Abstract
Background
Potassium (K+) supplementation of isotonic crystalloid fluids in daily fluid therapy is commonly performed, yet its accuracy in veterinary medicine is undetermined.
Objective
To investigate the accuracy of K+ supplementation in isotonic crystalloid fluids.
Animals
None.
Methods
Observational study. 210 bags of fluid supplemented with KCl being administered to hospitalized dogs and cats intravenously (IV) were sampled over a 3‐month period. Measured K+ concentration ([K+]) was compared to the intended [K+] of the bag. In a second experiment, 60 stock fluid bags were supplemented to achieve a concentration of 20 mmol/L K+, mixed well and [K+] was measured. In another 12 bags of 0.9% NaCl, K+ was added without mixing the bag, and [K+] of the delivered fluid was measured at regular time points during constant rate infusion.
Results
The measured [K+] was significantly higher than intended [K+] (mean difference 9.0 mmol/L, range 6.5 to >280 mmol/L, P < .0001). In 28% of clinical samples measured [K+] was ≥5 mmol/L different than intended [K+]. With adequate mixing, K+ supplementation of fluids can be accurate with the mean difference between measured and intended [K+] of 0.7 (95% CI −0.32 to 1.7) mmol/L. When not mixed, K+ supplementation of 20 mmol/L can lead to very high [K+] of delivered fluid (up to 1410 mmol/L).
Conclusions and Clinical Importance
Inadequate mixing following K+ supplementation of fluid bags can lead to potentially life threatening IV infused [K+]. Standard protocols for K+ supplementation should be established to ensure adequate mixing.
Keywords: Additives, Crystalloid fluids, Electrolytes, Hyperkalemia, Mixing
Abbreviations
- K+
potassium
- KCl
potassium chloride
- PVC
polyvinyl chloride
Isotonic crystalloid fluids are the most common fluid type used for intravenous (IV) fluid therapy in small animal medicine.1 There are several different commercially available isotonic crystalloid fluid types that differ in their composition of electrolyte and non‐electrolyte solutes. Fluid composition can be further modified by the addition of electrolytes and drugs, the most common of which is potassium (K+) supplementation.1, 2, 3, 4
Commercially available isotonic crystalloid fluids are usually deficient in K+ when used for maintenance fluid therapy and supplementation of crystalloid solutions with potassium chloride (KCl) is widely performed to meet maintenance K+ requirements and to prevent electrolyte depletion in both human and animal patients.1, 3, 5 An empirical scale to determine the amount of KCl to be added to commercially available crystalloid fluids in the treatment of small animal species has been established.6 Potassium supplementation is not without risk; life threatening and fatal hyperkalemia are reported adverse effects of inadvertent K+ overdosage in human medicine.2, 7, 8, 9 A case of transient subclinical hyperkalemia directly attributable to K+ supplementation of fluids administered IV has also been described in the veterinary literature.10 Prior to the initiation of routine quality control, concerning inaccuracies in K+ supplementation of fluids administered IV have been reported in human pharmacies, however, the accuracy of clinical K+ supplementation of fluids administered IV has not been investigated in veterinary medicine.11
This study was performed to investigate the accuracy of K+ supplementation in isotonic crystalloid fluids used in daily fluid therapy administered IV.
Materials and Methods
Potassium Supplementation in Hospitalized Dogs and Cats
To determine the accuracy of K+ administration in daily fluid therapy, samples were acquired from 500 mL, 1000 mL, 3000 mL, and 5000 mL flexible polyvinyl chloride (PVC) bags of lactated Ringer's®, 2 Plasma‐Lyte 148®, 2, and 0.9% NaCl2 that had K+ supplementation and were in clinical use (i.e. currently being administered to a dog or cat in hospital) in both the small animal intensive care unit and the general dog and cat wards at the University of California, Davis, William R. Pritchard Veterinary Medical Teaching Hospital between August 2013 and October 2013. Sampling was performed on a convenience basis; on a given day of sampling, all K+ supplemented bags in clinical use were included.
