Abstract
There have been many individual phytoplankton datasets collected across Australia since the mid 1900s, but most are unavailable to the research community. We have searched archives, contacted researchers, and scanned the primary and grey literature to collate 3,621,847 records of marine phytoplankton species from Australian waters from 1844 to the present. Many of these are small datasets collected for local questions, but combined they provide over 170 years of data on phytoplankton communities in Australian waters. Units and taxonomy have been standardised, obviously erroneous data removed, and all metadata included. We have lodged this dataset with the Australian Ocean Data Network (http://portal.aodn.org.au/) allowing public access. The Australian Phytoplankton Database will be invaluable for global change studies, as it allows analysis of ecological indicators of climate change and eutrophication (e.g., changes in distribution; diatom:dinoflagellate ratios). In addition, the standardised conversion of abundance records to biomass provides modellers with quantifiable data to initialise and validate ecosystem models of lower marine trophic levels.
Subject terms: Community ecology, Biodiversity, Marine biology
Background & Summary
Phytoplankton are microalgae at the base of the food web and directly or indirectly support all marine life. As highly efficient primary producers they are critical to maintaining biodiversity and supporting fisheries throughout the ocean1. Due to their high turnover rates and sensitivity to changes in environmental conditions phytoplankton are useful indicators of changing oceanographic conditions, climate change, and deterioration in water quality2,3. Some phytoplankton produce toxins which may be accumulated by filter feeding shellfish, causing irritation, serious illness or death to animals and humans.
A litre of seawater may contain up to one million algal cells, representing at least 100–150 different species4. These include the larger phytoplankton, dominated by the diatoms and dinoflagellates, but also include flagellates and the coccoid picoplanktonic forms. Currently, 537 infraspecific dinoflagellate and 938 diatom taxa are known to inhabit coastal and oceanic waters around Australia5,6. The fractions of nanoplankton flagellates and coccoid picoplankton, although smaller in size, can account for up to 90% of the total phytoplankton chlorophyll under low biomiass scenarios in offshore waters4. The latter are difficult, or impossible, to identify with light microscopy. Including a reliable estimate of biomass, along with cell abundance will provide more realistic information about the phytoplankton community structure at a particular point in time.
Many researchers have phytoplankton data sitting around on paper records or in spreadsheets, some published and some unpublished, which may eventually be lost or misplaced. Consultancies often hold data archives from several research projects from which data are released only to the client with no time and incentive to publish. These data, archived thoroughly, standardised and freely available to a wider audience, are an invaluable resource for the research community. Small, individual datasets with limited stand alone impact, can collectively provide valuable additions to large scale spatial and temporal studies. Many of these data have previously been published in some form. Journal articles, theses and reports and especially older publications, rarely include the relevant dataset so the data are not available for use after publication unless the author releases the data to a publically accessible platform. Even when data are released, they are often only presence data and are not explicit as they could be.
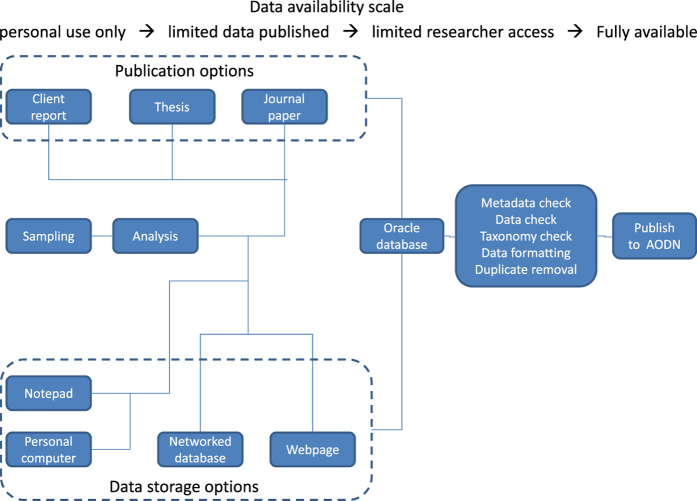
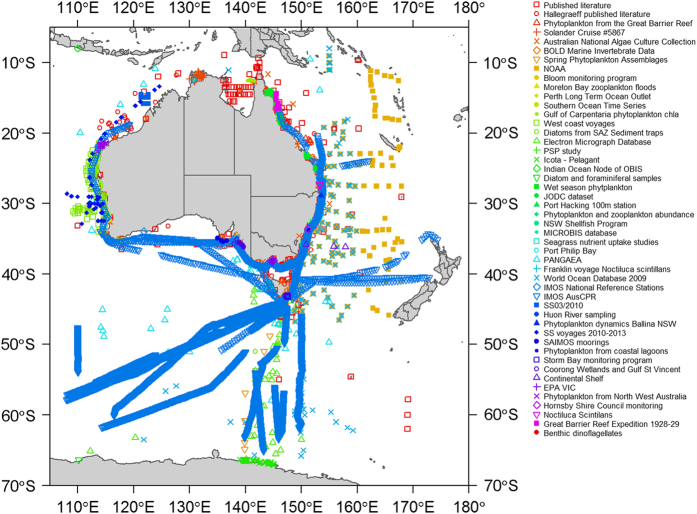
The Australian Phytoplankton Database has been collated from literature, active and retired researchers, consultancies, archives and databases (Fig. 1). Only data with the relevant corresponding metadata about collection location, date and methods has been included. Figure 2 shows the spatial extent of the records collected in this data set. Taxonomic identification of many phytoplankton taxa is fraught with difficulties, especially when limited to light microscopy. Whilst all have been standardised to the correct current classification as given by the World Register of Marine Species (WoRMS) (http://www.marinespecies.org/aphia.php?p=webservice), it is clearly not possible to verify every identification made by each analyst.
Figure 1. Flow diagram of data collation, verification and release to AODN.
Figure 2. Locations of data for each project in the Australian Phytoplankton Database.
The Australian Phytoplankton Database contains data on marine phytoplankton abundance, biomass and species composition. It can be used to:
determine community structures and range changes over time8,9 or oceanographic conditions10;
understand dynamics of harmful algal species to help inform the aquaculture industry11;
develop inputs to climate, ecosystem and fisheries models to inform management about resources12.
The Australian Phytoplankton Database is available through the Australian Ocean Data Network (AODN: http://portal.aodn.org.au) portal. This portal is the main repository for marine data in Australia. The Phytoplankton Database will be maintained in the CSIRO data centre and can be updated with new records, which will automatically upload to the AODN. Researchers wishing to submit new data should contact the corresponding author or the AODN. A snapshot of the Australian Phytoplankton Database as it is at the time of this publication has been assigned a DOI and will be maintained in perpetuity by the AODN (Data Citation 1).
