Abstract
Background
The single-stranded RNA Flavivirus, Zika virus (ZIKV), has recently re-emerged and spread rapidly across the western hemisphere’s equatorial countries, primarily through Aedes mosquito transmission. While symptoms in adult infections appear to be self-limiting and mild, severe birth defects, such as microcephaly, have been linked to infection during early pregnancy. Recently, Tang et al. (Cell Stem Cell 2016, doi: 10.1016/j.stem.2016.02.016) demonstrated that ZIKV efficiently infects induced pluripotent stem cell (iPSC) derived human neural progenitor cells (hNPCs), resulting in cell cycle abnormalities and apoptosis. Consequently, hNPCs are a suggested ZIKV target.
Methods
We analyzed the transcriptomic sequencing (RNA-seq) data (GEO: GSE78711) of ZIKV (Strain: MR766) infected hNPCs. For comparison to the ZIKV-infected hNPCs, the expression data from hNPCs infected with human cytomegalovirus (CMV) (Strain: AD169) was used (GEO: GSE35295). Utilizing a combination of Gene Ontology, database of human diseases, and pathway analysis, we generated a putative systemic model of infection supported by known molecular pathways of other highly related viruses.
Results
We analyzed RNA-sequencing data for transcript expression alterations in ZIKV-infected hNPCs, and then compared them to expression patterns of iPSC-derived hNPCs infected with CMV, a virus that can also induce severe congenital neurological defects in developing fetuses. We demonstrate for the first time that many of cellular pathways correlate with clinical pathologies following ZIKV infection such as microcephaly, congenital nervous system disorders and epilepsy. Furthermore, ZIKV activates several inflammatory signals within infected hNPCs that are implicated in innate and acquired immune responses, while CMV-infected hNPCs showed limited representation of these pathways. Moreover, several genes related to pathogen responses are significantly upregulated upon ZIKV infection, but not perturbed in CMV-infected hNPCs.
Conclusion
The presented study is the first to report enrichment of numerous pro-inflammatory pathways in ZIKV-infected hNPCs, indicating that hNPCs are capable of signaling through canonical pro-inflammatory pathways following viral infection. By defining gene expression profiles, new factors in the pathogenesis of ZIKV were identified which could help develop new therapeutic strategies.
Background
Zika Virus (ZIKV) is a single-stranded positive sense RNA Flavivirus [1] that is primarily transmitted though the Aedes mosquito [2]. Until recently this virus had remained relatively obscure, with the exception of a few scattered outbreaks [1–3]. Currently, 26 countries across South and Central America have reported widespread ZIKV infections, with cases also emerging in Europe and the United States [4]. Typically, adult ZIKV infections are mild or asymptomatic, with exanthema, fever, conjunctivitis, and joint pain being the most common symptoms [2, 5]. However, ZIKV infection is also believed to be related to the concurrent increase in adult Guillain–Barré syndrome (GBS) cases [4]. This is supported by a case controlled prospective study of the French Polynesian outbreak, where ZIKV was linked with increased incidence of GBS [3]. Aside from its effects in adults, ZIKV infection of pregnant women can result in birth defects ranging from low birth weight to craniofacial and eye abnormalities [6], as well as microcephaly [7, 8]. The detection of ZIKV in fetal brain tissues indicates that ZIKV can also cross the placental barrier [9]. Consequently, curbing the current ZIKV epidemic has become a top priority for a number of international health initiatives.
Prompt laboratory-based efforts by Tang et al. provided RNA-seq data (GEO: GSE78711) from ZIKV-infected human neural progenitor cells (hNPCs). They found that when compared to neurons and embryonic stem cells, ZIKV infects hNPCs with high efficiency, and that infection results in abnormal cell cycle dynamics and apoptosis [9]. For the current study, we analyzed this dataset using bioinformatic methods to uncover links between neuro-inflammatory pathways and ZIKV infection of hNPCs, as well as its correlation with clinical neurological symptoms. Since congenital CMV infection is also associated with higher incidence of neurological birth defects [10], a CMV-infected hNPC dataset (GEO: GSE19345) was used for comparison [11]. We demonstrate for the first time that ZIKV activates several inflammatory signals within infected hNPCs that are implicated in both innate and acquired immune responses. Moreover, several genes related to pathogen responses are significantly up-regulated in response to ZIKV infection, but not perturbed in CMV-infected hNPCs. Our approach offers novel insight into the potential mechanisms underlying ZIKV infection and provides useful directions for future clinical research.
Results
Comprehensive gene set gene ontology analysis
Comparison of the mock-infected samples to the ZIKV-infected samples using a modified t-test yields a total of 3443 genes up-regulated and 3421 down-regulated. The top 30 highest absolute fold-change (FC) genes are reported in Tables 1 and 2 along with their associated Entrez IDs and p-values. Gene Ontology (GO) term analysis showed that the up-regulated gene set was exclusively enriched in processes related to nucleic acid metabolism regulation (Fig. 1a). Since replication of the viral genome requires reallocation of host nucleotides [12], this finding was unsurprising. Also, the down-regulated gene set was enriched for DNA metabolism processes, although with emphasis on cell cycle regulation. These findings were consistent with evidence showing ZIKV infection results in the down-regulation of cell cycle genes, impaired cell cycle progression through G1 [9], and decreased proliferation [13] in vitro. Of the ZIKV down-regulated genes, the biological process most enriched proved to be chromosome segregation. This finding provides an interesting link to known causes of hereditary microcephaly, as abnormal brain development and reduced volume often are the result of dysregulation of chromosome segregation [14, 15].
Table 1.
