Abstract
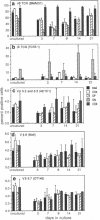
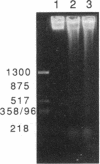
A recently described organ culture system for human thymocytes is shown to support the generation of a diverse T-cell receptor repertoire in vitro: thymocytes of the alpha beta lineage, including representatives of the V beta families 5.2/5.3, 6.7, and 8, accounted for the majority of T-cell receptor-positive cells throughout a 3-week culture period. Thymocytes bearing gamma delta receptors were also identified, particularly among the CD4 CD8 double-negative subset. The T-cell receptor repertoire expressed in organ culture responded to experimental manipulation with staphylococcal enterotoxins. Staphylococcal enterotoxin D (a powerful activator of human peripheral T cells expressing V beta 5.2/5.3 receptors) caused a marked reduction of V beta 5.2/5.3 expression, as determined with the V beta-specific antibody 42/1C1. Evidence is presented that this loss of V beta 5.2/5.3 expression resulted from the selective deletion of activated thymocytes by apoptosis, in concert with T-cell receptor modulation. These effects of staphylococcal enterotoxin D were specific (since staphylococcal enterotoxin E did not influence V beta 5.2/5.3 expression) and V beta-selective (since expression of V beta 6.7 remained unaffected by staphylococcal enterotoxin D). On the basis of these observations, we suggest that thymic organ culture provides a powerful approach to study the generation of the human T-cell repertoire.
Full text
PDF

Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Band H., Hochstenbach F., McLean J., Hata S., Krangel M. S., Brenner M. B. Immunochemical proof that a novel rearranging gene encodes the T cell receptor delta subunit. Science. 1987 Oct 30;238(4827):682–684. doi: 10.1126/science.3672118. [DOI] [PubMed] [Google Scholar]
- Ben-Nun A., Liblau R. S., Cohen L., Lehmann D., Tournier-Lasserve E., Rosenzweig A., Zhang J. W., Raus J. C., Bach M. A. Restricted T-cell receptor V beta gene usage by myelin basic protein-specific T-cell clones in multiple sclerosis: predominant genes vary in individuals. Proc Natl Acad Sci U S A. 1991 Mar 15;88(6):2466–2470. doi: 10.1073/pnas.88.6.2466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benoist C., Mathis D. Positive and negative selection of the T cell repertoire in MHC class II transgenic mice. Semin Immunol. 1989 Nov;1(2):117–124. [PubMed] [Google Scholar]
- Beverley P. C., Callard R. E. Distinctive functional characteristics of human "T" lymphocytes defined by E rosetting or a monoclonal anti-T cell antibody. Eur J Immunol. 1981 Apr;11(4):329–334. doi: 10.1002/eji.1830110412. [DOI] [PubMed] [Google Scholar]
- Born W., McDuffie M., Roehm N., Kushnir E., White J., Thorpe D., Stefano J., Kappler J., Marrack P. Expression and role of the T cell receptor in early thymocyte differentiation in vitro. J Immunol. 1987 Feb 15;138(4):999–1008. [PubMed] [Google Scholar]
- Boylston A. W., Borst J., Yssel H., Blanchard D., Spits H., de Vries J. E. Properties of a panel of monoclonal antibodies which react with the human T cell antigen receptor on the leukemic line HPB-ALL and a subset of normal peripheral blood T lymphocytes. J Immunol. 1986 Jul 15;137(2):741–744. [PubMed] [Google Scholar]
- Carrel S., Giuffrè L., Vacca A., Salvi S., Mach J. P., Isler P. Monoclonal antibodies against idiotypic determinant(s) of the T cell receptor from HPB-ALL cells induce IL2 production in Jurkat cells without apparent evidence of binding. Eur J Immunol. 1986 Jul;16(7):823–828. doi: 10.1002/eji.1830160717. [DOI] [PubMed] [Google Scholar]
- Choi Y. W., Kotzin B., Herron L., Callahan J., Marrack P., Kappler J. Interaction of Staphylococcus aureus toxin "superantigens" with human T cells. Proc Natl Acad Sci U S A. 1989 Nov;86(22):8941–8945. doi: 10.1073/pnas.86.22.8941. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fisher A. G., Larsson L., Goff L. K., Restall D. E., Happerfield L., Merkenschlager M. Human thymocyte development in mouse organ cultures. Int Immunol. 1990;2(6):571–578. doi: 10.1093/intimm/2.6.571. [DOI] [PubMed] [Google Scholar]
