Abstract
INTRODUCTION
Angelman syndrome (AS) is a neurodevelopmental disorder caused by deficiency of maternally inherited UBE3A, an ubiquitin E3 ligase. Despite recent progress in understanding the mechanism underlying UBE3A imprinting, there is no effective treatment. Further investigation of the roles played by UBE3A in the central nervous system (CNS) is needed for developing effective therapies.
AREA COVERED
This review covers the literature related to genetic classifications of AS, recent discoveries regarding the regulation of UBE3A imprinting, alterations in cell signaling in various brain regions, and potential therapeutic approaches. Since a large proportion of AS patients exhibit comorbid autism spectrum disorder (ASD), potential common molecular bases are discussed.
EXPERT OPINION
Advances in understanding UBE3A imprinting provide a unique opportunity to induce paternal UBE3A expression, thus targeting the syndrome at its “root.” However, such efforts have yielded less-than-expected rescue effects in AS mouse models, raising the concern that activation of paternal UBE3A after a critical period cannot correct all the CNS defects that developed in a UBE3A-deficient environment. On the other hand, targeting abnormal downstream cell signaling pathways has provided promising rescue effects in preclinical research. Thus, combined reinstatement of paternal UBE3A expression with targeting abnormal signaling pathways should provide better therapeutic effects.
1. Introduction
Angelman syndrome (AS) is a genetic neurodevelopmental disorder characterized by severe developmental delay, language and cognition deficits, motor dysfunction [1, 2], unusually happy demeanor and, in many AS patients, seizure activity [1, 3, 4] and autism-like behavior. The prevalence of AS is estimated at 1 in 10,000 to 20,000 live births [5, 6]. AS is caused by deficient expression of the maternally inherited UBE3A gene in neurons [7–9]. In most tissues, as well as in non-neuronal cells in the brain, UBE3A is expressed from both alleles, even though the maternal copy is favored [10]. In neurons, however, only the maternally inherited UBE3A is expressed, while the paternally inherited copy is silenced [11–13]. The imprinted expression of UBE3A is most likely regulated by a long noncoding antisense RNA transcript (UBE3A-ATS), which is part of a larger SNURF-SNRPN transcript [14–16]. The mechanism by which UBE3A-ATS blocks UBE3A transcription is unknown, but may involve histone-mediated repression, transcriptional interference, or repressive three-dimensional chromatin structure [16–19]. Maternal UBE3A deficiency has been attributed to four genetic etiologies [9, 20]: deletions of the maternal 15q11–q13 region (class I, approximately 70% of cases), paternal uniparental disomy of chromosome 15 (class II, 5%), imprinting defects (class III, 5%), and mutations in UBE3A (class IV, 10%). Another small group of clinically diagnosed AS patients (class V, 10%) presents with no detectable UBE3A abnormality. Some cases in this group are now recognized as Angelman-like syndromes with newly defined genetic causes [21] (discussed later).
The UBE3A gene encodes a protein, which is also known as E6-associated protein (E6AP), the founding member of the HECT (homologous to E6AP carboxy terminus) domain-containing E3 ligase family [22]. In humans, at least 29 members of the HECT ligase family have been identified and the shared HECT domain comprises a highly conserved sequence of 350 amino acid residues [23, 24]. This domain is responsible for E2~ubiquitin thioester binding and subsequent targeting for protein conjugation. The function of the amino-terminal domain is not clear, although it has been proposed that this domain may be involved in recruiting target protein substrates [23, 24]. Residue Cys820 within the HECT domain is the active site, which forms a high energy thioester bond with ubiquitin prior to transferring it to specific substrate proteins [23]. The UBE3A mRNA has at least seven reported variants, which arise from alternative splicing mostly within the 5′ end of the transcript [12]. Three of them produce three UBE3A isoforms, but the functional significance and tissue specific localization of these isoforms remain elusive. UBE3A functions both as an E3 ligase in the ubiquitin-proteasome pathway [25–27], and as a transcriptional co-activator for steroid hormone receptors [28]. It is not yet known which of these function(s) is/are responsible for the phenotypic manifestations of Angelman syndrome.
Mice with maternal Ube3a deficiency (AS mice) exhibit several features of the human disease, including reduced brain size, abnormal electroencephalogram, learning and memory deficits, motor dysfunction [29–31], as well as impairment in long-term potentiation (LTP) [30, 32–34]. Results from studying AS mice have begun to shed light on the pathophysiology of Angelman syndrome and reveal potential therapeutic targets, which are the focus of this review. There are several excellent reviews regarding the clinical and genetic aspects of Angelman syndrome, which will not be addressed here; interested readers are strongly encouraged to consult them for further discussion of those aspects (e.g. [35–40]).
2. Pathophysiology of AS
2.1. Alterations in synaptic plasticity in AS mice
In addition to smaller brains, AS mice exhibit reduced number and size of dendritic spines in cerebellar Purkinje neurons [41, 42], hippocampal pyramidal neurons [41], and visual cortical neurons [43]. Although baseline synaptic transmission is normal in hippocampus of AS mice, LTP is impaired [30, 32, 44], while mGlu5 receptor-dependent long-term depression (LTD) is enhanced [45]. In the visual cortex, AS mice exhibit deficits in both LTP and LTD, as well as decreased mEPSCs without clearly defined mechanisms [43, 46]. On the other hand, multiple molecular mechanisms have been proposed to account for synaptic plasticity impairment in hippocampus (Table I). Earlier experiments showed that, while the amount of CaMKII in hippocampus was not altered in AS mice, phosphorylation of the kinase at Thr286 (the autophosphorylation site) and Thr305 (the inhibitory phosphorylation site) was significantly increased [44]. Changes in CaMKII phosphorylation were associated with reduced activity, suggesting that changes at the inhibitory phosphorylation site dominated [44]. Subsequently, the same group showed that cross-breeding AS mice with mice expressing CaMKII-T305V, which prevents inhibitory phosphorylation, increased CaMKII activity and rescued behavioral deficits and LTP impairment [33], confirming that inhibitory phosphorylation of CaMKII was critically involved in AS pathogenesis. However, how Ube3a deficiency leads to increased CaMKII inhibitory phosphorylation remains unknown.
Table I.
Alterations in synaptic proteins in AS mice
| Synaptic protein | Changes in AS mice | Effects on LTP/LTD |
|---|---|---|
| CaMKII | Increased inhibitory phosphorylation | Impaired LTP induction and consolidation |
| Arc | Increased Arc reduces synaptic AMPARs | Impaired LTP consolidation |
| TrkB | Increased Arc interrupts PSD95 and TrkB association, and decreases BDNF signaling | Impaired LTP induction and maintenance |
| SK2 channel | Increased synaptic level | Impaired LTP, enhanced NMDAR-LTD, impaired memory |
| mGlu5-Homer 1b/c | Increased coupling of mGlu5 and Homer1b/c | Enhanced mGluR-LTD |
The immediate early gene product Arc (activity-regulated cytoskeletal protein) represents the first identified Ube3a target protein in the mammalian brain (Table I). Arc has been shown to be ubiquitinated and degraded in an Ube3a-dependent manner [25]. However, a recent study argues that Ube3a regulates Arc at the transcriptional level rather than through direct ubiquitination [47]. Nevertheless, increased Arc levels could account for LTP impairment, since Arc has been shown to promote AMPA receptor (AMPAR) internalization and reduce AMPAR-mediated synaptic transmission [48–50]. Brain-derived neurotrophic factor (BDNF)-induced signaling through its receptor, TrkB, is known to be essential for both LTP induction and maintenance; further, the association of TrkB with the postsynaptic density protein-95 (PSD-95) is critical for intact BDNF signaling [51]. In AS mice, increased Arc levels interrupt BDNF-TrkB signaling through its association with PSD-95, which may contribute to the observed LTP impairment [51].
Recent research has shown that LTP in field CA1 of hippocampus is also modulated by small conductance calcium-activated potassium channels (SK2) [52, 53], which are opened upon N-methyl-D-aspartate (NMDA) receptor activation and then repolarize the membrane, thereby terminating NMDA receptor function [54]. SK channels participate in various CNS functions, from regulating neuronal intrinsic excitability to network rhythmic activity and higher brain functions [55–57]. It has been recently shown that UBE3A ubiquitinates SK2 in its C-terminal domain, which results in internalization of the channels and decreased synaptic levels [58]. In AS mice, increased postsynaptic SK2 levels result in decreased NMDA receptor activation, thereby impairing hippocampal long-term synaptic plasticity. Impairments in both synaptic plasticity and fear conditioning memory in AS mice were significantly ameliorated by blocking SK2. These results provide a novel mechanism by which UBE3A directly influences cognitive function. LTD represents another form of synaptic plasticity, which involves removal/endocytosis of AMPARs at synapses [reviewed in [59–61]]. It has recently been shown that metabotropic glutamate receptor mGlu5-mediated LTD is enhanced in field CA1 of hippocampal slices from AS mice, as compared to wild-type (WT) mice [45]. Although Arc has been shown to be involved in mGluR-dependent LTD [62], Pignatelli et al. found that neither basal levels of expression nor LTD-associated increase in Arc levels differed between AS and WT mice. On the other hand, they found that enhanced LTD in AS mice was associated with enhanced coupling of mGlu5 with Homer proteins, which target group I mGluR to the postsynaptic density, thereby enhancing their signaling potency [63, 64]. Interestingly, in their recent paper Sun et al. showed that low-frequency-induced LTD was enhanced in hippocampal slices from AS mice, which was blocked by the SK2 channel blocker, apamin [58]. Furthermore, the rescue effects of apamin on both LTP impairment and LTD enhancement were NMDAR-dependent. These results suggest that by regulating NMDAR activity, increased synaptic levels of SK2 channels can influence the response of hippocampal circuits to different stimulation patterns and thus affect different cognitive functions, e.g. both LTP- and LTD-dependent learning behaviors.