The Australian Phytoplankton Database has been built with ease of use and minimising user error in mind. Therefore it provides the clean data at a level that requires minimal interpretation. The CSIRO database holds all the raw data, for example original species names and ambiguous records, from these datasets, and researchers can request further information from the corresponding author if required.
Methods
Samples were mainly collected from Niskin bottles, net drops or tows, and the Continuous Plankton Recorder (CPR). These are all standard methods of collecting phytoplankton samples13,14 and many are still largely reliant on a phytoplankton manual written in 1978 (ref. 15). A few samples were collected using automated samplers on moorings16. The sampling was done via research vessels, container ships and small boats by experienced researchers, students and volunteers. The majority of the samples were preserved using Lugol’s solution, although formalin, paraformaldehyde and glutaraldehyde have also been used. Different methods of preservation can affect the condition of the sample and which taxa are well preserved4,17. Samples were analysed with standard methods including light microscopy, transmission or scanning electron microscopy which are described in Hallegraeff, et al.4 All methodological variations within our phytoplankton database are detailed in the metadata where available and recorded for each data entry. Where available a citation is referenced for each project, which gives details on methodologies and limitations from that project (Table 1 (available online only)).
Table 1. Summary information on the project data sets, their location, time and taxonomic resolutions, numbers of samples and records available.
| Project_id | Project description | Custodian (affiliation) | Acknowledgement | Location | Start date | End date | No samples | No records | Taxonomic resolution |
|---|---|---|---|---|---|---|---|---|---|
| Organised by number of records (* ongoing datasets) | |||||||||
| 597* | IMOS AusCPR14 | Anthony Richardson (CSIRO) | IMOS | Australia | 2007-12-17 | 2016-01-20 | 13866 | 1375559 | Species where possible, zooplankton data available27 |
| 794* | NSW Shellfish Program29,30 | Steve Brett (Microalgal Services) | Microalgal Services | Estuaries in NSW | 2004-08-24 | 2014-08-11 | 11669 | 1340327 | HAB species only |
| 1065* | EPA VIC long term monitoring | Renee Patten (EPA VIC) | Microalgal Services | Victoria | 2008-02-21 | 2014-07-22 | 715 | 223266 | Species where possible |
| 599* | IMOS NRS31 | Anthony Richardson (CSIRO) | IMOS | Australia | 2008-09-29 | 2015-12-12 | 490 | 95625 | Species where possible, zooplankton and biogeochemical data available27 |
| 1054 | Phytoplankton dynamics | David Rissik (NCCARF) | Ballina NSW | 1998-11-24 | 2000-11-27 | 407 | 93632 | Species where possible | |
| 589 | Bloom monitoring program | Ian Jameson (CSIRO) | Tasmania | 1992-10-12 | 1993-04-15 | 388 | 62076 | Species of interest only | |
| 1051 | Huon River sampling | Peter Thompson (CSIRO) | Huon River TAS | 1996-07-02 | 1998-10-05 | 1705 | 44517 | Species where possible; chla | |
| 796 | Phytoplankton zooplankton NSW9,10,32 | Linda Armbrecht (Macquarie University) | IMOS, OEH, NMSC | Coffs Harbours NSW | 2011-05-27 | 2012-09-12 | 297 | 44248 | Species where possible, chla |
| 1064* | Hornsby Shire Council monitoring | Peter Coad (Hornsby Council) | Hornsby Council / Microalgal Services | Hornsby area NSW | 2003-05-06 | 2015-06-17 | 149 | 26969 | Species where possible; chla |
| 1068 | GBR expedition 1928-29 | Published literature | Great Barrier Reef QLD | 1928-07-25 | 1929-07-17 | 462 | 33675 | Species where possible | |
| 1066 | Phytoplankton NW Shelf | Miles Furnas (AIMS) | GBRMPA | North West Shelf WA | 1997-10-26 | 2002-04-11 | 141 | 25803 | Species where possible |
| 807 | Wet season phytoplankton | Michelle Devlin (JCU) | North Queensland | 2011-02-14 | 2014-05-17 | 206 | 25132 | Species where possible | |
| 609 | Phytoplankton dynamics | Sophie C. Leterme (Flinders University) | Coorong Wetlands Gulf St Vincent SA | 2010-07-01 | 2013-08-01 | 143 | 23171 | Selected taxa identified to highest resolution possible | |
| 509 | Gulf of Carpentaria phytoplankton33–35 | Michele Burford (Griffith University) | Gulf of Carpentaria QLD | 1986-05-08 | 1992-03-28 | 611 | 17856 | Genera where possible | |
| 786 | Voyage SS04/2007 | Peter Thompson (CSIRO) | West Coast Australia | 2007-05-16 | 2007-06-04 | 166 | 17264 | Species where possible, chla | |
| 1056* | SAIMOS moorings | Sophie C. Leterme (Flinders University) | IMOS | South Australia | 2010-07-01 | 2013-02-01 | 92 | 13858 | Species where possible |
| 479 | Moreton Bay zooplankton floods | Julian Uribe (CSIRO) | Moreton Bay QLD | 2011-01-19 | 2011-12-21 | 84 | 12936 | Species where possible, zooplankton data available27 | |
| 1059 | Continental Shelf | Sophie C. Leterme (Flinders University) | East Coast Aus | 2011-01-28 | 2013-07-31 | 208 | 12017 | Selected taxa identified to highest resolution possible | |
| 806 | Phytoplankton Great Barrier Reef | Miles Furnas (AIMS) | GBRMPA | Great Barrier Reef QLD | 1986-02-07 | 2001-08-28 | 88 | 11748 | Species where possible |
| 790 | Voyage FR10/2000 | Peter Thompson (CSIRO) | West Coast Australia | 2000-11-15 | 2000-11-26 | 102 | 10608 | Species where possible | |
