Abstract
The DAZ family genes boule, daz and dazl have conserved functions in primordial germ cell (PGC) migration, germ stem cell proliferation, differentiation and meiosis progression. It has remained unknown whether this family is required for PGC formation in developing embryos. Our recent study in the fish medaka (Oryzias latipes) has defined dnd as the critical PGC specifier and predicted the presence of additional factors essential for PGC formation. Here we report that dazl is a second key player for medaka PGC formation. Dazl knockdown did not prevent PGC formation even in the absence of normal somatic structures. It turned out that a high level of Dazl protein was maternally supplied and persisted until gastrulation, and hardly affected by two antisense morpholino oligos targeting the dazl RNA translation. Importantly, microinjection of a Dazl antibody remarkably reduced the number of PGCs and even completely abolished PGC formation without causing detectable somatic abnormality. Therefore, medaka PGC formation requires the Dazl protein as maternal germ plasm component, offering first evidence that dazl is a critical player in PGC formation in vivo. Our results demonstrate that antibody neutralization is a powerful tool to study the roles of maternal protein factors in PGC development in vivo.
In many animals, the germline is established early in development by primordial germ cell (PGC) specification. PGCs migrate into the gonad, and gonadal germ cells in the adult ovary and testis undergo stem cell self-renewal, differentiation, meiosis and post-meiotic morphogenesis, culminating in the production of eggs and sperm1. Defects in any of these processes will lead to reproductive deficiency and infertility. Male infertility represents 40~50% of human infertility and affects one-sixth of couples worldwide2,3. Male infertility is often associated with azoospermia or oligozoospermia as a consequence of genetic alterations4. The DAZ gene family is the best studied that encode infertility factors in animal models5 and human6,7. This family consists of daz, dazl and boule, which encode RNA-binding proteins that act as functional homologs across phyla8,9,10. The founder member Daz is encoded by the human Deleted-in-azoospermia and acts as a critical male fertility factor. Four Daz genes reside on the human Y chromosome. Daz is restricted to primates, its autosomal homolog Daz-like (Dazl) has been described in several vertebrates including the human11,12, mouse13,14, Xenopus15, axolotl16, newt17, zebrafish18 and medaka19. Boule has been considered as the ancestor of the DAZ family and its ortholog has been found in vertebrates5,7. Boule is the only DAZ family member in invertebrates including Drosophila20 and C. elegans21. In the rainbow trout, differential expression of boule and dazl reveals germ cell sex prior to meiosis22,23.
The DAZ family is exclusively required for germ cell development. The functions of the family members are, however, distinct and variable in different organisms. In invertebrates, boule is expressed only in the ovary and required for oogenesis of C. elegans21, but is essential for meiotic cell cycle in spermatogenesis of Drosophila, as male mutants are sterile and their spermatocytes are arrested at the G2/M phase20,24. In vertebrates, dazl depletion in Xenopus leads to defective PGC development8, targeted dazl disruption in mouse results in the sterility in both sexes, with the prime spermatogenic defect being a failure of spermatogonial differentiation as germ cells in the testis are arrested at spermatogonial stage14. Several studies in vitro have revealed a role of the DAZ family members in germ cell fate decision. In mouse ES cells in culture, forced dazl expression promotes germ cell formation25. In human ES cells, Dazl functions also in germ cell formation, whereas Daz and Boule promote later stages of meiosis and development of haploid gametes26. It has remained unknown whether Dazl functions PGC specification in developing embryos.
