Abstract
High frequency oscillations (HFOs) constitute a novel trend in neurophysiology that is fascinating neuroscientists in general, and epileptologists in particular. But what are HFOs? What is the frequency range of HFOs? Are there different types of HFOs, physiological and pathological? How are HFOs generated? Can HFOs represent temporal codes for cognitive processes? These questions are pressing and this symposium volume attempts to give constructive answers. As a prelude to this exciting discussion, we summarize the physiological high frequency patterns in the intact brain, concentrating mainly on hippocampal patterns, where the mechanisms of high frequency oscillations are perhaps best understood.
Keywords: Ripples, Hippocampus, Neocortex, Gamma, Sharp wave, Spike and wave, Epilepsy, Memory
To deal with the main questions of the symposium, it is useful to consider briefly how neurophysiologists have characterized neuronal oscillations, in general, as reflected in local field potentials (LFP) and therefore in EEG and MEG signals. Since the early days of human neurophysiology scientists have been fascinated by the variety of oscillations that may be recorded from the scalp and directly from within the brain (Buzsáki, 2006). Empirically a classification of these oscillation in a series of frequency bands emerged, which were designated by Greek letters (δ, θ, α/µ, β, γ) a classification that was supported by multivariate statistical analysis of EEG spectral values in the seventies (Lopes da Silva, 2011). In the early descriptions of EEG high frequency components did not occupy a prominent place; frequency components higher than about 30 Hz (gamma band and higher) was an unchartered continent.
Two main factors changed considerably this picture in the last decades:
the rise of broad-band digital EEG what made possible to record signals in human beyond the traditional low-pass filtered EEG at 70 Hz, extending the recordings to frequencies as high as 500 Hz and beyond
novel findings in animal neurophysiology showing the existence of oscillations at frequencies in the range of 30–600 Hz in several cortical and sub-cortical brain areas (for early literature on this subject see Bressler and Freeman, 1980).
Among the latter factors, a number of specific phenomena attracted particular attention: oscillations around 40 Hz in the visual cortex associated with visual perception (reviewed in Singer and Gray, 1995), and in the sensorimotor cortex related to motor activity (Murthy and Fetz, 1992). The former was proposed to form the mechanism by which various features of a visual scene may be bound together (“binding hypothesis”) into a percept. Beyond this observation, It was also shown that gamma oscillations may operate as a general mechanism that is capable of binding together, by a process of phase synchronization, not only the firing of neurons at the local level, but also neural activities of spatially separate cortical areas(Roelfsema et al., 1997).Several studies support the hypothesis that the gamma rhythm may play a role in encoding information (Lisman and Idiart, 1995; Tallon-Baudry et al., 1998; Sederberg et al., 2007; Fell et al., 2006). Such studies demonstrate that oscillations in the gamma range (30–90 Hz) are not only relevant to binding of visual perceptions but have a broader connotation and appear to underlie information processing, working- and long-term memory (Jensen et al., 2007). As discussed by Buzsáki, 2006 and Fries (2009), gamma-band synchronization should be considered a general operational mode of cortical processing of information working through a mechanism of entrainment of neuronal networks. This would promote the transfer of relevant information between distinct brain systems.
What defines the identity of a rhythm? LFP frequency bands have been traditionally and artificially defined by frequency border criteria rather than mechanisms. From the pen-recorder perspective of brain rhythm, virtually every pattern above 30 Hz or so has been referred to as high frequency oscillation. The term ‘fast gamma’ (Csicsvari et al., 1999a,b) or ‘high gamma’ (Canolty et al., 2006) has been introduced to describe the frequency band between 90 Hz and 140 Hz in the cortex (or even up to 600 Hz; Gaona et al., 2011). Recent findings suggest that although multiple gamma oscillations may be interacting in the 30– 90 Hz band, they share numerous common features (Tort et al., 2010; Belluscio et al., 2012). Frequencies above 90 Hz (epsilon band) also occupy several distinct bands with different physiological mechanism but they are quite different from the gamma band proper (30–90 Hz; Buzsáki and Wang, 2012). A further important note is that increased power in a given band does not warranty the presence of an oscillation. In each and every case, appropriate methods should be used to demonstrate the presence of genuine oscillations. High frequency power increase above 100 Hz most often represents the power of increased frequency and synchrononous neuronal spikes rather than synaptic potentials or intrinsic membrane potential changes (Buzsáki, 1986; Ray and Maunsell, 2011; Belluscio et al., 2012).
Among the family of true HFOs, hippocampal “ripples” (O’Keefe and Nadel, 1978; Buzsáki et al., 1983, 1992), kindled the interest for a variety of reasons discussed below, followed by several other HFOs in the intact cortex. Hippocampal ripples represent the most synchronous physiological patterns in the mammalian brain. Controlling such strong synchrony is a delicate process and any interference with the physiological recruitment of neurons into ripples may turn them into pathological patterns that can compromise the circuits from which they emanate. Indeed, even faster short transient oscillations, named “fast ripples” in the temporal cortex of epileptic humans and rodents were discovered subsequently (Bragin et al., 1999a,b,c). Our discussion below is confined though to physiological ‘ripple’ type oscillations.
1. Hippocampal sharp waves and ripples
Large amplitude local field potentials (‘sharp waves’; SPWs) occur irregularly in the hippocampal CA1 str. radiatum when the animal has minimal or no interaction with its environment, such as immobility, consummatory behaviors or slow wave sleep (Buzsáki et al., 1983). SPWs reflect the depolarization of the apical dendrites of CA1 and CA3 pyramidal cells, due to the synchronous bursting of CA3 pyramidal cells (Fig. 1). The population bursts in the CA3 region are supported by the spread of excitatory activity in the strongly recurrent CA3 collateral system at times when the burst-suppressive effects of the subcortical inputs are reduced (Buzsáki et al., 1983). The widespread axon collaterals of the CA3 pyramidal cells, recruited into a SPW event, can broadcast their collective excitation over a large volume of the CA3 and CA1 region (Ylinen et al., 1995). The CA3 output also discharges various CA1 and CA3 interneurons (Csicsvari et al., 1999a,b, 2000; Klausberger et al., 2004; Klausberger and Somogyi, 2008). In turn, the local interaction between pyramidal cells and interneurons triggers a short-lived ripple oscillation (O’Keefe and Nadel, 1978; Buzsáki et al., 1992; Ylinen et al., 1995; Csicsvari et al., 2000; Brunel and Wang, 2003; Foffani et al., 2007; Klausberger et al., 2004). Additional mechanisms, such as gap junction-mediated effects (Draguhn et al., 1998; Schmitz et al., 2001; Traub and Bibbig, 2000; Ylinen et al., 1995) and ephaptic entrainment of neurons by the large SPW field (Anastassiou et al., 2010), may also contribute to the highly spatially localized ripple episodes (Chrobak and Buzsáki, 1996; Csicsvari et al., 2000). The main generators of the LFP ripples are the synchronously discharging pyramidal cells responsible for the negative peaks of the ripples (‘mini populations spikes’, Buzsáki, 1986; Reichinnek et al., 2010) and synchronous IPSCs in nearby pyramidal cells brought about by basket neurons. The synchronously discharging dense axon terminals of basket cells in the pyramidal layer may also contribute to the LFP ripple but this possibility has not been tested rigorously. The spike content of the ripple may explain why increasing the frequency (e.g., from fast gamma to ripples) increases the amplitude of the ripple waves, opposite to what one might expect from summation of increasing frequency of PSPs. This may be so because at higher frequencies the temporal overlap of the spikes of neighboring neurons increases.
