Abstract
Background
Dorsal compressive lesions at the atlantoaxial junction (ie, AA bands) occur in dogs with Chiari‐like malformations (CMs), but their clinical relevance is unclear.
Objective
Investigate the influence of AA bands on clinical status and syringomyelia (SM) in mature cavalier King Charles spaniels (CKCS).
Animals
Thirty‐six CKCS, 5–12 years of age, including 20 dogs with neuropathic pain.
Methods
Dogs were examined and assigned a neurologic grade. Magnetic resonance imaging (MRI) of the craniocervical junction was performed with the craniocervical junction extended and flexed (ie, normal standing position). Imaging studies were assessed for the presence of an AA band, CM, SM or some combination of these findings. Band and SM severity were quantified using an objective compression index and ordinal grading scale, respectively.
Results
Of 36 CKCS imaged, 34 had CM. Atlantoaxial bands were present in 31 dogs and were more prominent in extended than flexed positions. Syringomyelia was found in 26 dogs, 23 of which also had AA bands. Bands were associated with both the presence (P = .0031) and severity (P = .008) of clinical signs and SM (P = .0147, P = .0311, respectively). Higher compression indices were associated with more severe SM (P = .0137).
Conclusions
Prevalence of AA bands in older CKCS is high. Positioning of dogs in extension during MRI enhances the sensitivity of the study for detecting this important abnormality. There were significant associations among AA bands, clinical signs, and SM in dogs with CM; additional work is needed to understand whether or not this relationship is causal.
Keywords: Chiari‐like, Compression index, Fibrous band, Magnetic resonance imaging
Abbreviations
- AA
atlantoaxial
- CKCS
cavalier King Charles spaniels
- CM
Chiari‐like malformation
- MRI
magnetic resonance imaging
- SAS
subarachnoid space
- SM
syringomyelia
Dorsal compressive lesions have been described at the atlantoaxial (AA) and atlantooccipital junctions in cavalier King Charles spaniels (CKCS), causing varying degrees of attenuation of the subarachnoid space (SAS) and the spinal cord. 1, 2, 3, 4, 5 These lesions are visible surgically as the foci area of whitened and thickened soft tissue dorsal to the AA and atlantooccipital junctions. 1, 6, 7, 8, 9 Histopathologically, atlantooccipital dorsal constrictive lesions are composed of areas of lymphoplasmacytic inflammation and fibrosis, with areas of mineralization, osseous metaplasia, or both. 2, 7 The presence of inflammatory cells suggests that these are not static lesions, but rather sites of ongoing inflammation. Atlantoaxial dorsal constrictive lesions are not as well described histopathologically as their more cranial counterparts, although they appear to involve the dorsal interarcuate ligament when visualized and resected at surgery.1, 4, 5 These lesions have been described using various terms, including AA dorsal compressive or constrictive lesions, and dural fibrous bands.1, 3, 4, 5, 6 In this report, they will be referred to as AA bands.
Atlantoaxial bands can be diagnosed preoperatively as areas of focal SAS compression on T2‐weighted magnetic resonance imaging (MRI) images; dilatation of the SAS also may be seen immediately caudal or cranial to the band site or in both locations on imaging and at surgery (Fig 1).1, 3, 8 These AA bands are present in 38% of small and toy breed dogs.3 Of these, CKCS represent 1 of the most commonly affected breeds, with a prevalence of craniocervical junction anomalies of 42% in a group of symptomatic and asymptomatic CKCS.1 They occur most commonly in conjunction with Chiari‐like malformations (CMs), although they may occur alone or with other craniocervical junction anomalies, such as dorsal angulation of the dens.1, 3, 6, 10
Figure 1.
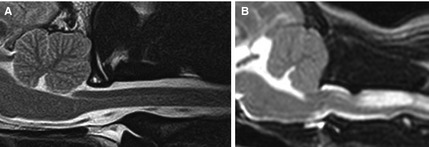
T2‐weighted sagittal MR images of the craniocervical junction in a cavalier King Charles spaniels. In comparison to the normal dorsal subarachnoid space (SAS) seen in figure (A), figure (B) demonstrates elimination of the dorsal SAS by a dorsal atlantoaxial band, alongside cervical spinal cord syringomyelia.