Top 30 genes up-regulated in ZIKV infected hNPCs compared to controls
| Gene symbol | Entrez | Gene name | Log2 FC | p value |
|---|---|---|---|---|
| IFIT2 | 3433 | Interferon-induced protein with tetratricopeptide repeats 2 | 5.22967 | 0.0051 |
| TNFRSF14 | 8764 | Tumor necrosis factor receptor superfamily, member 14 | 3.4467 | 0.00005 |
| BHLHE41 | 79365 | Basic helix-loop-helix family, member e41 | 3.41193 | 0.00005 |
| SNORD116-4 | 100033416 | Small nucleolar RNA, C/D box 116-4 | 3.3541 | 0.00065 |
| CEBPB | 1051 | CCAAT/enhancer binding protein (C/EBP), beta | 3.08608 | 0.00005 |
| SEC24D | 9871 | SEC24 family, member D (S. cerevisiae) | 3.06648 | 0.00005 |
| CHAC1 | 79094 | ChaC, cation transport regulator homolog 1 (E. coli) | 2.98802 | 0.00005 |
| CREB3L1 | 90993 | cAMP responsive element binding protein 3-like 1 | 2.9718 | 0.00005 |
| DDIT3 | 1649 | DNA-damage-inducible transcript 3 | 2.94172 | 0.00005 |
| STC2 | 8614 | Stanniocalcin 2 | 2.85446 | 0.00005 |
| IL20RB | 53833 | Interleukin 20 receptor beta | 2.74302 | 0.00005 |
| ADM2 | 79924 | Adrenomedullin 2 | 2.73791 | 0.00005 |
| DDR2 | 4921 | Discoidin domain receptor tyrosine kinase 2 | 2.68283 | 0.00005 |
| ULBP1 | 80329 | UL16 binding protein 1 | 2.6589 | 0.00005 |
| SLC7A11 | 23657 | Solute carrier family 7 (anionic amino acid transporter light chain, xc-system), member 11 | 2.55855 | 0.00005 |
| GABRR2 | 2570 | Gamma-aminobutyric acid (GABA) A receptor, rho 2 | 2.54697 | 0.00025 |
| FOSL1 | 8061 | FOS-like antigen 1 | 2.54626 | 0.00005 |
| RELB | 5971 | v-rel reticuloendotheliosis viral oncogene homolog B | 2.52167 | 0.00005 |
| BHLHE40 | 8553 | Basic helix-loop-helix family, member e40 | 2.50278 | 0.00005 |
| INHBE | 83729 | Inhibin, beta E | 2.48416 | 0.00005 |
| XBP1 | 7494 | X-box binding protein 1 | 2.48392 | 0.00005 |
| SELPLG | 6404 | Selectin P ligand | 2.47628 | 0.00005 |
| SLC7A5 | 8140 | Solute carrier family 7 (amino acid transporter light chain, L system), member 5 | 2.44283 | 0.00005 |
| ARHGAP9 | 64333 | Rho GTPase activating protein 9 | 2.42899 | 0.00005 |
| HERPUD1 | 9709 | Homocysteine-inducible, endoplasmic reticulum stress-inducible, ubiquitin-like domain member 1 | 2.38525 | 0.00005 |
| KLF15 | 28999 | Kruppel-like factor 15 | 2.36633 | 0.00005 |
| CDC42EP1 | 11135 | CDC42 effector protein (Rho GTPase binding) 1 | 2.35073 | 0.00005 |
| CEBPD | 1052 | CCAAT/enhancer binding protein (C/EBP), delta | 2.34979 | 0.00025 |
| GAL3ST2 | 64090 | Galactose-3-O-sulfotransferase 2 | 2.31589 | 0.0021 |
| FAM129A | 116496 | Family with sequence similarity 129, member A | 2.28808 | 0.00005 |
Table 2.
Top 30 genes down-regulated in ZIKV infected hNPCs compared to controls
| Gene symbol | Entrez | Gene name | Log2 FC | p-value |
|---|---|---|---|---|
| SIGLEC10 | 89790 | Sialic acid binding Ig-like lectin 10 | −3.60664 | 0.00005 |
| CDC20B | 166979 | Cell division cycle 20 homolog B (S. cerevisiae) | −3.44083 | 0.0002 |
| OR51E2 | 81285 | Olfactory receptor, family 51, subfamily E, member 2 | −3.05742 | 0.0001 |
| CCNO | 10309 | Cyclin O | −3.00562 | 0.00005 |
| SLCO4A1 | 28231 | Solute carrier organic anion transporter family, member 4A1 | −2.97508 | 0.0229 |
| SGPP2 | 130367 | Sphingosine-1-phosphate phosphatase 2 | −2.69025 | 0.00035 |
| RRM2 | 6241 | Ribonucleotide reductase M2 | −2.63783 | 0.00005 |
| SFXN2 | 118980 | Sideroflexin 2 | −2.61672 | 0.00005 |
| COLEC12 | 81035 | Collectin sub-family member 12 | −2.60458 | 0.00005 |
| CHRNA1 | 1134 | Cholinergic receptor, nicotinic, alpha 1 (muscle) | −2.5688 | 0.00005 |
| PRR22 | 163154 | Proline rich 22 | −2.54683 | 0.00045 |
| DHFR | 1719 | Dihydrofolate reductase | −2.53145 | 0.00005 |
| MFI2 | 4241 | Antigen p97 (melanoma associated) identified by monoclonal antibodies 133.2 and 96.5 | −2.50748 | 0.00005 |
| MAPK15 | 225689 | Mitogen-activated protein kinase 15 | −2.46203 | 0.00005 |
| C21orf58 | 54058 | Chromosome 21 open reading frame 58 | −2.39697 | 0.00005 |
| SPEF1 | 25876 | Sperm flagellar 1 | −2.39565 | 0.00005 |
| GHRL | 51738 | Ghrelin/obestatin prepropeptide | −2.35804 | 0.00805 |
| ATG16L2 | 89849 | Autophagy related 16-like 2 (S. cerevisiae) | −2.34225 | 0.00005 |
| CXCL14 | 9547 | Chemokine (C-X-C motif) ligand 14 | −2.33189 | 0.0015 |
| SUSD2 | 56241 | Sushi domain containing 2 | −2.28898 | 0.00005 |
| C6orf118 | 168090 | Chromosome 6 open reading frame 118 | −2.27987 | 0.00005 |
| DLX6-AS1 | 285987 | DLX6 antisense RNA 1 | −2.26662 | 0.00005 |
| ADAM28 | 10863 | ADAM metallopeptidase domain 28 | −2.25906 | 0.00005 |
| NXPH2 | 11249 | Neurexophilin 2 | −2.22856 | 0.00005 |
| AQP7P1 | 375719 | Aquaporin 7 pseudogene 1 | −2.2255 | 0.00005 |
| EFCAB1 | 79645 | EF-hand calcium binding domain 1 | −2.19918 | 0.00005 |
| WNT8B | 7479 | Wingless-type MMTV integration site family, member 8B | −2.19366 | 0.00005 |
| PI15 | 51050 | Peptidase inhibitor 15 | −2.17615 | 0.00005 |
| C1orf189 | 388701 | Chromosome 1 open reading frame 189 | −2.17531 | 0.01255 |
| OLFML3 | 56944 | Olfactomedin-like 3 | −2.16835 | 0.0016 |
Fig. 1.