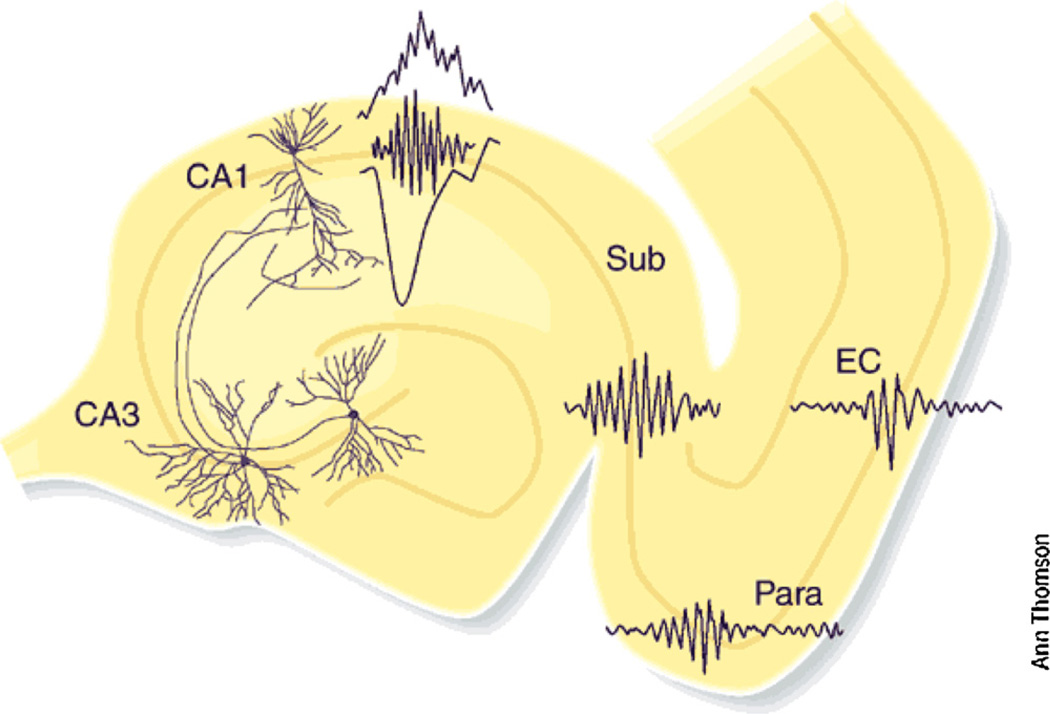
Fig. 1.
Self-organized burst of activity in the CA3 region of the hippocampus produces a sharp wave sink in the apical dendrites of CA1 pyramidal neurons and also discharge interneurons. The interactions between the discharging pyramidal cells and interneurons give rise to a short-lived fast oscillation (‘ripple’; 140–200 Hz), which can be detected as a field potential in the somatic layer. The strong CA1 population burst brings about strongly synchronized activity in the target populations of parahippocampal structures as well. These parahippocampal ripples are slower and less synchronous, compared to CA1 ripples. Reprinted from Buzsáki and Chrobak (2005).
While the spatially widespread SPWs and the locally confined ripples (and the related fast gamma bursts, 90–140 Hz; see below; Sullivan et al., 2011) are strongly linked in the intact brain, they are distinct physiological events and can be dissociated in both normal tissue and pathology (Csicsvari et al., 2000; Leinekugel et al., 2002; Bragin et al., 1999a,b,c, 2002; Buhl and Buzsáki, 2005; Engel et al., 2009; Nakashiba et al., 2009; see chapters of this volume). Their separation is amply demonstrated during development. Whereas sharp waves and associated population bursts of pyramidal neurons represent the first hippocampal pattern after birth in the rat (Leinekugel et al., 2002), ripples are among the latest maturing network patterns (Buhl and Buzsáki, 2005). The strong coincidence of SPWs and ripples (often called SPW-ripples, SPW-R) represent one of the most robust examples of cross-frequency coupling in the brain, with relatively well-understood coupling mechanisms.
SPWs and associated ripples (SPW-R) have been observed in every mammalian species investigated so far, including humans (Buzsáki et al., 1992, 2003; Bragin et al., 1999a,b; Le Van Quyen et al., 2010). SPW-Rs are the most synchronous events in the mammalian brain, associated with a robustly enhanced transient excitability in the hippocampus (Buzsáki, 1986). SPW-Rs are emergent population events; indeed observing single neurons separately does not indicate their occurrence, since in a given SPW-R event a single pyramidal neuron typically fires a single spike or a single burst (Buzsáki et al., 1992). However, the temporal coordination of firing across many neurons transpose the single neuron events into a robust population event. In the time window of SPW-R (50–150 ms), 50,000–100,000 neurons (10–18% of all neurons) discharge synchronously in the CA3-CA1-subicular complex-entorhinal cortex of the rat (Chrobak and Buzsáki, 1996; Csicsvari et al., 1999a,b), representing a several-fold increase of population synchrony compared to theta oscillations (Csicsvari et al., 1998). Largely because of this strong excitatory gain, SPW-Rs are an ideal mechanism for transferring information and inducing plasticity (Chrobak and Buzsáki, 1994, 1996).
2. Neuronal content of SPW-Rs and their role in memory consolidation
Neuronal participation in the population discharge of SPW-R is not random but shaped by previous experience of the animal (Buzsáki, 1989; McClelland et al., 1995; Kudrimoti et al., 1999; Nádasdy et al., 1999; Skaggs and McNaughton, 1996; Wilson and McNaughton, 1994; Lee and Wilson, 2002; Foster and Wilson, 2006; Diba and Buzsáki, 2007; O’Neill et al., 2006, 2008; Ponomarenko et al., 2008; Dupret et al., 2010; Foster and Wilson, 2006; Diba and Buzsáki, 2007; Karlsson and Frank, 2008, 2009; Davidson et al., 2009; Singer and Frank, 2009). Such replay of experience-related sequence can occur during both waking immobility and slow wave sleep. Fig. 2 illustrates that the replay of neuronal activity during SPW-Rs may depend on the behavioral events that surround the SPW-R. Prior to the journey on a linear track, the sequence of neuronal activity during SPW-Rs is similar to the sequence of the place-related firing while the rat traverses the track. After traversing the track, the sequences of place cells are often reactivated in the reverse of the order experienced behaviorally, as if the network was ‘backtracking’ the visited places. This reverse replay of events may serve to link behavioral trajec tories to their reward outcomes (Foster and Wilson, 2006). In contrast, forward replay that occur preferentially at the beginning of runs may serve to ‘rehearse’ the planned movement trajectories (Diba and Buzsáki, 2007).
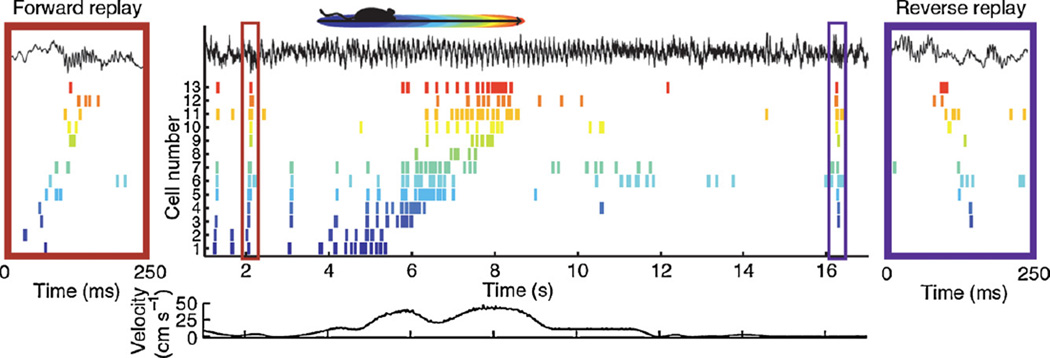
Fig. 2.
Place cell sequences experienced during behavior are replayed in both the forward and reverse direction during awake SPW-Rs. Spike trains for 13 place fields on the track are shown before, during and after a single traversal. Sequences that occur during track running are reactivated during SPW-Rs both prior to and after the run, when the rat stays immobile. Forward replay (left inset, red box) occurs before traversal of the environment and reverse replay (right inset, blue box) after. The CA1 local field potential is shown on top and the animal’s velocity is shown below.
Reprinted from Diba and Buzsáki (2007).