Diverse animal species make use of two distinct modes for PGC formation, namely preformation and epigenesis27,28. Preformation operates in egg-laying animals such as Drosophila29, C. elegans30 and Xenopus15. In these organisms, the cytoplasmic germ plasm is maternally supplied to the embryo, asymmetrically partitioned to one or few cells to intrinsically determine the PGC fate before or during cleavage divisions. Epigenesis prevails in mammals such as mouse31 and urodelean amphibians such as newt16. In epigenesis, maternal inheritance of germ plasm components is absent, and PGC formation is independent of germ plasm but extrinsically induced by cell-cell interactions during gastrulation31. In fish, PGC preformation has been demonstrated in zebrafish and medaka. In zebrafish, germ plasm components are maternally inherited and asymmetrically segregated into pPGCs during early cleavages32,33,34. In medaka (Oryzias latipes), embryo perturbation does not affect the PGC number, leading to the notion for PGC preformation in this organism35. Unusually, medaka maternal germ plasm components, such as the transcripts of boule and dazl5,19, vasa36,37,38 and piwi9, distribute widely during early development rather than localization into a small number of cells. In addition, knockdown of germ genes such as nanos39, vasa36 or piwi9 can reduce the number of PGCs and affect PGC migration but cannot completely preventing PGC formation. Direct evidence for medaka PGC preformation comes from the observation that associated single cells from midblastula embryos in culture are able to form PGCs in the absence of normal somatic structures and known inducing factors40. Most recently, we have identified dnd as the critical PGC specifier in medaka and predicted the presence of additional factors essential for PGC formation, as dnd overexpression can enhance the PGC number by up to 3 folds, and many dnd-expressing cells adopt somatic cell fates41. This study was aimed at analyzing the role of dazl in medaka PGC development. We show that injection of an anti-Dazl antibody is able to abolish PGC formation in medaka embryos, providing first evidence that maternal Dazl is required for PGC formation in vivo.
Results
Effect of dazl knockdown on PGC development
Transgenic medaka lines Ng and Vg were used for PGC observation, which express GFP from the medaka nanos3 and vasa promoter (olvas-gfp) exclusively in germ cells, respectively36. To trace PGCs specifically by zygotic GFP expression, hybrid embryos (referred NgVg embryos thereafter) between Ng females and Vg males were produced for monitoring PGC development36.
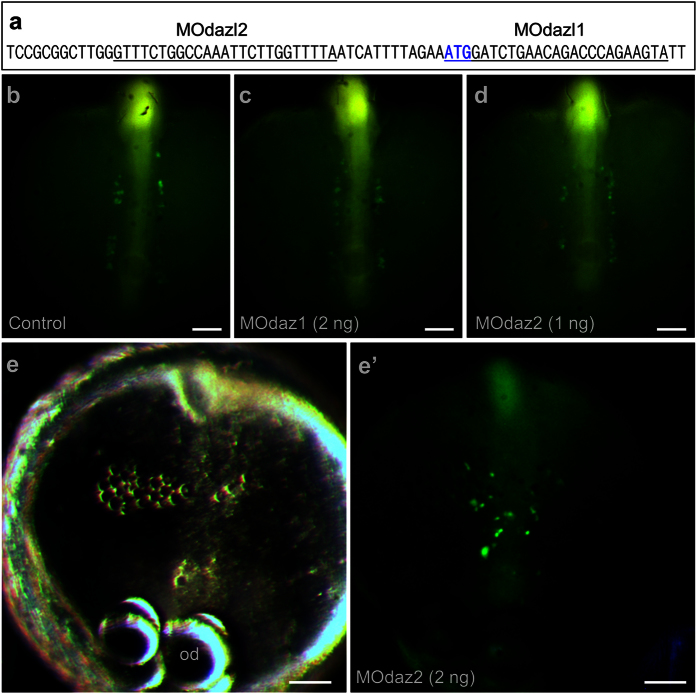
Several experiments have suggested that medaka PGC formation is independent on somatic development9,35,36. In zebrafish, microinjection of antisense morpholino oligos against germ plasm RNA components such as vasa42, nanos43 and dnd44 does not affect PGC formation. In medaka, microinjection of morpholinos against vasa45 and piwi9 affects PGC migration but does not prevent PGC formation. Most recently, we show that dnd acts as the medaka PGC specifier41. We extended our study to dazl for analyzing the role of a maternal factor in medaka PGC formation. The dazl RNA is a maternally supplied germ plasm component in medaka19. In mouse, forced dazl expression promotes germ cell formation from ES cells in culture25. In human, Dazl functions in germ cell formation from ES cells, whereas closely related genes Daz and Boule promote later stages of meiosis and development of