Majeed syndrome [OMIM #609628] is a syndromic form of CRMO which presents with early onset, severe CRMO and a microcytic dyserythropoietic anemia of variable severity. Onset in all reported cases has been in the first 2 years of life1–3. A minority of patients develop a neutrophilic dermatosis. The disease is due to mutations in LPIN2, which encodes LIPIN2, a phosphatidate phosphatase, important in lipid metabolism2–6.
Here we present the case histories of two male cousins of Indian heritage with Majeed syndrome. Both born to parents with consanguineous marriages; the proband presented with typical Majeed syndrome (onset of CRMO by 2 years of age, significant microcytic dyserythropoietic anemia and failure to thrive) but the cousin had later onset, milder disease more reminiscent of non-syndromic CRMO (onset at 8 years of age and minimal anemia). Both were found to be homozygous for the same novel LPIN2 mutation. This report details the natural history of the disease in these 2 boys, expands the at-risk ethnic and racial group of Majeed syndrome, and demonstrates that the disease can present in older children and have a milder phenotype similar to non-syndromic CRMO.
Case 1
A 15 y.o. boy presented with a history of monthly recurrence of painful swelling of the knees, ankles, elbows and wrists at 2 years of age lasting 10 to 15 days without rash or fever. Laboratory studies revealed chronic microcytic anemia (hemoglobin 7.9 – 9.9 g/dL, MCV 64.6 to 71.4), mild leukopenia (2840–4230 cells/mm3) and elevated ESR (53 – 140 mm/hr). He had been diagnosed with juvenile idiopathic arthritis and treated with methotrexate, naproxen and oral corticosteroids with some benefit. He was seen by the first author at 15 years of age, at which time he had severe failure to thrive (height 140 cm and weight 26 kg, both <<<3rd centiles) with absent secondary sexual characteristics. He had periarticular warmth and swelling in the lower extremities, hepatosplenomegaly but no rash. Bone marrow examination revealed marked erythroid hyperplasia with significant dyserythropoiesis (20% nuclear budding and karyorrhexis). Bone scan showed increased uptake in the distal femurs, proximal and distal tibiae, distal radii, and the left calcaneum. MRI of the lower extremity demonstrated increase T2 signal in the proximal and distal metaphyseal regions of the bilateral tibia suggestive of osteomyelitis. The parents did not consent for a bone biopsy; therefore, it was not performed.
A clinical diagnosis of Majeed syndrome was suspected. He was switched to parenteral methotrexate and given pamidronate 1 mg/kg/dose (except the first dose was 0.5 mg/kg) for 3 days every 3 monthly for over 2 years. His symptoms decreased by about 50 –60% (self-reported), he began to grow (from 145 cm at 16 yrs. to 159 cm at 19.5 yrs), gain weight (28 kg at 16 yrs. to 43 kg at 19.5 yrs) and had onset of puberty; hemoglobin increased to 9.5 g/dL and ESR fell from 107 mm/hr prior to pamidronate to ~ 60 mm/hr post treatment.
Case 2
The proband’s 13 year-old cousin, also born to a consanguineous parents, was asymptomatic until 8 years of age when he developed painful swelling at the metaphyseal regions of the long bones (symptoms were less severe and less frequent versus the proband). There was no fever or rash. His height and weight were between the 5th and 10th centiles. Painful juxta-articular inflammatory swelling was present at the ankles, knees and wrists. There was no organomegaly or rash. Investigation revealed a normal WBC, mild anemia (hemoglobin 11.2 g/dL, MCV of 86.8) and a moderately elevated ESR (45 mm/hr). Bone scan revealed symmetric uptake at the metaphyses of the long bones (Figure) and plain films showed osteolytic lesions at the fibular metaphysis bilaterally. MRI and bone marrow were not performed. He was started on weekly parenteral methotrexate with improvement in ESR (11–29 mm/hr) but no significant change in hemoglobin. He reported a 70–80% reduction in symptomatology.
