Abstract
Background:
There are numerous investigations on wide range of issues that disrupt regulatory spermatogenesis, individuals who are exposed to drug abuse faced infertility and immature spermatogenesis.
Objective:
The aim of this study was to evaluate the addiction effects of morphine and its derivatives on rats spermatogenesis.
Materials and Methods:
40 male Wistar rats were randomly divided into 5 equal groups, which were exposed either with intravenous morphine, naloxone, naloxone and morphine, sham (with normal saline injection) and a control group without infusion. Spermatogenesis was assessed after three months via histological sections with hematoxylin and eosin staining, using a light microscope based on measurement of spermatogonia, spermatocyte, spermatid, and spermatozoa.
Results:
Those rats that received opioids had changes in spermatogenesis function. The population of spermatogenesis cycle cells at spermatogonia, spermatocyte, spermatid, and spermatozoa stages was significantly decreased in those rats that received opioid in comparison to the control group (p<0.05). Histological studies revealed that changes in different groups of opioid application might affect sperm formation. Sperm count in morphine group was (0±0) and in naloxone group, naloxone+morphine, sham and control were 235±3.77, 220±3.81, 247.12±6.10 and 250±6.54, respectively (p<0.001).
Conclusion:
Morphine could affect all spermatogenesis stages.
Key Words: Opioids, Morphine, Spermatogenesis, Sperm, Rats
Introduction
Despite the large number of studies addressing the impacts of opioid consumption in women on pregnancy outcomes, few have found such an effect on male fertility along with paternal exposure relationship in development of offspring physiological and behavioral features (1-3). In some cases opioid drugs are used as pain reliever or to treat premature ejaculation, which may affect fertility (4). Herbal drugs such as Achillea millefolium or environmental factors such as Bisphenol-A might have same function and affect germinal epithelium in gonadal region (5). However, due to many involved cofounding variables, primary explanation for this relationship has never been well studied (1).
In contrast to the human subjects, animal models are available, allowing for more robust and much more biological assessment of underlying processes (1, 6-9). With this in mind, extensive amounts of evidence have supported the relationship between chronic exposures of adult male rodents to opiates with development of future generation (6, 10-13). No single study has been done to evaluate the exact mechanism of this phenomenon yet. Many studies have shown the effect of opioid on hypothalamic and gonadal system via evaluating gene expression and hormonal analysis (14, 15). However, the results are not conclusive because of lacking morphological studies.
Therefore, we aimed to evaluate the histological alterations in different stages of spermatogenesis in male rats. For this purpose, we evaluated adult male rat testis under a chronic exposure to opioids such as morphine to reveal the fertility status on paternity of animal model.
Materials and methods
Animal model establishment
This experimental animal study was conducted on forty adult male Wistar rats obtained from Pasteur Institute Iran with a mean age of three months and a weight range of 250-300 gr. The ethics committee of Tehran University of Medical Sciences approved the study and all protocols provided by Institutional Committee of Animal Care were exactly followed.
The rats were housed singly in a room in animal laboratory at 12 hr light/dark cycle provided with proper nutrition (Pars Animal Feed Factory, Iran). Temperature was set between 20-24oC. The rats were randomized into five equal groups (8 rats in each group) as follows: morphine, naloxone, naloxone and morphine, sham, and control. Sham group was injected only with normal saline serum and control group was only observed without any type of injection.
Morphine sulfate (TEMAD, Iran) was injected subcutaneously (5 mg/kg) and its effects was evaluated in 7 different time intervals, i.e. days 0, 10, 20, 30, 40, 70, 90 (16, 17). Because of short and long term effects of morphine, we evaluated it’s early and late effects by choosing these different timelines (18, 19). Withdrawal symptoms were checked subsequently including tremor, movements of limb off the baseline floor or diarrhea occurrence after 30 min of injection. Naloxone (Darupakhsh, Iran) which was chosen as antagonist for morphine addiction was administrated 1.5 mg/kg intra-peritoneally (20). Naloxone was injected 30 min before the test (17). Moderate dose of naloxone and morphine were administered in the naloxone+morphine group (1, 4 respectively), and normal saline was administered in the sham group (17).