haploid gametes26. Two series of experiments were performed. To this end, NgVg embryos at the 2-cell stage were subjected to microinjection of antisense morpholino oligos (MOs) against the medaka dazl. Two MOs were used: MOdaz1 targets the sequence spanning the ATG codon, MOdaz2 recognizes the sequence upstream of the ATG (Fig. 1a). Microinjection of MOdaz1 at 2 ng or MOdaz2 at 1 ng was permissive for normal somatic development and PGC formation (Fig. 1b–d). MO injection at higher doses, namely MOdaz2 at 2 ng prevented somatic development, resulting in a disorganized cell mass that lacked normal embryonic structures. Interestingly, even in these severely disorganized embryos, PGC formation was not prevented (Fig. 1e,e’). Similarly, coinjection of MOdaz1 and MOdaz2 at 1 ng led to abnormal somatic development and seemingly normal PGC formation (Figure S1). In a total of 83 MO-injected embryos, we failed to detect a remarkable reduction in the number of PGCs. As summarized in Table 1, a control embryo after water injection produces 31.8 PGCs at 40 hpf, which is not significantly different from 29.3 of those injected with MOdaz1 and 33.7 of those injected with MOdaz2. Although it is unclear whether abnormal somatic development is due to the toxicity of MOdaz1 and MOdaz2 or an essential role of dazl in somatic development of early medaka embryos, these data demonstrate that medaka PGCs can form in the absence of a normal somatic environment, conforming to the preformation mode in this organism.
Figure 1. dazl knockdown has little effect on PGC formation.
NgVg embryos were injected at the 1-cell stage and analyzed microscopically at stage 22 for PGCs (green). (a) Positions of MOdaz1 and MOdaz2. The target sequences on the medaka dazl cDNA are underlined. The ATG codon is shown in bold. (b–d) Normal somatic development and PGC formation after injection with water (b), MOdaz1 (c) and MOdaz2 (d). (e,e’) Abnormal somatic development and normal PGC formation after morpholino injection. The anterior is to the top. od, oil droplet. Scale bars, 100 μm.
Table 1. Effect of dazl morpholinos on the PGC number1.
| Injection | dose | Number of embryos observed | Number of PGCs per embryo2 |
|---|---|---|---|
| water | 52 | 31.8 ± 5.5 | |
| MOdaz1 | 2 ng | 47 | 29.3 ± 7.4 |
| MOdaz2 | 1 ng | 36 | 33.7 ± 8.0 |
1NgVg embryos were injected at the 1-cell stage. PGCs were scored by GFP expression at 40 hpf.
2Data are mean ± s.d. No significant difference was observed between water injection control and experimental groups injected with MOdaz1 or MOdaz2.
Embryonic Dazl protein expression
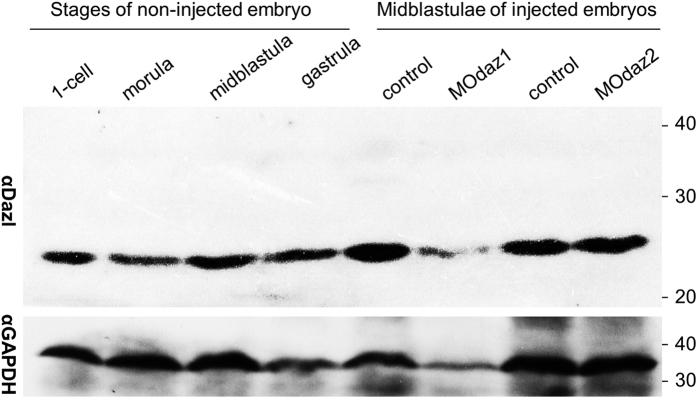
Injection of dazl MOs affects the soma but not PGC formation in medaka embryos is unusual, because dazl is sufficient to promote germ cell formation from mammalian ES cells25,26. Since MOs act through the inhibition of translation, we performed a Western analysis on Dazl protein expression in developing medaka embryos by using αDazl, a polyclonal anti-Dazl antiserum capable of specifically staining medaka germ cells in the adult testis and ovary19. The Dazl protein was seen at a high level already in 1-cell embryos and until gastrulation, and this level was not reduced significantly by MOdaz1 or MOdaz2 (Fig. 2). Therefore, the Dazl protein in medaka is maternally supplied at a high level and persists until gastrulation when PGC formation occurs, and it is not surprising that dazl MOs are inefficient to reduce the Dazl level and thus unable to exhibit effect on PGC development in this organism.
