Abstract
Objective
Our objectives were to determine the expression of EVC2 in craniofacial tissues and investigate the effect of Evc2 deficiency on craniofacial bones using Evc2 knockout (KO) mouse model.
Design
Evc2 KO mice were generated by introducing a premature stop codon followed by the Internal Ribosomal Entry Site fused to β-galactosidase (LacZ). Samples from wild-type (WT), heterozygous (Het) and homozygous Evc2 KO mice were prepared. LacZ staining and immunohistochemistry (IHC) with anti-β-galactosidase, anti-EVC2 and anti-SOX9 antibodies were performed. The craniofacial bones were stained with alcian blue and alizarin red.
Results
The LacZ activity in KO was mainly observed in the anterior parts of viscerocranium. The Evc2-expressing cells were identified in many cartilageous regions by IHC with anti-β-galactosidase antibody in KO and Het embryos. The endogenous EVC2 protein was observed in these areas in WT embryos. Double labeling with anti-SOX9 antibody showed that these cells were mainly chondrocytes. At adult stages, the expression of EVC2 was found in chondrocytes of nasal bones and spheno-occipital synchondrosis, and osteocytes and endothelial-like cells of the premaxilla and mandible. The skeletal double staining demonstrated that craniofacial bones, where the expression of EVC2 was observed, in KO had the morphological defects as compared to WT.
Conclusion
To our knowledge, our study was the first to identify the types of Evc2-expressing cells in craniofacial tissues. Consistent with the expression pattern, abnormal craniofacial bone morphology was found in the Evc2 KO mice, suggesting that EVC2 may be important during craniofacial growth and development.
Keywords: Ellis-van Creveld syndrome, EVC2, expression, craniofacial bone, development, knockout (KO) mouse
1. Introduction
Ellis-van Creveld (EvC) syndrome (OMIM #225500) or “chondro-ectodermal dysplasia” was named in 1940 by Drs. Richard Ellis and Simon van Creveld, who first described the detailed clinical manifestations of the disease-phenotype (1). EvC syndrome is a rare autosomal recessive syndrome characterized by four main congenital anomalies: chondral dysplasia of ribs and limbs, affecting more the distal bones and leading to a disproportionate dwarfism; ectodermal dysplasia of the nails, teeth and sometimes hair; polydactyly (postaxial sixth finger) of hands and feet; and congenital heart disease affecting up to 60% of all cases (1–3). Demographic data showed that the disease has a higher prevalence in the Old Order Amish community in Lancaster County, Pennsylvania than in any other areas worldwide.
Previously it has been reported that the genetic locus responsible for the EvC phenotype was narrowed down to chromosome 4p16 by the linkage and haplotype analyses (4). This finding further led to the identification of a novel gene, EVC that was mutated in EvC patients (5). Although the causative gene was identified, many of the EvC patients were not associated with EVC mutations. Mutations in the novel second gene, EVC2 were later identified (6, 7). Consequently, most of the EvC cases were associated with either gene mutation (8). Chondrodysplasia has been also found in many cattle breeds and the mutation has been identified in LIMBIN (LBN) gene, which is the bovine ortholog of EVC2, in the Japanese brown cattle suffering bovine chondrodysplasia (bcd) (9). The structural analysis of EVC and EVC2 revealed that these 2 genes were closely located in a head to head configuration with only 2.86 kilo bp of separation between their transcriptional start sites on chromosome 4p16 (10). It is now known that either EVC or EVC2 mutation results in EvC syndrome and the clinical phenotype of EvC appears to be similar to a high degree, regardless of mutations in either gene.
The molecular functions of EVC and EVC2 have been proposed, i.e. both EVC and EVC2 are positive regulators of the hedgehog (Hh) signaling pathway (10, 11). EVC can function as a component of the Hh signal transduction pathway required for normal endochondral ossification (12) and is proposed to act as a downstream molecule of Smoothened (Smo) to facilitate the transcription of the target genes, including Patched1 (Ptch1) and Gli1 (11). It has been also shown that EVC2 is required for Hh pathway activation in response to the Smo agonist by showing the decreased expression of Ptch1 in Evc2 KO cells (10). Recent studies showed that the EVC-EVC2 forms a protein complex with Smo at the base of the cilia followed by inhibition of Suppressor of Fused Homolog (SuFu) and Gli activation (13–15). Since many of the phenotypes seen in patients with ciliopathies can be attributed to defective Hh signaling (16), it is now well accepted that Hh signaling is essential for craniofacial development, suggesting that EVC2 could be also involved in craniofacial tissue development. To support this notion, it has been reported that EvC patients exhibit craniofacial growth and developmental phenotypes, including defective skull growth pattern such as enlarged skull, depressed nasal bridge, mandibular prognathism, skeletal class III (maxillary deficiency and mandibular prognathism), and skeletal open bite (1, 17, 18). A genome-wide association study revealed that human chromosome 4p16.1, which includes EVC and EVC2 genes, is strongly associated with mandibular prognathism (19). The presence of Evc2 mRNA in the craniofacial primordia during mouse development also indicates the importance of EVC2 function in the development of craniofacial tissues (9). However, it is not fully clear at this point which type(s) of cells and tissues in the craniofacial areas likely contribute to the craniofacial growth and developmental phenotypes in the EvC patients. Very recently, we reported the generation of a new Evc2/Lbn knockout (KO) mouse model where a premature stop codon was inserted in exon 12, which mimics the bcd cattle mutation (20). At birth, the Evc2 KO pups showed dwarfism mainly seen in limbs, although their body length and head size were not changed. At 1 week, the head size became different between Evc2 KO and littermate control mice. Therefore, in the current study, we aim to investigate the expression pattern of EVC2 at a late embryonic stage and postnatal stage, and characterize the craniofacial tissue morphology where EVC2 is expressed using Evc2 KO mouse model.
2. Materials and methods
2.1. Evc2 knockout (KO) mouse and sample preparation
Generation and genotyping of Evc2 KO mice have been previously reported (20). Briefly, a targeting vector was designed for Evc2 KO mice by introducing a premature stop codon in exon 12, followed by Internal Ribosomal Entry Site (IRES) fused to β-galactosidase (LacZ) under the control of the endogenous Evc2 promoter. The targeting cassette was introduced to AB2.2 embryonic stem (ES) cells and the correctly targeted ES cell clones were identified by Southern blot and genomic PCR strategies. After the germline transmission, resulted heterozygous (Het) Evc2 mice were intercrossed to generate homozygous Evc2 KO mice as well as the control wild-type (WT) and Het mice. All animal experiments were performed in accordance with guidelines in each institution covering the humane care and use of animals in research.
2.2. X-Gal staining
Embryos at embryonic (E) day 15.5 were collected and genotyped. The heads were dissected in cold phosphate buffered saline (PBS) followed by fixation with 4% paraformaldehyde (PFA) for 30 min on ice. Samples were rinsed with detergent-rinse buffer (2 mM MgCl2, 0.02% Nonidet P-40, 0.1 M PBS pH 7.3 and 0.01% sodium deoxycholate) for 30 min three times at room temperature (RT). Samples were then incubated with an embryo staining solution (1 mg/ml X-gal in DMSO, 5 mM potassium ferricyanide, 5 mM potassium ferrocyanide in the detergent rinse buffer) overnight at RT in the dark. On the following day, samples were re-fixed with 4% PFA for 30 min at RT, washed twice with PBS at RT and stored in 70% ethanol at 4°C until imaging.
2.3. Real-time PCR
Primary chondrocytes were isolated from ribs of Evc2 WT and KO embryos at E18.5 and cultured as previously described (20). To investigate the gene expression of Evc2 in primary chondrocytes, total RNA was extracted with TRIzol reagent (Life Technologies). Two μg of the total RNA extract was used for RT using the Omniscript RT Kit (Qiagen). Real-time PCR was performed in triplicate using the specific primers-probe for mouse Evc2 (Applied Biosystems, ABI assay number; Mm00507589_m1) or rodent glyceraldehyde-3-phosphate dehydrogenase (Gapdh; 04308313), and the expression levels were analyzed by the ABI 7300 real-time PCR system. The mean fold changes in the expression of Evc2 relative to that of Gapdh were calculated using the values obtained from the WT chondrocyte cDNA with Evc2 expression as a calibrator by means of 2−ΔΔCT method as previously reported (21).