Histological study
According to the mentioned timeline, each group of rats was scarified by chloroform and testes were removed and washed out for histological analysis. Tissue samples were added to fixative solutions (formalin) for 24 hr. The tissues were washed with alcohol, embedded in paraffin, sliced in a 5 µm thickness and finally stained with hematoxylin and eosin (Sigma, England). Using a light microscope (CX31- OLYMPUS, Japan) and a magnification field of 40×/0.65 to evaluate spermatogenesis in different stages.
Morphometry of spermatogenesis cycle in testis
Through the seminiferous location characterization, we can evaluate spermatogenesis stages in seminiferous tubules. In each cross section of seminiferous tubule, stage analysis was done through the tubules random selection. Cells were counted according to the shape of nuclei and their location to luminal part and were evaluated by Photo tools version 2 (Microsoft Corp.) (21).
Statistical analysis
Data were analyzed by IBM statistical package for the social sciences (SPSS, version 21, IBM Corp., New York, US) to show the fertility indices difference between experimental groups. Normal distribution of the data was evaluated using Kolmogorov-Smirnov test. In order to analyze continuous variables, t-test and one-way ANOVA were applied for parametric variables while Kruskal-Wallis and Mann-Whitney U-tests were used to analyze the nonparametric variables. The obtained values were deemed significant at p<0.05.
Results
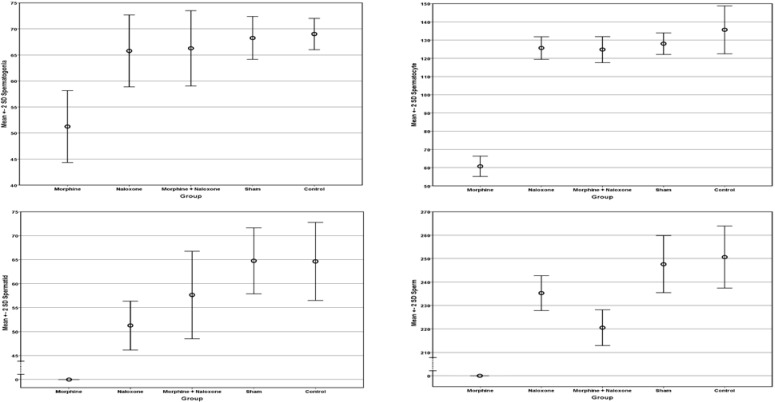
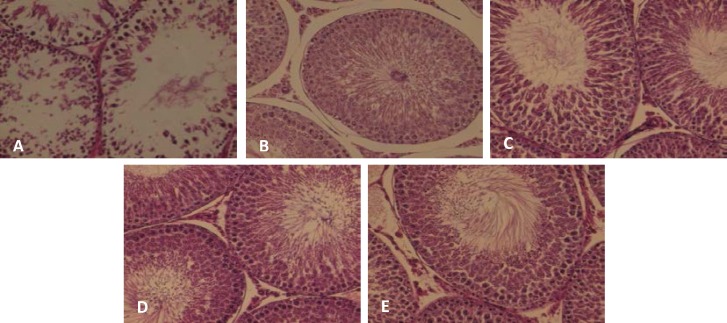
There were no casualties in rats. Fertility parameters characteristics and measurements are summarized in table I. Figure 1 also demonstrates parameters mean±SD according to different groups. The histological evaluation of spermatogenesis cycle in each group is shown in figure 2. The intergroup comparisons of different study groups according to fertility parameters are shown in table II. As clearly indicated, the morphine group had a statistically significant difference with all other groups in term of spermatogonia, spermatocyte, spermatid, and sperm counts, and also testes weight.
Table I.
