Abstract
Study Objectives:
To investigate behavioral aspects and quality of life in children and adolescents with type 1 narcolepsy (NT1).
Methods:
We performed a case-control study comparing 29 patients with NT1 versus sex- and age-matched patients with idiopathic epilepsy (n = 39) and healthy controls (n = 39). Behavior and quality of life were evaluated by self-administered questionnaires (Child Behavior Checklist, Pediatric Quality of Life Inventory). Patient groups were contrasted and scale results were correlated with clinical and polysomnographic parameters, and cerebrospinal fluid hypocretin-1 levels.
Results:
Young patients with NT1 showed increased internalizing problems associated with aggressive behavior. Emotional profile in patients with NT1 positively correlated with age at onset, diagnostic delay, and subjective sleepiness, whereas treatment and disease duration were associated with fewer behavioral problems (attention problems, aggressive behavior, and attention deficit/hyperactivity disorder). Psychosocial health domains of pediatric NT1 were worse than in healthy controls, whereas the physical health domains were comparable.
Conclusions:
Young NT1 patients show a discrete pattern of altered behavioral, thought, and mood profile in comparison with healthy controls and with idiopathic epilepsy patients thus suggesting a direct link with sleepiness. Further studies investigating behavior in patients with idiopathic hypersomnia or type 2 narcolepsy are needed to disentangle the role of REM sleep dysfunction and hypocretin deficiency in psychiatric disorders. Symptoms of withdrawal, depression, somatic complaints, thought problems, and aggressiveness were common, NT1 children perceived lower school competencies than healthy children, and their parents also reported worse psychosocial health. Our data suggest that early effective treatment and disease self-awareness should be promoted in NT1 children for their positive effect on behavior and psychosocial health.
Citation:
Rocca FL, Finotti E, Pizza F, Ingravallo F, Gatta M, Bruni O, Plazzi G. Psychosocial profile and quality of life in children with type 1 narcolepsy: a case-control study. SLEEP 2016;39(7):1389–1398.
Keywords: attention deficit/hyperactivity disorder, narcolepsy with cataplexy, psychiatry, quality of life
Significance.
Psychosocial profile was tested for the first time in young patients with Narcolepsy type 1 (NT1) in comparison with patients with Idiopathic Epilepsy (IE) and healthy controls. NT1 patients showed more psychological and behavioral problems that IE and control subjects, with a hierarchical order for the externalizing aspects (NT1 > IE > controls) and a peculiar profile in the internalizing features (NT1 > IE = controls). The psychological and behavioral aspects negatively influenced quality of life in NT1 patients. Pharmacological treatment of NT1 was associated with a better profile in the attention, aggressiveness, hyperactivity, and oppositional defiant areas. Further longitudinal studies including psychological and pharmacological interventions are warranted. Improving the psychosocial impairment in NT1 young patients could contribute to prevent major psychiatric comorbidities.
INTRODUCTION
Type 1 narcolepsy (NT1) is a chronic central disorder of hypersomnolence characterized by excessive daytime sleepiness (EDS), cataplexy, sleep paralyses, hypnagogic hallucinations, and disrupted nocturnal sleep.1 NT1 is linked to hypothalamic hypocretin deficiency of possible autoimmune origin as suggested by the genetic predisposition of human leukocyte antigen (HLA) alleles combined with triggering environmental factors.1,2 NT1 greatly affects patients' quality of life (QoL) in a homogeneous way across countries, independently from cultural and geographic provenience.3
In children and adolescents, NT1 displays a peculiar phenotype that contributes to frequent misdiagnosis, and to the remarkable diagnostic delay.4–11 Young patients can manifest EDS with sleep attacks as well as with long periods of daytime and nighttime sleep, or with hyperactive behavior and irritability that may resemble neurodevelopmental disturbances.12,13 Young NT1 patients often experience a widespread impairment in their education and social relationships, and may suffer from neuropsychiatric disorders encompassing anxiety, depressive mood, psychotic symptoms, and attention deficit hyperactivity disorder (ADHD), as reported in adults.13–20 Both the large overlap between NT1 and psychological problems and the possible involvement of the hypocretinergic system in psychiatric disorders raises the question whether the multifaceted psychological picture of childhood NT1 has to be considered and treated as an intrinsic dysfunction, reactive to NT1, or as a discrete real psychiatric comorbidity.21,22
Psychiatric comorbidity in young NT1 was mostly described in case reports or anecdotally reported as intrinsic susceptibility.23–31 Few studies used standardized tests to investigate different areas of psychosocial functioning, including social behavior, emotional and conduct problems, depressive mood, competences and behavioral problems, ADHD, schizophrenia, autism symptoms, and poor QoL outcomes.13,32–37 However, most studies were performed in small and heterogeneous NT1 groups in parallel with other patient groups, or thorough uncontrolled designs including a mixture of patients with narcolepsy with and without cataplexy.13,32–37
The aim of this study was to investigate behavioral aspects and QoL in young NT1 patients versus age- and sex- matched nonsleepy patients with epilepsy and healthy controls, using the Child Behavior Checklist (CBCL) and the Pediatric Quality of Life Inventory (PedsQL).
METHODS
Patients
Twenty-nine children and adolescents (age range 7–16 y) with a diagnosis of NT1 were recruited at the Outpatient Clinic for Narcolepsy of the Department of Biomedical and Neuromotor Sciences of the University of Bologna.1 Results of both 48 h polysomnographic continuous recording and Multiple Sleep Latency Test (MSLT) were available for all patients, whereas cerebrospinal fluid hypocretin-1 (CSF HCRT-1) levels were available only for 17 of 29 patients due to the parents' refusal to perform lumbar puncture. In all patients, brain magnetic resonance imaging (MRI) was normal. Patients were HLA-DQB1*06:02 positive, excluding a girl with proved CSF HCRT-1 deficiency. At study inclusion, patients completed the Pediatric Daytime Sleepiness Scale (PDSS).38
The parallel patients group consisted of 39 age- and sex-matched children and adolescents (age range, 7–15 y) with idiopathic epilepsies (IE) according to international diagnostic criteria, followed at the Department of Woman and Child Health, University of Padua. Inclusion criteria required absence of sleep disorders complaints (including EDS) at clinical interview, and normal neurological examination and brain MRI.39
The healthy control group consisted of 39 age- and sex-matched healthy children and adolescents (age range, 6–18 y) without complaints related to sleep disorders (including EDS). The control group was recruited among students attending three local schools during the academic year. After an informative interview, parents had a letter describing the research project, providing details of the test methods, and requesting that they sign a form to authorize the participation in compliance with current legislation on privacy. The same questionnaires as those administered to the cases were anonymously administered in class during normal school hours.
The study was approved by the internal review board of all the institutes.
Questionnaires
NT1 patients and controls were investigated for both behavioral (CBCL) and QoL (PedsQL) aspects, whereas the IE group served as pathological control group to compare the disease effect on behavioral domains.
Child Behavior Checklist 6–18
The CBCL 6–18, completed by parents, addresses skills and problems in patients aged 6 to 18 y and provides a picture of their social, emotional, and behavioral profile, divided in two parts.40–42 The first refers to the Competence and adaptive scales (daily activities, social interaction, and school performance), and the second to emotional and behavioral problems with eight different Syndrome scales: five of the aforementioned scales are further grouped into higher internalizing (anxious/ depressed, depressed-withdrawn, and somatic complaints) and externalizing (rule-breaking, and aggressive behavior) scales; the Total Problems scale comprised all 113 items. The six Diagnostic Statistical Manual (DSM)-Oriented scales consistent with DSM diagnostic categories completed the CBCL 2001 version: affective disorder, anxiety disorder, somatic disorder, ADHD, oppositional defiant disorder (ODD), and conduct disorder (CD).43
Pediatric Quality of Life Inventory (PedsQL)
The PedsQL” 4.0, copyright 1998 JW Varni, PhD All rights reserved, Generic Core Scales, Italian version is composed of a patient self-report form and of a parent-proxy report form. PedsQL consists of 23 items addressing 4 core domains: physical, emotional, social, and school functioning.44,45 PedsQL is summarized into the following two measures: physical health summary score and psychosocial health summary score.