Figure 2. Western blot analysis of Dazl protein expression.
Crude protein extract from three embryos was used for each lane. GAPDH was detected as a loading control. Size markers in kilodalton are shown to the right. MOdaz1 and MOdaz2 were injected at 2 ng and 1 ng to the1-cell embryos, respectively.
Medaka PGC development requires maternal Dazl
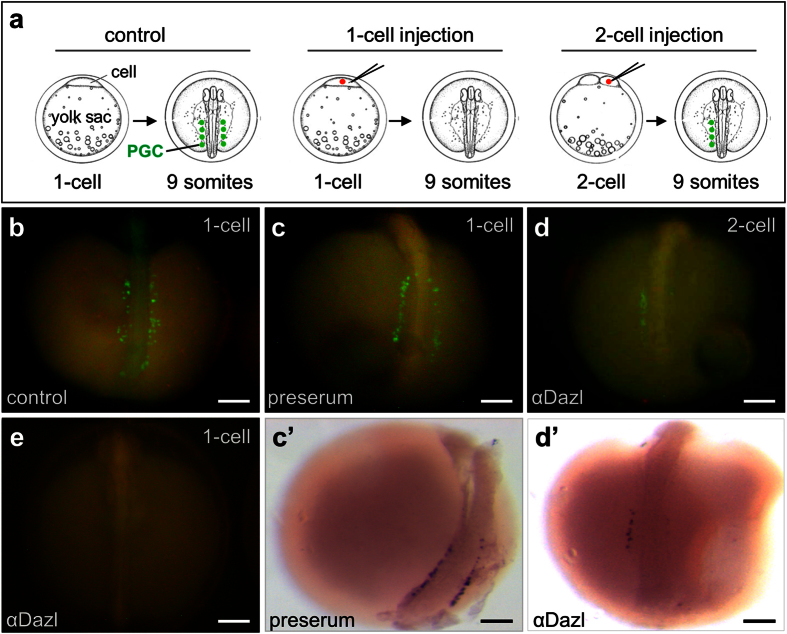
In Xenopus, injection of an anti-Vasa antibody perturbed the function resulted in failure of PGC differentiation at the tadpole stage46. The inefficiency of dazl MOs in reducing the Dazl protein level due to an abundant maternal supply provoked αDazl injection to neutralize the Dazl activity. To this end, NgVg embryos at the 2-cell stage were injected with αDazl or preserum as a control. When injected at high doses (5~10 ng per embryo), either antibody produced abnormal embryos. Upon injection with either antibody at 3 ng per embryo, the majority of embryos appeared normal. As expected, injection of water and a preserum did not affect PGC formation (Fig. 3a,b), and produced an average of 33.5 PGCs among 25 embryos at 2 dpf. A total of 52 embryos at the 1-cell stage were injected with αDazl, 43 survivors at 2 dpf exhibited seemingly normal development and were analyzed for PGC development. This revealed that the average PGC number decreased by 30.5% to 23.3 (Table 2). The inhibitory effect of αDazl on the PGC number became more evident when PGCs were examined for bilaterally asymmetric distribution. In controls, averages of PGCs were 14.5 and 19.0 at the left and right sides, respectively. These values became 7.9 and 15.4 in αDazl-injected embryos, giving rise to a reduction by 45.5% and 18.9%, respectively (Table 2). Upon αDazl injection at the 2-cell stage, the absence of PGCs on the side from the injected blastomeres was seen in certain Vg embryos (Fig. 3c). Most convincingly, two of the 43 Vg embryos injected with αDazl at the 1-cell stage were found to be completely free of PGCs (Fig. 3d). In situ hybridization by using an antisense dazl riboprobe revealed the presence of ~34 PGCs in preserum-injected control embryo (Fig. 3b’) but only 9 PGCs upon αDazl injection (Fig. 3c’). Collectively, medaka PGC formation requires the maternal Dazl protein.
