Abstract
Crude lecithin, a mixture of mainly phospholipids, potentially helps to increase the systemic availability of dietary omega-3 polyunsaturated fatty acids (n-3 PUFA), such as docosahexaenoic acid (DHA). Nevertheless, no clear data exist on the effects of prolonged combined dietary supplementation of DHA and lecithin on RBC and plasma PUFA levels. In the current experiments, levels of DHA and choline, two dietary ingredients that enhance neuronal membrane formation and function, were determined in plasma and red blood cells (RBC) from rats after dietary supplementation of DHA-containing oils with and without concomitant dietary supplementation of crude lecithin for 2–3 weeks. The aim was to provide experimental evidence for the hypothesized additive effects of dietary lecithin (not containing any DHA) on top of dietary DHA on PUFA levels in plasma and RBC. Dietary supplementation of DHA-containing oils, either as vegetable algae oil or as fish oil, increased DHA, eicosapentaenoic acid (EPA), and total n-3 PUFA, and decreased total omega-6 PUFA levels in plasma and RBC, while dietary lecithin supplementation alone did not affect these levels. However, combined dietary supplementation of DHA and lecithin increased the changes induced by DHA supplementation alone. Animals receiving a lecithin-containing diet also had a higher plasma free choline concentration as compared to controls. In conclusion, dietary DHA-containing oils and crude lecithin have synergistic effects on increasing plasma and RBC n-3 PUFA levels, including DHA and EPA. By increasing the systemic availability of dietary DHA, dietary lecithin may increase the efficacy of DHA supplementation when their intake is combined.
Keywords: Phospholipids, Docosahexaenoic acid, Plasma lipids, n-3 Fatty acids, Nutrition, Fish oil, Algal lipids
Introduction
Omega-3 polyunsaturated fatty acids (n-3 PUFA), such as docosahexaenoic acid (DHA), are important structural components of neuronal membranes and are functionally implied in various membrane-bound processes [1, 2]. Dietary supplementation of n-3 PUFA raises blood n-3 PUFA levels rapidly [3, 4] and increases their uptake into the brain and therefore their availability for incorporation into neuronal membranes [5, 6]. Thus, neuronal membrane structure and function are subject to alterations induced by nutritional compounds such as n-3 PUFA.
As a result of its biochemical and biophysical properties, dietary lecithin may help to increase the availability of dietary n-3 PUFA, with subsequent effects on neuronal membrane structure and function. Crude lecithin is a mixture of mainly phospholipids (up to approximately 75 % for de-oiled lecithin), i.e., phosphatidylcholine (PtdCho), phosphatidylethanolamine (PtdEtn), and phosphatidylinositol (PtdIns), and a smaller fraction of glycolipids, neutral lipids, and carbohydrates, that can be extracted from plant or animal food substances. Lecithin has low solubility in water, but is a very good emulsifier and is also a source of active compounds, such as choline, that can be released from PtdCho. Theoretically, dietary lecithin (i.e., phospholipids) could increase systemic availability of dietary n-3 PUFA, since (biliary) phospholipids are required for the absorption of fat from the gut lumen into the enterocytes and the lymph [7, 8]. Several preclinical studies have examined the potential additive effects of lecithin or phospholipids on the bioavailability of triglycerides after oral intake [9–15]. However, most of these studies investigated (1) post-prandial effects of a single dose and/or (2) effects on lymphatic accretion and/or (3) effects of emulsification, i.e., whether pre-emulsifying oil with lecithin enhances the oil’s bioavailability. For example, recently Couëdelo et al. showed in rats that the lymphatic accretion after single intra-gastric administration of an oil containing the omega-3 fatty acid alpha-linolenic acid (ALA) could be increased by pre-emulsifying the oil with lecithin [14]. No distinct data exist on the effects of prolonged combined dietary supplementation of n-3 PUFA, especially DHA, and phospholipids on plasma and red blood cell (RBC) PUFA levels. Systemic availability is functionally relevant as the availability of n-3 PUFA to tissues such as the brain is largely dependent on their levels in circulation.
The objective of the present study was to test the hypothesized additive effects of dietary crude lecithin (which itself does not contain DHA) on top of dietary supplementation with DHA-containing oils. Experimental diets were presented to rats for 2–3 weeks after which relative and quantitative plasma levels and relative RBC levels of DHA, eicosapentaenoic acid (EPA), total n-3 PUFA, and total omega-6 (n-6) PUFA were determined. Plasma choline was measured to confirm that dietary lecithin, being a source of PtdCho, increases plasma choline concentration.
Materials and Methods
To investigate the effects of dietary lecithin on the systemic availability of dietary DHA in adult rats, two experiments were conducted at the Centrum Kleine Proefdieren, Wageningen University (Wageningen, The Netherlands).
Animals
Twenty-four male Wistar rats (HsdCpb:WU) (Experiment A) and 24 Sprague–Dawley rats (Hsd:SD) (Experiment B) were obtained from Harlan (Harlan Nederland, Horst, The Netherlands). Animals aged 10 weeks on arrival were housed in groups in a temperature- and light-controlled room, under 12 h light–12 h dark cycles. Rats had free access to experimental diets and water. Body weight and food-intake was recorded at least once a week. All experimental protocols were conducted in accordance with international and national laws and institutional guidelines and approved by the local ethics committee [DEC Consult, Bilthoven, The Netherlands, protocol numbers DEC Nr. NR117 (Experiment A) and DEC Nr. DAN0190 (Experiment B)].
Diets
In Experiment A, four grain-based experimental diets with varying levels of vegetable algae DHA oil and crude lecithin were used: (1) control; (2) lecithin; (3) vegetable DHA oil; (4) vegetable DHA oil + lecithin. In Experiment B, three AIN-93M-based diets were used with varying levels of fish oil and crude lecithin: (1) control; (2) fish oil; (3) fish oil + lecithin. Within each experiment the diets were isocaloric and identical with respect to their protein, carbohydrate, fiber, and mineral contents. The diets fulfilled minimal dietary requirements for rats and were presented to the animals as pellets. The diets differed in composition with regard to the fat blends used, as well as the amount of lecithin added. The control diets and the lecithin diet did not contain DHA or EPA and the control diets did not contain any added crude lecithin. The lecithin-containing diets in Experiment A were supplemented with 400 mg de-oiled crude soy lecithin/100 g diet (0.4 %, Emulpur IP, Cargill Texturizing Solutions, The Netherlands), whereas the amount of de-oiled crude lecithin added to the lecithin-containing diet in Experiment B was 1000 mg/100 g diet (1.0 %). Average daily intake of crude lecithin per body weight was calculated based on the respective measured average body weight and food intake and was approximately 250 mg crude lecithin/kg BW/day (Experiment A) and 550 mg crude lecithin/kg BW/day (Experiment B). The de-oiled soy crude lecithin mainly contained phospholipids (77 g phospholipids/100 g lecithin) of which mainly PtdCho (20 g/100 g lecithin), PtdIns (14 g/100 g lecithin), and PtdEtn (13 g/100 g lecithin, as provided by the supplier), with mainly linoleic acid (18:2n-6), palmitic acid (16:0), and oleic acid (18:1n-9), and did not contain any DHA. The total fat content of the fish oil + lecithin diet in Experiment B was corrected for the fat content of the added lecithin. In Experiment A, the amount of fat from lecithin was considered to be negligible. The DHA content of the diets containing vegetable algae DHA oil (DHASCO oil, Martek Biosciences Corporation, USA) in Experiment A and the diets containing fish oil (Tuna RoPUFA, Nu-Mega, Australia) in Experiment B was equal and moderate, namely 0.2 DHA g/100 g. Average daily intake of the DHA per body weight was calculated based on the respective measured average body weight and food intake and was approximately 125 mg DHA/kg BW/day (Experiment A) and 110 mg DHA/kg BW/day (Experiment B). Average daily intake of the EPA per body weight was approximately 31 mg EPA/kg BW/day (Experiment A) and 27 mg EPA/kg BW/day (Experiment B). A detailed overview of the contents of diets is presented in Table 1. Diets were manufactured by Research Diet Services, Wijk bij Duurstede, The Netherlands (Experiment A) and Ssniff Spezialdiäten, Soest, Germany (Experiment B) and were stored at −20 °C until use, in order to prevent oxidation of lipids. Analysis of the diets confirmed the calculated fatty acid composition and confirmed that the control diets and the lecithin diet did not contain DHA or EPA.
Table 1.
Detailed compositions of the experimental diets used in Experiment A and Experiment B
| Ingredients (g/100 g diet) | Diets Experiment A | |||
|---|---|---|---|---|
| Control | Lecithin | Vegetable DHA oil | Vegetable DHA oil + lecithin | |
| Wheat | 25.87 | 25.87 | 25.87 | 25.87 |
| Barley | 25.00 | 25.00 | 25.00 | 25.00 |
| Semolina | 25.00 | 24.60 | 25.00 | 24.60 |
| Soybean meal | 8.50 | 8.50 | 8.50 | 8.50 |
| Bentonite | 1.00 | 1.00 | 1.00 | 1.00 |
| Whey powder | 5.00 | 5.00 | 5.00 | 5.00 |
| Soy oil | 1.90 | 1.90 | 1.76 | 1.76 |
| Corn oil | 2.20 | 2.20 | 1.90 | 1.90 |
| Coconut fat | 0.90 | 0.90 | 0.80 | 0.80 |
| Vegetable algae DHA oil (DHASCO oil) | – | – | 0.54 | 0.54 |
| Vitamin/mineral mix (including choline) | 2.20 | 2.20 | 2.20 | 2.20 |
| CaCO3 | 1.40 | 1.40 | 1.40 | 1.40 |
| Dicalcium phosphate | 0.30 | 0.30 | 0.30 | 0.30 |
| NaCl | 0.50 | 0.50 | 0.50 | 0.50 |
| l-Lysine HCl | 0.18 | 0.18 | 0.18 | 0.18 |
| dl-Methionine | 0.05 | 0.05 | 0.05 | 0.05 |
| Soy lecithin (Emulpur IP) | – | 0.40 | – | 0.40 |
| Sum | 100.00 | 100.00 | 100.00 | 100.00 |
| Ingredients (g/100 g diet) | Diets Experiment B | ||
|---|---|---|---|
| Control | Fish oil | Fish oil + lecithin | |
| Cornstarch, pre-gelatinized | 34.59 | 34.59 | 34.34 |
| Maltodextrin, 10 DE | 15.50 | 15.50 | 15.50 |
| Sucrose | 10.00 | 10.00 | 10.00 |
| Dextrose | 10.00 | 10.00 | 10.00 |
| Cellulose powder | 5.00 | 5.00 | 5.00 |
| Casein | 14.00 | 14.00 | 14.00 |
| Soy oil | 3.00 | 3.00 | 2.25 |
| Corn oil | 2.10 | 1.50 | 1.50 |
| Coconut fat | 0.90 | 0.70 | 0.70 |
| Fish oil (Tuna RoPUFA) | – | 0.80 | 0.80 |
| Mineral mix (AIN-93M-MX) | 3.50 | 3.50 | 3.50 |
| Vitamin mix (AIN-93-VX) | 1.00 | 1.00 | 1.00 |
| Choline chloride (50 %) | 0.23 | 0.23 | 0.23 |
| l-Cystine | 0.18 | 0.18 | 0.18 |
| Tert-butylhydroquinone | 0.0008 | 0.0008 | 0.0008 |
| Soy lecithin (Emulpur IP) | – | – | 1.00 |
| Sum | 100.00 | 100.00 | 100.00 |
Experimental Design
In both experiments, all rats were provided with the respective control diet for 2 weeks before starting the intervention. Subsequently, animals were divided into the experimental groups that were matched according to their body weight and food intake and were placed on one of the experimental diets for 2 weeks (Experiment A) or 3 weeks (Experiment B).
