Pachymeningeal enhancement and brain sagging are recognized neuroimaging manifestations of craniospinal hypovolemia (CSH).1 Typically, CSF leaks from a dural defect, pools in the epidural space, and is absorbed into epidural veins. It is subsequently seen as early radiotracer activity on cisternography or as contrast in the renal pelvis on CT myelography. Not infrequently, despite extensive investigations, a dural defect is not identified in patients with CSH.2 Many of these patients undergo repeated single or multilevel blood patches with limited benefit.
We report 3 patients with CSH in whom the etiology of CSF leak was eventually identified to be a spinal CSF venous fistula (SCVF). In these cases, the CSF does not leak through a dural defect but drains from the subarachnoid space into the venous system due to a fistulous communication.
Case reports.
Case 1.
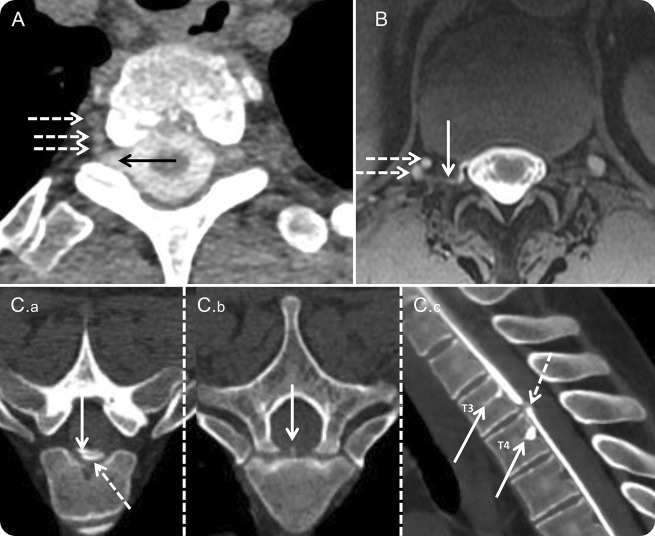
A 58-year-old man with a long-standing history of orthostatic headaches and neuroimaging evidence of CSH was evaluated for a 2-year history of progressive imbalance, cognitive difficulties, hypersomnolence, and severe dysphagia. Despite 2 cisternograms, 2 CT myelograms, and 1 positive-pressure intrathecal gadolinium magnetic resonance (MR) myelogram, the site of CSF leak was not identified. Five epidural blood patches, including 2 multilevel patches with blood and fibrin glue, did not result in an enduring benefit. An additional positive-pressure dynamic CT myelogram (figure, A) performed under anesthesia with intrathecal infusion of preservative-free saline showed a direct communication between the right T2-3 nerve root sleeve and adjacent paravertebral veins. At surgery, the right T2-3 nerve root was ligated. Dilated venous channels were noted along the course of the nerve root. At 2 months follow-up there was significant clinical improvement and normalization of neuroimaging evidence of CSH.
Figure. Imaging in spinal CSF venous fistula.
(A) Axial CT myelography image in case 1 shows a direct communication (arrow) between the right T2-3 nerve root sleeve and paravertebral veins (dotted arrows). (B) Axial intrathecal gadolinium-enhanced T1 magnetic resonance myelography image in case 2 shows contrast in the right T12-L1 foramen (arrow) draining into paravertebral veins (dotted arrows). (C) Axial (C.a. and C.b) CT myelography images in case 3 show intrathecal contrast (C.a: arrow) and contrast extravasation (C.a: dotted arrow) at T3-4. The dural defect and initial site of contrast extravasation are at the site of a spiculated osteophyte (C.b: arrow). Also shown is contrast entering the basivertebral veins at T3 and T4 (C.c: arrows). The dotted arrow in C.c shows the dural defect.
Case 2.
A 70-year-old man with an 8-year history of cough headaches and brain MRI evidence of CSH was evaluated with a CT myelogram that showed a CSF leak at the right T12-L1 level. It was mistakenly presumed that this was through a dural defect. Over the preceding year he had received 9 blood patches (5 of these were with fibrin glue, 3 were directed at the right T12-L1 level, and 3 were multilevel). With each intervention the benefit was definite but short-lived. A positive-pressure intrathecal gadolinium MR myelogram showed curvilinear contrast in the right T12-L1 foramen that drained into an adjacent paravertebral vein (figure, B). Contrast did not persist on delayed imaging, suggesting the presence of a SCVF. At surgery, the right T12 nerve root was amputated and the venous complex in the foramen was obliterated. Postoperative follow-up at 10 weeks showed resolution of symptoms and normalization of his brain MRI.
Case 3.