2.4. Generation of anti-EVC2 antibody and its characterization by Western blot analysis
An affinity-purified polyclonal anti-EVC2 antibody was generated by immunizing rabbits with a synthetic peptide corresponding to the sequence in the N-terminal region of mouse EVC2, i.e. residues 117–133; DEITGLAVKESPGNNTQ (Bethyl Laboratories, Inc.).
The EVC2 mammalian expression plasmid containing full coding sequence of mouse Evc2 without any tag (pCMV-SPORT6-Evc2) was purchased from Life Technologies (MGC clone: 4237472). Mouse chondrogenic ATDC5 cells were transiently transfected with pCMV-SPORT6-Evc2 plasmid as a positive control using X-tremeGENE 9 DNA transfection reagent (Roche Life Science). Primary chondrocytes of Evc2 WT and KO embryos were cultured. Cell lysates from primary chondrocytes and ATDC5 cells were prepared using cold lysis buffer containing 150 mM NaCl, 20 mM Tris-HCl, pH 7.5, 10 mM EDTA, 1% Triton-X-100, 1% deoxycholate, and protease inhibitor cocktail for 30 min on ice followed by centrifugation at 12,000 rpm for 15 minutes. The quantity of protein concentration was measured by the detergent compatible (DC) protein assay kit (Bio-Rad), identical amount of total cell lysates from each cell type of primary chondrocytes (40 μg each) and cell lysates from the transfected ATDC5 cells (30 μg) were resolved by 4–12% SDS-PAGE. Western blotting analysis was performed with rabbit anti-EVC2 and anti-β-ACTIN (Cell Signaling Technology) antibodies. Chemiluminescent detection of bound antibodies was determined using the ECL Western blotting detection reagent (GE Healthcare Life Sciences).
2.5. Immunohistochemistry (IHC) and fluorescence IHC
The Heads of Evc2 WT, Het and KO mice were collected and fixed in 10% formalin. Samples were dehydrated, embedded in paraffin, and sectioned at a thickness of 5 μm and at least 6 sections per each sample at each time point were used for each of the following staining.
Since the IRES-LacZ cassette was introduced in the targeting construct, the Evc2-expressing cells could be visualized in KO and Het embryos by means of IHC with anti-β-galactosidase (β-gal) antibody (US Biological) and WT embryos were used as a negative control. Sections were deparaffinized, treated with citric acid solution for the antigen retrieval and then incubated with anti-β-gal antibody (diluted in 1:250) or non-immune mouse Immunoglobulin (Ig; Santa Cruz Biotechnology, Inc.) as a negative control. IHC staining was performed using the VECTASTAIN Elite ABC Kit (Vector Laboratories Inc.) as previously reported (22). Sections were counterstained with hematoxylin and mounted.
IHC staining was performed with rabbit anti-EVC2 antibody (1:2,000) or non-immune rabbit Ig as a negative control using WT embryos in the same manner as described above. Samples of KO embryos were used as a negative control.
Samples at postnatal (P) stages were also investigated for IHC staining with anti-β-gal antibody (for KO and Het samples) and anti-EVC2 antibody (for WT samples). The head of each mouse was sagittally dissected into two equal halves, followed by decalcification with EDTA. Serial X-rays were taken on a weekly basis to confirm proper decalcification. Decalcified samples were then embedded in paraffin, sectioned, and IHC staining was performed in the same manner as described above.
To identify the type of Evc2-expressing cells, fluorescence IHC was performed with anti-β-gal and anti-SOX9 (EMD Millipore) antibodies using Het and KO embryos at E16.5 with DAPI nuclear staining as previously described (20). For staining with WT embryos at E16.5, anti-EVC2 (sc-28393, Santa Cruz Biotechnology, Inc) and anti-SOX9 antibodies were used. Alexa Fluor 488 (for anti-SOX9) and 594 (for anti-β-gal or anti-EVC2) (both Life Technologies) were used as secondary antibodies and mounted with ProLong Gold antifaint mountant with DAPI (Life Technologies).
2.6. Skeletal double staining
The heads of Evc2 WT and KO mice at P0, P18, and P24 were collected and the skin was then carefully peeled off in hot water at 70°C followed by fixation in 95% ethanol overnight. Samples were incubated with acetone overnight for the removal of fat tissues, rinsed with deionized water and stained for cartilage with alcian blue 8GX. Samples were washed with 70% ethanol, transferred to 1% potassium hydroxide (KOH) solution and incubated overnight for tissue clearing. Samples were then incubated for staining bone with alizarin red, placed in 1% KOH / 20% glycerol for further tissue clearing and kept in 50% glycerol / 50% ethanol until being photographed.
2.7. Imaging
The embryos stained with X-Gal and postnatal mouse samples with skeletal double staining were visualized using a stereomicroscope (SZX16 Research Stereo Microscope, Olympus). Slide sections were viewed with an upright microscope (BX61 Upright Microscope, Olympus), and images were captured with an assistance of a high-resolution camera (Q-Color5TM, Olympus). Fluorescence images were taken under a confocal microscope (Nikon C1).
3. Results
3.1. Evc2 is highly expressed in chondrocytes in the developing craniofacial structures as well as in osteocytes within the adult craniofacial bones
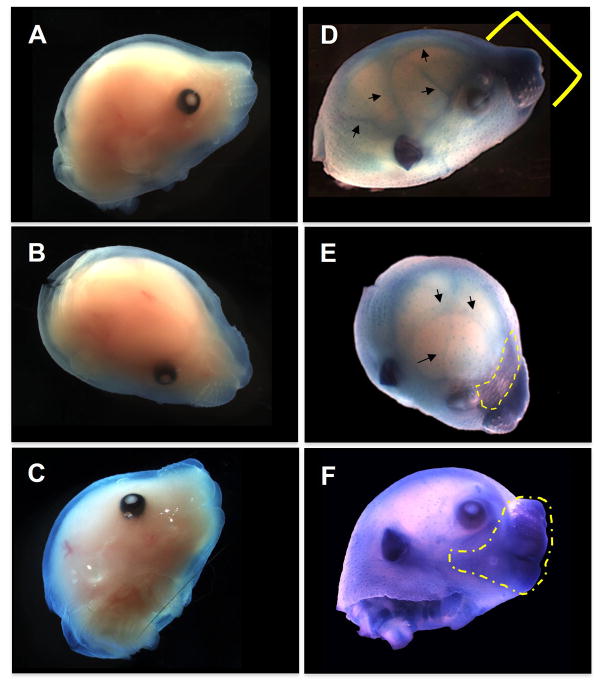
In order to track the spatiotemporal expression of Evc2, a reporter cassette (IRES-LacZ) was engineered as a part of the targeting vector where LacZ was knocked in under the control of the endogenous Evc2 promoter. This IRES-LacZ cassette allows us to identify the Evc2-expressing cells. The staining pattern of Evc2 KO head at E15.5 by X-gal was similar to that of Het (data not shown) with higher intensity in KO embryos, and no staining was detected in WT (Fig. 1A–C). In the KO embryos, the viscerocranium including the nasal maxilla-premaxilla (Fig. 1D, indicated by a bracket) and the anterior part of the mandibular area (Fig. 1F, indicated by a dotted area) showed the highest LacZ activity. The activity was also observed in neurocranium including the frontal bone area (Fig. 1E, indicated by a dotted area) and the cranial sutures (Figs. 1D & E, indicated by arrows).
Figure 1. Expression pattern of Evc2 in the craniofacial tissues at the embryonic stage.