Sample Preparation
After the supplementation period, animals were food-deprived for 3–4 h and euthanized by inhalation of isoflurane and subsequent decapitation by guillotine. Trunk blood was collected through a funnel into EDTA-containing tubes. After centrifugation at 1300×g at 20 °C for 10 min (Experiment A) or 1750×g at 4 °C for 10 min (Experiment B), plasma and RBC (Experiment B only) were collected and analyzed for plasma fatty acid composition (Experiments A and B) and for RBC fatty acid composition and free choline (Experiment B only). Plasma sample preparation in Experiment A was not adequate for choline analysis as plasma free choline is not stable at room temperature and therefore these results are lacking. Based on the results of Experiment A, RBC samples were also collected in Experiment B.
Plasma Fatty Acid Composition and Plasma Free Choline Analyses
Plasma and RBC total lipid fatty acid composition was detected using gas chromatography (GC). Total lipids were extracted from plasma and RBC using a modified Bligh and Dyer protocol [16] by adding methanol, 1 % EDTA solution, and dichloromethane to 100 µL plasma or approximately 150 µL RBC (non-quantitative). After vortexing for 5 min, samples were centrifuged at 1750×g for 10 min and the organic phase (dichloromethane and lipids) was collected. The dichloromethane layer was dried using a SpeedVac® concentrator. Next, 2.0 mL methanol and 40 μL concentrated sulfuric acid (2 v/v %) were added to the dried extract and samples were heated at 100 °C for 60 min [17]. After cooling, 2 mL hexane and 0.5 mL 2.5 mol/L sodium hydroxide solution were added. Samples were subsequently vortexed and centrifuged for 5 min at 1750×g after which the upper layer was collected and dried using a SpeedVac®. Dried samples were subsequently dissolved in 125 μL iso-octane and analyzed by GC (Shimadzu Corporation, Kyoto, Japan) using flame ionization detection with a CP-SIL88 column (50 m × 0.25 mm id. 0.20 μm film thickness; Agilent Technologies, Inc., Santa Clara, CA, USA). The carrier gas was hydrogen and the make-up gas was nitrogen. GC oven temperature settings were as follows: initial temperature 150 °C for 3.75 min; temperature increment to 220 °C at 22 °C/min; and subsequent constant temperature of 220 °C for 14.07 min (in-house settings). Fatty acids were identified based on retention time using an external reference standard (GLC-461, Nu-Chek Prep, Inc., Elysian, MN, USA). Peak area was used as a measure of relative percent (mol%). An internal standard (1,2-dinonadecanoyl-sn-glycero-3-phosphocholine, Avanti Polar Lipids, Inc., AL, USA) was used for absolute quantification of fatty acids (µmol/L) in plasma. RBC fatty acids were not quantitatively pipetted and were therefore not absolutely quantified.
High performance liquid chromatography (HPLC) coupled to electrochemical detection of plasma free choline was performed using a commercially available choline assay HPLC method from Thermo Fisher Scientific (former ESA Biosciences, Chelmsford, MA, USA). Briefly, after protein precipitation, samples were centrifuged to remove proteins. The supernatant was injected into the HPLC (Thermo Fisher Scientific, former ESA Biosciences, Chelmsford, MA, USA) using a post-column immobilized enzyme reactor, in an on-line enzyme reaction to produce H2O2, which was detected electrochemically.
Statistical Analyses
All statistical analyses were performed using SPSS (version 19, SPSS Inc., Chicago, IL, USA). Data were expressed as means ± SEM. P values <0.05 were considered significant. Effects of dietary treatments on body weight and food intake were analyzed using repeated-measures ANOVA with Diet (Experiment A) or dietary DHA and dietary lecithin (Experiment B) as the between-subject factor(s) and day as the within-subject factor. Plasma and RBC fatty acids relative and quantitative levels (DHA, EPA, total n-3 PUFA and n-6 PUFA) and choline concentrations were compared between rats fed the different experimental diets using two-way ANOVA with dietary DHA and dietary lecithin as between-subject factors (Experiment A) or one-way ANOVA (Experiment B). Post hoc comparisons were performed where appropriate.
Results
Experiment A
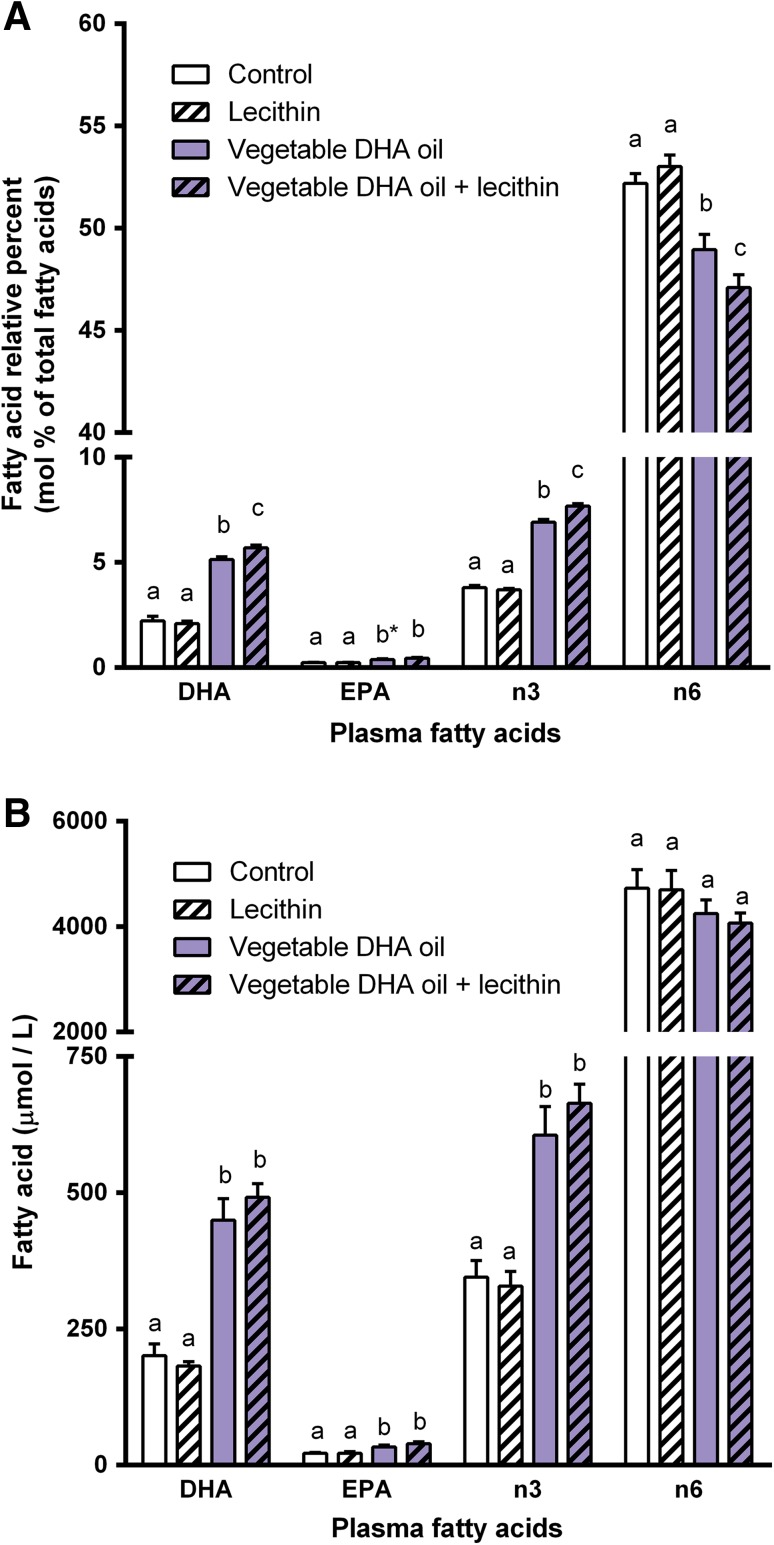
After feeding all animals the control diet for 2 weeks, they were divided into four experimental groups that were matched according to their body weight and food intake. During the supplementation period of 2 weeks body weight was neither affected by dietary DHA supplementation (P = 0.55) nor by dietary lecithin supplementation (P = 0.72). Also no effects on food intake were found (dietary DHA: P = 0.89); dietary lecithin: P = 0.99). Dietary DHA supplementation increased relative plasma levels of DHA (P < 0.001), EPA (P < 0.001), and total n-3 PUFA (P < 0.001), and decreased total n-6 PUFA (P < 0.001). Plasma levels of DHA (P = 0.20), EPA (P = 0.21), and total n-6 PUFA (P = 0.41) were not affected by dietary lecithin supplementation, whereas plasma total n-3 PUFA level (P = 0.007) was affected. Furthermore, interactions between dietary DHA and lecithin supplementation were found for plasma DHA (P = 0.039), total n-3 PUFA (P = 0.001), and total n-6 PUFA (P = 0.041), however not for EPA (P = 0.19). These main and interaction effects indicate that combined dietary supplementation of DHA oil and lecithin increased the changes induced by DHA supplementation alone (Fig. 1a). Quantitative plasma concentrations of DHA (P < 0.001), EPA (P < 0.001), and total n-3 PUFA (P < 0.001) were also affected by dietary DHA supplementation, whereas total n-6 PUFA (P = 0.078) was not. No effects of dietary lecithin or interactions between dietary DHA and lecithin were found when expressing the plasma fatty acid levels as quantitative concentrations, although trends were apparent (Fig. 1b). Tables 2 and 3 give a complete overview of all plasma fatty acids measured, expressed as relative percent levels (Table 2) and quantitative concentrations (Table 3).
Fig. 1.
Experiment A: effects of dietary supplementation of vegetable DHA oil, lecithin or the combination of both on a relative percent levels (mol%) and b quantitative concentrations (µmol/L) of DHA, EPA, total n-3 PUFA, and total n-6 PUFA in plasma of rats. Plasma DHA, EPA, total n-3 PUFA, and total n-6 PUFA levels in rats that received one of the four experimental diets (control, lecithin, vegetable DHA oil, vegetable DHA oil + lecithin) for 2 weeks. Values are means, with the SEM represented by vertical bars. Different letters indicate mean values were significantly different (P < 0.05). *P = 0.077 vs. vegetable DHA oil + lecithin. n = 6 per experimental group
Table 2.