In humans, dorsal AA compressive bands have been found to play an important role in the development of clinical signs and syringomyelia (SM), particularly in persons also diagnosed with Chiari malformations.7, 11 In veterinary medicine, AA bands are suspected of causing neuropathic pain, similar to other craniocervical junction anomalies, including head, craniocervical junction and cervical hyperesthesia, generalized dysesthesia and allodynia. Clinical signs related to cervical myelopathy are also described.3, 4, 6, 8 In addition, improvement in clinical signs can result from surgical band excision in dogs.4, 5 However, the relationship between these bands and both clinical status and SM is not fully understood. In a group of 64 CKCS screened for craniocervical junction disorders, no relationship was found between degree of compression caused by AA bands and the presence or severity of clinical signs or SM.1 A separate screening study described an objective method of assessing degree of dorsal compression caused by AA bands (ie, dorsal compression index), but did not evaluate the clinical relevance of this measurement.3
Our study aims to expand upon the current understanding of the problem by evaluating the relationships among AA bands, clinical signs, CM and SM in CKCS > 5 years of age.
Materials and Methods
Inclusion Criteria
A group of 36 dogs was prospectively recruited from various sources, including a group of CKCS evaluated previously in a separate study,1 CKCS clubs, online breed‐associated groups, and CKCS presented to the Cornell University and North Carolina State University veterinary teaching hospitals as patients. Inclusion criteria were as follows: >5 years of age, normal CBC and serum biochemistry panel results (within 7 days of imaging) and absence of physical examination findings contraindicating anesthesia, such as heart murmur grade >4 of 6, or evidence of clinically apparent cardiac disease (eg, coughing, tachypnea).
Clinical and Magnetic Resonance Imaging Assessment
Dogs were assessed for pain, dysesthesia, and neurologic dysfunction by neurologic examination performed by 1 of the investigators (SCG or NJO) in addition to owner questionnaires assessing clinical signs seen at home. In the latter, owners were asked if their dogs had a history of scratching; rubbing their head, neck or shoulders on objects; episodes of crying out after play; decreased interaction with littermates or housemates; or, evidence of neck or head pain at home (eg, limited movement of the neck, blepharospasm, head‐shy behavior). The area scratched, the frequency of scratching, factors precipitating its occurrence (eg, excitement, play, changes in environmental temperature or barometric pressure, neck leads, contact with the skin, or hair on the neck), and response to medications, surgery, or both also were evaluated, where applicable. Lastly, owners were asked if they had noted any changes in their dog's gait or ability to climb stairs. Information acquired from the questionnaires was then confirmed and supplemented at an in‐person interview at the time of imaging.
This information was used to assign a neurologic grade between 0 and 5.1 Dogs were anesthetized with fentanyl (premedication), propofol (induction), and either sevoflurane or isoflurane (maintenance). They were positioned in sternal recumbency for MRI,1 first using padding to achieve a craniocervical junction posture approximating a normal standing position1 and then with their craniocervical junction extended and their neck flat on the table, in a more typical posture used for MRI scanning of the cervical spine and brain. Head angles in flexion and extension were measured using a previously described method.12
Acquired MRI sequences included T1‐ and T2‐weighted sagittal images and T2‐weighted transverse images of the craniocervical junction. These were uploaded into OsiriX Medical Imaging Software (open source software, www.osirix-viewer.com) and evaluated (by SCG) for the following: presence of a CM (ie, cerebellar indentation and herniation through the foramen magnum and loss of cerebrospinal fluid at the craniocervical junction); presence of SM; and presence of dorsal compression of the SAS, spinal cord, or both at the level of the first and second cervical vertebrae (ie, an AA band). In dogs with SM, an AA band, or both a severity grade was assigned (Table 1). This grade was assigned separately from images of the cervical spine in both the flexed and extended positions. A compression index also was generated to provide an objective assessment of severity of compression secondary to AA band formation. This index was determined using T2‐weighted sagittal images with the craniocervical junction in an extended position, using a method described previously.3 Specifically, the distance was measured between the dorsal‐most aspect of the dorsal SAS at the AA junction and the ventral‐most point of band‐related compression. This distance was then divided by the height of the nearest normal spinal cord. Extended images were used to assess the full extent of compression that might occur in a physiologic range of motion; positioning in flexion decreased the apparent degree of compression in most cases (Fig 2).