(A–F) Visualization of Evc2-expressing cell distribution by X-gal staining at E15.5. Evc2 WT (AC) and KO (D–F) embryos were stained with X-gal. The KO embryo exhibited a higher LacZ activity in the anterior portion of the viscerocranium in the nasal, premaxilla, maxilla, and mandibular (D and F; indicated by a bracket and a dotted line) areas. Expression was also detected in the anterior part of frontal bones (E; indicated by a dotted line) and in the suture area of the neurocranium (D and E; indicated by arrows). WT embryos used as a negative control showed no LacZ activity (A–C). At least 3 samples per each genotype have been investigated and the representative head images were acquired and presented.
As we found that there was no obvious morphological difference between Evc2 WT and Het embryo heads, we took an advantage of the IRES-LacZ cassette and IHC analysis with anti-β-gal antibody was performed to identify the Evc2-expressing cells using Het embryos at E15.5 (Sup-Fig. 1). The immunoreactivity was observed in the areas of nasal capsule (Sup-Fig. 1C) and nasal septum (Sup-Fig. 1G), paranasal (maxillary and ethmoid) sinuses (Sup-Fig. 1L), palatal mesenchyme (Sup-Fig. 1E) and Meckel’s cartilage (Sup-Fig. 1J). The results showed that major cell types positive for β-gal antibody staining in Het were chondrocytes (Sup-Figs. 1C, 1G, 1J and 1L, indicated by filled arrows), and to a lesser extent mesenchymal cells (Sup-Fig. 1E, indicated by arrowheads) and lining epithelial cells (Sup-Fig. 1L, indicated by open arrows). No immunoreactivity was observed when non-immune immunoglobulin (Ig) was used (Sup-Figs. 1B, 1D, 1F, 1I and 1K), showing the specificity of the antibody used.
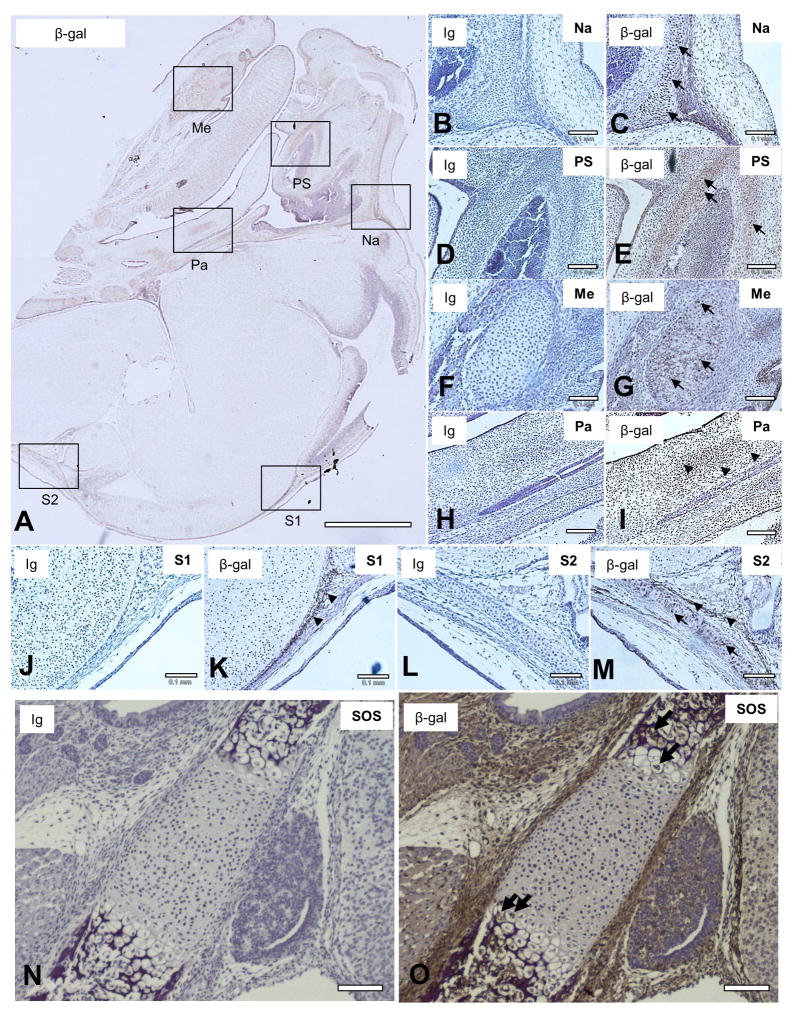
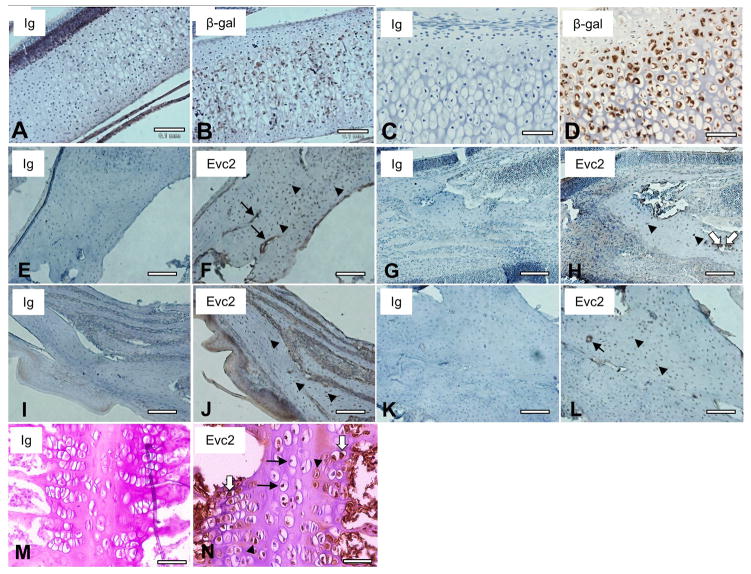
There is a possibility that deficiency of the gene causes structural changes. Therefore, we next investigated types of Evc2-expressing cells in KO embryos and IHC analysis using anti-β-gal antibody was performed at the same time point as Het (E15.5) (Fig. 2). The immunoreactivity was observed in the areas of nasal capsule (Fig. 2C), paranasal (maxillary and ethmoid) sinuses (Fig. 2E), palatal mesenchyme (Fig. 2I), Meckel’s cartilage (Fig. 2G), and cranial suture tissues (Figs. 2K & 2M). Consistent with the IHC analysis with Het embryos, the cell types which expressed Evc2 included mainly chondrocytes (Figs. 2C, 2E, 2G, and 2M, indicated by arrows) and mesenchymal cells in some areas (Fig. 2I, 2K and 2M, indicated by arrowheads) using KO embryos. We also investigated the Evc2-expressing cells in cranial base synchondrosis using the Evc2 KO embryo at E18.5, and hypertrophic chondrocytes of spheno-occipital synchondrosis (SOS) were positively stained (Fig. 2O). No immunoreactivity was observed when non-immune immunoglobulin (Ig) was used (Figs. 2B, 2D, 2F, 2H, 2J, 2L and 2N), showing the specificity of the antibody used. These histological data also confirmed that the staining pattern of Evc2 KO was similar to that of Het.
Figure 2. Detection of Evc2-expressing cells in the homozygous Evc2 KO embryo.
IHC staining using anti-β-gal antibody was performed to visualize the Evc2-expressing cells in the craniofacial structures of homozygous Evc2 KO embryos at E15.5 (A–M) and E18.5 (N, O) (brown). Sagittal sections of KO head were prepared and the area of nasal capsule (Na), paranasal sinus (PS), Meckel’s cartilage (Me), posterior palate (Pa), or cranial suture (S1, S2) was indicated by an open box (A), respectively. Expression of β-gal was detected in chondrocytes (indicated by arrows) of Na (C), PS (E), Me (G), S2 (M), and spheno-occipital synchondrosis (SOS) (O) as well as in mesenchymal cells (indicated by arrowheads) of Pa (I) and S1 (K). Serial sections of the KO head were also stained with non-immune immunoglobulin (Ig) as a negative control and no immunoreactivity in the corresponding craniofacial areas was observed (Na in (B), PS in (D), Me in (F), Pa in (H), S1 in (J), S2 in (L), SOS in (N), respectively). Sections were counterstained with hematoxylin (blue). At least 3 head samples have been investigated using at least 6 sections per sample and the representative images were acquired and presented. Scale bars; A = 1 mm, B–O = 0.1 mm.