Experiment A: effects of dietary supplementation of vegetable DHA oil, lecithin or the combination of both on plasma fatty acid composition (mol%) in rats
| Plasma fatty acids (mol% of total fatty acids) | Experiment A | |||
|---|---|---|---|---|
| Dietary intervention | ||||
| Control | Lecithin | Vegetable DHA oil | Vegetable DHA oil + lecithin | |
| 6:0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 |
| 8:0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 |
| 10:0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 |
| 12:0 | 0.6 ± 0.2 | 0.7 ± 0.1 | 0.7 ± 0.1 | 0.6 ± 0.1 |
| 14:0 | 0.9 ± 0.1 | 0.9 ± 0.1 | 1.0 ± 0.1 | 1.0 ± 0.1 |
| 15:0 | 0.3 ± 0.0 | 0.3 ± 0.0 | 0.3 ± 0.0 | 0.3 ± 0.0 |
| 16:0 | 18.6 ± 0.2 | 18.6 ± 0.2 | 19.1 ± 0.3 | 19.8 ± 0.2 |
| 17:0 | 0.3 ± 0.0 | 0.3 ± 0.0 | 0.3 ± 0.0 | 0.3 ± 0.0 |
| 18:0 | 7.8 ± 0.4 | 7.6 ± 0.5 | 7.4 ± 0.4 | 7.1 ± 0.3 |
| 20:0 | 0.1 ± 0.0 | 0.1 ± 0.0 | 0.1 ± 0.0 | 0.1 ± 0.0 |
| 22:0 | 0.2 ± 0.0 | 0.2 ± 0.0 | 0.2 ± 0.0 | 0.2 ± 0.0 |
| 23:0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 |
| 24:0 | 0.2 ± 0.0 | 0.2 ± 0.0 | 0.2 ± 0.0 | 0.2 ± 0.0 |
| 16:1n-7 | 1.0 ± 0.1 | 0.8 ± 0.1 | 1.1 ± 0.1 | 1.3 ± 0.2 |
| 18:1n-7 | 2.2 ± 0.1 | 2.0 ± 0.1 | 2.1 ± 0.2 | 2.3 ± 0.2 |
| 18:1n-9 | 11.2 ± 0.5 | 10.9 ± 0.5 | 11.1 ± 0.5 | 11.5 ± 0.4 |
| 20:1n-9 | 0.3 ± 0.0 | 0.3 ± 0.0 | 0.3 ± 0.0 | 0.3 ± 0.0 |
| 22:1n-9 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 |
| 24:1n-9 | 0.1 ± 0.0 | 0.1 ± 0.0 | 0.1 ± 0.0 | 0.1 ± 0.0 |
| 20:3n-9 | 0.1 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 |
| 18:2n-6 | 26.5 ± 1.4 | 28.3 ± 1.0 | 27.6 ± 0.7 | 28.1 ± 0.8 |
| 18:3n-6 | 0.4 ± 0.0 | 0.3 ± 0.0 | 0.2 ± 0.0 | 0.2 ± 0.0 |
| 20:2n-6 | 0.6 ± 0.0 | 0.6 ± 0.0 | 0.5 ± 0.0 | 0.6 ± 0.0 |
| 20:3n-6 | 0.6 ± 0.1 | 0.6 ± 0.0 | 0.7 ± 0.0 | 0.8 ± 0.0 |
| 20:4n-6 | 22.4 ± 1.5 | 21.8 ± 1.5 | 19.1 ± 1.3 | 16.6 ± 1.1 |
| 22:4n-6 | 1.1 ± 0.1 | 1.0 ± 0.1 | 0.5 ± 0.0 | 0.6 ± 0.0 |
| 22:5n-6 | 0.6 ± 0.1 | 0.4 ± 0.0 | 0.2 ± 0.0 | 0.2 ± 0.0 |
| 24:2n-6 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 |
| 18:3n-3 | 0.7 ± 0.1 | 0.8 ± 0.1 | 0.9 ± 0.1 | 0.9 ± 0.1 |
| 18:4n-3 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 |
| 20:5n-3 | 0.2 ± 0.0 | 0.2 ± 0.0 | 0.4 ± 0.0 | 0.5 ± 0.0 |
| 22:5n-3 | 0.6 ± 0.1 | 0.5 ± 0.0 | 0.5 ± 0.0 | 0.6 ± 0.0 |
| 22:6n-3 | 2.2 ± 0.2 | 2.1 ± 0.1 | 5.1 ± 0.1 | 5.7 ± 0.1 |
| Total SFA | 29.1 ± 0.2 | 29.1 ± 0.1 | 29.4 ± 0.3 | 29.6 ± 0.2 |
| Total MUFA | 14.8 ± 0.5 | 14.2 ± 0.7 | 14.7 ± 0.8 | 15.5 ± 0.6 |
| Total PUFA | 56.1 ± 0.5 | 56.7 ± 0.6 | 55.9 ± 0.7 | 54.8 ± 0.6 |
| Total n-3 PUFA | 3.8 ± 0.1 | 3.7 ± 0.1 | 6.9 ± 0.1 | 7.7 ± 0.1 |
| Total n-6 PUFA | 52.2 ± 0.5 | 53.0 ± 0.6 | 49.0 ± 0.7 | 47.1 ± 0.6 |
| Total fatty acids | 100.0 | 100.0 | 100.0 | 100.0 |
Values are means ± SEM
Table 3.
Experiment A: effects of dietary supplementation of vegetable DHA oil, lecithin or the combination of both on plasma fatty acid quantitative concentrations (µmol/L) in rats
| Plasma fatty acids (µmol/L) | Experiment A | |||
|---|---|---|---|---|
| Dietary intervention | ||||
| Control | Lecithin | Vegetable DHA oil | Vegetable DHA oil + lecithin | |
| 6:0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 |
| 8:0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 |
| 10:0 | 2.3 ± 1.2 | 2.4 ± 0.9 | 2.7 ± 0.6 | 1.9 ± 0.6 |
| 12:0 | 57.9 ± 18.5 | 70.2 ± 18.6 | 65.4 ± 10.8 | 51.6 ± 6.6 |
| 14:0 | 80.5 ± 15.3 | 80.9 ± 14.2 | 88.0 ± 10.9 | 86.9 ± 7.0 |
| 15:0 | 27.0 ± 2.5 | 29.7 ± 3.0 | 25.8 ± 2.2 | 26.8 ± 1.3 |
| 16:0 | 1700.2 ± 160.4 | 1658.4 ± 152.8 | 1669.9 ± 130.7 | 1711.4 ± 92.3 |
| 17:0 | 28.2 ± 1.5 | 29.3 ± 1.1 | 26.6 ± 1.6 | 26.3 ± 1.4 |
| 18:0 | 698.2 ± 35.0 | 661.1 ± 17.6 | 639.6 ± 32.0 | 615.4 ± 38.9 |
| 20:0 | 9.6 ± 1.2 | 10.2 ± 1.4 | 9.4 ± 0.8 | 8.5 ± 0.4 |
| 22:0 | 18.1 ± 1.7 | 17.6 ± 1.8 | 14.1 ± 1.1 | 14.5 ± 0.4 |
| 23:0 | 3.2 ± 0.8 | 3.2 ± 0.5 | 2.9 ± 0.5 | 3.1 ± 0.2 |
| 24:0 | 18.3 ± 1.2 | 19.5 ± 2.3 | 17.4 ± 1.0 | 17.4 ± 1.0 |
| 16:1n-7 | 91.5 ± 14.0 | 72.3 ± 12.5 | 95.7 ± 17.9 | 110.2 ± 17.5 |
| 18:1n-7 | 205.2 ± 21.5 | 184.1 ± 22.1 | 189.7 ± 27.5 | 201.4 ± 17.6 |
| 18:1n-9 | 1031.2 ± 132.8 | 984.0 ± 125.9 | 981.3 ± 114.0 | 989.7 ± 51.5 |
| 20:1n-9 | 26.7 ± 3.7 | 27.6 ± 4.2 | 25.9 ± 3.7 | 24.5 ± 1.0 |
| 22:1n-9 | 2.3 ± 0.5 | 2.5 ± 0.5 | 2.1 ± 0.3 | 2.2 ± 0.3 |
| 24:1n-9 | 11.0 ± 0.4 | 12.1 ± 1.3 | 11.1 ± 0.7 | 12.2 ± 0.7 |
| 20:3n-9 | 6.7 ± 3.3 | 2.6 ± 0.4 | 2.8 ± 1.3 | 2.8 ± 0.6 |
| 18:2n-6 | 2427.3 ± 292.0 | 2551.9 ± 303.6 | 2419.2 ± 202.0 | 2424.5 ± 126.3 |
| 18:3n-6 | 33.6 ± 3.8 | 27.9 ± 3.0 | 21.0 ± 2.3 | 20.4 ± 2.4 |
| 20:2n-6 | 50.6 ± 5.2 | 51.6 ± 4.0 | 44.9 ± 5.1 | 50.0 ± 2.4 |
| 20:3n-6 | 58.5 ± 7.0 | 56.4 ± 7.3 | 61.3 ± 6.4 | 70.4 ± 3.4 |
| 20:4n-6 | 1996.4 ± 110.1 | 1887.3 ± 74.0 | 1635.1 ± 72.7 | 1433.5 ± 116.6 |
| 22:4n-6 | 102.1 ± 12.6 | 84.5 ± 5.5 | 48.3 ± 6.0 | 50.7 ± 2.6 |
| 22:5n-6 | 57.9 ± 11.9 | 36.6 ± 1.6 | 16.0 ± 2.9 | 16.9 ± 1.0 |
| 24:2n-6 | 3.0 ± 0.5 | 2.9 ± 0.4 | 2.2 ± 0.3 | 1.9 ± 0.2 |
| 18:3n-3 | 69.1 ± 13.4 | 76.1 ± 14.4 | 77.4 ± 10.1 | 76.8 ± 6.4 |
| 18:4n-3 | 2.8 ± 1.0 | 1.5 ± 0.9 | 1.3 ± 0.9 | 0.2 ± 0.2 |
| 20:5n-3 | 21.7 ± 1.6 | 21.7 ± 3.0 | 33.5 ± 3.4 | 39.2 ± 3.7 |
| 22:5n-3 | 50.7 ± 5.0 | 47.2 ± 3.3 | 43.6 ± 3.4 | 56.1 ± 5.0 |
| 22:6n-3 | 200.8 ± 21.6 | 181.8 ± 7.8 | 449.8 ± 39.1 | 491.7 ± 25.0 |
| Total SFA | 2643.4 ± 223.7 | 2582.5 ± 211.7 | 2561.9 ± 180.8 | 2563.8 ± 132.2 |