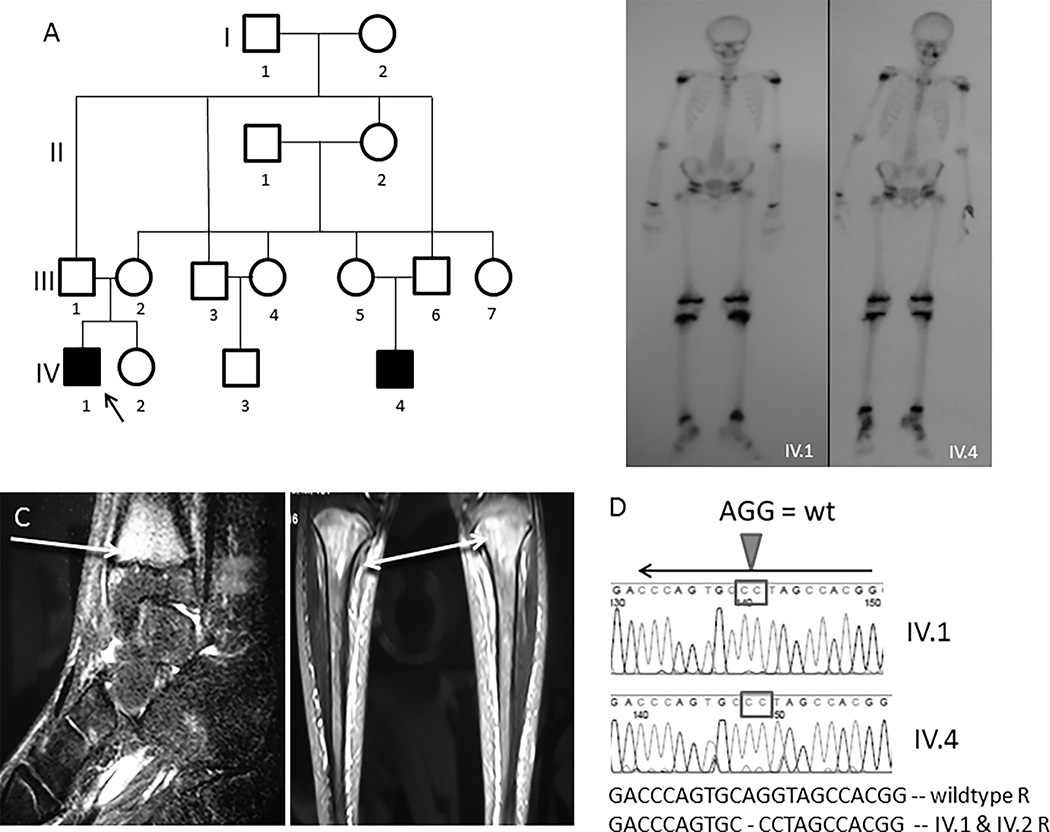
Figure.
Majeed Syndrome with phenotypic variability: A) Pedigree. Affected IV.1 (proband) and IV.4 (cousin) are in solid black; B) Tc99 bone scan (proband – IV.1) shows increased uptake in the knees, ankles and the right calcaneus in the proband; his cousin IV.4 shows symmetric uptake in the epiphyses of the long bones of the lower extremities C) MRI shows increased signal intensity in the tibial metaphyses in the proband, D) Sequencing on IV.1 and IV.2 show homozygous c.2241_2243delinsGG LPIN2 mutation.
LPIN2 was sequenced as previously described4 and a homozygous deleterious mutation was identified (c.2241_2243delinsGG) which results in a premature truncation of the protein at amino acid 747 [p.Tyr747*] in both the proband and his affected cousin while the parents were heterozygous.
Majeed syndrome is an exceedingly rare autoinflammatory bone disease. The disease has only been described in 9 children, all of Middle Eastern heritage1, 7, 8. All have had severe disease with onset in infancy to age 2 years. Here we report the first cases of Majeed syndrome in individuals of Indian heritage and with variable severity. The proband had typical Majeed syndrome with failure to thrive, hepatosplenomegaly, delayed puberty, dyserythropoietic anemia with onset of severe CRMO by 2 years of age. Yet, the proband’s cousin had only very mild anemia and mild CRMO with onset at age 8 years; demonstrating for the first time, that Majeed syndrome can present later in childhood with less severe disease mimicking non-syndromic CRMO.