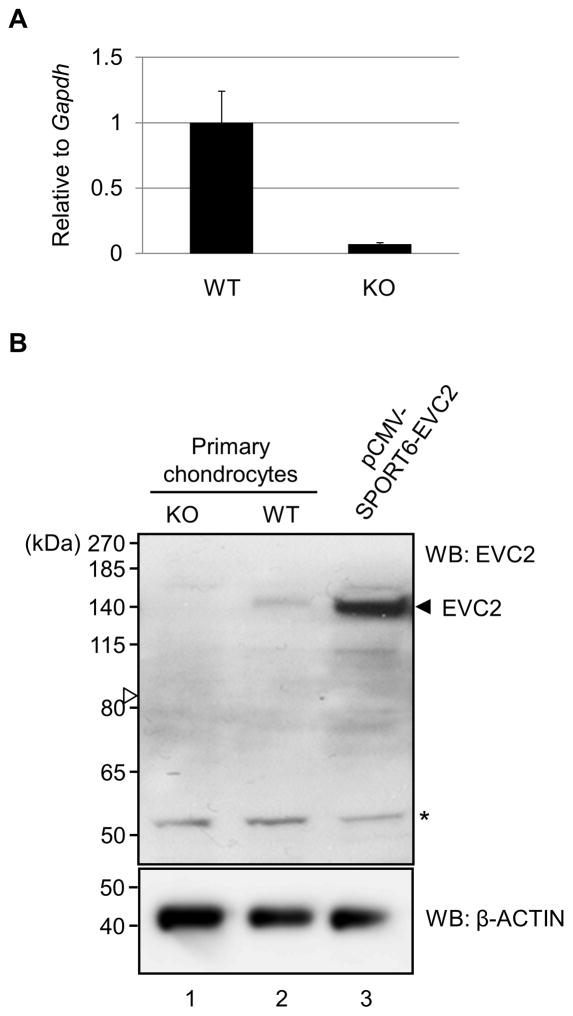
To further determine whether Evc2 is expressed in chondrocytes, the gene expression level of Evc2 in primary chondrocytes from Evc2 WT and KO embryos at E18.5 was determined by real-time PCR analysis using the Evc2-specific primers-probe which detects amplicons spanning exon 6 and 7. Our results demonstrated that Evc2 expression was detected in WT primary chondrocytes, while that of Evc2 KO primary chondrocytes was markedly reduced (~13.8 times lowered) as compared to WT (Fig. 3A). We were able to detect β-gal staining and LacZ activity suggesting that this level of reduction can still generate enough levels of β-gal protein for aforementioned detection.
Figure 3. Expression of EVC2 in Evc2 WT and KO primary chondrocytes.
(A) Gene expression of Evc2 in primary chondrocytes. Quantitative real-time PCR analysis was performed using Evc2 WT and KO primary chondrocytes. The mean fold change in the expression of Evc2 was calculated based on the normalization to that of glyceraldehyde-3-phosphate dehydrogenase (Gapdh) using the value of Evc2 in WT chondrocytes as a calibrator. The values are shown as the mean + S.D. based on triplicate assays. (B) Analysis of EVC2 protein levels in Evc2 WT and KO chondrocytes by rabbit anti-EVC2 antibody. Western blot (WB) analysis showed the detection of EVC2 in WT chondrocytes (upper panel, lane 2, at ~140kDa) and a lack of detection of EVC2 in Evc2 KO chondrocytes (upper panel, lane 1). Immunodetection of a band at ~140kDa in a positive control (i.e. pCMV-SPORT6-Evc2 plasmid-transfected ATDC5 chondrogenic cells, indicated by an arrowhead) indicated successful detection and validation of rabbit anti-EVC2 antibody. An open arrowhead indicated the expected molecular weight of the truncated EVC2 protein, which was not identified. β-ACTIN was used as loading control. *; non-specific band.
We then determined the levels of EVC2 protein in chondrocytes by characterizing a rabbit anti-EVC2 antibody (Bethyl Laboratories, Inc). Cell lysates from primary chondrocytes of Evc2 WT and KO embryos were obtained and Western blot analysis was performed using the rabbit anti-EVC2 antibody. The ATDC5 chondrogenic cell lysates transfected with mouse Evc2 expression plasmid were used as a positive control to compare the molecular weight of EVC2 protein with the endogenous EVC2 protein in primary chondrocytes of Evc2 WT. The results showed that an immunoreactive band to anti-EVC2 antibody was detected at ~140kDa when Evc2 expression plasmid was transfected (indicated by an arrowhead; Fig. 3B, upper panel, lane 3), demonstrating the successful detection of EVC2 protein by rabbit anti-EVC2 antibody and successful transfection of Evc2 expression plasmid in ATDC5 chondrogenic cells. The immunoreactive band at the same molecular weight as overexpressed EVC2 protein was identified when cell lysates of Evc2 WT primary chondrocytes were applied (Fig. 3B, upper panel, lane 2), showing the successful detection of endogenous EVC2 protein in primary chondrocytes. However, no immunoreactive band was observed when cell lysates of Evc2 KO primary chondrocytes were applied at the expected size of the truncated EVC2 protein (indicated by an open arrowhead; Fig. 3B, upper panel, lane 1). Our results showed that anti-EVC2 antibody was successfully generated and characterized, and also that the truncated form of EVC2 protein, unlike β-gal protein, was not detectable in Evc2 KO primary chondrocytes.
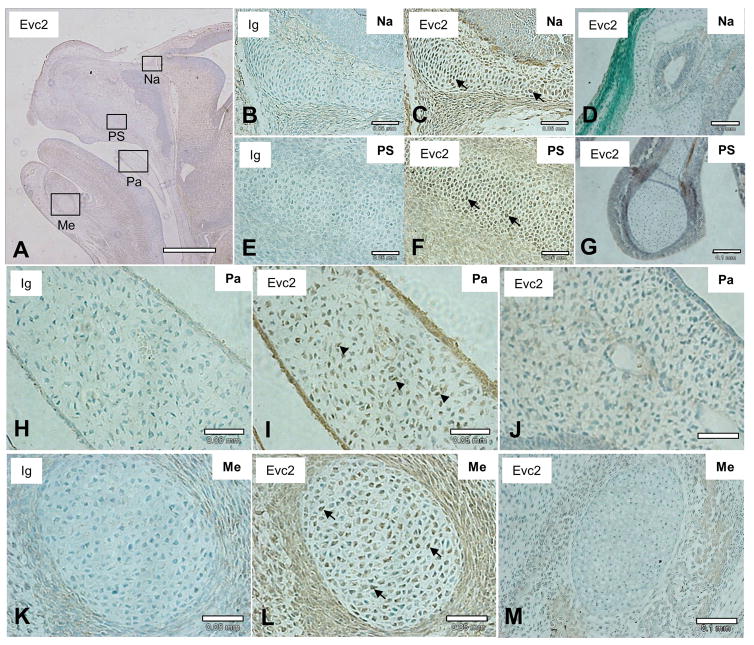
To investigate the expression of EVC2 protein in the craniofacial tissues, IHC with the rabbit anti-EVC2 antibody was performed using WT embryos at E15.5 (Fig. 4). The results demonstrated that EVC2 was mainly produced in chondrocytes (indicated by filled arrows) of the nasal capsule (Fig. 4C), paranasal (maxillary and ethmoid) sinuses (Fig. 4F) and Meckel’s cartilage (Fig. 4L). EVC2 was also expressed in mesenchymal cells of the palate (Fig. 4I, indicated by arrowheads). No immunoreactivty was observed when KO samples at E15.5 were stained with the rabbit anti-EVC2 antibody in the areas of nasal capsule (Fig. 4D), paranasal (ethmoid) sinus (Fig. 4G), palate (Fig. 4J) and Meckel’s cartilage (Fig. 4M), reproducing the data obtaining from Western blot analyses (Fig. 3). Our data confirmed that endogenous EVC2 protein was detected in the same areas where β-gal staining and LacZ activity were observed in Evc2 Het/KO embryos.