Statistical Analyses
Categorical and continuous data were reported as frequencies or mean (SD) for each group. CBCL raw scores were transformed into T-scores to allow comparisons. CBCL results have been contrasted between the three groups with Kruskall-Wallis analysis of variance followed by post hoc comparisons among groups with Mann-Whitney U test for continuous variables, and with chi-square tests for categorical variables. A subanalysis was also performed to compare treated versus untreated NT1 patients with Mann-Whitney U test.
Pearson correlation coefficient analyses were used to address the relations between CBCL and PedsQL scores in NT1 and control subjects, and between CBCL scores and clinical and polysomnographic data in NT1 and IE subjects.
A value of P ≤ 0.05 was considered statistically significant.
RESULTS
Participants' Characteristics
At study inclusion, NT1 patients (55% males) had a mean age of 11.52 (2.89) y (range 7–16). NT1 patients reported EDS and cataplexy (100%), hypnagogic hallucinations (45%), sleep paralyses (28%), disturbed nocturnal sleep (79%), automatic behaviors (45%), and relevant weight gain (79%) occurring closely after NT1 symptoms onset. The mean PDSS score was 15.24 (4.66). The mean age at onset of first symptom (either sleepiness or cataplexy) was 8.90 (2.35) y, and the mean disease duration was approximately 2.8 (2.95) y. The mean age at diagnosis was 10.45 (2.43) y, with a mean diagnostic delay of 1.55 (2.13) y. The mean number of schooling years was 6.66 (2.74).
MSLT showed a mean sleep latency of 3.02 (2.09) min with 4.41 (0.87) sleep onset rapid eye movement (REM) periods. Nocturnal polysomnography showed a mean total sleep time (TST) of 489.71 (72.64) min with a mean sleep latency of 5.89 (3.97) min, sleep efficiency of 87.54 (14.24) %, and a mean REM sleep latency of 39.96 (56.92) min. During daytime polysomnography, patients showed a mean number of naps of 2.64 (1.75) with a mean TST of 117.11 (85.47) min. The mean CSF HCRT-1 levels were 29.93 (28.91) pg/mL.
Twenty-one patients (72%) were undergoing treatment (modafinil 69%, sodium oxybate 28%, and venlafaxine 7%) whereas eight patients (28%) were drug-naïve.
The IE group (51% males) mean age was 10.64 (2.02) y. The mean age at IE onset was 5.31 (1.54) y, and the mean disease duration was 5.36 (2.51) y. Twenty IE patients (51%) had localization-related epilepsy and 19 (49%) idiopathic generalized epilepsy; 14 participants (36%) were receiving antiepileptic drug treatment, and 25 (64%) were drug-free. In the treated IE group of patients, 12 patients were under antiepileptic drug monotherapy (valproic acid, levetiracetam, carbamazepine, lamotrigine, clobazam), and 2 patients received an antiepileptic drug politherapy (valproic acid + ethosuximide; lamotrigine + ethosuximide, respectively).
The control group mean age was 11.21 (3.44) y; there were 20 males (51%) and 19 females (49%).
Behavior
Table 1 summarizes CBCL results as T-scores (mean, standard deviation) and as pathological findings in each group, expressed as the percentage of subject with pathological scores.
Table 1.
Child Behavioral Checklist T-scores and pathological scores (%) in type 1 narcolepsy patients, idiopathic epilepsy patients, and healthy control patients, with statistical comparison.
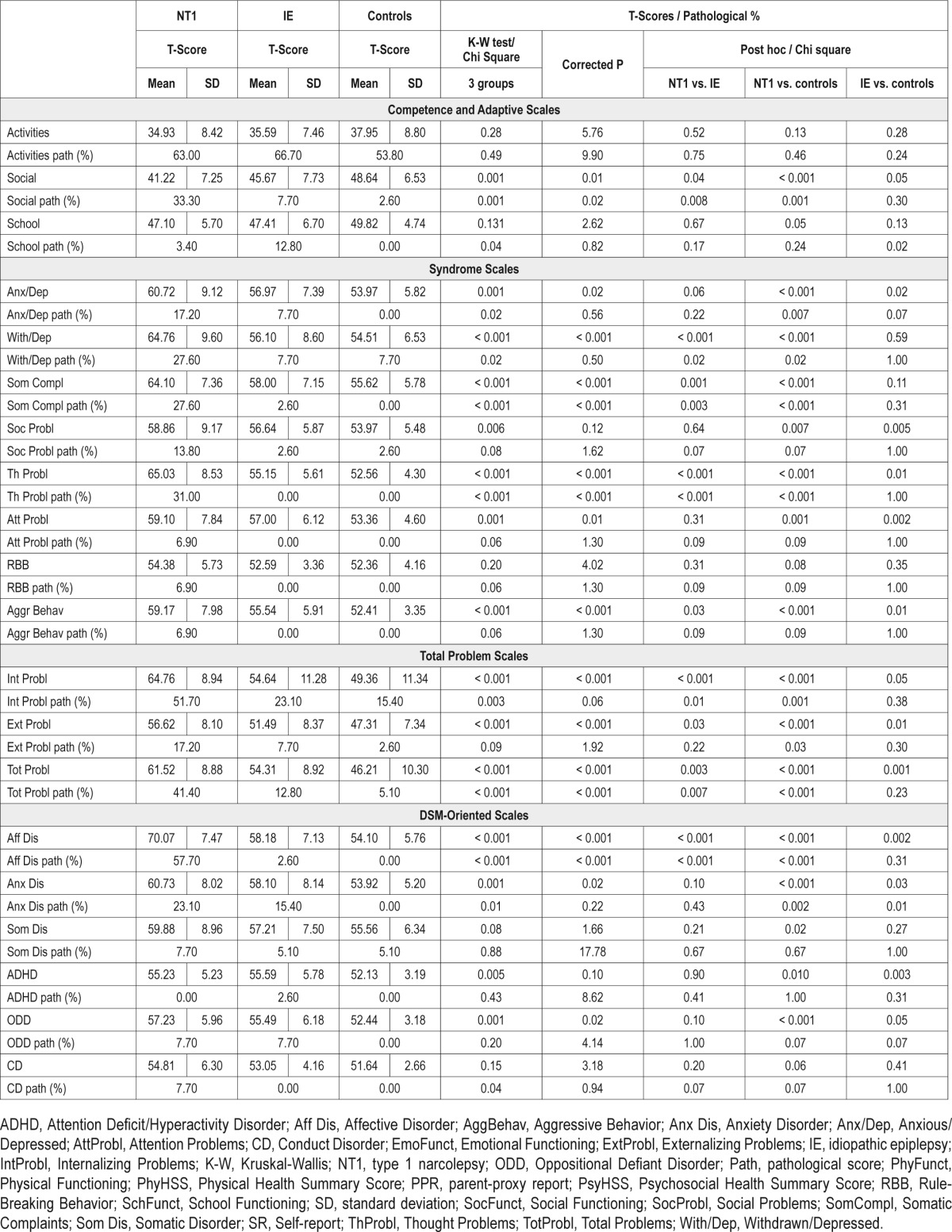
In Competence scales the three groups differed exclusively for social activities, with the NT1 patients having the lowest scores.
In withdrawn/depressed and somatic complaints areas, NT1 patients had the highest scores and they more frequently had pathological scores versus IE and controls.
Both NT1 and IE groups showed higher scores than the control group for anxious/depressed, social and attention problems, without significant differences between NT1 and IE groups. NT1 patients had pathological scores than controls exclusively in the anxious/depressed domain.
Thought and aggressive behavior scales showed the highest values in patients with NT1, intermediate in patients with IE, and lowest in healthy controls. Pathological values at thought scale were most prevalent among NT1 patients.
The three groups had comparable scores for rule-breaking behavior.
In total problem scales, the NT1 group showed higher internalizing and total problems scores as T-scores than the other two groups, and NT1 patients had more frequently pathological scores when compared with controls and IE patients. Finally, externalizing problems scores were highest in the NT1 group, intermediate in the IE group, and lowest in controls (Figure 1).
Figure 1.
