Abstract
The maintenance of genome integrity and the generation of biological diversity are important biological processes, and both involve homologous recombination. In yeast and animals, homologous recombination requires the function of the RAD51 recombinase. In vertebrates, RAD51 seems to have acquired additional functions in the maintenance of genome integrity, and rad51 mutations cause lethality, but it is not clear how widely these functions are conserved among eukaryotes. We report here a loss-of-function mutant in the Arabidopsis homolog of RAD51, AtRAD51. The atrad51-1 mutant exhibits normal vegetative and flower development and has no detectable abnormality in mitosis. Therefore, AtRAD51 is not necessary under normal conditions for genome integrity. In contrast, atrad51-1 is completely sterile and defective in male and female meioses. During mutant prophase I, chromosomes fail to synapse and become extensively fragmented. Chromosome fragmentation is suppressed by atspo11-1, indicating that AtRAD51 functions downstream of AtSPO11-1. Therefore, AtRAD51 likely plays a crucial role in the repair of DNA double-stranded breaks generated by AtSPO11-1. These results suggest that RAD51 function is essential for chromosome pairing and synapsis at early stages in meiosis in Arabidopsis. Furthermore, major aspects of meiotic recombination seem to be conserved between yeast and plants, especially the fact that chromosome pairing and synapsis depend on the function of SPO11 and RAD51.
Homologous recombination and DNA-damage repair are fundamental biological processes found in all life forms. Homologous recombination plays a major role in both maintaining genome stability (DNA-damage repair) and the generation of genetic variability. Defects in DNA-damage repair generally lead to genome instability and are increasingly found to be associated with cancer in mammals. The active surveillance mechanisms that can recognize and precisely repair DNA damage to prevent the accumulation of errors are meanwhile thought to be intimately involved in the prevention of cancer and the delaying of aging (1, 2). Genes playing critical roles in homologous recombination are important for these processes.
Homologous recombination has been intensively studied in budding yeast Saccharomyces cerevisiae, and a number of genes have been identified that function in this process. Some of these genes, including RAD51, were identified based on the hypersensitivity of their mutants to radiation (3). RAD51 and another yeast gene, DMC1, share significant sequence homology with the bacterial recA gene (4). Similar to the bacterial RecA protein, the yeast RAD51 protein acts in homology searching, DNA pairing, and strand exchange (5), activities important for both DNA-damage repair and meiosis. RAD51 homologs have been found in all eukaryotic organisms thus far and are well studied in the vertebrates human, mouse, and chicken. In contrast to yeast, the loss of RAD51 function is lethal in both chicken DT40 and mouse cells (4). These RAD51-deficient cells arrest during the mitotic cell cycle, show signs of chromosome fragmentation, and undergo programmed cell death. Lethality of rad51 mutant cells is delayed by a mutation in the p53 tumor suppressor gene. In addition, RAD51 and p53 proteins interact directly, and expression of a dominant negative RAD51 gene that interferes with homologous recombination but not general DNA repair does not cause lethality in hamster cells (6). Furthermore, RAD51 and the cancer-susceptibility protein BRCA2 interact directly and colocalize to nuclear foci formed after DNA damage (7, 8), and defects in BRCA2 affect homologous recombination and DNA-damage repair. These data suggest that the lethality caused by rad51 defects in vertebrate cells is connected primarily to the effect of DNA damage on cell-cycle progression and not directly related to homologous recombination. However, it is unknown how conserved this feature is among higher eukaryotes.
Yeast rad51 mutations also cause meiotic defects including an accumulation of meiosis-specific double-stranded breaks (DSBs) and reduced formation of physical recombinants (9). RAD51 also plays a role in meiosis of fission yeast, Aspergillus nidulans, Caenorhabditis elegans, and Drosophila (10–13). In addition, RAD51 homologs are expressed at relatively high levels in the reproductive organs or meiotic cells of several organisms including chicken and mouse (14, 15). Furthermore, the RAD51 protein is localized to recombination foci along early meiotic chromosomes in yeast, maize, and mouse (16–19) and might be a component of early recombination nodules in lily (18). Detailed analysis of RAD51 foci localization during normal and mutant maize meiosis supports the idea that RAD51 is important for homologous chromosome pairing in addition to its role in recombination (20, 21). These findings suggest that RAD51 operates in meiosis in a variety of organisms and plays important roles in multiple processes including homologous chromosome pairing and meiotic recombination.
Homologous recombination in meiosis is required for proper chromosome segregation and generation of genetic diversity (22). In budding yeast, SPO11 plays a major role in the initiation of meiotic recombination (23) by catalyzing the formation of DSBs via a topoisomerase-like transesterase activity (24). This mechanism to initiate meiotic recombination seems to be conserved among eukaryotes because homologs of the SPO11 gene have been identified in both animals and plants; additionally, AtSPO11-1, the Arabidopsis SPO11 homolog, is also required for normal meiotic recombination and synapsis (25). However, little is known about the interplay between RAD51 and SPO11 in multicellular eukaryotes.
Although RAD51 function has been studied in organisms from fungi through animals, large gaps in our knowledge remain, especially for plants. Previously, an Arabidopsis RAD51 homolog, AtRAD51, was described (26). To analyze the function of AtRAD51, we identified a knockout mutation in this gene, atrad51-1, and studied its role in development and meiosis. Unexpectedly, the homozygous atrad51-1 plants were fully viable and developed normally under standard conditions. However, mutant plants were completely male- and female-sterile, and cytological and genetic analyses indicated that AtRAD51 is required for repair of meiotic DNA DSBs generated by AtSPO11-1. Our results demonstrate that RAD51 function in a higher plant is significantly different from that in vertebrates but may be similar to that in yeast and invertebrate animals.