The bags were not disturbed or shaken prior to acquiring the fluid sample. The injection port of the fluid bag was wiped with alcohol and a 1 ml sample of the bag's content was obtained with a 22GX 19 mm needle,3 MA fully inserted into the injection port (located at the most gravity dependent portion of the bag), with the bag in the hanging position. The fluid type, quantity of KCl added, whether KCl was added to a full or partial bag, and intended target K+ concentration of the fluid as noted on the medication additive label, were recorded. Sampled bags were subsequently marked to prevent repeat sampling. Samples were transferred to a plain blood collection tube with no additives and 100 μL of the sample was diluted with 400 μL of deionized water to assess K+ concentration. If the K+ concentrations were too high to measure, samples were then diluted by adding 100 μL of original sample to 900 μL of deionized water. All potassium samples were measured on a point of care blood gas analyzer.4 Deionized water was determined to be free of K+ by analysis.
Standardized Potassium Supplementation with Mixing
To determine the accuracy of potassium supplementation under controlled circumstances, 60 stock fluid bags (twenty 1 L bags each of lactated Ringer's® 2, Plasma‐Lyte 148® 2, and 0.9% NaCl2) whose baseline K+ concentrations were measured using the same technique as above were weighed5 and weight recorded in grams. The weight of an empty bag of the respective fluid type in grams was subtracted from each measurement. Each bag was supplemented to a target concentration of 20 mmol/L of K+ according to its preexisting K+ concentration and weight using a 2 mmol/mL KCl solution.6 For example, a 1000 mL bag of LRS has a [K+] of 4 mmol/L. To target a final [K+] of 20 mmol/L it is necessary to add 16 mmol of K+. If (following subtraction of the weight of an empty bag) a bag of LRS weighed 1050 g, 16.8 mmol of K+ was added to the bag. To reflect clinical practice, the effect of added volume on the calculated [K+] was disregarded in this experiment. Potassium chloride was added through the injection port of the bags with the bag in the inverted position (injection port uppermost). The injection port was squeezed and the bags were inverted 4 times to ensure adequate mixing. Following K+ supplementation, 1 mL of fluid was sampled from each bag for postsupplementation measurements of K+ concentration4. To assess K+ within the analyzer's range of highest precision, 100 μL of each sample were diluted with 400 μL of deionized water using a manual pipette.7
Potassium Supplementation without Mixing
The effect of not mixing a bag following K+ supplementation was further investigated in 12 additional 1L bags of 0.9% NaCl2. All bags were supplemented to 20 mmol/L of K+ by injection of 10 mL of 2 mmol/mL of KCl6. All bags were hung from an infusion stand at the time of supplementation and KCl was injected through the injection port while in the hanging position. KCl injection was performed over 30 seconds (slow) in 6 bags and over 10 seconds (rapid) in 6 bags and no attempt was made to mix the bags. Administration sets were inserted to the bags prior to KCl injection and primed with the fluid immediately after injection. Bags injected at a slow speed were infused into a receptacle at a rate of 50 mL/h and bags with rapid KCl injection were infused at 100 mL/h, using a fluid pump.8 Aliquots for K+ concentration measurements were collected during fluid infusion from the end of the administration set at specific time points. For the slow injection bags running at 50 mL/h, fluid samples were collected at 0, 15, 30, 45, 60, 90, 120, 150, 180, 240, 300 minutes. For the fast injection bags running at 100 mL/h, fluid samples were collected at 0, 30, 60, 120, 180, 240, 300, 360, and 420 respectively. Potassium concentration in the fluid discharged was measured in samples diluted with deionized water as described above. The fluid infusion rates and sample times were designed to allow acquisition of sufficient data points for area under the curve analysis (see statistics section). The high concentration of K+ in the slow injection bag was such that a slower infusion rate was necessary for adequate sampling time to be achieved.
Accuracy of Potassium Measurement
In this study, K+ concentration was measured on a point of care blood gas analyzer4 using an ion‐selective electrode (measurement range 2–8 mmol/L, bias −0.03 to 0.04).9 The analyzer used for this study measures potassium using a direct ion specific electrode that is calibrated to account for the impact of protein on the determination of ion concentration. When determining potassium concentration in nonprotein containing fluid, as was performed in this study, there maybe a difference of up to 1.5% between the determined value and the true potassium ion concentration.12 This very small difference was not considered relevant to the results of this study.