| Total MUFA | 1367.9 ± 169.1 | 1282.6 ± 164.0 | 1305.9 ± 161.6 | 1340.2 ± 72.6 |
| Total PUFA | 5081.3 ± 380.6 | 5029.9 ± 392.7 | 4856.2 ± 308.3 | 4735.0 ± 220.5 |
| Total n-3 PUFA | 345.1 ± 30.2 | 328.3 ± 27.2 | 605.5 ± 52.6 | 664.0 ± 35.3 |
| Total n-6 PUFA | 4729.4 ± 352.0 | 4699.0 ± 365.4 | 4247.9 ± 256.8 | 4068.2 ± 188.2 |
| Total fatty acids | 9092.6 ± 771.1 | 8895.0 ± 765.0 | 8724.0 ± 647.0 | 8639.0 ± 399.5 |
Values are means ± SEM
Experiment B
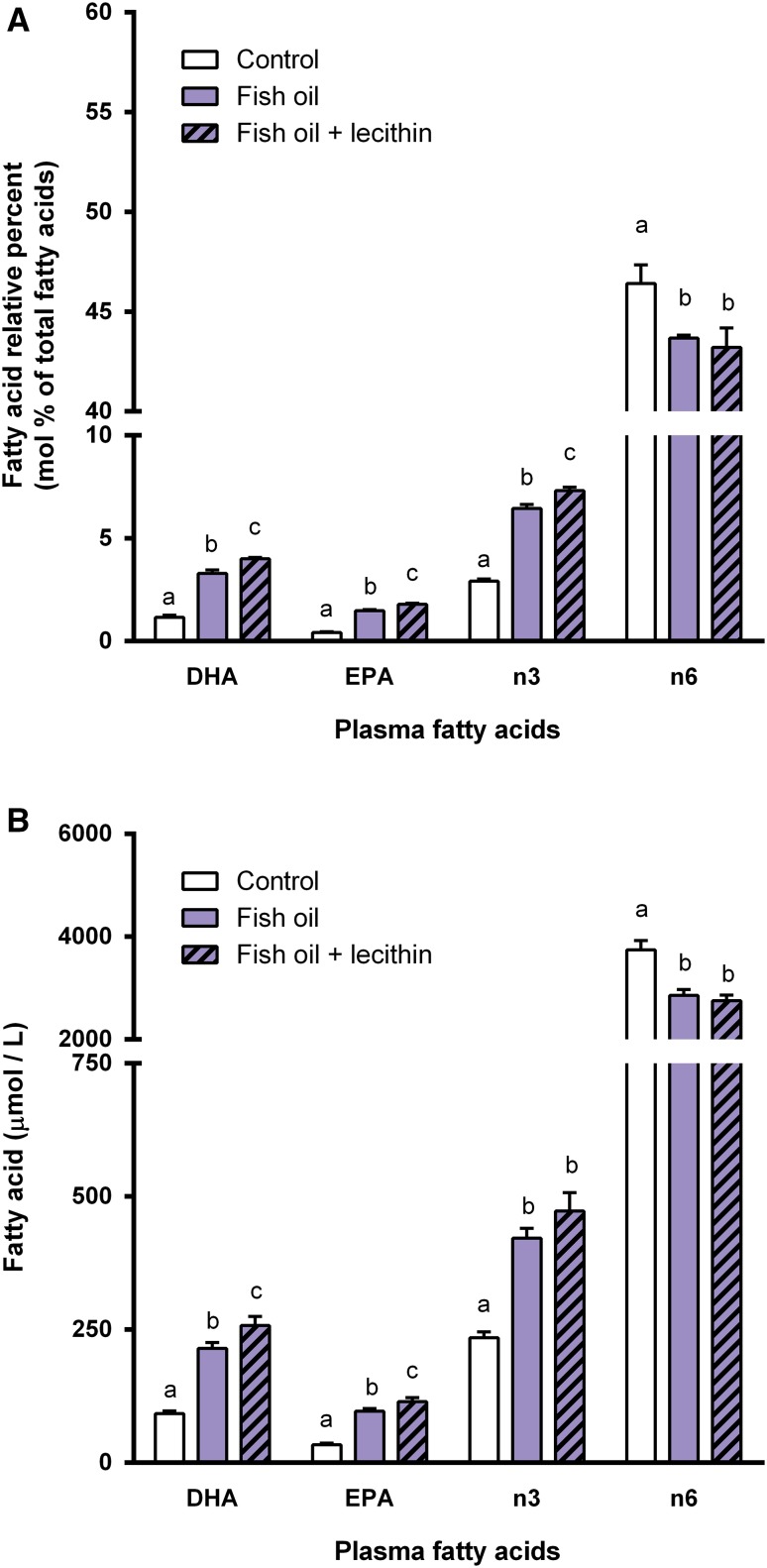
Animals were divided into three experimental groups that were matched according to their body weights and food intake after feeding the animals the control diet for 2 weeks. Body weight (P = 0.78) and food intake (P = 0.47) did not differ between the experimental groups over the course of the 3-week intervention period. Relative plasma levels of DHA (P < 0.001), EPA (P < 0.001), total n-3 PUFA (P < 0.001), and total n-6 PUFA (P = 0.023) were affected by the experimental diets (Fig. 2a). Animals fed the fish oil diet displayed increased plasma levels of DHA (P < 0.001), EPA (P < 0.001), and total n-3 PUFA (P < 0.001), and a decreased plasma total n-6 PUFA level (P = 0.031) as compared to the control diet. The increases in plasma DHA, EPA, total n-3 PUFA were augmented by additional supplementation with lecithin, i.e., plasma DHA (P < 0.001), EPA (P < 0.001), and total n-3 PUFA (P = 0.001) were higher in rats fed the fish oil + lecithin diet as compared to those fed the fish oil diet. Expressing the plasma fatty acid levels as quantitative concentrations revealed similar effects of the fish oil diet and the fish-oil + lecithin diet. Plasma concentrations of DHA (P < 0.001), EPA (P < 0.001), total n-3 PUFA (P < 0.001), and total n-6 PUFA (P < 0.001) differed between rats fed one of the three experimental diets (Fig. 2b). Compared to animals fed the control diet or the Fish oil diet, those fed the fish oil + lecithin diet displayed increased plasma concentrations of DHA (P < 0.001 vs. control diet; P = 0.022 vs. fish oil diet) and EPA (P < 0.001 vs. control diet; P = 0.028 vs. fish oil diet). Tables 4 and 5 give a complete overview of all plasma fatty acids measured, expressed as relative percent levels (Table 4) and quantitative concentrations (Table 5).
Fig. 2.
Experiment B: effects of dietary supplementation of fish oil or combined dietary supplementation of fish oil and lecithin on a relative percent levels (mol%) and b quantitative concentrations (µmol/L) of DHA, EPA, total n-3 PUFA, and total n-6 PUFA in plasma of rats. Plasma DHA, EPA, total n-3 PUFA, and total n-6 PUFA levels in rats that received one of the three experimental diets (control, fish oil, fish oil + lecithin) for 3 weeks. Values are means, with the SEM represented by vertical bars. Different letters indicate mean values were significantly different (P < 0.025). n = 7–8 per experimental group, 1 outlier was omitted from the data
Table 4.
Experiment B: effects of dietary supplementation of fish oil or combined dietary supplementation of fish oil and lecithin on plasma fatty acid composition (mol%) in rats
| Plasma fatty acids (mol% of total fatty acids) | Experiment B | ||
|---|---|---|---|
| Dietary intervention | |||
| Control | Fish oil | Fish oil + lecithin | |
| 6:0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 |
| 8:0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 |
| 10:0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 |
| 12:0 | 0.4 ± 0.1 | 0.2 ± 0.0 | 0.2 ± 0.0 |
| 14:0 | 1.2 ± 0.1 | 0.9 ± 0.0 | 1.0 ± 0.1 |
| 15:0 | 0.2 ± 0.0 | 0.2 ± 0.0 | 0.3 ± 0.0 |
| 16:0 | 21.2 ± 0.2 | 22.4 ± 0.2 | 22.9 ± 0.3 |
| 17:0 | 0.1 ± 0.0 | 0.1 ± 0.0 | 0.2 ± 0.0 |
| 18:0 | 6.8 ± 0.4 | 7.2 ± 0.2 | 7.2 ± 0.3 |
| 20:0 | 0.1 ± 0.0 | 0.1 ± 0.0 | 0.1 ± 0.0 |
| 22:0 | 0.2 ± 0.0 | 0.2 ± 0.0 | 0.2 ± 0.0 |
| 23:0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 |
| 24:0 | 0.3 ± 0.0 | 0.3 ± 0.0 | 0.3 ± 0.0 |
| 16:1n-7 | 2.8 ± 0.3 | 2.4 ± 0.1 | 2.4 ± 0.3 |
| 18:1n-7 | 3.5 ± 0.2 | 2.9 ± 0.1 | 2.7 ± 0.1 |
| 18:1n-9 | 13.3 ± 0.7 | 12.5 ± 0.2 | 11.6 ± 0.5 |
| 20:1n-9 | 0.2 ± 0.0 | 0.1 ± 0.0 | 0.1 ± 0.0 |
| 22:1n-9 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 |
| 24:1n-9 | 0.1 ± 0.0 | 0.1 ± 0.0 | 0.2 ± 0.0 |
| 20:3n-9 | 0.2 ± 0.0 | 0.1 ± 0.0 | 0.2 ± 0.0 |
| 18:2n-6 | 26.4 ± 0.7 | 27.4 ± 0.5 | 27.6 ± 0.4 |
| 18:3n-6 | 0.4 ± 0.0 | 0.2 ± 0.0 | 0.2 ± 0.0 |
| 20:2n-6 | 0.3 ± 0.0 | 0.3 ± 0.0 | 0.2 ± 0.0 |
| 20:3n-6 | 0.4 ± 0.0 | 0.5 ± 0.0 | 0.5 ± 0.0 |
| 20:4n-6 | 18.4 ± 1.1 | 14.8 ± 0.5 | 14.3 ± 0.8 |
| 22:4n-6 | 0.3 ± 0.1 | 0.4 ± 0.1 | 0.3 ± 0.1 |
| 22:5n-6 | 0.1 ± 0.0 | 0.1 ± 0.0 | 0.1 ± 0.0 |
| 24:2n-6 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 |
| 18:3n-3 | 0.9 ± 0.0 | 1.0 ± 0.0 | 0.8 ± 0.1 |
| 18:4n-3 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 |
| 20:5n-3 | 0.4 ± 0.0 | 1.5 ± 0.1 | 1.8 ± 0.1 |
| 22:5n-3 | 0.5 ± 0.0 | 0.7 ± 0.0 | 0.8 ± 0.0 |
| 22:6n-3 | 1.2 ± 0.1 | 3.3 ± 0.2 | 4.0 ± 0.1 |
| Total SFA | 30.6 ± 0.3 | 31.7 ± 0.3 | 32.4 ± 0.3 |
| Total MUFA | 19.8 ± 1.1 | 18.0 ± 0.3 | 16.9 ± 0.9 |
| Total PUFA | 49.5 ± 1.0 | 50.3 ± 0.2 | 50.7 ± 0.9 |
| Total n-3 PUFA | 2.9 ± 0.1 | 6.5 ± 0.2 | 7.3 ± 0.2 |
| Total n-6 PUFA | 46.4 ± 0.9 | 43.7 ± 0.1 | 43.2 ± 1.0 |
| Total fatty acids | 100.0 | 100.0 | 100.0 |
Values are means ± SEM
Table 5.