Interleukin-1 inhibitors were not available to treat these boys; however, for severe disease, IL-1β blockade has been reported to be highly effective at controlling osseous and systemic inflammation3. These cases demonstrate that there can be phenotypic variability in Majeed syndrome and expands the at-risk ethnic/racial groups that can be affected by this disease.
Acknowledgments
Funding Source: This work was supported by the National Institute of Arthritis and Musculoskeletal and Skin Diseases at the National Institutes of Health [R01AR059703 PJF]
Abbreviations
- CDA
congenital dyserythropoietic anemia
- CRMO
chronic recurrent multifocal osteomyelitis
- IL-1
interleukin-1
Footnotes
Financial Disclosure: The authors have no financial relationships relevant to this article to disclose.
Conflict of Interest: The authors have no conflicts of interest to disclose.
Contributor’s Statement:
Anand Prahalad Rao, MD, Dharmanand Balebail Gopalakrishna, DM: Drs. Anand and Dharmanand identified the patients, requested the proper gene testing, drafted the initial case report, provided images for the manuscript and approved the final manuscript as submitted.
Xinyu Bing: Ms. Bing performed the sequencing and analysis of the data.
Polly J. Ferguson: Dr. Ferguson coordinated and supervised the data collection, oversaw the analysis, wrote the introduction and conclusion, revised the case report, and approved the final manuscript as submitted.
Bibliography
- 1.Majeed HA, Al-Tarawna M, El-Shanti H, Kamel B, Al-Khalaileh F. The syndrome of chronic recurrent multifocal osteomyelitis and congenital dyserythropoietic anaemia. Report of a new family and a review. Eur J Pediatr. 2001;160:705–710. doi: 10.1007/s004310100799. [DOI] [PubMed] [Google Scholar]
- 2.Al-Mosawi ZS, Al-Saad KK, Ijadi-Maghsoodi R, El-Shanti HI, Ferguson PJ. A splice site mutation confirms the role of LPIN2 in Majeed syndrome. Arthritis Rheum. 2007;56:960–964. doi: 10.1002/art.22431. [DOI] [PubMed] [Google Scholar]
- 3.Herlin T, Fiirgaard B, Bjerre M, Kerndrup G, Hasle H, Bing X, et al. Efficacy of anti-IL-1 treatment in Majeed syndrome. Ann Rheum Dis. 2013;72:410–413. doi: 10.1136/annrheumdis-2012-201818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Ferguson PJ, Chen S, Tayeh MK, Ochoa L, Leal SM, Pelet A, et al. Homozygous mutations in LPIN2 are responsible for the syndrome of chronic recurrent multifocal osteomyelitis and congenital dyserythropoietic anaemia (Majeed syndrome) J Med Genet. 2005;42:551–557. doi: 10.1136/jmg.2005.030759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Donkor J, Sariahmetoglu M, Dewald J, Brindley DN, Reue K. Three mammalian lipins act as phosphatidate phosphatases with distinct tissue expression patterns. J Biol Chem. 2007;282:3450–3457. doi: 10.1074/jbc.M610745200. [DOI] [PubMed] [Google Scholar]
- 6.Reue K. The lipin family: mutations and metabolism. Curr Opin Lipidol. 2009;20:165–170. doi: 10.1097/MOL.0b013e32832adee5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Majeed HA, Kalaawi M, Mohanty D, Teebi AS, Tunjekar MF, al-Gharbawy F, et al. Congenital dyserythropoietic anemia and chronic recurrent multifocal osteomyelitis in three related children and the association with Sweet syndrome in two siblings. J Pediatr. 1989;115:730–734. doi: 10.1016/s0022-3476(89)80650-x. [DOI] [PubMed] [Google Scholar]
- 8.Majeed HA, El-Shanti H, Al-Rimawi H, Al-Masri N. On mice and men: An autosomal recessive syndrome of chronic recurrent multifocal osteomyelitis and congenital dyserythropoietic anemia. J Pediatr. 2000;137:441–442. doi: 10.1067/mpd.2000.107613. [DOI] [PubMed] [Google Scholar]