To further investigate the accuracy of K+ concentration measurements on crystalloid fluid samples, K+ concentration was measured in 40 samples of 0.9% NaCl2 supplemented to a known K+ concentration. Twenty samples each of 4 mmol/L and 8 mmol/L of K+ were prepared by adding 2 μL or 4 μL of a 2 mmol/mL KCl solution6 to 998 μL or 996 μL of 0.9% NaCl2 using a manual pipette7 respectively. Samples were inverted 3 times in a plain blood collection tube and K+ concentration measured and compared to the intended concentration using Bland‐Altman analysis.
Statistical Analysis
Intended K+ concentrations as noted on the fluid bags' adhesive label for fluid additives and measured K+ concentrations were assessed for normality by a Shapiro‐Wilk normality test and were not normally distributed. A Wilcoxon matched‐pairs signed rank test was used to compare the median intended K+ to median measured K+ concentration in clinical samples. The agreement of expected and measured K+ concentrations in stock solutions supplemented with KCl under controlled circumstances were compared using Bland‐Altman analysis. For potassium supplementation without mixing area under the curve analysis was performed for the first hour of potential delivery. Statistical analyses were performed using commercially available software.10 Results were considered statistically significant when P < .05.
Results
Potassium Supplementation in Hospitalized Dogs and Cats
A total of 210 bags of fluids administered IV in clinical use were evaluated. Nine of the 210 samples needed to be diluted 10‐fold in order to be able to measure K+ concentration. Crystalloid fluid types included lactated Ringer's® (n = 170), Plasma‐Lyte 148® (n = 38), and 0.9% NaCl (n = 2). Fluid bag sizes were 5000 mL (n = 1), 3000 mL (n = 1), 1000 mL (n = 185), and 500 mL (n = 23). Nine of the sampled bags were supplemented with KCl as partial bag volumes, at time of supplementation ranging from 300 mL to 985 mL. Of the 210 samples of fluids administered to hospitalized dogs and cats, the intended K+ concentration following supplementation ranged from 10 mmol/L to 40 mmol/L (10 mmol/L, n = 10; 20 mmol/L, n = 190; 24 mmol/L, n = 1; 25 mmol/L, n = 1; 30 mmol/L, n = 3; 34 mmol/L, n = 1; 40 mmol/L, n = 4). The concentration of KCl used for K+ supplementation was stated on the medication additive labels in 118 of the 210 cases. However, during the study period at our institution only one concentration of KCL was available (2 mmol/mL). On those labels that did not state the concentration of KCl used, either the total volume of KCl added to the bag, the total mEq of K+ added, or a combination thereof was noted.
In 3 samples, K+ concentration remained too high to be measured even when the samples were diluted 10‐fold, indicating K+ concentrations of >300 mmol/L. Two of these samples had intended K+ concentrations of 20 mmol/L and one of 25 mmol/L. Although the median K+ concentration of the measured samples was the same as for the intended K+ concentration, they were statistically significantly different because of the large variation in range, with the median measured K+ concentration being (20 mmol/L, range 9 to >300 mmol/L) compared to the median intended K+ concentration of (20 mmol/L, range 10 to 40 mmol/L; P < .0001 n = 207). Differences between measured and intended K+ concentrations ranged from −6.5 mmol/L to >280 mmol/L (mean difference 9.0 mmol/L). In 72.4% of the samples, the difference between measured and intended K+ ranged from −4.99 mmol/L to 4.99 mmol/L and in 25.7% of the samples the difference ranged from a K+ of 5 mmol/L to 179 mmol/L. Differences in measured and expected K+ concentration are shown in Table 1.
Table 1.
