Abstract
High-grade serous cancer (HGSC) progresses to advanced stages without symptoms and the 5-year survival rate is a dismal 30%. Recent studies of ovaries and Fallopian tubes in patients with BRCA1 or BRCA2 mutations have documented a pre-metastatic intramucosal neoplasm that is found almost exclusively in the Fallopian tube, termed ‘serous tubal intraepithelial carcinoma’ or STIC. Moreover, other proliferations, termed p53 signatures, secretory cell outgrowths (SCOUTs), and lower-grade serous tubal intraepithelial neoplasms (STINs) fall short of STIC but share similar alterations in expression, in keeping with an underpinning of genomic disturbances involved in, or occurring in parallel with, serous carcinogenesis. To gain insight into the cellular origins of this unique tubal pathway to high-grade serous cancer, we cloned and both immortalized and transformed Fallopian tube stem cells (FTSCs). We demonstrated that pedigrees of FTSCs were capable of multipotent differentiation and that the tumours derived from transformed FTSCs shared the histological and molecular features of HGSC. We also demonstrated that altered expression of some biomarkers seen in transformed FTSCs and HGSCs (stathmin, EZH2, CXCR4, CXCL12, and FOXM1) could be seen as well in immortalized cells and their in vivo counterparts SCOUTs and STINs. Thus, a whole-genome transcriptome analysis comparing FTSCs, immortalized FTSCs, and transformed FTSCs showed a clear molecular progression sequence that is recapitulated by the spectrum of accumulated perturbations characterizing the range of proliferations seen in vivo. Biomarkers unique to STIC relative to normal tubal epithelium provide a basis for novel detection approaches to early HGSC, but must be viewed critically given their potential expression in lesser proliferations. Perturbations shared by both immortalized and transformed FTSCs may provide unique early targets for prevention strategies. Central to these efforts has been the ability to clone and perpetuate multipotent FTSCs.
Keywords: Fallopian tubes, ovary, cell culture, neoplasia
Introduction
Epithelial ovarian cancer (EOC) is the fifth most common cause of death from cancer in women, and the most common type – high-grade serous carcinoma or HGSC – is the most lethal. One in 200 women will develop ovarian cancer between their 50th and 70th birthdays. Worldwide, there are 225 000 new cases of ovarian cancer diagnosed annually and an estimated 140 163 disease-related deaths [1]. Up to 80% of women present with stages III/IV disease, and the 5-year survival rate is a dismal 30%. Mortality for this disease has not markedly changed since the 1930s [2], because ovarian cancer cannot be detected at low stage by current screening programmes. Resolving this dilemma will require effective tools and methods to interrupt the carcinogenic sequence at a point that permits either cure or prevention of tumour-specific mortality.
There is cogent clinical and molecular evidence to suggest that many, if not all, cases of HGSC arise from the Fallopian tube. From the clinical end, it has been known that reducing the risk of BRCA1 and BRCA2 patients required removal of the Fallopian tube in addition to the ovary [3]. Molecular analyses have shown that HGSC has gene expression profiles more akin to those of Fallopian tube epithelium than to ovarian surface epithelium [4]. Finally, and most significantly, the pathological examination of risk reduction salpingo-oophorectomies for germ-line BRCA1 and BRCA2 mutations has uncovered pre-metastatic stages of HGSC (serous tubal intraepithelial carcinoma or STIC) as well as premalignant tubal intraepithelial neoplasia (or serous tubal intraepithelial lesions) [5,6]. In the Fallopian tube model, STIC is considered the earliest morphological manifestation of serous carcinoma. STICs are composed of ‘secretory cells’, the non-ciliated population of the endosalpinx. These cells, when neoplastic, exhibit features including variable stratification, increased proliferation, and loss of nuclear polarity [7].
Most STICs are marked by mutant p53, as are their metastatic form, high-grade serous cancers. Further analyses of BRCA1/BRCA2 mutation-associated Fallopian tubes have revealed the presence as well of a ‘latent precancer’ – the ‘p53 signature’, which has mutant p53 overexpression but retains cell polarity and lacks excessive cell proliferation. Interestingly, p53 signatures have been found adjacent to STICs and in several revealing examples have been shown to share the same p53 mutation as HGSC, suggesting a lineage relationship [8]. These compelling results demonstrate that the Fallopian tube is a site of origin of HGSC, the development of which follows the classic multi-step carcinogenesis model. Importantly, latent precancers are common in the tubes of women who are not at genetic risk, and between 40% and 60% of the serous cancers in BRCA mutation-negative women also co-exist with STIC [7,8] with a genetic link between the two [9,10]. Thus, STIC represents the earliest phase of most pelvic serous cancers and targeted treatment or prevention of STIC is a valid goal in cancer prevention. In parallel with the serous carcinogenic sequence is one characterized by putative stem cell outgrowths, termed SCOUTs. These proliferations lack p53 mutations but share many attributes with intraepithelial neoplasms, one being altered expression levels of genes including ALDH1, PAX2, EZH2, LEF1, and others. The impression from these collective entities is that the tube is prone to both self-limited and potentially malignant intraepithelial proliferations.
HGSCs presumably arise from non-ciliated cells of the Fallopian tube (secretory cells) but the precise relationship between these cells and stem cells in the tube is not understood, in part because of a lack of detailed in vitro studies of putative stem cells. Herein, we report a Fallopian tube stem cell model based on a cell culture paradigm of both limited (immortalization) and aggressive (transformation) cell outgrowth. This model is superimposed on a similar in vivo paradigm of proliferative lesions seen in the Fallopian tube. The goal of this exercise was to discern not only molecular perturbations marking the transition from STIC to metastatic disease but also those that highlight the loss of growth control in the early phases of neoplasia.
Materials and methods
Case material
This study was approved by the Brigham and Women’s Human Investigation Committee and involved the use of discarded fresh and archived tissues. Case material for gene expression analysis and histology consisted of the following epithelia/lesions: (1) normal oviduct and HGSC paired samples (n = 10) and (2) normal oviduct, STIC, and invasive HGSC lesions from each patient section (n = 6). Cases for immunohistochemistry were selected by one of us (CPC) using criteria that have been previously described [10].
Stem cell culture and differentiation
Fimbriae of Fallopian tube tissue were obtained from discarded surgical specimens of women undergoing benign procedures. Discarded fetal Fallopian tube tissues were obtained under an approved IRB protocol. Tissues were digested in 2 mg/ml collagenase A (Roche, Indianapolis, IN, USA) at 37 °C for 1.5 h. Disaggregated cells were cultured on a feeder layer of lethally irradiated 3 T3-J2 cells in stem cell culturing media (SCM-6 F8) [11]. Clonal analysis and in vitro ALI differentiation were based on previously described methods for lung epithelial stem cells [12].