Figure 3. Dazl is required for medaka PGC formation.
Vg embryos were monitored for PGCs by GFP expression and in situ hybridization at 40 hpf. (a) Schematic microinjection at the 1-cell or 2-cell stage and PGC detection at the 9-somite stage. (b,c) Control Vg embryos, showing many PGCs (green) in two bilateral clusters along the embryo axis without (b) or with preserum injection. (d) Embryo after αDazl injection at the 2-cell stage, showing the absence of PGCs in the right side from the injected cell. (e) Embryo after αDazl injection at the 1-cell stage, showing the absence of PGCs. (c’,d’) Embryos shown in (c,d) after in situ hybridization with an antisense dazl riboprobe, showing the presence of ~34 PGCs on both sides (c’) and only 9 PGCs in the left side from the non-injected cell (d’). 1-cell, microinjection at the 1-cell stage; 2-cell, microinjection into one of the 2 cells at the 2-cell stage. Scale bars, 100 μm.
Table 2. Dazl depletion blocks PGC formation1.
| Serum injected | Number of embryos | Number of PGCs | ||
|---|---|---|---|---|
| Total | Side 12 | Side 22 | ||
| Preserum | 25 | 33.5 ± 5.3 | 14.5 ± 3.2 | 19.0 ± 3.1 |
| αDazl | 43 | 23.3 ± 6.4 | 7.9 ± 3.5 | 15.4 ± 4.6 |
1Preserum or αDazl was injected with 1.5 ng per NgVg embryo into one of the two cells at the 2-cell stage, and PGCs were scored by GFP expression at 40 hpf.
2Side 1 is the injected side, which was labeled by a co-injected fluorescent dye, and side 2 is the noninjected side. Significant difference was observed between preserum injection and αDazl injection in the injected side but not noninjected side.
Discussion
The transcripts and protein products of germ genes are often germ plasm components that are maternally supplied in many egg-laying organisms. In zebrafish, maternal RNA inheritance has been known for vasa32,34, nanos43, dnd44, zili47 and ziwi48, and microinjection of antisense morpholino oligos against some of them including nanos43 and dnd44 leads to abnormal PGC development, ranging from a reduced PGC number over defective PGC migration to survival. In medaka, maternal RNA inheritance has been reported for vasa and piwi, and microinjection of their antisense morpholino oligos results in a reduced PGC number and defective PGC migration9,36. Interestingly, accumulated data in fish show that antisense morpholino oligos of germ genes cannot completely prevent PGC formation but merely affect subsequent steps of PGC development, compared to their requirement for PGC formation as illustrated by vasa loss-of-function mutations in Drosophila46. A difference in phenotype between morpholino-mediated translation inhibition and loss-of-function mutations has been ascribed to a high level of maternal protein supply in combination with incomplete translation inhibition36. In this study, we provide first evidence in medaka that the Dazl protein is indeed maternally supplied at a high level and persists until gastrulation when PGC formation occurs. Consequently, dazl morpholino oligos have little effect on the Dazl protein level and thus on PGC development. We demonstrate that αDazl injection is sufficient to remarkably reduce the PGC number and even to abolish PGC formation in certain cases, perhaps via neutralizing the activity of Dazl protein. Our data suggest that antibody injection offers an alternative tool to study the earliest event of PGC development, namely PGC formation in fish, as has been reported in Xenopus49.