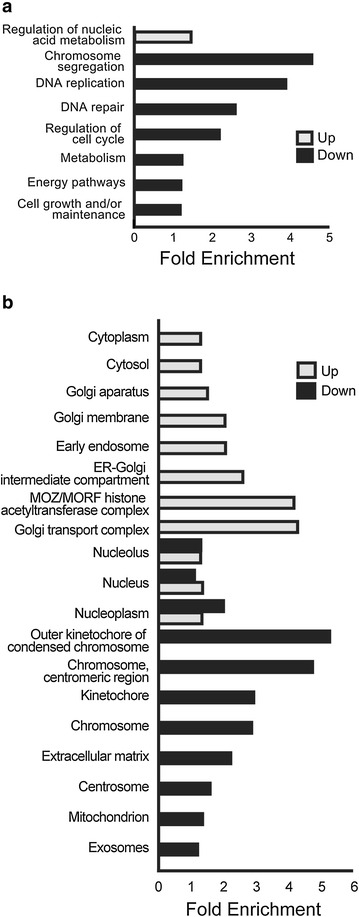
Comprehensive gene set gene ontology analysis. Gene ontology analysis was performed on hNPC genes whose expression was significantly altered following infection with ZIKV for 56 h. Transcript enrichment significance was determined using the human protein coding genome as the reference background. a Regulation of nucleic acid metabolism was the only biological process sub-ontology enriched with ZIKV up-regulated genes. Down-regulated genes were enriched for pathways related to cell cycle regulation and general metabolism. All significant terms are presented. b Cellular component enrichment indicated down-regulated genes generally associated with chromosomal regions, the extracellular matrix, and centrosomes. Up-regulated genes showed the most enrichment for the Golgi apparatus. Both up- and down-regulated genes were significantly enriched in nuclear sites. Enrichment scores were sorted by the most significant terms, and the top 11 compartments are represented. (q < 0.05)
By also assessing cellular compartment sub-ontology, we elucidated the cellular location of the infection induced gene expression alterations (Fig. 1b). Genes down-regulated by ZIKV were highly enriched in centromeric and nuclear sites, the extracellular matrix, and mitochondria. Furthermore, down-regulated genes were associated with exosomes, suggesting that the infection can influence exosome biology. Up-regulated genes were enriched specifically in the Golgi complex, the cytoplasm, and the monocytic leukemia zinc-finger protein/related factor (MOZ/MORF) histone acetyltransferase complex, which serves as a regulator of neural and hematopoietic stem-cell identity [16].
ZIKV DEGs represented in canonical human neurological diseases
To investigate whether ZIKV induced differentially expressed genes (DEGs) are related to the observed clinical phenotypes, comparisons were made between significant ZIKV DEGs and those related to the pathogenesis of human neurological diseases (Fig. 2) (Table 3). Considering that hNPCs were used for the RNA-seq study, our inquiry was limited to diseases of the nervous system. Using the MalaCards [17] integrated searchable database of human diseases, we selected six conditions for comparison. Each condition is a parent disease relating sub-categories of more specific diseases. For each condition, the number of ZIKV DEGs present in the MalaCards reference list is reported as the represented percentage. The number of total genes in each MalaCards reference list is plotted along the second y-axis (scatterplot). The condition with the most representation proved to be microcephaly, with 70 % of the 30 reference list genes present in the ZIKV DEGs. Since, epilepsy is a common microcephaly co-morbidly [18], it was unsurprising to discover there was 43 % representation of related genes. The more general group of congenital nervous system disorders also displayed a similarly high representation of ZIKV DEGs at 60 % of the total reference list. Since, there has also been an increase in reported cases of adult GBS in ZIKV affected areas [2, 3], we investigated encephalitis, demyelinating diseases, and GBS specifically. While representation of ZIKV DEGs in these disease classifications were present, it was significantly less than congenital central nervous system (CNS) diseases.
Fig. 2.

ZIKV DEGs mapped to canonical human disease gene sets. Using the MalaCards database of human diseases, diseases with established clinical significance to ZIKV infection were queried. ZIKV DEGs were then compared to reference gene list for each disease to determine a percentage of representation. Diseases related to congenital CNS disorders were most highly represented with ZIKV DEGs. Although less, representation of adult inflammatory and demyelinating diseases were also observed. (Bars % of ZIKV induced genes present in the reference list) (Open circles total number of genes in the disease reference list)
Table 3.
Neurological clinical phenotypes associated with ZIKV DEGs
| Disease | Gene symbol | Gene name |
|---|---|---|
| Microcephaly | DYRK1A | Dual-specificity tyrosine-(Y)-phosphorylation regulated kinase 1A |
| MCPH1 | Microcephalin 1 | |
| CASK | Calcium/calmodulin-dependent serine protein kinase (MAGUK family) | |
| PHGDH | Phosphoglycerate dehydrogenase | |
| ARFGEF2 | ADP-ribosylation factor guanine nucleotide-exchange factor 2 | |
| ASPM | Asp (abnormal spindle) homolog, microcephaly associated | |
| CENPJ | Centromere protein J | |
| CEP152 | Centrosomal protein 152 kDa | |
| SLC25A19 | Solute carrier family 25 (mitochondrial thiamine pyrophosphate carrier), member 19 | |
| WDR62 | WD repeat domain 62 | |
| CDK5RAP2 | CDK5 regulatory subunit associated protein 2 | |
| CASC5 | Cancer susceptibility candidate 5 | |
| NBN | Nibrin | |
| EFTUD2 | Elongation factor Tu GTP binding domain containing 2 | |
| IER3IP1 | Immediate early response 3 interacting protein 1 | |
| STIL | SCL/TAL1 interrupting locus | |
| CEP135 | Centrosomal protein 135 kDa | |
| ZNF335 | Zinc finger protein 335 | |
| NDE1 | nudE nuclear distribution E homolog 1 | |
| DIAPH1 | Diaphanous homolog 1 | |
| KIF11 | Kinesin family member 11 | |
| Congenital nervous system disorders | MCPH1 | Microcephalin 1 |
| ARFGEF2 | ADP-ribosylation factor guanine nucleotide-exchange factor 2 (brefeldin A-inhibited) | |
| TGIF1 | TGFB-induced factor homeobox 1 | |
| GRIN2B | Glutamate receptor, ionotropic, N-methyl d-aspartate 2B | |
| SLC12A5 | Solute carrier family 12 (potassium/chloride transporter), member 5 | |
| FLNA | Filamin A, alpha | |
| SIX3 | SIX homeobox 3 | |
| SHH | Sonic hedgehog | |
| CDK5RAP2 | CDK5 regulatory subunit associated protein 2 | |
| NDE1 | nudE nuclear distribution E homolog 1 | |
| WDR62 | WD repeat domain 62 | |
| ASPM | Asp (abnormal spindle) homolog, microcephaly associated | |
| CENPJ | Centromere protein J | |
| STIL | SCL/TAL1 interrupting locus | |