Materials and Methods
Plant Materials. Both wild-type and atrad51-1 mutant plants are of the Columbia ecotype. Unless indicated otherwise, plants were grown under long-day conditions (16-h day/8-h night) at 22 ± 2°C.
Phenotypic Analysis. Tetrads were dissected and stained with 0.1% toluidine blue before being photographed. Viability of pollen grains was examined by staining with Alexander's solution (27). Mitosis was examined by using root tips of 4-day-old seedlings; spindle structure was examined by using antibodies against β-tubulin (Sigma). For mitotic index determination, cells that have a preprophase microtubule band and a perinuclear envelope were regarded as preprophase/prophase cells; cells with an established spindle structure and aligned condensed chromosomes but lack a preprophase microtubule band and a nuclear envelope were grouped as metaphase cells; anaphase cells were those having an anaphase spindle and newly separated chromosomes; and telophase cells were those with clustered and partially decondensed chromosomes (28). Meiotic chromosomal behavior in meiocytes was observed by preparing chromosome spreads (29) stained with 4′,6-diamidino-2-phenylindole (1 μg/ml). Female meiosis was analyzed essentially according to Armstrong et al. (30) by using floral buds at stages 10–11 (31).
RT-PCR. RNA was extracted from wild-type and atrad51-1 leaves and young inflorescences and treated with RQ1 DNase (Promega). cDNA was synthesized by using Superscript II and random priming (Invitrogen) according to manufacturer instructions. Quantitative RT-PCR was performed by combining gene-specific primers atrad51-1-1 (5′-GGTTCCATCACGGAGTTATATGG-3′) and atrad51-2-1 (5′-AGCCATGATATTCCCAC CAATC-3′) with plant-specific 18S rRNA primers (Quantum RNA 18S internal standards) (1:10 dilution; primer/competimer, 3:5) according to manufacturer instructions (Competimer primer kit, Ambion, Austin, TX).
Complementation with a CaMV 35S::MYC::AtRAD51 Construct. The AtRAD51 cDNA was cloned by PCR amplification of a wild-type young inflorescence cDNA sample with gene-specific primers oMC858 and oMC859 and inserted into a binary MYC vector (pROK2-MYC vector). An Agrobacterium C3581 strain containing that the confirmed construct was used to transform AtRAD51/atrad51-1 plants. Transgenic plants that carried the atrad51-1 allele but not the wild-type AtRAD51 allele were identified, and the expression of the MYC::AtRAD51 transgene was tested. Expression of the constitutive APT1 gene was examined as a control (32) (for details, see Supporting Materials and Methods, which is published as supporting information on the PNAS web site).
Generation of the atrad51-1 atspo11-1 Double Mutant. A cross was performed between AtSPO11-1/atspo11-1 and AtRAD51/atrad51-1 plants, and resulting F1 double-heterozygous plants (AtRAD51/atrad51-1; AtSPO11-1/atspo11-1) were identified by using PCR with gene-specific primers. Progeny of the double-heterozygous plants were analyzed to identify double mutants by PCR (see Supporting Materials and Methods for details).
RNA in Situ Hybridization. Nonradioactive RNA in situ hybridization was performed essentially as described (33). The antisense and sense RNAs of AtRAD51 were labeled with digoxigenin through in vitro transcription of linearized pTA-AtRAD51, which carries a fragment of the AtRAD51 cDNA, amplified with gene-specific primers oMC1479 and oMC1480 (5′-CTGAATAATTTCCTGTCTCTGAGCATG-3′).
Transmission Electron Microscopy. For transmission electron microscopy, the fixation and infiltration procedures were performed according to ref. 34 with minor modifications. To determine appropriate meiotic stages, thick cross sections through the buds were cut, collected on glass slides, stained with 0.1% toluidine blue, and observed with a light microscope. Ultrathin sections were examined with a JEOL 1200 EXII transmission electron microscope, and at least 50 ultrathin sections were analyzed for each bud. Additional details can be found in Supporting Materials and Methods.
Results
Identification of an AtRAD51 T-DNA Insertional Mutant. A T-DNA (portion of the tumor-inducing plasmid that is transferred to plant cells) insertion line containing an insert in the AtRAD51 gene (line 134A01 [see Fig. 7A, which is published as supporting information on the PNAS web site]) was identified in the GABI-KAT population (35). This line contained two T-DNA inserts that segregated independently. From this line, plants that contained only the T-DNA insertion in AtRAD51 were obtained by outcrossing, and this insertion was designated as the atrad51-1 allele. The insert is a highly truncated copy of the pAC161 tagging vector (GenBank accession no. AJ537514) located between nucleotide positions 2830 and 2831 in the DNA sequence of the AtRAD51 gene (26). Southern blotting and long-template PCR indicated that the atrad51-1 insert did not cause any additional rearrangements of the AtRAD51 sequence (data not shown). DNA sequencing of the PCR product showed that the insertion carried neither a complete resistance marker nor an intact right border. atrad51-1 is likely a loss-of-function allele, because the AtRAD51 mRNA was not detectable in plants homozygous for the T-DNA insertion (see Fig. 7B) by using quantitative RT-PCR that was sensitive enough to detect a <100-fold lower level of wild-type AtRAD51 transcript.