Comparison of the different spermatogenesis cells counts between study groups (8 rats in each group
| Parameters | Morphine | Naloxone | Morphine + Naloxone | Sham | Control | p-value |
| Spermatogonia | 51 ± 3.46 | 65 ± 3.46 | 66 ± 3.62 | 68 ± 2 | 69 ± 1.40 | <0.001 |
| Spermatocyte | 60 ± 2.77 | 125 ± 3.16 | 124 ± 3.58 | 128 ± 2.87 | 135 ± 6.54 | <0.001 |
| Spermatid | 0±0 | 51 ± 21.59 | 57 ± 4.59 | 64 ± 3.46 | 64 ± 4.06 | <0.001 |
| Sperm | 0±0 | 235 ± 3.77 | 220 ± 3.81 | 247 ± 6.10 | 250 ± 6.54 | <0.001 |
| Testes Weight (mg) | 1.12 ± 0.04 | 1.35 ± 0.03 | 1.32 ± 0.01 | 1.38 ± 0.02 | 1.39 ± 0.05 | <0.001 |
Data are presented as mean±SD. Mann-Whitney U test was used.
Figure 1.
Comparison of the means of the spermatogenesis cycle cells counts according to different study groups
Figure 2.
Microscopic view of seminiferous tubules.
A)Morphine group. B) Control group. C) Sham group. D) Naloxone group. E) Naloxone+Morphine group (Staining, H&E ×40).
Table II.
Intergroup Comparisons of different study groups according to fertility parameters
| Parameters | M-N | M-M+N | M-S | M-C | N-M+N | N-S | N-C | M+N-S | M+N-C | S-C |
|---|---|---|---|---|---|---|---|---|---|---|
| Spermatogonia | 0.001 | 0.001 | 0.001 | 0.001 | 0.526 | 0.064 | 0.011 | 0.187 | 0.030 | 0.151 |
| Spermatocyte | 0.001 | <0.001 | 0.001 | 0.001 | 0.526 | 0.081 | 0.004 | 0.039 | 0.002 | 0.030 |
| Spermatid | <0.001 | <0.001 | <0.001 | <0.001 | 0.532 | 0.013 | 0.011 | 0.004 | 0.003 | 0.741 |
| Sperm | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | 0.372 |
| Testes Weight | 0.001 | 0.001 | <0.001 | <0.001 | 0.442 | 0.092 | 0.026 | 0.003 | <0.001 | 0.652 |
Data are presented as mean±SD. Mann-Whitney U test was used.
M: Morphine
N: Naloxone
M+N: Morphine + Naloxone
S: Sham
C: Control.
Discussion
Opioid can be applied for medical purposes; also in current decade, its prevalence usage among young people in society led us to run out this work to represent its effect on male reproductive system via spermatogenesis evaluation. The opioid system as a biological exchange apparatus is modulated by opioid peptides (22). These endogenous peptides play an important role in the control of pituitary luteinizing hormone (LH) secretion, acting on releasing of gonadotropin hormone (GnRH) release from the hypothalamus (23, 24). Administration of exogenous opiates such as morphine has been shown to decrease the levels of LH in contrast to blockade of endogenous opioid peptide receptor (25).
Wang et al studied the effect of heroin addiction on pituitary-testicular function. They indicated that testicular function had decreased in opioid addiction (26). Some other studies evaluated the effect of opioids on other organs, the most important on cerebellar structure, and found these compounds increase the neurotoxic factors and seems to disrupt the main neural tract (27). In the literature, multiple cases of toxicity and abuse of opioid like morphine and tramadol have been reported, and they figured out these drugs effect on reproductive dysfunction, our findings are in line with recent studies carried out by El Sawy et al and El-Ghawet et al who reported that injections of tramadol would face with disorganization of seminiferous tubules with almost missing of fertility and comparatively decreased spermatogenic cells in rats (28).