A 46-year-old man was seen for a 12-year history of exertion-induced spells that were characterized by nausea, vomiting, imbalance, sense of ear fullness, and altered sensorium. Two years prior to his evaluation, he was noted to have neuroimaging and CSF evidence of superficial siderosis. A CT myelogram had identified a defect at T3-4 but 2 directed blood patches did not result in an enduring response. A CT myelogram done at our institution identified a CSF leak at T3-4 (figure, C.a) (adjacent to a small central spiculated osteophyte—figure, C.b). Contrast was then noted to promptly leak into the basivertebral veins at T3 and T4 (figure, C.c), suggesting the presence of a fistulous communication. At surgery, the dural defect was repaired and the spiculated osteophyte excised.
Discussion.
Immunohistochemical studies have conclusively shown that spinal arachnoid villi exist and are morphologically associated with radicular veins.3 They may be the sites of CSF resorption in neonates prior to the development of intracranial arachnoid granulations and likely have an active role in CSF resorption in adults. In some cases, they may predispose to the development of SCVFs. Intravascular Pantopaque infusion during myelography is likely due to SCVFs.4 Iatrogenic SCVFs may develop following myelography.5 Digital subtraction myelography may show SCVFs in patients with CSH.6 Dural defects commonly present as craniospinal hypovolemia or superficial siderosis (as in case 3).1 Not infrequently, a CSF leak through a dural defect is not identified in CSH.2
Our cases suggest that in some cases of CSH where the source of leak is not identified (cases 1 and 2), a SCVF is likely causative. Furthermore, case 3 suggests that a CSF leak through a dural defect may predispose to the formation of a SCVF. This may result from a communication between spinal arachnoid granulations and dilated perispinal vascular channels that frequently accompany CSH. These lesions require innovative imaging including positive-pressure CT myelography, often with a dynamic or ultrafast component, at times under general anesthesia.7 Additional investigations may include intrathecal gadolinium MR myelography and digital subtraction myelography.8 The prevalence of SCVF in the CSH population and the optimal imaging strategy are yet to be defined. These cases did not show an enduring response to targeted blood patches. Treatment options include sacrificing a nonappendicular nerve root or venous communication obliteration. The latter would be the preferred option with appendicular nerve roots that cannot be sacrificed without resulting deficits.
Footnotes
Author contributions: Dr. Kumar contributed to design and conceptualization of the study and to drafting and revising the manuscript. Dr. Diehn contributed to analysis and interpretation of data and to revising the manuscript. Dr. Carr contributed to analysis and interpretation of data and to revising the manuscript. Dr. Verdoorn contributed to analysis and interpretation of data and to revising the manuscript. Dr. Garza contributed to revising the manuscript and to study supervision. Dr. Luetmer contributed to analysis and interpretation of data and to revising the manuscript. Dr. Atkinson contributed to analysis and interpretation of data, revising the manuscript, and study supervision. Dr. Morris contributed to design and conceptualization of the study, analysis and interpretation of data, revising the manuscript, and study supervision.
Study funding: No targeted funding reported.
Disclosure: The authors report no disclosures relevant to the manuscript. Go to Neurology.org for full disclosures.
References
- 1.Kumar N. Beyond superficial siderosis: introducing “duropathies.” Neurology 2012;78:1992–1999. [DOI] [PubMed] [Google Scholar]
- 2.Wicklund MR, Mokri B, Drubach DA, Boeve BF, Parisi JE, Josephs KA. Frontotemporal brain sagging syndrome: an SIH-like presentation mimicking FTD. Neurology 2011;76:1377–1382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Tubbs RS, Hansasuta A, Stetler W, et al. Human spinal arachnoid villi revisited: immunohistological study and review of the literature. J Neurosurg Spine 2007;7:328–331. [DOI] [PubMed] [Google Scholar]
- 4.Lin PM, Clarke J. Spinal fluid-venous fistula: a mechanism for intravascular pantopaque infusion during myelography: report of two cases. J Neurosurg 1974;41:773–776. [DOI] [PubMed] [Google Scholar]
- 5.Maillot C. The space surrounding the spinal cord: constitution, organization and relationship with the cerebrospinal fluid. J Radiol 1990;71:539–547. [PubMed] [Google Scholar]
- 6.Schievink WI, Moser FG, Maya MM. CSF-venous fistula in spontaneous intracranial hypotension. Neurology 2014;83:472–473. [DOI] [PubMed] [Google Scholar]
- 7.Thielen KR, Sillery JC, Morris JM, et al. Ultrafast dynamic computed tomography myelography for the precise identification of high-flow cerebrospinal fluid leaks caused by spiculated spinal osteophytes. J Neurosurg Spine 2015;22:324–331. [DOI] [PubMed] [Google Scholar]
- 8.Akbar JJ, Luetmer PH, Schwartz KM, Hunt CH, Diehn FE, Eckel LJ. The role of MR myelography with intrathecal gadolinium in localizaton of spinal CSF leaks in patients with spontaneous intracranial hypotension. AJNR Am J Neuroradiol 2012;33:535–540. [DOI] [PMC free article] [PubMed] [Google Scholar]