AtRAD51 Function Is Dispensable for Vegetative Growth and Root Mitosis. Homozygous atrad51-1 plants showed no apparent abnormal phenotype during the vegetative phase. atrad51-1 homozygous plants were identified by PCR, grown under standard long- and short-day conditions, and indistinguishable from heterozygous and wild-type plants until they started to set seeds (see Fig. 8, which is published as supporting information on the PNAS web site). In particular, homozygous plants grew with a similar rate and developed a similar number of rosette and cauline leaves and a similar biomass as their wild-type neighboring plants (data not shown). Therefore, the loss of AtRAD51 function did not cause detectable defects on vegetative development.
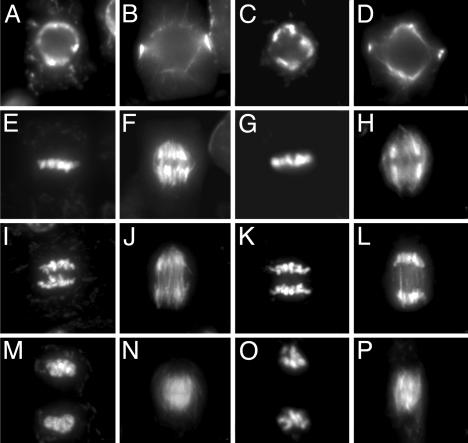
Loss of the RAD51 function in mouse and chicken severely affects cell proliferation, and RAD51-defective cells have a severe defect in mitosis that is accompanied by chromosome fragmentation (reviewed in ref. 4). Because growth experiments might not detect minor defects in mitosis, a direct cytological analysis was performed. Mitosis in 4-day-old root tips of mutant plants (Fig. 1 C, D, G, H, K, L, O, and P) was indistinguishable from wild-type plants (Fig. 1 A, B, E, F, I, J, M, and N) in all parameters analyzed. No difference was observed in the congression of the chromosomes at metaphase and the segregation at anaphase that result in the formation of two daughter nuclei at the end of mitosis. Moreover, the typical ratio of representative mitotic stages was not altered in atrad51 compared with wild type [wild type: 56.9% preprophase/prophase, 12.6% metaphase, 6.5% anaphase, and 24.0% telophase cells (n = 462); atrad51-1: 58.0% prophase/preprophase, 11.7% metaphase, 7.7% anaphase, and 22.6% telophase cells (n = 350)].
Fig. 1.
Mitosis in root tips of wild-type (A, B, E, F, I, J, M, and N) and atrad51-1 (C, D, G, H, K, L, O, and P) seedlings. Chromosomes (Left) and microtubule structures (Right) in the same cells were visualized with 4′,6-diamidino-2-phenylindole, and antibodies against β-tubulin were visualized at different stages during the mitotic cell cycle (preprophase, A and C; metaphase, E and G; anaphase, I and K; and telophase, M and O).
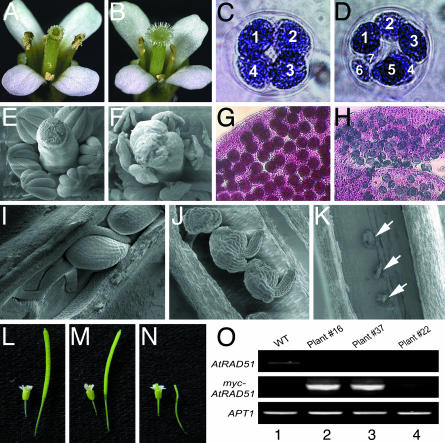
The atrad51-1 Mutant Is Male- and Female-Sterile. In contrast to the normal vegetative development, the atrad51-1 mutant plants were sterile (Fig. 2B). The mutant pollen grains were nonviable (as shown by staining with Alexander's solution; Fig. 2H), variable in size, and usually smaller than wild-type ones (data not shown). Although wild-type meioses (>99%, 230 meioses from four plants) produced four spores (Fig. 2C), mutant meioses produced multiple spores ranging in number from five to nine, and most tetrads contained six or seven spores (Fig. 2D). Although atrad51-1 flowers have normal carpels (Fig. 2 B and F) and produce wild-type-like ovules (Fig. 2 J), the ovules did not develop further after meiosis and subsequently degenerated (Fig. 2K), preventing further seed development (Fig. 2N). Female sterility is confirmed by the complete failure to obtain seed set after the mutant stigma was pollinated with wild-type pollen grains (10 stigma from four mutant plants).
Fig. 2.
Phenotypes of wild-type (A, C, E, G, I, and L), atrad51-1 (B, D, F, H, J, K, and N), and atrad51-1/atrad51-1 35S::MYC::AtRAD51 transgenic plants (M). Shown are opened flowers (A and B), stamens and carpels dissected from unopened flowers (E and F), tetrad (C and D) with numbered microspores, pollen grain (G and H), ovule (I–K; arrows point at aborted ovules), and young siliques (L and N). Two independent 35S::MYC::AtRAD51 transgenic lines (plants 16 and 37) produced siliques (M) that were similar in size to the wild-type ones (L). RT-PCR analysis of these two lines and another sterile transgenic line was performed and shown in O. Lanes: 1, wild type; 2, T1 transgenic plant 16; 3, T1 transgenic plant 37; 4, T1 transgenic plant 22. APT1 expression was determined as a positive control.
The sterility phenotype is genetically linked to the atrad51-1 mutation, because in a population of 149 plants derived from a heterozygote, only the 38 homozygous atrad51-1 plants were sterile. In addition, the defect can be complemented by the introduction of a CaMV 35S::MYC::AtRAD51 gene, which completely restored fertility in homozygous mutants (Fig. 2M). Fertility was correlated with high-level expression of the introduced transgene (plants 16 and 37), whereas lines with low-level expression remained sterile (e.g., plant 22; Fig. 2O). Expression of the endogenous AtRAD51 gene was not detectable in any of these plants (Fig. 2O), consistent with their atrad51 mutant background. Complementation in the next generation strictly cosegregated with the transgene (data not shown). Therefore, the loss of AtRAD51 function is the cause of the sterility phenotype.