Singer et al reported that application of hashish, heroin or morphine has direct effect on vitality, morphology and motility of sperm. They evaluated the semen smear and observed that 20-30% of spermatozoa had weak motility, 40% of spermatogenic cells had pathologic abnormality with decreased number. Finally, they found that morphine family drugs result in oligospermia and azoospermia (29). Furthermore, administration of tramadol and morphine lead to structural abnormalities and could disrupt the normal histological structure of rat testis (30). Our study indicated that all spermatogenic cell populations had decreased levels under the effect of chronic exposure to morphine sulphate. Same results have been already reported by James and colleagues that 13 weeks after withdrawing morphine sulfate the quantitative reductions in the population of spermatogenic cells and levels of pituitary gland hormones were reversed (31).
In addition, in the current study, administration of naloxone was associated with modification of spermatogenic cells count compared with previously exposure to morphine sulphate. Nevertheless, the cell counts did not reach to physiologic level in each group. This could be explained by the results of Bablok et al in which elevation of serum levels of LH, FSH, prolactin, and estradiol were observed while the amount of free testosterone was significantly diminished after performing of naloxone test administering 0.4 mg naloxone intravenously (32). However, in contrast with present study, Cicero et al have concluded that sperm counts and motility are not affected by administration of morphine sulphate (1). In addition, the response of LH to naloxone seems to be related to the concentrations of circulating gonadal steroids in healthy men and women (25).
Effects of morphine and other opiates on reproductive system as well as germ cells and gonadal cell population have been thoroughly studied. Many alterations in reproductive systems of rats have been reported about opioid derivatives. The lower pregnancy rate in female rats mated with opiate exposed male seems to be affected by decreasing effects of morphine on sex organ secretions including seminal vesicles and prostate which together provide a transporting pathway for sperm cells (33-35). It seems that morphine and its families could act directly on opioid receptors and have a negative regulatory impact on serum levels of sex and other gonadal hormones at the same time. In addition, direct effect of opiates on morphology and function of sperm and its progenitors should never be neglected.
It is of interest if further studies evaluate morphology and motility of sperm cells.In addition, evaluating the effects of opioid derivatives on other part of reproductive system can declare the main reason of infertility.
Conclusion
In conclusion, we showed that exposure to morphine reduces the number of all spermatogenesis cell population in male rats. In addition, administration of naloxone would have modulatory effect on spermatogenic cells population.
Acknowledgment
No funds, grants, or other supports were received in any forms.
Note
This article extracted from M.D. thesis. (Ali Lashkari)
Conflict of interest
There is no conflict of interest.
References
- 1.Cicero TJ, Davis LA, LaRegina MC, Meyer ER, Schlegel MS. Chronic opiate exposure in the male rat adversely affects fertility. Pharmacol Biochem Behav. 2002;72:157–163. doi: 10.1016/s0091-3057(01)00751-1. [DOI] [PubMed] [Google Scholar]