| FKTN | Fukutin | |
| POMGNT1 | Protein O−linked mannose beta1,2−N−acetylglucosaminyltransferase | |
| CEP152 | Centrosomal protein 152 kDa | |
| POMT2 | Protein−O−mannosyltransferase 2 | |
| GLI2 | GLI family zinc finger 2 | |
| ZNF335 | Zinc finger protein 335 | |
| Epilepsy syndrome | CDKL5 | Cyclin−dependent kinase−like 5 |
| CSTB | Cystatin B (stefin B) | |
| TSC1 | Tuberous sclerosis 1 | |
| CACNB4 | Calcium channel, voltage−dependent, beta 4 subunit | |
| CLCN2 | Chloride channel, voltage−sensitive 2 | |
| PCDH19 | Protocadherin 19 | |
| KCNQ2 | Potassium voltage−gated channel, KQT−like subfamily, member 2 | |
| CLN5 | Ceroid−lipofuscinosis, neuronal 5 | |
| ATP1A2 | ATPase, Na +/K + transporting, alpha 2 polypeptide | |
| LGI1 | Leucine−rich, glioma inactivated 1 | |
| SCN1B | Sodium channel, voltage−gated, type I, beta subunit | |
| ALDH5A1 | Aldehyde dehydrogenase 5 family, member A1 | |
| EFHC1 | EF−hand domain (C−terminal) containing 1 | |
| Encephalitis | DDX58 | DEAD (Asp−Glu−Ala−Asp) box polypeptide 58 |
| RPSA | Ribosomal protein SA | |
| TREX1 | Three prime repair exonuclease 1 | |
| Demyelinating disease | PLP1 | Proteolipid protein 1 |
| RTN4 | Reticulon 4 | |
| GALC | Galactosylceramidase | |
| GBS | FAS | Fas (TNF receptor superfamily, member 6) |
GO network mapping reveals connections between hNPC ZIKV infection and the immune system
A wide variety of cell types outside the traditional immune system can respond to the threat of pathogens [19]. Thus, investigations were conducted to determine if infected hNPCs presented a pattern of gene expression consistent with a defense response. To visualize the intracellular networks regulating expression changes, a GO network map was generated (Fig. 3). All significant ZIKV DEGs greater than 0.5 absolute log fold-change were loaded into Cytoscape [20], and gene ontology clustering was performed with the ClueGO plugin [21]. The human genome was used as the background with GO biological process and immune system terms queried for enrichment. GO terms for biological processes are denoted with a circle, while GO immune system terms are denoted with a triangle. As expected, nucleic acid metabolic processes were heavily enriched and connected, representing the majority of the network and indicating that they are not a directly connected to the immune system terms. Of the eight main networks identified, four were associated with immune system response. The identified networks were immune response, cytokine production, leukocyte activation, and defense response to other organisms. Additionally, cell cycle, negative regulation of macromolecule biosynthetic process, and cellular macromolecule metabolic process terms were represented.
Fig. 3.

Gene ontology functional network of biological process and immune system terms. All significantly altered genes greater than 0.5 fold-change were imported into Cytoscape and Gene Ontology clustering was performed with the ClueGO plugin. The human genome was used as the background with the GO biological process and immune system terms queried for enrichment. Terms were generated such that GO Term Fusion was implemented on pathways with a less than 0.01 q-value. Groupings with less than 3 connections were excluded from the final list of networks. Terms related to cytokine and chemokine production as well as general pathogen response formed networks independent of nucleic acid and macromolecule metabolic processes
Gene expression in ZIKV-infected hNPCs differs from the expression profile of CMV-infected hNPCs
A number of common human viruses are known to infect fetal NPCs, such as Herpes simplex virus 1 (HSV-1), CMV, human immunodeficiency virus (HIV), and Varicella-zoster virus (VZV), and are linked to the development of severe birth defects [6, 9, 11, 13, 22–24]. To explore if the alterations in immune related gene expression seen in ZIKV-infected hNPCs was consistent with other viral infections, we compared the ZIKV expression profile to that of CMV-infected human induced pluripotent stem (iPS) derived NPCs. This gene set provided well matched data for comparison, since the hNPCs in both studies were derived from iPS cells and implement Illumina transcript analysis [9, 23]. Additionally, hNPCs have demonstrated permissiveness to both viruses and infection in early fetal development with either virus has been linked to similar congenital neurological defects [9, 11, 23, 24].
The significant CMV related DEGs were analyzed in the same manner as the ZIKV related DEGs, such that the significant biological pathways are reported as fold enrichment. Clustering the pathways enriched by either CMV or ZIKV infection revealed apparent differences in the functional pathway enrichment patterns (Fig. 4). Forced clustering by functional pathway was used with hierarchical clustering within the groups implemented by rows. When hierarchical clustering is applied to columns, ZIKV up- and down-regulated gene pathways showed stronger similarity to each other than to CMV altered pathways (Fig. 4a). Examination of the cytokine/chemokine specific pathways likewise indicated divergent expression profiles between viruses (Fig. 4b). Several cytokine-regulated pathways were activated in ZIKV-infected NPCs, while CMV-infected hNPCs showed limited representation of these pathways. Interleukin-5 (IL-5), granulocyte-macrophage colony-stimulating factor (GM-CSF), and interleukin-3 (IL-3) mediated signaling were comparably enriched but associated with the down regulated genes in CMV but with the up regulated genes in the CMV set. Additionally, the pathogen response pathways highly represented in the ZIKV altered genes are not significantly enriched in either the CMV up or down regulated gene sets (Fig. 4c). While both viruses can infect hNPCs and cause comparable birth defects, the apparent mechanism by which these birth defects are caused appears to be unrelated between viruses.
Fig. 4.