Xenografts of transformed FTSCs
Fallopian tube stem cells were infected with retroviruses expressing c-Myc, hTERT, and SV40 large T antigen (SV40 large T and hTERT for immortalization; and SV40 large T, hTERT, and c-Myc for transformation). The PMN-MYC-IRES-GFP retroviral vector expressing full-length human c-MYC was a gift from Yu Qiang [13]. PBABE-puro SV40 LT was a gift from Thomas Roberts (Addgene plasmid #13970; Addgene, Cambridge, MA, USA) [14] and PBABE-puro-hTERT was a gift from Bob Weinberg (Addgene plasmid #1771) [15]. Recombinant retroviral particles were prepared by transient transfection of GP2-293 T cells (ATCC) along with packaging plasmids (pCMV-VSVG). The medium containing recombinant retrovirus was harvested 48 h after transfection. Cellular debris was removed by centrifugation and filtration through a 0.45 μm filter (Millipore, Billerica, MA, USA). For infection, in brief, 200 000 stem cells were plated onto a lawn of feeder cells in 3 cm culture dishes and transduced 3 days later. After 48 h, cells were split 1:5 onto new lawns and grown and passaged for 4 weeks before plating onto culture plates without feeder cells for an additional 4 weeks. Individual colonies were selected and tested for growth in soft agar, and positive colonies selected for expansion and transplantation. Two thousand transformed cells (expressing SV40 large T antigen, hTERT, and c-myc) were injected subcutaneously into 6-week-old female NSG (NOD.Cg-Prkdcscid Il2rgtm1Wjl/SzJ) mice following the protocols approved by BRC IACUC #110643 at the Agency for Science Technology and Research (A*STAR) Singapore. Visible tumours appeared typically at 2 weeks and were harvested following euthanasia and analysed by histology and expression microarray.
Histology and immunofluorescence
Histology, immunohistochemistry (IHC), and immunofluorescence (IF) were performed following standard laboratory protocols. All the primary antibodies used in this study and staining conditions are summarized in Supplementary Table 1. For IHC, detection was completed with the Vectastain ABC kit (Catalog No PK-6102; Vector Laboratories, Inc, Burlingame, CA, USA) with a liquid DAB-plus substrate kit (Catalog No 00-2020). Slides were counterstained with Hematoxylin Stain 3 (Catalog No CS402-1D). Stained slides were stored at 4 °C in the dark and all images for section slides were captured by using an Inverted Eclipse Ti-Series microscope (Nikon, Japan) with Lumencor SOLA light engine and Andor Technology Clara Interline CCD camera and NIS-Elements Advanced Research v.4.13 software (Nikon, Japan) or an LSM 780 confocal microscope (Carl Zeiss, Germany) with LSM software. Bright field cell culture images were obtained on an Eclipse TS100 microscope (Nikon, Japan) with a Digital Sight DSFi1 camera (Nikon, Japan) and NIS-Elements F3.0 software (Nikon, Japan).
Laser captured microdissection
Fresh surgical specimens from ten independent women for normal oviduct and HGSC and six independent women for normal, STIC, and invasive STIC were embedded in OCT, sectioned on a cryostat, and stained with haematoxylin to morphologically identify each region. Twelve serial frozen sections of each tissue sample were microdissected using a PALM microbeam instrument (Carl Zeiss, Germany) and each selected cell population from different slides of the same patient was pooled. Total RNAs were extracted using the Pico Pure RNA extraction kit (Life Technologies, Grand Island, NY, USA).
Microarray
For normal Fallopian tube epithelium and paired HGSC samples from ten patients and FTSC (stem cells, immortalized, transformed, and xenograft) samples, total RNA processing and hybridization were performed on Affymetrix human U133 plus 2.0 Array chips (Affymetrix, CA, USA). For normal Fallopian tube epithelium, STIC, and invasive STIC from six patients, total RNAs were amplified using the WT Pico RNA Amplification System V2 and Encore Biotin Module (NuGEN Technologies, San Carlos, CA, USA). Amplified DNA samples were prepared according to the manufacturer’s instructions and hybridized onto a GeneChip Human Exon 1.0 ST Array (Affymetrix). GeneChip operating software was used to process all the Cel files and calculate probe intensity values. To validate sample quality, a quality check was conducted using Affymetrix Expression Console software. The intensity values were log2-transformed and imported into the Partek Genomics Suite 6.6 (Partek Inc, Chesterfield, MO, USA). For the GeneChip Human Exon 1.0 ST Array, exons were summarized to genes and a one-way ANOVA was performed to identify differentially expressed genes. p values and fold-change numbers were calculated for each analysis.
Bioinformatics for gene expression
Unsupervised clustering and heatmap generation were performed with sorted datasets by Euclidean distance on average linkage clustering with selected probe sets by Partek Genomics Suite 6.6. Gene set enrichment analysis (GSEA) [16] was performed to compare (1) STIC, invasive cancer, and normal; and (2) FTSCs and immortalized and transformed FTSCs (FTSCi and FTSCt). DAVID bioinformatics resources (http://david.abcc.ncifcrf.gov/) were used to find enriched pathways [17]. In Figure 1g, 1.5-fold progressively up-regulated genes are selected from FTSCs to FTSTi to FTSCt in order to create a heatmap. In Figures 2d, 4a and 4c, 2-fold or more and p < 0.05 differentially expressed genes are chosen as significantly changed genes for further data analysis. Datasets generated for this study have been submitted to the National Center for Biotechnology Information Gene Expression Omnibus (GEO) database under GSE69428 for normal oviduct and HGSC from ten patients; GSE69453 for FTSC-ALI and transformed FTSCs (stem cell, immortalized, transformed, and xenografts); and GSE69429 for normal Fallopian tubal epithelium, STIC, and invasive STIC from six patients.
Figure 1.
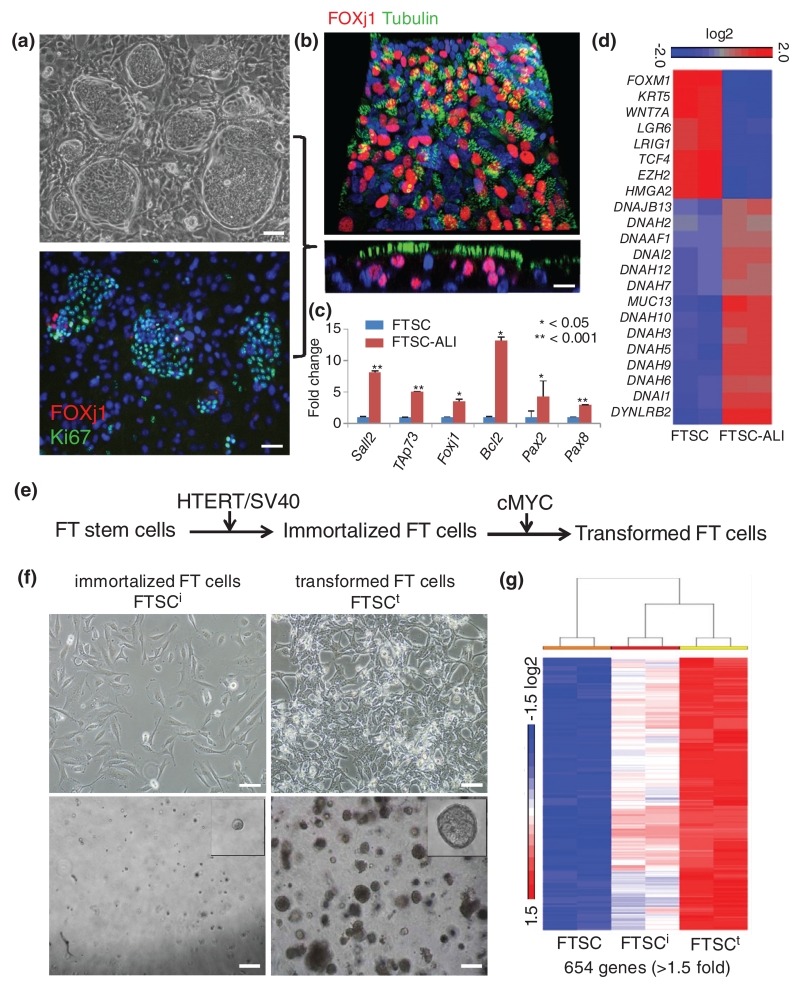
Cloning, immortalization, and transformation of the Fallopian tube stem cells. (a) Cloned FTSCs with proliferation marker Ki67 (green) and ciliated marker FOXJ1 (red). Scale bar = 50 μm. (b) ALI differentiation culture of FTSCs stained with FOXJ1 (red) and acetylated tubulin (green). Scale bar = 25 μm. (c) RT-PCR of selected markers (n = 2; error bars, SD) (d) Heatmap of selected genes from whole-genome transcriptome analysis. (e) Schematic of FTSC immortalization and transformation in vitro. (f) Morphology of immortalized (FTSCi) and transformed FT stem cells (FTSCt) on plastic culture dishes and in 3D Matrigel assay. Scale bar = 25 μm. (g) Progressive change of gene expression among FTSCs, FTSCi, and FTSCt (n = 2 each). Genes with increased expression (> 1.5-fold and p < 0.05, 654 genes) following transformation were selected for heatmap production.