- 2.Mattson S, Riley E. A review of the neurobehavioral deficits in children with fetal alcohol syndrome or prenatal exposure to alcohol. Clin Exp Res. 1998;22:304–312. doi: 10.1111/j.1530-0277.1998.tb03651.x. [DOI] [PubMed] [Google Scholar]
- 3.Smeriglio V HC W. Prenatal drug exposure and child outcome Past, present, future. Clin Perinatol. 1999;26:1–16. [PubMed] [Google Scholar]
- 4.El Sawy MM, Malak HWA. Effect of tramadol abuse on testicular tissue of adult albino rats: a light and electron microscopic study. Egypt J Histol. 2015;38:356–366. [Google Scholar]
- 5.Takzare N, Hosseini MJ, Hamideh Mortazavi S, Safaie S, Moradi R. The effect of Achillea millefolium extract on spermatogenesis of male Wistar rats. Hum Exp Toxicol. 2011;30:328–334. doi: 10.1177/0960327110372401. [DOI] [PubMed] [Google Scholar]
- 6.Cicero T, Adams M, Giordano A, Miller B, O’Connor L, Nock B. Influence of morphine exposure during adolescence on the sexual maturation of male rats and the development of their offspring. J Pharmacol Exp Ther. 1991;256:1086–993. [PubMed] [Google Scholar]
- 7.Cicero T, Meyer E, Weist W, Olney J RD B. Effects of chronic morphine administration of the reproductive system of the male rat. J Pharmacol Exp Ther. 1975;192:542–548. [PubMed] [Google Scholar]
- 8.Cicero T, Nock B, O’Connor L, Adams M, Meyer E. Adverse effects of paternal opiate exposure on offspring development and sensitivity to morphine-induced analgesia. J Pharmacol Exp Ther. 1995;273:386–392. [PubMed] [Google Scholar]
- 9.Sharpe RM. Environmental/lifestyle effects on spermatogenesis. Biol Sci. 2010;365:1697–1712. doi: 10.1098/rstb.2009.0206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Friedler G, Cicero T. Paternal pregestational opiate exposure in male mice: neuroendocrine deficits in their offspring. Res Commun Subst Abuse. 1987;8:109–116. [Google Scholar]
- 11.Joffe J LF S. Paternal drug exposure: effects on reproduction and progeny. Semin Perinatol. 1982;6:116–124. [PubMed] [Google Scholar]
- 12.Smith D MJ J. Increased neonatal mortality in offspring of male rats treated with methadone or morphine before mating. Nature. 1974;253:202–203. doi: 10.1038/253202a0. [DOI] [PubMed] [Google Scholar]
- 13.Wang YH, Liu HM. The histological effect of morphine-dependence on male rat germ cell. Chin J Lab Diagnos. 2011;2:010. [PubMed] [Google Scholar]
- 14.Ghowsi M, Yousofvand N. Impact of morphine dependency and detoxification by methadone on male’s rat reproductive system. Iran JReprod Med. 2015;13:275–282. [PMC free article] [PubMed] [Google Scholar]
- 15.Siddiqui A, Haq S, Shah BH. Perinatal exposure to morphine disrupts brain norepinephrine, ovarian cyclicity, and sexual receptivity in rats. Pharmacol Biochem Behav. 1997;58:243–248. doi: 10.1016/s0091-3057(97)00012-9. [DOI] [PubMed] [Google Scholar]
- 16.Yilmaz B, Konar V, Kutlu S, Sandal S, Canpolat S, Gezen M, et al. Influence of chronic morphine exposure on serum LH, FSH, testosterone levels, and body and testicular weights in the developing male rat. SystBiol Reprod Med. 1999;43:189–196. doi: 10.1080/014850199262481. [DOI] [PubMed] [Google Scholar]
- 17.Karimi S, Karami M, Zardooz H, Salimi SH, Sahraei H. Biphasic Effects of Naloxone in the Rats Receiving Morphine Overdose A Place Preference Study. Iran JPharm Res. 2011;10:605. [PMC free article] [PubMed] [Google Scholar]
- 18.Bhutta AT, Rovnaghi C, Simpson PM, Gossett JM, Scalzo FM, Anand K. Interactions of inflammatory pain and morphine in infant rats: long-term behavioral effects. Physiolv Behav. 2001;73:51–58. doi: 10.1016/s0031-9384(01)00432-2. [DOI] [PubMed] [Google Scholar]