2.2. Motor dysfunction and potential mechanisms
2.2.1. Dysfunction of dopaminergic and GABAergic neurons
In contrast to hippocampus-dependent learning and memory and synaptic plasticity, motor dysfunction has not been widely studied, even though tremor, gait disturbance, and ataxia are major symptoms associated with AS and are also present in AS mice. An earlier study found that motor impairment in AS mice was associated with a loss of dopaminergic neurons [65]. However, a more recent study reported elevated levels of dopamine in the striatum, midbrain and frontal cortex in both AS mice and mice with Ube3a duplication [66]. It has also been reported that AS mice exhibit increased dopamine release in the mesolimbic pathway and decreased dopamine release in the nigrostriatal pathway [67], which highlights the current uncertainty of the effect of UBE3A deficiency on dopaminergic signaling.
The cerebellum is essential for coordinating purposeful and smooth movement. In autopsy studies of a 21-year-old AS woman, there was marked cerebellar atrophy with loss of Purkinje and granule cells and extensive Bergmann gliosis [68]. Likewise, dendritic spine dystrophy is also prominent in hippocampus and cerebellum of AS mice [41, 42]. Recently, Egawa et al. showed that tonic inhibition is reduced in cerebellar granule cells in AS mice, possibly due to increased levels of γ-aminobutyric acid (GABA) transporter 1 (GAT1) [69]. The same group also demonstrated that treatment of AS mice with low doses of the selective extrasynaptic GABAA receptor agonist 4,5,6,7-tetrahydroisothiazolo-[5,4-c]pyridin-3-ol (THIP) normalized firing properties of Purkinje neurons and improved motor function [69].
2.2.2. Abnormal mTOR signaling in cerebellum of AS mice
Abnormal signaling of the mechanistic target of rapamycin (mTOR) pathway has been recently reported to be responsible for the dendritic spine abnormalities in Purkinje neurons and motor dysfunction observed in AS mice [42]. Dysregulation of the mTOR pathway has also been associated with fragile X and Rett syndromes, and tuberous sclerosis complex (TSC) (see [70, 71] for recent reviews). Through its integration of signals from trophic factors, NMDARs, mGluRs, and energy status, mTOR plays critical roles in both brain development and synaptic plasticity [72–74]. mTOR exists in two distinct functional complexes: mTORC1 coupled to raptor and responding to a number of signals, including growth factors, amino acids, oxygen, and energy status; and mTORC2 coupled to rictor and mainly stimulated by growth factors [75]. It has been recently shown that in cerebellum of AS mice, mTORC1 activity is increased while mTORC2 activity is decreased, and that mTORC2 inhibition is due to S6K1-mediated inhibitory phosphorylation of its regulator, rictor [42]. The functional significance of this mTORC1/mTORC2 dysregulation is underscored by the fact that rapamycin treatment not only normalized mTORC1 and mTORC2 activities, but also improved dendritic spine morphology of Purkinje neurons and motor function in AS mice [42].
While the roles of mTORC1 in cell growth and proliferation have been well characterized [75, 76], those of mTORC2 are not as clear. Emerging experimental data indicate that mTORC2 may play critical roles in regulating the actin network and cell motility [77, 78]. Early studies in yeast showed that mTORC2 regulates actin cytoskeleton through activation of Rho family GTPases [79]. Studies with mammalian cells showed that the same pathway is also responsible for mTORC2-mediated regulation of actin filaments [77, 80]. Additionally, it has been shown that mTORC2 regulates the actin cytoskeleton through PKC and Akt activation, although the effects of Akt on cell motility may be cell and tissue specific [81]. Akt has been additionally shown to play important roles in neuronal survival and synaptic plasticity [82]. Deletion of the mTORC2 component, rictor, in the CNS results in severe microcephaly with decreased neuronal size, altered neurite organization, and motor function impairment [83]. Furthermore, selective knockout of rictor in Purkinje neurons leads to abnormal morphology of these neurons and motor dysfunction associated with loss of activation of Akt, PKC, and SGK1 (serum/glucocorticoid regulated kinase 1), without effects on mTORC1 activity [83]. Conditional deletion of rictor in postnatal murine forebrain reduced mTORC2 activity and actin polymerization in a Rac1/PAK/cofilin-dependent manner in hippocampus [84]. It is thus conceivable that decreased mTORC2 activity together with increased mTORC1 activity are at least partially responsible for the reported immature dendritic spine phenotype [41] and the decreased spine density of Purkinje neurons in AS mice [42]. On the other hand, mTORC1-induced protein synthesis of RhoA has also been shown to increase actin polymerization and LTP consolidation in hippocampus [85]. Whether mTORC1 and mTORC2 differentially regulate actin polymerization in different brain regions remains to be determined. Nevertheless, these studies indicate that imbalanced mTORC1 and mTORC2 activation contributes, at least in part, to motor dysfunction in AS. It is noteworthy that mTOR activity has been shown to be increased in fragile X and Down syndromes, and TSC, but decreased in Rett syndrome (reviewed in [70]), although these syndromes all show varying degrees of intellectual disability. These results warrant further investigation of the regulation and function of mTOR in these disorders, especially with respect to the different roles of mTORC1 and mTORC2 and their downstream signaling cascades.
2.2.3. AS and Angelman-like syndrome: abnormal lysosome-dependent mTOR activation as the molecular link?
Interestingly, recent research indicates that at least some of the class V AS cases can be attributed to mutations in solute carrier family 9 isoform 6 (SLC9A6) [86], which encodes NHE6, a member of the endosomal subtype of Na+/H+ exchangers (NHEs). NHEs have also been implicated in other neurodevelopmental and neuropsychiatric disorders, including autism, X-linked intellectual disability (XLID), epilepsy, addiction, and attention deficit hyperactivity disorder (ADHD) (reviewed in [87]). Slc9a6 knockout in mice results in lysosomal dysfunction in neurons in the basolateral nuclei of the amygdala, CA3 and CA4 regions and dentate gyrus of the hippocampus, and in some areas of the cerebral cortex [88]. It also leads to axonal dystrophy and Purkinje cell death in the cerebellum. NHE6 regulates endosomal/lysosomal function by transporting Na+ in and H+ out of these compartments. It also regulates endosomal/lysosomal volume and the rate of surface receptor recycling [89]. Endosomal/lysosomal pH is precisely tuned by a combination of acidification through the proton pump V-ATPase and alkalization via NHE6; loss-of-function mutations of NHE6 would cause hyperacidification of the endosomal lumen [87]. Of note, emerging evidence has demonstrated that mTORC1 activity is regulated by lysosomal amino acids, possibly through V-ATPase [90] and another lysosomal membrane protein, SLC38A9 [91–93]. Whether abnormal mTORC1 activation is the molecular converging point for AS and Angelman-like syndrome condition remains an interesting question.
2.3. Additional changes in neuronal properties
2.3.1. Abnormal cell contact signaling
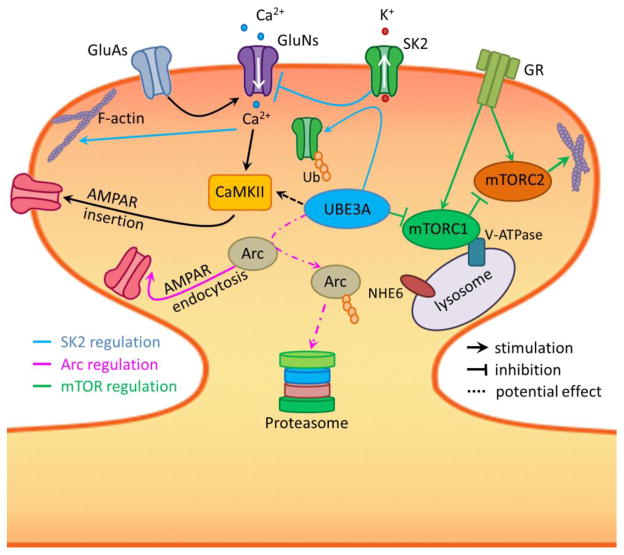
Another recently identified potential brain Ube3a substrate is a guanine nucleotide exchange factor (GEF) of the Rho family small GTPases, Ephexin5, which negatively regulates excitatory synapse development by interrupting EphB/EphrinB signaling (reviewed in [94]). Ephexin5 levels are increased in AS mouse brains, which may impede EphB receptor signaling and maturation of excitatory synapses [26]. However, whether increased Ephexin5 contributes to AS pathogenesis remains to be determined. Signaling of neuregulin 1 through the receptor tyrosine kinase ErbB4 is also enhanced in AS mice, and ErbB inhibitors reverse LTP impairment and memory deficits in AS mice [34]. Neuregulin-1/ErbB4 has been shown to activate the PI3K-mTOR pathway [95], but whether this is the case in AS mice requires further studies. Figure 1 summarizes the potential alterations in synaptic proteins and signaling pathways that can impair synaptic plasticity and dendritic spine maturation.