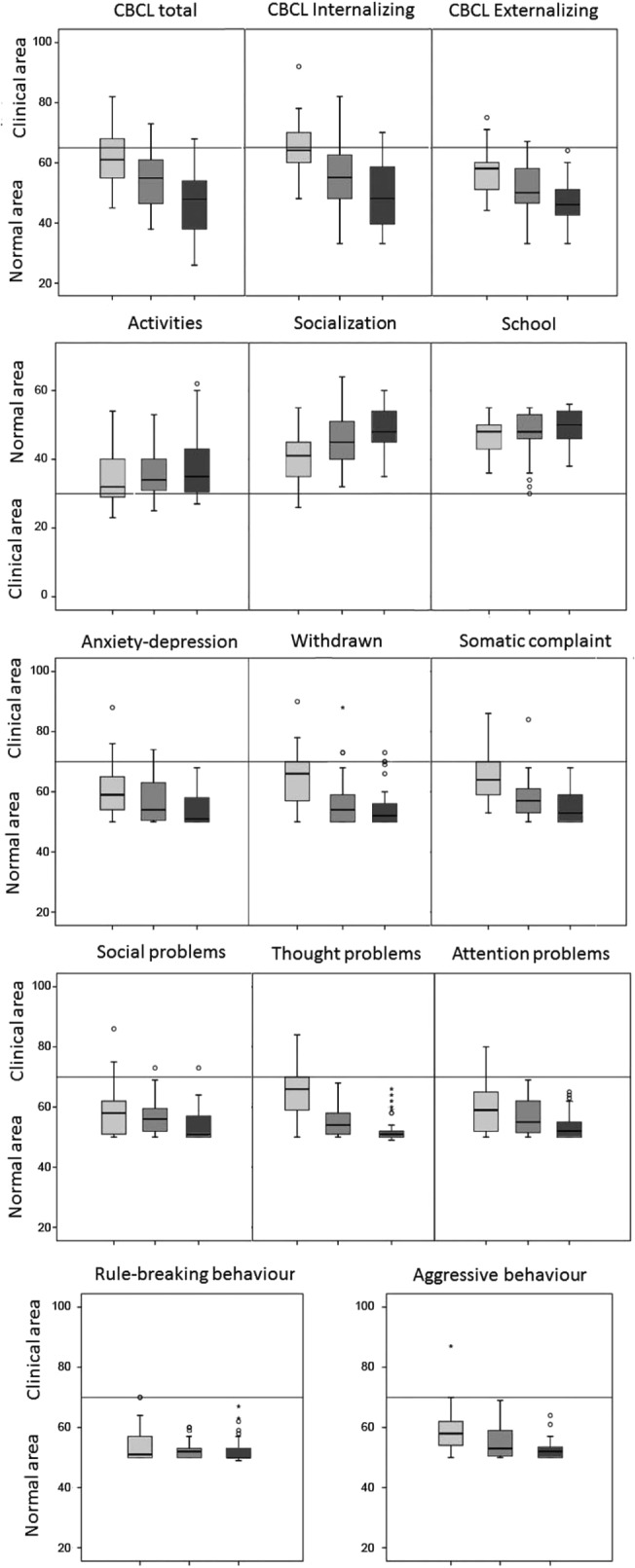
Distribution of T-scores in Syndrome scales. Distribution of T-scores for NT1 (gray box), IE patients (dark gray box), and controls (black box) in Syndrome scales. In the box plot graph, box's sides mark the first and third quartiles, the central line represents the median and the whiskers account for the maximum and minimum; outliers points lie outside the fences. The horizontal lines mark clinical/normal boundaries. CBCL, Child Behavior Checklist; IE, idiopathic epilepsies; NT1, narcolepsy type 1.
In DSM-oriented scales, the three groups differed for affective disorder, anxiety disorder, ADHD, ODD, and CD scores.
NT1 patients had significantly higher scores than IE patients who in turn differed from controls in the affective disorder scale, but only NT1 patients reached pathological score values at this scale.
Both the NT1 and IE groups showed higher scores in the anxiety disorder scale, and NT1 and IE patients had significant pathological scores compared to controls, without any difference between NT1 and IE.
ADHD and ODD scores were higher in both patients groups than in healthy controls, without differences in pathological scores. Neither somatic nor conduct disorder areas showed significant differences among groups (Figure 2).
Figure 2.
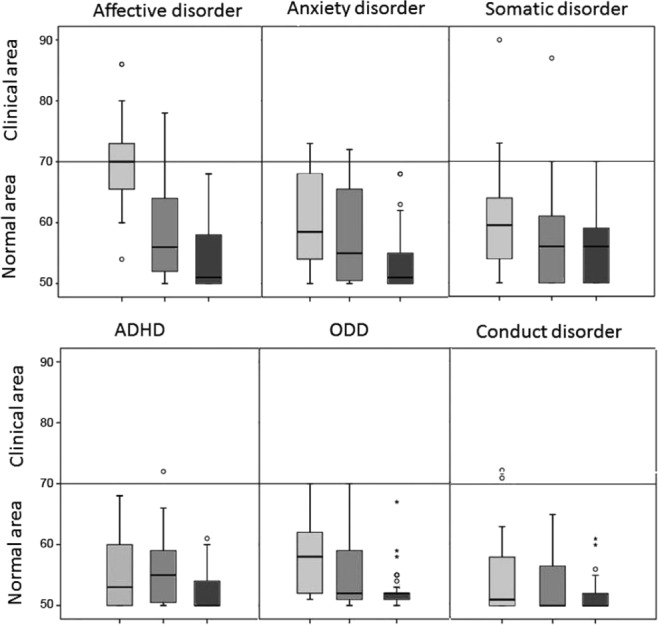
Distribution of T-scores in Diagnostic and Statistical Manual Oriented scales. Distribution of T-scores for NT1 (gray box), IE patients (dark gray box), and controls (black box) in Diagnostic and Statistical Manual Oriented scales. In the box plot graph, box's sides mark the first and third quartiles, the central line represents the median and the whiskers account for the maximum and minimum; outliers points lie outside the fences. The horizontal lines mark clinical/normal boundaries. ADHD, attention deficit/hyperactivity disorder; CBCL, Child Behavior Checklist; IE, idiopathic epilepsies; NT1, narcolepsy type 1; ODD, oppositional/defiant disorder.
Quality of Life
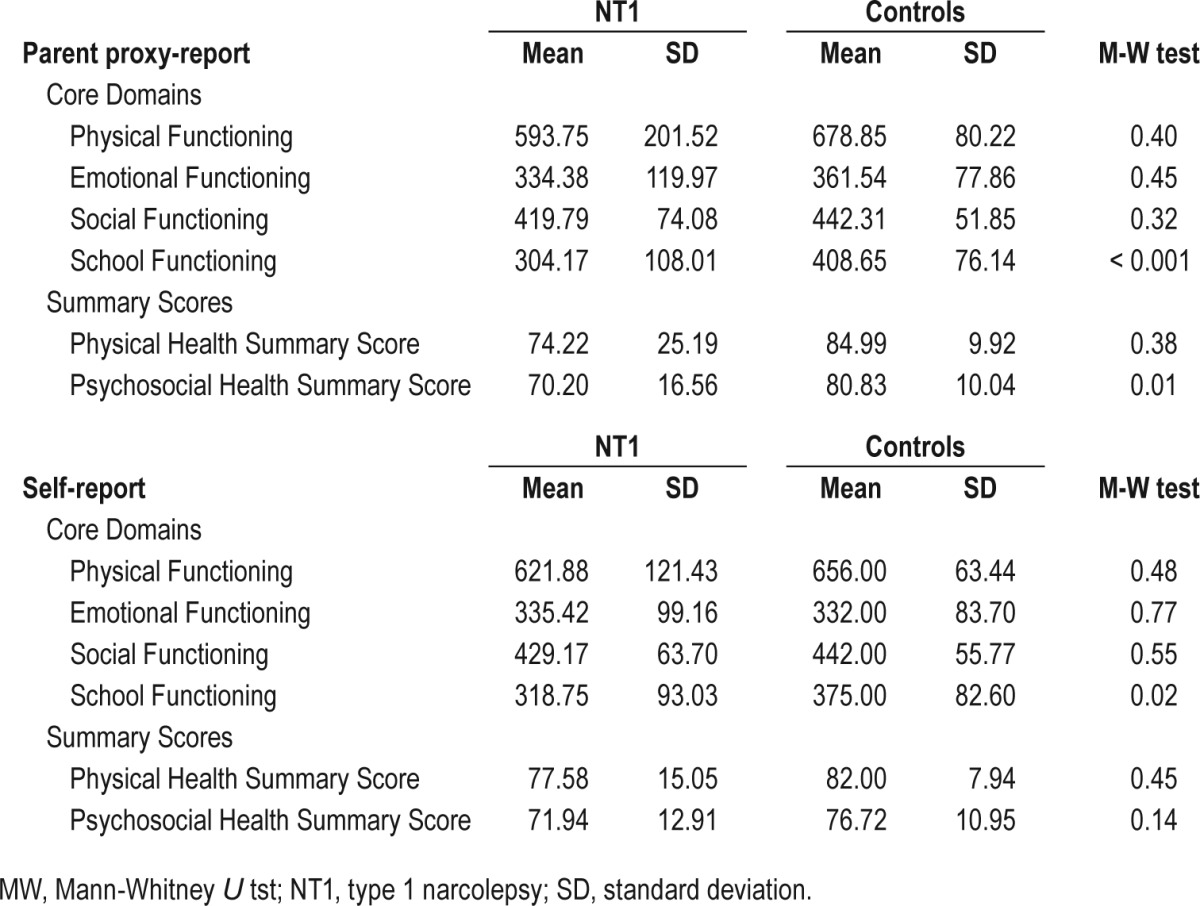
PedsQL results in NT1 patients and healthy controls are reported in Table 2. NT1 patients had physical, emotional and social subscales and physical health summary scores comparable to controls considering either self-reported or parent-proxy report. Conversely, NT1 patients had lower school functioning and psychosocial health summary scores than controls, the latter finding exclusively according to parent-proxy report.