Figure 2.
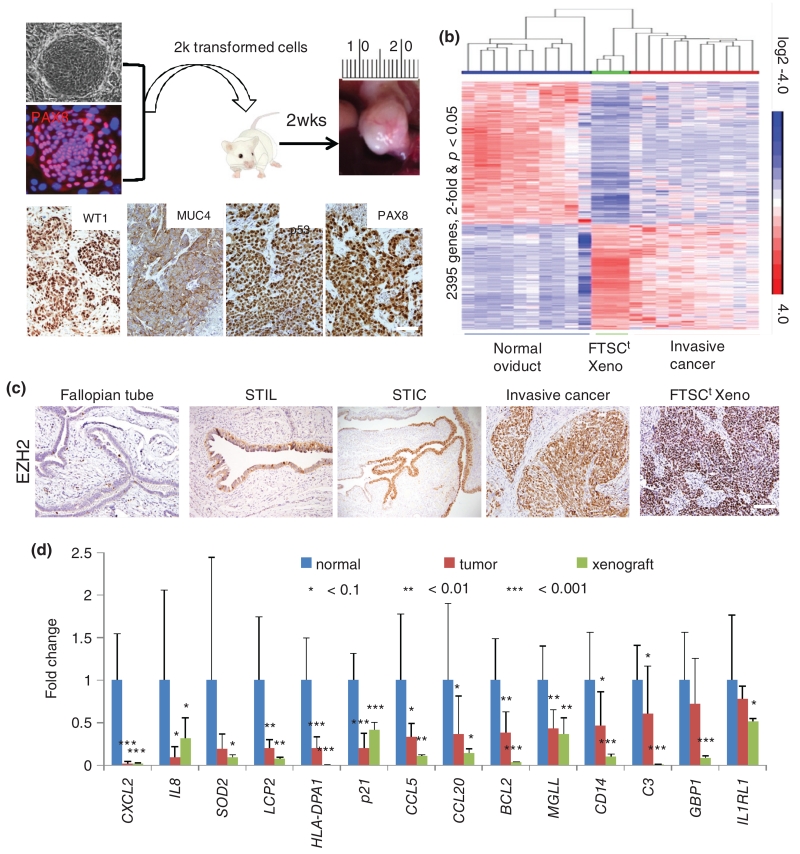
FTSCt xenograft tumour resembles human high-grade serous cancer. (a) Upper panel: 2000 FTSCt cells (PAX8, red) were injected into NSG mice and palpable tumour was observed at 2 weeks. Lower panel: xenograft tumour expressed HGSC hallmarks MUC4, p53, and PAX8. Scale bar = 50 μm. (b) Heatmap showing that FTSCt xenograft tumours and invasive SC share similar gene expression profiles (FTSCt tumour: n = 3; invasive SC: n = 10; and paired normal oviduct: n = 10; 2395 genes selected, > 2-fold and p < 0.05). (c) EZH2 protein in multiple stages of HGSC development. Scale bar = 1 mm. (d) EZH2 target genes in FTSCt xenograft tumours and invasive SC (n > 3; error bars, SD).
Figure 4.
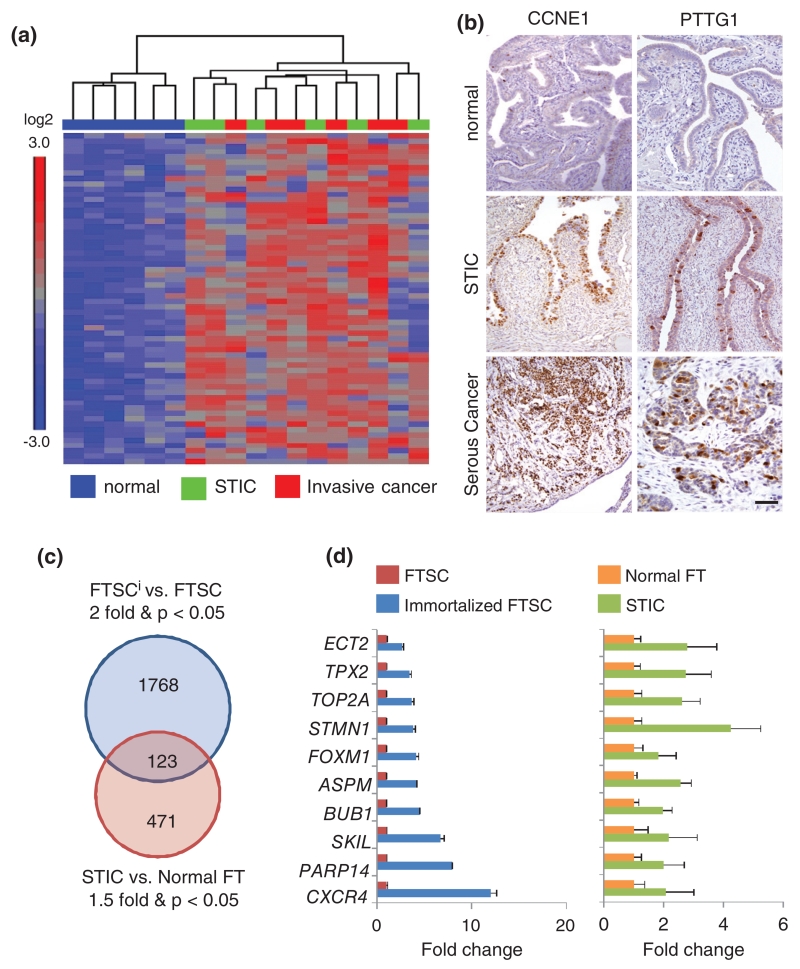
Early molecular changes associated with FTSC immortalization and STIC. (a) Heatmap showing 62 genes (> 2-fold, p < 0.05) commonly overexpressed between STIC and matched invasive serous cancer. (b) Representative images of CCNE1 and PTTG1 immunostaining on normal FT epithelium, STIC, and invasive serous cancer. Scale bar = 1 mm. (c) Venn diagram of genes overexpressed in STIC (> 1.5-fold, p < 0.05) and immortalized FTSCs (> 2-fold, p < 0.05). (d) Selected overlapping genes and fold change. n = 2; error bars: SD.