The AtRAD51 Gene Is Highly Expressed in Meiotic Cells. AtRAD51 was shown previously by Northern blotting to be expressed widely at low levels, and the highest expression was in the floral buds (26). We examined the AtRAD51 expression in detail in reproductive organs by using RNA in situ hybridization. AtRAD51 expression was first detected in flower primordia at a rather low level (Fig. 3A). An increased level was observed in young anthers before meiosis (Fig. 3B), and its expression became even stronger in flowers at the time of male meiosis, particularly in the meiocytes (Fig. 3 C and D). AtRAD51 was expressed also in the female meiotic cells (Fig. 3E). However, AtRAD51 expression could not be detected in developing male gametophytes (Fig. 3F) or in carpels (Fig. 3G) after meiosis. The expression of AtRAD51 in both male and female meiocytes is consistent with an important function in meiosis.
Fig. 3.
AtRAD51 gene expression pattern in floral organs. AtRAD51 expression was detected in floral primordia (A) and at moderate levels in very young flower buds (B). In later anthers, the expression is strong in meiocytes (C) and restricted in the anther locules (D). Expression of AtRAD51 in carpels is restricted to ovules (E). There was no detectable expression in pollen grains (F) or gynoecium after meiosis (G) or when a sense probe was used (H).
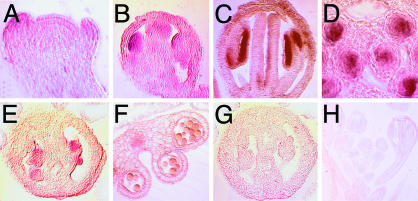
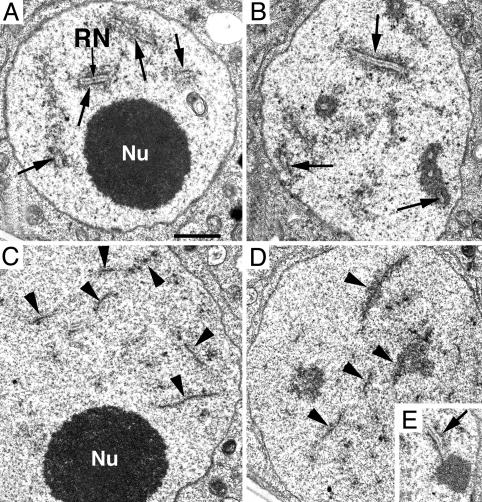
Male Meiosis Is Disrupted in atrad51-1 Mutant Plants. Because the atrad51-1 mutant tetrads indicated that male meiosis is defective, we compared male meiosis in wild-type and atrad51-1 mutant plants. Wild-type and mutant male meiocytes were similar at early prophase I (leptotene; Fig. 4 A and G). However, at zygotene, homologous chromosome pairing can be seen as pairing forks in wild type (Fig. 4B) but not in atrad51-1 (Fig. 4H). In wild type, meiosis proceeds by completing synapsis of homologous chromosomes at pachytene (Fig. 4C) and further condensation at diplotene (Fig. 4D), leading to the formation of five highly condensed bivalents at diakinesis (Fig. 4E). These bivalents further condense and align at the division plane at metaphase I (Fig. 4F). In atrad51-1, no typical fully synapsed chromosomes were observed among >1,000 meiocytes. Additional analysis with transmission electron microscopy verified that the vast majority of atrad51-1 meiocytes did not have any synaptonemal complexes (SCs) (Fig. 5C), although an occasional SC was observed (Fig. 5E). At the stages corresponding to zygotene through late pachytene, axial elements remained unpaired in the mutant cells when chromosomes appeared much more condensed (Fig. 5D), whereas in wild-type zygotene cells, most axial elements were associated into SCs (Fig. 5A). Extension of SCs was increased during progression of chromosome condensation, and by late pachytene no unpaired axial elements were found (Fig. 5B).
Fig. 4.
Male and female meiosis in wild-type (male, A–F; female, M–R) and atrad51-1 (male, G–L; female, S–X) plants. For male meiosis, stages included for wild-type (A–F) and atrad51 (G–L) meiosis are leptotene (A and G), zygotene (B and H), pachytene (C and I), diplotene (D and J), diakinesis (E and K), and metaphase I (F and L). For female meiosis, stages included for wild-type (M–R) and atrad51-1 (S–X) meiosis are leptotene (M and S), zygotene (N and T), pachytene (O and U), diakinesis (P and V), metaphase I (Q and W), and anaphase I (R and X). The arrow in B indicates a pairing fork.
Fig. 5.
Transmission electron micrographs of male meiocyte nuclei in wild-type (A and B) and atrad51-1 (C–E) plants. SCs (arrows) were observed at zygotene (A) and pachytene (B) stages in wild type. In atrad51-1, the axial elements (arrowheads) remained unpaired in nuclei corresponding to zygotene (C) or pachytene (D) stages; an occasional SC was observed (E). Nu, nucleolus; RN, recombination nodule. (Scale bar, 100 nm.)