Table 2.
Scale descriptives for Pediatric Quality of Life Inventory in children with type 1 narcolepsy self-report and parent proxy-report and comparisons with healthy children scores.
Correlations of CBCL Results
The correlational analyses among CBCL results, PedsQL, and clinical/demographic data are reported separately in the different groups in Table 3 and Table 4.
Table 3A.
Correlations between Child Behavioral Checklist scores and Pediatric Quality of Life Inventory scores in patients with type 1 narcolepsy.
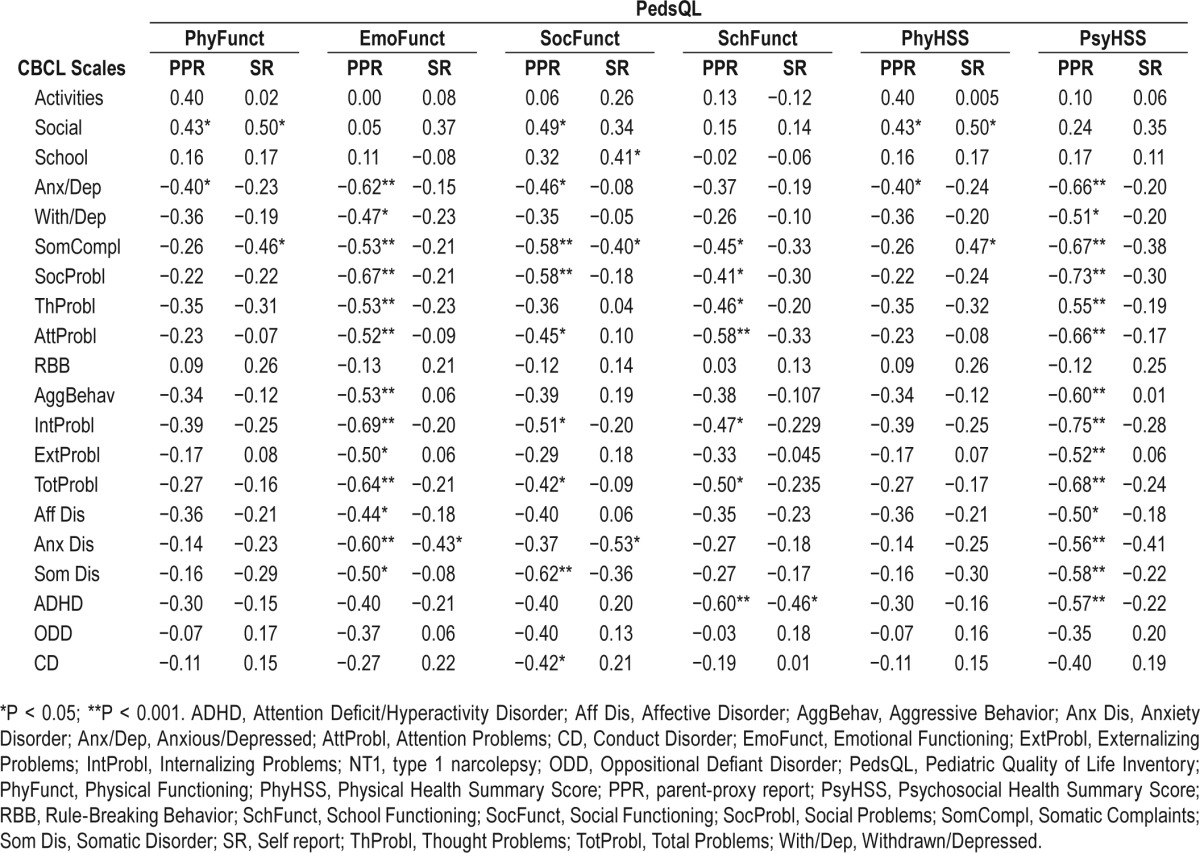
Table 4A.
Correlations between Child Behavioral Checklist scores and clinical correlates in patients with type 1 narcolepsy.
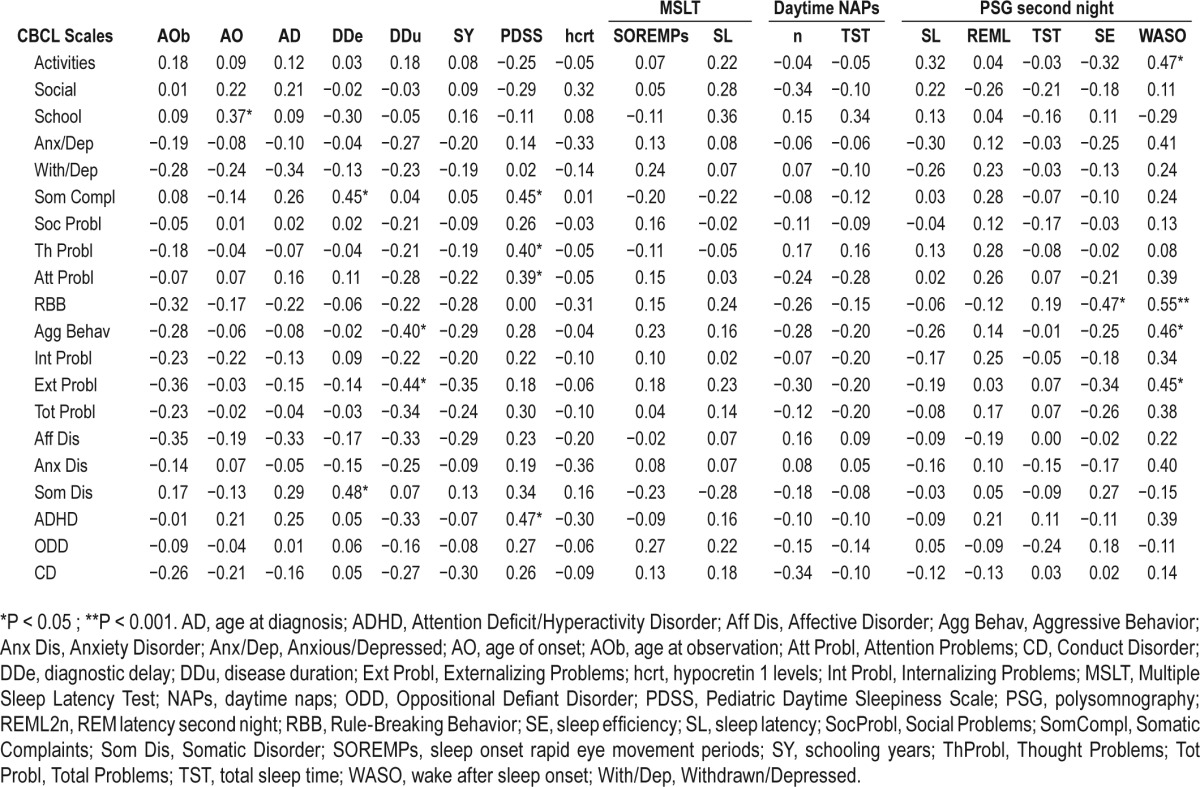
Table 3B.