Experiment B: effects of dietary supplementation of fish oil or combined dietary supplementation of fish oil and lecithin on plasma fatty acid quantitative concentrations (µmol/L) in rats
| Plasma fatty acids (µmol/L) | Experiment B | ||
|---|---|---|---|
| Dietary intervention | |||
| Control | Fish oil | Fish oil + lecithin | |
| 6:0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 |
| 8:0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 |
| 10:0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 |
| 12:0 | 40.3 ± 13.8 | 14.9 ± 2.3 | 12.9 ± 2.8 |
| 14:0 | 104.2 ± 19.2 | 61.3 ± 4.5 | 62.7 ± 7.5 |
| 15:0 | 15.0 ± 1.1 | 16.0 ± 0.9 | 17.0 ± 1.4 |
| 16:0 | 1726.0 ± 111.3 | 1462.0 ± 50.1 | 1471.8 ± 81.1 |
| 17:0 | 9.0 ± 1.3 | 9.7 ± 2.2 | 11.6 ± 1.3 |
| 18:0 | 544.7 ± 26.4 | 470.0 ± 18.4 | 456.3 ± 13.8 |
| 20:0 | 6.9 ± 0.7 | 6.3 ± 0.4 | 6.7 ± 0.6 |
| 22:0 | 18.5 ± 1.1 | 14.1 ± 1.0 | 14.8 ± 1.0 |
| 23:0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 |
| 24:0 | 22.2 ± 0.7 | 18.5 ± 0.8 | 21.5 ± 1.2 |
| 16:1n-7 | 230.3 ± 36.0 | 155.7 ± 7.8 | 158.2 ± 23.6 |
| 18:1n-7 | 282.9 ± 29.9 | 187.8 ± 7.7 | 176.0 ± 16.3 |
| 18:1n-9 | 1101.0 ± 124.7 | 815.0 ± 38.0 | 750.9 ± 68.1 |
| 20:1n-9 | 12.6 ± 1.9 | 8.3 ± 0.5 | 5.9 ± 1.1 |
| 22:1n-9 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 |
| 24:1n-9 | 9.3 ± 0.5 | 9.5 ± 0.2 | 9.8 ± 0.8 |
| 20:3n-9 | 17.6 ± 1.3 | 9.3 ± 0.8 | 9.6 ± 0.9 |
| 18:2n-6 | 2151.2 ± 165.7 | 1793.2 ± 85.1 | 1768.8 ± 96.2 |
| 18:3n-6 | 29.6 ± 1.2 | 16.0 ± 1.6 | 14.0 ± 1.3 |
| 20:2n-6 | 27.4 ± 2.4 | 17.4 ± 1.4 | 15.9 ± 1.3 |
| 20:3n-6 | 35.8 ± 2.2 | 34.2 ± 1.7 | 33.1 ± 2.1 |
| 20:4n-6 | 1463.9 ± 45.1 | 963.2 ± 45.5 | 902.7 ± 16.3 |
| 22:4n-6 | 28.1 ± 9.0 | 26.9 ± 10.4 | 17.0 ± 6.9 |
| 22:5n-6 | 9.1 ± 2.9 | 6.6 ± 0.4 | 3.7 ± 1.4 |
| 24:2n-6 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 |
| 18:3n-3 | 71.9 ± 8.5 | 63.7 ± 3.9 | 50.6 ± 8.4 |
| 18:4n-3 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 |
| 20:5n-3 | 33.8 ± 2.7 | 96.6 ± 5.0 | 115.0 ± 7.3 |
| 22:5n-3 | 37.1 ± 2.0 | 46.7 ± 2.8 | 49.2 ± 5.3 |
| 22:6n-3 | 92.1 ± 5.0 | 214.5 ± 11.2 | 257.7 ± 16.9 |
| Total SFA | 2486.9 ± 156.9 | 2072.6 ± 66.0 | 2075.2 ± 96.7 |
| Total MUFA | 1636.1 ± 187.7 | 1176.4 ± 52.0 | 1100.9 ± 107.6 |
| Total PUFA | 3997.7 ± 189.1 | 3288.2 ± 130.7 | 3237.4 ± 139.2 |
| Total n-3 PUFA | 234.9 ± 10.9 | 421.5 ± 18.7 | 472.5 ± 35.0 |
| Total n-6 PUFA | 3745.2 ± 178.2 | 2857.4 ± 115.9 | 2755.2 ± 106.6 |
| Total fatty acids | 8120.6 ± 510.0 | 6537.3 ± 243.6 | 6413.5 ± 327.5 |
Values are means ± SEM
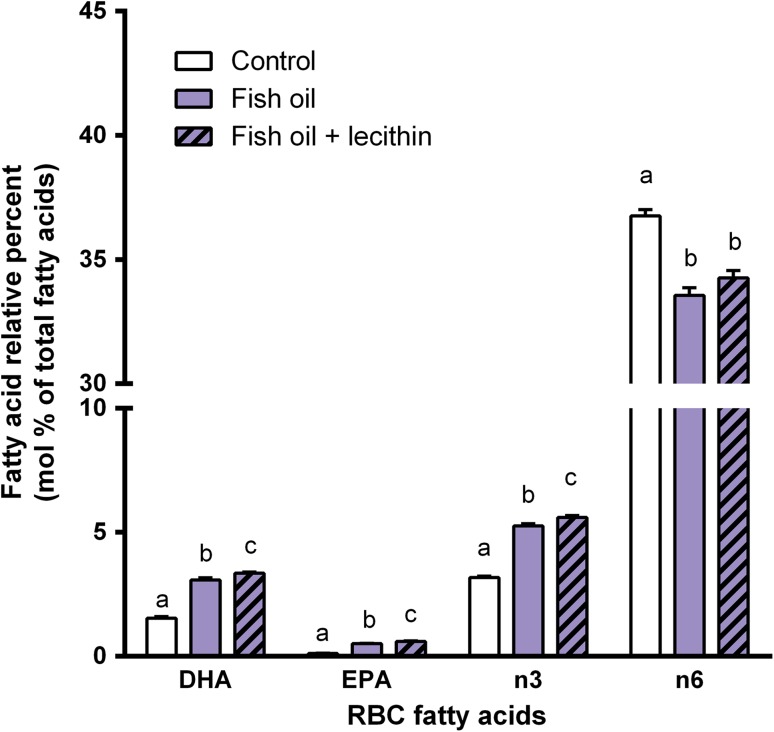
In RBC, similar differences in levels of DHA, EPA, n-3 PUFA, and n-6 PUFA between the experimental groups were found as obtained in plasma. Relative RBC levels of DHA (P < 0.001), EPA (P < 0.001), total n-3 PUFA (P < 0.001), and total n-6 PUFA (P < 0.001) also were affected by the experimental diets (Fig. 3). Compared to animals fed the control diet or the fish oil diet, those fed the fish oil + lecithin diet displayed increased RBC levels of DHA (P < 0.001 vs. control diet; P = 0.009 vs. fish oil diet), EPA (P < 0.001 vs. control diet; P = 0.006 vs. fish oil diet), and total n-3 PUFA (P < 0.001 vs. control diet; P = 0.004 vs. fish oil diet). Table 6 gives a complete overview of all RBC fatty acids measured, expressed as relative percent levels.
Fig. 3.
Experiment B: effects of dietary supplementation of fish oil or combined dietary supplementation of fish oil and lecithin on relative percent levels (mol%) of DHA, EPA, total n-3 PUFA, and total n-6 PUFA in RBC of rats. RBC DHA, EPA, total n-3 PUFA, and total n-6 PUFA levels in rats that received one of the three experimental diets (control, fish oil, fish oil + lecithin) for 3 weeks. Values are means, with the SEM represented by vertical bars. Different letters indicate mean values were significantly different (P < 0.025). n = 8 per experimental group
Table 6.
Experiment B: effects of dietary supplementation of fish oil or combined dietary supplementation of fish oil and lecithin on RBC fatty acid composition (mol%) in rats
| RBC fatty acids (mol% of total fatty acids) | Experiment B | ||
|---|---|---|---|
| Dietary intervention | |||
| Control | Fish oil | Fish oil + lecithin | |
| 6:0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 |
| 8:0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 |
| 10:0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 |
| 12:0 | 0.1 ± 0.0 | 0.1 ± 0.0 | 0.1 ± 0.0 |
| 14:0 | 0.6 ± 0.0 | 0.5 ± 0.0 | 0.5 ± 0.0 |
| 15:0 | 0.2 ± 0.0 | 0.3 ± 0.0 | 0.3 ± 0.0 |
| 16:0 | 34.7 ± 0.3 | 36.4 ± 0.4 | 35.5 ± 0.3 |
| 17:0 | 0.3 ± 0.0 | 0.3 ± 0.0 | 0.4 ± 0.0 |
| 18:0 | 10.1 ± 0.3 | 10.1 ± 0.2 | 10.4 ± 0.2 |
| 20:0 | 0.1 ± 0.0 | 0.1 ± 0.0 | 0.1 ± 0.0 |
| 22:0 | 0.4 ± 0.0 | 0.4 ± 0.0 | 0.4 ± 0.0 |
| 23:0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 |
| 24:0 | 1.1 ± 0.0 | 1.0 ± 0.0 | 1.1 ± 0.0 |
| 16:1n-7 | 0.9 ± 0.1 | 0.8 ± 0.0 | 0.8 ± 0.1 |
| 18:1n-7 | 3.5 ± 0.1 | 3.2 ± 0.1 | 3.0 ± 0.1 |
| 18:1n-9 | 7.2 ± 0.2 | 7.0 ± 0.1 | 6.6 ± 0.2 |
| 20:1n-9 | 0.1 ± 0.0 | 0.1 ± 0.0 | 0.1 ± 0.0 |
| 22:1n-9 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 |
| 24:1n-9 | 0.7 ± 0.0 | 0.7 ± 0.0 | 0.7 ± 0.0 |
| 20:3n-9 | 0.2 ± 0.0 | 0.1 ± 0.0 | 0.1 ± 0.0 |
| 18:2n-6 | 14.2 ± 0.5 | 14.5 ± 0.3 | 15.4 ± 0.3 |
| 18:3n-6 | 0.1 ± 0.0 | 0.1 ± 0.0 | 0.1 ± 0.0 |
| 20:2n-6 | 0.4 ± 0.0 | 0.4 ± 0.0 | 0.4 ± 0.0 |
| 20:3n-6 | 0.4 ± 0.0 | 0.5 ± 0.0 | 0.5 ± 0.0 |
| 20:4n-6 | 19.6 ± 0.3 | 16.6 ± 0.2 | 16.5 ± 0.3 |
| 22:4n-6 | 1.6 ± 0.0 | 1.2 ± 0.0 | 1.2 ± 0.0 |
| 22:5n-6 | 0.3 ± 0.0 | 0.2 ± 0.0 | 0.2 ± 0.0 |
| 24:2n-6 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 |
| 18:3n-3 | 0.2 ± 0.0 | 0.3 ± 0.0 | 0.2 ± 0.0 |
| 18:4n-3 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 |
| 20:5n-3 | 0.1 ± 0.0 | 0.5 ± 0.0 | 0.6 ± 0.0 |
| 22:5n-3 | 1.3 ± 0.0 | 1.4 ± 0.0 | 1.4 ± 0.0 |
| 22:6n-3 | 1.5 ± 0.1 | 3.1 ± 0.1 | 3.4 ± 0.0 |
| Total SFA | 47.5 ± 0.4 | 49.2 ± 0.3 | 48.7 ± 0.3 |
| Total MUFA | 12.5 ± 0.4 | 11.9 ± 0.2 | 11.3 ± 0.3 |
| Total PUFA | 40.1 ± 0.3 | 38.9 ± 0.3 | 40.0 ± 0.3 |
| Total n-3 PUFA | 3.2 ± 0.1 | 5.3 ± 0.1 | 5.6 ± 0.1 |
| Total n-6 PUFA | 36.8 ± 0.3 | 33.6 ± 0.3 | 34.3 ± 0.3 |
| Total fatty acids | 100.0 | 100.0 | 100.0 |
Values are means ± SEM
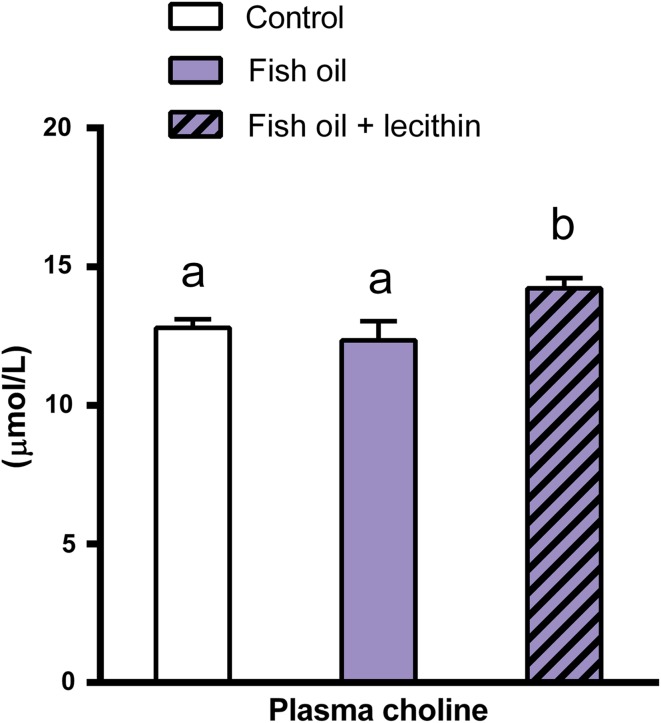
Plasma free choline concentration was also affected by the experimental diets (P = 0.030, Fig. 4). In animals receiving the lecithin-containing diet, plasma free choline concentration was higher as compared to those receiving the diets without lecithin (P = 0.049 vs. control diet; P = 0.012 vs. fish oil diet).