Table 1.
Grading of severity of dorsal compressive AA bands and SM
| Grade | AA Band Severity | Severity of SM |
|---|---|---|
| 0 | None | None |
| 1 | Underlying SAS reduced but not eliminated | <33% of spinal cord |
| 2 | Underlying SAS eliminated, questionable spinal cord compression | 33–60% of spinal cord |
| 3 | Underlying spinal cord compressed/deformed | >60% of spinal cord |
AA, atlantoaxial; SM, syringomyelia; SAS, subarachnoid space.
Figure 2.
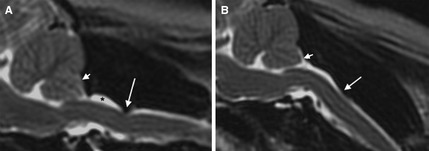
T2‐weighted sagittal MR images of the craniocervical junction in a cavalier King Charles spaniels positioned in an extended/straight position (A) and in a flexion, resembling a standing posture (B). Dorsal atlantoaxial (AA) band‐associated compression (long arrow) is more prominent in flexion than in extension. A Chiari‐like malformation is also present (short arrow), along with dorsal subarachnoid space dilation cranial to the AA band (*).
Statistical Analysis
Data were analyzed using SAS software.2 Data analyzed included the presence of a CM (Y or N); clinical signs (Y or N); severity of neurologic signs graded from 0 to 5; presence of SM (Y or N); severity of SM graded from 0 to 3; presence of an AA band (Y or N); severity of AA band compression graded from 0 to 3; and compression index. Contingency tables were constructed to investigate the relationship between the presence of an AA band and the presence of neurologic signs and of SM. Ordinal factors, such as neurologic grade, SM grade, and compression grade also were examined by construction of contingency tables. Significance of relationships between pairs of ordinal variables was established using Chi Square tests. Spearman correlation coefficients were used to investigate associations between disease severity and continuous measurements (eg, compression index). Wilcoxon nonparametric tests were used to compare continuous measurements across 2 groups (ie, presence of SM), and Kruskal‐Wallace tests were used for comparisons across more than 2 groups (eg, severity of neurologic signs). A logistic regression model was developed to examine the relationship among the presence of a CM, AA bands and the presence of clinical signs or SM. To control for the increased chance of false positives caused by multiple comparisons, the alpha level was decreased from 0.10 to 0.035.
Results
Patient Characteristics
Thirty‐six CKCS ranging in age from 5 to 12 years (mean, 8.8; median, 9) were evaluated; 15/36 (41.7%) were male. Twenty of the dogs (56%) had varying degrees of scratching, neck pain, and dysesthesia (Table 2). Six dogs had neck pain alone but no other sensory or neurologic signs. Twelve dogs (33%) were being treated for their clinical signs at the time of imaging. These were treated with pain medications (gabapentin, pregabalin, or tramadol), omeprazole, prednisone, or some combination of these drugs. All but 1of these were being treated for neuropathic pain; the remaining dog was being treated for osteoarthritis. A neurologic grade of 5 (ie, ataxia and tetraparesis) was recorded in 1 dog, in which severe postoperative scar tissue and displacement of an implant appeared to cause compression of the SAS and spinal cord at both the atlantooccipital and AA junctions. This dog was excluded from further analysis because of the secondary (ie, postoperative) nature of its compression. Two additional dogs had a history of foramen magnum decompression surgery. In these, although scar tissue was not outwardly apparent, its presence could not be excluded. Consequently, to eliminate the risk of these dogs introducing bias to the study, they were excluded from statistical analyses investigating the relationship among AA bands, clinical signs, and SM.
Table 2.