Correlations between Child Behavioral Checklist scores and Pediatric Quality of Life Inventory scores in healthy control patients.
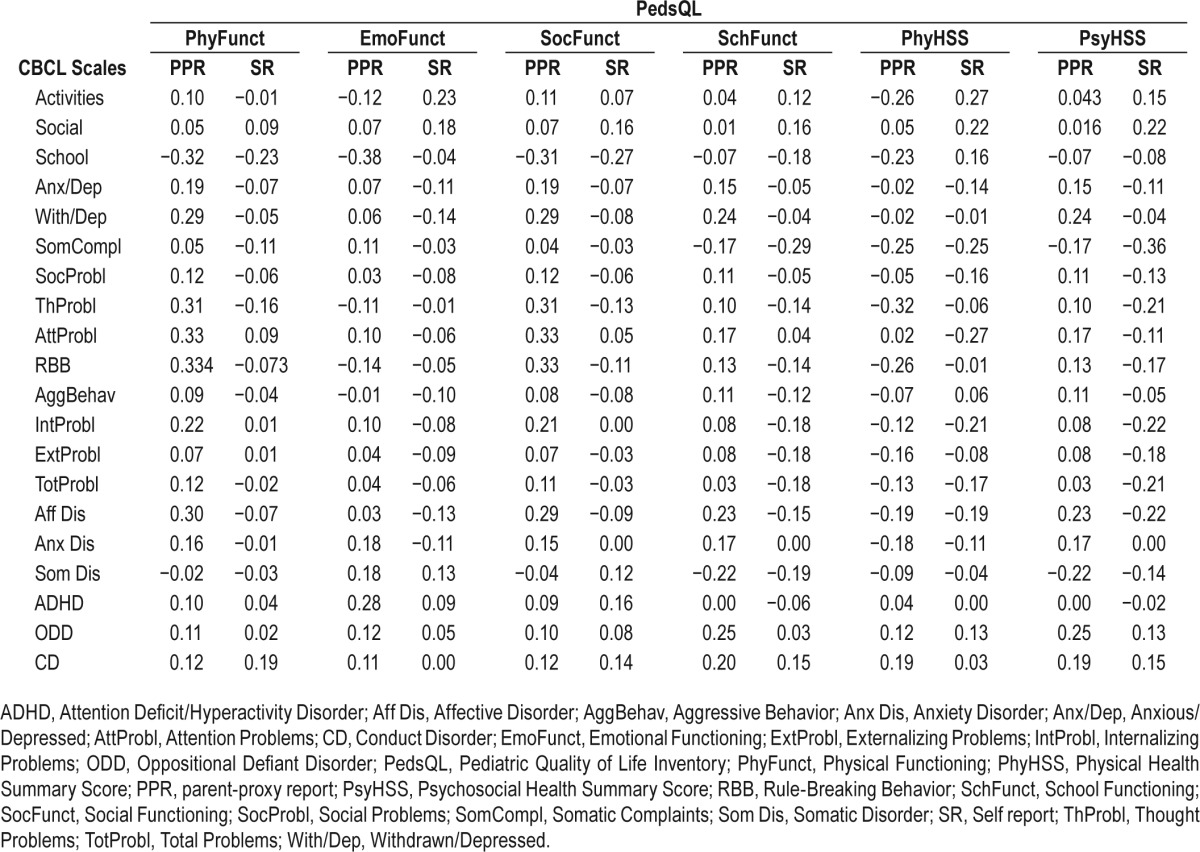
Table 4B.
Correlations of Child Behavioral Checklist scores and clinical correlates in patients with idiopathic epilepsy.
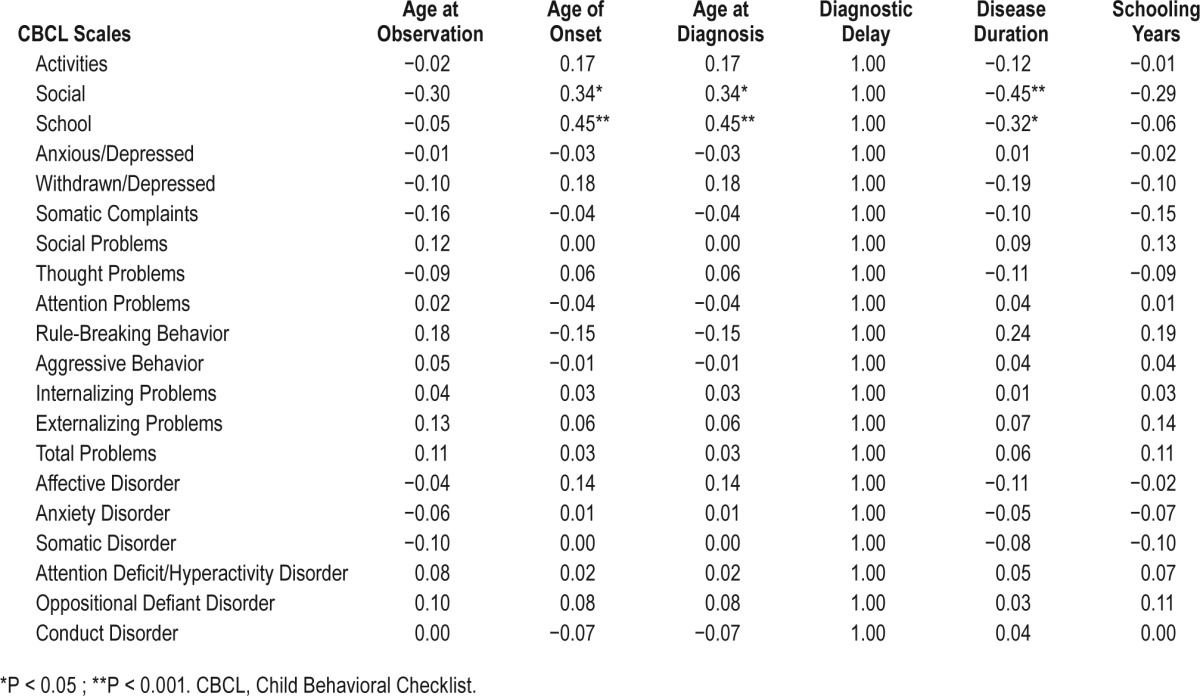
In NT1 patients, social scale score was positively correlated with physical (parent and self-report), and social functioning (parent), as well as with physical health summary score (parent and self report), whereas school scale score positively correlated only with self-reported social functioning.
No significant correlations were found in controls for the adaptive scales.
In NT1, physical functioning and health summary scores were inversely correlated with anxious/depressed and with somatic complaints according to parents and patients, respectively. Emotional functioning was inversely correlated with most of the CBCL syndromes and DSM-oriented scales according to parents, a cluster of findings confirmed by self-reported assessment only for the anxiety disorder scale.
Similarly, social and school functioning were inversely correlated with somatic, social, attention, internalizing, and total problem scores, with additional associations between social functioning, anxious/depressed, somatic, and CD as well as between school functioning, thought, and ADHD according to parent proxy-report. Patients' self-report confirmed the aforementioned associations only between social and school functioning versus somatic complaints and ADHD, but showed an inverse correlation also between social functioning and anxiety disorder. Overall, psychosocial health summary had a strong negative association with most CBCL results according to parents, a finding not mirrored by patients' self-reports.
In healthy controls, CBCL and PedsQL results were not significantly correlated.
In NT1 patients, a longer diagnostic delay was associated with more somatic complaints/disorder, whereas longer disease duration was associated with fewer aggressive and externalizing problems. EDS was positively correlated with somatic, thought, attention problems, and ADHD. Conversely, objective sleep propensity at the MSLT, CSF HCRT-1, and nocturnal sleep features did not show significant correlations, but a negative relation between sleep efficiency and rule-breaking behavior. In IE patients, CBCL results were not correlated with clinical features.