Results
Cloning, immortalizing, and transforming Fallopian tube stem cells
If the Fallopian tube is the origin of serous cancer, one possible mechanism for the evolution of cancer is a dysregulation of indigenous stem cells. We therefore set out to clone the stem cells of the Fallopian tube using methods to clone columnar epithelial stem cells such as human intestinal stem cells [11]. Using this method, we were able to generate clones of Fallopian tube stem cells which contained many small, undifferentiated, and highly proliferative (Ki67+) cells that can propagate through multiple passages (Figure 1a). These stem cell clones show strong and consistent staining with markers of Fallopian tube epithelial cells (PAX8) (Figure 2a). In our recent work on cloning adult stem cells from human airway and human intestine [11,12], we established a pedigree analysis method to examine the multipotential differentiation ability from a single stem cell. The high clonogenic capacity of Fallopian tube stem cells (consistently 10–15% throughout passages; data not shown) allowed us to use the same approach to rapidly generate single cell ‘pedigree’ lines of expansion and characterization of their lineage fates upon induced differentiation in air–liquid interface (ALI) cultures. Following FTSC differentiation, we found through immune staining with specific antibodies and RT-PCR with specific primers that one single Fallopian tube stem cell (Foxj1−, acetylated tubulin−, TAp73−, Sall2−, BCL2−, PAX2−) can give rise to both ciliated cells (Foxj1+, acetylated tubulin+, TAp73+, Sall2+) and secretory cells (Foxj1−, acetylated tubulin−, BCL2+, PAX2+) [18] (Figures 1b and 1c). Moreover, the same FTSC pedigree line can be induced to differentiate into squamous metaplasia (p63+/Krt5+) in 3D Matrigel assay [19]. We next compared the FTSCs and their differentiated structure in ALI by gene expression. FTSCs showed high expression of several known adult stem cell markers such as Lrig1 [20] and Lgr6 [21] and regulators of self-renewal such as EZH2 [22], FOXM1 [23], and TCF4 [24]. Interestingly, we did not find high expression of Lgr5 [25] in FTSCs. While the differentiated cells lost expression of stem cell markers, they showed increased expression of genes associated with ciliated cell and secretory cell differentiation such as genes in the dynein family [26] (DNAH3, DNAH2, DNAI2, DNAH12, DNAH7, DNAH10, DNAH5, DNAH9, DNAH6, DNAI1, and DYNLRB2) and MUC13 [27] (Figure 1d and Supplementary Table 2).
To examine whether the Fallopian tube stem cells are the cell of origin of high-grade serous cancer, we introduced SV40/hTERT or SV40/hTERT/c-MYC into these cells by retroviral infection to induce immortalization or transformation of these cells (Figure 1e). We showed that while both immortalized and transformed FTSCs gained the new property of growing without the support of an irradiated 3 T3-J2 fibroblast feeder, the transformed FTSCs lost contact inhibition and showed fibroblast-like morphology (Figure 1f, upper). Moreover, both immortalized and transformed FTSCs formed sphere structures in growth factor-reduced Matrigel in 5 days, but transformed FTSCs generated the irregular structures around ten times larger in comparison with small and round spheres derived from immortalized FTSCs (Figure 1f, lower). A heatmap of differentially expressed genes in whole-genome transcriptome analysis of FTSCs and immortalized and transformed FTSCs showed distinct expression profile differences between normal FTSCs and transformed FTSCs (Figure 1d and Supplementary Table 2). Interestingly, immortalized FTSCs expressed at the moderate level many genes that are highly expressed in transformed FTSCs (Figure 1g and Supplementary Table 3). Gene ontology analysis was performed to identify the gene pathways significantly enriched in transformed cells including DNA replication and DNA repair (Supplementary Figure 1a). In addition, a highly amplified gene in ovarian cancer, c-MYC, was used as the transforming agent [28]. Consistently, we observed that downstream genes of c-MYC are highly enriched in transformed FTSCs but not in immortalized FTSCs (Supplementary Figure 1b).
Transformed Fallopian tube stem cells gave rise to high-grade serous cancer
To examine whether the transformed Fallopian tube stem cells belong in the serous carcinogenic pathway, we injected 2000 transformed Fallopian tube stem cells subcutaneously into immunodeficient (NOD.Cg-Prkdcscid Il2rgtm1Wjl/SzJ) mice [29] and observed the formation of palpable tumour in 2 weeks. The xenografted tumours demonstrate all the pathological and immunological hallmarks of human high-grade serous cancer, such as gain of p53, WT1, EZH2, and MUC4 expression (Figure 2a) [19,30-32]. To determine whether FTSCt xenograft tumours resemble human high-grade serous cancer at the level of gene expression, we first compared the RNA from ten microdissected histologically normal Fallopian tube epithelium and paired high-grade serous cancer tumour samples on expression microarray chips. These data revealed a significant two-or-more-fold alteration (p < 0.5) of the levels of 2395 genes, which is presented in a heatmap (Figure 2b and Supplementary Table 4). Of the 2395 genes, 1017 are up-regulated and 1378 are down-regulated in tumour samples. Further analysis showed that FTSCt xenograft tumours, just like HGSC, also expressed these HGSC-related genes in a similar manner.
To uncover the genes or pathways that could be targeted to lead to tumour cell death for therapeutic purposes, we uncovered a number of druggable targets in HGSC such as enhancer of zeste homolog 2 (EZH2). EZH2 is a histone methyl transferase (HMT) and a member of the polycomb group of genes (PcG) regulating (suppressing) transcription through nucleosome modification, chromatin remodeling, and interaction with other transcription factors. Several studies have demonstrated that EZH2 is involved in oncogenesis, and high EZH2 transcript and protein levels have consistently been associated with aggressive tumour behavior, chemo-resistant tumour stem-like side populations, and overall poor clinical outcome in ovarian, breast, prostate, and bladder cancer patients [33]. We showed that the protein expression of EZH2 is up-regulated in precancerous lesions of HSGC (p53 signature) [34], STIC, invasive serous cancer, and FTSCt xenograft tumours (Figure 2c). Consistent with the increased expression of EZH2 in HGSC and xenograft tumours, the expression of downstream targets of EZH2 was significantly down-regulated compared with normal Fallopian tube epithelium (Figure 2d).
Resolving molecular alterations in STIC and its progression to invasive serous cancer
Although advanced serous cancer has been proposed to result from STIC progression, there has not been any transcriptome analysis to demonstrate the molecular progression from STIC to advanced serous cancer. To better understand the potential links between these entities via markers of progression, we used the LCM approach to isolate normal Fallopian tube epithelium, STIC, and advance serous cancer from the same patient (Figure 3a). A heatmap of differentially expressed genes in these datasets showed a distinct expression profile between normal Fallopian tubal epithelium and advanced serous cancer among six patients. In contrast, STIC showed a significant overlap with both normal epithelium and advanced cancer (Figure 3b). Microarray and gene set enrichment analysis (GSEA) were performed to identify the genes and pathways significantly enriched in both STIC and advanced cancer (Supplementary Figures 2c and 3b and Supplementary Table 9) or uniquely changed in advanced serous cancer (Figure 3c, Supplementary Figures 2a and 2b, and Supplementary Tables 5 and 8). The pathways involved in cell proliferation, genomic instability, and survival are aberrantly expressed at the early stage of serous carcinogenesis and are followed by deregulation of the pathways involved in cell migration and cell adhesion.
Figure 3.