Figure 1. Potential molecular basis for synaptic plasticity impairment and abnormal spine morphology in AS mice.
Under normal conditions, AMPAR (GluA) activation-induced membrane depolarization leads to NMDAR (GluN) channel opening and Ca2+ influx. Increased intracellular Ca2+ levels trigger several mechanisms, including CaMKII activation, AMPAR insertion, and actin network reorganization. Ube3a activation results in degradation of Arc and endocytosis of SK2 channels, facilitating LTP induction and enhancing consolidation. Ube3a also regulates mTORC1 and mTORC2 activation, leading to balanced protein synthesis and cytoskeletal formation. Additionally, lysosomes provide a platform for nutrient-induced mTORC1 activation through V-ATPase and other regulatory proteins. NHE6 regulates lysosomal pH and its loss of function results in Angelman-like syndrome. GR: growth factor receptors.
2.3.2. Neuronal intrinsic excitability and excitation/inhibition imbalance
CA1 pyramidal neurons in AS mice exhibit increased expression of the α1 subunit of Na/K-ATPase (α1-NaKA), the voltage-gated sodium channel NaV1.6, and the axon initial segment (AIS) protein ankyrin-G, which could contribute to alterations in intrinsic membrane properties [96]. Genetic manipulation experiments have shown that these changes as well as the deficits in LTP and hippocampus-dependent memory are prevented by deletion of α1-NaKA, suggesting that increased expression of α1-NaKA during early development plays an important role in AS pathogenesis [97]. However, how Ube3a deficiency could result in increased α1-NaKA expression is currently unknown.
Imbalance between excitation and inhibition has been implicated in autism spectrum disorders, schizophrenia and other neurological disorders. Wallace et al. recently showed that in the visual cortex of AS mice, although excitatory and inhibitory synaptic inputs onto inhibitory interneurons are mostly normal, the inhibitory innervation of neocortical pyramidal neurons is severely decreased, possibly due to defective presynaptic vesicle cycling in interneurons [98]. Although deficits in inhibitory transmission could result in excitatory/inhibitory imbalance at the cellular and circuit levels, whether this is the case in AS remains to be determined.
3. Development of therapeutic approaches
3.1 Un-silencing the paternal UBE3A gene
Since the majority of AS cases are caused by UBE3A deficiency, several groups have attempted to restore UBE3A expression by direct gene therapy or by un-silencing the paternal allele. Injection of recombinant adeno-associated virus (AAV) carrying the mouse Ube3a into the hippocampus of AS mice restored local Ube3a expression and improved hippocampus-dependent learning and memory [99]. However, the viral vectors showed limited distribution beyond the hippocampus and there was no effect on motor dysfunction. Another concern of this approach is the precise control of UBE3A expression, since high UBE3A levels are a risk factor for ASD [100–102].
Several lines of evidence have indicated that paternal UBE3A silencing in both neurons and induced Pluripotent Stem Cells (iPSC) from AS patients is due to the expression of a long non-coding antisense RNA, UBE3A-ATS [14–16, 103]. Recent research with AS mice has shown that Ube3a-ATS is an atypical RNAPII transcript, which functions to suppress paternal Ube3a expression [18]. Therefore, suppression of UBE3A-ATS expression becomes an attractive approach to reactivate the paternal allele. Earlier attempts used dietary supplements to increase DNA methylation, based on the rationale that increasing methylation might reduce UBE3A-ATS expression, thereby increasing UBE3A expression from the paternal gene; however, these attempts were not successful [104, 105]. It was then shown that the topoisomerase I inhibitor, topotecan, inhibited transcriptional progression of Ube3a-ATS, leading to un-silencing of the paternal copy of Ube3a in AS mice [106]. Since topotecan is an FDA-approved anti-cancer drug, the results gave hope for rapidly developing the drug as a potential therapy for AS. However, topotecan’s lack of specificity and toxicity have impeded further advancement of the drug as an AS treatment.
Meanwhile, genetic truncation of Ube3a-ATS transcription has also been shown to increase paternal Ube3a expression and improve memory performance and motor skills, which conceptually validates the idea of targeting UBE3A-ATS [107]. To develop a more therapeutically relevant intervention, Meng et al. administered antisense oligonucleotides (ASOs) against Ube3a-ATS in AS mice via intracerebroventricular (ICV) injection. The ASO treatment was generally well tolerated, reduced Ube3a-ATS expression, and produced sustained expression of paternal Ube3a (up to four months), although the restoration was only partial [108]. Behavioral studies performed four weeks after ASO treatment showed that memory impairment in the fear-conditioning paradigm was significantly improved, while abnormal open field behavior was not rescued, nor were alterations in marble burying and accelerating rotarod performance. The authors postulated that a complete phenotypic reversal might require treatment before a critical developmental window, a longer recovery time, or a higher UBE3A induction level. However, this developmental window may be shorter than expected. Although AS symptoms generally become noticeable at 6- to 12-months of age in humans, by this age the brain has already established connections under UBE3A-deficient conditions. It is also possible that, by this age, compensatory epigenetic and biochemical modifications have taken place, limiting the efficacy of this therapeutic approach.
The fact that genetic truncation of UBE3A-ATS more effectively rescues phenotypic defects [107] than ASOs treatment in AS mice has two therapeutic implications: i) long-lasting unsilencing is required; and ii) early, prenatal, UBE3A expression is critical. This notion is supported by a recent study, which systematically investigated the effects of reinstating Ube3a expression during distinct neurodevelopmental windows. The study found that Ube3a reinstatement only during a very early time window, in the embryonic stage, fully rescued AS-relevant phenotypes. Ube3a reinstatement during adolescence was semi-effective, while during adulthood only hippocampal synaptic plasticity was restored [109]. Thus, targeting UBE3A reinstatement should be attempted as early as possible during postnatal development and even so, this type of treatment alone may not be sufficient. It is noteworthy that a recent report indicates that UBE3A activity is reduced by PKA-mediated phosphorylation and disruption of this regulation by an autism-linked mutation in UBE3A results in enhanced UBE3A activity and excessive dendritic spine development [110]. These new results warrant further research on the regulation of UBE3A activity in addition to its expression levels.
3.2 Mechanism-based therapies directed at functional corrections
In parallel to attempts at increasing UBE3A expression, several groups have explored alternative approaches to rescue synaptic plasticity and cognitive functions. These alternative approaches are based on findings related to the molecular underpinnings of abnormal synaptic plasticity in AS mice and to evidence that autism-related monogenetic syndromes share alterations in a common set of synaptic mechanisms (Table II). It was first reported that cross-breeding AS mice with mice carrying mutations preventing the inhibitory phosphorylation of CaMKII could correct abnormal CaMKII inhibition, seizure propensity, and deficits in motor performance and hippocampus-dependent learning and plasticity found in AS mice [33]. Although increases in Arc have been hypothesized as a cause of synaptic plasticity impairment through the stimulation of AMPAR internalization [25, 47], there is no information regarding whether direct down-regulation of Arc ameliorates synaptic plasticity or learning and motor function deficits. However, increased association of Arc with PSD-95 was also found in AS mice, which perturbed BDNF-TrkB signaling. Treatment with a PSD-95 peptidomimetic compound, CN2097, increased BDNF signaling and rescued LTP in hippocampus of AS mice [51].
Table II.
Effects of targeting different pathways on phenotypic manifestations of AS-related disorders.
| Target proteins | Manipulation/Drug | Effects |
|---|---|---|
| CaMKII | Genetic deletion of inhibitory phosphorylation | Rescues LTP, hippocampus-dependent learning and motor function |
| TrkB | Peptidomimetic inhibits PSD-95/Arc interaction | Rescues hippocampal LTP |
| AMPAR | Ampakine enhances AMPAR activation, BDNF release, and actin polymerization | Rescues hippocampal LTP, spine actin polymerization, and hippocampus-dependent learning |
| SK2 channel | Channel blocker apamin | Rescues hippocampal LTP, LTD, spine actin polymerization, and hippocampus-dependent learning |
| mTOR | Rapamycin inhibits mTORC1 and disinhibits mTORC2 | Rescues motor function |
| GAT1 | THIP, a selective extrasynaptic GABAA receptor agonist | Rescues Purkinje neuron firing and reduces cerebellar ataxia |
| ErbB | ErbB inhibitors | Rescues hippocampal LTP and hippocampus-dependent learning |
Multiple lines of evidence indicate that the plasticity defects in several developmental disorders reflect a failure of activity-driven signaling pathways that are critical for the reorganization of the spine cytoskeleton required for consolidation of LTP and memory (reviewed in [111]). In particular, LTP impairment in AS mice is associated with defects in activity-dependent actin polymerization, which has been shown to be critical for LTP stabilization [112–114]. This result indicates that AS shares a specific spine dysfunction with rodent models of two other human conditions associated with memory and cognitive deficits: low estrogen levels and early stage Huntington’s disease [115, 116]. In vivo treatment of AS mice with ampakines, positive AMPAR modulators known to increase BDNF release [117, 118], not only rescued LTP and actin polymerization, but also significantly improved hippocampus-dependent learning behavior [32]. Ampakine treatment has also been shown to improve social interactions in the BTBR mouse model of autism [119], and improve respiratory function in a mouse model of Rett syndrome [120]. Although BDNF application reverses LTP impairment in fragile X syndrome [121] and ampakines increase BDNF release, it has recently been reported in a phase II clinical trial, that the ampakine CX516 produced minimal beneficial effects, possibly due to the low dose tested [122]. It is noteworthy that blocking SK2 channels not only rescued LTP and normalized LTD, but also reactivated plasticity-induced actin polymerization [58]. Systemic injection of a SK2 channel blocker immediately before training also rescued fear conditioning learning. As mentioned earlier, administration of low doses of THIP, a selective extrasynaptic GABAA receptor agonist, rescued Purkinje neuron firing patterns and improved motor performance [69].