Effect of Pharmacological Treatment
Treated NT1 patients (Table S1 in the supplemental material) showed lower scores than untreated patients in attention problems, particularly when treated with sodium oxybate, and in ADHD scale. NT1 patients receiving sodium oxybate treatment had significantly higher school functioning than drug-naïve ones, and those treated with venlafaxine showed better scores in Aggressive scale and ODD. Modafinil therapy did not show significant associations, and no differences in CBCL profile were observed comparing treated and untreated IE patients (Table S2 in the supplemental material).
DISCUSSION
We documented that young patients with NT1 had more behavioral problems than IE patients and healthy controls: a higher incidence of anxious/depressed, withdrawn/depressed, social, thought, attention problems, somatic complaints, and aggressive behaviors have been recorded. However, we should highlight that NT1 patients' mean T scores fell within the normal ranges on each of these summary and syndrome scales, but for withdrawn/depressed behavior, somatic complaints, thought and internalizing problems. These specific psychological features can be useful to better manage NT1 at a young age: avoidance of situations that would precipitate cataplexy or draw attention to sleepiness is common in patients with narcolepsy, but extreme withdrawal associated with thought disorder should suggest a depressive or even a psychotic disorder.46
Previous reports disclosed a high prevalence of anxiety and depressive disorders in children with narcolepsy. Dorris et al.34 found that children with narcolepsy presented high scores in internalizing problems, especially anxious/depressed and withdrawn/depressed behavior on the CBCL, and Stores et al.32 reported that children with narcolepsy scored significantly higher than controls on the Child Depression Inventory. Szakács et al.35 reported that 20% of children with narcolepsy had a major depression, and 10% of them had a general anxiety disorder, while Inocente et al.33 described depressive feelings in up to 25% of a sample of 117 young patients with narcolepsy.
Several reports showed that adults with narcolepsy have a high rate of psychiatric symptoms and disorders compared to patients with symptomatic or idiopathic hypersomnia: mood disorders occur in up to 57% of patients with narcolepsy, although self-reported depressive symptoms are frequently insufficient to satisfy the diagnostic criteria for a major depressive disorder.17,47–49 Anxiety disorders, especially panic and social phobia, occur more commonly in NT1 patients than in the general population.17,19
Psychotic disorders have often been suspected in adult patients with narcolepsy, and the disease may also be misdiagnosed as a psychiatric disorder due to hallucinations.16,50–55 Although psychotic symptoms in narcolepsy may reflect a stimulant-induced psychosis, and in these situations the psychiatric symptoms resolve when medication dose is lowered, reports on adults and children with dual narcolepsy and schizophrenia are frequent.36,56–64 Finally, the presence of hypnagogic hallucinations seems positively related to the development of psychosis in vulnerable individuals.65 ADHD symptoms are more prevalent in adults with narcolepsy compared to healthy controls, and recent data confirmed this association in children with narcolepsy.13,20
In our study, QoL was worse in young patients with NT1 than in controls according to their parents, a finding partially confirmed by children with NT1. Previous studies investigating the effect of narcolepsy on QoL showed greater impairment in the mental health subscale versus controls, with no differences between children with narcolepsy or EDS, and that the domains energy/vitality and physical and psychological well-being were mostly impaired in young patients with narcolepsy.32,37
In contrast with a previous study, we found significant correlations between behavioral and QoL aspects, as well as with clinical characteristics: observation close to disease onset, diagnostic delay, and EDS (at PDSS) were correlated with behavior worsening, suggesting the importance of routinely assessing QoL and psychological features in young patients with NT1 for early detection of behavioral problems.32 These findings confirm the importance of both an early NT1 diagnosis and treatment in order to reduce the disease burden.11 Indeed, our data indicate that pharmacological treatment, that is off-label in the pediatric population, was associated with better outcomes in some CBCL scales: sodium oxybate improved attention, and venlafaxine reduced aggressive and oppositive-defiant behaviors. Although several studies have shown antiepileptic drugs effects on cognitive processes and behavior, we did not observe significant differences in CBCL scores between treated and untreated IE patients, probably due to the small number of patients involved.66 Conversely, our study disclosed statistically significant differences between treated and untreated NT1 patients thus suggesting a stronger effect of symptomatic treatment in NT1 versus IE patients.
Chronic disorders are associated with increased risk of psychiatric comorbidity with high levels of internalizing disorders disclosed by the CBCL: a recent meta-analysis showed that externalizing problems are both illness-specific and often associated with impaired brain function, as in epilepsy and in migraine/headache.67 Therefore, the psychopathological profile of childhood NT1 is similar to other chronic diseases for anxiety-depressive aspects, somatic complaints, and thought and attention problems, but it is peculiar for aspects of withdrawn/depression, social problems, and aggressive behavior. Further studies are needed to address the correlations among NT1 specific psychological traits, EDS, and emotions given the recently discovered role of hypocretinergic transmission during proactive behavior, and the intrinsic link between positive emotions and cataplexy.68,69
We acknowledge that our study has limitations. First, the cross-sectional design cannot demonstrate causation, but only compare findings in NT1 versus controls and IE patients. Second, the absence of sleep parameters in the IE and control group limits the comparison with those obtained in NT1 children to disentangle the potential relationship between behavioral and sleep patterns. Third, we used the parent version of CBCL, and parallel information from the children themselves and/or data from behavioral observation would strengthen the current findings. Acknowledging that the CBCL includes some items concerning sleep, we also calculated the results excluding ambiguous items without obtaining different findings (data not shown), thus confirming the high internal consistency of the CBCL itself. Finally, the CBCL is a screening tool that does not allow discrete psychiatric diagnosis.
CONCLUSIONS
Children suffering from NT1 had a high representation of internalizing problems, especially depressed-anxious, aspects of withdrawal, somatic complaints, and thought disorders that may be common in chronic diseases. Conversely, the presence of externalizing problems, such as attention problems and aggressive behavior, counted as anecdotal and confirmed in our study, should be considered as part of the spectrum of the NT1 behavioral phenotype.
Future interventional-longitudinal studies are needed to characterize the behavioral evolution, to assess the effect of risk factors for the development of psychiatric disorders, and to evaluate the effectiveness of NT1 pharmacological and/or psychological treatments.
DISCLOSURE STATEMENT
This was not an industry supported study. No funding was secured for this study. Dr. Plazzi has participated on advisory boards for UCB Pharma, Bioproject, and Jazz Pharmaceuticals. The other authors have indicated no financial conflicts of interest.
ACKNOWLEDGMENTS
The authors thank Dr. James Varni who kindly provided us the permission to use the PedsQL” (for information contact: Mapi Research Trust, Lyon, France. Email: PROinformation@mapi-trust.org – Internet: www.proqolid.org and www.pedsql.org).