- Herman A., Croteau G., Sekaly R. P., Kappler J., Marrack P. HLA-DR alleles differ in their ability to present staphylococcal enterotoxins to T cells. J Exp Med. 1990 Sep 1;172(3):709–717. doi: 10.1084/jem.172.3.709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Herman A., Kappler J. W., Marrack P., Pullen A. M. Superantigens: mechanism of T-cell stimulation and role in immune responses. Annu Rev Immunol. 1991;9:745–772. doi: 10.1146/annurev.iy.09.040191.003525. [DOI] [PubMed] [Google Scholar]
- Hori T., Cupp J., Wrighton N., Lee F., Spits H. Identification of a novel human thymocyte subset with a phenotype of CD3- CD4+ CD8 alpha + beta-1. Possible progeny of the CD3- CD4- CD8- subset. J Immunol. 1991 Jun 15;146(12):4078–4084. [PubMed] [Google Scholar]
- Jenkinson E. J., Franchi L. L., Kingston R., Owen J. J. Effect of deoxyguanosine on lymphopoiesis in the developing thymus rudiment in vitro: application in the production of chimeric thymus rudiments. Eur J Immunol. 1982 Jul;12(7):583–587. doi: 10.1002/eji.1830120710. [DOI] [PubMed] [Google Scholar]
- Jenkinson E. J., Kingston R., Smith C. A., Williams G. T., Owen J. J. Antigen-induced apoptosis in developing T cells: a mechanism for negative selection of the T cell receptor repertoire. Eur J Immunol. 1989 Nov;19(11):2175–2177. doi: 10.1002/eji.1830191132. [DOI] [PubMed] [Google Scholar]
- Kappler J. W., Wade T., White J., Kushnir E., Blackman M., Bill J., Roehm N., Marrack P. A T cell receptor V beta segment that imparts reactivity to a class II major histocompatibility complex product. Cell. 1987 Apr 24;49(2):263–271. doi: 10.1016/0092-8674(87)90567-8. [DOI] [PubMed] [Google Scholar]
- Kappler J., Kotzin B., Herron L., Gelfand E. W., Bigler R. D., Boylston A., Carrel S., Posnett D. N., Choi Y., Marrack P. V beta-specific stimulation of human T cells by staphylococcal toxins. Science. 1989 May 19;244(4906):811–813. doi: 10.1126/science.2524876. [DOI] [PubMed] [Google Scholar]
- Kruisbeek A. M., Hodes R. J., Singer A. Cytotoxic T lymphocyte responses by chimeric thymocytes. Self-recognition is determined early in T cell development. J Exp Med. 1981 Jan 1;153(1):13–29. doi: 10.1084/jem.153.1.13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kurrle R., Lang W., Kanzy E. J., Seiler F. R. Distinction of two CD3-monoclonal antibodies in respect to the effectivity to modulate the induction of IgM- and gamma-interferon synthesis. Behring Inst Mitt. 1986 Jun;(80):48–58. [PubMed] [Google Scholar]
- MacDonald H. R., Howe R. C., Pedrazzini T., Lees R. K., Budd R. C., Schneider R., Liao N. S., Zinkernagel R. M., Louis J. A., Raulet D. H. T-cell lineages, repertoire selection and tolerance induction. Immunol Rev. 1988 Aug;104:157–182. doi: 10.1111/j.1600-065x.1988.tb00762.x. [DOI] [PubMed] [Google Scholar]
- MacDonald H. R., Schneider R., Lees R. K., Howe R. C., Acha-Orbea H., Festenstein H., Zinkernagel R. M., Hengartner H. T-cell receptor V beta use predicts reactivity and tolerance to Mlsa-encoded antigens. Nature. 1988 Mar 3;332(6159):40–45. doi: 10.1038/332040a0. [DOI] [PubMed] [Google Scholar]
- Merkenschlager M., Fisher A. G. CD45 isoform switching precedes the activation-driven death of human thymocytes by apoptosis. Int Immunol. 1991 Jan;3(1):1–7. doi: 10.1093/intimm/3.1.1. [DOI] [PubMed] [Google Scholar]
- Merkenschlager M., Fisher A. G. Human postnatal thymocytes generate phenotypically immature CD3dim, CD5dim, CD1abright progeny in organ culture. J Immunol. 1992 Feb 15;148(4):1012–1015. [PubMed] [Google Scholar]
- Merkenschlager M., Ikeda H., Wilkinson D., Beverly P. C., Trowsdale J., Fisher A. G., Altmann D. M. Allorecognition of HLA-DR and -DQ transfectants by human CD45RA and CD45R0 CD4 T cells: repertoire analysis and activation requirements. Eur J Immunol. 1991 Jan;21(1):79–88. doi: 10.1002/eji.1830210113. [DOI] [PubMed] [Google Scholar]