Since dysregulation of mTOR pathways has been implicated in several monogenetic neurodevelopmental disorders, manipulation of this pathway has been attempted for these disorders. In mouse models of TSC, treatment with rapamycin, an inhibitor of mTOR, prolonged survival and prevented epilepsy [123–125]. Rapamycin treatment also rescued hippocampal LTP and behavioral deficits in Tsc2(+/−) mice [126]. Further research has shown that rapamycin treatment has beneficial effects in ASD associated with mutations in PTEN (phosphatase and tensin homolog deleted on chromosome ten), which regulates PI3K (phosphoinositide 3-kinase)/Akt signaling upstream of TSC/mTOR (reviewed in [71]). In the BTBR mouse model of autism, rapamycin treatment has been shown to improve sociability but not stereotypic behaviors [127]. Although abnormal mTOR activation has been associated with fragile X syndrome [128–130], the effect of rapamycin treatment in this disorder is not clear. Rapamycin treatment of AS mice normalized imbalanced mTORC1 and mTORC2 signaling in the cerebellum and significantly improved cerebellum-dependent motor learning [42]. Collectively, the current literature seems to suggest that the mTOR signaling pathway is the converging point in several neurodevelopmental disorders, especially autism-related diseases. However, whether rapamycin or its analogs have any beneficial effects in AS or other autism-related diseases awaits further preclinical research and clinical trials.
3.3 Therapies inferred from other monogenetic neurodevelopmental disorders
Minocycline, a member of the tetracycline family, has been shown to improve behavioral performance in humans with cognitive dysfunction, including schizophrenia [131], depression [132, 133] and fragile X syndrome (FXS) [122, 134]. In human trials with a small number of FXS patients, minocycline treatment improved language ability, behavior, and cognition [122]. Based on these promising results and the shared pathophysiological mechanisms between AS and FXS, an open-label prospective trial of minocycline was conducted in 25 AS patients for 16 weeks. The pilot study showed that minocycline was well tolerated and that there were improvements in language ability, fine motor skills and adaptive behaviors (Table III; [135]). However, since this was an uncontrolled study, the clinical outcomes of minocycline in AS patients need to be further validated. As summarized in Table III, to date the few clinical trials that intended to find a curative treatment instead of managing symptoms for AS, have either failed to show significant effects or have been inconclusive.
Table III.
Clinical trials in AS patients
| Compound | Target | Trial results | References |
|---|---|---|---|
| Betaine/folic acid | methylation | No significant improvement | [104, 105] |
| Levodopa/carbidopa | CaMKII | Completed but results were not posted | ClinicalTrials.gov Identifier: NCT01281475 |
| Minocycline | MMP-9 | Ongoing | ClinicalTrials.gov Identifier: NCT01531582 |
The mechanisms underlying the potential therapeutic effects of minocycline in FXS and other neurologic disorders are unknown. Minocycline has been found to be neuroprotective through anti-inflammatory, anti-oxidant, anti-apoptotic and anti-excitotoxic effects [131]. However, research with mouse models of FXS has indicated that minocycline normalized neuronal morphology via interaction with matrix metalloproteinases (MMPs), which are zinc-dependent endopeptidases that degrade extracellular matrix proteins [134]. Knockout of MMP9 in FXS mice rescued dendritic spine abnormalities, abnormal mGluR5-dependent LTD, as well as aberrant behaviors in open field and social novelty tests [136]. Notably, MMP9 deficiency also corrected abnormal mTOR activation in FXS [136], although whether there is dysregulation of MMPs in AS remains to be determined.
4. Conclusion
Since the discovery that UBE3A deficiency is the cause for AS in 1997, considerable efforts have been spent on understanding the molecular/cellular basis underlying AS-relevant phenotypes. A couple of proteomic studies have proposed several potential UBE3A substrates [137, 138], though whether the identified proteins are directly or indirectly affected by UBE3A and whether they play critical roles in AS pathogenesis remain to be determined. On the other hand, using AS mouse models, several groups have reported abnormalities in cell signaling and have identified UBE3A substrates that are critically involved in AS-relevant phenotypes. The literature reviewed in this article supports the notion that UBE3A deficiency-induced changes converge at synapses, and result in abnormal spine morphology and functions. Accordingly, normalization of these synaptic alterations should be beneficial, as should the reinstatement of paternal UBE3A gene expression.
Of note, maternal duplication of chromosome region 15q11-q13 is associated with a wide range of neuropsychiatric disorders, including ASD; a recent study of a female patient with duplication of only the UBE3A gene suggests that UBE3A is the key etiologic factor in the phenotypes of 15q11-q13 duplications [139]. Therefore, further investigation of the roles of UBE3A in the CNS will not only elucidate AS pathogenesis but may also benefit our understanding of ASD and other psychiatric disorders.
5. Expert opinion
Although Angelman syndrome has been known for over 50 years, there is still no effective treatment. Current therapies are mainly supportive, directed at mitigating symptoms and enhancing quality of life, but even these approaches have limited success. With the establishment of UBE3A deficiency as the cause for AS, a better understanding of the subsequent pathophysiology, and the unveiling of the molecular mechanism for paternal UBE3A gene silencing, there is hope that new avenues of treatment may be developed in the near future. Current AS therapeutic research consists of two major approaches: 1) increasing UBE3A levels by either viral infections carrying UBE3A genetic information or un-silencing the paternal copy of UBE3A and 2) targeting downstream proteins. Given that there are likely many potential UBE3A targets, the latter approach may be less effective than the former. However, as reviewed here, increasing UBE3A levels only partially reverses AS-associated behavioral deficits, while several approaches targeting downstream proteins provide better rescue effects. Limitations in the first type of approach could be related to insufficient levels of reintroduced UBE3A. However, it is also possible that expressing UBE3A after a critical time period may not be sufficient to completely correct brain abnormalities that developed under UBE3A-deficient conditions before this critical period. On the other hand, accumulating evidence indicates that there are shared pathways and alterations in synaptic structures among autism-related monogenetic syndromes, which may provide a rationale for effective interventions common to all of these disorders. Future approaches may need to combine targeting specific etiologic genes and downstream cell signaling pathways, which are shared by these multiple disorders and responsible for most of their symptoms.
Highlights.
Understanding UBE3A gene imprinting provides opportunities to treat the root cause of Angelman syndrome.
The relatively short time window to reactivate paternal UBE3A limits this therapeutic strategy.
Identification of several signaling pathways regulated by UBE3A offers alternative targets for effective treatment of AS pathology.
Combination of gene reactivation with pharmacological targeting of specific pathways may represent an optimal strategy.