During slow wave sleep, SPW-Rs occur at relatively high density and intensity. Typically, the spike sequences are forward, meaning that the temporal order of spikes reflect the order of the events experienced in the waking state animal (Nádasdy et al., 1999; Skaggs and McNaughton, 1996; Lee and Wilson, 2002). Currently, it is not clear why in the waking state the sequences can move both forward and backward in time, whereas during sleep the direction is mainly forward. In fact, no current neurophysiological mechanism can explain satisfactorily how neurons in the complex matrix of the CA3 recurrent system can generate a forward trajectory at one moment and reverse the direction of activity propagation at another moment. Furthermore, no study has examined the content of SPW-Rs during sleep rigorously enough to dismiss the possibility of reverse replay during sleep.
Direct evidence for a causal role of SPW-Rs in memory consolidation comes from studies in which ripples were selectively suppressed after learning. In those experiments, the animals were trained daily in a hippocampus-dependent reference memory task, after which they were allowed to sleep. During post-learning sleep, CA1 ripples were detected bilaterally in the CA1 pyramidal layer. Whenever a ripple was detected, it was aborted by timed electric stimulation delivered to the hippocampal commissure. Such targeted interference did not affect other aspects of sleep but produced large performance impairment in daily performance, comparable to that observed after surgically damaging the hippocampus (Girardeau et al., 2009). The causal role of SPW-Rs assisting learning and memory was demonstrated in another experiment as well (Ego-Stengel and Wilson, 2010). It should be point out thought that while these pattern perturbation and elimination experiments demonstrates the vital importance of SPW-Rs, they do not provide information about the nature of the physiological processes that underlie their role in memory transfer and plasticity. A main problem is that eliminating SPW-Rs interferes not only with the spike contribution of wake-active neurons but with the entire dynamic of the population events with many neuronal contributors. No experiments have yet been performed to ‘erase’ waking SPW-Rs. Such experiments may elucidate whether the physiological functions of waking and sleep related ripples are similar or different. They may also elucidate for example whether SPW-Rs are essential modifying hippocampal networks during exploration of novel environments.
3. Constructive role SPW-Rs
While most studies discuss SPW-R replays in terms of place cell activation of spatial navigation, ample evidence is available to demonstrate that such off-line activity is not simply a replay part of a memory consolidation. Several recent experiments point to the direction that the spike content of SPW-Rs does not exclusively relate to the current situation. For example, when the rat sits in the home cage or the home base of a training apparatus, spike sequences rarely correspond to the place cells active in that environment but to those sequences experienced during the course of recent learning in a different environment (Dupret et al., 2010; cf., Carr et al., 2011; Derdikman and Moser, 2010). Thus, new experience rather than simple repetition might be critical for the selections of neurons for SPW-Rs. These and other experiments (Mehta, 2007; Gupta et al., 2010) also indicate that recency is also not the most critical ingredient of the neuron selection mechanism. Finally, new evidence is accumulating, which shows a constructive, in addition to the replay role of SPW-Rs. The reverse replay of is already an indication that the pattern of activity during SPW-Rs is not simply a compressed version of the experienced sequence of cell assembly firing. Many more sequences occur in SPW-Rs than could be accounted for by the forward or reverse sequences of experienced place cells (Nádasdy et al., 1999). Sequences that predict the place cell sequences on not-yet experienced tracks have been detected during both waking (Gupta et al., 2010) and sleep prior to the visits to the novel routes (Dragoi and Tonegawa, 2011). These findings can be interpreted in multiple new ways. First, that during exploration the experienced places are anchored by the emerging place cells but the flexible routes among all possible places are laid down by the combinatorial sequences that occur during SPW-Rs, in line with the graph theory of map-based navigation (Muller et al., 1996). The other possibility is that the SPW-R-constructed neuronal paths during sleep constrain the combinatorial possibilities in the large recurrent system of CA3 pyramidal cells (Wittner et al., 2007) and consequently the possible neuronal sequences during exploration of novel environments. Such constructive or anterograde role of SPW-R is also supported by the observation that SPW-Rs are the first organized patterns in the hippocampus, present at birth (Leinekugel et al., 2002), that is much before the emergence of place cells and entorhinal grid cells (Wills et al., 2010; Langston et al., 2010). Importantly, it should be emphasized that while most of these experiments have been discussed in the framework of spatial navigation, they may equally apply to ‘cognitive navigation’ during episodic memory recall and planning (Pastalkova et al., 2008). The constructive nature of SPW-Rs can be tested by ‘inserting’ synthetic sequences of neurons during sleep using optogenetic methods.
4. SPW-Rs are embedded in slow events and affect wide cortical areas
SPWs emerge in the recurrent collateral system of the CA3 hippocampal region as results of releasing these excitatory fibers from the relatively tonic subcortical suppression during theta oscillation states (Buzsáki et al., 1983). However, the exact timing of the population events can be biased by various inputs impinging upon the hippocampus. When the CA3 subregion is isolated from the rest of the brain and kept in vitro, SPW-like bursts occur super-regularly at 2–4 Hz (Papatheodoropoulos and Kostopoulos, 2002; Kubota et al., 2003; Maier et al., 2003), in contrast to the irregular SPW-R intervals typical in vivo. Thus, the in vitro observations imply that a low frequency oscillator can trigger SPW-related population events. In contrast, in the intact brain the occurrence of subsequent SPW-R events can be advanced by the hippocampal inputs. Both the dentate gyrus and the entorhinal cortex can effectively bias the CA3-CA1 events by transferring neocortical patterns. As a result, the occurrence of SPW-Rs is phase-modulated by sleep spindles (12–16 Hz; Siapas and Wilson, 1998), both of them by slow oscillations (0.5–1.5 Hz; Steriade et al., 1993), and each of the three rhythms by the ultraslow (0.1 Hz) oscillation (Sirota et al., 2003; Isomura et al., 2006; Mölle et al., 2006; Isomura et al., 2006; Sullivan et al., 2011). Such multiple time scale, cross-frequency coupling can effectively generate irregular patterns because of the interference between the hypothesized SPW-pacing oscillator in the CA3 network and the entorhinal cortex-mediated amplitude varying inputs, and possibly subcortical afferents.
At the output side, the super-synchronous SPW-Rs can affect large territories of the neocortex. These effects are mediated largely by the entorhinal cortex and the medial prefrontal cortex (Chrobak and Buzsáki, 1994, 1996; Battaglia et al., 2004; Wierzynski et al., 2009). The exact impact of SPW-Rs on neocortical activity is hard to assess because there appears to be a functional topographical organization between the hippocampus and neocortex (Royer et al., 2010). CA1 ripples are strongly localized (Chrobak and Buzsáki, 1996; Csicsvari et al., 2003) and therefore ripples, e.g., in the ventral hippocampus may not affect entorhinal neurons in its medial dorsolateral part, a region that does not receive inputs from the ventral hippocampus. Conversely, ripples confined to the dorsal hippocampus may not strongly affect neurons in the prefrontal cortex since hippocampo-frontal cortex afferents arise from the posterior-ventral segments of the hippocampus.
The subcortical impact of SPW-Rs is rarely considered. This is surprising since subcortical structures, such as the lateral septum, hypothalamus and nucleus accumbens, are major targets of the CA3-CA1-subicular systems. Although the important controlling role of the hippocampus on the hypothalamic-pituitary system is well-known, it is much less clear how under what physiological conditions do hippocampal neurons exert their effects. The strongly synchronous hippocampal SPW-Rs may be the appropriate neuronal substrate to play such control (Buzsáki, 1998).