Neurologic grades of dogs: distribution, and number of dogs with Chiari‐like malformations, dorsal compressive AA bands, or SM, per neurologic grade
| Neurologic Grade | Number of Dogs, Overall | Number of Dogs with CM | Number of Dogs with AA Band | Number of Dogs with SM |
|---|---|---|---|---|
| 0 | 15 | 14 | 11 | 8 |
| 1 | 4 | 4 | 4 | 3 |
| 2 | 3 | 2 | 3 | 2 |
| 3 | 13 | 12 | 13 | 12 |
| 4 | 0 | 0 | 0 | 0 |
| 5 | 0 | 0 | 0 | 0 |
| Total | 35 | 32 | 31 | 25 |
AA, atlantoaxial; CM, Chiari‐like malformation; SM, syringomyelia.
Imaging Findings
Mean head angles were 142 and 196 degrees in flexion and extension, respectively, and positioning differed by an average of 57 degrees between flexion and extension. On MRI analysis, 33 dogs had a CM present (94%). Thirty‐one dogs (88.6%) showed AA band compression on T2‐weighted sagittal and transverse images, with severity ranging from minimal deformation of the dorsal SAS at the level of the AA junction to complete elimination of the SAS coupled with dorsolateral compression of the underlying spinal cord (Fig 3, Table 3). Twenty‐nine of these had a concurrent CM; of the remaining 3 dogs, 2 had signs of neuropathic pain.
Figure 3.
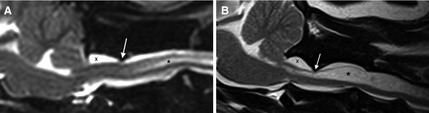
T2‐weighted sagittal MR images of the craniocervical junction in cavalier King Charles spaniels demonstrating variability in atlantoaxial band severity. In Figure 1A, focal compression (arrow) of the dorsal subarachnoid space (SAS) is present (grade 1). In Figure 1B the SAS is eliminated, and ventral displacement of the underlying spinal cord is seen (arrow; grade 3), along with atlanto‐occipital overlapping, dilation of the SAS cranial to the dural band (X), and syringomyelia (*).
Table 3.
Frequency of occurrence of dorsal compressive AA band grades
| AA Band Grade | Number of Dogs |
|---|---|
| 0 | 4 |
| 1 | 10 |
| 2 | 17 |
| 3 | 5 |
| Total | 36 |
AA, atlantoaxial.
When comparing each individual dog's extended and flexed sequences, dorsal compression of the SAS and spinal cord at the AA junction was less prominent in flexed positions than in extension (Fig 2). In extension, the degree of compression ranged from a dorsal compression index of 0–46.7%, with the majority ranging between 20 and 30% (mean, 20.6; median, 20).
Relationship Between Atlantoaxial Bands and Syringomyelia
Syringomyelia was present in 26 (74.3%) dogs overall, 23 of which also had AA bands. The cranial‐most extent of SM was located either at the level of the first cervical vertebra (C1 l11, 42.3%), C2 (10, 38.5%), or C3 (5, 19.2%). All grades of SM severity were seen: grade 1, 28%; grade 2, 47%; and, grade 3, 14%. In evaluating the relationship between the presence of AA bands and SM, AA bands were significantly associated with the presence (P = .029) and severity (ie, grade; P = .046) of SM. Objective measurements of compression severity (ie, compression index) were associated with both SM presence (P = .039) and severity (ie, grade; P = .0458; Fig 3). In a logistic regression model evaluating the relationship between the presence of a CM and AA bands as independent variables and the presence of SM as the dependent variable, the presence of AA bands was significantly predictive of the presence of SM (0.007). In contrast, the presence of a CM was not significant as a predictor of the presence of SM (P = .055), although, the number of dogs included in the study may not have been sufficient to identify this relationship.
Relationship Between Atlantoaxial Bands and Clinical Signs
In evaluating clinical signs, the presence of an AA band was found to be associated with their presence (P = .0024) but not their severity (P = .132). In evaluating the relationship between compression indexes and clinical signs, a higher index was associated with the presence (P = .028), but not the severity (P = .095) of clinical signs. In a logistic regression model evaluating the relationship between the presence of CM and AA bands as independent variables, and the presence of clinical signs as the dependent variable, only the presence of AA bands was found to be significantly predictive of the presence of clinical signs (P = .008).