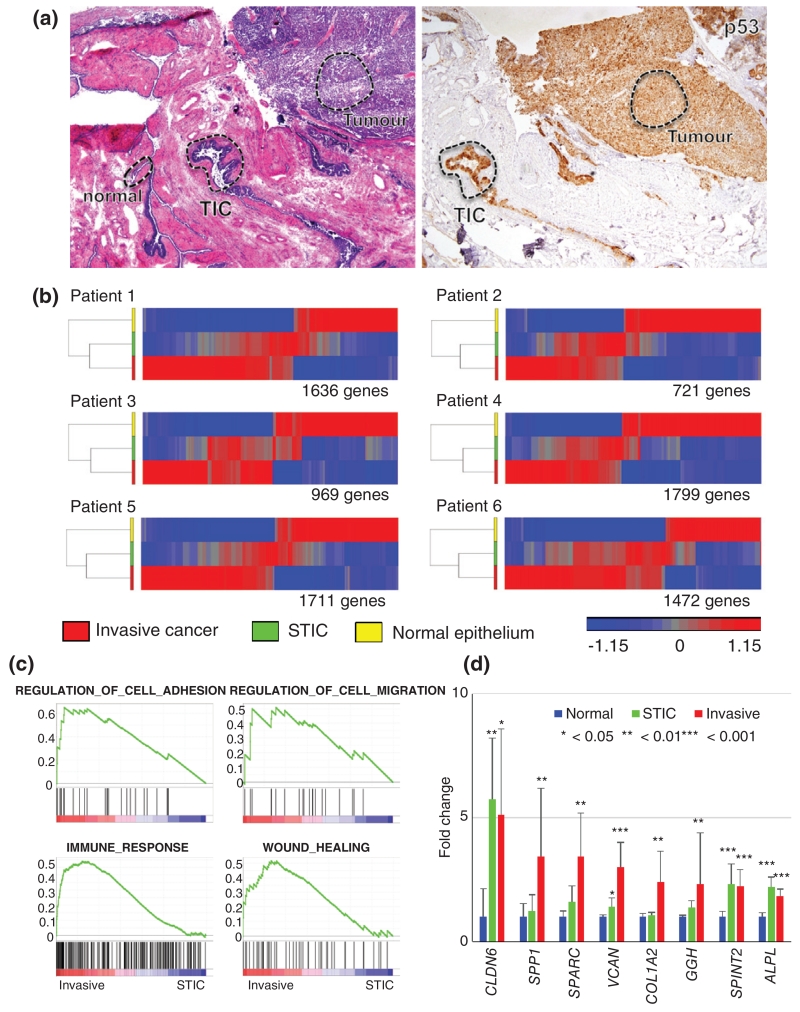
Molecular correlates of progression from STIC to invasive cancer. (a) Left: histology of the sections used for laser captured microdissection (LCM) of normal Fallopian tube epithelium, STIC, and invasive cancer. Right: p53 antibody staining showing high levels in STIC and invasive cancer. Scale bar = 1 mm. (b) Heatmaps showing progressive gene expression from STIC to invasive cancer in six individual patients (genes differentially expressed in invasive cancer compared with normal FT epithelium were selected, > 2-fold and p < 0.05). (c) Gene set enrichment analysis (GSEA) of invasive cancer versus STIC highlighting angiogenesis and regulation of cell adhesion in invasive cancer. (d) Plots of selected genes highly expressed in STIC and invasive cancer (normal Fallopian tube: n = 6; STIC: n = 6; invasive SC: n = 6; error bars: SD).
Furthermore, we particularly focused on the secreted proteins that are highly expressed in STIC or invasive serous cancer with the goal to use them as biomarkers for early detection of HGSC. We identified eight genes significantly up-regulated in invasive cancer and three of them are already up-regulated in the localized tubal tumour (STIC) (Figure 3d). Among them, SPP1 (osteopontin), SPARC (osteonectin), and VCAN (versican) have been reported to be overexpressed in various human cancers. In particular, osteopontin levels in plasma were significantly higher in patients with epithelial ovarian cancer compared with those of healthy controls and patients with other gynaecological cancers [35].
Uncovering early molecular changes associated with STIC
The data presented here support the existing hypothesis that the Fallopian tube is the site of origin of high-grade serous cancer and that STIC is the non-invasive, pre-metastatic form of high-grade serous cancer. A heatmap including 62 genes (>2-fold, p < 0.05) was generated to show genes up-regulated in common between STIC and matched invasive cancer (Figure 4a and Supplementary Table 6). Among these 62 genes, pituitary tumour-transforming gene (PTTG1) and cyclin E1 (CCNE1) (Supplementary Figure 3a) are particularly interesting because they have been implicated in early oncogenesis through their driving role in cellular transformation [36,37]. To validate the expression of some of these genes, we next performed the immuno-histochemistry using antibodies for PTTG1 and CCNE1 on patient-matched sections of normal Fallopian tube epithelium, STIC, and invasive cancer. While these two markers are barely detectable in normal Fallopian tube epithelium, they are highly expressed in STIC and invasive cancer (Figure 4b). We next hypothesized that among the aberrantly expressed genes in STIC, there is a group expressed during cellular immortalization, which is the first step towards malignancy [38]. To test this, we compared up-regulated genes in immortalized FTSCs and STIC and uncovered 123 genes (> 2-fold, p < 0.05) that overlap in these two entities (Figure 4c and Supplementary Table 7). Amongst these, stathmin 1, a microtubule destabilizing protein [39]; Ect2, a Rho guanine nucleotide exchange factor [40]; and forkhead box M1 (FOXM1), a transcription factor regulating cell cycle [41], have been suggested to play critical roles in HGSC initiation (Figure 4d).
Discussion
The perception of ovarian epithelial carcinogenesis is changing rapidly since the proposal that many of these tumours appear to originate in the Fallopian tube [34]. In 2012, Gilbert et al further supported this hypothesis – among patients with ‘early’ HGSC, the cancer had originated from the Fallopian tube, peritoneum, or both in 78% [42]. Further evidence for the tubal origin of high-grade serous carcinomas comes from a recent report noting that non-uterine high-grade serous carcinomas incidentally discovered in the general patient population arise in the Fallopian tube in most cases [43].