Bibliography
- 1.Barry RJ, Leitner RP, Clarke AR, et al. Behavioral aspects of Angelman syndrome: a case control study. Am J Med Genet A. 2005;132A(1):8–12. doi: 10.1002/ajmg.a.30154. [DOI] [PubMed] [Google Scholar]
- 2.Chamberlain SJ, Chen PF, Ng KY, et al. Induced pluripotent stem cell models of the genomic imprinting disorders Angelman and Prader-Willi syndromes. Proc Natl Acad Sci U S A. 2010;107(41):17668–73. doi: 10.1073/pnas.1004487107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Dan B. Angelman syndrome: current understanding and research prospects. Epilepsia. 2009;50(11):2331–9. doi: 10.1111/j.1528-1167.2009.02311.x. [DOI] [PubMed] [Google Scholar]
- 4.Peters SU, Beaudet AL, Madduri N, et al. Autism in Angelman syndrome: implications for autism research. Clin Genet. 2004;66(6):530–6. doi: 10.1111/j.1399-0004.2004.00362.x. [DOI] [PubMed] [Google Scholar]
- 5.Petersen MB, Brondum-Nielsen K, Hansen LK, et al. Clinical, cytogenetic, and molecular diagnosis of Angelman syndrome: estimated prevalence rate in a Danish county. Am J Med Genet. 1995;60(3):261–2. doi: 10.1002/ajmg.1320600317. [DOI] [PubMed] [Google Scholar]
- 6.Kyllerman M. On the prevalence of Angelman syndrome. Am J Med Genet. 1995;59(3):405. doi: 10.1002/ajmg.1320590331. author reply 403–4. [DOI] [PubMed] [Google Scholar]
- 7**.Kishino T, Lalande M, Wagstaff J. UBE3A/E6-AP mutations cause Angelman syndrome. Nat Genet. 1997;15(1):70–3. doi: 10.1038/ng0197-70. One of the first papers identifying UBE3A mutations as the cause of Angelman syndrome. [DOI] [PubMed] [Google Scholar]
- 8**.Matsuura T, Sutcliffe JS, Fang P, et al. De novo truncating mutations in E6-AP ubiquitin-protein ligase gene (UBE3A) in Angelman syndrome. Nat Genet. 1997;15(1):74–7. doi: 10.1038/ng0197-74. One of the first papers identifying UBE3A mutations as the cause of Angelman syndrome. [DOI] [PubMed] [Google Scholar]
- 9*.Lalande M, Calciano MA. Molecular epigenetics of Angelman syndrome. Cell Mol Life Sci. 2007;64(7–8):947–60. doi: 10.1007/s00018-007-6460-0. Interesting review summarizing molecular types of Angelman syndrome. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Gustin RM, Bichell TJ, Bubser M, et al. Tissue-specific variation of Ube3a protein expression in rodents and in a mouse model of Angelman syndrome. Neurobiol Dis. 2010;39(3):283–91. doi: 10.1016/j.nbd.2010.04.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11*.Albrecht U, Sutcliffe JS, Cattanach BM, et al. Imprinted expression of the murine Angelman syndrome gene, Ube3a, in hippocampal and Purkinje neurons. Nat Genet. 1997;17(1):75–8. doi: 10.1038/ng0997-75. One of the first papers showing that UBE3A expression is imprinted. [DOI] [PubMed] [Google Scholar]
- 12*.Rougeulle C, Glatt H, Lalande M. The Angelman syndrome candidate gene, UBE3A/E6-AP, is imprinted in brain. Nat Genet. 1997;17(1):14–5. doi: 10.1038/ng0997-14. One of the first papers showing that UBE3A expression is imprinted. [DOI] [PubMed] [Google Scholar]
- 13.Vu TH, Hoffman AR. Imprinting of the Angelman syndrome gene, UBE3A, is restricted to brain. Nat Genet. 1997;17(1):12–3. doi: 10.1038/ng0997-12. [DOI] [PubMed] [Google Scholar]
- 14.Landers M, Bancescu DL, Le Meur E, et al. Regulation of the large (approximately 1000 kb) imprinted murine Ube3a antisense transcript by alternative exons upstream of Snurf/Snrpn. Nucleic Acids Res. 2004;32(11):3480–92. doi: 10.1093/nar/gkh670. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Martins-Taylor K, Hsiao JS, Chen PF, et al. Imprinted expression of UBE3A in non-neuronal cells from a Prader-Willi syndrome patient with an atypical deletion. Hum Mol Genet. 2014;23(9):2364–73. doi: 10.1093/hmg/ddt628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Runte M, Huttenhofer A, Gross S, et al. The IC-SNURF-SNRPN transcript serves as a host for multiple small nucleolar RNA species and as an antisense RNA for UBE3A. Hum Mol Genet. 2001;10(23):2687–700. doi: 10.1093/hmg/10.23.2687. [DOI] [PubMed] [Google Scholar]
- 17.Rougeulle C, Cardoso C, Fontes M, et al. An imprinted antisense RNA overlaps UBE3A and a second maternally expressed transcript. Nat Genet. 1998;19(1):15–6. doi: 10.1038/ng0598-15. [DOI] [PubMed] [Google Scholar]
- 18.Meng L, Person RE, Beaudet AL. Ube3a-ATS is an atypical RNA polymerase II transcript that represses the paternal expression of Ube3a. Hum Mol Genet. 2012;21(13):3001–12. doi: 10.1093/hmg/dds130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Chamberlain SJ. RNAs of the human chromosome 15q11-q13 imprinted region. Wiley Interdiscip Rev RNA. 2013;4(2):155–66. doi: 10.1002/wrna.1150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lossie AC, Whitney MM, Amidon D, et al. Distinct phenotypes distinguish the molecular classes of Angelman syndrome. J Med Genet. 2001;38(12):834–45. doi: 10.1136/jmg.38.12.834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Tan WH, Bird LM, Thibert RL, et al. If not Angelman, what is it? A review of Angelman-like syndromes. Am J Med Genet A. 2014;164A(4):975–92. doi: 10.1002/ajmg.a.36416. [DOI] [PubMed] [Google Scholar]
- 22.Huibregtse JM, Scheffner M, Beaudenon S, et al. A family of proteins structurally and functionally related to the E6-AP ubiquitin-protein ligase. Proc Natl Acad Sci U S A. 1995;92(11):5249. doi: 10.1073/pnas.92.11.5249-b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Rotin D, Kumar S. Physiological functions of the HECT family of ubiquitin ligases. Nat Rev Mol Cell Biol. 2009;10(6):398–409. doi: 10.1038/nrm2690. [DOI] [PubMed] [Google Scholar]
- 24.Huang L, Kinnucan E, Wang G, et al. Structure of an E6AP-UbcH7 complex: insights into ubiquitination by the E2-E3 enzyme cascade. Science. 1999;286(5443):1321–6. doi: 10.1126/science.286.5443.1321. [DOI] [PubMed] [Google Scholar]
- 25.Greer PL, Hanayama R, Bloodgood BL, et al. The Angelman Syndrome protein Ube3A regulates synapse development by ubiquitinating arc. Cell. 2010;140(5):704–16. doi: 10.1016/j.cell.2010.01.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Margolis SS, Salogiannis J, Lipton DM, et al. EphB-mediated degradation of the RhoA GEF Ephexin5 relieves a developmental brake on excitatory synapse formation. Cell. 2010;143(3):442–55. doi: 10.1016/j.cell.2010.09.038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Scheffner M, Huibregtse JM, Vierstra RD, et al. The HPV-16 E6 and E6-AP complex functions as a ubiquitin-protein ligase in the ubiquitination of p53. Cell. 1993;75(3):495–505. doi: 10.1016/0092-8674(93)90384-3. [DOI] [PubMed] [Google Scholar]
- 28.Nawaz Z, Lonard DM, Smith CL, et al. The Angelman syndrome-associated protein, E6-AP, is a coactivator for the nuclear hormone receptor superfamily. Mol Cell Biol. 1999;19(2):1182–9. doi: 10.1128/mcb.19.2.1182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Heck DH, Zhao Y, Roy S, et al. Analysis of cerebellar function in Ube3a-deficient mice reveals novel genotype-specific behaviors. Hum Mol Genet. 2008;17(14):2181–9. doi: 10.1093/hmg/ddn117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Jiang YH, Armstrong D, Albrecht U, et al. Mutation of the Angelman ubiquitin ligase in mice causes increased cytoplasmic p53 and deficits of contextual learning and long-term potentiation. Neuron. 1998;21(4):799–811. doi: 10.1016/s0896-6273(00)80596-6. [DOI] [PubMed] [Google Scholar]
- 31.Huang HS, Burns AJ, Nonneman RJ, et al. Behavioral deficits in an Angelman syndrome model: effects of genetic background and age. Behav Brain Res. 2013;243:79–90. doi: 10.1016/j.bbr.2012.12.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32*.Baudry M, Kramar E, Xu X, et al. Ampakines promote spine actin polymerization, long-term potentiation, and learning in a mouse model of Angelman syndrome. Neurobiol Dis. 2012;47(2):210–5. doi: 10.1016/j.nbd.2012.04.002. Interesting study showing that a positive AMPA receptor modulator can improve learning and memory in Angelman syndrome mice. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.van Woerden GM, Harris KD, Hojjati MR, et al. Rescue of neurological deficits in a mouse model for Angelman syndrome by reduction of alphaCaMKII inhibitory phosphorylation. Nat Neurosci. 2007;10(3):280–2. doi: 10.1038/nn1845. [DOI] [PubMed] [Google Scholar]