5. Ripples and fast gamma oscillations
Recent experiments demonstrate strong physiological similarities between ripples (~140–220 Hz) and fast gamma (or ‘epsilon’ band; 90–150 Hz) oscillations in the rodent hippocampus (Sullivan et al., 2011). These observations are important because different hippocampal regions can oscillate at different frequencies during a given SPW-R event and oscillations at adjacent frequencies can occur in the same region (Sullivan et al., 2011; Le Van Quyen et al., 2010). For example, during SPWs, the fastest (ripple; 140–220 Hz) oscillations are present in the CA1 pyramidal layer, whereas slower (fast gamma; 90–150 Hz) oscillations dominate the CA3 region and parahippocampal structures (Chrobak and Buzsáki, 1996; Sullivan et al., 2011). These findings illustrate that in the absence of theta oscillations, the CA1 region can respond in two different ways to CA3 inputs: either with a transient, fast gamma oscillation (~90–150 Hz) or in the form of a ripple (~140–220 Hz). Such regional and state differences can be understood in light of the different resonant properties of the CA1 and CA3 networks (Sullivan et al., 2011). The SPW-related population burst provides the necessary depolarization force for network resonance and both CA3 and CA1 networks respond with oscillations. Both regions possess resonance in two frequency ranges, centered at approximately 110 Hz in CA3 and 170 Hz in CA1 (Sullivan et al., 2011). Under strong but still physiological excitatory conditions, the CA3 region resonates at <110 Hz while the CA1 at 170 Hz. When excitation is weaker, as reflected by smaller SPWs, both regions resonate at approximately 110-Hz. This hypothesis also predicts that excessive excitation of the CA3 network should also generate ripples at the same frequency as in CA1, as has been shown under challenged conditions in vitro, where both CA3 and CA1 regions generate ripple or higher frequency oscillations (de la Prida and Gal, 2004; Behrens et al., 2005; Draguhn et al., 1998; Dzhala and Staley, 2004; Bragin et al., 1999a,b, 2000; Both et al., 2008; Liotta et al., 2011).
The presence of epsilon (or fast/high gamma; 90–150 Hz) frequency patterns during non-theta and theta oscillations (Csicsvari et al., 1999b; Colgin et al., 2009; Sullivan et al., 2011) begs the question about the mechanisms of these oscillations in different states. A distinct and puzzling difference between these similar rhythms is that while during theta oscillations epsilon band episodes occur near the trough of the CA1 pyramidal layer theta cycle (Colgin et al., 2009; Belluscio et al., 2012), in the absence of theta epsilon patterns occur at the peak of the underlying CA1 pyramidal layer wave (Sullivan et al., 2011), corresponding to a smaller amplitude SPW. Thus, the mechanisms of cross-frequency coupling, i.e., the inducing process may be different but the mechanisms of the epsilon band oscillations in the two states may be similar.
6. Limiting the propagation of SPW-Rs by inhibition
An elaborate temporal coordination of inhibitory interneurons secures the orderly recruitment of pyramidal cells into SPW-Rs. In any given SPW-R event, approximately 10–15% of the local pyramidal cells discharge, whereas the majority of pyramidal neurons are suppressed by inhibition. Perisomatic basket, chandelier neurons and trilaminar cells are strongly recruited SPW-Rs and their elevated discharge may be responsible for both preventing the recruitment of a much larger fraction of pyramidal cells and terminate the population event underlying SPW-Rs (Csicsvari et al., 1999a,b). In contrast, other neurons, such as O-LM cells, are transiently silenced, whereas bistratified inteneurons show biphasic effects (Klausberger et al., 2003).
The strong population synchrony underlying SPW-R bursts can propagate simultaneous into multiple directions, including the subicular complex, cortical sites (Chrobak and Buzsáki, 1994, 1996; Battaglia et al., 2004; Wierzynski et al., 2009; Peyrache et al., 2009) and subcortical structures (Buzsáki, 1998). In the hippocampus-entorhinal loop, the excitatory gain is dissipated by the increased recruitment of inhibition at subsequent stages of the hippocampus-entorhinal system. A main function of the SPW-R is to transiently shift the balance of excitation and inhibition so that the hippocampal output can have a large impact on its target structures. In the time window of the SPW-R, CA1 pyramidal cells increase their discharge rates by 5 to 6-fold, whereas interneurons increase their rates only by 2–3-fold, creating an effective 2–3-fold gain of excitation. This gain dissipates in the subiculum, largely balanced in layer 5 of the entorhinal cortex and in layers 2 and 3 an inhibitory gain dominates (Fig. 3). Such inhibitory gating or filtering mechanism may serve to route the spread of hippocampal excitation during SPW-Rs to various neocortical sites, return the pattern to the hippocampus or prevent further propagation of activity altogether.
Fig. 3.
Gain and loss of excitation in different hippocampal-entorhinal regions during SPW-Rs. Population means of ripple-unit cross-correlograms in CA1, CA3 and dentate gyrus (DG) of the hippocampus and layers II, III and V of the entorhinal cortex (EC2, EC3, EC5). Principal cells and putative interneurons are shown in the left and middle columns, respectively. Peak of the ripple episode is time 0. Right column, Relative increase of neuronal discharge, normalized to the baseline (−200 to 200 ms) for both pyramidal cells (pyr, gray line) and interneurons (int, black line). The ratio between the relative peaks of pyramidal cells and interneurons is defined as ‘gain’. Note largest excitatory gain in CA1, flowed by CA3 and EC5. Gain is balanced in DG and EC2, whereas in EC3 inhibition dominates. Data from Mizuseki et al. (2009).
The complex orchestration of SPW-Rs by the inhibitory interneuron system demonstrates that large resources are dedicated by the hippocampus to allow the recruitment of large yet regulated fraction of pyramidal cells for an expected important benefit for the brain. At the same time, this complex system is highly vulnerable and may be affected in multiple pathological diseases, such as epilepsy.
7. Physiological fast ripples in the intact neocortex
The fast field oscillations also are present also in the neocortex and the evoking conditions are similar to those described in the hippocampus. Prerequisite of the emergence of such oscillations is strong depolarization of cortical pyramidal cells. Transient fast oscillations (‘neocortical ripples’; 400–600 Hz) occur spontaneously during sleep spindles and especially during high voltage spindles (HVS) in rodents (Fig. 4; Kandel and Buzsáki, 1997). HVSs are thalamocortical rhythms, which involve strong depolarization and discharge of neurons in both supragranular and infragranular neurons. The maximum amplitude of the ripple oscillation occurs in layer V, associated with phase-locked discharge of pyramidal cells and putative interneurons. The physiological mechanisms and significance of the fast network oscillation has yet to be clarified. The critical role of the thalamus in driving neocortical ripples is shown by the effectiveness of thalamic stimulation in inducing ripples (Kandel and Buzsáki, 1997). Thus besides excitatory connections, inhibitory processes probably play a major role in the pacing of ripples (Grenier et al., 2001). High frequency ripples can also be evoked in the somatosensory cortex by strong whisker stimulation in the anesthetized rat (Staba et al., 2005) or by stimulation of the median nerve in humans (Curio, 1999).
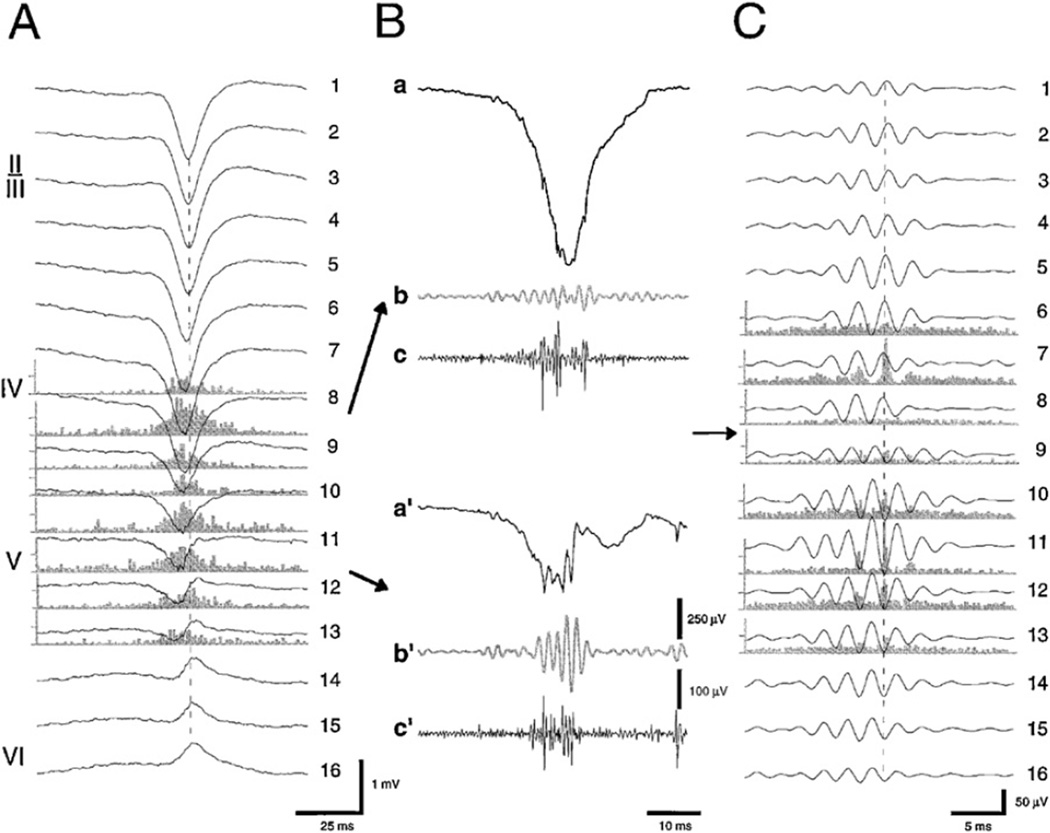
Fig. 4.