| 1067 | NW Shelf Phyto | Paul Thomson (UWA) | IMOS | North West Shelf WA | 2015-01-18 | 2015-01-24 | 46 | 8234 | Species where possible |
| 788 | Voyage SS07/2005 | Peter Thompson (CSIRO) | West Coast Australia | 2005-07-21 | 2005-08-10 | 72 | 7488 | Species where possible | |
| 784 | Voyage SS08/2003 | Peter Thompson (CSIRO) | West Coast Australia | 2003-10-02 | 2003-10-21 | 57 | 5928 | Species where possible | |
| 795 | Perth Long Term Ocean Outlet | Peter Thompson (CSIRO) | Perth | 1999-03-24 | 2000-03-30 | 64 | 5687 | Species where possible | |
| 1057 | Phytoplankton from coastal lagoons in Sydney | Shauna Murray (UTS) | Sydney NSW | 2011-11-02 | 2014-05-02 | 259 | 5222 | Species where possible | |
| 575 | NOAA | NOAA | Australia | 1947-03-11 | 1984-02-16 | 138 | 4786 | Species where possible | |
| 1058 | Storm Bay | Ruth Eriksen (IMAS) | Storm Bay TAS | 2009-11-09 | 2015-04-22 | 85 | 4703 | Species where possible; chla | |
| 801 | PSP study | Tim Ingleton (EnvNSW) | Morpeth NSW | 2001-09-03 | 2001-11-12 | 87 | 3306 | Species where possible, chla | |
| 780 | World Ocean Database 200936 | OBIS World Ocean Database 2009 | Australia | 1946-12-30 | 1984-02-16 | 118 | 2961 | Species where possible | |
| 782 | Phytoplankton Port Hacking 100 m station37,38 | Penelope Ajani (Macquarie University) | Port Hacking NSW | 1997-04-03 | 2009-12-07 | 113 | 2944 | Species where possible, chla | |
| 559 | Wood (1954).39 | Published literature | Australia | 1902-01-01 | 1955-01-01 | 33 | 2924 | Species where possible | |
| 511 | Crosby & Wood (1958).40 | Published literature | Australia | 1938-11-01 | 1958-01-01 | 32 | 2670 | Species where possible | |
| 804 | Solander cruise #5867 | Diane Purcell-Meyerink (NAMRA & AIMS) | NAMRA / AIMS | Darwin & Van Diemen Gulf, NT | 2013-09-09 | 2013-09-22 | 34 | 2530 | Species where possible, chla |
| 805* | Southern Ocean Time Series | Ruth Eriksen (CSIRO) | Southern Ocean Time Series mooring | 2010-09-12 | 2011-04-07 | 24 | 2409 | Species where possible | |
| 792 | Voyage FRxx/2005 | Peter Thompson (CSIRO) | West Coast Australia | 1995-06-13 | 1995-06-14 | 23 | 2392 | Species where possible | |
| 1070 | Benthic dinoflagellates41 | Michael Holmes (DSITI) | East Coast Queensland | 1982-12-02 | 1990-02-28 | 889 | 2186 | Selected genera only, units are no per g substrate | |
| 1053 | SS voyages 2010-201342,43 | Anya Waite (UWA/AWI) | West Coast Australia | 2010-07-10 | 2013-07-21 | 255 | 2111 | Genera where possible; chla | |
| 752 | Icota—Pelagant | Icota—Pelagant Icota—Pelagant | Australia | 2004-01-19 | 2004-01-28 | 54 | 1749 | Species where possible | |
| 1069 | Voyage FR05/199544 | Peter Thompson (CSIRO) | West Coast Australia | 1995-06-04 | 1995-06-15 | 24 | 1562 | Species where possible | |
| 800 | Franklin voyage CTD Noctiluca scintillans45 | Jocelyn Dela-Cruz (UNSW) | NSW | 1998-11-14 | 1999-02-01 | 1046 | 1046 | Noctiluca scintillans only | |
| 571 | CSIRO (1965). | Published literature | Port Hacking NSW | 1965-04-14 | 1965-12-20 | 1 | 925 | Species where possible | |
| 555 | Rothlisberg & Pollard, et al. (1994).46 | Peter Rothlisberg (CSIRO) | Gulf of Carpentaria | 1988-01-01 | 1988-01-01 | 4 | 744 | Genera where possible | |
| 778 | Spring Phytoplankton Assemblages36 | OBIS CLIVAR-SR3 | Southern Ocean | 2001-11-03 | 2001-12-10 | 751 | 751 | Species where possible | |
| 799 | Franklin voyage underway Noctiluca scintillans45 | Jocelyn Dela-Cruz (UNSW) | NSW | 1998-11-14 | 1999-01-30 | 504 | 504 | Noctiluca scintillans only | |
| 573 | Jeffrey & Carpenter (1974).47 | Published literature | Port Hacking NSW | 1971-09-01 | 1972-05-16 | 423 | 423 | Species where possible | |
| 567 | ANZ-DiatomsII | Published literature | Australia and New Zealand | 1844-01-01 | 1959-01-01 | 21 | 366 | Species where possible | |
| 553 | Revelante et al. (1982).48 | Published literature | Australia | 1 | 343 | Species where possible | |||
| 746 | Diatoms from SAZ Sediment traps | OBIS Diatoms from SAZ Sediment traps | Australia | 1997-09-04 | 1997-09-20 | 3 | 304 | Species where possible | |
| 1050 | SS03/2010 | Peter Thompson (CSIRO) | NW shelf WA | 2010-04-16 | 2010-04-25 | 12 | 304 | Species where possible | |
| 529 | Hallegraeff & Jeffrey (1984).49 | Gustaaf Hallegraeff (UTAS) | Australia | 1980-06-08 | 1982-05-19 | 1 | 299 | Species of interest only | |
| 803 | Port Philip Bay50–52 | Sasi Nayar (SARDI) | Port Philip VIC | 2012-12-11 | 2012-12-13 | 9 | 297 | Genera where possible | |
| 587 | Hallegraeff’s notebook | Gustaaf Hallegraeff (UTAS) | Australia | 1982-07-05 | 1984-09-13 | 22 | 291 | Species of interest only | |
| 537 | Australian National Algae Culture Collection | Ian Jameson (CSIRO) | Australia | 1962-01-01 | 2008-01-01 | 24 | 288 | Species of interest only | |
| 772 | Ocean Drilling Program | PANGAEA | Australia | 11 | 264 | Selected species only | |||
| 766 | International marine global change study | PANGAEA | Australia | 1 | 222 | Species where possible | |||
| 563 | Wood (1961).53 | Published literature | Australia | 1961-01-01 | 1961-01-01 | 14 | 192 | Species where possible | |
| 565 | Wood, Crosby & Cassie (1959).54 | Published literature | Australia | 1875-01-01 | 1959-01-01 | 14 | 192 | Species where possible | |
| 533 | Hiramatsu & De Deckker (1996). | Published literature | Southern Tasmania | 1994-01-10 | 1994-01-10 | 5 | 185 | Species where possible | |