Difference between K+ concentration measured in the fluid bag minus the reported ‘target’ value written on the bag label, from fluids in clinical use (n = 210 samples).
| Difference in K+ (mmol/L) | n | % |
|---|---|---|
| −10.0 to −5.0 | 1 | 0.5 |
| −4.9 to + 4.9 | 152 | 72.4 |
| 5.0 to 19.9 | 31 | 14.8 |
| 20 to 59.9 | 12 | 5.7 |
| 60 to 99.9 | 4 | 1.9 |
| 100 to 199.9 | 7 | 3.3 |
| 200 to 279.9 | 0 | 0 |
| >280 | 3 | 1.4 |
Standardized Potassium Supplementation with Mixing
Bland‐Altman analysis comparing measured minus intended K+ concentration showed a mean bias of 0.7 mmol/L (95% limits of agreement −0.32 to 1.7 mmol/L).
Potassium Supplementation without Mixing
Figures 1, 2 show the mean K+ concentration of the infused fluid after slow and rapid KCl injection into fluid bags with no mixing. Maximal K+ concentration identified was 1410 mmol/L in the slow injection group and 230 mmol/L in the rapid injection group. Area under the curve analysis for the first hour of fluid discharged after slow KCl injection and no mixing show that 13.3 mmol of K+ was cleared from the bags within the first hour. After rapid injection of 20 mmol/L KCl and no mixing, area under the curve analysis shows 5.7 mmol of K+ was cleared from the bags within the first hour of delivery.
Figure 1.
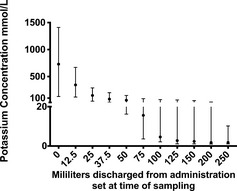
Potassium concentration of discharged fluid over time of 6 1 L bags of 0.9% NaCl supplemented with 20 mmol of potassium chloride via slow injection (≈30 seconds) and no mixing performed. The circle represents the median value and the error bars represent the range. Fluid administration rate was 50 mL/h.
Figure 2.
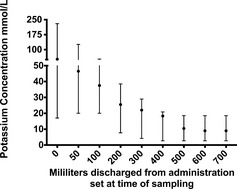
Potassium concentration of discharged fluid over time of 6 1 L bags of 0.9% NaCl supplemented with 20 mmol of potassium chloride via rapid injection (≈10 seconds) and no mixing performed. The circle represents the median value and the error bars represent the range. Fluid administration rate was 100 mL/h.
Accuracy of Potassium Measurement
Analysis of the twenty samples each of 0.9% NaCl6 supplemented to 4 mmol/L and 8 mmol/L of K+ showed a mean bias of −0.18 mmol/L (95% limits of agreement −0.45 to 0.11 mmol/L).
Discussion
In the 210 clinical samples, the comparison of measured to intended K+ concentration showed measured K+ was significantly higher although undersupplementation was also present. In 28% of the samples, measured K+ concentration was ≥5 mmol/L above or below the intended concentration. In 11% of all samples, measured K+ exceeded the target K+ concentration by 20 mmol/L to 179 mmol/L and in 3 of 210 samples, the K+ concentration was >280 mmol/L more than desired. As inadvertent administration of K+ boluses from fluid bags with such high K+ concentrations can lead to life threatening or even fatal impairment of cardiac conduction, this variability in K+ concentration could have an important clinical impact.2, 7, 8, 9 Several factors likely contribute to inaccuracies in K+ supplementation, such as inadequate mixing of additives in nonrigid fluid containers, manufacturer variability in bag fluid volumes and K+ addition, KCl injection technique, as well as human error such as miscalculations or labeling errors leading to repeat supplementation of the same bag.
Of the 210 clinical fluid bags sampled, 9 had KCl added to a partially used bag and 6 of these bags were over supplemented with excess in K+ ranging from 8 mmol/L to 154 mmol/L. Two of the 3 highest K+ concentrations found in all clinical samples were measured in bags supplemented as partial bags at 174 mmol/L and 156 mmol/L respectively. The setting of adding KCl to a bag that is already in use may make it more likely that calculation errors to determine the quantity of KCl for addition will occur. It also seems more likely that KCl addition would be performed with the bag in a hanging position when bags are already in use and that insufficient attempts to adequately mix these bags would occur.