Divergent gene expression patterns in CMV and ZIKV-infected hNPCs. Genes significantly different between mock and cells infected with either CMV or ZIKV were processed identically and resulting significant biological pathways were mapped. Clustering was performed on the resulting pathways such that similar functions were grouped for clarity. The Cytokine/Chemokine and Pathogen Response clusters were enlarged to demonstrate the divergent pathway enrichment between ZIKV and CMV infection
Immune and inflammatory responses in ZIKV-infected hNPCs
Given that the immune response gene enrichment seen in ZIKV-infected hNPCs differed from CMV infection, we further investigated the potential inflammatory pathways involved. Inspection of the biological pathways significantly enriched for ZIKV DEGs revealed a pattern of immunological term enrichment in genes up-regulated by the virus. Using search terms classifying immune related pathways our query returned 24 immune related pathways out of the 178 total in the ZIKV up-regulated genes (Fig. 5a). Of the 119 significantly enriched pathways of genes down-regulated by ZIKV infection, none were related to immune function (Fig. 5a). Among the inflammatory pathways identified from the up-regulated genes, were the endosomal viral pathogen recognizing Toll-like receptor (TLR) 9 and TLR7/8 pathways which recognize CpG containing DNA and ssRNA, respectively [25]. TLR receptor signaling is primarily mediated through the adaptor protein myeloid differentiation primary response gene 88 (MyD88) in response to infection [26]. Other TLRs are located on the plasma membrane and recognize a wide variety of pathogen associated molecules [25] and were thus classified as pathogen response pathways. Additionally, several canonical inflammatory pathways were activated, including tumor necrosis factor (TNF) receptor pathways, interferon gamma (IFN-γ) signaling pathway, pathways involved in interleukin (IL)-1, IL-2, IL-3, IL-5, IL-6, IL-12, IL-23, C-X-C motif chemokine 12 (CXCL12)/CXCR4, pathway and GM-CSF mediated pathway (Fig. 5b).
Fig. 5.
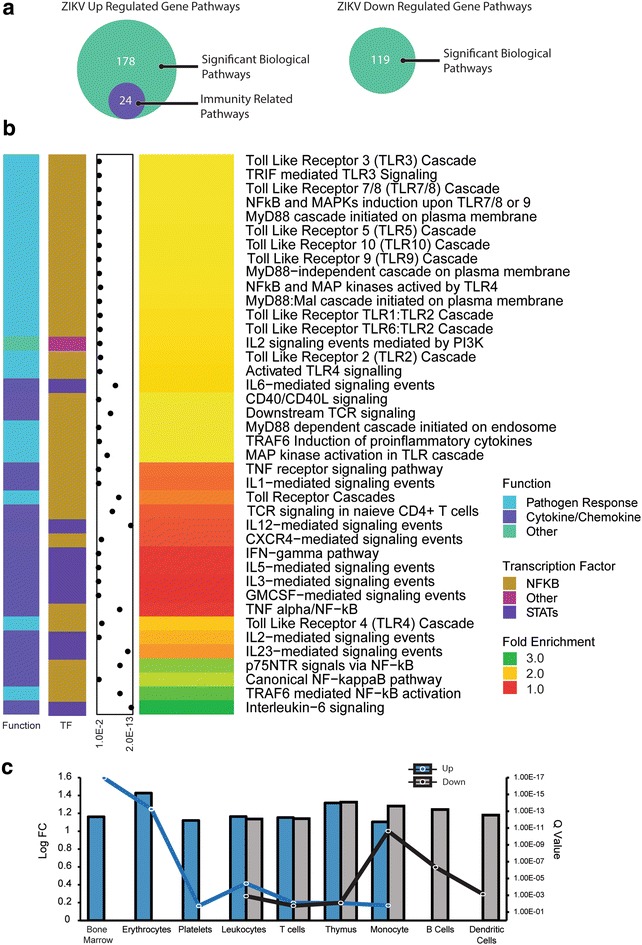
Immune specific biological pathways enriched in up-regulated genes. a The biological pathways associated with ZIKV transcript changes were filtered to include top immune specific pathways. b Using a FDR q-value <0.05, several top immune function pathways were enriched for the up-regulated gene set, although none of the pathways were enriched for significant down-regulated transcripts. The q-values for the pathway enrichment are annotated along the left panel as the dot plot. c Using tissue specific expression databases, up- and down-regulated genes were probed for immune related cells and tissues. Innate and adaptive immune system components were represented in both lists. (q < 0.05)
Cell and tissue type analysis of the whole ZIKV DEG gene set revealed representation of tissue types associated with immune and hematological function (Fig. 5c). Tissue and cell types exclusively associated with ZIKV up-regulated genes included the bone marrow, erythrocytes, and platelets. B-cells and dendritic cells were the only cell types exclusive to the down-regulated gene set. Both up- and down-regulated genes were enriched for monocytes, T-cells, leukocytes, and the thymus. The immune specific enrichment of the genes differentially expressed in ZIKV-infected hNPCs indicate involvement of both the innate and adaptive immune system. Dendritic cells are important antigen-presenting cells (APC) while T- and B-cells are primarily responsible for adaptive response. This enrichment points to a role for ZIKV inducing NPCs to take on a yet unclear immune-modulating phenotype.
Analysis of cytokine/chemokine associated intracellular signaling pathways
The two major immune cell pathways that control the production of inflammatory cytokines can be classified as being nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) and signal transducer and activator of transcription (STAT) protein family dependent. Both NF-κB and STAT family-members are transcription factors that induce gene expression upon stimulation by wide variety of ligands. Figure 6 illustrates simplified versions of these pathways activated in ZIKV-infected NPCs. The pathogens (represented as pathogen-associated molecular patterns, PAMP) and TLR 7/8/9 pathways enriched in the ZIKV up-regulated genes (Fig. 4), induce gene expression primarily through nuclear translocation of NF-κB. Other NF-κB mediated pathways enriched in ZIKV up-regulated genes were C-X-C chemokine receptor type 4 (CXCR4), CD40, TNF, and IL-1 signaling (Fig. 4). STAT family induced genes can also result in inflammatory cytokine production upon activation of plasma membrane receptors. The enriched STAT family related signaling pathways were IFN-γ, IL-6, and IL-12 in the ZIKV up-regulated genes (Fig. 4).
Fig. 6.

Activation of NF-κB and STATs signaling pathway in ZIKV-infected hNPCs. Cytokines (IL-1 and TNF-α), PAMPs, CD40 and CXCL12 interact with their receptors resulting in activation of the NF-κB pathway. IFN-γ, IL-6 and IL-12 binding to their receptors induces STAT dimerization and translocation to the nucleus. Activation of both the canonical NF-κB and STAT signaling pathways, results in the production of numerous inflammatory cytokines.