Figure 4. Expression of endogenous EVC2 protein in the craniofacial tissues.
(A, C, F, I, L) The endogenous EVC2 expression pattern was visualized by IHC analysis using anti-EVC2 antibody with WT embryo at E15.5. Evc2 KO embryo at E15.5 was also used as a negative control for anti-EVC2 antibody (D, G, J, M). The immunoreactivity was detected in the areas of the nasal capsule (Na), paranasal sinus (PS), palate (Pa) and Meckel’s cartilage (Me). The EVC2 protein expression was clearly observed in chondrocytes in these areas (Na in (C), PS in (F), Me in (L)) as well as in mesenchymal cells of Pa in (I). Serial section of the WT head was also stained with non-immune Ig as a negative control and no immunoreactivity in the corresponding craniofacial areas was observed (Na in (B), PS in (E), Pa in (H), Me in (K), respectively). No immunoreactivity was detected using Evc2 KO embryo with anti-EVC2 antibody in areas of Na (D), PS (G), Pa (J) and Me (M). Sections were counterstained with hematoxylin. At least 3 samples per each genotype have been investigated using at least 6 sections per sample and the representative images were acquired and presented. Scale bars; A= 1 mm, B, C, E, F, H, I, K, L = 0.05 mm, D, G, J, M = 0.1 mm.
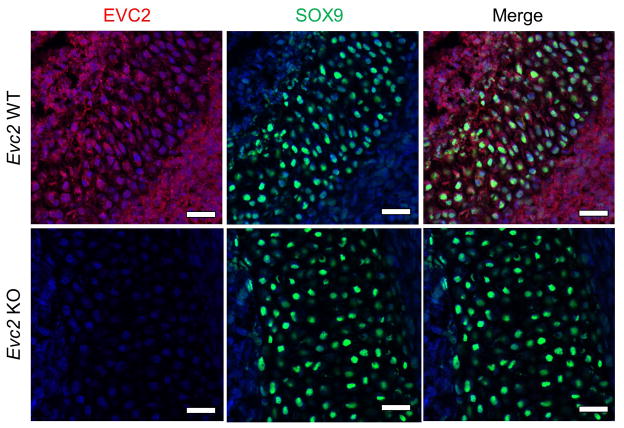
To further identify the cell type that mainly expresses EVC2, the expression of SRY (sex determining region Y)-box 9 (SOX9), a well-known chondrocyte differentiation marker, was investigated by fluorescence IHC with double labeling using anti-β-gal and anti-SOX9 antibodies. As shown in Sup-Fig. 2, the majority of β-gal-positive cells (Sup-Fig. 2, left panels, red) in paranasal sinus were overlapped with SOX-9 positive cells (Sup-Fig. 2, middle panels, green) in both Het (Sup-Fig. 2, upper right panel) and KO (Sup-Fig. 2, lower right panel) embryos. Moreover, we also performed the double labeling with anti-EVC2 (Fig. 5, left panels, red) and anti-SOX9 (Fig. 5, middle panels, green) antibodies using WT (Fig. 5, upper panels) and KO (Fig. 5, lower panels) embryos and the data clearly showed that cells that express endogenous EVC2 in paranasal sinus were positively stained and overlapped with anti-SOX9 antibody (Fig. 5, upper right panel). No immunoreactivity to anti-EVC2 antibody was detected when KO embryos were used (Fig. 5, lower left panel), showing the specificity of anti-EVC2 antibody used. The results thus confirm that the cell type that mainly expresses EVC2 is chondrocyte in the mouse craniofacial tissues.
Figure 5. The cell type that expresses EVC2 is chondrocyte.
Fluorescence IHC with double labeling was performed to identify the cell type that expresses EVC2. Sagittal sections of WT and KO heads at E16.5 were prepared. The endogenous EVC2 expression was visualized by fluorescence IHC analysis with anti-EVC2 (left panels, red) and anti-SOX9 (middle panels, green) antibodies using WT (upper panels) and KO (lower panels) embryos. The immunoreactivity was detected in the area of paranasal sinus (PS) and is shown. No immunoreactivity was detected using Evc2 KO embryo with anti-EVC2 antibody (lower left panel). Sections were counterstained with DAPI. Scale bars; 0.04 mm.
We next investigated the postnatal expression of Evc2 (Fig. 6) using Het and KO samples at 3 weeks by IHC with anti-β-gal antibody (Fig. 6A–D). The results showed that the immunoreactive cells were identified only in the cartilaginous structures of the nasal septum (Fig. 6B in Het, 6D in KO) and the cells immunostained were hypertrophic chondrocytes. No immunoreactivity was observed when non-immune Ig was used (Figs. 6A & 6C). We then investigated the endogenous EVC2 protein expression by IHC with anti-EVC2 antibody using WT samples at 6 weeks. The results demonstrated that the expression of EVC2 was observed in osteocytes (indicated by arrowheads) of the premaxilla (Fig. 6F), paranasal (ethmoid) sinus (Fig. 6H), palate (Fig. 6J), and mandible (Fig. 6L). In spheno-occipital synchondrosis, EVC2 was highly expressed in resting, proliferative and hypertrophic chondrocytes (Fig. 6N). The expression of EVC2 was also observed in endothelial-like cells (Figs. 6F and 6L, indicated by filled arrowheads), osteoblasts (Fig. 6H, indicated by open arrows) and hematopoietic cells (Fig. 6N). No immunoreactivity was observed in these areas when non-immune Ig was used (Figs. 6A, 6C, 6E, 6G, 6I, 6K and 6M).
Figure 6. EVC2 expression at postnatal stages.
Sagittal sections of Het (A, B) and KO (C, D) heads at 3 weeks were used to visualize Evc2-expressing cells. IHC staining with anti-β-gal antibody showed Evc2-expressing cells (brown) in hypertrophic chondrocytes in the nasal septum area in Het (B) and KO (D). Corresponding areas to (B, D), respectively, were stained with non-immune Ig as negative controls (A, C). (E–N) Sagittal sections of WT heads at 6 weeks were used to perform IHC staining by anti-EVC2 antibody. Expression of EVC2 was detected in osteocytes of the premaxilla (F, arrowheads), paranasal (ethmoid) sinus (H, arrowheads), palate (J, arrowheads) and mandible (L, arrowheads). Expression was also detected in osteoblasts of paranasal sinuses (H; indicated by open arrows), endothelial-like cells (F, L, filled arrows) and hematopoietic cells (N). In addition, EVC2 expression was observed in chondrocytes in the resting (arrows), proliferative (arrowheads) and hypertrophic (open arrows) zones at spheno-occipital synchondrosis (N). No immunoreactivity was observed with non-immune Ig as a negative control in the corresponding areas (E, G, I, K, M). Sections were counterstained with hematoxylin. At least 3 head samples have been investigated using at least 6 sections per sample and the representative images were acquired and presented. Scale bars; A, B, E–L = 0.1mm, C, D = 0.05 mm, M, N = 0.04 mm.
