Abstract
Dermatofibrosarcoma protuberans (DFSP) is a rare cutaneous tumor with a tendency towards local recurrence. A 26-year-old woman presented with a 3 × 2 cm raised, purple-hued lesion on her left breast. Excisional biopsy identified the lesion as a DFSP. She underwent two additional operations to achieve widely clear margins. Operative excision is the primary treatment of dermatofibrosarcoma protuberans of the breast.
Dermatofibrosarcoma protuberans (DFSP) is a rare, locally aggressive cutaneous tumor. It is characterized by its slow, infiltrative growth and marked tendency towards local recurrence after surgical resection (1). The incidence rate is 4.2 to 4.5 cases per million persons per year in the United States (2). The most common location of DFSP is the trunk, with 25% of cases affecting the chest and shoulder areas (1). At least 40 case reports of DFSP in the breast have been reported, but less than half address the surgical management (3–17), which we describe here.
CASE DESCRIPTION
A 26-year-old Hispanic woman presented to the surgery clinic at Baylor University Medical Center at Dallas complaining of a left breast lesion. The lesion had been present since an incision and drainage procedure in the area approximately 8 years earlier. She noted that it had slowly grown over this period. The lesion was approximately 2 × 3 cm in size. It was located 5 cm from the nipple at the 7 o'clock position on the left breast. The lesion had a keloid-like appearance in that it was purple-hued, protuberant, and irregular. She had no ulceration of the lesion or nipple discharge. She was not taking any hormone therapy. There was no family history of breast, ovarian, skin, or other cancers. An ultrasound revealed a 3 cm lesion of the dermis corresponding with the area of concern.
An excisional biopsy of the lesion with gross margins using a radially oriented elliptical incision disclosed it to be DFSP (Figure 1). The lesion had a low mitotic index, but there was invasion into the underlying adipose tissue. All margins were positive for residual disease. She subsequently had wide local excision with a 2 cm margin of tissue taken circumferentially around the previous incision. Initial frozen section evaluation revealed that the deep margin was positive, so a new margin was taken. Frozen section analysis of the new margin was negative at the time of surgery, but on final analysis DFSP was seen at the lateral edge and another focus 2 mm away from the new deep margin. The patient underwent a third operation for wide local excision, again with 2 cm circumferential margins. All margins were clear by at least 1 cm on histological evaluation. The patient has done well postoperatively with a satisfactory cosmetic result (Figure 2).
Figure 1.

Histologic appearance of the lesion consistent with dermatofibrosarcoma protuberans.
Figure 2.
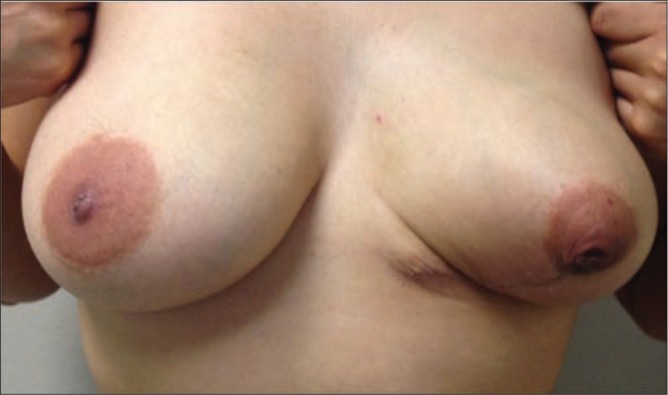
Postsurgical appearance following wide local excision and primary closure.
DISCUSSION
DFSP is known to have a high rate of recurrence after surgical excision. The propensity for this tumor to develop subclinical, tentacle-like extensions of tumor cells into the underlying deep tissue makes complete excision difficult. Prior to the 1980s, recurrence rates for DFSP after local excision ranged from 10% to 60%. Pooled data from the last 20 years reveal that the local recurrence rate now ranges from 0% to 21%, with more recent studies advocating the use of wide surgical margins (18). “Wide local excision,” however, has not been well defined. The National Comprehensive Cancer Network guidelines recommend excision of DFSP with 2 to 4 cm margins including the deep investing fascia of muscle, which is in line with recommendations for sarcomas in general. Data from the application of Mohs surgery for DFSP have shown that 75% of tumors are cleared with 1 cm margins, 80% with 1.5 cm margins, 85% with 2 cm margins, and 95% with 2.5 cm margins. Tumors that were <2 cm are usually cleared with a 1.5 cm margin, while 2.5 cm is required to clear tumors >2 cm (19).