In addition to the absence of SCs, mutant meiocytes contain multiple brightly stained spots, ranging from 20 to 50 per meiocyte from diakinesis through the end of meiosis I (Fig. 4 J–L). These observations indicate that severe chromosome fragmentation had occurred at an early stage. From anaphase I through the end of meiosis II, the atrad51-1 meiocytes are abnormal (see Fig. 9, which is published as supporting information on the PNAS web site), largely as a consequence of earlier mutant defects. Chromosome fragmentation together with the absence of normal chromosomal pairing and synapsis indicates an essential role of AtRAD51 in Arabidopsis meiosis.
Female Meiosis in atrad51-1 Is Defective Also. The defects observed in mutant female meiocytes were similar to those in male meiosis (Fig. 4). In atrad51-1, homologous chromosome paring at zygotene was absent (Fig. 4T). In addition, we could not find fully synapsed chromosomes, and thus no typical pachytene stage was observed (Fig. 4U). Mutant female meiocytes contained >10 brightly stained chromosomal structures, indicating that chromosome fragmentation also occurred in female meiocytes (Fig. 4 V–X).
DSB Formation by SPO11 Is a Prerequisite for Chromosome Fragmentation. To investigate the relationship of SPO11-induced DSBs and chromosome fragmentation in atrad51-1 cells, we analyzed atspo11-1 atrad51-1 double mutants (Fig. 6). The chromosomal behavior of double-mutant plants (atrad51-1/atrad51-1; atspo11-1/atspo11-1) was similar (Fig. 6 G–L) to that of the atspo11-1 single mutant (Fig. 6 A–F). In particular, chromosome fragmentation was absent, and no meiocytes with >10 brightly stained dots were observed (Fig. 6 H and I). In addition, the absence of typical zygotene chromosome structures characteristic for atspo11-1 male meiosis was observed also in the atrad51-1 atspo11-1 double mutant (Fig. 6 A and G). Moreover, similar to those seen in the atspo11-1 mutant (Fig. 6 A–D), multiple unpaired chromosomes (6–10 per meiocyte) with only occasional chromosomal pairs were seen in the atrad51-1 atspo11-1 double mutant (Fig. 6 H–J and data not shown). Consistently, both atspo11-1 and the double mutant had 20 sister chromatids at anaphase II (Fig. 6 F and L). The epistatic analysis showed that AtRAD51 functions downstream of AtSPO11-1 and suggests that a major function of AtRAD51 is to repair DSBs generated by AtSPO11-1. The atrad51-1 spo11-1 double mutant can also produce a small number of seeds (data not shown), similar to the spo11-1 single mutant, suggesting that segregation of the univalents can yield a low percentage of viable pollen.
Fig. 6.
Male meiosis in atspo11-1 (A–F) and atspo11-1 atrad51-1 (G–L). Stages included are zygotene (A and G), diakinesis (B and H), metaphase I (C and I), anaphase I (D and J), metaphase II (E and K), and anaphase II (F and L). The arrows in D and I indicate the presence of bivalents. See Fig. 4 for a comparison of the wild-type and atrad51-1 meioses.
Discussion
A T-DNA insertion was identified that caused a loss-of-function mutation in the Arabidopsis AtRAD51 gene. Phenotypic characterization showed that RAD51 is dispensable for normal vegetative development in Arabidopsis but has an essential function in male and female meiosis. The phenotype of this T-DNA-generated mutant is different from a report that used RNA interference to suppress RAD51 function in meiosis, and no effect on fertility was observed (36). These data imply that RNA-interference-generated data need to be treated with caution and support our conclusion that the T-DNA insertion is likely to have caused loss of RAD51 function.
RAD51 Is Not Necessary for Vegetative Development in Plants. rad51 mutant yeast cells are hypersensitive to ionizing radiation and DNA-damaging chemicals (9). Even more severely, rad51 mutations in vertebrates cause a severe defect in cell proliferation that eventually causes cell death. In the absence of data from a larger variety of organisms, different scenarios were discussed to explain this difference in lethality between yeast and vertebrates. One scenario was the need for a more efficient recombination apparatus in vertebrates, because such cells have a more complex genome and a different chromatin structure. Our data show that RAD51 is normally not required for vegetative and flower development in Arabidopsis. This fact indicates that an organism with high genome complexity and chromatin structure can survive without RAD51 function, and thus genome complexity is not likely to be the cause of lethality in vertebrates.
Another possibility is that RAD51 in higher organisms has acquired an additional function that links quality control in genome maintenance to cell-cycle progression (4, 37). Such a link might exist in human cells, in which RAD51 has been shown to interact with tumor suppressor genes p53, BRCA1, and BRCA2 (4). However, it is still unclear whether the relationship of RAD51 to cell-cycle control or recombination functions more downstream are the primary cause of lethality in vertebrates. To resolve this question, additional data from other complex, multicellular organisms such as Arabidopsis are needed. Arabidopsis differs in major aspects in the signal transduction chain linking genome integrity to cell-cycle progression. The genome contains no homolog for p53, and the BRCA1 (38) and the BRCA2 (36) homologs that exist in Arabidopsis may differ from vertebrate BRCA proteins. Homology of AtBRCA1 to the vertebrate proteins is rather low, and AtRAD51 differs in amino acid sequence from vertebrate proteins in one of the interaction domains with BRCA2 that is considered important for regulation (data not shown). Therefore, no effect on normal mitotic growth together with major differences in the signal transduction pathway may indicate that plants differ from animals in the link of RAD51 to cell-cycle control and apoptosis and thus mitotic progression and DNA-damage control. RAD51 knockouts in other organisms, including multicellular animals C. elegans (39) and Drosophila (12), also do not cause lethality. Therefore, viability is the more common phenotype, clearly indicating that the link of RAD51 to cell-cycle control is the cause of lethality in vertebrates. In addition, these data suggest that the additional functions of RAD51 have mostly evolved in vertebrates, possibly because they need a more stringent control of DNA-damage repair.