| 768 | Paleoenvironmental reconstructions | PANGAEA | Australia | 33 | 176 | Selected species only | |||
| 748 | Electron Micrograph Database | Electron Micrograph Database Electron Micrograph Database | Australia | 1998-03-04 | 2005-03-03 | 45 | 165 | Species where possible | |
| 541 | Jeffrey & Hallegraeff (1987). | Gustaaf Hallegraeff (UTAS) | Eddy Mario NSW | 1981-05-01 | 1981-05-01 | 1 | 152 | Species where possible | |
| 770 | Deep Sea Drilling Project | PANGAEA | Australia | 8 | 134 | Selected species only | |||
| 543 | LeRoi & Hallegraeff (2004). | Gustaaf Hallegraeff (UTAS) | Australia | 1994-06-01 | 1995-05-30 | 3 | 87 | Species where possible | |
| 523 | Hallegraeff (1984).55 | Gustaaf Hallegraeff (UTAS) | Australia | 1979-04-01 | 1984-01-01 | 7 | 84 | Species of interest only | |
| 591 | Zooplankton community dynamics | Sarah Pausina (UQ) | Healthy Waterways | Moreton Bay QLD | 2009-02-04 | 2011-09-28 | 82 | 82 | Noctiluca Scintilans |
| 527 | Hallegraeff & Lucas (1988).56 | Gustaaf Hallegraeff (UTAS) | Australia | 1983-06-01 | 1988-01-01 | 6 | 81 | Species of interest only | |
| 539 | Jeffrey & Hallegraeff (1980).57 | Gustaaf Hallegraeff (UTAS) | East Australian Current | 1978-12-08 | 1978-12-08 | 1 | 79 | Species where possible | |
| 802 | Seagrass nutrient uptake studies58 | Sasi Nayar (SARDI) | South Australia | 2005-06-29 | 2006-02-27 | 9 | 75 | Genera where possible | |
| 577 | Wood (1963b).59 | Published literature | Australia | 1963-01-01 | 1963-01-01 | 7 | 65 | Species where possible | |
| 545 | LeRoi & Hallegraeff (2006).60 | Gustaaf Hallegraeff (UTAS) | Tasmania Australia | 1994-06-01 | 1995-05-30 | 3 | 58 | Species of interest only | |
| 547 | McMinn (1990).61 | Published literature | Australia | 1990-01-01 | 1990-01-01 | 9 | 58 | Selected species only | |
| 569 | CSIRO (1959). | Published literature | Port Hacking NSW | 1959-02-10 | 1959-12-15 | 1 | 51 | Species where possible | |
| 585 | Hallegraeff’s Coral Sea Notebook | Gustaaf Hallegraeff (UTAS) | Coral Sea | 1986-01-23 | 1986-02-02 | 49 | 49 | Species of interest only | |
| 549 | O’Connor et al (1996). | Published literature | Macquarie Harbour NSW | 1995-09-04 | 1995-09-04 | 1 | 45 | Species where possible | |
| 758 | MICROBIS database36 | MICROBIS database | Australia | 12 | 44 | Selected species only | |||
| 525 | Hallegraeff & Reid (1986).62 | Gustaaf Hallegraeff (UTAS) | Australia | 1978-03-28 | 1979-04-30 | 1 | 32 | Species of interest only | |
| 531 | Hallegraeff & Jeffrey (1993).63 | Gustaaf Hallegraeff (UTAS) | NSW and TAS | 1981-10-01 | 1984-09-15 | 5 | 32 | Species of interest only | |
| 551 | Revelante & Gilmartin (1982).64 | Published literature | Australia | 1 | 32 | Species where possible | |||
| 581 | CSIRO Voyage: SP3 | Published literature | Australia | 1982-03-09 | 1982-03-17 | 6 | 29 | Species where possible | |
| 521 | Hallegraeff (1983).65 | Gustaaf Hallegraeff (UTAS) | Australia | 1981-10-01 | 1982-03-01 | 1 | 28 | Species of interest only | |
| 583 | CSIRO Voyage: SP7 | Published literature | Australia | 1982-07-05 | 1982-07-05 | 4 | 24 | Genera where possible | |
| 744 | Diatom and foraminiferal samples36 | OBIS (IOC) | Southern Ocean | 1997-02-11 | 1997-02-11 | 1 | 21 | Species where possible | |
| 517 | Grant & Kerr (1970).66 | Published literature | Port Hacking NSW | 1966-04-01 | 1966-04-01 | 1 | 20 | Species where possible | |
| 513 | Cummins et al. (2004).67 | Published literature | Tuggerah Lakes NSW | 1999-01-01 | 1999-01-01 | 1 | 19 | Genera where possible | |
| 515 | Gottschalk et al. (2007).68 | Published literature | Queensland | 2007-01-01 | 2007-01-01 | 3 | 19 | Species where possible | |
| 742 | BOLD Marine Invertebrate Data36 | OBIS BOLD Marine Invertebrate Data | Australia | 12 | 17 | HAB species only | |||
| 519 | Hallegraeff (1981).69 | Gustaaf Hallegraeff (UTAS) | Port Hacking NSW | 1978-01-01 | 1978-01-01 | 15 | 15 | Species of interest only | |
| 579 | CSIRO Voyage: G01 | Published literature | Australia | 1960-02-02 | 1960-02-04 | 2 | 12 | Species where possible | |
| 764 | Climate | PANGAEA | Australia | 9 | 9 | One species only | |||
| 754 | Indian Ocean Node of OBIS | OBIS IndOBIS—Indian Ocean Node of OBIS | Indian Ocean | 1903-07-02 | 1930-07-02 | 1 | 6 | Selected species only | |
| 756 | JODC dataset | JODC dataset | Australia | 1977-06-28 | 1977-06-29 | 1 | 6 | Species where possible | |
| 561 | Wood (1963).70 | Published literature | North West Australia | 1963-01-01 | 1963-01-01 | 1 | 6 | Selected species only | |
| 557 | Thompson et al. (2008).71 | Published literature | Huon River TAS | 1997-01-01 | 2005-01-01 | 1 | 5 | Selected species only |
There were three stages in data gathering. The first stage was to conduct a literature review of Australian phytoplankton data. Any literature that contained abundance or presence data was digitised and uploaded into the CSIRO maintained Oracle database. The second stage was to scan already existing databases, such as the CSIRO data trawler, the Ocean Biogeographic Ocean System (OBIS) and the Atlas of Living Australia (AOLA). These repositories only store presence records. Relevant data were selected and uploaded into the database. The third stage was to ask researchers to contribute any other data sets that they had. All data were organised into a standard format and uploaded into the database. Data were then served to and hosted by the AODN.