Spontaneously occurring fast ‘ripple’ oscillations (400–500 Hz) in the neocortex of the rat during high-voltage spindles. (A) Averaged high-voltage spindles and associated unit firing histograms from layers IV–VI. (B) Wide-band (a and a′; 1 Hz–5 kHz), filtered field (b and b′; 200–800 Hz), and filtered unit (c and c′; 0.5–5 kHz) traces from layers IV and V, respectively. (C) Averaged fast waves and corresponding unit histograms. The field ripples are filtered (200–800 Hz) derivatives of the wide-band signals recorded from 16 sites. Note the sudden phase-reversal of the oscillating waves (arrows) but locking of unit discharges (dashed lines). These phase reversed dipoles likely reflect synchronous discharge of layer 5 neurons in the vicinity of the recording electrode.
Reprinted from Kandel and Buzsáki (1997).
8. Fast oscillations in disease
Physiological population patterns in the brain are characterized by their strict bounds of both duration and synchrony. Although their dynamics occasionally bear features of scale-free behavior, both their magnitude and recruitment speed are effectively controlled by multiple physiological mechanisms. Hippocampal SPWs vary in both parameters but SPWs in the rodent never exceed 3 mV s or involve more than 18% of the pyramidal neurons under physiological conditions. Because of the effective buffer mechanisms that curb scale-free dynamics, our brains can operate for decades without a single supersynchronous event (Buzsaki, 2006). This organization is in sharp contrast to epileptic patterns in which scale-free behavior dominates and, therefore, event magnitudes vary several orders of magnitude. In principle, such pathological patterns can be brought about by two different classes of mechanisms. In the first case, the homeostatic regulatory mechanisms break down. For example, in the case of impaired inhibitory control, recruitment of pyramidal neurons occurs much faster and may involve excess number of neurons. These patterns reflect an ‘exaggerated’ version of physiological activity. Alternatively, an entirely novel mechanism(s) can give rise to e.g., a superfast oscillation. The emerging pathological patterns, in turn, can bring about two kinds of changes in the brain. First, they compete and interfere with the native patterns locally. For example, they can interfere with the neuronal sequences brought about by the normal SPW-Rs and add to them an arbitrary set of neurons, which do not represent the previously experienced patterns of activity. Second, the hypersynchronous events can broadcast the arbitrary neuron content to wide areas of the brain. Such mechanisms may underlie hallucinations and paranoid behavior, often present in patients with complex partial seizures. They may also affect hypothalamic and other subcortical circuits in unintended ways, owing to their powerfully synchronizing features. As the discussions of the various chapters of this volume amply indicate, there is no shortage of mechanisms that can go wrong in the network control of neuronal interactions. Controlling the emergence of pathological patterns by drugs or other therapeutic means, therefore, requires an understanding of the physiological mechanisms that are responsible for organizing population synchrony.
Acknowledgments
This work was supported by the J.D. McDonnell Foundation, NSF Grant SBE 0542013, National Institutes of Health Grant NS034994, National Institute of Mental Health Grant MH54671 and the Human Frontiers Science Program.
Abbreviations
- HFO
high frequency oscillations
- LFP
local field potentials
- EEG
electroencephalogram
- MEG
magnetoencephalogram
- SPW
sharp wave
- SPW-R
sharp wave ripple
- O-LM cells
oriens-lacunosum-moleculare interneurons
References
- Anastassiou CA, Montgomery SM, Barahona M, Buzsáki G, Koch C. The effect of spatially inhomogeneous extracellular electric fields on neurons. J. Neurosci. 2010;30:1925–1936. doi: 10.1523/JNEUROSCI.3635-09.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Battaglia FP, Sutherland GR, McNaughton BL. Hippocampal sharp wave bursts coincide with neocortical up-state transitions. Learn. Mem. 2004;11:697–704. doi: 10.1101/lm.73504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Behrens CJ, van den Boom LP, de Hoz L, Friedman A, Heinemann U. Induction of sharp wave-ripple complexes in vitro and reorganization of hippocampal networks. Nat. Neurosci. 2005;8:1560–1567. doi: 10.1038/nn1571. [DOI] [PubMed] [Google Scholar]
- Belluscio MA, Mizuseki K, Schmidt R, Kempter R, Buzsáki G. Cross-frequency phase-phase coupling between θ and γ oscillations in the hippocampus. J. Neurosci. 2012;32(2):423–435. doi: 10.1523/JNEUROSCI.4122-11.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Both M, Bähner F, von Bohlen und Halbach O, Draguhn A. Propagation of specific network patterns through the mouse hippocampus. Hippocampus. 2008;18:899–908. doi: 10.1002/hipo.20446. [DOI] [PubMed] [Google Scholar]
- Bragin A, Engel J, Jr, Wilson CL, Fried I, Mathern GW. Hippocampal and entorhinal cortex high-frequency oscillations (100–500 Hz) in human epileptic brain and in kainic acid - treated rats with chronic seizures. Epilepsia. 1999a;40:127–137. doi: 10.1111/j.1528-1157.1999.tb02065.x. [DOI] [PubMed] [Google Scholar]
- Bragin A, Engel J, Jr, Wilson CL, Vizentin E, Mathern GW. Electrophysiologic analysis of a chronic seizure model after unilateral hippocampal KA injection. Epilepsia. 1999b;40:1210–1221. doi: 10.1111/j.1528-1157.1999.tb00849.x. [DOI] [PubMed] [Google Scholar]
- Bragin A, Engel J, Wilson CL, Fried I, Buzsáki G. High-frequency oscillations in human brain. Hippocampus. 1999c;9:137–142. doi: 10.1002/(SICI)1098-1063(1999)9:2<137::AID-HIPO5>3.0.CO;2-0. [DOI] [PubMed] [Google Scholar]
- Bragin A, Mody I, Wilson CL, Engel J. Local generation of fast ripples in epileptic brain. J. Neurosci. 2002;22:2012–2021. doi: 10.1523/JNEUROSCI.22-05-02012.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bragin A, Wilson CL, Engel J., Jr Chronic epileptogenesis requires development of a network of pathologically interconnected neuron clusters: a hypothesis. Epilepsia. 2000;41(Suppl. 6):S144–S152. doi: 10.1111/j.1528-1157.2000.tb01573.x. [DOI] [PubMed] [Google Scholar]
- Bressler SL, Freeman WJ. Frequency analysis of olfactory system EEG in cat, rabbit and rat. Electroencephalogr. Clin. Neurophysiol. 1980;50:19–24. doi: 10.1016/0013-4694(80)90319-3. [DOI] [PubMed] [Google Scholar]
- Brunel N, Wang X-J. What determines the frequency of fast network oscillations with irregular neural discharges? I. Synaptic dynamics and excitation-inhibition balance. J. Neurophysiol. 2003;90:415–430. doi: 10.1152/jn.01095.2002. [DOI] [PubMed] [Google Scholar]
- Buhl DL, Buzsáki G. Developmental emergence of hippocampal fast-field ripple oscillations in the behaving rat pups. Neuroscience. 2005;134:1423–1430. doi: 10.1016/j.neuroscience.2005.05.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buzsáki G. Hippocampal sharp waves: their origin and significance. Brain Res. 1986;398:242–252. doi: 10.1016/0006-8993(86)91483-6. [DOI] [PubMed] [Google Scholar]