A number of proteins with limited sequence homology to RAD51 are found in all eukaryotic genomes (reviewed in ref. 4). Although their function in homologous recombination is less well understood, they seem to be nonredundant with RAD51 but have partially overlapping functions in vertebrates. Recently, such a paralog, AtXRCC3, has been characterized in Arabidopsis (40). In contrast to vertebrates, loss of AtXRCC3 function also does not impair viability in Arabidopsis. Therefore, it is unlikely that paralogs can substitute for RAD51 function in vegetative development.
AtRAD51 Is Required for Normal Meiosis. The fact that the atrad51-1 mutant is viable and healthy is in sharp contrast to its severe meiotic defects. Light-microscopic analysis of both male and female meioses showed that meiosis in atrad51-1 proceeded normally through leptotene. However, in contrast to wild type, atrad51-1 chromosomes were not paired at zygotene and did not form SCs. Transmission electron microscopy strongly supported the conclusion that atrad51-1 is defective in synapsis. In addition, chromosomes become fragmented at the onset of diakinesis, and fragmentation remains visible through the rest of meiosis. The extensive chromosome fragmentation is likely to be the cause of abnormal segregation of chromosomes and, consistently, of complete sterility. The failure of atrad51-1 meiocytes in chromosome pairing and synapsis strongly suggests that RAD51 is important for interhomolog recognition in Arabidopsis meiosis. In maize and lily, RAD51 is localized to numerous loci at early zygotene and associated with pairing, then subsequently found at greatly reduced foci during pachytene (18, 19). Furthermore, RAD51 foci are greatly reduced in many maize mutants that are defective in pairing (20). Our analysis of atrad51-1 strongly supports the conclusion drawn from these results that RAD51-mediated homology search is critical for pairing and synapsis in plants. This situation may be different from animals, because a knockout of RAD51 in C. elegans resulted in abnormal chromosomal morphology and univalent formation at diakinesis but did not affect meiotic homology recognition and synapsis (39). In addition, the atrad51-1 defects in pairing and recombination do not seem to activate a checkpoint that could prevent progression through meiosis. This phenotype is in contrast to Drosophila melanogaster, in which a meiotic checkpoint is directly involved in the generation of the meiotic phenotype (9, 12), and Tetrahymena thermophila, in which rad51-null individuals resulted in an arrest during meiosis I (41). These data suggest that RAD51 deficiencies affect meiosis differently in different organisms and that meiosis in Arabidopsis is more similar to budding yeast than to animals. Our results indicate a direct involvement of RAD51 in chromosome pairing and synapsis in Arabidopsis; however, further experiments are needed to show that this role for RAD51 also applies to other organisms.
AtRAD51-1 Likely Acts Downstream of AtSPO11-1. In yeast, meiosis is initiated by a DSB introduced by SPO11, and RAD51 is involved in the repair of such DSBs (9, 12). Chromosome fragmentation caused by the atrad51-1 mutation is suppressed by an atspo11-1 mutation, implying that RAD51 also functions in meiotic DSB repair in Arabidopsis. Furthermore, RAD51 acts downstream of SPO11 in the Arabidopsis meiotic recombination pathway. Therefore, major aspects of meiotic recombination are conserved between yeast and plants, not only the function of key players but also the molecular connections between them.
A variety of mutants affecting meiosis have been reported in Arabidopsis. Mutants defective in synapsis include asy1, syn1, solo dancers, atspo11-1, and dmc1 (25, 42–44). SYN1 is especially interesting, because the mutation also to leads chromosome fragmentation in meiosis. SYN1 encodes a cohesin that possibly is required for the establishment of the chromosomal structure during prophase I. Therefore the syn1 phenotype suggests that a close interaction between homologous chromosomes is a prerequisite for meiotic DSB repair, in which RAD51 is involved. The yeast DMC1 protein, a RAD51 paralog, has roles both overlapping with and distinct from those of RAD51 in meiotic recombination and synapsis. The yeast dmc1 mutant exhibited extensive meiotic chromosome fragmentation, and DMC1 may also function at a later stage than does RAD51 (45); however, dmc1 knockouts in animals and plants lack chromosome fragmentation, although they were sterile and defective in meiotic chromosome synapsis, indicating that these proteins have separated functions in meiosis (46, 47). In contrast to yeast in which both RAD51 and DMC1 are critical for the repair of SPO11-dependent DSBs, RAD51 alone may have this function in multicellular organisms, whereas the plant and animal DMC1 is not required and may be specific for late functions in meiotic recombination and synapsis.
Defects in AtXRCC3 also lead to chromosome fragmentation during meiosis I in Arabidopsis (40), suggesting that AtXRCC3 is involved in repair of meiotic DSBs as well. Apparently, however, RAD51 paralogs cannot fully substitute for RAD51 in meiosis. Therefore, AtRAD51 and its paralogs are likely to play distinct roles in plant meiosis, and possibly both are required for a certain process (AtRAD51 and AtXRCC3) or act in succession (AtRAD51 and AtDMC1). Knockout phenotypes of other recombination genes including AtRAD50 are similar to atrad51-1 in Arabidopsis. In contrast to animals, knockouts of AtXRCC3 and the AtRAD50 show no phenotype in vegetative development; however, meiosis is severely affected (40, 48). These results suggest that homologous recombination or major genes operating in this pathway have evolved in plants to function mainly in meiosis rather than DNA-damage repair.