The occurrence of inadequate mixing when supplementing fluid containers with additives such as KCl has long been recognized and has been associated with adverse drug reactions in human patients as well as companion animals.1, 3, 7, 9, 10, 13, 14, 15, 16, 17, 18 Several studies have confirmed that non‐rigid fluid containers are most susceptible to inadequate mixing of additives and have investigated different techniques to ensure more adequate mixing.1, 7, 9, 13, 14, 17 One time inversion of the infusion bag into a hanging position after additives were added to the bag in a bottom up position have been shown to provide adequate mixing when compared to injecting additives to a flexible bag already hanging in the infusion position.7, 9, 14 Other experimental studies have shown that adequate mixing can be achieved with one or two double inversions of PVC containers held by their ends.16 Studies on adequate mixing of magnesium additives to non‐rigid fluid containers show that adequate mixing can be achieved with 3 inversions of the bag and that inversing the bag 15 times provides no additional advantage.19 Shaking of PVC containers has been shown to provide less mixing of KCl than the double inversion technique and is not recommended, and squeezing the bag after additives are injected in an infusion position has even been shown to pump the additives from the dependent injection port into the infusion chamber.9, 16 No standardized mixing protocol was in place at our facility at the time of this study and a multitude of people including senior veterinary students, veterinary technicians, and veterinarians are involved in preparation of bags of fluids for IV administration and supplementation with KCl. It is therefore possible that the high K+ concentrations obtained from the bottom of PVC containers could be attributed to layering of KCl because of inadequate injection or mixing techniques.
Following K+ supplementation of stock fluids to 20 mmol/L under standardized conditions with mixing in this study, the mean difference (95% limits of agreement) was 0.7 (−0.32 to 1.7) mmol/L K+. These results show that under ideal circumstances including adequate mixing techniques, a target K+ concentration can be attained with a clinically acceptable accuracy. Although not evaluated in this study, empiric K+ supplementation based on the labeled bag volume, not the bag weight is also likely to provide K+ concentrations that are in a clinically acceptable range as long as effective mixing is performed.
Our results confirm that adequate mixing of additives can be achieved when a nonrigid fluid container is inverted 4 times, a technique most closely resembling the previously described two “rapid double inversions” technique.16 We therefore recommend implementing inversion 4 times as a standard protocol following KCl addition to a fluid bag intended for IV administration. While lower numbers of bag inversions could still provide sufficient mixing, no comparison has been made in the present study and other techniques can therefore not be recommended. Once proper mixing is able to achieve a homogenous solution of an additive within a bag, it is important to note that storage of the bag does not promote renewed layering of the additive and ions do remain in solution.17, 20
Inadequate mixing of fluid containers after supplementation with additives as described above can lead to variability in drug concentrations within that container.1, 17 Additive underdosage and overall concentration fluctuations can be as important as overdoses as the initial solution of an unmixed bag contains the majority of the additive, whereas the subsequent amounts of the infused solution contain very low to detectable concentration of the additive.1, 17 In the case of KCl, especially the accidental administration of a bolus when starting a fresh, unmixed bag could lead to life threatening adverse effects of hyperkalemia, such as cardiac arrhythmias, collapse, or sudden death.2
An uneven delivery of the supplemented K+ was evident when KCl was injected into bags in the infusion (hanging) position and no attempt was made to mix the bags. This effect was more pronounced with a slower injection speed of KCl into the bag, indicating that rapid speed of injection does provide some turbulence that contributes to mixing of the additives in addition to the higher velocity propelling the K+ solution higher into the fluid bag. Potassium concentration measurements from slowly injected KCl in unmixed bags show, that potentially lethal K+ concentrations as high as 1410 mmol/L could be administered to a dog or cat in the initially delivered fluid. Area under the curve analysis for the first 50 mL of fluid from the bag confirm, that a 10 kg dog run at an approximately twice maintenance rate of fluids administered IV at 50 mL/h could be administered a potentially lethal dose of K+ of 1.3 mmol/kg in the first hour. A positive influence of higher injection speed on mixing of fluid additives has been described, however, this was previously thought to be a property of rigid glass containers and to be ineffective in nonrigid fluid containers.1, 7, 14, 18 High discharged K+ concentrations are still found with high injection speeds and an appropriate mixing technique is still essential.