Fig. 4.
Experiment B: effects of dietary supplementation of fish oil or combined dietary supplementation of fish oil and lecithin on rat plasma free choline concentration (µmol/L). Plasma free choline concentration in rats that received one of the three experimental diets for 3 weeks (control, fish oil, fish oil + lecithin). Values are means, with the SEM represented by vertical bars. Different letters indicate mean values were significantly different (P < 0.05). n = 8 per experimental group
Discussion
The present experiments indicate that dietary crude lecithin can increase the systemic availability of dietary DHA with combined intake. Dietary supplementation of lecithin, which itself does not contain any DHA, increased levels of DHA, EPA, and total n-3 PUFA in plasma and RBC, and decreased levels of total n-6 PUFA in plasma of rats fed DHA oil-containing diets. Thus, while dietary lecithin supplementation alone did not affect DHA levels in plasma and RBC, lecithin further increased the rise in DHA induced by dietary DHA supplementation (vs. control diets) with up to 61 % (relative levels) and 47 % (quantitative concentration) in plasma, and with 18 % (relative level) in RBC. Furthermore, dietary lecithin, being a source of PtdCho, increased plasma choline concentration with 11 %.
Dietary supplementation of moderate levels of DHA (i.e., 0.2 %), either as vegetable algae oil or as fish oil, affected relative and quantitative plasma levels and relative RBC fatty acid levels as expected. Previous studies, both experimental [4, 5, 18–21] and clinical [3, 22, 23], repeatedly demonstrated that plasma and RBC levels of DHA increase relatively fast in a dose-dependent manner after oral administration of DHA. In human, steady state plasma concentrations of DHA after supplementation is reached within 1 week up to 1 month, whereas for RBC this takes several months [3, 22]. In rats, fatty acid levels in plasma and RBC represents dietary intake of fats over a relatively short-term period of 1–4 weeks [24]. Hence, the plasma and RBC samples from the rats in the current study reflect the effects of the changes in dietary pattern (i.e., changes dietary DHA-containing oils and lecithin content) over the weeks of the applied supplementation period.
Dietary crude lecithin is a source of active compounds as it is digested into fatty acids, lysophospholipids, phosphatidic acid, glycerol, monoglycerides, and other compounds, including choline and ethanolamine. These digestion products are subsequently absorbed and further metabolized. For example, the intake of lecithin has been shown to increase serum levels of choline in humans [25] and rats [26]. In Experiment B, it was confirmed that lecithin supplementation increases plasma choline concentration in rats.
Systemic DHA is known to be transferred across the blood–brain barrier, as shown in both experimental [27, 28] and clinical studies [6, 29, 30]. In fact, dietary supplementation with DHA (in various formulas) has repeatedly been shown to affect membrane composition [5, 18, 31–39], thereby influencing its biophysical properties. A high level of n-3 PUFA in neuronal membranes is associated with favorable effects on numerous membrane-dependent processes and thus neuronal functioning [1, 2]. Likewise, small increases in plasma choline can exert significant effects on brain choline levels [40], and in fact, lecithin intake has been shown to increase both plasma and brain choline levels in the rat [26]. Brain choline levels in turn control the rates at which it is utilized to form acetylcholine [41, 42]. In addition, DHA, EPA and choline [43–45] are known precursors for phospholipid synthesis needed for membrane formation and integrity. By increasing the systemic availability of dietary DHA and by increasing the systemic levels of choline, dietary lecithin putatively leads to increased uptake of DHA and choline into the brain, with subsequent effects on neuronal membrane formation and function.
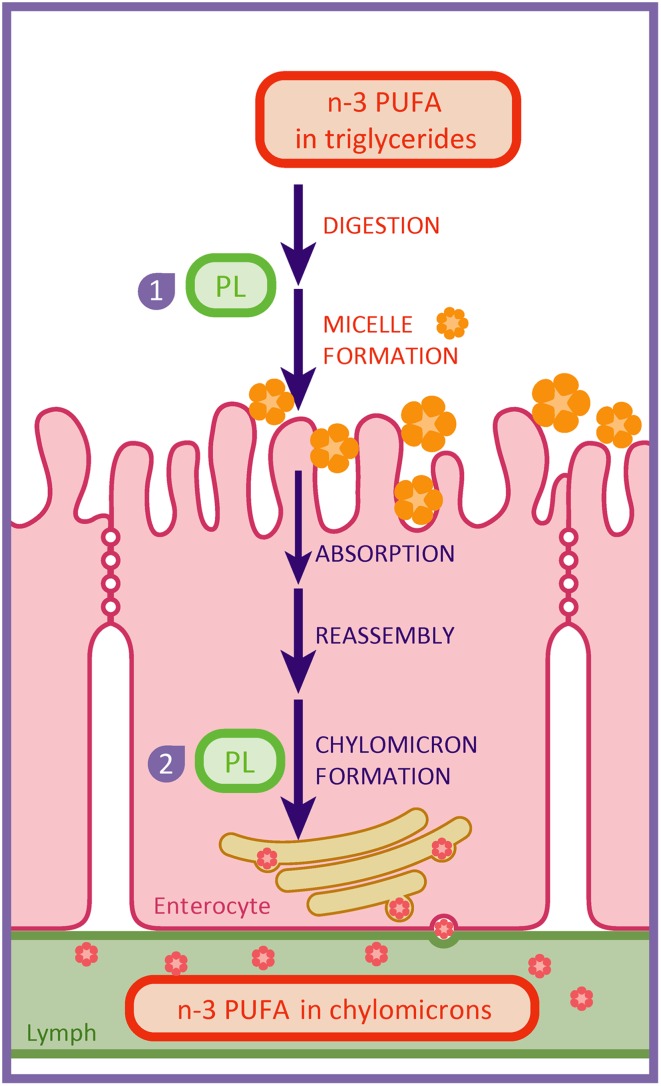
The effects of lecithin supplementation on the systemic availability of dietary n-3 PUFA might be explained by an increased absorption from the gut into the enterocytes and the lymph. Dietary phospholipids could enhance the absorption of n-3 PUFA from the gut, since they are known to facilitate the emulsification of dietary fat in the lumen and subsequent micelle formation for absorption into the enterocytes [8, 46]. Moreover, dietary phospholipids may increase the intestinal uptake of fat by increasing the formation of chylomicrons in enterocytes and their secretion into the lymph [7, 10]. In the current study, the highest percentage increases in DHA were found in Experiment B in which a higher dose of crude lecithin was used (0.4 % in Experiment A and 1.0 % in Experiment B), possibly indicating dose-dependency. Figure 5 shows a schematic representation of these effects of dietary phospholipids.
Fig. 5.
Schematic representation of the effects of dietary phospholipids on enhancing the absorption of dietary n-3 PUFA from the gut into the enterocytes and the lymph. Dietary phospholipids may increase the bioavailability of dietary n-3 PUFA by facilitating emulsification (1) and by increasing chylomicron formation (2). Figure is adapted from van Wijk et al. [54] with permission from IOS press. PL dietary phospholipids
Several investigators have previously attempted to demonstrate additive effects of lecithin or phospholipids or other emulsifiers on the bioavailability of triglycerides after oral intake in various models and using different approaches. Various studies thereby focused on the effects of pre-emulsification of oil with various emulsifiers, including lecithin [13–15, 47–49]. These studies distinctly show that single oral administration of pre-emulsified oil has a better bioavailability than non-emulsified oil. For example, Nishimukai et al. showed that duodenal infusion of an emulsion of triglycerides plus lecithin induces higher portal plasma triglycerides as compared to infusion of triglycerides alone [10]. In one study, the incorporation of radiolabeled DHA into various rat tissues were measured after a single dose of orally gavaged radiolabeled DHA, with or without administration of phospholipids (without emulsification), and showed no differential effects on the level of radiolabeled DHA in whole blood after 24 h [11]. Studies examining the effects of combined dietary supplementation of lecithin or phospholipids and (non-emulsified) triglycerides on systemic availability of triglycerides are scarcer. An experiment in fish (carps) demonstrated stimulated intestinal triglyceride uptake into plasma when dietary triglycerides were combined with dietary phospholipids [9]. In contrast, Lim et al. did not find increased amounts of DHA in plasma after combined dietary supplementation with DHA and phospholipids compared to supplementation with DHA alone [18]. However, in this study DHA content of the diet with both DHA and phospholipid was half of the DHA diet, which makes the diets’ efficacy impossible to compare. Other investigators have tried to show superiority of DHA-containing phospholipids (i.e., phospholipids with DHA as one of the fatty acid chains) over DHA-containing triglycerides to increase tissue levels of DHA in both preclinical and clinical experiments [11, 21, 50–53]. None of these studies showed differences in plasma DHA levels after supplementing with DHA-containing phospholipids as compared to DHA-containing triglycerides, although some studies did reveal superiority of DHA-containing phospholipids on DHA levels in other tissues, such as RBC [21, 53] and the brain [11, 53]. Nevertheless, most studies concluded that the absorption into the bloodstream is independent of type of DHA (as phospholipid or as triglyceride). The present study actually shows that supplementation of lecithin, i.e., phospholipids, further increases the rise in systemic DHA induced by dietary DHA supplementation.