All taxa have been verified as accepted species and given the currently accepted name as defined by the World Register of Marine Species database (WoRMS—http://www.marinespecies.org/aphia.php?p=webservice). If any taxa could not be verified, then a second check was done through AlgaeBase (http://www.algaebase.org/). If this did not verify that taxa as a valid name then the taxonomic level of identification was decreased to a satisfactory level or the entry removed. All abundance values were standardised to cells.l−1 or are given as presence only. Records of the original identifications and units were archived so any records can be checked.
Identification of the smaller phytoplankton is often to a coarser taxonomic level as many cannot be distinguished to species using light microscopy. In some studies electron microscopy has been used to determine species, but in other studies functional groups have been identified. This data set does not include accessory pigment data which can help resolve these smaller taxa18 although it can be thought of as a complementary dataset. Over 20 years of pigment data are available in Australia via the AEsOP database (http://aesop.csiro.au/).
Cell biovolume is calculated as per Hillebrand, et al.19 following the suggested shape factors for each genera. Size parameters were estimated from measurements taken from Australian samples or Australian references where available4,20 and other sources where not21–23. In some data sets, direct measurements of size classes (e.g., P599 the Australian Continuous Plankton Recorder Survey and P597 the IMOS National Reference Stations), were used in preference to literature values. For some taxa there was insufficient information available to estimate a biovolume, these were generally the rare taxa. Rather than estimate a size class without any information available, these have been left blank.
Data Records
Each data record represents the abundance or presence of a phytoplankton taxa at a certain point in space and time and has been given a unique record identification number, P(project_id)_(sample_id)_(record_id). Each data record belongs to a project, with each project having a unique identification number, Pxxx. A project is defined as a set of data records which have been collected together, usually as a cruise or study with the same sampling method and having the same person counting the samples. Metadata ascribed to a project relates to all the data records within that project. Details to identify each separate project are given in Table 1 (available online only). Each sample within that project has a unique sample_id. The sample_id has not been changed from the original data set to maintain traceability. So these may be duplicated between projects but P(project_id)_(sample_id) will be a unique entity in space and time. Species abundance records within the sample are given a unique record_id.
The majority of these projects have been uploaded as part of the collation of data for this database. None have been previously published as datasets but The IMOS National Reference Stations, P599, and Continuous Plankton Recorder Survey, P597, which together constitute half the data in this database, are continually updated and available through the AODN (https://portal.aodn.org.au/search?uuid=dfef238f-db69-3868-e043-08114f8c8a94 and https://portal.aodn.org.au/search?uuid=c1344979-f701-0916-e044-00144f7bc0f4 respectively).
Table 1 (available online only) gives summary information on the project data sets, their space, time and taxonomic resolutions, numbers of samples and records available. Users can select data sets from this information and download as desired through the AODN.
Technical Validation
The Phytoplankton Database will provide an extensive resource for phytoplankton researchers, although there are some caveats due to the variety of the sampling and analysis protocols.
The various sample collection methods infer that abundances might not always be directly comparable across projects. For example, quantitative methods such as bottle sampling (e.g., Project 599) will collect all but the rarest phytoplankton and will include the whole size spectrum, whereas semi-quantitative methods such as net sampling are selective and dependent on tow method, mesh size and the mix of species present in the water as some species may clog the net and trap smaller species that would otherwise go through the mesh (e.g., Project 509)21. By including all data collected using different methods and including this information as meta-data, researchers are able to analyse the relative abundance of each taxa within a project and compare across compatible projects. Metadata includes as much detail as is available about sampling methods and limitations and provides guidance to the users about the potential of each data set. Users should consider collection methods, preservation techniques and microscopic limitations when comparing datasets. All datasets have been standardised to taxa/m3 of water except P1070 where the units are taxa per gram of substrate measured. This project collected the phytoplankton by collecting substrates and analysing parts of the substrate. It is included here as the only data set on Gambierdiscus and associated benthic dinoflagellates from Australia which are important to the studies of the ciguatera.
All datasets submitted can be interpreted as confirming the presence of those species recorded. In some datasets, e.g., time series, it is possible also to infer absences, assuming that all species are looked for on each sampling occasion. Absences have been included in the data sets where the project information available allowed us the confidence to interpret such absences correctly. The interpretation of absences from other projects is at the discretion of the user. We suggest that a project-by-project approach should be taken. If a taxa is not observed at all in a project, then the absence could be due to the taxa not being present, that taxa not being of interest to the analyst, or the inability to identify that taxa. Thus, a real absence should not be inferred. If a taxa is observed in some samples of a project, it can most likely be assumed that the microscopist could identify the taxa, and that a real absence may be inferred in samples within that project where the taxa was not marked as present.
Some data records were removed when there was ambiguity as to the identification of the taxa, i.e., when the taxonomic traceability, usually from older sources, was confused or when spelling mistakes make it unclear which one of two species was meant. Species known as freshwater species were removed as the methods used to collect data were not aimed at freshwater environments and the inclusion of the odd records of these species would not be comprehensive or meaningful. Estuarine species were captured and the records kept. Data records with positions on land, with an unreal number for abundance or with impossible dates were also removed or converted to presence records.
Usage Notes
The database contains information on the functional group of the species, which can aid analysis. Functional groups include diatoms, dinoflagellates, flagellates, ciliates (including tintinnids), silicoflagellates, and cyanobacteria. Once downloaded and binned as required, data are suitable for use in the creation of ecological indicators. For example:
Total diatoms, dinoflagellates
Diatom:Dinoflagellate ratio
Total phytoplankton abundance or biomass per degree square
Abundance (cells.l−1) for the phytoplankton counts is given where it is available, providing more information about the productivity of an area than presence data alone. A low cell abundance may indicate a low level or production, but this may not be the case if these are large cells. The biomass data helps to show productivity of an area. The biovolume has been calculated for each cell count and when converted to biomass, is available for use by modellers and to assist in interpretation of an area’s productivity. An accepted method of converting biovolume to biomass is to assume that the cell has the density of water (1 mm3.l−1=1 mg.l−1)13. Another useful conversion is to carbon biomass; full methods are readily found in the literature24–26. Table 2 gives details conversions of phytoplankton size data to carbon biomass.
Table 2. Information for converting biovolume V (μm3) to carbon biomass B (pgC.cell−1).
Some of the data records are missing dates or coordinates. It was considered useful to keep these records as the presence of a taxa in a location may still be of value. The user may estimate coordinates from the location given and would then also be aware that these would not be the exact coordinates of the sample.