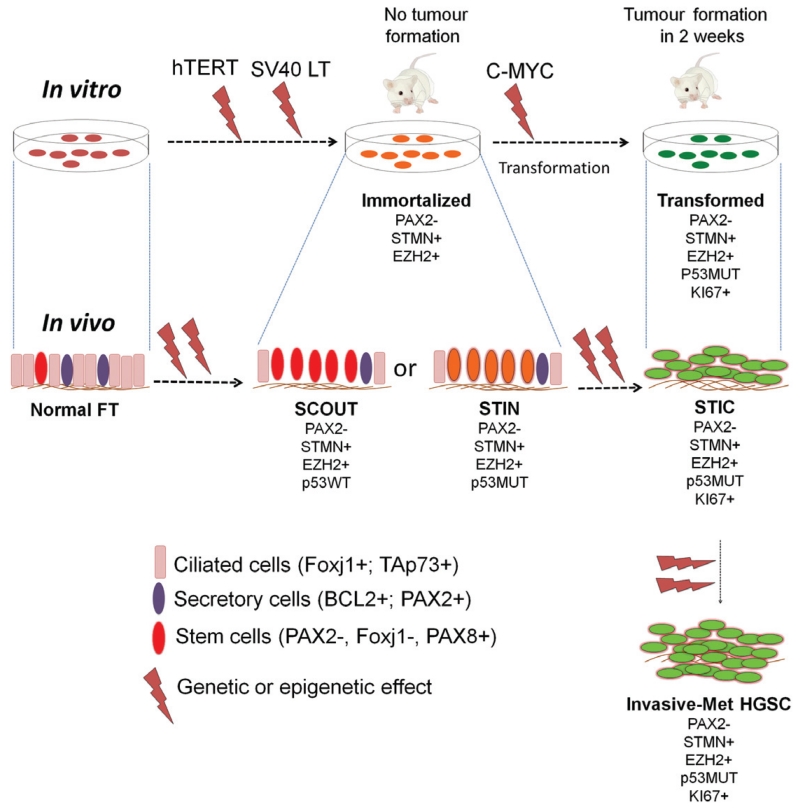
In this study, we cloned stem cells from human Fallopian tube and demonstrated that transformed Fallopian tube stem cells (FTSCs) can develop to aggressive HGSC in mouse xenograft models in a short time. The xenografted tumour shared all the hallmark features with HGSC, further supporting the Fallopian tube as the site of origin of serous cancer. In our working model (Figure 5), we hypothesize that the immortalized FTSCs correlate with STIN/SCOUTs and the transformed FTSCs correlate with STIC. Gene expression array and genomic analysis of cloned cells from STIN/SCOUTs or STIC will help to further examine this hypothesis and provide valuable information of the multi-step carcinogenesis of HGSC in vivo.
Figure 5.
In vitro and in vivo correlations proposing a model of multi-step development of HGSC originating from Fallopian tube stem cells.
Attempts to culture oviductal epithelial cells have been made previously, including efforts to model HGSC [14,44-47]. In addition, a study using a mouse model specifically targeting BRCA, p53, and pTEN in Fallopian tube further supports it as the site of origin for high-grade serous cancer [48]. However, none of these studies addressed the existence of FTSCs and their role in serous carcinogenesis. Herein, we report that stem cells of Fallopian tube can be maintained in culture in their elemental state and, using the pedigree approach, are capable of multipotent differentiation from one single stem cell in the Fallopian tube. Importantly, this platform of culturing FTSCs faithfully and robustly in vitro provided us a unique opportunity to functionally study putative oncogenes or tumour suppressors discovered in recent cancer genome analyses through genetic editing of patient-derived FTSCs. Moreover, in this study, we found that several putative oncogenes were significantly overexpressed at the step of FTSC immortalization prior to the occurrence of transformation. Among them, stathmin 1 has been proposed as a marker expressed in early pelvic serous carcinomas [39]; CXCR4 and its ligand CXCL12 have been suggested as the key determinants of tumour initiation and metastasis of ovarian cancer [49]; and forkhead box M1 (FOXM1) has been reported as a key regulator of tumourigenesis by increasing proliferative activity and leading to uncontrolled cell division [41]. EZH2, a negative regulator of transcription, was also up-regulated two-fold with immortalization. Interestingly, our previous study showed that the PAX2-null progenitor cell growth (secretory cell outgrowth, SCOUTs) in the Fallopian tube also strongly expressed both EZH2 and stathmin 1 [19]. This correlation led us to propose the link between FTSC immortalization and the development of early proliferations in the tube, either SCOUTs or lower-grade STINs. Based on these correlations, it appears that these gene perturbations associated with immortalization might occur prior to or even in the absence of the serous cancer, in which case their value as predictors of malignancy could be limited by their lack of specificity as actionable values. However, they could conceivably be targeted as co-determinants of neoplastic progression, with the goal of depriving the serous carcinogenic sequence of a participating pathway.
The stage at which an ovarian cancer is detected is the single most important factor influencing outcome, and interrupting ovarian cancer when it is curable will require addressing early disease in the distal Fallopian tube. Early molecular signatures that are specific for these neoplasms are of paramount importance given their potential value in detecting neoplasia via analysis of fluids in the lower genital tract [50]. Given the fact that many STICs do not have a HGSC outcome, we believe that there is a window of opportunity where patients with potentially lethal precursors can be identified through screening and spared death from this malignancy. It is hoped that a molecular analysis of early lesions might provide an array of targets that are either secreted by these cells or presented on the cell surface for screening and therapeutic value, respectively. Monoclonal antibodies to secreted proteins have the potential to form the basis of population-wide screening methods from blood or cervical fluid for those at risk who might benefit from salpingectomy. Monoclonal antibodies to cell surface markers in these lesions might assist in alternative detection via imaging technologies as the technology evolves. An important question that must be addressed is whether the parallels between immortalized cells in vitro and proliferations in vivo signify a background of accrued biological events that precede – and are needed for – progression to malignancy. Thus, the challenge will be to tease out those molecular events that are biologically significant and, when intervened, will prevent subsequent malignancy.
Supplementary Material
Figure S2. GSEA and heatmaps of differentially expressed genes.
Table S9. List of genes overexpressed in normal Fallopian tube epithelium.
Figure S3. PTTG1 and CCNE1 expression and enriched pathways in STIC and invasive cancer versus normal epithelium.
Table S1. List of antibodies.
Figure S1. Pathway enrichment analyses.
Table S2. Differentially expressed genes in FTSC and ALI structures.
Table S3. Genes with progressively increased expression in immortalized and transformed FTSC.
Table S4. Common genes expressed in xenograft tumour and human HGSC.
Table S5. Gene sets for pathways enriched in invasive cancer versus STIC.
Table S6. Genes overexpressed in STIC and invasive cancer.
Table S7. Genes overexpressed in both immortalized FTSC and STIC.
Table S8. List of genes specifically overexpressed in invasive cancer.
Acknowledgments
This work was supported by grants from Connecticut Innovations (WX and FM); the Joint Council Office of the Agency for Science Technology Research Agency (ASTAR), Singapore (WX and FM); the National Medical Research Council, Singapore (BnB11dec063 to NN, MC, FM, and WX); the Department of Defense (W81XWH-10-1-0289 to CPC); the Cancer Prevention & Research Institute of Texas (WX and FM); and the NIH (R01 CA188025 to ST). We thank J Hammer for assistance with figure preparation and the Division of Gynecologic Oncology at Brigham and Women’s Hospital and Dana Farber Cancer Institute for their contribution to the study. We also thank Drs Ju Yuan and In Young Hwang for assistance in cell culture. This study was approved by the Brigham and Women’s Hospital Institutional Review Board.
Abbreviations
- ALI
air–liquid interface culture
- FTSCs
Fallopian tube stem cells
- HGSC
high-grade serous cancer
- STIC
serous tubal intraepithelial carcinoma
Footnotes
No conflicts of interest were declared.
Author contribution statement
The authors contributed in the following way: design: YY, GN, MC, FMcK, CPC, and WX; data collection: YY, GN, BEH, KM, LW, XW, YH, TSW, TZ, DB FK, and WX; data analysis and interpretation: YY, GN, BEH, KM, LW, XW, YH, TSW, TZ, FK, NN, MC, ST, MB, FMcK, CPC, and WX; literature search: YY, MC, FMcK, CPC, and WX; figures: FMcK, CPC, and WX; manuscript writing: YY, MC, ST, MB, FMcK, CPC, and WX.
SUPPORTING INFORMATION ON THE INTERNET
The following supporting information may be found in the online version of this article:
Figure S1. Pathway enrichment analyses.
Figure S2. GSEA and heatmaps of differentially expressed genes.
Figure S3. PTTG1 and CCNE1 expression and enriched pathways in STIC and invasive cancer versus normal epithelium.