In this study, we have revealed that αDazl injection leads to severe reduction in the PGC number and even a complete loss of PGCs, demonstrating that dazl plays an essential role in PGC formation. The PGC absence may be due to the absence of PGC formation or disappearance of PGCs by death prior to observation. Three observations favor the absence of PGC formation in certain αDazl-injected embryos. First, zebrafish PGC survival requires the function of nanos43 and dnd44, and PGCs are visible by transient GFP expression during somitogenesis and begin to die afterwards upon nanos or dnd knockdown. Second, vasa or piwi knockdown in medaka does not affect PGC survival even at ectopic sites of advanced embryos or in culture9,36. Finally, GFP or RFP is fairly stable and its fluorescence can persist in dead cells for 3 days, as illustrated by cell culture in the presence of puromycin40. Medaka PGCs are visible by transient GFP expression until 13 hpf39, and PGC observation in this study has been made from 40 hpf onwards. Well-specified PGCs, either live or dead, should be identifiable by GFP expression, suggesting that observation in medaka at 40 hpf is able to detect the majority–if not all–of previously formed PGCs. The fact that αDazl injection leads to a reduction or even loss of PGCs suggests a role for dazl in medaka PGC formation. Previously, we have shown in medaka that vasa or piwi knockdown reduces the PGC number9,36. Most recently, we have identified dnd as the critical PGC specifier and predicted the presence of additional factors in medaka PGC formation41. Results in this study reveals dazl as the second key player in PGC formation. In mammals, forced dazl expression in vitro promotes germ cell formation from ES cells of mouse25 and human26. Hence, dazl plays a conserved role for PGC development from fish to mammals.
Materials and Methods
Animals
Work with animals was carried out in strict accordance with the recommendations in the Guide for the Care and Use of Laboratory Animals of the National Advisory Committee for Laboratory Animal Research in Singapore and approved by this committee (Permit Number: 27/09). Medaka strains HB32C and af were maintained under an artificial photoperiod of 14-h light to 10-h darkness at 26 °C50,51,52. Transgenic line Vg was described previously51, which expresses GFP from the medaka vasa promoter36. Heterozygous Vg embryos were produced by crossing homozygous Vg males to non-transgenic females and used for microinjection and cell culture. In certain experiments, heterozygous Vg males were crossed with non-transgenic females, and resultant embryos were used for cell cultures.
Morpholino oligos
Morpholino antisense oligos were purchased from Gene Tools (Oregon) and dissolved in water. MOdaz1 (TACTTCTGGGTCTGTTCAGATCCAT) and MOdaz2 (TAAAACCAAGAATTTGGCCAGAAAC) target the medaka dazl RNA (Accession number AY973274), the former spans the initiation codon (underlined), and latter is positioned 12 nt upstream of the initiation codon.
Antibodies
Control preserum and polyclonal anti-Dazl antisera (αDazl) were produced and used as previously described19.
Embryo injections
Embryos were injected at the 1- or 2-cell stages as described36. MOdaz1 and MOdaz2 were dissolved at 0.1~5 mg/ml, which corresponds to 0.1~5 ng per injection. Preserum and αDazl were diluted in water at 1:3 before injection, corresponding to 3 ng protein per embryo as determined by using the BioRad protein assay kit (#500-0006). Successful injection was monitored on the basis of co-injected fluorescent dye Texas red.
In situ hybridization
Embryos were fixed and subjected to in situ hybridization with an antisense dazl riboprobe as described5,19.
Western blot analysis
Homogenates of whole embryos at representative stages were resolved in 10% SDS-PAGE and blotted as described (Xu et al., 2005). The blots were incubated with αDazl or αGAPDH, the latter being a monoclonal mouse antibody against the human glyceraldehyde-3-phosphate dehydrogenase (GAPDH) at a 1:1000 dilution (sc-47724, Santa Cruz Biotechnology, Inc.). After washing, the blots were incubated with secondary antibodies (A0545 or A9044, Sigma) at a 10,000 dilution and visualized by the ECL detection reagents (Pierce, USA).