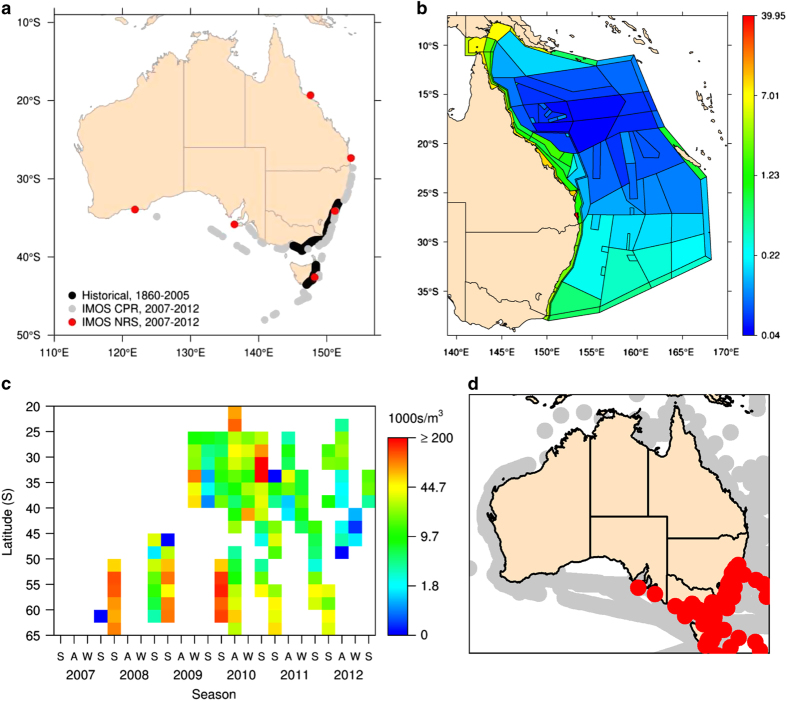
Data can be analysed in many different ways and in many software applications (e.g., R, Matlab). We include here some figures created in The R Project for Statistical Computing (https://www.r-project.org) to demonstrate some potential uses of the data (Fig. 3).
Figure 3. Demonstrated uses of phytoplankton data in the Australian Phytoplankton Database.
(a) Map showing the range extension of Noctiluca scintillans over the past 150 years28; (b) Parameterisation of an ecosystem model (Atlantis) of diatom abundance using satellite chlorophyll Phytoplankton Functional Type analysis and the Continuous Plankton Recorder species ratios; (c) Diatom diversity by latitude from the Continuous Plankton Recorder; (d) Distribution of potentially harmful algal bloom species Dinophysis tripos.
In some cases, notably project 599, the IMOS National Reference Stations, the phytoplankton component of the survey is only a part of the data available. Additional biogeochemical data are available for this data set via the AODN. Some of the projects, viz. P479, P599, P597, have corresponding zooplankton data freely available in The Australian Zooplankton Database27. These data sets can be matched by the project_id and the sample_id which are consistent across databases. The list of citations referenced in Table 1 (available online only) will also give users information as to how this data has been previously used from the discrete projects.
Additional information
How to cite this article: Davies, C. H. et al. A database of marine phytoplankton abundance, biomass and species composition in Australian waters. Sci. Data 3:160043 doi: 10.1038/sdata.2016.43 (2016).
Supplementary Material
Acknowledgments
We acknowledge the contributions from all collaborators and their institutions. If data from multiple projects are used, please acknowledge this publication; if individual project data are used, please acknowledge use of data as per the custodian information. We would also encourage the data user to enter into collaboration with the researchers involved in the data they use—understanding the history of a project will add value to the research. The relevant acknowledgement data is available from the metadata files attached to the data.
If using data from Project 599 the National Reference Stations or Project 597 the Australian Continuous Plankton Recorder, please use the following acknowledgement:
‘Data sourced from the Integrated Marine Observing System (IMOS)—IMOS is a national collaborative research infrastructure, supported by the Australian Government.’
Similarly for Projects 806 and 1066, please use the following acknowledgement:
‘A part of the data included in the database was obtained with support from the Great Barrier Reef Marine Park Authority, through funding from the Australian Government Reef Program and from the Australian Institute of Marine Science’.
Footnotes
The authors declare no competing financial interests.
Data Citations
- Davies C. H. 2016. Australian Ocean Data Network. http://dx.doi.org/10.4225/69/56454b2ba2f79
References
- Field C. B., Behrenfeld M. J., Randerson J. T. & Falkowski P. Primary production of the biosphere: Integrating terrestrial and oceanic components. Science 281, 237–240 (1998). [DOI] [PubMed] [Google Scholar]
- Poloczanska E. S. et al. Global imprint of climate change on marine life. Nat. Clim. Chang 3, 919–925 (2013). [Google Scholar]
- Beaugrand G. Decadal changes in climate and ecosystems in the North Atlantic Ocean and adjacent seas. Deep Sea Res. (II Top. Stud. Oceanogr.) 56, 656–673 (2009). [Google Scholar]
- Hallegraeff G. M. et al. Algae of Australia: Phytoplankton of temperate coastal waters (ABRS, Canberra & CSIRO publishing, 2010). [Google Scholar]
- McCarthy P. M. Census of Australian Marine Dinoflagellates. http://www.anbg.gov.au/abrs/Dinoflagellates/index_Dino.html (2013).