Discussion
Our results show that collectively, ZIKV induced transcriptomic alterations are associated with established clinical pathologies. Following viral infection there is a reallocation of host nucleic acids and other metabolites towards viral specific replication processes [12], thus the enrichment of biological processes related to nucleic acid metabolism is an expected finding, as well as the enrichment of cell cycle regulation. The cell cycle perturbations of infected cells suggest the initiation of a G1/S checkpoint and impaired progression through G1 [9]. A possible explanation for these cell cycle abnormalities could be inhibition of S-phase progression due to limited nucleic acid reserves as the virus reallocates them for its own replication. Down-regulation of critical epigenetic markers represents a possible driver for abnormal gene expression in infected cells. If NPCs in the developing human brain are diverted from normal development, it can result in conditions like microcephaly. Moreover, this raises questions about the possible long-term neurological effects in adult following ZIKV infection of adult neural progenitors. Although, the ZIKV associated clinical phenotypes were highly enriched for congenital CNS disorders, comparatively few genes were associated with adult CNS inflammation and demyelination diseases. This may be the due to the fact that the current study implemented the MR766 Ugandan isolate which has not been associated with an increased incidence of GBS and myelitis. As a result, future studies that employ more neoteric strains will likely observe increased representation of related genes.
The significant enrichment of canonical immune system pathways in ZIKV-infected hNPCs proved to be another unexpected finding. Our study provides evidence at the transcriptional level that two major mediators of inflammation, NF-κB and the STAT family of proteins, regulate inflammatory response in hNPCs. NF-κB is one of the main transcriptional factors activated in response to pro-inflammatory cytokines, such as TNF-α and IL-1. It is also triggered by TLRs via pathogen-associated molecular patterns (PAMPs). The Janus Kinase (JAK)-Signal transducer and activator of transcription (STAT) pathway is also a major signaling pathway that regulates inflammation and mediates the responses of target cells to inflammatory cytokines. Activation of these pathways has a central role in inflammation through the regulation of genes encoding pro-inflammatory cytokines, chemokines, and inducible enzymes such as cyclooxygenase-2 (COX2) and inducible nitric oxide synthase (iNOS) [27]. Moreover, it has recently been reported that ZIKV-infected fetal brains display a number of inflammatory markers, including diffuse astrogliosis, and activations of macrophages and microglia [9]. It is conceivable that ZIKV-infected NPCs may exert immunomodulatory and inflammatory effects on CNS cells such as neurons as well as astrocytes in an autocrine, paracrine, and juxtacrine manner, thus amplifying brain inflammation. Interestingly, although congenital CMV infection can also result in microcephaly and general impaired brain development [24], the inflammatory signals were not observed in CMV-infected hNPCs. The significance of this is still under investigation.
With the currently limited molecular data regarding transcriptomic alterations in infected host cells, large scale analysis is difficult. In spite of this, we present analysis of the currently available data in an effort to guide future studies. The RNA-seq was performed on only one infected cell type, hNPCs, thus we implemented stringent Benjamini Hochberg (BH) [28] false discovery rate (FDR) q-value cut-offs to limit false positives in enriched pathways. The use of the older MR766 isolate for the experiments likewise limits extrapolation of the results, even though it shares 88.9 % nucleotide and 96.5 % amino acid identity with the current strain [29]. Considering that reported pathogenicity has been increasing over time, the presented data likely represents a milder host response should any host transcript differences exist.
Conclusion
In conclusion, we report that ZIKV infection induces alterations to NPC immune response gene expression. While the primary infection typically occurs through the bite of an infected mosquito, the mechanism for entry into the restricted CNS is currently unclear. Presumably the specific physiology of the fetal blood brain barrier is permissible to ZIKV as it is to other viruses [24]. Regardless, it has been shown that the virus can infect NPCs with high efficiency leading to increased apoptosis [9]. In addition, the presented study is the first to report enrichment of numerous pro-inflammatory pathways in ZIKV-infected hNPCs, indicating that hNPCs are capable of activating pro-inflammatory immune system pathways following viral infections. This represents, in addition to direct activation of apoptotic pathways, a mechanism by which the virus could induce cell death via the propagation a cytotoxic pro-inflammatory environment in the CNS. Although it is possible that ZIKV can infect immune cells in a similar manner as the related Dengue virus [30–33], the ability of ZIKV to infect hNPCs may exacerbate CNS inflammation. Consequently, a better understanding of the cellular and molecular mechanisms regulation of the cross-talk between NPCs, CNS resident cells, and immune cells may be crucial for the development of effective strategies to treat ZIKV infection.
Methods
Gene sets and expression analysis
We analyzed the transcriptomic sequencing (RNA-seq) data (GEO: GSE78711) of ZIKV (MR766) infected human neural progenitor cells (hNPCs). Briefly, Tang et al. infected hNPCs derived from human induced pluripotent stem (iPS) cells with ZIKV. The infected hNPCs were incubated for 56 h, a time frame already proven sufficient for a greater than 60 % infection rate. RNA extracted from ZIKV-infected samples and mock infected samples was used for library generation and sequencing [9]. For comparison to the ZIKV-infected hNPCs, the expression data from iPCS derived NPCs infected with human CMV (Ad169) was implemented (GEO: GSE35295). The cells were infected with CMV, and the RNA was extracted 24 after infection for analysis on an Illumina HumanHT-12 V4.0 expression beadchip. Three biological replicates for control and infected cells were used, and a list of significantly altered genes were generated with a BH FDR q-value cut-off of < 0.05 [23].
Availability of data and materials
The datasets supporting the conclusions of this article are available in the gene expression omnibus (GEO) repository, http://www.ncbi.nlm.nih.gov/geo/.
Biological process and cellular compartment analysis
Using FunRich [34], up- and down-regulated genes with associated Log2 FCs were analyzed for enrichment against the human annotated genome. Biological process, cellular compartment, and biological pathway significance was defined as a FDR q-value less than 0.05. All significant biological processes were presented. All 11 significant cellular compartments for the ZIKV up-regulated genes were presented, along with the top 11 most significant pathways enriched for ZIKV down-regulated genes.
Subsetting for immune specific biological pathway enrichment
The significant biological pathways associated with both ZIKV up- and down-regulated genes were filtered such that non-immune specific biological pathways were excluded. Significance was defined as a FDR q-value less than 0.05.