3.2. Skeletal staining reveals defects of the mineralized structures in Evc2 KO mice
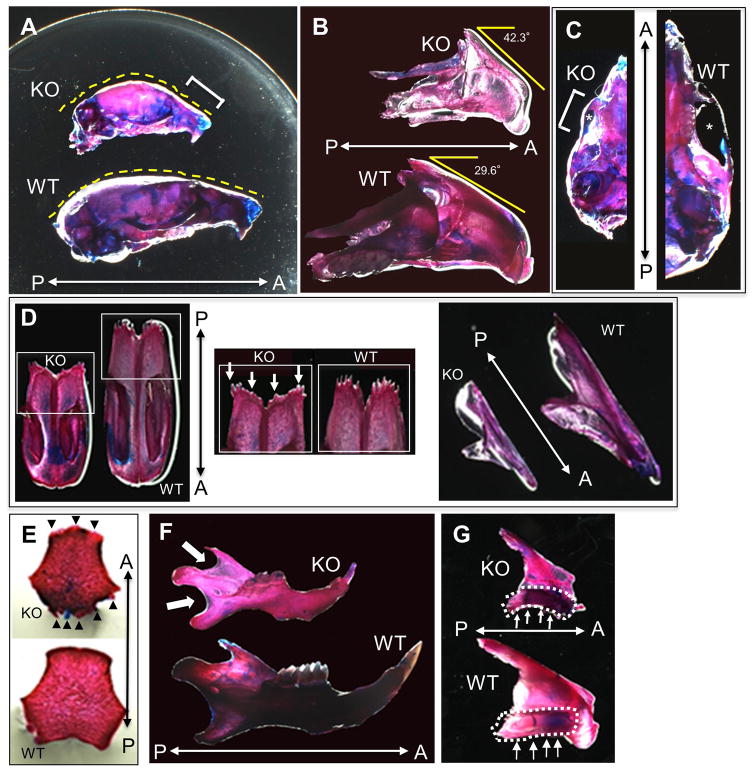
We have previously reported that EVC2 is important for normal skeletogenesis and skull bones including nasal and frontal bones that were smaller and less mineralized by micro CT analysis (20). To investigate the developing bones and cartilages of the craniofacial structures in Evc2 WT and KO mice, skeletal double staining was performed in the manner as we previously reported (23). The size of the upper skull in KO was significantly smaller than that of WT at postnatal day (P)18 (Fig. 7A and 7C). In addition to the difference in size, the shape of craniofacial bones in KO including nasal bone (Fig. 7A, indicated by a bracket), premaxilla (Fig. 7B) and zygomatic arch (Fig. 7C, indicated by a bracket) was deformed as compared to that in WT. The profile of nasal-cranial bone outline in the nasal-premaxilla area was retruded and depressed in KO as compared to WT (Fig. 7A, outlined with dotted lines). Moreover, the growth of the retruded premaxilla in KO appeared to be in a more vertical direction indicated by the more obtuse angle of the premaxilla to the horizontal plane as compared to WT (Fig. 7B, 42.3° in KO vs. 29.6° in WT). The zygomatic arch of WT (Fig. 7C, right) was properly curved showing a convex shape, and there was a considerable amount of space between the zygomatic bone and the infra-temporal surface of the skull (Fig. 7C, right, indicated by an asterisk). On the other hand, the zygomatic bone in KO (Fig. 7C, left, indicated by a bracket) appeared to be underdeveloped and there was a decrease of the space between the zygomatic bone and infra-temporal surface likely due to the growth failure of the arch bone (Fig. 7C, indicated by an asterisk).
Figure 7.
Skeletal double staining of Evc2 WT and KO craniofacial tissues. The heads of Evc2 WT and KO mice at postnatal day (P) 0, P18, and P24 were stained with alizarin red and alcian blue, and craniofacial bones were further dissected for comparison. (A) Lateral view of WT and KO mouse skulls at P18. The size of the KO surface area was smaller than that of WT. The nasal-premaxilla area in KO (bracket) is more depressed and retruded with an overall convex skull outline (yellow dotted line) as compared to WT. A; anterior, P; posterior. (B) Lateral view of the premaxilla-maxilla-palatine bone complex at P18, showing that KO exhibited the retruded premaxilla with a larger degree than WT. A; anterior, P; posterior. (C) Ventral view of WT and KO mouse skulls at P18 showing depressed zygomatic arch in KO (bracket). Note that the infra-temporal space (asterisk) below the arch in KO was smaller than that in WT. A; anterior, P; posterior. (D) Ventral (left and middle panels) and lateral (right panel) view of nasal bones at P24. Irregular and wide border of the frontal process of the nasal bone (middle panel, arrows) was found in KO as compared to WT. A; anterior, P; posterior. (E) Ventral view of basioccipital bones. Irregular borders and abnormal shape of the basioccipital bone in KO (arrowheads) at P0 as compared to WT. A; anterior, P; posterior. (F) Lateral view of mandibles at P18. KO showed transparent areas at the mandibular notch and the posterior mandibular bone border (arrows) compared to WT. A; anterior, P; posterior. (G) Lateral view of the palatine bones at P24. The palatine bone in KO was deformed in a more curved shape as compared to WT (indicated by dotted circle areas and arrows). A; anterior, P; posterior.
The extent of alizarin red staining was lesser in KO than that in WT, which was observed in the premaxilla-maxilla complex (Fig. 7B) and mandibular notch and posterior border of the mandibular bones (Fig. 7F, indicated by arrows), suggesting the thinner bone and/or poorer quality of bone/minerals present in KO. These craniofacial bone samples were further dissected, and the nasal (Fig. 7D), basioccipital (Fig. 7E) and palatine (Fig. 7G) bones from WT and KO were compared. Both nasal and basioccipital bones in KO exhibited irregular bony borders (indicated by arrows) and abnormal shapes. The palatine bone in KO exhibited a more curved shape than WT (Fig. 7G, indicated by dotted areas and arrows).
These data indicate that Evc2 deficiency led to the impairment of certain craniofacial structures, which is consistent and overlapping with the areas where we observed Evc2 expression (Figs. 2 and 4–6, and Sup-Fig. 1 and 2).
4. Discussion
Chondrocytes, the primary cell type of cartilage, are the main units of the growth plate that contribute to bone development by endochondral ossification. Cartilage develops through chondrocyte proliferation, differentiation followed by apoptosis and replacement by bone formation (24). In our study, Evc2 is mainly detected in chondrocytes positively stained with SOX9 (Fig. 5). These cells that express EVC2 are observed in the areas of nasal capsule, nasal septum, paranasal (maxillary and ethmoid) sinuses, Meckel’s cartilage and spheno-occipital synchondrosis. Our results by skeletal double staining showed abnormal shape of nasal and basioccipital bones with irregular borders in Evc2 KO mice (Figs. 7D & 7E). Consistent with our finding, Evc (not Evc2) KO mice also demonstrated the irregular margins between bones and cartilages in the basioccipital junction (11), indicating that Evc2 as well as Evc are required for chondrocyte cell function(s) in the developing cranial growth plates at the cartilage bone interface, which subsequently lead to the recruitment and invasion of bone trabeculae. Therefore, the clinical phenotype where EvC patients had mid-facial deficiency may be likely due to the effect of EVC2/EVC on these craniofacial bone structure growth.
It has been reported that many inherited diseases resulting from non-sense mutations activate nonsense-mediated mRNA decay (NMD), leading to the degradation of such premature termination codon (PTC)-containing mRNAs (25). In some cases such as β-thalasemia, mutations in β-GLOBIN do not elicit NMD pathway, resulting in the presence of truncated β-GLOBIN protein which functions as a dominant-negative form (26). Since the remarkably reduced amount of Evc2 mRNA was detected in Evc2 KO chondrocytes (Fig. 3A), this indicates that the aberrant Evc2 mRNA with PTC in exon 12 likely activates NMD pathway. This also, at least in part, explains the reason why we could not detect any truncated EVC2 protein in Evc2 KO chondrocytes (Fig. 3B).
Our IHC staining with anti-EVC2 antibody demonstrated that EVC2 protein is expressed in the resting, proliferating and hypertrophic chondrocytes of spheno-occipital synchondrosis (Fig. 6N), suggesting the presence of potential chondrocyte phenotype in Evc2 KO mice. Consistent with this, the Evc2-expressiing cells were present in hypertrophic chondrocytes at an earlier cranial base synchondrosis development (Fig. 2O) and it has been demonstrated that a disorganized and irregular arrangement of proliferative chondrocytes and premature chondrocyte differentiation with premature growth plate closure were observed in the growth plates of long bones in Japanese brown cattle with bovine chondrodysplastic dwarfism (bcd) (27), which was identified to have mutations in the LBN gene (bovine ortholog of EVC2) (9). Furthermore, Tyrolean Grey cattle with bcd due to LBN mutations demonstrated similar histopathological features in long bones to the Japanese brown cattle (28). Our recent report in Evc2 KO mice also showed the skeletal defects, manifesting the dwarfism phenotype (20). All of these bcd and Evc2 limb phenotype is in agreement with our Evc2 expression data. Furthermore, it has been previously reported that Evc KO mice, generated by replacing the 1st exon with a LacZ cassette (11), showed a positive LacZ activity in the craniofacial region in both mesenchymal (maxilla, mandible and cranial sutures) and cartilaginous (nasal capsule, septum and growth plate of the base of the skull) structures. Evc-expressing cells were observed in the resting and proliferating chondrocytes of the epiphyseal growth plate in long bones (11) and Evc KO mice showed premature chondrocyte differentiation in the growth plates of long bones (11). Taken together, our and previous data suggest that EVC/EVC2 plays a role in cranial base expansion and endochondral ossification.