- Murphy K. M., Heimberger A. B., Loh D. Y. Induction by antigen of intrathymic apoptosis of CD4+CD8+TCRlo thymocytes in vivo. Science. 1990 Dec 21;250(4988):1720–1723. doi: 10.1126/science.2125367. [DOI] [PubMed] [Google Scholar]
- Nakayama T., Singer A., Hsi E. D., Samelson L. E. Intrathymic signalling in immature CD4+CD8+ thymocytes results in tyrosine phosphorylation of the T-cell receptor zeta chain. Nature. 1989 Oct 19;341(6243):651–654. doi: 10.1038/341651a0. [DOI] [PubMed] [Google Scholar]
- O'Hehir R. E., Lamb J. R. Induction of specific clonal anergy in human T lymphocytes by Staphylococcus aureus enterotoxins. Proc Natl Acad Sci U S A. 1990 Nov;87(22):8884–8888. doi: 10.1073/pnas.87.22.8884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ohashi P. S., Pircher H., Bürki K., Zinkernagel R. M., Hengartner H. Distinct sequence of negative or positive selection implied by thymocyte T-cell receptor densities. Nature. 1990 Aug 30;346(6287):861–863. doi: 10.1038/346861a0. [DOI] [PubMed] [Google Scholar]
- Paliard X., West S. G., Lafferty J. A., Clements J. R., Kappler J. W., Marrack P., Kotzin B. L. Evidence for the effects of a superantigen in rheumatoid arthritis. Science. 1991 Jul 19;253(5017):325–329. doi: 10.1126/science.1857971. [DOI] [PubMed] [Google Scholar]
- Posnett D. N., Wang C. Y., Friedman S. M. Inherited polymorphism of the human T-cell antigen receptor detected by a monoclonal antibody. Proc Natl Acad Sci U S A. 1986 Oct;83(20):7888–7892. doi: 10.1073/pnas.83.20.7888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schönrich G., Kalinke U., Momburg F., Malissen M., Schmitt-Verhulst A. M., Malissen B., Hämmerling G. J., Arnold B. Down-regulation of T cell receptors on self-reactive T cells as a novel mechanism for extrathymic tolerance induction. Cell. 1991 Apr 19;65(2):293–304. doi: 10.1016/0092-8674(91)90163-s. [DOI] [PubMed] [Google Scholar]
- Scott S., Pandolfi F., Kurnick J. T. Fibroblasts mediate T cell survival: a proposed mechanism for retention of primed T cells. J Exp Med. 1990 Dec 1;172(6):1873–1876. doi: 10.1084/jem.172.6.1873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith C. A., Williams G. T., Kingston R., Jenkinson E. J., Owen J. J. Antibodies to CD3/T-cell receptor complex induce death by apoptosis in immature T cells in thymic cultures. Nature. 1989 Jan 12;337(6203):181–184. doi: 10.1038/337181a0. [DOI] [PubMed] [Google Scholar]
- White J., Herman A., Pullen A. M., Kubo R., Kappler J. W., Marrack P. The V beta-specific superantigen staphylococcal enterotoxin B: stimulation of mature T cells and clonal deletion in neonatal mice. Cell. 1989 Jan 13;56(1):27–35. doi: 10.1016/0092-8674(89)90980-x. [DOI] [PubMed] [Google Scholar]
- Wraith D. C., McDevitt H. O., Steinman L., Acha-Orbea H. T cell recognition as the target for immune intervention in autoimmune disease. Cell. 1989 Jun 2;57(5):709–715. doi: 10.1016/0092-8674(89)90786-1. [DOI] [PubMed] [Google Scholar]
- Wucherpfennig K. W., Ota K., Endo N., Seidman J. G., Rosenzweig A., Weiner H. L., Hafler D. A. Shared human T cell receptor V beta usage to immunodominant regions of myelin basic protein. Science. 1990 May 25;248(4958):1016–1019. doi: 10.1126/science.1693015. [DOI] [PubMed] [Google Scholar]
- Wyllie A. H., Morris R. G., Smith A. L., Dunlop D. Chromatin cleavage in apoptosis: association with condensed chromatin morphology and dependence on macromolecular synthesis. J Pathol. 1984 Jan;142(1):67–77. doi: 10.1002/path.1711420112. [DOI] [PubMed] [Google Scholar]
- Zanders E. D., Lamb J. R., Feldmann M., Green N., Beverley P. C. Tolerance of T-cell clones is associated with membrane antigen changes. Nature. 1983 Jun 16;303(5918):625–627. doi: 10.1038/303625a0. [DOI] [PubMed] [Google Scholar]
- von Boehmer H. Developmental biology of T cells in T cell-receptor transgenic mice. Annu Rev Immunol. 1990;8:531–556. doi: 10.1146/annurev.iy.08.040190.002531. [DOI] [PubMed] [Google Scholar]