- 34.Kaphzan H, Hernandez P, Jung JI, et al. Reversal of impaired hippocampal long-term potentiation and contextual fear memory deficits in Angelman syndrome model mice by ErbB inhibitors. Biol Psychiatry. 2012;72(3):182–90. doi: 10.1016/j.biopsych.2012.01.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Bird LM. Angelman syndrome: review of clinical and molecular aspects. Appl Clin Genet. 2014;7:93–104. doi: 10.2147/TACG.S57386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Williams CA, Driscoll DJ, Dagli AI. Clinical and genetic aspects of Angelman syndrome. Genet Med. 2010;12(7):385–95. doi: 10.1097/GIM.0b013e3181def138. [DOI] [PubMed] [Google Scholar]
- 37.Gurrieri F, Accadia M. Genetic imprinting: the paradigm of Prader-Willi and Angelman syndromes. Endocr Dev. 2009;14:20–8. doi: 10.1159/000207473. [DOI] [PubMed] [Google Scholar]
- 38.Clayton-Smith J, Laan L. Angelman syndrome: a review of the clinical and genetic aspects. J Med Genet. 2003;40(2):87–95. doi: 10.1136/jmg.40.2.87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Laan LA, v Haeringen A, Brouwer OF. Angelman syndrome: a review of clinical and genetic aspects. Clin Neurol Neurosurg. 1999;101(3):161–70. doi: 10.1016/s0303-8467(99)00030-x. [DOI] [PubMed] [Google Scholar]
- 40.Khan NL, Wood NW. Prader-Willi and Angelman syndromes: update on genetic mechanisms and diagnostic complexities. Curr Opin Neurol. 1999;12(2):149–54. doi: 10.1097/00019052-199904000-00004. [DOI] [PubMed] [Google Scholar]
- 41.Dindot SV, Antalffy BA, Bhattacharjee MB, et al. The Angelman syndrome ubiquitin ligase localizes to the synapse and nucleus, and maternal deficiency results in abnormal dendritic spine morphology. Hum Mol Genet. 2008;17(1):111–8. doi: 10.1093/hmg/ddm288. [DOI] [PubMed] [Google Scholar]
- 42*.Sun J, Liu Y, Moreno S, et al. Imbalanced mechanistic target of rapamycin c1 and c2 activity in the cerebellum of angelman syndrome mice impairs motor function. J Neurosci. 2015;35(11):4706–18. doi: 10.1523/JNEUROSCI.4276-14.2015. First study implicating abnormal mTOR signaling in Angelman syndrome pathogenesis. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Yashiro K, Riday TT, Condon KH, et al. Ube3a is required for experience-dependent maturation of the neocortex. Nat Neurosci. 2009;12(6):777–83. doi: 10.1038/nn.2327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Weeber EJ, Jiang YH, Elgersma Y, et al. Derangements of hippocampal calcium/calmodulin-dependent protein kinase II in a mouse model for Angelman mental retardation syndrome. J Neurosci. 2003;23(7):2634–44. doi: 10.1523/JNEUROSCI.23-07-02634.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Pignatelli M, Piccinin S, Molinaro G, et al. Changes in mGlu5 receptor-dependent synaptic plasticity and coupling to homer proteins in the hippocampus of Ube3A hemizygous mice modeling angelman syndrome. J Neurosci. 2014;34(13):4558–66. doi: 10.1523/JNEUROSCI.1846-13.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Sato M, Stryker MP. Genomic imprinting of experience-dependent cortical plasticity by the ubiquitin ligase gene Ube3a. Proc Natl Acad Sci U S A. 2010;107(12):5611–6. doi: 10.1073/pnas.1001281107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Kuhnle S, Mothes B, Matentzoglu K, et al. Role of the ubiquitin ligase E6AP/UBE3A in controlling levels of the synaptic protein Arc. Proc Natl Acad Sci U S A. 2013;110(22):8888–93. doi: 10.1073/pnas.1302792110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Chowdhury S, Shepherd JD, Okuno H, et al. Arc/Arg3.1 interacts with the endocytic machinery to regulate AMPA receptor trafficking. Neuron. 2006;52(3):445–59. doi: 10.1016/j.neuron.2006.08.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Rial Verde EM, Lee-Osbourne J, Worley PF, et al. Increased expression of the immediate-early gene arc/arg3.1 reduces AMPA receptor-mediated synaptic transmission. Neuron. 2006;52(3):461–74. doi: 10.1016/j.neuron.2006.09.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Shepherd JD, Rumbaugh G, Wu J, et al. Arc/Arg3.1 mediates homeostatic synaptic scaling of AMPA receptors. Neuron. 2006;52(3):475–84. doi: 10.1016/j.neuron.2006.08.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Cao C, Rioult-Pedotti MS, Migani P, et al. Impairment of TrkB-PSD-95 signaling in Angelman syndrome. PLoS Biol. 2013;11(2):e1001478. doi: 10.1371/journal.pbio.1001478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Hammond RS, Bond CT, Strassmaier T, et al. Small-conductance Ca2+-activated K+ channel type 2 (SK2) modulates hippocampal learning, memory, and synaptic plasticity. J Neurosci. 2006;26(6):1844–53. doi: 10.1523/JNEUROSCI.4106-05.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Lin MT, Lujan R, Watanabe M, et al. SK2 channel plasticity contributes to LTP at Schaffer collateral-CA1 synapses. Nat Neurosci. 2008;11(2):170–7. doi: 10.1038/nn2041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Ngo-Anh TJ, Bloodgood BL, Lin M, et al. SK channels and NMDA receptors form a Ca2+-mediated feedback loop in dendritic spines. Nat Neurosci. 2005;8(5):642–9. doi: 10.1038/nn1449. [DOI] [PubMed] [Google Scholar]
- 55.Adelman JP, Maylie J, Sah P. Small-conductance Ca2+-activated K+ channels: form and function. Annu Rev Physiol. 2012;74:245–69. doi: 10.1146/annurev-physiol-020911-153336. [DOI] [PubMed] [Google Scholar]
- 56.Colgin LL, Jia Y, Sabatier JM, et al. Blockade of NMDA receptors enhances spontaneous sharp waves in rat hippocampal slices. Neurosci Lett. 2005;385(1):46–51. doi: 10.1016/j.neulet.2005.05.005. [DOI] [PubMed] [Google Scholar]
- 57.Ohtsuki G, Piochon C, Adelman JP, et al. SK2 channel modulation contributes to compartment-specific dendritic plasticity in cerebellar Purkinje cells. Neuron. 2012;75(1):108–20. doi: 10.1016/j.neuron.2012.05.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58*.Sun J, Zhu G, Liu Y, et al. UBE3A regulates synaptic plasticity and learning and memory by controlling SK2 channel endocytosis. Cell Rep. 2015;12:1–13. doi: 10.1016/j.celrep.2015.06.023. First paper identifying SK2 as an UBE3A substrate and discussing potential implications for Angelman syndrome pathogenesis. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Braunewell KH, Manahan-Vaughan D. Long-term depression: a cellular basis for learning? Rev Neurosci. 2001;12(2):121–40. doi: 10.1515/revneuro.2001.12.2.121. [DOI] [PubMed] [Google Scholar]
- 60.Poschel B, Stanton PK. Comparison of cellular mechanisms of long-term depression of synaptic strength at perforant path-granule cell and Schaffer collateral-CA1 synapses. Prog Brain Res. 2007;163:473–500. doi: 10.1016/S0079-6123(07)63026-X. [DOI] [PubMed] [Google Scholar]
- 61.Malenka RC, Bear MF. LTP and LTD: an embarrassment of riches. Neuron. 2004;44(1):5–21. doi: 10.1016/j.neuron.2004.09.012. [DOI] [PubMed] [Google Scholar]
- 62.Waung MW, Pfeiffer BE, Nosyreva ED, et al. Rapid translation of Arc/Arg3.1 selectively mediates mGluR-dependent LTD through persistent increases in AMPAR endocytosis rate. Neuron. 2008;59(1):84–97. doi: 10.1016/j.neuron.2008.05.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Brakeman PR, Lanahan AA, O’Brien R, et al. Homer: a protein that selectively binds metabotropic glutamate receptors. Nature. 1997;386(6622):284–8. doi: 10.1038/386284a0. [DOI] [PubMed] [Google Scholar]
- 64.Ronesi JA, Collins KA, Hays SA, et al. Disrupted Homer scaffolds mediate abnormal mGluR5 function in a mouse model of fragile X syndrome. Nat Neurosci. 2012;15(3):431–40. S1. doi: 10.1038/nn.3033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Mulherkar SA, Jana NR. Loss of dopaminergic neurons and resulting behavioural deficits in mouse model of Angelman syndrome. Neurobiol Dis. 2010;40(3):586–92. doi: 10.1016/j.nbd.2010.08.002. [DOI] [PubMed] [Google Scholar]
- 66.Farook MF, DeCuypere M, Hyland K, et al. Altered serotonin, dopamine and norepinepherine levels in 15q duplication and Angelman syndrome mouse models. PLoS One. 2012;7(8):e43030. doi: 10.1371/journal.pone.0043030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Riday TT, Dankoski EC, Krouse MC, et al. Pathway-specific dopaminergic deficits in a mouse model of Angelman syndrome. J Clin Invest. 2012;122(12):4544–54. doi: 10.1172/JCI61888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Jay V, Becker LE, Chan FW, et al. Puppet-like syndrome of Angelman: a pathologic and neurochemical study. Neurology. 1991;41(3):416–22. doi: 10.1212/wnl.41.3.416. [DOI] [PubMed] [Google Scholar]
- 69.Egawa K, Kitagawa K, Inoue K, et al. Decreased tonic inhibition in cerebellar granule cells causes motor dysfunction in a mouse model of Angelman syndrome. Sci Transl Med. 2012;4(163):163ra157. doi: 10.1126/scitranslmed.3004655. [DOI] [PubMed] [Google Scholar]
- 70.Troca-Marin JA, Alves-Sampaio A, Montesinos ML. Deregulated mTOR-mediated translation in intellectual disability. Prog Neurobiol. 2012;96(2):268–82. doi: 10.1016/j.pneurobio.2012.01.005. [DOI] [PubMed] [Google Scholar]
