor and FGFR-mediated signalling has been evaluated in a phase 3 trial in HCC, but failed to improve survival.29 Although in patients with advanced HCC it may not be relevant clinically, it would be interesting from a biological point of view to see if HCV viral load declined under brivanib. However, despite the link between FGF-2 signalling and cell proliferation we found that the enhancing effect on HCV replication was similar in non-dividing cells suggesting that a different mechanism is responsible. Moreover, our data show that FGF-2 stimulation of HCV replication can be blocked by inhibition of the tyrosine kinase activity of FGFRs and that it is dependent on the presence of FGFR3, one of four distinct FGFRs that are stimulated by FGF-2. High expression of FGFR3 compared with other FGFRs detected by microarray in human PHHs suggests that this mechanism might also be active in the human liver in vivo. Which exact downstream events triggered by FGF-2 and FGFR3 are required for HCV replication is the subject of ongoing studies.
In conclusion, we provide a characterisation of the cytokine milieu immediately following OLT when HCV graft infection occurs and show that in many cases its sum effect is proviral with regards to HCV replication. Moreover, we identify extracellular FGF-2 and the cellular signalling cascade triggered by it through FGFR3 as a previously unknown precondition of efficient HCV replication.
Supplementary Material
Acknowledgments
The authors thank Oliver Dittrich-Breiholz and the MHH Transcriptomics Core Facility for performance of the microarray analysis and Katrin Rohrmann for technical support.
Footnotes
Correction notice: This article has been corrected since it published Online First. Lisa Sandmann has been added as co-author.
Contributors: NDV performed the experiments. SC and TvH performed the overall statistical analyses, designed and wrote the paper. CSF analysed the Luminex data. FWRV provided the primary human hepatocytes. FH, MPM and HW provided patient serum samples.
Funding: This work was supported by a grant from the TUI Foundation to TvH and by the Germany Center for Infection Research (DZIF).
Competing interests: None.
Patient consent: Obtained.
Ethics approval: Ethics Committee Approval MHH.
Provenance and peer review: Not commissioned; externally peer reviewed.
References
- 1.Lavanchy D. Evolving epidemiology of hepatitis C virus. Clin Microbiol Infect 2011;17:107–15. 10.1111/j.1469-0691.2010.03432.x [DOI] [PubMed] [Google Scholar]
- 2.Brown RS. Hepatitis C and liver transplantation. Nature 2005;436:973–8. 10.1038/nature04083 [DOI] [PubMed] [Google Scholar]
- 3.Mercer DF, Schiller DE, Elliott JF, et al. . Hepatitis C virus replication in mice with chimeric human livers. Nat Med 2001;7:927–33. 10.1038/90968 [DOI] [PubMed] [Google Scholar]
- 4.Garcia-Retortillo M, Forns X, Feliu A, et al. . Hepatitis C virus kinetics during and immediately after liver transplantation. Hepatology (Baltimore, MD) 2002;35:680–7. 10.1053/jhep.2002.31773 [DOI] [PubMed] [Google Scholar]
- 5.Prieto M, Berenguer M, Rayon JM, et al. . High incidence of allograft cirrhosis in hepatitis C virus genotype 1b infection following transplantation: relationship with rejection episodes. Hepatology (Baltimore, MD) 1999;29:250–6. 10.1002/hep.510290122 [DOI] [PubMed] [Google Scholar]
- 6.Forman LM, Lewis JD, Berlin JA, et al. . The association between hepatitis C infection and survival after orthotopic liver transplantation. Gastroenterology 2002;122:889–96. 10.1053/gast.2002.32418 [DOI] [PubMed] [Google Scholar]
- 7.Taga SA, Washington MK, Terrault N, et al. . Cholestatic hepatitis C in liver allografts. Liver Transpl Surg 1998;4:304–10. 10.1002/lt.500040401 [DOI] [PubMed] [Google Scholar]
- 8.Lupberger J, Zeisel MB, Xiao F, et al. . EGFR and EphA2 are host factors for hepatitis C virus entry and possible targets for antiviral therapy. Nat Med 2011;17:589–95. 10.1038/nm.2341 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Goto K, Lin W, Zhang L, et al. . The AMPK-related kinase SNARK regulates hepatitis C virus replication and pathogenesis through enhancement of TGF-beta signaling. J Hepatol 2013;59:942–8. 10.1016/j.jhep.2013.06.025 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Friedman BH, Wolf JH, Wang L, et al. . Serum cytokine profiles associated with early allograft dysfunction in patients undergoing liver transplantation. Liver Transpl 2012;18:166–76. 10.1002/lt.22451 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hassan L, Bueno P, Ferron-Celma I, et al. . Early postoperative response of cytokines in liver transplant recipients. Transplant Proc 2006;38:2488–91. 10.1016/j.transproceed.2006.08.054 [DOI] [PubMed] [Google Scholar]