Table S1. List of antibodies.
Table S2. Differentially expressed genes in FTSC and ALI structures.
Table S3. Genes with progressively increased expression in immortalized and transformed FTSC.
Table S4. Common genes expressed in xenograft tumour and human HGSC.
Table S5. Gene sets for pathways enriched in invasive cancer versus STIC.
Table S6. Genes overexpressed in STIC and invasive cancer.
Table S7. Genes overexpressed in both immortalized FTSC and STIC.
Table S8. List of genes specifically overexpressed in invasive cancer.
Table S9. List of genes overexpressed in normal Fallopian tube epithelium.
References
- 1.Auersperg N, Wong AS, Choi KC, et al. Ovarian surface epithelium: biology, endocrinology, and pathology. Endocr Rev. 2001;22:255–288. doi: 10.1210/edrv.22.2.0422. [DOI] [PubMed] [Google Scholar]
- 2.Vaughan S, Coward JI, Bast RC, Jr, et al. Rethinking ovarian cancer: recommendations for improving outcomes. Nature Rev Cancer. 2011;11:719–725. doi: 10.1038/nrc3144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Piek JM, van Diest PJ, Zweemer RP, et al. Dysplastic changes in prophylactically removed Fallopian tubes of women predisposed to developing ovarian cancer. J Pathol. 2001;195:451–456. doi: 10.1002/path.1000. [DOI] [PubMed] [Google Scholar]
- 4.Marquez RT, Baggerly KA, Patterson AP, et al. Patterns of gene expression in different histotypes of epithelial ovarian cancer correlate with those in normal fallopian tube, endometrium, and colon. Clin Cancer Res. 2005;11:6116–6126. doi: 10.1158/1078-0432.CCR-04-2509. [DOI] [PubMed] [Google Scholar]
- 5.Finch A, Shaw P, Rosen B, et al. Clinical and pathologic findings of prophylactic salpingo-oophorectomies in 159 BRCA1 and BRCA2 carriers. Gynecol Oncol. 2006;100:58–64. doi: 10.1016/j.ygyno.2005.06.065. [DOI] [PubMed] [Google Scholar]
- 6.Lowell CB, Swisher EM, Cass I, et al. Long term follow up of BRCA1 and BRCA2 mutation carriers with unsuspected neoplasia identified at risk reducing salpingo-oophorectomy. Gynecol Oncol. 2013;129:364–371. doi: 10.1016/j.ygyno.2013.01.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Carlson JW, Miron A, Jarboe EA, et al. Serous tubal intraepithelial carcinoma: its potential role in primary peritoneal serous carcinoma and serous cancer prevention. J Clin Oncol. 2008;26:4160–4165. doi: 10.1200/JCO.2008.16.4814. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kindelberger DW, Lee Y, Miron A, et al. Intraepithelial carcinoma of the fimbria and pelvic serous carcinoma: evidence for a causal relationship. Am J Surg Pathol. 2007;31:161–169. doi: 10.1097/01.pas.0000213335.40358.47. [DOI] [PubMed] [Google Scholar]
- 9.Salvador S, Rempel A, Soslow RA, et al. Chromosomal instability in fallopian tube precursor lesions of serous carcinoma and frequent monoclonality of synchronous ovarian and fallopian tube mucosal serous carcinoma. Gynecol Oncol. 2008;110:408–417. doi: 10.1016/j.ygyno.2008.05.010. [DOI] [PubMed] [Google Scholar]
- 10.Mehrad M, Ning G, Chen EY, et al. A pathologist’s road map to benign, precancerous, and malignant intraepithelial proliferations in the fallopian tube. Adv Anat Pathol. 2010;17:293–302. doi: 10.1097/PAP.0b013e3181ecdee1. [DOI] [PubMed] [Google Scholar]
- 11.Wang X, Yamamoto Y, Wilson LH, et al. Cloning and variation of ground state intestinal stem cells. Nature. 2015;522:173–178. doi: 10.1038/nature14484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kumar PA, Hu Y, Yamamoto Y, et al. Distal airway stem cells yield alveoli in vitro and during lung regeneration following H1N1 influenza infection. Cell. 2011;147:525–538. doi: 10.1016/j.cell.2011.10.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Tan J, Li Z, Lee PL, et al. PDK1 signaling toward PLK1–MYC activation confers oncogenic transformation, tumor-initiating cell activation, and resistance to mTOR-targeted therapy. Cancer Discov. 2013;10:1156–1171. doi: 10.1158/2159-8290.CD-12-0595. [DOI] [PubMed] [Google Scholar]
- 14.Zhao JJ, Gjoerup OV, Subramanian RR, et al. Human mammary epithelial cell transformation through the activation of phosphatidylinositol 3-kinase. Cancer Cell. 2003;3:483–495. doi: 10.1016/s1535-6108(03)00088-6. [DOI] [PubMed] [Google Scholar]
- 15.Counter CM, Hahn WC, Wei W, et al. Dissociation among in vitro telomerase activity, telomere maintenance, and cellular immortalization. Proc Natl Acad Sci U SA. 1998;95:14723–14728. doi: 10.1073/pnas.95.25.14723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Subramanian A, Tamayo P, Mootha VK, et al. Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc Natl Acad Sci U S A. 2005;102:15545–15550. doi: 10.1073/pnas.0506580102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Huang da W, Sherman BT, Lempicki RA. Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nature Protoc. 2009;4:44–57. doi: 10.1038/nprot.2008.211. [DOI] [PubMed] [Google Scholar]
- 18.Levanon K, Ng V, Piao HY, et al. Primary ex vivo cultures of human fallopian tube epithelium as a model for serous ovarian carcinogenesis. Oncogene. 2010;29:1103–1113. doi: 10.1038/onc.2009.402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ning G, Bijron J, Yamamoto Y, et al. The PAX2-null immunophenotype defines multiple lineages with common expression signatures in benign and neoplastic oviductal epithelium. J Pathol. 2014;234:478–487. doi: 10.1002/path.4417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Wong VW, Stange DE, Page ME, et al. Lrig1 controls intestinal stem-cell homeostasis by negative regulation of ErbB signalling. Nature Cell Biol. 2012;14:401–408. doi: 10.1038/ncb2464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Oeztuerk-Winder F, Guinot A, Ochalek A, et al. Regulation of human lung alveolar multipotent cells by a novel p38α MAPK/miR-17-92 axis. EMBO J. 2012;31:3431–3441. doi: 10.1038/emboj.2012.192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Juan AH, Derfoul A, Feng X, et al. Polycomb EZH2 controls self-renewal and safeguards the transcriptional identity of skeletal muscle stem cells. Genes Dev. 2011;25:789–794. doi: 10.1101/gad.2027911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Bella L, Zona S, Nestal de Moraes G, et al. FOXM1: a key oncofoetal transcription factor in health and disease. Semin Cancer Biol. 2014;29:32–39. doi: 10.1016/j.semcancer.2014.07.008. [DOI] [PubMed] [Google Scholar]
- 24.van Es JH, Haegebarth A, Kujala P, et al. A critical role for the Wnt effector Tcf4 in adult intestinal homeostatic self-renewal. Mol Cell Biol. 2012;32:1918–1927. doi: 10.1128/MCB.06288-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ng A, Tan S, Singh G, et al. Lgr5 marks stem/progenitor cells in ovary and tubal epithelia. Nature Cell Biol. 2014;16:745–757. doi: 10.1038/ncb3000. [DOI] [PubMed] [Google Scholar]