Discussion
Atlantoaxial bands were found in a larger proportion of CKCS in this study (83.8%) than has been reported previously in this breed (42%)1 or in small and toy breeds in general (38%).3 This finding may be explained by the maturity of dogs in this study, which specifically examined >5 years of age. Although previous study populations were predominantly young, with median ages of 1.83 and 2.51 years, the median age of dogs in our study was 9 years. The higher prevalence of AA bands in older CKCS populations may reflect progression of the disease over time, as described for SM,13, 14 but longitudinal studies are needed to confirm this hypothesis. Excessive vertebral movement at the AA junction has been proposed as a contributing factor for dorsal compressive band formation in dogs.3, 4 Although this relationship is not yet confirmed, if present, such an effect could lead to cumulative compression over time.
Compression was, in general, more pronounced in extended (ie, straight) positions, compared to each dog's flexed views. In fact, in a small number of cases, AA bands that were noticeable in extension were not apparent in flexed views. Thus, imaging in flexion alone could underestimate the prevalence of AA bands, and may have played a role in their apparently lower prevalence in previous studies. In contrast, other craniocervical junction anomalies, such as cerebellar herniation, become more pronounced in flexed views.12 For this reason, it may be optimal to image the canine craniocervical junction in both flexion and extension to obtain maximal diagnostic information.
An unconfirmed association has been suspected between AA bands and neuropathic pain signs consistent with cervical myelopathies.1, 7, 8, 10 In our study, AA bands appeared to be independently associated with the presence of clinical signs, which primarily manifested as neuropathic pain. In addition, despite the high prevalence of CM, AA bands had a stronger relationship with the presence of clinical signs than did CM. When severity of signs was considered, it was not found to be associated with the severity of band compression, which ranged from minimal indentation to complete obliteration of the SAS, with concurrent underlying spinal cord compression. Neuropathic pain associated with this condition may result either from primary neural compression or as a consequence of SM. The latter, present in our study in 92.3% of dogs with AA bands, previously has been identified as an important factor in determining the presence of neurologic signs in dogs with craniocervical junction anomalies.1, 15, 16
Atlantoaxial bands cause variable degrees of attenuation of the SAS at the AA junction, as can be seen on MRI studies and at surgery.1, 3, 8, 10 Compression of the SAS throughout the neuraxis, in turn, has been suspected of playing a role in SM formation by influencing local cerebrospinal fluid flow dynamics, as described in numerous hydrodynamic models.15, 17, 18, 19, 20 For this reason, AA bands have been suspected of playing a role in SM formation since they were first identified.1, 8, 10, 15 Our study confirms the existence of an association between AA bands and SM. Specifically, the presence of an AA band was associated with the presence of SM, regardless of the presence of CM. In addition, more severe compression was positively associated with both the presence and severity of SM. The pathophysiology underlying SM associated with AA bands remains to be elucidated, however, and must be considered in light of other craniocervical junction anomalies commonly found in this area such as CM.
Several factors have been proposed to play a role in the presence of SM associated with CM, including differences in parenchyma21, 22 and caudal fossa1, 23 sizes, changes in cerebrospinal fluid flow characteristics,17, 24 cerebellar pulsation,25 and abnormal jugular foramina size leading to venous congestion.26
Atlantoaxial bands may play a role in the development of SM, and if so, they would be expected to alter local cerebrospinal fluid flow dynamics in a manner similar to that described in the spinal thecal sac constriction model (ie, as observed in spinal ligation studies). In this model, focal iatrogenic constriction of the SAS results in SM formation, both cranially and caudally to the point of obstruction.19 The fluid pulse pressure theory, generated in response to this model, theorizes that the accumulation of extracellular fluid within the spinal cord parenchyma (ie, SM formation) results from pressure differentials cranial and caudal to the focal point of obstruction. Thus, as a result of systolic pulse pressure waves located within the dorsal SAS caudal to the obstruction, relatively lower pressures are thought to occur within the underlying spinal cord parenchyma. It is hypothesized that these lower pressures encourage movement of extracellular fluid into the caudal spinal cord parenchyma. Conversely, it is thought that during valsalva maneuvers, lower pressures exist within the spinal cord parenchyma cranial to the point of obstruction (compared with the SAS), leading to cranial SM formation.19
In our study, the cranial‐most extent of SM was found overlying the first cervical vertebra in the majority of cases, and extending caudal to the AA band. The presence of SM both cranial and caudal to this focal compression suggests that dynamics similar to those observed in spinal ligations studies could be at play in AA band‐related SM, or simply could be a reflection of syrinx progression associated with age. Studies evaluating cerebrospinal fluid flow within this area are needed to better understand the processes influencing the location and extent of syrinx formation.