- Buzsáki G. Two-stage model of memory trace formation. A role for “Noisy” brain states. Neuroscience. 1989;31:551–570. doi: 10.1016/0306-4522(89)90423-5. [DOI] [PubMed] [Google Scholar]
- Buzsáki G. Memory consolidation during sleep: a neurophysiological perspective. J. Sleep Res. 1998;7:17–23. doi: 10.1046/j.1365-2869.7.s1.3.x. [DOI] [PubMed] [Google Scholar]
- Buzsáki G. Rhythms of the Brain. Oxford University Press; 2006. [Google Scholar]
- Buzsáki G, Chrobak JJ. Synaptic plasticity and self-organization in the hippocampus. Nat. Neurosci. 2005;8:1418–1420. doi: 10.1038/nn1105-1418. [DOI] [PubMed] [Google Scholar]
- Buzsáki G, Wang X-J. Large-scale recording of neurons by movable silicon probes in behaving rodents. Ann Rev Neurosci. 2012 doi: 10.3791/3568. in press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buzsáki G, Horvath Z, Urioste R, Hetke J, Wise K. High-frequency network oscillation in the hippocampus. science. 1992;256:1025–1027. doi: 10.1126/science.1589772. [DOI] [PubMed] [Google Scholar]
- Buzsáki G, Lai-Wo SL, Vanderwolf CH. Cellular bases of hippocampal EEG in the behaving rat. Brain Res. Rev. 1983;6:139–171. doi: 10.1016/0165-0173(83)90037-1. [DOI] [PubMed] [Google Scholar]
- Buzsáki G, Buhl DL, Harris KD, Csicsvari J, Cze´h B, Morozov A. Hippocampal network patterns of activity in the mouse. Neuroscience. 2003;116:201–211. doi: 10.1016/s0306-4522(02)00669-3. [DOI] [PubMed] [Google Scholar]
- Canolty R, Edwards E, Dalal S, Soltani M, Nagarajan S, et al. High gamma power is phase-locked to theta oscillations in human neocortex. science. 2006;313:1626–1628. doi: 10.1126/science.1128115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carr MF, Jadhav SP, Frank LM. Hippocampal replay in the awake state: a potential substrate for memory consolidation and retrieval. Nat. Neurosci. 2011;14:147–153. doi: 10.1038/nn.2732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chrobak J, Buzsáki G. Selective activation of deep layer (V-VI) retrohippocampal cortical neurons during hippocampal sharp wavesin the behaving rat. J. Neurosci. 1994;14:6160–6170. doi: 10.1523/JNEUROSCI.14-10-06160.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chrobak JJ, Buzsáki G. High-frequency oscillations in the output networks of the hippocampal-entorhinal axis of the freely behaving rat. J. Neurosci. 1996;16:3056–3066. doi: 10.1523/JNEUROSCI.16-09-03056.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Colgin LL, Denninger T, Fyhn M, Hafting T, Bonnevie T, Jensen O, Moser MB, Moser EI. Frequency of gamma oscillations routes flow of information in the hippocampus. Nature. 2009;462:353–357. doi: 10.1038/nature08573. [DOI] [PubMed] [Google Scholar]
- Csicsvari J, Hirase H, Czurko A, Buzsáki G. Reliability and state dependence of pyramidal cell-interneuron synapses in the hippocampus: an ensemble approach in the behaving rat. Neuron. 1998;21:179–189. doi: 10.1016/s0896-6273(00)80525-5. [DOI] [PubMed] [Google Scholar]
- Csicsvari J, Hirase H, Czurkó A, Mamiya A, Buzsáki G. Oscillatory coupling of hippocampal pyramidal cells and interneurons in the behaving rat. J. Neurosci. 1999a;19:274–287. doi: 10.1523/JNEUROSCI.19-01-00274.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Csicsvari J, Hirase H, Czurkó A, Mamiya A, Buzsáki G. Fast network oscillations in the hippocampal CA1 region of the behaving rat. J. Neurosci. 1999b;19:RC20. doi: 10.1523/JNEUROSCI.19-16-j0001.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Csicsvari J, Hirase H, Mamiya A, Buzsáki G. Ensemble patterns of hippocampal CA3-CA1 neurons during sharp wave - associated population events. Neuron. 2000;28:585–594. doi: 10.1016/s0896-6273(00)00135-5. [DOI] [PubMed] [Google Scholar]
- Csicsvari J, Jamieson B, Wise KD, Buzsáki G. Mechanisms of gamma oscillations in the hippocampus of the behaving rat. Neuron. 2003;37:311–322. doi: 10.1016/s0896-6273(02)01169-8. [DOI] [PubMed] [Google Scholar]
- Curio G. High frequency (600 Hz) bursts of spike-like activities generated in the human cerebral somatosensory system. Electroencephalogr. Clin. Neurophysiol. Suppl. 1999;49:56–61. [PubMed] [Google Scholar]
- Davidson TJ, Kloosterman F, Wilson MA. Hippocampal replay of extended experience. Neuron. 2009;63:497–507. doi: 10.1016/j.neuron.2009.07.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de la Prida LM, Gal B. Synaptic contributions to focal and widespread spatiotemporal dynamics in the isolated rat subiculum in vitro. J. Neurosci. 2004;24:5525–5536. doi: 10.1523/JNEUROSCI.0309-04.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Diba K, Buzsáki G. Forward and reverse hippocampal place-cell sequences during ripples. Nat. Neurosci. 2007;10:1241. doi: 10.1038/nn1961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Derdikman D, Moser MB. A dual role for hippocampal replay. Neuron. 2010;65:582–584. doi: 10.1016/j.neuron.2010.02.022. [DOI] [PubMed] [Google Scholar]
- Dragoi G, Tonegawa S. Preplay of future place cell sequences by hippocampal cellular assemblies. Nature. 2011 Jan;469(7330):397–401. doi: 10.1038/nature09633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Draguhn A, Traub R, Schmitz D, Jefferys J. Electrical coupling underlies high-frequency oscillations in the hippocampus in vitro. Nature. 1998;394:189–192. doi: 10.1038/28184. [DOI] [PubMed] [Google Scholar]
- Dupret D, O’Neill J, Pleydell-Bouverie B, Csicsvari J. The reorganization and reactivation of hippocampal maps predict spatial memory performance. Nat. Neurosci. 2010;13:995–1002. doi: 10.1038/nn.2599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dzhala VI, Staley KJ. Mechanisms of fast ripples in the hippocampus. J. Neurosci. 2004;24:8896–8906. doi: 10.1523/JNEUROSCI.3112-04.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ego-Stengel V, Wilson MA. Disruption of ripple-associated hippocampal activity during rest impairs spatial learning in the rat. Hippocampus. 2010;20:1–10. doi: 10.1002/hipo.20707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Engel J, Jr, Bragin A, Staba R, Mody I. High-frequency oscillations: what is normal and what is not? Epilepsia. 2009;50:598–604. doi: 10.1111/j.1528-1167.2008.01917.x. [DOI] [PubMed] [Google Scholar]
- Fell J, Fernández G, Lutz MT, Kockelmann E, Burr W, Schaller C, Elger CE, Helmstaedter C. Rhinal-hippocampal connectivity determines memory formation during sleep. Brain. 2006;129(Pt 1):108–114. doi: 10.1093/brain/awh647. [DOI] [PubMed] [Google Scholar]
- Fries P. Neuronal gamma-band synchronization as a fundamental process in cortical computation. Annu. Rev. Neurosci. 2009;32:209–224. doi: 10.1146/annurev.neuro.051508.135603. [DOI] [PubMed] [Google Scholar]
- Foffani G, Uzcategui YG, Gal B, de la Prida LM. Reduced spike-timing reliability correlates with the emergence of fast ripples in the rat epileptic hippocampus. Neuron. 2007;55:930–941. doi: 10.1016/j.neuron.2007.07.040. [DOI] [PubMed] [Google Scholar]
- Foster DJ, Wilson MA. Reverse replay of behavioural sequences in hippocampal place cells during the awake state. Nature. 2006;440:680–683. doi: 10.1038/nature04587. [DOI] [PubMed] [Google Scholar]