- McCarthy P. M. Census of Australian Marine Diatoms. http://www.anbg.gov.au/abrs/Marine_Diatoms/index.html (2013). [Google Scholar]
- Jeffrey S. W. & Hallegraeff G. M. Phytoplankton ecology of Australian waters 310–348 (Longman, 1990). [Google Scholar]
- Buchanan P. J., Swadling K. M., Eriksen R. S. & Wild-Allen K. New evidence links changing shelf phytoplankton communities to boundary currents in southeast Tasmania. Rev. Fish Biol. Fish 24, 427–442 (2014). [Google Scholar]
- Armbrecht L. H., Schaeffer A., Roughan M. & Armand L. K. Interactions between seasonality and oceanic forcing drive the phytoplankton variability in the tropical-temperate transition zone (similar to 30 degrees S) of Eastern Australia. J. Mar. Syst 144, 92–106 (2015). [Google Scholar]
- Armbrecht L. H. et al. Phytoplankton composition under contrasting oceanographic conditions: Upwelling and downwelling (Eastern Australia). Cont. Shelf Res. 75, 54–67 (2014). [Google Scholar]
- Hallegraeff G. M. Ocean climate change, phytoplankton community responses, and harmful algal blooms: A formidable predictive challenge. J. Phycol. 46, 220–235 (2010). [Google Scholar]
- Brown C. J. et al. Effects of climate-driven primary production change on marine food webs: implications for fisheries and conservation. Global Change Biol 16, 1194–1212 (2010). [Google Scholar]
- Suthers I. & Rissik D. Plankton: a guide to their ecology and monitoring for water quality (CSIRO Publishing, 2009). [Google Scholar]
- Richardson A. J. et al. Using continuous plankton recorder data. Prog. Oceanogr. 68, 27–74 (2006). [Google Scholar]
- Sournia A. Phytoplankton manual (United Nations Educational, Scientific and Cultural Organization, 1978). [Google Scholar]
- Trull T. W. et al. The Australian Integrated Marine Observing System Southern Ocean Time Series facility. in OCEANS 2010. (Sydney, NSW, Australia, 2010). [Google Scholar]
- Williams O. J., Beckett R. E. & Maxwell D. L. Marine phytoplankton preservation with Lugol’s: a comparison of solutions. J. Appl. Phycol. 28, 1705–1712 (2016). [Google Scholar]
- Thompson P. A. et al. Contrasting oceanographic conditions and phytoplankton communities on the east and west coasts of Australia. Deep Sea Res. (II Top. Stud. Oceanogr.) 58, 645–663 (2011). [Google Scholar]
- Hillebrand H., Durselen C. D., Kirschtel D., Pollingher U. & Zohary T. Biovolume calculation for pelagic and benthic microalgae. J. Phycol. 35, 403–424 (1999). [Google Scholar]
- Scott F. J. & Marchant H. J. Antarctic Marine Protists 572 (Australian Biological Resources Study, 2005). [Google Scholar]
- Tomas C. R. Idenitfying marine phytoplankton 858 (Academic Press, 1997). [Google Scholar]
- Olenina I. et al. Biovolumes and size-classes of phytoplankton in the Baltic Sea 144 (Baltic Marine Environment Protection Commission, 2006). [Google Scholar]
- Leblanc K. et al. A global diatom database—abundance, biovolume and biomass in the world ocean. Earth System Science Data 4, 149–165 (2012). [Google Scholar]
- Menden-Deuer S. & Lessard E. J. Carbon to volume relationships for dinoflagellates, diatoms, and other protist plankton. Limnol. Oceanogr. 45, 569–579 (2000). [Google Scholar]
- Verity P. G. & Langdon C. Relationships between lorica volume, carbon, nitrogen and ATP content of Tintinnids in Narragansett Bay. J. Plankton Res. 6, 859–868 (1984). [Google Scholar]
- Vogt M. et al. Global marine plankton functional type biomass distributions: Phaeocystis spp. Earth System Science Data 4, 107–120 (2012). [Google Scholar]
- Davies C. H. et al. Over 75 years of zooplankton data from Australia. Ecology 95, 3229–3229 (2014). [Google Scholar]
- McLeod D. J., Hallegraeff G. M., Hosie G. W. & Richardson A. J. Climate-driven range expansion of the red-tide dinoflagellate Noctiluca scintillans into the Southern Ocean. J. Plankton Res. 34, 332–337 (2012). [Google Scholar]
- Farrell H., Brett S., Ajani P. & Murray S. Distribution of the genus Alexandrium (Halim) and paralytic shellfish toxins along the coastline of New South Wales, Australia. Mar. Pollut. Bull. 72, 133–145 (2013). [DOI] [PubMed] [Google Scholar]
- Ajani P. et al. The risk of harmful algal blooms (HABs) in the oyster-growing estuaries of New South Wales, Australia. Environ. Monit. Assess. 185, 5295–5316 (2013). [DOI] [PubMed] [Google Scholar]
- Lynch T. P. et al. in Oceans 2008, Vols 1-4 Oceans-Ieee 367–374 (Ieee, 2008). [Google Scholar]
- Armbrecht L. H. et al. Comparison of the cross-shelf phytoplankton distribution of two oceanographically distinct regions off Australia. J. Mar. Syst 148, 26–38 (2015). [Google Scholar]
- Burford M. A., Rothlisberg P. C. & Wang Y. G. Spatial and temporal distribution of tropical phytoplankton species and biomass in the Gulf of Carpentaria, Australia. Mar. Ecol. Prog. Ser. 118, 255–266 (1995). [Google Scholar]
- Burford M. A. & Rothlisberg P. C. Factors limiting phytoplankton production in a tropical continental shelf ecosystem. Estuar Coast Shelf S 48, 541–549 (1999). [Google Scholar]
- Burford M. A., Rothlisberg P. C. & Revill A. T. Sources of nutrients driving production in the Gulf of Carpentaria, Australia: a shallow tropical shelf system Mar. Freshwater Res. 60, 1044–1053 (2009). [Google Scholar]
- IOC. The Ocean Biogeographic Information System. http://www.iobis.org (2014).
- Ajani P. A., Allen A. P., Ingleton T. & Armand L. Erratum. A decadal decline in relative abundance and a shift in microphytoplankton composition at a long-term coastal station off southeast Australia. Limnol. Oceanogr 59, 2240–2242 (2014). [Google Scholar]
- Ajani P. A., Allen A. P., Ingleton T. & Armand L. A decadal decline in relative abundance and a shift in microphytoplankton composition at a long-term coastal station off southeast Australia. Limnol. Oceanogr. 59, 519–531 (2014). [Google Scholar]
- Wood E. J. F. Dinoflagellates in the Australian region. Aust. J. Mar. Freshw. Res 5, 171–351 (1954). [Google Scholar]
- Crosby L. H. & Wood E. J. F. Studies on Australian and New Zealand diatoms. I. Planktonic and allied species. Trans. R. Soc. N.Z 85, 483–530 (1958). [Google Scholar]
- Gillespie N. C., Holmes M. J., Burke J. B. & Doley J. Distribution and periodicity of Gambierdiscus toxicus in Queensland, Australia (1985).