Plant genomes are constantly challenged by genotoxic stresses such as oxidative damage or stalled replication forks that arise with cell division. The absence of a phenotype in the vegetative phase of Arabidopsis atrad51-1, atxrcc3, and atrad50 mutants suggests that plants might use pathways other than homologous recombination for DNA-damage repair as suggested by the high efficiency of nonhomologous DNA integration routinely observed in flowering plants (49). However, it is hardly conceivable that such imprecise repair mechanisms are used exclusively for DNA-damage repair in plants. Therefore, other homologous recombination mechanisms might also function in somatic cells or in mitosis, or yet-uncharacterized protein(s) might substitute for RAD51 function. Furthermore, the finding that AtRAD51 is important for chromosome pairing and synapsis during meiosis supports the idea that AtRAD51-dependent homology search is important for pairing and synapsis, in addition to its role in meiotic recombination (20, 21, 50). Additional investigations are needed to understand the function of AtRAD51 and also its relationship between its paralogs, such as AtXRCC3 and AtDMC1, in pairing, synapsis, and recombination.
Supplementary Material
Acknowledgments
We thank Dr. R. Cyr, E. Kemper, E. Wendeler, and E. Wonderlich for technical assistance; M. Rosso (Max-Planck-Institut für Züchtungsforschung, Köln, Germany) and B. Weisshaar (Max-Planck-Institut für Züchtungsforschung, Köln, Germany) for providing the atrad51-1 mutant; A. Omeis and J. Wang for plant care; and C. Hendrix and the reviewers for helpful comments on this manuscript. This work was supported by National Institutes of Health Grant R01 GM63871-01, National Science Foundation Grant MCB-0092075, U.S. Department of Energy Grant DE-FG02-02ER15332 (to H.M.), and European Commission Grants QLK3-CT-2000-00367 and LSHG-CT-2003-503303 (to B.R.). W.L. was partially supported by the Intercollege Graduate Program in Plant Physiology at Pennsylvania State University.
Abbreviations: DSB, double-stranded break; T-DNA, portion of the tumor-inducing plasmid that is transferred to plant cells; SC, synaptonemal complex.
References
- 1.Hickson, I. D. (2003) Nat. Rev. Cancer 3, 169–178. [DOI] [PubMed] [Google Scholar]
- 2.Lieber, M. R., Ma, Y., Pannicke, U. & Schwarz, K. (2003) Nat. Rev. Mol. Cell Biol. 4, 712–720. [DOI] [PubMed] [Google Scholar]
- 3.Game, J. (2000) Mutat. Res. 451, 277–293. [DOI] [PubMed] [Google Scholar]
- 4.Thacker, J. (1999) Trends Genet. 15, 166–168. [DOI] [PubMed] [Google Scholar]
- 5.Haber, J. (2000) Trends Genet. 16, 259–264. [DOI] [PubMed] [Google Scholar]
- 6.Lambert, S. & Lopez, B. S. (2000) EMBO J. 19, 3090–3099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Pellegrini, L., Yu, D. S., Lo, T., Anand, S., Lee, M., Blundell, T. L. & Venkitaraman, A. R. (2002) Nature 420, 287–293. [DOI] [PubMed] [Google Scholar]
- 8.Yu, D. S., Sonoda, E., Takeda, S., Huang, C. L., Pellegrini, L., Blundell, T. L. & Venkitaraman, A. R. (2003) Mol. Cell 12, 1029–1041. [DOI] [PubMed] [Google Scholar]
- 9.Shinohara, A., Ogawa, H. & Ogawa, T. (1992) Cell 69, 457–470. [DOI] [PubMed] [Google Scholar]
- 10.Muris, D. F., Vreeken, K., Schmidt, H., Ostermann, K., Clever, B., Lohman, P. H. & Pastink, A. (1997) Curr. Genet. 31, 248–254. [DOI] [PubMed] [Google Scholar]
- 11.van Heemst, D., Swart, K., Holub, E. F., van Dijk, R., Offenberg, H. H., Goosen, T., van den Broek, H. W. & Heyting, C. (1997) Mol. Gen. Genet. 254, 654–664. [DOI] [PubMed] [Google Scholar]
- 12.Staeva-Vieira, E., Yoo, S. & Lehmann, R. (2003) EMBO J. 22, 5863–5874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Takanami, T., Mori, A., Takahashi, H., Horiuchi, S. & Higashitani, A. (2003) Chromosome Res. 11, 125–135. [DOI] [PubMed] [Google Scholar]
- 14.Bezzubova, O., Shinohara, A., Mueller, R. G., Ogawa, H. & Buerstedde, J. M. (1993) Nucleic Acids Res. 21, 1577–1580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Yamamoto, A., Taki, T., Yagi, H., Habu, T., Yoshida, K., Yoshimura, Y., Yamamoto, K., Matsushiro, A., Nishimune, Y. & Morita, T. (1996) Mol. Gen. Genet. 251, 1–12. [DOI] [PubMed] [Google Scholar]
- 16.Bishop, D. K. (1994) Cell 79, 1081–1092. [DOI] [PubMed] [Google Scholar]
- 17.Ashley, T., Plug, A. W., Xu, J., Solari, A. J., Reddy, G., Golub, E. I. & Ward, D. C. (1995) Chromosoma 104, 19–28. [DOI] [PubMed] [Google Scholar]