- 26.Kikkawa M. Big steps toward understanding dynein. J Cell Biol. 2013;202:15–23. doi: 10.1083/jcb.201304099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Chauhan SC, Vannatta K, Ebeling MC, et al. Expression and functions of transmembrane mucin MUC13 in ovarian cancer. Cancer Res. 2009;69:765–774. doi: 10.1158/0008-5472.CAN-08-0587. [DOI] [PubMed] [Google Scholar]
- 28.Hahn WC, Counter CM, Lundberg AS, et al. Creation of human tumour cells with defined genetic elements. Nature. 1999;400:464–468. doi: 10.1038/22780. [DOI] [PubMed] [Google Scholar]
- 29.Shultz LD, Goodwin N, Ishikawa F, et al. Human cancer growth and therapy in immunodeficient mouse models. Cold Spring Harb Protoc. 2014;7:694–708. doi: 10.1101/pdb.top073585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Crum CP, Herfs M, Gang N, et al. Through the glass darkly: intraepithelial neoplasia, top down differentiation, and the road to ovarian cancer. J Pathol. 2013;231:402–412. doi: 10.1002/path.4263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Chauhan SC, Singh AP, Ruiz F, et al. Aberrant expression of MUC4 in ovarian carcinoma: diagnostic significance alone and in combination with MUC1 and MUC16 (CA125) Mod Pathol. 2006;9:1386–1394. doi: 10.1038/modpathol.3800646. [DOI] [PubMed] [Google Scholar]
- 32.Madore J, Ren F, Filali-Mouhim A, et al. Characterization of the molecular differences between ovarian endometrioid carcinoma and ovarian serous carcinoma. J Pathol. 2010;220:392–400. doi: 10.1002/path.2659. [DOI] [PubMed] [Google Scholar]
- 33.Rizzo S, Hersey JM, Mellor P, et al. Ovarian cancer stem cell-like side populations are enriched following chemotherapy and overexpress EZH2. Mol Cancer Ther. 2011;10:325–335. doi: 10.1158/1535-7163.MCT-10-0788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Lee Y, Miron A, Drapkin R, et al. A candidate precursor to serous carcinoma that originates in the distal fallopian tube. J Pathol. 2007;211:26–35. doi: 10.1002/path.2091. [DOI] [PubMed] [Google Scholar]
- 35.Kim JH, Skates SJ, Uede T, et al. Osteopontin as a potential diagnostic biomarker for ovarian cancer. J Am Med Assoc. 2002;287:1671–1679. doi: 10.1001/jama.287.13.1671. [DOI] [PubMed] [Google Scholar]
- 36.Pei L, Melmed S. Isolation and characterization of a pituitary tumor-transforming gene (PTTG) Mol Endocrinol. 1997;11:433–441. doi: 10.1210/mend.11.4.9911. [DOI] [PubMed] [Google Scholar]
- 37.Karst AM, Jones PM, Vena N, et al. Cyclin E1 deregulation occurs early in secretory cell transformation to promote formation of fallopian tube-derived high-grade serous ovarian cancers. Cancer Res. 2014;74:1141–1152. doi: 10.1158/0008-5472.CAN-13-2247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144:646–674. doi: 10.1016/j.cell.2011.02.013. [DOI] [PubMed] [Google Scholar]
- 39.Karst AM, Levanon K, Duraisamy S, et al. Stathmin 1, a marker of PI3K pathway activation and regulator of microtubule dynamics, is expressed in early pelvic serous carcinomas. Gynecol Oncol. 2011;123:5–12. doi: 10.1016/j.ygyno.2011.05.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Huff LP, Decristo MJ, Trembath D, et al. The role of Ect2 nuclear RhoGEF activity in ovarian cancer cell transformation. Genes Cancer. 2013;4:460–475. doi: 10.1177/1947601913514851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Wierstra I, Alves J. FOXM1, a typical proliferation-associated transcription factor. Biol Chem. 2007;388:1257–1274. doi: 10.1515/BC.2007.159. [DOI] [PubMed] [Google Scholar]
- 42.Gilbert L, Basso O, Sampalis J, et al. Assessment of symptomatic women for early diagnosis of ovarian cancer: results from the prospective DOvE pilot project. Lancet Oncol. 2012;13:285–291. doi: 10.1016/S1470-2045(11)70333-3. [DOI] [PubMed] [Google Scholar]
- 43.Gilks CB, Irving J, Köbel M, et al. Incidental nonuterine high-grade serous carcinomas arise in the fallopian tube in most cases: further evidence for the tubal origin of high-grade serous carcinomas. Am J Surg Pathol. 2015;39:357–364. doi: 10.1097/PAS.0000000000000353. [DOI] [PubMed] [Google Scholar]
- 44.Kervancioglu ME, Saridogan E, Martin JE, et al. A simple technique for the long-term non-polarised and polarised culture of human fallopian tube epithelial cells. Biol Cell. 1994;82:103–107. doi: 10.1016/s0248-4900(94)80012-x. [DOI] [PubMed] [Google Scholar]
- 45.Karst AM, Levanon K, Drapkin R. Modeling high-grade serous ovarian carcinogenesis from the fallopian tube. Proc Natl Acad Sci U S A. 2011;108:7547–7552. doi: 10.1073/pnas.1017300108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Jazaeri AA, Bryant JL, Park H, et al. Molecular requirements for transformation of fallopian tube epithelial cells into serous carcinoma. Neoplasia. 2011;13:899–911. doi: 10.1593/neo.11138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Shan W, Mercado-Uribe I, Zhang J, et al. Mucinous adenocarcinoma developed from human fallopian tube epithelial cells through defined genetic modifications. Cell Cycle. 2012;11:2107–2113. doi: 10.4161/cc.20544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Perets R, Wyant GA, Muto KW, et al. Transformation of the fallopian tube secretory epithelium leads to high-grade serous ovarian cancer in Brca;Tp53;Pten models. Cancer Cell. 2013;24:751–765. doi: 10.1016/j.ccr.2013.10.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Kajiyama H, Shibata K, Terauchi M, et al. Involvement of SDF-1α/CXCR4 axis in the enhanced peritoneal metastasis of epithelial ovarian carcinoma. Int J Cancer. 2008;122:91–99. doi: 10.1002/ijc.23083. [DOI] [PubMed] [Google Scholar]
- 50.Kinde I, Bettegowda C, Wang Y, et al. Evaluation of DNA from the Papanicolaou test to detect ovarian and endometrial cancers. Sci Transl Med. 2013;5:167ra4. doi: 10.1126/scitranslmed.3004952. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Figure S2. GSEA and heatmaps of differentially expressed genes.
Table S9. List of genes overexpressed in normal Fallopian tube epithelium.
Figure S3. PTTG1 and CCNE1 expression and enriched pathways in STIC and invasive cancer versus normal epithelium.
Table S1. List of antibodies.
Figure S1. Pathway enrichment analyses.
Table S2. Differentially expressed genes in FTSC and ALI structures.
Table S3. Genes with progressively increased expression in immortalized and transformed FTSC.
Table S4. Common genes expressed in xenograft tumour and human HGSC.
Table S5. Gene sets for pathways enriched in invasive cancer versus STIC.
Table S6. Genes overexpressed in STIC and invasive cancer.
Table S7. Genes overexpressed in both immortalized FTSC and STIC.
Table S8. List of genes specifically overexpressed in invasive cancer.