- 71.Ehninger D, Silva AJ. Rapamycin for treating Tuberous sclerosis and Autism spectrum disorders. Trends Mol Med. 2011;17(2):78–87. doi: 10.1016/j.molmed.2010.10.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Gong R, Park CS, Abbassi NR, et al. Roles of glutamate receptors and the mammalian target of rapamycin (mTOR) signaling pathway in activity-dependent dendritic protein synthesis in hippocampal neurons. J Biol Chem. 2006;281(27):18802–15. doi: 10.1074/jbc.M512524200. [DOI] [PubMed] [Google Scholar]
- 73.Hou L, Klann E. Activation of the phosphoinositide 3-kinase-Akt-mammalian target of rapamycin signaling pathway is required for metabotropic glutamate receptor-dependent long-term depression. J Neurosci. 2004;24(28):6352–61. doi: 10.1523/JNEUROSCI.0995-04.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Takei N, Inamura N, Kawamura M, et al. Brain-derived neurotrophic factor induces mammalian target of rapamycin-dependent local activation of translation machinery and protein synthesis in neuronal dendrites. J Neurosci. 2004;24(44):9760–9. doi: 10.1523/JNEUROSCI.1427-04.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Laplante M, Sabatini DM. mTOR signaling in growth control and disease. Cell. 2012;149(2):274–93. doi: 10.1016/j.cell.2012.03.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Wullschleger S, Loewith R, Hall MN. TOR signaling in growth and metabolism. Cell. 2006;124(3):471–84. doi: 10.1016/j.cell.2006.01.016. [DOI] [PubMed] [Google Scholar]
- 77.Jacinto E, Loewith R, Schmidt A, et al. Mammalian TOR complex 2 controls the actin cytoskeleton and is rapamycin insensitive. Nat Cell Biol. 2004;6(11):1122–8. doi: 10.1038/ncb1183. [DOI] [PubMed] [Google Scholar]
- 78.Zhou HY, Huang SL. Current development of the second generation of mTOR inhibitors as anticancer agents. Chin J Cancer. 2012;31(1):8–18. doi: 10.5732/cjc.011.10281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Schmidt A, Bickle M, Beck T, et al. The yeast phosphatidylinositol kinase homolog TOR2 activates RHO1 and RHO2 via the exchange factor ROM2. Cell. 1997;88(4):531–42. doi: 10.1016/s0092-8674(00)81893-0. [DOI] [PubMed] [Google Scholar]
- 80.He Y, Li D, Cook SL, et al. Mammalian target of rapamycin and Rictor control neutrophil chemotaxis by regulating Rac/Cdc42 activity and the actin cytoskeleton. Mol Biol Cell. 2013;24(21):3369–80. doi: 10.1091/mbc.E13-07-0405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Garcia-Martinez JM, Alessi DR. mTOR complex 2 (mTORC2) controls hydrophobic motif phosphorylation and activation of serum- and glucocorticoid-induced protein kinase 1 (SGK1) Biochem J. 2008;416(3):375–85. doi: 10.1042/BJ20081668. [DOI] [PubMed] [Google Scholar]
- 82.Wang Y, Briz V, Chishti A, et al. Distinct roles for mu-calpain and m-calpain in synaptic NMDAR-mediated neuroprotection and extrasynaptic NMDAR-mediated neurodegeneration. J Neurosci. 2013;33(48):18880–92. doi: 10.1523/JNEUROSCI.3293-13.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Thomanetz V, Angliker N, Cloetta D, et al. Ablation of the mTORC2 component rictor in brain or Purkinje cells affects size and neuron morphology. J Cell Biol. 2013;201(2):293–308. doi: 10.1083/jcb.201205030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Huang W, Zhu PJ, Zhang S, et al. mTORC2 controls actin polymerization required for consolidation of long-term memory. Nat Neurosci. 2013;16(4):441–8. doi: 10.1038/nn.3351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Briz V, Zhu G, Wang Y, et al. Activity-dependent rapid local RhoA synthesis is required for hippocampal synaptic plasticity. J Neurosci. 2015;35(5):2269–82. doi: 10.1523/JNEUROSCI.2302-14.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Gilfillan GD, Selmer KK, Roxrud I, et al. SLC9A6 mutations cause X-linked mental retardation, microcephaly, epilepsy, and ataxia, a phenotype mimicking Angelman syndrome. Am J Hum Genet. 2008;82(4):1003–10. doi: 10.1016/j.ajhg.2008.01.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Kondapalli KC, Prasad H, Rao R. An inside job: how endosomal Na(+)/H(+) exchangers link to autism and neurological disease. Front Cell Neurosci. 2014;8:172. doi: 10.3389/fncel.2014.00172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Stromme P, Dobrenis K, Sillitoe RV, et al. X-linked Angelman-like syndrome caused by Slc9a6 knockout in mice exhibits evidence of endosomal-lysosomal dysfunction. Brain. 2011;134(Pt 11):3369–83. doi: 10.1093/brain/awr250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Brett CL, Wei Y, Donowitz M, et al. Human Na(+)/H(+) exchanger isoform 6 is found in recycling endosomes of cells, not in mitochondria. Am J Physiol Cell Physiol. 2002;282(5):C1031–41. doi: 10.1152/ajpcell.00420.2001. [DOI] [PubMed] [Google Scholar]
- 90.Zoncu R, Bar-Peled L, Efeyan A, et al. mTORC1 senses lysosomal amino acids through an inside-out mechanism that requires the vacuolar H(+)-ATPase. Science. 2011;334(6056):678–83. doi: 10.1126/science.1207056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Wang S, Tsun ZY, Wolfson RL, et al. Metabolism. Lysosomal amino acid transporter SLC38A9 signals arginine sufficiency to mTORC1. Science. 2015;347(6218):188–94. doi: 10.1126/science.1257132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Rebsamen M, Pochini L, Stasyk T, et al. SLC38A9 is a component of the lysosomal amino acid sensing machinery that controls mTORC1. Nature. 2015;519(7544):477–81. doi: 10.1038/nature14107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Jung J, Genau HM, Behrends C. Amino Acid-Dependent mTORC1 Regulation by the Lysosomal Membrane Protein SLC38A9. Mol Cell Biol. 2015;35(14):2479–94. doi: 10.1128/MCB.00125-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Klein R. Bidirectional modulation of synaptic functions by Eph/ephrin signaling. Nat Neurosci. 2009;12(1):15–20. doi: 10.1038/nn.2231. [DOI] [PubMed] [Google Scholar]
- 95.Mei L, Xiong WC. Neuregulin 1 in neural development, synaptic plasticity and schizophrenia. Nat Rev Neurosci. 2008;9(6):437–52. doi: 10.1038/nrn2392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Kaphzan H, Buffington SA, Jung JI, et al. Alterations in intrinsic membrane properties and the axon initial segment in a mouse model of Angelman syndrome. J Neurosci. 2011;31(48):17637–48. doi: 10.1523/JNEUROSCI.4162-11.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Kaphzan H, Buffington SA, Ramaraj AB, et al. Genetic reduction of the alpha1 subunit of Na/K-ATPase corrects multiple hippocampal phenotypes in Angelman syndrome. Cell Rep. 2013;4(3):405–12. doi: 10.1016/j.celrep.2013.07.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Wallace ML, Burette AC, Weinberg RJ, et al. Maternal loss of Ube3a produces an excitatory/inhibitory imbalance through neuron type-specific synaptic defects. Neuron. 2012;74(5):793–800. doi: 10.1016/j.neuron.2012.03.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Daily JL, Nash K, Jinwal U, et al. Adeno-associated virus-mediated rescue of the cognitive defects in a mouse model for Angelman syndrome. PLoS One. 2011;6(12):e27221. doi: 10.1371/journal.pone.0027221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Guffanti G, Strik Lievers L, Bonati MT, et al. Role of UBE3A and ATP10A genes in autism susceptibility region 15q11-q13 in an Italian population: a positive replication for UBE3A. Psychiatry Res. 2011;185(1–2):33–8. doi: 10.1016/j.psychres.2010.04.057. [DOI] [PubMed] [Google Scholar]
- 101.Smith SE, Zhou YD, Zhang G, et al. Increased gene dosage of Ube3a results in autism traits and decreased glutamate synaptic transmission in mice. Sci Transl Med. 2011;3(103):103ra97. doi: 10.1126/scitranslmed.3002627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Schanen NC. Epigenetics of autism spectrum disorders. Hum Mol Genet. 2006;15(2):R138–50. doi: 10.1093/hmg/ddl213. [DOI] [PubMed] [Google Scholar]
- 103.Yamasaki K, Joh K, Ohta T, et al. Neurons but not glial cells show reciprocal imprinting of sense and antisense transcripts of Ube3a. Hum Mol Genet. 2003;12(8):837–47. doi: 10.1093/hmg/ddg106. [DOI] [PubMed] [Google Scholar]