- Gaona CM, Sharma M, Freudenburg ZV, Breshears JD, Bundy DT, Roland J, Barbour DL, Schalk G, Leuthardt EC. Nonuniform high-gamma (60–500 Hz) power changes dissociate cognitive task and anatomy in human cortex. J. Neurosci. 2011;31(6):2091–2100. doi: 10.1523/JNEUROSCI.4722-10.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Girardeau G, Benchenane K, Wiener SI, Buzsáki G, Zugaro MB. Selective suppression of hippocampal ripples impairs spatial memory. Nat. Neurosci. 2009;12:1222–1223. doi: 10.1038/nn.2384. [DOI] [PubMed] [Google Scholar]
- Grenier F, Timofeev I, Steriade M. Focal synchronization of ripples (80–200 Hz) in neocortex and their neuronal correlates. J. Neurophysiol. 2001;86:1884–1898. doi: 10.1152/jn.2001.86.4.1884. [DOI] [PubMed] [Google Scholar]
- Gupta AS, van der Meer MA, Touretzky DS, Redish AD. Hippocampal replay is not a simple function of experience. Neuron. 2010 Mar;65(5):695–705. doi: 10.1016/j.neuron.2010.01.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Isomura Y, Sirota A, Ozen S, Montgomery S, Mizuseki K, Henze DA, Buzsaki G. Integration and segregation of activity in entorhinal-hippocampal subregions by neocortical slow oscillations. Neuron. 2006;52:871–882. doi: 10.1016/j.neuron.2006.10.023. [DOI] [PubMed] [Google Scholar]
- Jensen O, Kaiser J, Lachaux JP. Human gamma-frequency oscillations associated with attention and memory. Trends Neurosci. 2007;30 (i7):317–324. doi: 10.1016/j.tins.2007.05.001. [DOI] [PubMed] [Google Scholar]
- Kandel A, Buzsáki G. Cellular-synaptic generation of sleep spindles, spike-and-wave discharges, and evoked thalamocortical responses in the neocortex of the rat. J. Neurosci. 1997;17:6783. doi: 10.1523/JNEUROSCI.17-17-06783.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karlsson MP, Frank LM. Network dynamics underlying the formation of sparse, informative representations in the hippocampus. J. Neurosci. 2008;28:14271–14281. doi: 10.1523/JNEUROSCI.4261-08.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karlsson MP, Frank LM. Awake replay of remote experiences in the hippocampus. Nat. Neurosci. 2009;12:913–918. doi: 10.1038/nn.2344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klausberger T, Magill PJ, Márton LF, Roberts JDB, Cobden PM, Buzsaki G, Somogyi P. Brain-state- and cell-type-specific firing of hippocampal interneurons in vivo. Nature. 2003;421:844–848. doi: 10.1038/nature01374. [DOI] [PubMed] [Google Scholar]
- Klausberger T, Márton LF, Baude A, Roberts JDB, Magill PJ, Somogyi P. Spike timing of dendrite-targeting bistratified cells during hippocampal network oscillations in vivo. Nat. Neurosci. 2004;7:41–47. doi: 10.1038/nn1159. [DOI] [PubMed] [Google Scholar]
- Klausberger T, Somogyi P. Neuronal diversity and temporal dynamics: the unity of hippocampal circuit operations. science. 2008;321:53–57. doi: 10.1126/science.1149381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kubota D, Colgin LL, Casale M, Brucher FA, Lynch G. Endogenous waves in hippocampal slices. J. Neurophysiol. 2003;89:81–89. doi: 10.1152/jn.00542.2002. [DOI] [PubMed] [Google Scholar]
- Kudrimoti HS, Barnes CA, McNaughton BL. Reactivation of hippocampal cell assemblies: effects of behavioral state, experience, and EEG dynamics. J. Neurosci. 1999;19:4090. doi: 10.1523/JNEUROSCI.19-10-04090.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Langston RF, Ainge JA, Couey JJ, Canto CB, Bjerknes TL, Witter MP, Moser EI, Moser MB. Development of the spatial representation system in the rat. science. 2010;328:1576–1580. doi: 10.1126/science.1188210. [DOI] [PubMed] [Google Scholar]
- Lee AK, Wilson MA. Memory of sequential experience in the hippocampus during slow wave sleep. Neuron. 2002;36:1183–1194. doi: 10.1016/s0896-6273(02)01096-6. [DOI] [PubMed] [Google Scholar]
- Leinekugel X, Khazipov R, Cannon R, Hirase H, Ben-Ari Y, Buzsaki G. Correlated bursts of activity in the neonatal hippocampus in vivo. science. 2002;296:2049–2052. doi: 10.1126/science.1071111. [DOI] [PubMed] [Google Scholar]
- Liotta A, çalış¸kan G, ul Haq R, Hollnagel JO, Rösler A, Heinemann U, Behrens CJ. Partial disinhibition is required for transition of stimulus-induced sharp wave-ripple complexes into recurrent epileptiform discharges in rat hippocampal slices. J. Neurophysiol. 2011;105:172–187. doi: 10.1152/jn.00186.2010. [DOI] [PubMed] [Google Scholar]
- Lisman JE, Idiart MA. Storage of 7±2 short-term memories in oscillatory subcycles. science. 1995;267:1512–1515. doi: 10.1126/science.7878473. [DOI] [PubMed] [Google Scholar]
- Lopes da Silva FH. Computer-Assisted EEG Diagnosis: Pattern Recognition and Brain Mapping. In: Schomer D, da Lopes Silva F, editors. Niedermeyer’s Electroencephalography. Basic Principles, clinical applications, and related fields. 6th. Lippincott Williams & Wilkins; 2011. p. 1256. [Google Scholar]
- Maier N, Nimmrich V, Draguhn A. Cellular and network mechanisms underlying spontaneous sharp wave-ripple complexes in mouse hippocampal slices. J. Physiol. (Lond.) 2003;550:873–887. doi: 10.1113/jphysiol.2003.044602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McClelland JL, McNaughton BL, O’Reilly RC. Why there are complementary learning systems in the hippocampus and neocortex: insights from the successes and failures of connectionist models of learning and memory. Psychol. Rev. 1995;102:419–457. doi: 10.1037/0033-295X.102.3.419. [DOI] [PubMed] [Google Scholar]
- Mehta M. Cortico-hippocampal interaction during up-down states and memory consolidation. Nat. Neurosci. 2007;10:13–15. doi: 10.1038/nn0107-13. [DOI] [PubMed] [Google Scholar]
- Mizuseki K, Sirota A, Pastalkova E, Buzsáki G. Theta oscillations provide temporal windows for local circuit computation in the entorhinal-hippocampal loop. Neuron. 2009;64:267–280. doi: 10.1016/j.neuron.2009.08.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mölle M, Yeshenko O, Marshall L, Sara SJ, Born J. Hippocampal sharp wave-ripples linked to slow oscillations in rat slow-wave sleep. J. Neurophysiol. 2006;96:62–70. doi: 10.1152/jn.00014.2006. [DOI] [PubMed] [Google Scholar]
- Muller RU, Stead M, Pach J. The hippocampus as a cognitive graph. J. Gen. Physiol. 1996;107:663–694. doi: 10.1085/jgp.107.6.663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murthy VN, Fetz EE. Coherent 25- to 35-Hz oscillations in the sensori-motor cortex of awake behaving monkeys. Proc. Natl. Acad. Sci. U.S.A. 1992;89(12):5670–5674. doi: 10.1073/pnas.89.12.5670. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nádasdy Z, Hirase H, Czurkó A, Csicsvari J, Buzsáki G. Replay and time compression of recurring spike sequences in the hippocampus. J. Neurosci. 1999;19:9497–9507. doi: 10.1523/JNEUROSCI.19-21-09497.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakashiba T, Buhl DL, McHugh TJ, Tonegawa S. Hippocampal CA3 output is crucial for ripple-associated reactivation and consolidation of memory. Neuron. 2009;62:781–787. doi: 10.1016/j.neuron.2009.05.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O’Keefe J, Nadel L. The Hippocampus as a Cognitive Map. USA: Oxford University Press; 1978. [Google Scholar]