This study has several limitations. The electrolyte measurements performed in this study were performed on a blood gas and electrolyte analyzer intended for electrolyte measurements in heparinized human whole blood and not validated for electrolyte measurements in crystalloid fluids and the measurements were not repeated on a second analyzer for verification. However, our validation experiments measuring K+ concentration in samples of known K+ supplementation confirm a satisfactory level of agreement between known and measured K+ concentration with a mean bias of −0.18 mmol/L and 95% limits of agreement of −0.45 to 0.11 mmol/L. Potassium values were measured by dilution (5 to 10‐fold for samples of hospitalized dogs and cats and 5 to 100‐fold for potassium supplementation without mixing) which could exacerbate these errors by the dilution factor. The maximum underestimation and overestimation of samples with K+ concentrations high enough to necessitate 100‐fold dilutions (no mixing samples only) would therefore be 45 mmol/L and 11 mmol/L, respectively, leaving inaccuracies in K+ measurement technique unable to explain differences of up to 1390 mmol/L between measured to intended K+ concentrations found in the present study. For samples obtained from bags in clinical use, the dilutions were mostly 5‐fold, the 95% confidence limits of the samples variability from technique on a sample measuring 20 mmol/L K+ would be 17.75 to 20.55 mmol/L which would not alter the interpretation of our results.
A commercially available KCl solution with a concentration of 2 mmol/mL was used through out this study but we could not confirm the concentration of the product used for supplementation of the fluids administered IV to hospitalized dogs and cats. Although some variability in the solution is possible, the accuracy of K+ supplementation achieved in this study under standardized conditions suggests such variability is not of any clinically relevant degree. Lastly, the clinical samples were only collected from the region of the injection port in the hanging bag. To differentiate inadequate mixing from incorrect K+ addition samples from different levels within the bag or sampling before and after mixing the bag would have been ideal.
In conclusion, the measured K+ concentration was significantly higher than the intended K+ concentration of fluids administered IV in clinical use at our institution during the 3‐month study period. Many factors could have contributed to this finding but inadequate mixing following K+ supplementation was suspected to be a major cause. The experimental studies presented here show that inadequate mixing of fluid bags intended for IV administration of fluids following K+ supplementation can lead to dangerously high infused K+ concentrations and demonstrate the importance of standard protocols in conjunction with appropriate staff education to ensure adequate mixing and labeling of bags. A protocol such as injection of potassium into the bag while held in an inverted position, followed by a double inversion, as we performed in this study, is a relatively easy and reliable method for potassium supplementation.
Acknowledgments
Funding: There was no funding source for this study.
Conflict of Interest Declaration: Authors disclose no conflict of interest.
Off‐label Antimicrobial Declaration: Authors declare no off‐label use of antimicrobials.
Partial results of this study have been presented as an oral abstract at the 36th annual Gerald V. Ling House Officer Seminar Day, School of Veterinary Medicine, University of California, Davis, CA and as a poster at the 20th International Veterinary Emergency & Critical Care Symposium, Indianapolis, IN.