ZIKV genes associated with clinical phenotypes
Clinically relevant diseases associated with ZIKV DEGs were queried using the MalaCards [17] integrated database of human diseases. The absolute percentage of ZIKV DEGs present in the reference list derived from MalaCards was reported, as well as the total number of genes in the reference list.
Gene ontology network analysis
DEGs with an absolute fold change greater than 0.5 were imported to Cytoscape [20], where Gene Ontology clustering was performed using the ClueGO plugin [21]. The human genome was used as the background, and the GO biological process and GO immune system terms were queried for enrichment. Terms were generated such that GO Term Fusion was implemented for pathways with a less than 0.01 FDR q-value. Groups with less than three connections were excluded from the final network.
R packages
Heatmaps generated with the Bioconductor package ComplexHeatmap [35] within the R statistical computing environment [36].
Authors’ contributions
AR and YR conceived and designed the study. AR analyzed the data. AR, DB, JW, RN, JF and YR interpreted the results and discussed the study. AR, DB and YR wrote the paper. All authors read and approved the final manuscript.
Acknowledgements
Thanks to Stephanie Hurwitz for review of the manuscript. This work was supported by the National Institutes of Health (R01GM100474 and R01GM072611).
Competing interests
The authors declare that they have no competing interests.
Abbreviations
- ZIKV
Zika virus
- iPSC
induced pluripotent stem cell
- hNPC
human neural progenitor cell
- CMV
cytomegalovirus
- GBS
Guillain–Barré syndrome
- GO
gene ontology
- FC
fold-change
- MOZ/MORF
monocytic leukemia zinc-finger protein/related factor
- DEG
differentially expressed genes
- CNS
central nervous system
- HSV-1
herpes simplex virus 1
- HIV
human immunodeficiency virus
- VZV
varicella-zoster virus
- GM-CSF
granulocyte–macrophage colony stimulating factor
- IL
interleukin
- TLR
toll-like receptor
- MyD88
myeloid differentiation primary response gene 88
- TNF
tumor necrosis factor
- IFN
interferon
- CXC
C-X-C motif chemokine
- APC
antigen-presenting cells
- STAT
signal transducer and activator of transcription
- NF-κB
nuclear factor kappa-light-chain-enhancer of activated B cells
- PAMP
pathogen-associated molecular patterns
- JAK
Janus kinase
- COX2
cyclooxygenase-2
- iNOS
inducible nitric oxide synthase
Contributor Information
Alyssa J. Rolfe, Email: alyssa.rolfe@med.fsu.edu
Dale B. Bosco, Email: dale.bosco@med.fsu.edu
Jingying Wang, Email: jingying.wang0618@gmail.com.
Richard S. Nowakowski, Email: richard.nowakowski@med.fsu.edu
Jianqing Fan, Email: jqfan@princeton.edu.
Yi Ren, Phone: 850 645 2013, Email: yi.ren@med.fsu.edu.
References
- 1.Faye O, Freire CC, Iamarino A, Faye O, de Oliveira JV, Diallo M, et al. Molecular evolution of Zika virus during its emergence in the 20(th) century. PLoS Negl Trop Dis. 2014;8(1):e2636. doi: 10.1371/journal.pntd.0002636. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Lazear HM, Diamond MS. Zika virus: new clinical syndromes and its emergence in the western hemisphere. J Virol. 2016 doi: 10.1128/JVI.00252-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Cao-Lormeau V-M, Blake A, Mons S, Lastère S, Roche C, Vanhomwegen J, et al. Guillain-Barre syndrome outbreak associated with Zika virus infection in French Polynesia: a case-control study. Lancet. 2016;387(10027):1531–1539. doi: 10.1016/S0140-6736(16)00562-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Zammarchi L, Tappe D, Fortuna C, Remoli ME, Gunther S, Venturi G, et al. Zika virus infection in a traveller returning to Europe from Brazil, March 2015. Euro Surveill. 2015;20(23). [DOI] [PubMed]
- 5.Olson JG, Ksiazek TG, Suhandiman, Triwibowo Zika virus, a cause of fever in Central Java, Indonesia. Trans R Soc Trop Med Hyg. 1981;75(3):389–393. doi: 10.1016/0035-9203(81)90100-0. [DOI] [PubMed] [Google Scholar]
- 6.Ventura CV, Maia M, Bravo-Filho V, Góis AL, Belfort R., Jr Zika virus in Brazil and macular atrophy in a child with microcephaly. Lancet. 2016;387(10015):228. doi: 10.1016/S0140-6736(16)00006-4. [DOI] [PubMed] [Google Scholar]
- 7.Mlakar J, Korva M, Tul N, Popović M, Poljšak-Prijatelj M, Mraz J, et al. Zika virus associated with microcephaly. N Engl J Med. 2016;374(10):951–958. doi: 10.1056/NEJMoa1600651. [DOI] [PubMed] [Google Scholar]
- 8.Kleber de Oliveira W, Cortez-Escalante J, De Oliveira WT, do Carmo GM, Henriques CM, Coelho GE, et al. Increase in reported prevalence of microcephaly in infants born to women living in areas with confirmed Zika virus transmission during the first trimester of pregnancy—Brazil, 2015. MMWR Morb Mortal Wkly Rep. 2016;65(9):242–247. doi: 10.15585/mmwr.mm6509e2. [DOI] [PubMed] [Google Scholar]
- 9.Tang H, Hammack C, Ogden Sarah C, Wen Z, Qian X, Li Y, et al. Zika virus infects human cortical neural progenitors and attenuates their growth. Cell Stem Cell. 2016 doi: 10.1016/j.stem.2016.02.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Dollard SC, Grosse SD, Ross DS. New estimates of the prevalence of neurological and sensory sequelae and mortality associated with congenital cytomegalovirus infection. Rev Med Virol. 2007;17(5):355–363. doi: 10.1002/rmv.544. [DOI] [PubMed] [Google Scholar]