- 12.Tilg H, Vogel W, Aulitzky WE, et al. . Evaluation of cytokines and cytokine-induced secondary messages in sera of patients after liver transplantation. Transplantation 1990;49:1074–80. 10.1097/00007890-199006000-00009 [DOI] [PubMed] [Google Scholar]
- 13.Sainz B Jr, Chisari FV. Production of infectious hepatitis C virus by well-differentiated, growth-arrested human hepatoma-derived cells. J Virol 2006;80:10253–7. 10.1128/JVI.01059-06 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Gehring G, Rohrmann K, Atenchong N, et al. . The clinically approved drugs amiodarone, dronedarone and verapamil inhibit filovirus cell entry. J Antimicrob Chemother 2014;69:2123–31. 10.1093/jac/dku091 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.von Hahn T, Schiene-Fischer C, Van ND, et al. . Hepatocytes that express variants of cyclophilin A are resistant to HCV infection and replication. Gastroenterology 2012;143:439–47. 10.1053/j.gastro.2012.04.053 [DOI] [PubMed] [Google Scholar]
- 16.Ciesek S, Westhaus S, Wicht M, et al. . Impact of occludin intra- and inter-species variation on its co-receptor function for authentic hepatitis C virus particles. J Virol 2011;85:7613–21. 10.1128/JVI.00212-11 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.von Hahn T, Schulze A, Chicano Wust I, et al. . The novel immunosuppressive protein kinase C inhibitor sotrastaurin has no pro-viral effects on the replication cycle of hepatitis B or C virus. PloS One 2011;6:e24142 10.1371/journal.pone.0024142 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ciesek S, Steinmann E, Iken M, et al. . Glucocorticosteroids increase cell entry by hepatitis C virus. Gastroenterology 2010;138:1875–84. 10.1053/j.gastro.2010.02.004 [DOI] [PubMed] [Google Scholar]
- 19.Lindner V, Reidy MA. Proliferation of smooth muscle cells after vascular injury is inhibited by an antibody against basic fibroblast growth factor. Proc Natl Acad Scie U S A 1991;88:3739–43. 10.1073/pnas.88.9.3739 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Abraham JA, Mergia A, Whang JL, et al. . Nucleotide sequence of a bovine clone encoding the angiogenic protein, basic fibroblast growth factor. Science 1986;233:545–8. 10.1126/science.2425435 [DOI] [PubMed] [Google Scholar]
- 21.Klagsbrun M, Smith S, Sullivan R, et al. . Multiple forms of basic fibroblast growth factor: amino-terminal cleavages by tumor cell- and brain cell-derived acid proteinases. Proc Natl Acad Sci U S A 1987;84:1839–43. 10.1073/pnas.84.7.1839 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Okada-Ban M, Thiery JP, Jouanneau J. Fibroblast growth factor-2. Int J Biochem Cell Biol 2000;32:263–7. 10.1016/S1357-2725(99)00133-8 [DOI] [PubMed] [Google Scholar]
- 23.Graham BM, Richardson R. Memory of fearful events: the role of fibroblast growth factor-2 in fear acquisition and extinction. Neuroscience 2011;189:156–69. 10.1016/j.neuroscience.2011.05.041 [DOI] [PubMed] [Google Scholar]
- 24.Liao S, Bodmer J, Pietras D, et al. . Biological functions of the low and high molecular weight protein isoforms of fibroblast growth factor-2 in cardiovascular development and disease. Dev Dyn 2009;238:249–64. 10.1002/dvdy.21677 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Fei Y, Gronowicz G, Hurley MM. Fibroblast growth factor-2, bone homeostasis and fracture repair. Curr Pharm Des 2013;19:3354–63. 10.2174/1381612811319190002 [DOI] [PubMed] [Google Scholar]
- 26.Yoshiji H, Kuriyama S, Yoshii J, et al. . Synergistic effect of basic fibroblast growth factor and vascular endothelial growth factor in murine hepatocellular carcinoma. Hepatology (Baltimore, MD) 2002;35:834–42. 10.1053/jhep.2002.32541 [DOI] [PubMed] [Google Scholar]
- 27.Wang L, Park H, Chhim S, et al. . A novel monoclonal antibody to fibroblast growth factor 2 effectively inhibits growth of hepatocellular carcinoma xenografts. Mol Cancer Ther 2012;11:864–72. 10.1158/1535-7163.MCT-11-0813 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Finn RS. Development of molecularly targeted therapies in hepatocellular carcinoma: where do we go now? Clin Cancer Res 2010;16:390–7. 10.1158/1078-0432.CCR-09-2084 [DOI] [PubMed] [Google Scholar]
- 29.Llovet JM, Decaens T, Raoul JL, et al. . Brivanib in patients with advanced hepatocellular carcinoma who were intolerant to sorafenib or for whom sorafenib failed: results from the randomized phase III BRISK-PS study. J Clin Oncol 2013;31:3509–16. 10.1200/JCO.2012.47.3009 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.