REFERENCES
- 1.American Academy of Sleep Medicine. International Classification of Sleep Disorders. 3rd ed. Darien. IL: American Academy of Sleep Medicine; 2014. [Google Scholar]
- 2.Partinen M, Kornum BR, Plazzi G, Jennum P, Julkunen I, Vaarala O. Narcolepsy as an autoimmune disease: the role of H1N1 infection and vaccination. Lancet Neurol. 2014;13:600–13. doi: 10.1016/S1474-4422(14)70075-4. [DOI] [PubMed] [Google Scholar]
- 3.Ingravallo F, Gnucci V, Pizza F, et al. The burden of narcolepsy with cataplexy: how disease history and clinical features influence socioeconomic outcomes. Sleep Med. 2012;3:1293–300. doi: 10.1016/j.sleep.2012.08.002. [DOI] [PubMed] [Google Scholar]
- 4.Kryger MH, Walid R, Manfreda J. Diagnoses received by narcolepsy patients in the year prior to diagnosis by a sleep specialist. Sleep. 2002;25:36–41. doi: 10.1093/sleep/25.1.36. [DOI] [PubMed] [Google Scholar]
- 5.Macleod S, Ferrie C, Zuberi SM. Symptoms of narcolepsy in children misinterpreted as epilepsy. Epileptic Disord. 2005;7:13–7. [PubMed] [Google Scholar]
- 6.Stores G. The protean manifestations of childhood narcolepsy and their misinterpretation. Dev Med Child Neurol. 2006;48:307–10. doi: 10.1017/S0012162206000661. [DOI] [PubMed] [Google Scholar]
- 7.Singh AK, Saha A, Dutt N, Mohapatra PR, Janmeja AK, Bansal A. Narcolepsy presenting as refractory epilepsy. Indian J Pediatr. 2009;76:1073. doi: 10.1007/s12098-009-0211-1. [DOI] [PubMed] [Google Scholar]
- 8.Kauta SR, Marcus CL. Cases of pediatric narcolepsy after misdiagnoses. Pediatr Neurol. 2012;47:362–5. doi: 10.1016/j.pediatrneurol.2012.08.005. [DOI] [PubMed] [Google Scholar]
- 9.Zhou J, Zhang X, Dong Z. Case report of narcolepsy in a six-year-old child initially misdiagnosed as atypical epilepsy. Shanghai Arch Psychiatry. 2014;26:232–5. doi: 10.3969/j.issn.1002-0829.2014.04.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Morrish E, King MA, Smith IE, Shneerson JM. Factors associated with a delay in the diagnosis of narcolepsy. Sleep Med. 2004;5:37–41. doi: 10.1016/j.sleep.2003.06.002. [DOI] [PubMed] [Google Scholar]
- 11.Thorpy MJ, Krieger AC. Delayed diagnosis of narcolepsy: characterization and impact. Sleep Med. 2014;15:502–7. doi: 10.1016/j.sleep.2014.01.015. [DOI] [PubMed] [Google Scholar]
- 12.Rocca FL, Pizza F, Ricci E, Plazzi G. Narcolepsy during childhood: an update. Neuropediatrics. 2015;46:181–98. doi: 10.1055/s-0035-1550152. [DOI] [PubMed] [Google Scholar]
- 13.Lecendreux M, Lavault S, Lopez R, et al. Attention-deficit/ hyperactivity disorder (ADHD) symptoms in pediatric narcolepsy: a cross-sectional study. Sleep. 2015;38:1285–95. doi: 10.5665/sleep.4910. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Broughton WA, Broughton RJ. Psychosocial impact of narcolepsy. Sleep. 1994;17(8 Suppl):S45–9. doi: 10.1093/sleep/17.suppl_8.s45. [DOI] [PubMed] [Google Scholar]
- 15.Posar A, Pizza F, Parmeggiani A, Plazzi G. Neuropsychological findings in childhood narcolepsy. J Child Neurol. 2014;29:1370–6. doi: 10.1177/0883073813508315. [DOI] [PubMed] [Google Scholar]
- 16.Fortuyn HA, Lappenschaar GA, Nienhuis FJ, et al. Psychotic symptoms in narcolepsy: phenomenology and a comparison with schizophrenia. Gen Hosp Psychiatry. 2009;31:146–54. doi: 10.1016/j.genhosppsych.2008.12.002. [DOI] [PubMed] [Google Scholar]
- 17.Fortuyn HA, Lappenschaar MA, Furer JW, et al. Anxiety and mood disorders in narcolepsy: a case-control study. Gen Hosp Psychiatry. 2010;32:49–56. doi: 10.1016/j.genhosppsych.2009.08.007. [DOI] [PubMed] [Google Scholar]
- 18.Fortuyn HA, Mulders PC, Renier WO, Buitelaar JK, Overeem S. Narcolepsy and psychiatry: an evolving association of increasing interest. Sleep Med. 2011;12:714–9. doi: 10.1016/j.sleep.2011.01.013. [DOI] [PubMed] [Google Scholar]
- 19.Ohayon MM. Narcolepsy is complicated by high medical and psychiatric comorbidities: a comparison with the general population. Sleep Med. 2013;14:488–92. doi: 10.1016/j.sleep.2013.03.002. [DOI] [PubMed] [Google Scholar]
- 20.Modestino EJ, Winchester J. A retrospective survey of childhood ADHD symptomatology among adult narcoleptics. J Atten Disord. 2013;17:574–82. doi: 10.1177/1087054713480033. [DOI] [PubMed] [Google Scholar]
- 21.Pizza F, Magnani M, Indrio C, Plazzi G. The hypocretin system and psychiatric disorders. Curr Psychiatry Rep. 2014;16:433. doi: 10.1007/s11920-013-0433-9. [DOI] [PubMed] [Google Scholar]
- 22.Chen Q, de Lecea L, Hu Z, Gao D. The hypocretin/orexin system: an increasingly important role in neuropsychiatry. Med Res Rev. 2015;35:152–97. doi: 10.1002/med.21326. [DOI] [PubMed] [Google Scholar]
- 23.Kotagal S, Hartse KM, Walsh JK. Characteristics of narcolepsy in preteenaged children. Pediatrics. 1990;85:205–9. [PubMed] [Google Scholar]
- 24.Allsopp MR, Zaiwalla Z. Narcolepsy. Arch Dis Child. 1992;67:302–6. doi: 10.1136/adc.67.3.302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Dahl RE, Holttum J, Trubnick L. A clinical picture of child and adolescent narcolepsy. J Am Acad Child Adolesc Psychiatry. 1994;33:834–41. doi: 10.1097/00004583-199407000-00009. [DOI] [PubMed] [Google Scholar]
- 26.Guilleminault C, Pelayo R. Narcolepsy in prepubertal children. Ann Neurol. 1998;43:135–42. doi: 10.1002/ana.410430125. [DOI] [PubMed] [Google Scholar]
- 27.Han F, Chen E, Wei H, et al. Childhood narcolepsy in North China. Sleep. 2001;24:321–4. doi: 10.1093/sleep/24.3.321. [DOI] [PubMed] [Google Scholar]
- 28.Kotagal S, Krahn LE, Slocumb N. A putative link between childhood narcolepsy and obesity. Sleep Med. 2004;5:147–50. doi: 10.1016/j.sleep.2003.10.006. [DOI] [PubMed] [Google Scholar]
- 29.Aran A, Einen M, Lin L, Plazzi G, Nishino S, Mignot E. Clinical and therapeutic aspects of childhood narcolepsy-cataplexy: a retrospective study of 51 children. Sleep. 2010;33:1457–64. doi: 10.1093/sleep/33.11.1457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Peraita-Adrados R, García-Peñas JJ, Ruiz-Falcó L, et al. Clinical. polysomnographic and laboratory characteristics of narcolepsy cataplexy in a sample of children and adolescents. Sleep Med. 2011;12:24–7. doi: 10.1016/j.sleep.2010.02.017. [DOI] [PubMed] [Google Scholar]
- 31.Nevsimalova S, Jara C, Prihodova I, Kemlink D, Sonka K, Skibova J. Clinical features of childhood narcolepsy. Can cataplexy be foretold? Eur J Paediatr Neurol. 2011;15:320–5. doi: 10.1016/j.ejpn.2011.01.008. [DOI] [PubMed] [Google Scholar]
- 32.Stores G, Montgomery P, Wiggs L. The psychosocial problems of children with narcolepsy and those with excessive daytime sleepiness of uncertain origin. Pediatrics. 2006;118:e1116–23. doi: 10.1542/peds.2006-0647. [DOI] [PubMed] [Google Scholar]
- 33.Inocente CO, Gustin MP, Lavault S, et al. Depressive feelings in children with narcolepsy. Sleep Med. 2014;15:309–14. doi: 10.1016/j.sleep.2013.08.798. [DOI] [PubMed] [Google Scholar]
- 34.Dorris L, Zuberi SM, Scott N, Moffat C, McArthur I. Psychosocial and intellectual functioning in childhood narcolepsy. Dev Neurorehabil. 2008;11:187–94. doi: 10.1080/17518420802011493. [DOI] [PubMed] [Google Scholar]