While it would be ideal to capture all tissue down through the investing fascia of the muscle, this might not be practical in every case of DFSP of the breast. Given that the breast is a unique area of the body where intervening tissue lies between the subcutaneous adipose tissue and the underlying muscle fascia, this begs the question as to whether the additional tissue resection is necessary. In our case, approximately 4 cm of deep margin was excised without encountering the fascia of the pectoralis major, and a negative margin was accepted as an adequate wide local excision.
The optimal surgical treatment to achieve negative margins can be variable based on tumor size and location on the breast. A review of the literature revealed a variety of surgical approaches to address DFSP of the breast. One patient underwent a mastectomy, one patient underwent a wide local excision with concomitant breast reduction, three patients underwent a wide local excision with flap reconstruction, one patient underwent a wide local excision with skin graft, and nine patients underwent wide local excision with primary closure (Table 1, 3–17). The reconstructive challenge in DFSP of the breast is to balance the oncologic principles of adequate resection with the cosmetic goals of acceptable symmetry and contour. In our case, we were able to achieve adequate cosmetic results with a wide local excision and primary closure, but this might not be feasible in all patients. The National Comprehensive Cancer Network recommends that any reconstruction that involves extensive undermining or tissue movement should be delayed until histologic margins are verified (2). Due to the inaccuracy of intraoperative pathologic assessments with frozen section, staged reconstruction should be considered.
Table 1.
Cases of dermatofibrosarcoma protuberans of the breast addressing surgical management
| First author (ref) | Age (yr) | Size of lesion (cm) | Recurrence or primary | Surgical management | Margin size (cm) | Deep fascia excision |
|---|---|---|---|---|---|---|
| Yeniay (3) | 48 | 10 | Recurrence | Mastectomy | Unknown | Yes |
| Cavus¸og˘lu (4) | 26 | 2 | Primary | Wide local excision with breast reduction pattern | 5 | Yes |
| Swan (5) | 39 | 5 | Recurrence | Wide local excision with latissimus dorsi flap | 5 | Yes |
| Dagregorio (6) | 66 | 21 | Primary | Wide local excision with reverse abdominal plasty | 5 | Yes |
| Castle (7) | 30 | Unknown | Primary | Wide local excision with pectoralis flap | Unknown | Unknown |
| McGregor (8) | 33 | Unknown | Primary | Wide local excision with skin graft | “Wide” | Yes |
| Dragoumis (9) | 48 | 2 | Recurrence | Wide local excision with primary closure | 3 | No |
| Park (10) | 28 | 3.8 | Primary | Wide local excision with primary closure | 3 | No |
| Kamiya (11) | 75 | 1.2 | Primary | Wide local excision with primary closure | 3 | Yes |
| Cottier (12) | 75 | 1.8 | Primary | Wide local excision with primary closure | 5 | Yes |
| Mikhail (13) | 19 | <3 | Primary | Wide local excision with primary closure | 2–3 | Unknown |
| Jiang (14) | 26 | 3.2 | Primary | Wide local excision with primary closure | 3 | Unknown |
| Koh (15) | 42 | 5.5 | Primary | Wide local excision with primary closure | Unknown | Unknown |
| Lin (16) | 49 | 2.3 | Primary | Wide local excision with primary closure | Unknown | Unknown |
| Roy (17) | 102 | 8 | Recurrence | Wide local excision with primary closure | Unknown | Unknown |
References
- 1.Gloster HM., Jr Dermatofibrosarcoma protuberans. J Am Acad Dermatol. 1996;35(3 Pt 1):355–374. doi: 10.1016/s0190-9622(96)90597-6. [DOI] [PubMed] [Google Scholar]
- 2.Miller SJ. Alam M. Andersen JS. Berg D. Bichakjian CK. Bowen GM. Cheney RT. Glass LF. Grekin RC. Ho AL. Kessinger A. Liegeois N. Lydiatt DD. Michalski J. Morrison WH. Nehal KS. Nelson KC. Nghiem P. Olencki T. Perlis CS. Shaha AR. Tuli M. Urist MM. Wang LC. Zic JA. National Comprehensive Cancer Network. Dermatofibrosarcoma protuberans. J Natl Compr Canc Netw. 2012;10(3):312–318. doi: 10.6004/jnccn.2012.0032. [DOI] [PubMed] [Google Scholar]