- 18.Anderson, L. K., Offenberg, H. H., Verkuijlen, W. M. & Heyting, C. (1997) Proc. Natl. Acad. Sci. USA 94, 6868–6873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Franklin, A. E., McElver, J., Sunjevaric, I., Rothstein, R., Bowen, B. & Cande, W. Z. (1999) Plant Cell 11, 809–824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Pawlowski, W. P., Golubovskaya, I. N. & Cande, W. Z. (2003) Plant Cell 15, 1807–1816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Franklin, A. E., Golubovskaya, I. N., Bass, H. W. & Cande, W. Z. (2003) Chromosoma 112, 17–25. [DOI] [PubMed] [Google Scholar]
- 22.Romanienko, P. J. & Camerini-Otero, R. D. (1999) Genomics 61, 156–169. [DOI] [PubMed] [Google Scholar]
- 23.Klapholz, S., Waddell, C. S. & Esposito, R. E. (1985) Genetics 110, 187–216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Keeney, S., Giroux, C. N. & Kleckner, N. (1997) Cell 88, 375–384. [DOI] [PubMed] [Google Scholar]
- 25.Grelon, M., Vezon, D., Gendrot, G. & Pelletier, G. (2001) EMBO J. 20, 589–600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Doutriaux, M. P., Couteau, F., Bergounioux, C. & White, C. (1998) Mol. Gen. Genet. 257, 283–291. [DOI] [PubMed] [Google Scholar]
- 27.Alexander, M. P. (1969) Stain Technol. 44, 117–122. [DOI] [PubMed] [Google Scholar]
- 28.Marc, J., Granger, C. L., Brincat, J., Fisher, D. D., Kao, T., McCubbin, A. G. & Cyr, R. J. (1998) Plant Cell 10, 1927–1940. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Ross, K. J., Fransz, P. & Jones, G. H. (1996) Chromosome Res. 4, 507–516. [DOI] [PubMed] [Google Scholar]
- 30.Armstrong, S. J., Franklin, F. C. & Jones, G. H. (2001) J. Cell Sci. 114, 4207–4217. [DOI] [PubMed] [Google Scholar]
- 31.Smyth, D. R., Bowman, J. L. & Meyerowitz, E. M. (1990) Plant Cell 2, 755–767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Moffatt, B. A., McWhinnie, E. A., Agarwal, S. K. & Schaff, D. A. (1994) Gene 143, 211–216. [DOI] [PubMed] [Google Scholar]
- 33.Yong, W. D., Xu, Y. Y., Xu, W. Z., Wang, X., Li, N., Wu, J. S., Liang, T. B., Chong, K., Xu, Z. H., et al. (2003) Planta 217, 261–270. [DOI] [PubMed] [Google Scholar]
- 34.Owen, H. A. & Makaroff, C. A. (1995) Protoplasma 185, 7–21. [Google Scholar]
- 35.Rosso, M. G., Li, Y., Strizhov, N., Reiss, B., Dekker, K. & Weisshaar, B. (2003) Plant Mol. Biol. 53, 247–259. [DOI] [PubMed] [Google Scholar]
- 36.Siaud, N., Dray, E., Gy, I., Gerard, E., Takvorian, N. & Doutriaux, M. P. (2004) EMBO J. 23, 1392–1401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Sonoda, E., Takata, M., Yamashita, Y. M., Morrison, C. & Takeda, S. (2001) Proc. Natl. Acad. Sci. USA 98, 8388–8394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Lafarge, S. & Montane, M. H. (2003) Nucleic Acids Res. 31, 1148–1155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Alpi, A., Pasierbek, P., Gartner, A. & Loidl, J. (2003) Chromosoma 112, 6–16. [DOI] [PubMed] [Google Scholar]
- 40.Bleuyard, J. Y. & White, C. I. (2004) EMBO J. 23, 439–449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Marsh, T. C., Cole, E. S., Stuart, K. R., Campbell, C. & Romero, D. P. (2000) Genetics 154, 1587–1596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Caryl, A. P., Armstrong, S. J., Jones, G. H. & Franklin, F. C. (2000) Chromosoma 109, 62–71. [DOI] [PubMed] [Google Scholar]
- 43.Bai, X., Peirson, B. N., Dong, F., Xue, C. & Makaroff, C. A. (1999) Plant Cell 11, 417–430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Azumi, Y., Liu, D., Zhao, D., Li, W., Wang, G., Hu, Y. & Ma, H. (2002) EMBO J. 21, 3081–3095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Shinohara, A., Gasior, S., Ogawa, T., Kleckner, N. & Bishop, D. K. (1997) Genes Cells 2, 615–629. [DOI] [PubMed] [Google Scholar]
- 46.Pittman, D. L., Cobb, J., Schimenti, K. J., Wilson, L. A., Cooper, D. M., Brignull, E., Handel, M. A. & Schimenti, J. C. (1998) Mol. Cell 1, 697–705. [DOI] [PubMed] [Google Scholar]
- 47.Couteau, F., Belzile, F., Horlow, C., Grandjean, O., Vezon, D. & Doutriaux, M. P. (1999) Plant Cell 11, 1623–1634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Gallego, M. E., Jeanneau, M., Granier, F., Bouchez, D., Bechtold, N. & White, C. I. (2001) Plant J. 25, 31–41. [DOI] [PubMed] [Google Scholar]
- 49.Reiss, B. (2003) Int. Rev. Cytol. 228, 85–139. [DOI] [PubMed] [Google Scholar]
- 50.Pawlowski, W. P., Golubovskaya, I. N., Timofejeva, L., Meeley, R. B., Sheridan, W. F. & Cande, W. Z. (2004) Science 303, 89–92. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.