- 35.Szakács A, Hallböök T, Tideman P, Darin N, Wentz E. Psychiatric comorbidity and cognitive profile in children with narcolepsy with or without association to the H1N1 influenza vaccination. Sleep. 2015;38:615–21. doi: 10.5665/sleep.4582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Huang YS, Guilleminault C, Chen CH, Lai PC, Hwang FM. Narcolepsy-cataplexy and schizophrenia in adolescents. Sleep Med. 2014;15:15–22. doi: 10.1016/j.sleep.2013.09.018. [DOI] [PubMed] [Google Scholar]
- 37.Inocente CO, Gustin MP, Lavault S, et al. Quality of life in children with narcolepsy. CNS Neurosci Ther. 2014;20:763–71. doi: 10.1111/cns.12291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Drake C, Nickel C, Burduvali E, et al. The pediatric daytime sleepiness scale (PDSS): sleep habits and school outcomes in middleschool children. Sleep. 2003;26:455–8. [PubMed] [Google Scholar]
- 39.Commission on Classification and Terminology of the International League Against Epilepsy. Proposal for revised classification of epilepsies and epileptic syndromes. Epilepsia. 1989;30:389–99. doi: 10.1111/j.1528-1157.1989.tb05316.x. [DOI] [PubMed] [Google Scholar]
- 40.Achenbach TM. Burlington: Department of Psychiatry, University of Vermont; 1991. Integrative guide for the CBCL/4-18, YSR and TRF profiles. [Google Scholar]
- 41.Achenbach TM, Rescorla LA. Burlington: Research Centre for Children, Youth and Families, University of Vermont; 2001. Manual for the ASEBA school-age forms & profiles. [Google Scholar]
- 42.Achenbach TM. CBCL (Child Behavior Checklist 4-18) Adattamento italiano e standardizzazione a cura di M. Molteni e A. Frigerio. 2001. I.R.C.C.S. Medea. Italia. [Google Scholar]
- 43.American Psychiatric Association. 4th ed. Washington, DC: American Psychiatric Association; 1994. Diagnostic and Statistical Manual of Mental Disorders. [Google Scholar]
- 44.Varni JW, Seid M, Rode CA. The PedsQL: measurement model for the pediatric quality of life inventory. Med Care. 1999;37:126–39. doi: 10.1097/00005650-199902000-00003. [DOI] [PubMed] [Google Scholar]
- 45.Trapanotto M, Giorgino D, Zulian F, Benini F, Varni JW. The Italian version of the PedsQL in children with rheumatic diseases. Clin Exp Rheumatol. 2009;27:373–80. [PubMed] [Google Scholar]
- 46.Simeonova DI, Nguyen T, Walker EF. Psychosis risk screening in clinical high-risk adolescents: a longitudinal investigation using the Child Behavior Checklist. Schizophr Res. 2014;159:7–13. doi: 10.1016/j.schres.2014.07.046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Roth B, Nevsimalova S. Depression in narcolepsy and hypersomnia. Schweiz Arch Neurol Neurochir Psych. 1975;116:291–300. [PubMed] [Google Scholar]
- 48.Reynolds CF, Christiansen CL, Taska LS, et al. Sleep in narcolepsy and depression. Does it look alike? J Nerv Ment Dis. 1983;171:290–5. doi: 10.1097/00005053-198305000-00005. [DOI] [PubMed] [Google Scholar]
- 49.Dauvilliers Y, Paquereau J, Bastuji H, Drouot X, Weil JS, Viot-Blanc V. Psychological health in central hypersomnias: the French Harmony study. J Neurol Neurosurg Psychiatry. 2009;80:636–41. doi: 10.1136/jnnp.2008.161588. [DOI] [PubMed] [Google Scholar]
- 50.Coren HZ, Strain JJ. A case of narcolepsy with psychosis (paranoid state of narcolepsy) Compr Psychiatry. 1965;6:191–9. doi: 10.1016/s0010-440x(65)80036-0. [DOI] [PubMed] [Google Scholar]
- 51.Shapiro B, Spitz H. Problems in the differential diagnosis of narcolepsy versus schizophrenia. Am J Psychiatry. 1976;133:1321–3. doi: 10.1176/ajp.133.11.1321. [DOI] [PubMed] [Google Scholar]
- 52.Douglass AB, Hays P, Pazderka F, Russell JM. Florid refractory schizophrenias that turn out to be treatable variants of HLA-associated narcolepsy. J Nerv Ment Dis. 1991;179:12–7. doi: 10.1097/00005053-199101000-00003. discussion 18. [DOI] [PubMed] [Google Scholar]
- 53.Douglass AB, Shipley JE, Haines RF, Scholten RC, Dudley E, Tapp A. Schizophrenia. narcolepsy. and HLADr15. Dq6. Biol Psychiatry. 1993;34:773–80. doi: 10.1016/0006-3223(93)90066-m. [DOI] [PubMed] [Google Scholar]
- 54.Jackson C, Bachman D. Narcolepsy-related psychosis misinterpreted as schizophrenia. Neuropsychiatry Neuropsychol Behav Neurol. 1996;9:139–140. [Google Scholar]
- 55.Bhat SK, Galang R. Narcolepsy presenting as schizophrenia. Am J Psychiatry. 2002;159:1245. doi: 10.1176/appi.ajp.159.7.1245. [DOI] [PubMed] [Google Scholar]
- 56.Vourdas A, Shneerson JM, Gregory CA, et al. Narcolepsy and psychopathology: is there an association? Sleep Med. 2002;3:353–60. doi: 10.1016/s1389-9457(02)00062-x. [DOI] [PubMed] [Google Scholar]
- 57.Ishiguro T, Miyasaka M. Narcolepsy with chronic hallucinatory and paranoid state. Seishin Igaku. 1969;11:885–92. [Google Scholar]
- 58.Ullman KC. Narcolepsy and schizophrenia. Am J Psychiatry. 1977;134:822. doi: 10.1176/ajp.134.7.822b. [DOI] [PubMed] [Google Scholar]
- 59.Honda Y. DSM-III diagnoses for patients with concomitant narcolepsy and hallucinatory paranoid state. Jpn J Clin Psychiatry. 1982;11:239–46. [Google Scholar]
- 60.Cadieux RJ, Kales JD, Kales A, Biever J, Mann LD. Pharmacologic and psychotherapeutic issues in coexistent paranoid schizophrenia and narcolepsy: case report. J Clin Psychiatry. 1985;46:191–3. [PubMed] [Google Scholar]
- 61.Silvestri R, Montagnese C, De Domenico P, et al. Narcolepsy and psychopathology. A case report. Acta Neurol (Napoli) 1991;13:275–8. [PubMed] [Google Scholar]
- 62.Takeuchi N, Mukai M, Uchimura N, Satomura T, Sakamoto T, Maeda H. A narcoleptic patient exhibiting hallucinations and delusion. Psychiatry Clin Neurosci. 2000;54:321–2. doi: 10.1046/j.1440-1819.2000.00696.x. [DOI] [PubMed] [Google Scholar]
- 63.Kishi Y, Konishi S, Koizumi S, Kudo Y, Kurosawa H, Kathol RG. Schizophrenia and narcolepsy: a review with a case report. Psychiatry Clin Neurosci. 2004;58:117–24. doi: 10.1111/j.1440-1819.2003.01204.x. [DOI] [PubMed] [Google Scholar]
- 64.Talih FR. Narcolepsy presenting as schizophrenia: a literature review and two case reports. Innov Clin Neurosci. 2011;8:30–4. [PMC free article] [PubMed] [Google Scholar]
- 65.Ohayon MM, Priest RG, Caulet M, Guilleminault C. Hypnagogic and hypnopompic hallucinations: pathological phenomena? Br J Psychiatry. 1996;169:459–67. doi: 10.1192/bjp.169.4.459. [DOI] [PubMed] [Google Scholar]
- 66.Palac S, Meador KJ. Antiepileptic drugs and neurodevelopment: an update. Curr Neurol Neurosci Rep. 2011;11:423–7. doi: 10.1007/s11910-011-0194-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Pinquart M, Teubert D. Academic. physical. and social functioning of children and adolescents with chronic physical illness: a meta-analysis. J Pediatr Psychol. 2012;37:376–89. doi: 10.1093/jpepsy/jsr106. [DOI] [PubMed] [Google Scholar]
- 68.Blouin AM, Fried I, Wilson CL, et al. Human hypocretin and melanin-concentrating hormone levels are linked to emotion and social interaction. Nat Commun. 2013;4:1547. doi: 10.1038/ncomms2461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Meletti S, Vaudano AE, Pizza F, et al. The brain correlates of laugh and cataplexy in childhood narcolepsy. J Neurosci. 2015;35:11583–94. doi: 10.1523/JNEUROSCI.0840-15.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