- 3.Yeniay L. Unalp O. Sezak M. Yilmaz R. Dermatofibrosarcoma protuberans of the breast. Breast J. 2012;18(5):493–494. doi: 10.1111/j.1524-4741.2012.01282.x. [DOI] [PubMed] [Google Scholar]
- 4.Cavuşoğlu T. Yavuzer R. Tuncer S. Dermatofibrosarcoma protuberans of the breast. Aesthetic Plast Surg. 2003;27(2):104–106. doi: 10.1007/s00266-003-0107-9. [DOI] [PubMed] [Google Scholar]
- 5.Swan MC. Banwell PE. Hollowood K. Goodacre TE. Late recurrence of dermatofibrosarcoma protuberans in the female breast: a case report. Br J Plast Surg. 2005;58(1):84–87. doi: 10.1016/j.bjps.2004.06.017. [DOI] [PubMed] [Google Scholar]
- 6.Dagregorio G. Darsonval V. Aesthetic surgery techniques after excision of dermatofibrosarcoma protuberans: a case report. Br J Plast Surg. 2005;58(4):556–560. doi: 10.1016/j.bjps.2004.10.008. [DOI] [PubMed] [Google Scholar]
- 7.Castle KO. Guadagnolo BA. Tsai CJ. Feig BW. Zagars GK. Dermatofibrosarcoma protuberans: long-term outcomes of 53 patients treated with conservative surgery and radiation therapy. Int J Radiat Oncol Biol Phys. 2013;86(3):585–590. doi: 10.1016/j.ijrobp.2013.02.024. [DOI] [PubMed] [Google Scholar]
- 8.McGregor JK. Role of surgery in the management of dermatofibrosarcoma protuberans. Ann Surg. 1961;154(2):255–258. doi: 10.1097/00000658-196108000-00010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Dragoumis DM. Katsohi LA. Amplianitis IK. Tsiftsoglou AP. Late local recurrence of dermatofibrosarcoma protuberans in the skin of female breast. World J Surg Oncol. 2010;8(1):48. doi: 10.1186/1477-7819-8-48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Park TH. Seo SW. Kim JK. Chang CH. Reconstructive challenge of dermatofibrosarcoma protuberans in the female breast. World J Surg Oncol. 2011;9(1):1. doi: 10.1186/1477-7819-9-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kamiya T. Saga K. Kaneko R. Ono I. Kawada M. Maeda Y. Postradiation dermatofibrosarcoma protuberans. Acta Derm Venereol. 2006;86(2):152–153. doi: 10.2340/00015555-0012. [DOI] [PubMed] [Google Scholar]
- 12.Cottier O. Fiche M. Meuwly JY. Delaloye JF. Dermatofibrosarcoma presenting as a nodule in the breast of a 75-year-old woman: a case report. J Med Case Reports. 2011;5(1):503. doi: 10.1186/1752-1947-5-503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Mikhail GR. Lynn BH. Dermatofibrosarcoma protuberans. J Dermatol Surg Oncol. 1978;4(1):81–84. doi: 10.1111/j.1524-4725.1978.tb00384.x. [DOI] [PubMed] [Google Scholar]
- 14.Jiang JQ. Huang Z. Wang LH. Shen SD. Lu H. Dermatofibrosarcoma protuberans of the breast: a case report. Oncol Lett. 2014;8(3):1202–1204. doi: 10.3892/ol.2014.2291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Koh CK. Ko CB. Bury HP. Wyatt EH. Dermatofibrosarcoma protuberans. Int J Dermatol. 1995;34(4):256–260. doi: 10.1111/j.1365-4362.1995.tb01591.x. [DOI] [PubMed] [Google Scholar]
- 16.Lin JY. Sheen-Chen SM. Hsu W. Huang CC. Ko SF. Eng HL. Dermatofibrosarcoma protuberans of the breast. Tumori. 2008;94(6):861–863. doi: 10.1177/030089160809400616. [DOI] [PubMed] [Google Scholar]
- 17.Roy AD. Nishant K. Joshi D. Painful late recurrence of dermatofibrosarcoma protuberans of breast in a centurian female. J Clin Diagn Res. 2013;7(10):2278–2279. doi: 10.7860/JCDR/2013/5808.3494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Bogucki B. Neuhaus I. Hurst EA. Dermatofibrosarcoma protuberans: a review of the literature. Dermatol Surg. 2012;38(4):537–551. doi: 10.1111/j.1524-4725.2011.02292.x. [DOI] [PubMed] [Google Scholar]
- 19.Parker TL. Zitelli JA. Surgical margins for excision of dermatofibrosarcoma protuberans. J Am Acad Dermatol. 1995;32(2 Pt 1):233–236. doi: 10.1016/0190-9622(95)90132-9. [DOI] [PubMed] [Google Scholar]