Footnotes
Unpublished data, Hopper, K, University of California, Davis, CA
Baxter Healthcare Corporation, Deerfield, IL
Covidien, Mansfield
ABL 815, Radiometer Medical A/S, Copenhagen, Denmark
Pelouze®, Pelstar LLC, Bridgeview, IL
Hospira, Inc., Lake Forest, IL
Eppendorf North America, Inc., Hauppauge, NY
Baxter Flo Gard®, Baxter Healthcare Corporation, Deerfield, IL
ABL 800 FLEX reference manual, Radiometer Medical ApS, Bronshoj, Denmark
GraphPad Prism 6.0, Graph Pad Software, La Jolla, CA
References
- 1. Gong H Jr, King CY. Inadequate drug mixing: a potential hazard in continuous intravenous administration. Heart Lung J Crit Care 1983;12:528–532. [PubMed] [Google Scholar]
- 2. Lawson DH. Adverse reactions to potassium chloride. Q J Med 1974;43:433–440. [PubMed] [Google Scholar]
- 3. Lawson DH. The clinical use of potassium supplements. J Maine Med Assoc 1975;66:166–169. [PubMed] [Google Scholar]
- 4. Anon . Mixing drugs with intravenous infusions. Drug Ther Bull 1970;8:53–56. [PubMed] [Google Scholar]
- 5. DiBartola SP, De Morais HA. Disorders of potassium: hypokalemia and hyperkalemia In: DiBartola SP, ed. Fluid, Electrolyte, and Acid‐Base Disorders in Small Animal Practice, 4th ed St. Louis: Elsevier Saunders; 2012:92–119. [Google Scholar]
- 6. Feldman EC, Church DB. Electrolytes disorders: potassium (hyper/hypokalemia) In: Ettinger SJ, Feldman EC, eds. Textbook of Veterinary Internal Medicine, 7th ed St. Louis: Elsevier Saunders; 2010:303–307. [Google Scholar]
- 7. Lankton JW, Siler JN, Neigh JL. Letter: hyperkalemia after administration of potassium from nonrigid parenteral‐fluid containers. Anesthesiology 1973;39:660–661. [DOI] [PubMed] [Google Scholar]
- 8. Shapiro S. Fatal drug reactions among medical inpatients. J Am Med Assoc 1971;216:467. [PubMed] [Google Scholar]
- 9. Williams RH. Potassium overdosage: a potential hazard of non‐rigid parenteral fluid containers. Br Med J 1973;1:714–715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Dhein CR, Wardrop KJ. Hyperkalemia associated with potassium chloride administration in a cat. J Am Vet Med Assoc 1995;206:1565–1566. [PubMed] [Google Scholar]
- 11. Sanders LH, Mabadeje SA, Avis KE, et al. Evaluation of compounding accuracy and aseptic techniques for intravenous admixtures. Am J Hosp Pharm 1978;35:531–536. [PubMed] [Google Scholar]
- 12. Külpmann WR, Höbbel T. International consensus on the standardization of sodium and potassium measurements by ion‐selective electrodes in undiluted samples. Scand J Clin Lab Invest Suppl 1996;224:145–160. [DOI] [PubMed] [Google Scholar]
- 13. Bergman N, Vellar ID. Potential life‐threatening variations of drug concentrations in intravenous infusion systems: potassium chloride, insulin, and heparin. Med J Aust 1982;2:270–272. [PubMed] [Google Scholar]
- 14. Bighley LD, Wille J, Lach JL. Mixing of additives in glass and plastic intravenous fluid containers. Am J Hosp Pharm 1974;31:736–739. [PubMed] [Google Scholar]
- 15. Deardorff DL, Schmidt CN, Wiley RA. Effect of preparation techniques on mixing of additives in intravenous fluids in nonrigid containers. Hosp Pharm 1993;28:306, 309–310, 312–313. [PubMed] [Google Scholar]
- 16. Deardorff DL, Schmidt CN. Mixing additives in plastic LVPs. Am J Hosp Pharm 1980;37:1610, 1613. [PubMed] [Google Scholar]
- 17. Thompson WL, Feer TD. Incomplete mixing of drugs in intravenous infusions. Crit Care Med 1980;8:603–607. [DOI] [PubMed] [Google Scholar]
- 18. Todd B. Intravenous drug hazards: interactions, adsorption, and inadequate mixing. Geriatr Nurs 1988;9:20, 22. [DOI] [PubMed] [Google Scholar]
- 19. Whang R, Papper S, Fryer A. Intravenous magnesium–potential hazard of inadequate mixing. J Am Coll Nutr 1983;2:97–100. [DOI] [PubMed] [Google Scholar]
- 20. Donaldson TM, Mani V, Wheeler DW. Factors affecting the concentration of electrolyte infusions prepared from stock solutions. Postgrad Med J 2011;87:83–88. [DOI] [PubMed] [Google Scholar]