- Raes E. J. et al. Changes in latitude and dominant diazotrophic community alter N-2 fixation. Mar. Ecol. Prog. Ser. 516, 85–102 (2014). [Google Scholar]
- Raes E. J. et al. Sources of new nitrogen in the Indian Ocean. Global Biogeochem. Cy. 29, 1283–1297 (2016). [Google Scholar]
- Griffin D. A., Thompson P. A., Bax N. J., Bradford R. W. & Hallegraeff G. M. The 1995 mass mortality of pilchard: No role found for physical or biological oceanographic factors in Australia. Mar. Freshwater Res. 48, 27–42 (1997). [Google Scholar]
- Dela-Cruz J., Middleton J. H. & Suthers I. M. The influence of upwelling, coastal currents and water temperature on the distribution of the red tide dinoflagellate, Noctiluca scintillans, along the east coast of Australia. Hydrobiologia 598, 59–75 (2008). [Google Scholar]
- Rothlisberg P. C. et al. Phytoplankton community structure and productivity in relation to the hydrological regime of the Gulf of Carpentaria, Australia, in summer. Aust. J. Mar. Freshw. Res 45, 265–282 (1994). [Google Scholar]
- Jeffrey S. W. & Carpenter S. M. Seasonal succession of phytoplankton at a coastal station off Sydney. Aust. J. Mar. Freshwater Res 25, 361–369 (1974). [Google Scholar]
- Revelante N., Williams W. T. & Bunt J. S. Temporal and spatial distribution of diatoms, dinoflagellates and Trichodesmium in waters of the Great Barrier Reef. J. Exp. Mar. Biol. Ecol 63, 27–45 (1982). [Google Scholar]
- Hallegraeff G. M. & Jeffrey S. W. Tropical phytoplankton species and pigments of continental shelf waters of north and northwest Australia. Mar. Ecol. Prog. Ser. 20, 59–74 (1984). [Google Scholar]
- Nayar S., Collings G., Pfennig P. & Royal M. Managing nitrogen inputs into seagrass meadows near a coastal city: Flow-on from research to environmental improvement plans. Mar. Pollut. Bull. 64, 932–940 (2012). [DOI] [PubMed] [Google Scholar]
- Nayar S., Collings G. J., Miller D. J., Bryars S. & Cheshire A. C. Uptake and resource allocation of inorganic carbon by the temperate seagrasses Posidonia and Amphibolis. J. Exp. Mar. Biol. Ecol 373, 87–95 (2009). [DOI] [PubMed] [Google Scholar]
- Nayar S., Collings G. J., Miller D. J. & Bryars S. & Cheshire, A. C. Uptake and resource allocation of ammonium and nitrate in temperate seagrasses Posidonia and Amphibolis. Mar. Pollut. Bull. 60, 1502–1511 (2010). [DOI] [PubMed] [Google Scholar]
- Wood E. J. F. Studies on Australian and New Zealand diatoms. IV. Descriptions of further sedentary species. Trans. R. Soc. N.Z 88, 669–698 (1961). [Google Scholar]
- Wood E. J. F., Crosby L. H. & Cassie V. Studies on Australian and New Zealand diatoms. III. Descriptions of further discoid species. Trans. R. Soc. N.Z 87, 211–219 (1959). [Google Scholar]
- Hallegraeff G. M. Coccolithophorids (Calcareous nanoplankton) from Australian waters. Bot. Mar. 27, 229–247 (1984). [Google Scholar]
- Hallegraeff G. M. & Lucas I. A. N. The marine dinoflagellate genus Dinophysis (Dinophyceae): photosynthetic, neritic and non-photosynthetic, oceanic species. Phycologia 27, 25–42 (1988). [Google Scholar]
- Jeffrey S. W. & Hallegraeff G. M. Studies of phytoplankton species and photosynthetic pigments in a warm core eddy of the East Australian Current. I. Summer populations. Mar. Ecol. Prog. Ser. 3, 285–294 (1980). [Google Scholar]
- Nayar S. & Bott K. Uptake and translocation of ammonium and nitrate by temperate seagrass Zostera nigricaulis in Port Phillip Bay 51 (SARDI, 2015). [Google Scholar]
- Wood E. J. F. Dinoflagellates in the Australian region. III. Further collections. Technical Papers Division of Fisheries Australia 17, 1–20 (1963). [Google Scholar]
- LeRoi J. M. & Hallegraeff G. M. Scale-bearing nanoflagellates from southern Tasmanian coastal waters, Australia. I. Species of the genus Chrysochromulina (Haptophyta). Bot. Mar. 47, 73–102 (2004). [Google Scholar]
- McMinn A. Recent dinoflagellate cyst distribution in eastern Australia. Rev. Palaeobot. Palynol. 65, 305–310 (1990). [Google Scholar]
- Hallegraeff G. M. & Reid D. D. Phytoplankton species successions and their hydrological environment at a coastal station off Sydney. Aust. J. Mar. Freshw. Res 37, 361–377 (1986). [Google Scholar]
- Hallegraeff G. M. & Jeffrey S. W. Annually recurrent diatom blooms in spring along the New South Wales coast of Australia. Aust. J. Mar. Freshw. Res 44, 325–334 (1993). [Google Scholar]
- Revelante N. & Gilmartin M. Dynamics of phytoplankton in the Great Barrier Reef Lagoon. J. Plankton Res. 4, 47–76 (1982). [Google Scholar]
- Hallegraeff G. M. Scale-bearing and loricate nanoplankton from the East Australian Current. Bot. Mar. 26, 493–515 (1983). [Google Scholar]
- Grant B. R. & Kerr J. D. Phytoplankton numbers and species at Port Hacking station and their relationship to physical environment. Aust. J. Mar. Freshw. Res. 21, 35-& (1970). [Google Scholar]
- Cummins S. P., Roberts D. E., Ajani P. & Underwood A. J. Comparisons of assemblages of phytoplankton between open water and seagrass habitats in a shallow coastal lagoon. Mar. Freshwater Res. 55, 447–456 (2004). [Google Scholar]
- Gottschalk S., Uthicke S. & Heimann K. Benthic diatom community composition in three regions of the Great Barrier Reef, Australia. Coral Reefs 26, 345–357 (2007). [Google Scholar]
- Hallegraeff G. M. Seasonal Study of Phytoplankton Pigments and Species at a Coastal Station off Sydney: Importance of Diatoms and the Nanoplankton. Mar. Biol. 61, 2–3 (1981). [Google Scholar]
- Wood E. J. F. Checklist of diatoms recorded from the Indian Ocean (Cronulla, 1963). [Google Scholar]
- Thompson P. A., Bonham P. I. & Swadling K. M. Phytoplankton blooms in the Huon Estuary, Tasmania: top-down or bottom-up control? J. Plankton Res. 30, 735–753 (2008). [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Citations
- Davies C. H. 2016. Australian Ocean Data Network. http://dx.doi.org/10.4225/69/56454b2ba2f79