- 104.Bird LM, Tan WH, Bacino CA, et al. A therapeutic trial of pro-methylation dietary supplements in Angelman syndrome. Am J Med Genet A. 2011;155A(12):2956–63. doi: 10.1002/ajmg.a.34297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Peters SU, Bird LM, Kimonis V, et al. Double-blind therapeutic trial in Angelman syndrome using betaine and folic acid. Am J Med Genet A. 2010;152A(8):1994–2001. doi: 10.1002/ajmg.a.33509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106*.Huang HS, Allen JA, Mabb AM, et al. Topoisomerase inhibitors unsilence the dormant allele of Ube3a in neurons. Nature. 2012;481(7380):185–9. doi: 10.1038/nature10726. First study demonstrating that paternal Ube3a allele can be unsilenced. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Meng L, Person RE, Huang W, et al. Truncation of Ube3a-ATS unsilences paternal Ube3a and ameliorates behavioral defects in the Angelman syndrome mouse model. PLoS Genet. 2013;9(12):e1004039. doi: 10.1371/journal.pgen.1004039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108*.Meng L, Ward AJ, Chun S, et al. Towards a therapy for Angelman syndrome by targeting a long non-coding RNA. Nature. 2015;518(7539):409–12. doi: 10.1038/nature13975. Interesting study showing the possibility of unsilencing paternal Ube3a by using antisense oligonucleotides. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Silva-Santos S, van Woerden GM, Bruinsma CF, et al. Ube3a reinstatement identifies distinct developmental windows in a murine Angelman syndrome model. J Clin Invest. 2015;125(5):2069–76. doi: 10.1172/JCI80554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Yi JJ, Berrios J, Newbern JM, et al. An Autism-Linked Mutation Disables Phosphorylation Control of UBE3A. Cell. 2015;162(4):795–807. doi: 10.1016/j.cell.2015.06.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Baudry M, Bi X, Gall C, et al. The biochemistry of memory: The 26year journey of a ‘new and specific hypothesis’. Neurobiol Learn Mem. 2011;95(2):125–33. doi: 10.1016/j.nlm.2010.11.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Kramar EA, Lin B, Rex CS, et al. Integrin-driven actin polymerization consolidates long-term potentiation. Proc Natl Acad Sci U S A. 2006;103(14):5579–84. doi: 10.1073/pnas.0601354103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Lin B, Kramar EA, Bi X, et al. Theta stimulation polymerizes actin in dendritic spines of hippocampus. J Neurosci. 2005;25(8):2062–9. doi: 10.1523/JNEUROSCI.4283-04.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Lynch G, Rex CS, Chen LY, et al. The substrates of memory: defects, treatments, and enhancement. Eur J Pharmacol. 2008;585(1):2–13. doi: 10.1016/j.ejphar.2007.11.082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Kramar EA, Babayan AH, Gall CM, et al. Estrogen promotes learning-related plasticity by modifying the synaptic cytoskeleton. Neuroscience. 2012 doi: 10.1016/j.neuroscience.2012.10.038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Simmons DA, Rex CS, Palmer L, et al. Up-regulating BDNF with an ampakine rescues synaptic plasticity and memory in Huntington’s disease knockin mice. Proc Natl Acad Sci U S A. 2009;106(12):4906–11. doi: 10.1073/pnas.0811228106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Lauterborn JC, Truong GS, Baudry M, et al. Chronic elevation of brain-derived neurotrophic factor by ampakines. J Pharmacol Exp Ther. 2003;307(1):297–305. doi: 10.1124/jpet.103.053694. [DOI] [PubMed] [Google Scholar]
- 118.Jourdi H, Hsu YT, Zhou M, et al. Positive AMPA receptor modulation rapidly stimulates BDNF release and increases dendritic mRNA translation. J Neurosci. 2009;29(27):8688–97. doi: 10.1523/JNEUROSCI.6078-08.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Silverman JL, Oliver CF, Karras MN, et al. AMPAKINE enhancement of social interaction in the BTBR mouse model of autism. Neuropharmacology. 2013;64:268–82. doi: 10.1016/j.neuropharm.2012.07.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Ogier M, Wang H, Hong E, et al. Brain-derived neurotrophic factor expression and respiratory function improve after ampakine treatment in a mouse model of Rett syndrome. J Neurosci. 2007;27(40):10912–7. doi: 10.1523/JNEUROSCI.1869-07.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Lauterborn JC, Rex CS, Kramar E, et al. Brain-derived neurotrophic factor rescues synaptic plasticity in a mouse model of fragile X syndrome. J Neurosci. 2007;27(40):10685–94. doi: 10.1523/JNEUROSCI.2624-07.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Hagerman R, Lauterborn J, Au J, et al. Fragile X syndrome and targeted treatment trials. Results Probl Cell Differ. 2012;54:297–335. doi: 10.1007/978-3-642-21649-7_17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Carson RP, Van Nielen DL, Winzenburger PA, et al. Neuronal and glia abnormalities in Tsc1-deficient forebrain and partial rescue by rapamycin. Neurobiol Dis. 2012;45(1):369–80. doi: 10.1016/j.nbd.2011.08.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Zeng LH, Xu L, Gutmann DH, et al. Rapamycin prevents epilepsy in a mouse model of tuberous sclerosis complex. Ann Neurol. 2008;63(4):444–53. doi: 10.1002/ana.21331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Goto J, Talos DM, Klein P, et al. Regulable neural progenitor-specific Tsc1 loss yields giant cells with organellar dysfunction in a model of tuberous sclerosis complex. Proc Natl Acad Sci U S A. 2011;108(45):E1070–9. doi: 10.1073/pnas.1106454108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Ehninger D, Han S, Shilyansky C, et al. Reversal of learning deficits in a Tsc2+/− mouse model of tuberous sclerosis. Nat Med. 2008;14(8):843–8. doi: 10.1038/nm1788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Burket JA, Benson AD, Tang AH, et al. Rapamycin improves sociability in the BTBR T(+)Itpr3(tf)/J mouse model of autism spectrum disorders. Brain Res Bull. 2014;100:70–5. doi: 10.1016/j.brainresbull.2013.11.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Vanderklish PW, Edelman GM. Differential translation and fragile X syndrome. Genes Brain Behav. 2005;4(6):360–84. doi: 10.1111/j.1601-183X.2005.00134.x. [DOI] [PubMed] [Google Scholar]
- 129.Sharma A, Hoeffer CA, Takayasu Y, et al. Dysregulation of mTOR signaling in fragile X syndrome. J Neurosci. 2010;30(2):694–702. doi: 10.1523/JNEUROSCI.3696-09.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Gross C, Nakamoto M, Yao X, et al. Excess phosphoinositide 3-kinase subunit synthesis and activity as a novel therapeutic target in fragile X syndrome. J Neurosci. 2010;30(32):10624–38. doi: 10.1523/JNEUROSCI.0402-10.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Zhang L, Zhao J. Profile of minocycline and its potential in the treatment of schizophrenia. Neuropsychiatr Dis Treat. 2014;10:1103–11. doi: 10.2147/NDT.S64236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Soczynska JK, Mansur RB, Brietzke E, et al. Novel therapeutic targets in depression: minocycline as a candidate treatment. Behav Brain Res. 2012;235(2):302–17. doi: 10.1016/j.bbr.2012.07.026. [DOI] [PubMed] [Google Scholar]
- 133.Dean OM, Data-Franco J, Giorlando F, et al. Minocycline: therapeutic potential in psychiatry. CNS Drugs. 2012;26(5):391–401. doi: 10.2165/11632000-000000000-00000. [DOI] [PubMed] [Google Scholar]
- 134.Bilousova TV, Dansie L, Ngo M, et al. Minocycline promotes dendritic spine maturation and improves behavioural performance in the fragile X mouse model. J Med Genet. 2009;46(2):94–102. doi: 10.1136/jmg.2008.061796. [DOI] [PubMed] [Google Scholar]
- 135.Grieco JC, Ciarlone SL, Gieron-Korthals M, et al. An open-label pilot trial of minocycline in children as a treatment for Angelman syndrome. BMC Neurol. 2014;14(1):232. doi: 10.1186/s12883-014-0232-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Sidhu H, Dansie LE, Hickmott PW, et al. Genetic removal of matrix metalloproteinase 9 rescues the symptoms of fragile X syndrome in a mouse model. J Neurosci. 2014;34(30):9867–79. doi: 10.1523/JNEUROSCI.1162-14.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Jensen L, Farook MF, Reiter LT. Proteomic profiling in Drosophila reveals potential Dube3a regulation of the actin cytoskeleton and neuronal homeostasis. PLoS One. 2013;8(4):e61952. doi: 10.1371/journal.pone.0061952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Loon LH, Chen C-FJ, Chen C-CK, et al. Angelman syndrome: proteomics analysis of an UBE3A knockout mouse and its implications. 2015 http://www.intechopen.com/books/advanced-topics-in-neurological-disorders/angelman-syndrome-proteomics-analysis-of-an-ube3a-knockout-mouse-and-its-implications.
- 139.Noor A, Dupuis L, Mittal K, et al. 15q11.2 Duplication Encompassing Only the UBE3A Gene Is Associated with Developmental Delay and Neuropsychiatric Phenotypes. Hum Mutat. 2015;36(7):689–93. doi: 10.1002/humu.22800. [DOI] [PubMed] [Google Scholar]