Conclusions
Our study confirms the high prevalence of AA bands in older CKCS and demonstrates that positioning of dogs in extension during MRI enhances the sensitivity of the study for detecting this important abnormality. There was a significant association among AA bands, clinical signs and SM in dogs with CM but additional work is needed to understand whether this relationship is causal or not.
Acknowledgments
The authors thank Cavalier King Charles Spaniel owners and breeders as well as the American Cavalier King Charles Spaniel Club without whom this study could not have been completed. This work was funded by a grant from the American Cavalier King Charles Spaniel Club Charitable Trust.
Conflict of Interest Declaration: Authors disclose no conflict of interest.
Off‐label Antimicrobial Declaration: Authors declare no off‐label use of antimicrobials.
This study was performed at both Cornell University and the North Carolina State University.
Findings were presented as a Research Report at the 2014 ACVIM Forum, Nashville, TN.
Footnotes
Imaging was performed using the following 1.5 T MRI units: Vantage Atlas, Toshiba America Medical Systems, Tustin, CA; Siemens Medical Solutions USA Inc, Malvern, PA
SAS Software Version 9.3, Cary, NC
References
- 1. Cerda‐Gonzalez S, Olby NJ, McCullough S, et al. Morphology of the Caudal Fossa in Cavalier King Charles Spaniels. Vet Radiol Ultrasound 2009;50:37–45. [DOI] [PubMed] [Google Scholar]
- 2. Dewey CW, Berg JM, Barone G, et al. Foramen magnum decompression for treatment of caudal occipital malformation syndrome in dogs. J Am Vet Med Assoc 2005;227:1250–1251. [DOI] [PubMed] [Google Scholar]
- 3. Marino DJ, Loughin CA, Dewey CW, et al. Morphometric features of the craniocervical junction region in dogs with suspected Chiari‐like malformation determined by combined use of magnetic resonance imaging and computed tomography. Am J Vet Res 2012;73:105–111. [DOI] [PubMed] [Google Scholar]
- 4. Dewey CW, Marino DJ, Loughin CA. Craniocervical junction abnormalities in dogs. N Z Vet J 2013;61:202–211. [DOI] [PubMed] [Google Scholar]
- 5. Cerda‐Gonzalez S, Dewey CW, Scrivani PV, et al. Imaging features of atlanto‐occipital overlapping in dogs. Vet Radiol Ultrasound 2009;50:264–268. [DOI] [PubMed] [Google Scholar]
- 6. Cerda‐Gonzalez S, Dewey CW. Congenital diseases of the craniocervical junction in the dog. Vet Clin North Am Small Anim Pract 2010;40:121–141. [DOI] [PubMed] [Google Scholar]
- 7. Nakamura N, Iwasaki Y, Hida K, et al. Dural band pathology in syringomyelia with Chiari type I malformation. Neuropathology 2000;1:38–43. [DOI] [PubMed] [Google Scholar]
- 8. Tagaki S, Kadosawa T, Ohsaki T, et al. Hindbrain decompression in a dog with scoliosis associated with syringomyelia. J Am Vet Med Assoc 2005;226:1359–1363. [DOI] [PubMed] [Google Scholar]
- 9. Vermeersch K, Van Ham L, Caemaert J, et al. Suboccipital craniectomy, dorsal laminectomy of C1, durotomy and dural graft placement as a treatment for syringohydromyelia with cerebellar tonsil herniation in Cavalier King Charles spaniels. Vet Surg 2004;33:355–360. [DOI] [PubMed] [Google Scholar]
- 10. Bynevelt M, Rusbridge C, Britton J. Dorsal dens angulation and a chiari type malformation in a Cavalier King Charles spaniel. Vet Radiol Ultrasound 2000;41:521–524. [DOI] [PubMed] [Google Scholar]