It is well known that the neural crest cells (NCCs) are required for proper craniofacial development and they contribute to the formation of most viscerocranium structures such as frontonasal process and teeth, while the posterior parts of viscerocranium are from mesoderm (29, 30). In the current study, abnormal craniofacial bone morphology is consistently associated with Evc2 expression patterns. To support this, our recent study also showed that Evc2 KO mice had the reduced incisor length (20). Since many of NCC-derived cranial bones are hypomorphic in Evc2 KO mice such as the nasal bone and premaxilla-maxilla complex, it is an interesting future effort to understand how EVC2 differentially regulates proliferation and/or migration of osteo- and odonto-progenitor cells between neural crest-derived and mesodermal-derived cell populations.
In summary, our data demonstrated that Evc2 deficiency led to the impairment of proper craniofacial bone morphology, particularly in the anterior craniofacial structures. Although the expression of EVC2 was mainly found in chondrocytes, we also observed the EVC2 expression in other cell types including mesenchymal cells, osteoblasts/osteocytes, endothelial-like cells, and hematopoietic cells, suggesting that the craniofacial bone phenotypes in Evc2 KO mice were likely cell type context-dependent. Further study is clearly warranted to investigate the effect of Evc2 deficiency on craniofacial bone phenotypes in a tissue-specific fashion to identify the contribution of EVC2 to the specific cell lineage-derived craniofacial bone formation.
Supplementary Material
IHC staining using anti-β-gal antibody was performed to visualize the Evc2-expressing cells in the craniofacial structures of Het Evc2 KO embryos at E15.5 (brown) (A–L). Sagittal sections of Het heads at E15.5 were used (A, H) and the area of nasal capsule (Na), palate (Pa), nasal septum (NS), Meckel’s cartilage (Me), and paranasal sinus (PS) was indicated by an open box, respectively. Expression of β-gal was detected in chondrocytes (indicated by filled arrows) of Na (C), NS (G), Me (J) and PS (L) as well as in the mesenchymal cells of Pa (E, indicated by arrowheads) and lining epithelial cells (L, indicated by open arrows). Serial sections of the Het heads were also stained with non-immune Ig as a negative control and no immunoreactivity in the corresponding craniofacial areas was observed (Na in (B), Pa in (D), NS in (F), Me in (I) and PS in (K), respectively). Sections were counterstained with hematoxylin (blue). Scale bars; A, H = 1 mm, B–G, I–L = 0.05 mm. At least 3 head samples have been investigated using at least 6 sections per sample and the representative images were acquired and presented.
Sagittal sections of Het and KO heads at E16.5 were prepared. Fluorescence IHC staining with anti-β-gal (left panels, red) and anti-SOX9 (middle panels, green) antibodies was performed followed by incubating with secondary Alexa fluor 594 and 488 antibodies using Het (upper panels) and KO (lower panels) embryos. Merged images were shown on the right panels. Sections were counterstained with DAPI. Scale bars; 0.04 mm.
Highlights.
Evc2 knockout mouse was generated by introducing a stop codon followed by IRES-LacZ.
This IRES-LacZ cassette allows us to identify the Evc2-expressing cells.
Evc2-expressing cells were mainly chondrocytes in the anterior viscerocranium.
Endogenous EVC2 protein was observed in these corresponding cells.
Evc2 deficiency showed abnormal craniofacial bones in the anterior viscerocranium.
Acknowledgments
We thank Dr. Michael T. Kirber for his expertise and assistance in imaging studies (Cellular Imaging Core at Boston University Medical Campus).
Funding: This study was supported by NIDCR grants DE019527 (Y. Mochida.), DE020843 (Y. Mishina) and in part by CTSA UL1-TR000157. This study was also supported by the Intramural Research Program of the NIEHS/NIH to Y. Mishina (ES071003-0).
Abbreviations
- EvC
Ellis-van Creveld
- EVC2
Ellis-van Creveld 2
- WT
wild type
- KO
knockout
- Het
heterozygous
- IRES
Internal Ribosomal Entry Site
- β-gal
β-galactosidase
- IHC
immunohistochemistry
- Hh
hedgehog
- Smo
Smoothened
- Ptch1
Patched1
- SuFu
Suppressor of Fused Homolog
- E
embryonic
- P
postnatal
- PBS
phosphate buffered saline
- PFA
paraformaldehyde
- RT
room temperature
- SOX9
sex determining region Y-box 9
- Ig
Immunoglobulin
- KOH
potassium hydroxide
- NCC
neural crest cell
- LBN
LIMBIN
Footnotes
Competing interests: The authors declare no potential conflicts of interest with respect to the authorship and/or publication of this article.
Ethical approval: All animal procedures were approved by the IACUC at Boston University Medical Campus (Boston, MA, USA) and University of Michigan (Ann Arbor, MI, USA) and performed in accordance with the NIH Guide for the Care and Use of Laboratory Animals.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Ellis RW, van Creveld S. A Syndrome Characterized by Ectodermal Dysplasia, Polydactyly, Chondro-Dysplasia and Congenital Morbus Cordis: Report of Three Cases. Arch Dis Child. 1940;15(82):65–84. doi: 10.1136/adc.15.82.65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Baujat G, Le Merrer M. Ellis-van Creveld syndrome. Orphanet J Rare Dis. 2007;2:27. doi: 10.1186/1750-1172-2-27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Metrakos JD, Fraser FC. Evidence for a hereditary factor in chondroectodermal dysplasia (Ellis-van Creveld syndrome) American journal of human genetics. 1954;6(2):260–269. [PMC free article] [PubMed] [Google Scholar]
- 4.Polymeropoulos MH, Ide SE, Wright M, Goodship J, Weissenbach J, Pyeritz RE, et al. The gene for the Ellis-van Creveld syndrome is located on chromosome 4p16. Genomics. 1996;35(1):1–5. doi: 10.1006/geno.1996.0315. [DOI] [PubMed] [Google Scholar]
- 5.Ruiz-Perez VL, Ide SE, Strom TM, Lorenz B, Wilson D, Woods K, et al. Mutations in a new gene in Ellis-van Creveld syndrome and Weyers acrodental dysostosis. Nat Genet. 2000;24(3):283–286. doi: 10.1038/73508. [DOI] [PubMed] [Google Scholar]
- 6.Galdzicka M, Patnala S, Hirshman MG, Cai JF, Nitowsky H, Egeland JA, et al. A new gene, EVC2, is mutated in Ellis-van Creveld syndrome. Mol Genet Metab. 2002;77(4):291–295. doi: 10.1016/s1096-7192(02)00178-6. [DOI] [PubMed] [Google Scholar]
- 7.Ruiz-Perez VL, Tompson SW, Blair HJ, Espinoza-Valdez C, Lapunzina P, Silva EO, et al. Mutations in two nonhomologous genes in a head-to-head configuration cause Ellis-van Creveld syndrome. Am J Hum Genet. 2003;72(3):728–732. doi: 10.1086/368063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ruiz-Perez VL, Goodship JA. Ellis-van Creveld syndrome and Weyers acrodental dysostosis are caused by cilia-mediated diminished response to hedgehog ligands. American journal of medical genetics Part C, Seminars in medical genetics. 2009;151C(4):341–351. doi: 10.1002/ajmg.c.30226. [DOI] [PubMed] [Google Scholar]