- 19.Cicero TJ, Meyer ER, Wiest WG, Olney JW, Bell R. Effects of chronic morphine administration on the reproductive system of the male rat. J Pharmacol Exp Ther. 1975;192:542–548. [PubMed] [Google Scholar]
- 20.Sokol RZ. Hormonal effects of lead acetate in the male rat: mechanism of action. Biol Reprod. 1987;37:1135–1138. doi: 10.1095/biolreprod37.5.1135. [DOI] [PubMed] [Google Scholar]
- 21.Almeida FF, Leal MC, França LR. Testis morphometry, duration of spermatogenesis, and spermatogenic efficiency in the wild boar (Sus scrofa scrofa) Biol Reprod. 2006;75:792–799. doi: 10.1095/biolreprod.106.053835. [DOI] [PubMed] [Google Scholar]
- 22.Burstein SR, Williams TJ, Lane DA, Knudsen MG, Pickel VM, McEwen BS, et al. The influences of reproductive status and acute stress on the levels of phosphorylated delta opioid receptor immunoreactivity in rat hippocampus. Brain Res. 2013;1518:71–81. doi: 10.1016/j.brainres.2013.03.051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Genazzani AR, Petraglia F. Opioid control of luteinizing hormone secretion in humans. J Steroid Biochem. 1989;33:751–755. doi: 10.1016/0022-4731(89)90487-1. [DOI] [PubMed] [Google Scholar]
- 24.Kalra SP. Mandatory neuropeptide-steroid signaling for the preovulatory luteinizing hormone-releasing hormone discharge. Endocr Rev. 1993;14:507–538. doi: 10.1210/edrv-14-5-507. [DOI] [PubMed] [Google Scholar]
- 25.Quigley ME, Yen SSC. The role of endogenous opiates in LH secretion during the menstrual cycle. J Clin Endocrinol Metab. 1980;51:179–181. doi: 10.1210/jcem-51-1-179. [DOI] [PubMed] [Google Scholar]
- 26.WANG C, Chan V, Yeung RT. The effect of heroin addiction on pituitary‐testicular function. Clin Endocrinol. 1978;9:455–461. doi: 10.1111/j.1365-2265.1978.tb03585.x. [DOI] [PubMed] [Google Scholar]
- 27.El-Bermawy MI, Salem MF. Histological changes of the albino rat cerebellar cortex under the effect of different doses of tramadol administration. Egypt J Histol. 2015;38:143–155. [Google Scholar]
- 28.El-Ghawet HA. Effects of tramadol on the reproductive function of wistar albino rats. EurJ Exp Biol. 2015;5:56–64. [Google Scholar]
- 29.Singer R, Ben-Bassat M, Malik Z, Sagiv M, Ravid A, Shohat B, et al. Oligozoospermia, asthenozoospermia, and sperm abnormalities in ex-addict to heroin, morphine, and hashish. SystBiol Reprod Med. 1986;16:167–174. doi: 10.3109/01485018608986938. [DOI] [PubMed] [Google Scholar]
- 30.Abdellatief RB, Elgamal DA, Mohamed EE. Effects of chronic tramadol administration on testicular tissue in rats: an experimental study. Andrologia. 2015;47:674–679. doi: 10.1111/and.12316. [DOI] [PubMed] [Google Scholar]
- 31.James RW, Heywood R, Crook D. Effects of morphine sulphate on pituitary-testicular morphology of rats. Toxicol Lett. 1980;7:61–70. doi: 10.1016/0378-4274(80)90086-7. [DOI] [PubMed] [Google Scholar]
- 32.Bablok L, Fracki S, Wielgos M, Czaplicki M, Marianowski L. [The naloxone test in the degenerative changes of seminiferous tubules] Ginekol Pol. 1998;69:374–379. (In Polish) [PubMed] [Google Scholar]
- 33.Blandau R. On the factors involved in sperm transport through the cervix uteri of the albino rat. Am J Anat. 1945;77:253–272. [Google Scholar]
- 34.Carballada R, Esponda P. Structure of the vaginal plugs generated by normal rats and by rats with partially removed seminal vesicles. J Exp Zoo. 1993;256:61–68. doi: 10.1002/jez.1402650109. [DOI] [PubMed] [Google Scholar]
- 35.Matthews L, Adler N. Systematic interrelationship of mating, vaginal plug position and sperm transport in the rat. Physiol Behav. 1978;20:303–309. doi: 10.1016/0031-9384(78)90224-x. [DOI] [PubMed] [Google Scholar]