It should be noted that the present experiments were each conducted under different experimental conditions. Most important differences were the amounts of lecithin supplemented to the diet (0.4 vs. 1.0 %), supplementation periods (2 vs. 3 weeks), DHA-containing oils (vegetable algae DHA oil vs. fish oil), rat strain (Wistar vs. Sprague–Dawley), diet base (grain-based vs. AIN-93M-based), and diet manufacturer (Research Diet Services and Ssniff Spezialdiäten). However, despite the differences in experimental conditions, the results from both experiments are highly consistent with some small differences in effects sizes. For example, in Experiment B a significant additional effect of dietary lecithin supplementation on top of dietary DHA supplementation on relative plasma DHA level and quantitative plasma DHA concentration were observed, whereas in Experiment A this additional effect was only observed on relative plasma DHA level. Overall, the experiments demonstrate a substantial effect of combined dietary supplementation of lecithin and DHA on plasma and RBC levels of DHA, EPA, n-3 PUFA, and n-6 PUFA.
Important to consider when extrapolating the current results to a human setting is the physiological difference between rats and human. Rats have a continuous release of hepatic bile as opposed to burst release of gall bile in humans. Whether this physiological difference has an impact on the effects of lecithin supplementation is difficult to predict. In addition, for a better interpretation of the current results, future preclinical experiments should also incorporate measurements in other important lipid pools that either influence the plasma lipid pool (e.g., the liver) or that are influenced by the plasma lipid pool (e.g., the brain).
Dietary DHA-containing oils and crude lecithin have synergistic effects on increasing plasma and RBC n-3 PUFA levels, including DHA and EPA. By increasing the systemic availability of dietary DHA, dietary lecithin may increase the efficacy of DHA supplementation when their intake is combined. Lecithin itself also raises plasma choline concentration. Therefore, combined supplementation of lecithin and DHA may be relevant in conditions that are associated with lower levels of plasma DHA and choline.
Acknowledgments
The authors thank Dr. M. C. de Wilde and for useful discussions and for carefully reading the manuscript. The research as described in this paper was funded by Nutricia Research.
Abbreviations
- DHA
Docosahexaenoic acid
- EPA
Eicosapentaenoic acid
- GC
Gas chromatography
- HPLC
High performance liquid chromatography
- MUFA
Monounsaturated fatty acid(s)
- n-3
Omega-3
- n-6
Omega-6
- PUFA
Polyunsaturated fatty acid(s)
- PtdCho
Phosphatidylcholine
- PtdEtn
Phosphatidylethanolamine
- PtdIns
Phosphatidylinositol
- RBC
Red blood cell(s)
- SFA
Saturated fatty acid(s)
Compliance with Ethical Standards
Conflict of interest
NvW, MB, JWCS, and LMB are employees of Nutricia Research. MC is a scientific consultant to Nutricia Research. TJM has received a reimbursement for costs of analyses.
Contributor Information
Nick van Wijk, Phone: +31 30 2095000, Email: nick.vanwijk@nutricia.com.
Martin Balvers, Email: martin.balvers@nutricia.com.
Mehmet Cansev, Email: mcansev@uludag.edu.tr.
Timothy J. Maher, Email: timothy.maher@mcphs.edu
John W. C. Sijben, Email: john.sijben@nutricia.com
Laus M. Broersen, Email: laus.broersen@nutricia.com
References
- 1.Mielke MM, Lyketsos CG. Lipids and the pathogenesis of Alzheimer’s disease: is there a link? Int Rev Psychiatry. 2006;18:173–186. doi: 10.1080/09540260600583007. [DOI] [PubMed] [Google Scholar]
- 2.Yehuda S, Rabinovitz S, Carasso RL, Mostofsky DI. The role of polyunsaturated fatty acids in restoring the aging neuronal membrane. Neurobiol Aging. 2002;23:843–853. doi: 10.1016/S0197-4580(02)00074-X. [DOI] [PubMed] [Google Scholar]
- 3.Arterburn LM, Hall EB, Oken H. Distribution, interconversion, and dose response of n-3 fatty acids in humans. Am J Clin Nutr. 2006;83:1467S–1476S. doi: 10.1093/ajcn/83.6.1467S. [DOI] [PubMed] [Google Scholar]
- 4.Hashimoto M, Tanabe Y, Fujii Y, Kikuta T, Shibata H, Shido O. Chronic administration of docosahexaenoic acid ameliorates the impairment of spatial cognition learning ability in amyloid beta-infused rats. J Nutr. 2005;135:549–555. doi: 10.1093/jn/135.3.549. [DOI] [PubMed] [Google Scholar]
- 5.Connor WE, Neuringer M, Lin DS. Dietary effects on brain fatty acid composition: the reversibility of n-3 fatty acid deficiency and turnover of docosahexaenoic acid in the brain, erythrocytes, and plasma of rhesus monkeys. J Lipid Res. 1990;31:237–247. [PubMed] [Google Scholar]
- 6.Umhau JC, Zhou W, Carson RE, Rapoport SI, Polozova A, Demar J, Hussein N, Bhattacharjee AK, Ma K, Esposito G, Majchrzak S, Herscovitch P, Eckelman WC, Kurdziel KA, Salem N., Jr Imaging incorporation of circulating docosahexaenoic acid into the human brain using positron emission tomography. J Lipid Res. 2009;50:1259–1268. doi: 10.1194/jlr.M800530-JLR200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.O’Doherty PJ, Kakis G, Kuksis A. Role of luminal lecithin in intestinal fat absorption. Lipids. 1973;8:249–255. doi: 10.1007/BF02531899. [DOI] [PubMed] [Google Scholar]
- 8.Werner A, Havinga R, Perton F, Kuipers F, Verkade HJ. Lymphatic chylomicron size is inversely related to biliary phospholipid secretion in mice. Am J Physiol Gastrointest Liver Physiol. 2006;290:G1177–G1185. doi: 10.1152/ajpgi.00127.2005. [DOI] [PubMed] [Google Scholar]
- 9.Geurden I, Kaushik S, Corraze G. Dietary phosphatidylcholine affects postprandial plasma levels and digestibility of lipid in common carp (Cyprinus carpio) Br J Nutr. 2008;100:512–517. doi: 10.1017/S0007114508904396. [DOI] [PubMed] [Google Scholar]
- 10.Nishimukai M, Hara H. Soybean phosphatidylcholine-induced enhancement of lymphatic absorption of triglyceride depends on chylomicron formation in rats. Biosci Biotechnol Biochem. 2007;71:1192–1197. doi: 10.1271/bbb.60654. [DOI] [PubMed] [Google Scholar]
- 11.Graf BA, Duchateau GS, Patterson AB, Mitchell ES, van Bruggen P, Koek JH, Melville S, Verkade HJ. Age dependent incorporation of 14C-DHA into rat brain and body tissues after dosing various 14C-DHA-esters. Prostaglandins Leukot Essent Fatty Acids. 2010;83:89–96. doi: 10.1016/j.plefa.2010.05.004. [DOI] [PubMed] [Google Scholar]
- 12.Lim SY, Suzuki H. Intakes of dietary docosahexaenoic acid ethyl ester and egg phosphatidylcholine improve maze-learning ability in young and old mice. J Nutr. 2000;130:1629–1632. doi: 10.1093/jn/130.6.1629. [DOI] [PubMed] [Google Scholar]
- 13.Sugasini D, Devaraj VC, Ramesh M, Lokesh BR. Lymphatic transport of α-linolenic acid and its conversion to long chain n-3 fatty acids in rats fed microemulsions of linseed oil. Lipids. 2014;49:225–233. doi: 10.1007/s11745-013-3873-4. [DOI] [PubMed] [Google Scholar]
- 14.Couedelo L, Amara S, Lecomte M, Meugnier E, Monteil J, Fonseca L, Pineau G, Cansell M, Carriere F, Michalski MC, Vaysse C. Impact of various emulsifiers on ALA bioavailability and chylomicron synthesis through changes in gastrointestinal lipolysis. Food Funct. 2015;6:1726–1735. doi: 10.1039/C5FO00070J. [DOI] [PubMed] [Google Scholar]
- 15.Couedelo L, Boue-Vaysse C, Fonseca L, Montesinos E, Djoukitch S, Combe N, Cansell M. Lymphatic absorption of α-linolenic acid in rats fed flaxseed oil-based emulsion. Br J Nutr. 2011;105:1026–1035. doi: 10.1017/S000711451000454X. [DOI] [PubMed] [Google Scholar]
- 16.Bligh EG, Dyer WJ. A rapid method of total lipid extraction and purification. Can J Biochem Physiol. 1959;37:911–917. doi: 10.1139/o59-099. [DOI] [PubMed] [Google Scholar]
- 17.Christie WW. Preparation of ester derivatives of fatty acids for chromatographic analysis. In: Christie WW, editor. Advances in lipid methodology. Dundee: Oily Press; 1993. [Google Scholar]
- 18.Lim SY, Suzuki H. Effect of dietary docosahexaenoic acid and phosphatidylcholine on maze behavior and fatty acid composition of plasma and brain lipids in mice. Int J Vitam Nutr Res. 2000;70:251–259. doi: 10.1024/0300-9831.70.5.251. [DOI] [PubMed] [Google Scholar]
- 19.Lim SY, Suzuki H. Dose–response effect of docosahexaenoic acid ethyl ester on maze behavior and brain fatty acid composition in adult mice. Int J Vitam Nutr Res. 2002;72:77–84. doi: 10.1024/0300-9831.72.2.77. [DOI] [PubMed] [Google Scholar]
- 20.Gamoh S, Hashimoto M, Sugioka K, Shahdat HM, Hata N, Misawa Y, Masumura S. Chronic administration of docosahexaenoic acid improves reference memory-related learning ability in young rats. Neuroscience. 1999;93:237–241. doi: 10.1016/S0306-4522(99)00107-4. [DOI] [PubMed] [Google Scholar]
- 21.Valenzuela A, Nieto S, Sanhueza J, Nunez MJ, Ferrer C. Tissue accretion and milk content of docosahexaenoic acid in female rats after supplementation with different docosahexaenoic acid sources. Ann Nutr Metab. 2005;49:325–332. doi: 10.1159/000087337. [DOI] [PubMed] [Google Scholar]
- 22.Kuratko CN, Salem N., Jr Biomarkers of DHA status. Prostaglandins Leukot Essent Fatty Acids. 2009;81:111–118. doi: 10.1016/j.plefa.2009.05.007. [DOI] [PubMed] [Google Scholar]