- O’Neill J, Senior T, Csicsvari J. Place-selective firing of CA1 pyramidal cells during sharp wave/ripple network patterns in exploratory behavior. Neuron. 2006;49:143–155. doi: 10.1016/j.neuron.2005.10.037. [DOI] [PubMed] [Google Scholar]
- O’Neill J, Senior TJ, Allen K, Huxter JR, Csicsvari J. Reactivation of experience-dependent cell assembly patterns in the hippocampus. Nat. Neurosci. 2008;11:209–215. doi: 10.1038/nn2037. [DOI] [PubMed] [Google Scholar]
- Papatheodoropoulos C, Kostopoulos G. Spontaneous GABA(A)-dependent synchronous periodic activity in adult rat ventral hippocampal slices. Neurosci. Lett. 2002;319:17–20. doi: 10.1016/s0304-3940(01)02505-8. [DOI] [PubMed] [Google Scholar]
- Pastalkova E, Itskov V, Amarasingham A, Buzsáki G. Internally generated cell assembly sequences in the rat hippocampus. science. 2008;321:1322–1327. doi: 10.1126/science.1159775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peyrache A, Khamassi M, Benchenane K, Wiener SI, Battaglia FP. Replay of rule-learning related neural patterns in the prefrontal cortex during sleep. Nat. Neurosci. 2009;12:919–926. doi: 10.1038/nn.2337. [DOI] [PubMed] [Google Scholar]
- Ponomarenko AA, Li J-S, Korotkova TM, Huston JP, Haas HL. Frequency of network synchronization in the hippocampus marks learning. Eur. J. Neurosci. 2008;27:3035–3042. doi: 10.1111/j.1460-9568.2008.06232.x. [DOI] [PubMed] [Google Scholar]
- Ray S, Maunsell JH. Different origins of gamma rhythm and high-gamma activity in macaque visual cortex. PLoS Biol. 2011;9:e1000610. doi: 10.1371/journal.pbio.1000610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reichinnek S, Künsting T, Draguhn A, Both M. Field potential signature of distinct multicellular activity patterns in the mouse hippocampus. J. Neurosci. 2010;30:15441–15449. doi: 10.1523/JNEUROSCI.2535-10.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roelfsema PR, Engel AK, König P, Singer W. Visuomotor integration is associated with zero time-lag synchronization among cortical areas. Nature. 1997;385(6612):157–161. doi: 10.1038/385157a0. [DOI] [PubMed] [Google Scholar]
- Royer S, Sirota A, Patel J, Buzsáki G. Distinct representations and theta dynamics in dorsal and ventral hippocampus. J. Neurosci. 2010;30:1777. doi: 10.1523/JNEUROSCI.4681-09.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schmitz D, Schuchmann S, Fisahn A, Draguhn A, Buhl EH, Petrasch-Parwez E, Dermietzel R, Heinemann U, Traub RD. Axo-axonal coupling: a novel mechanism for ultrafast neuronal communication. Neuron. 2001;31:831–840. doi: 10.1016/s0896-6273(01)00410-x. [DOI] [PubMed] [Google Scholar]
- Sederberg PB, Schulze-Bonhage A, Madsen JR, Bromfield EB, McCarthy DC, Brandt A, Tully MS, Kahana MJ. Hippocampal and neocortical gamma oscillations predict memory formation in humans. Cereb. Cortex. 2007;17(5):1190–1196. doi: 10.1093/cercor/bhl030. [DOI] [PubMed] [Google Scholar]
- Singer W, Gray C. Visual feature integration and the temporal correlation hypothesis. Annu. Rev. Neurosci. 1995;18:555–586. doi: 10.1146/annurev.ne.18.030195.003011. [DOI] [PubMed] [Google Scholar]
- Singer AC, Frank LM. Rewarded outcomes enhance reactivation of experience in the hippocampus. Neuron. 2009;64:910–921. doi: 10.1016/j.neuron.2009.11.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sirota A, Csicsvari J, Buhl D, Buzsáki G. Communication between neocortex and hippocampus during sleep in rodents. Proc. Natl. Acad. Sci U.S.A. 2003;100:2065. doi: 10.1073/pnas.0437938100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Skaggs WE, McNaughton BL. Replay of neuronal firing sequences in rat hippocampus during sleep following spatial experience. science. 1996;271:1870–1873. doi: 10.1126/science.271.5257.1870. [DOI] [PubMed] [Google Scholar]
- Siapas AG, Wilson MA. Coordinated interactions between hippocampal ripples and cortical spindles during slow-wave sleep. Neuron. 1998;21(5):1123–1128. doi: 10.1016/s0896-6273(00)80629-7. [DOI] [PubMed] [Google Scholar]
- Staba RJ, Ard TD, Benison AM, Barth DS. Intracortical pathways mediate nonlinear fast oscillation (>200 Hz) interactions within rat barrel cortex. J. Neurophysiol. 2005;93:2934–2939. doi: 10.1152/jn.01101.2004. [DOI] [PubMed] [Google Scholar]
- Steriade M, McCormick DA, Sejnowski TJ. Thalamocortical oscillations in the sleeping and aroused brain. science. 1993;262:679. doi: 10.1126/science.8235588. [DOI] [PubMed] [Google Scholar]
- Sullivan D, Csicsvari J, Mizuseki K, Montgomery S, Diba K, Buzsáki G. Relationships between hippocampal sharp waves ripples, and fast gamma oscillation: influence of dentate and entorhinal cortical activity. J. Neurosci. 2011;31:8605–8616. doi: 10.1523/JNEUROSCI.0294-11.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tallon-Baudry C, Bertrand O, Peronnet F, Pernier J. Induced gamma-band activity during the delay of a visual short-term memory task in humans. J. Neurosci. 1998;18(11):4244–4254. doi: 10.1523/JNEUROSCI.18-11-04244.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tort AB, Komorowski R, Eichenbaum H, Kopell N. Measuring phase-amplitude coupling between neuronal oscillations of different frequencies. J. Neurophysiol. 2010;104(2):1195–1210. doi: 10.1152/jn.00106.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Traub RD, Bibbig A. A model of high-frequency ripples in the hippocampus based on synaptic coupling plus axon-axon gap junctions between pyramidal neurons. J. Neurosci. 2000;20:2086–2093. doi: 10.1523/JNEUROSCI.20-06-02086.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Le Van Quyen M, Staba R, Bragin A, Dickson C, Valderrama M, Fried I, Engel J. Large-scale microelectrode recordings of high-frequency gamma oscillations in human cortex during sleep. J. Neurosci. 2010;30:7770–7782. doi: 10.1523/JNEUROSCI.5049-09.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wierzynski CM, Lubenov EV, Gu M, Siapas AG. State-dependent spike-timing relationships between hippocampal and prefrontal circuits during sleep. Neuron. 2009;61:587–596. doi: 10.1016/j.neuron.2009.01.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilson M, McNaughton B. Reactivation of hippocampal ensemble memories during sleep. science. 1994;265:676–679. doi: 10.1126/science.8036517. [DOI] [PubMed] [Google Scholar]
- Wills TJ, Cacucci F, Burgess N, O’Keefe J. Development of the hippocampal cognitive map in preweanling rats. science. 2010 Jun 18;328(5985):1573–1576. doi: 10.1126/science.1188224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wittner L, Henze DA, Záborszky L, Buzsáki G. Three-dimensional reconstruction of the axon arbor of a CA3 pyramidal cell recorded and filled in vivo. Brain Struct. Funct. 2007;212:75–83. doi: 10.1007/s00429-007-0148-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ylinen A, Bragin A, Nádasdy Z, Jandó G, Szabó I, Sik A, Buzsáki G. Sharp wave-associated high-frequency oscillation (200 Hz) in the intact hippocampus: network and intracellular mechanisms. J. Neurosci. 1995;15:30–46. doi: 10.1523/JNEUROSCI.15-01-00030.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]