- 9.Takeda H, Takami M, Oguni T, Tsuji T, Yoneda K, Sato H, et al. Positional cloning of the gene LIMBIN responsible for bovine chondrodysplastic dwarfism. Proc Natl Acad Sci U S A. 2002;99(16):10549–10554. doi: 10.1073/pnas.152337899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Blair HJ, Tompson S, Liu Y-N, Campbell J, MacArthur K, Ponting CP, et al. Evc2 is a positive modulator of Hedgehog signalling that interacts with Evc at the cilia membrane and is also found in the nucleus. BMC biology. 2011;9:14. doi: 10.1186/1741-7007-9-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ruiz-Perez VL, Blair HJ, Rodriguez-Andres ME, Blanco MJ, Wilson A, Liu YN, et al. Evc is a positive mediator of Ihh-regulated bone growth that localises at the base of chondrocyte cilia. Development. 2007;134(16):2903–2912. doi: 10.1242/dev.007542. [DOI] [PubMed] [Google Scholar]
- 12.Pacheco M, Valencia M, Caparros-Martin JA, Mulero F, Goodship JA, Ruiz-Perez VL. Evc works in chondrocytes and osteoblasts to regulate multiple aspects of growth plate development in the appendicular skeleton and cranial base. Bone. 2012;50(1):28–41. doi: 10.1016/j.bone.2011.08.025. [DOI] [PubMed] [Google Scholar]
- 13.Dorn KV, Hughes CE, Rohatgi R. A Smoothened-Evc2 complex transduces the Hedgehog signal at primary cilia. Developmental cell. 2012;23(4):823–835. doi: 10.1016/j.devcel.2012.07.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Yang C, Chen W, Chen Y, Jiang J. Smoothened transduces Hedgehog signal by forming a complex with Evc/Evc2. Cell research. 2012;22(11):1593–1604. doi: 10.1038/cr.2012.134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Caparros-Martin JA, Valencia M, Reytor E, Pacheco M, Fernandez M, Perez-Aytes A, et al. The ciliary Evc/Evc2 complex interacts with Smo and controls Hedgehog pathway activity in chondrocytes by regulating Sufu/Gli3 dissociation and Gli3 trafficking in primary cilia. Hum Mol Genet. 2013;22(1):124–139. doi: 10.1093/hmg/dds409. [DOI] [PubMed] [Google Scholar]
- 16.Goetz SC, Anderson KV. The primary cilium: a signalling centre during vertebrate development. Nature reviews Genetics. 2010;11(5):331–344. doi: 10.1038/nrg2774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Goor D, Rotem Y, Friedman A, Neufeld HN. Ellis-van Creveld syndrome in identical twins. Br Heart J. 1965;27(6):797–804. doi: 10.1136/hrt.27.6.797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Susami T, Kuroda T, Yoshimasu H, Suzuki R. Ellis-van Creveld syndrome: craniofacial morphology and multidisciplinary treatment. Cleft Palate Craniofac J. 1999;36(4):345–352. doi: 10.1597/1545-1569_1999_036_0345_evcscm_2.3.co_2. [DOI] [PubMed] [Google Scholar]
- 19.Li Q, Zhang F, Li X, Chen F. Genome scan for locus involved in mandibular prognathism in pedigrees from China. PLoS One. 2010;5(9) doi: 10.1371/journal.pone.0012678. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Zhang H, Takeda H, Tsuji T, Kamiya N, Rajderkar S, Louie K, et al. Generation of Evc2/Limbin global and conditional KO mice and its roles during mineralized tissue formation. Genesis. 2015 doi: 10.1002/dvg.22879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Mochida Y, Parisuthiman D, Kaku M, Hanai J, Sukhatme VP, Yamauchi M. Nephrocan, a novel member of the small leucine-rich repeat protein family, is an inhibitor of transforming growth factor-beta signaling. J Biol Chem. 2006;281(47):36044–36051. doi: 10.1074/jbc.M604787200. [DOI] [PubMed] [Google Scholar]
- 22.Mochida Y, Kaku M, Yoshida K, Katafuchi M, Atsawasuwan P, Yamauchi M. Podocan-like protein: a novel small leucine-rich repeat matrix protein in bone. Biochem Biophys Res Commun. 2011;410(2):333–338. doi: 10.1016/j.bbrc.2011.05.150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Kamiya N, Ye L, Kobayashi T, Mochida Y, Yamauchi M, Kronenberg HM, et al. BMP signaling negatively regulates bone mass through sclerostin by inhibiting the canonical Wnt pathway. Development. 2008;135(22):3801–3811. doi: 10.1242/dev.025825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kronenberg HM. Developmental regulation of the growth plate. Nature. 2003;423(6937):332–336. doi: 10.1038/nature01657. [DOI] [PubMed] [Google Scholar]
- 25.Maquat LE. Nonsense-mediated mRNA decay in mammals. J Cell Sci. 2005;118(Pt 9):1773–1776. doi: 10.1242/jcs.01701. [DOI] [PubMed] [Google Scholar]
- 26.Neu-Yilik G, Amthor B, Gehring NH, Bahri S, Paidassi H, Hentze MW, et al. Mechanism of escape from nonsense-mediated mRNA decay of human beta-globin transcripts with nonsense mutations in the first exon. RNA. 2011;17(5):843–854. doi: 10.1261/rna.2401811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Soeta S, Shimoura H, Hatakeyama N, Kodaka T, Amasaki H, Yamano S, et al. Immunohistochemical examination of epiphyseal growth plates of Japanese Brown cattle with chondrodysplasia. Journal of comparative pathology. 2007;136(2–3):145–155. doi: 10.1016/j.jcpa.2007.01.008. [DOI] [PubMed] [Google Scholar]
- 28.Murgiano L, Jagannathan V, Benazzi C, Bolcato M, Brunetti B, Muscatello LV, et al. Deletion in the EVC2 gene causes chondrodysplastic dwarfism in Tyrolean Grey cattle. PLoS One. 2014;9(4):e94861. doi: 10.1371/journal.pone.0094861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Chai Y, Maxson RE., Jr Recent advances in craniofacial morphogenesis. Developmental dynamics: an official publication of the American Association of Anatomists. 2006;235(9):2353–2375. doi: 10.1002/dvdy.20833. [DOI] [PubMed] [Google Scholar]
- 30.Snider TN, Mishina Y. Cranial neural crest cell contribution to craniofacial formation, pathology, and future directions in tissue engineering. Birth defects research Part C, Embryo today: reviews. 2014;102(3):324–332. doi: 10.1002/bdrc.21075. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
IHC staining using anti-β-gal antibody was performed to visualize the Evc2-expressing cells in the craniofacial structures of Het Evc2 KO embryos at E15.5 (brown) (A–L). Sagittal sections of Het heads at E15.5 were used (A, H) and the area of nasal capsule (Na), palate (Pa), nasal septum (NS), Meckel’s cartilage (Me), and paranasal sinus (PS) was indicated by an open box, respectively. Expression of β-gal was detected in chondrocytes (indicated by filled arrows) of Na (C), NS (G), Me (J) and PS (L) as well as in the mesenchymal cells of Pa (E, indicated by arrowheads) and lining epithelial cells (L, indicated by open arrows). Serial sections of the Het heads were also stained with non-immune Ig as a negative control and no immunoreactivity in the corresponding craniofacial areas was observed (Na in (B), Pa in (D), NS in (F), Me in (I) and PS in (K), respectively). Sections were counterstained with hematoxylin (blue). Scale bars; A, H = 1 mm, B–G, I–L = 0.05 mm. At least 3 head samples have been investigated using at least 6 sections per sample and the representative images were acquired and presented.
Sagittal sections of Het and KO heads at E16.5 were prepared. Fluorescence IHC staining with anti-β-gal (left panels, red) and anti-SOX9 (middle panels, green) antibodies was performed followed by incubating with secondary Alexa fluor 594 and 488 antibodies using Het (upper panels) and KO (lower panels) embryos. Merged images were shown on the right panels. Sections were counterstained with DAPI. Scale bars; 0.04 mm.