- 11. Hida K, Iwasaki Y, Koyanagi I, et al. Surgical indication and results of foramen magnum decompression versus syringosubarachnoid shunting for syringomyelia associated with Chiari I malformation. Neurosurgery 1995;37:673–678; discussion 678–9. [DOI] [PubMed] [Google Scholar]
- 12. Upchurch JJ, McGonnell IM, Driver CJ, et al. Influence of head positioning on the assessment of Chiari‐like malformation in Cavalier King Charles spaniels. Vet Rec 2011;169:277. [DOI] [PubMed] [Google Scholar]
- 13. Driver CJ, De RL, Hamilton S, et al. Changes over time in craniocerebral morphology and syringomyelia in cavalier King Charles spaniels with Chiari‐like malformation. BMC Vet Res 2012;8:215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Parker JE, Knowler SP, Rusbridge C, et al. Prevalence of asymptomatic syringomyelia in Cavalier King Charles spaniels. Vet Rec 2011;168:667–669. [DOI] [PubMed] [Google Scholar]
- 15. Driver CJ, Volk HA, Rusbridge C, et al. An update on the pathogenesis of syringomyelia secondary to Chiari‐like malformations in dogs. Vet J 2013;198:551–559. [DOI] [PubMed] [Google Scholar]
- 16. Rusbridge C, Jeffery ND. Pathophysiology and treatment of neuropathic pain associated with syringomyelia. Vet J 2008;175:164–172. [DOI] [PubMed] [Google Scholar]
- 17. Cerda‐Gonzalez S, Olby NJ, Broadstone R, et al. Characteristics of cerebrospinal fluid flow in Cavalier King Charles Spaniels analyzed using phase velocity contrast cine magnetic resonance imaging. Vet Radiol Ultrasound 2009;50:467–476. [DOI] [PubMed] [Google Scholar]
- 18. Gardner WJ. Hydrodynamic mechanism of syringomyelia: Its relationship to myelocele. J Neurol Neurosurg Psychiatry 1965;28:247–259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Josephson A, Greitz D, Klason T, et al. A spinal thecal sac constriction model supports the theory that induced pressure gradients in the cord cause edema and cyst formation. Neurosurgery 2001;48:636–645; discussion 645–6. [DOI] [PubMed] [Google Scholar]
- 20. Stoodley MA, Gutschmidt B, Jones NR. Cerebrospinal fluid flow in an animal model of noncommunicating syringomyelia. Neurosurgery 1999;44:1065–1075; discussion 1075–6. [DOI] [PubMed] [Google Scholar]
- 21. Shaw TA, McGonnell IM, Driver CJ, et al. Caudal cranial fossa partitioning in Cavalier King Charles spaniels. Vet Rec 2013;172:341. [DOI] [PubMed] [Google Scholar]
- 22. Driver CJ, Rusbridge C, Cross HR, et al. Relationship of brain parenchyma within the caudal cranial fossa and ventricle size to syringomyelia in cavalier King Charles spaniels. J Small Anim Pract 2010;51:382–386. [DOI] [PubMed] [Google Scholar]
- 23. Rusbridge C, Knowler SP, Pieterse L, et al. Chiari‐like malformation in the Griffon Bruxellois. J Small Anim Pract 2009;50:386–393. [DOI] [PubMed] [Google Scholar]
- 24. March PA, Abramson CJ, Smith M, et al. CSF flow abnormalities in caudal occipital malformation syndrome. J Vet Intern Med 2005;19:418–419. [Google Scholar]
- 25. Driver CJ, Watts V, Bunck AC, et al. Assessment of cerebellar pulsation in dogs with and without Chiari‐like malformation and syringomyelia using cardiac‐gated cine magnetic resonance imaging. Vet J 2013;198:88–91. [DOI] [PubMed] [Google Scholar]
- 26. Schmidt MJ, Ondreka N, Sauerbrey M, et al. Volume reduction of the jugular foramina in Cavalier King Charles Spaniels with syringomyelia. BMC Vet Res 2012;8:158. [DOI] [PMC free article] [PubMed] [Google Scholar]