- 11.Luo MH, Hannemann H, Kulkarni AS, Schwartz PH, O’Dowd JM, Fortunato EA. Human cytomegalovirus infection causes premature and abnormal differentiation of human neural progenitor cells. J Virol. 2010;84(7):3528–3541. doi: 10.1128/JVI.02161-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Martin EM, Malec J, Sved S, Work TS. Studies on protein and nucleic acid metabolism in virus-infected mammalian cells. 1. Encephalomyocarditis virus in Krebs II mouse-ascites-tumour cells. Biochem J. 1961;80(3):585–597. doi: 10.1042/bj0800585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Garcez PP, Loiola EC, Madeiro da Costa R, Higa LM, Trindade P, Delvecchio R, et al. Zika virus impairs growth in human neurospheres and brain organoids. Science. 2016 doi: 10.1126/science.aaf6116. [DOI] [PubMed] [Google Scholar]
- 14.Mahmood S, Ahmad W, Hassan MJ. Autosomal recessive primary microcephaly (MCPH): clinical manifestations, genetic heterogeneity and mutation continuum. Orphanet J Rare Dis. 2011;6:39. doi: 10.1186/1750-1172-6-39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Bond J, Roberts E, Springell K, Lizarraga SB, Scott S, Higgins J, et al. A centrosomal mechanism involving CDK5RAP2 and CENPJ controls brain size. Nat Genet. 2005;37(4):353–355. doi: 10.1038/ng1539. [DOI] [PubMed] [Google Scholar]
- 16.Yang XJ, Ullah M. MOZ and MORF, two large MYSTic HATs in normal and cancer stem cells. Oncogene. 2007;26(37):5408–5419. doi: 10.1038/sj.onc.1210609. [DOI] [PubMed] [Google Scholar]
- 17.Rappaport N, Nativ N, Stelzer G, Twik M, Guan-Golan Y, Iny Stein T, et al. MalaCards: an integrated compendium for diseases and their annotation. Database (Oxford). 2013;bat018. [DOI] [PMC free article] [PubMed]
- 18.Aggarwal A, Mittal H, Patil R, Debnath S, Rai A. Clinical profile of children with developmental delay and microcephaly. J Neurosci Rural Pract. 2013;4(3):288–291. doi: 10.4103/0976-3147.118781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Anders HJ, Banas B, Schlondorff D. Signaling danger: toll-like receptors and their potential roles in kidney disease. J Am Soc Nephrol. 2004;15(4):854–867. doi: 10.1097/01.ASN.0000121781.89599.16. [DOI] [PubMed] [Google Scholar]
- 20.Shannon P, Markiel A, Ozier O, Baliga NS, Wang JT, Ramage D, et al. Cytoscape: a software environment for integrated models of biomolecular interaction networks. Genome Res. 2003;13(11):2498–2504. doi: 10.1101/gr.1239303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Bindea G, Mlecnik B, Hackl H, Charoentong P, Tosolini M, Kirilovsky A, et al. ClueGO: a cytoscape plug-into decipher functionally grouped gene ontology and pathway annotation networks. Bioinformatics. 2009;25(8):1091–1093. doi: 10.1093/bioinformatics/btp101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Nowakowski TJ, Pollen AA, Di Lullo E, Sandoval-Espinosa C, Bershteyn M, Kriegstein AR. Expression analysis highlights AXL as a candidate Zika virus entry receptor in neural stem cells. Cell Stem Cell. 2016 doi: 10.1016/j.stem.2016.03.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.D’Aiuto L, Di Maio R, Heath B, Raimondi G, Milosevic J, Watson AM, et al. Human induced pluripotent stem cell-derived models to investigate human cytomegalovirus infection in neural cells. PLoS One. 2012;7(11):e49700. doi: 10.1371/journal.pone.0049700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Cheeran MCJ, Lokensgard JR, Schleiss MR. Neuropathogenesis of congenital cytomegalovirus infection: disease mechanisms and prospects for intervention. Clin Microbiol Rev. 2009;22(1):99–126. doi: 10.1128/CMR.00023-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Takeda K, Akira S. TLR signaling pathways. Curr Protoc Immunol. 2004;16(1):3–9. doi: 10.1016/j.smim.2003.10.003. [DOI] [PubMed] [Google Scholar]
- 26.Akira S, Takeda K. Toll-like receptor signalling. Nat Rev Immunol. 2004;4(7):499–511. doi: 10.1038/nri1391. [DOI] [PubMed] [Google Scholar]
- 27.Baud V, Karin M. Is NF-[kappa]B a good target for cancer therapy? hopes and pitfalls. Nat Rev Drug Discov. 2009;8(1):33–40. doi: 10.1038/nrd2781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Benjamini Y, Hochberg Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc Series B Stat Methodol. 1995 [Google Scholar]
- 29.Lanciotti RS, Kosoy OL, Laven JJ, Velez JO, Lambert AJ, Johnson AJ, et al. Genetic and serologic properties of Zika virus associated with an epidemic, Yap State, Micronesia, 2007. Emerg Infect Dis. 2008;14(8):1232–1239. doi: 10.3201/eid1408.080287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Simmons CP, Farrar JJ, van Vinh Chau N, Wills B. Dengue. N Engl J Med. 2012;366(15):1423–1432. doi: 10.1056/NEJMra1110265. [DOI] [PubMed] [Google Scholar]
- 31.Nakao S, Lai CJ, Young NS. Dengue virus, a flavivirus, propagates in human bone marrow progenitors and hematopoietic cell lines. Blood. 1989;74(4):1235–1240. [PubMed] [Google Scholar]
- 32.Kelley JF, Kaufusi PH, Volper EM, Nerurkar VR. Maturation of dengue virus nonstructural protein 4B in monocytes enhances production of dengue hemorrhagic fever-associated chemokines and cytokines. Virology. 2011;418(1):27–39. doi: 10.1016/j.virol.2011.07.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.La Russa VF, Innis BL. Mechanisms of dengue virus-induced bone marrow suppression. Baillieres Clin Haematol. 1995;8(1):249–270. doi: 10.1016/S0950-3536(05)80240-9. [DOI] [PubMed] [Google Scholar]
- 34.Pathan M, Keerthikumar S, Ang CS, Gangoda L, Quek CY, Williamson NA, et al. FunRich: an open access standalone functional enrichment and interaction network analysis tool. Proteomics. 2015;15(15):2597–2601. doi: 10.1002/pmic.201400515. [DOI] [PubMed] [Google Scholar]
- 35.Zuguang G. ComplexHeatmap: making complex heatmaps. R package version 1.6.0 ed2015.
- 36.R Core Team . R: a language and environment for statistical computing. Vienna: R Foundation for Statistical Computing; 2015. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets supporting the conclusions of this article are available in the gene expression omnibus (GEO) repository, http://www.ncbi.nlm.nih.gov/geo/.