- 23.Plourde M, Chouinard-Watkins R, Vandal M, Zhang Y, Lawrence P, Brenna JT, Cunnane SC. Plasma incorporation, apparent retroconversion and beta-oxidation of 13C-docosahexaenoic acid in the elderly. Nutr Metab (Lond) 2011;8:5. doi: 10.1186/1743-7075-8-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Calviello G, Palozza P, Franceschelli P, Bartoli GM. Low-dose eicosapentaenoic or docosahexaenoic acid administration modifies fatty acid composition and does not affect susceptibility to oxidative stress in rat erythrocytes and tissues. Lipids. 1997;32:1075–1083. doi: 10.1007/s11745-997-0139-4. [DOI] [PubMed] [Google Scholar]
- 25.Wurtman RJ, Hirsch MJ, Growdon JH. Lecithin consumption raises serum-free-choline levels. Lancet. 1977;2:68–69. doi: 10.1016/S0140-6736(77)90067-8. [DOI] [PubMed] [Google Scholar]
- 26.Magil SG, Zeisel SH, Wurtman RJ. Effects of ingesting soy or egg lecithins on serum choline, brain choline and brain acetylcholine. J Nutr. 1981;111:166–170. doi: 10.1093/jn/111.1.166. [DOI] [PubMed] [Google Scholar]
- 27.Brossard N, Croset M, Lecerf J, Pachiaudi C, Normand S, Chirouze V, Macovschi O, Riou JP, Tayot JL, Lagarde M. Metabolic fate of an oral tracer dose of [13C] docosahexaenoic acid triglycerides in the rat. Am J Physiol. 1996;270:R846–R854. doi: 10.1152/ajpregu.1996.270.4.R846. [DOI] [PubMed] [Google Scholar]
- 28.Rapoport SI, Chang MC, Spector AA. Delivery and turnover of plasma-derived essential PUFAs in mammalian brain. J Lipid Res. 2001;42:678–685. [PubMed] [Google Scholar]
- 29.Freund Levi Y, Vedin I, Cederholm T, Basun H, Faxen Irving G, Eriksdotter M, Hjorth E, Schultzberg M, Vessby B, Wahlund LO, Salem N, Jr, Palmblad J. Transfer of omega-3 fatty acids across the blood–brain barrier after dietary supplementation with a docosahexaenoic acid-rich omega-3 fatty acid preparation in patients with Alzheimer’s disease: the OmegAD study. J Intern Med. 2014;275:428–436. doi: 10.1111/joim.12166. [DOI] [PubMed] [Google Scholar]
- 30.Quinn JF, Raman R, Thomas RG, Yurko-Mauro K, Nelson EB, Van Dyck C, Galvin JE, Emond J, Jack CR, Jr, Weiner M, Shinto L, Aisen PS. Docosahexaenoic acid supplementation and cognitive decline in Alzheimer disease: a randomized trial. JAMA. 2010;304:1903–1911. doi: 10.1001/jama.2010.1510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Aid S, Vancassel S, Linard A, Lavialle M, Guesnet P. Dietary docosahexaenoic acid [22:6(n-3)] as a phospholipid or a triglyceride enhances the potassium chloride-evoked release of acetylcholine in rat hippocampus. J Nutr. 2005;135:1008–1013. doi: 10.1093/jn/135.5.1008. [DOI] [PubMed] [Google Scholar]
- 32.Favreliere S, Perault MC, Huguet F, De Javel D, Bertrand N, Piriou A, Durand G. DHA-enriched phospholipid diets modulate age-related alterations in rat hippocampus. Neurobiol Aging. 2003;24:233–243. doi: 10.1016/S0197-4580(02)00064-7. [DOI] [PubMed] [Google Scholar]
- 33.Hashimoto M, Hossain S, Shimada T, Shido O. Docosahexaenoic acid-induced protective effect against impaired learning in amyloid beta-infused rats is associated with increased synaptosomal membrane fluidity. Clin Exp Pharmacol Physiol. 2006;33:934–939. doi: 10.1111/j.1440-1681.2006.04467.x. [DOI] [PubMed] [Google Scholar]
- 34.Hiratsuka S, Koizumi K, Ooba T, Yokogoshi H. Effects of dietary docosahexaenoic acid connecting phospholipids on the learning ability and fatty acid composition of the brain. J Nutr Sci Vitaminol (Tokyo) 2009;55:374–380. doi: 10.3177/jnsv.55.374. [DOI] [PubMed] [Google Scholar]
- 35.Shahdat H, Hashimoto M, Shimada T, Shido O. Synaptic plasma membrane-bound acetylcholinesterase activity is not affected by docosahexaenoic acid-induced decrease in membrane order. Life Sci. 2004;74:3009–3024. doi: 10.1016/j.lfs.2003.10.028. [DOI] [PubMed] [Google Scholar]
- 36.Vaisman N, Pelled D. n-3 phosphatidylserine attenuated scopolamine-induced amnesia in middle-aged rats. Prog Neuropsychopharmacol Biol Psychiatry. 2009;33:952–959. doi: 10.1016/j.pnpbp.2009.04.021. [DOI] [PubMed] [Google Scholar]
- 37.Broersen LM, Kuipers AA, Balvers M, van Wijk N, Savelkoul PJ, de Wilde MC, van der Beek EM, Sijben JW, Hageman RJ, Kamphuis PJ, Kiliaan AJ. A specific multi-nutrient diet reduces Alzheimer-like pathology in young adult AbetaPPswe/PS1dE9 mice. J Alzheimers Dis. 2013;33:177–190. doi: 10.3233/JAD-2012-112039. [DOI] [PubMed] [Google Scholar]
- 38.de Wilde MC, Hogyes E, Kiliaan AJ, Farkas T, Luiten PG, Farkas E. Dietary fatty acids alter blood pressure, behavior and brain membrane composition of hypertensive rats. Brain Res. 2003;988:9–19. doi: 10.1016/S0006-8993(03)03320-1. [DOI] [PubMed] [Google Scholar]
- 39.Carrie I, Clement M, de Javel D, Frances H, Bourre JM. Specific phospholipid fatty acid composition of brain regions in mice. Effects of n-3 polyunsaturated fatty acid deficiency and phospholipid supplementation. J Lipid Res. 2000;41:465–472. [PubMed] [Google Scholar]
- 40.Klein J, Koppen A, Loffelholz K. Small rises in plasma choline reverse the negative arteriovenous difference of brain choline. J Neurochem. 1990;55:1231–1236. doi: 10.1111/j.1471-4159.1990.tb03129.x. [DOI] [PubMed] [Google Scholar]
- 41.Cohen EL, Wurtman RJ. Brain acetylcholine: control by dietary choline. Science. 1976;191:561–562. doi: 10.1126/science.1251187. [DOI] [PubMed] [Google Scholar]
- 42.Wurtman RJ, Hefti F, Melamed E. Precursor control of neurotransmitter synthesis. Pharmacol Rev. 1980;32:315–335. [PubMed] [Google Scholar]
- 43.Cansev M, Wurtman RJ. Chronic administration of docosahexaenoic acid or eicosapentaenoic acid, but not arachidonic acid, alone or in combination with uridine, increases brain phosphatide and synaptic protein levels in gerbils. Neuroscience. 2007;148:421–431. doi: 10.1016/j.neuroscience.2007.06.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Cansev M, van Wijk N, Turkyilmaz M, Orhan F, Sijben JW, Broersen LM. A specific multi-nutrient enriched diet enhances hippocampal cholinergic transmission in aged rats. Neurobiol Aging. 2015;36:344–351. doi: 10.1016/j.neurobiolaging.2014.07.021. [DOI] [PubMed] [Google Scholar]
- 45.Wurtman RJ, Cansev M, Sakamoto T, Ulus IH. Use of phosphatide precursors to promote synaptogenesis. Annu Rev Nutr. 2009;29:59–87. doi: 10.1146/annurev-nutr-080508-141059. [DOI] [PubMed] [Google Scholar]
- 46.Jones DB, Hancock JD, Harmon DL, Walker CE. Effects of exogenous emulsifiers and fat sources on nutrient digestibility, serum lipids, and growth performance in weanling pigs. J Anim Sci. 1992;70:3473–3482. doi: 10.2527/1992.70113473x. [DOI] [PubMed] [Google Scholar]
- 47.Garaiova I, Guschina IA, Plummer SF, Tang J, Wang D, Plummer NT. A randomised cross-over trial in healthy adults indicating improved absorption of omega-3 fatty acids by pre-emulsification. Nutr J. 2007;6:4. doi: 10.1186/1475-2891-6-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Raatz SK, Redmon JB, Wimmergren N, Donadio JV, Bibus DM. Enhanced absorption of n-3 fatty acids from emulsified compared with encapsulated fish oil. J Am Diet Assoc. 2009;109:1076–1081. doi: 10.1016/j.jada.2009.03.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Vors C, Pineau G, Gabert L, Drai J, Louche-Pelissier C, Defoort C, Lairon D, Desage M, Danthine S, Lambert-Porcheron S, Vidal H, Laville M, Michalski MC. Modulating absorption and postprandial handling of dietary fatty acids by structuring fat in the meal: a randomized crossover clinical trial. Am J Clin Nutr. 2013;97:23–36. doi: 10.3945/ajcn.112.043976. [DOI] [PubMed] [Google Scholar]
- 50.Sala-Vila A, Campoy C, Castellote AI, Garrido FJ, Rivero M, Rodriguez-Palmero M, Lopez-Sabater MC. Influence of dietary source of docosahexaenoic and arachidonic acids on their incorporation into membrane phospholipids of red blood cells in term infants. Prostaglandins Leukot Essent Fatty Acids. 2006;74:143–148. doi: 10.1016/j.plefa.2005.10.003. [DOI] [PubMed] [Google Scholar]
- 51.Sala-Vila A, Castellote AI, Campoy C, Rivero M, Rodriguez-Palmero M, Lopez-Sabater MC. The source of long-chain PUFA in formula supplements does not affect the fatty acid composition of plasma lipids in full-term infants. J Nutr. 2004;134:868–873. doi: 10.1093/jn/134.4.868. [DOI] [PubMed] [Google Scholar]
- 52.Vaisman N, Kaysar N, Zaruk-Adasha Y, Pelled D, Brichon G, Zwingelstein G, Bodennec J. Correlation between changes in blood fatty acid composition and visual sustained attention performance in children with inattention: effect of dietary n-3 fatty acids containing phospholipids. Am J Clin Nutr. 2008;87:1170–1180. doi: 10.1093/ajcn/87.5.1170. [DOI] [PubMed] [Google Scholar]
- 53.Liu L, Bartke N, Van Daele H, Lawrence P, Qin X, Park HG, Kothapalli K, Windust A, Bindels J, Wang Z, Brenna JT. Higher efficacy of dietary DHA provided as a phospholipid than as a triglyceride for brain DHA accretion in neonatal piglets. J Lipid Res. 2014;55:531–539. doi: 10.1194/jlr.M045930. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.van Wijk N, Broersen LM, de Wilde MC, Hageman RJ, Groenendijk M, Sijben JW, Kamphuis PJ. Targeting synaptic dysfunction in Alzheimer’s disease by administering a specific nutrient combination. J Alzheimers Dis. 2014;38:459–479. doi: 10.3233/JAD-130998. [DOI] [PubMed] [Google Scholar]