Microscopy
Microscopy was done as described5,50,53. Briefly, live embryos and fry were visualized using a Leica MZFLIII stereo microscope equipped with a Fluo III UV-light system and a GFP2 filter and photographed by using a Nikon E4500 digital camera (Nikon Corp). For documentation at larger magnification, live embryos and fry were observed and photographed on Zeiss Axiovert2 invert microscope equipped with a Zeiss AxioCam MRc digital camera and AxioVision 4 software.
Statistics
Statistical analyses were calculated by using GraphPad Prism v4.0. Data consolidated were presented as mean ± s.d.
Additional Information
How to cite this article: Li, M. et al. Dazl is a critical player for primordial germ cell formation in medaka. Sci. Rep. 6, 28317; doi: 10.1038/srep28317 (2016).
Supplementary Material
Acknowledgments
We thank Jiaorong Deng for fish breeding, Veronica Wong and Choy Mei Foong for laboratory management. This work was supported by grants to M.L. from the National Natural Science Foundation of China (31372520), Shanghai Pujiang Program (15PJ1403100), Shanghai Universities Young Teachers Training Program (A1-2035-15-0021-04) and the Doctor Startup Fund of Shanghai Ocean University (A2-0302-14-300061) and to Y.H. from the National Research Foundation of Singapore (NRF-CRP7-2010-03).
Footnotes
The authors declare no competing financial interests.
Author Contributions M.L. designed the study. M.L., F.Z., Z.L. and N.H. did research. M.L. and Y.H. wrote the paper.
References
- Wylie C. Germ cells. Cell 96, 165–74 (1999). [DOI] [PubMed] [Google Scholar]
- Brugh V. M. 3rd & Lipshultz L. I. Male factor infertility: evaluation and management. Med Clin North Am 88, 367–85 (2004). [DOI] [PubMed] [Google Scholar]
- Hirsh A. Male subfertility. BMJ 327, 669–72 (2003). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferlin A. et al. Male infertility: role of genetic background. Reprod Biomed Online 14, 734–45 (2007). [DOI] [PubMed] [Google Scholar]
- Xu H., Li Z., Li M., Wang L. & Hong Y. Boule is present in fish and bisexually expressed in adult and embryonic germ cells of medaka. PLoS One 4, e6097 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ma K. et al. A Y chromosome gene family with RNA-binding protein homology: candidates for the azoospermia factor AZF controlling human spermatogenesis. Cell 75, 1287–95 (1993). [DOI] [PubMed] [Google Scholar]
- Xu E. Y., Moore F. L. & Pera R. A. A gene family required for human germ cell development evolved from an ancient meiotic gene conserved in metazoans. Proc Natl Acad Sci USA 98, 7414–9 (2001). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Houston D. W. & King M. L. A critical role for Xdazl, a germ plasm-localized RNA, in the differentiation of primordial germ cells in Xenopus. Development 127, 447–56 (2000). [DOI] [PubMed] [Google Scholar]
- Slee R. et al. A human DAZ transgene confers partial rescue of the mouse Dazl null phenotype. Proc Natl Acad Sci USA 96, 8040–5 (1999). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu E. Y. et al. Human BOULE gene rescues meiotic defects in infertile flies. Hum Mol Genet 12, 169–75 (2003). [DOI] [PubMed] [Google Scholar]
- Shan Z. et al. A SPGY copy homologous to the mouse gene Dazla and the Drosophila gene boule is autosomal and expressed only in the human male gonad. Hum Mol Genet 5, 2005–11 (1996). [DOI] [PubMed] [Google Scholar]
- Yen P. H. Putative biological functions of the DAZ family. Int J Androl 27, 125–9 (2004). [DOI] [PubMed] [Google Scholar]
- Cooke H. J., Lee M., Kerr S. & Ruggiu M. A murine homologue of the human DAZ gene is autosomal and expressed only in male and female gonads. Hum Mol Genet 5, 513–6 (1996). [DOI] [PubMed] [Google Scholar]
- Ruggiu M. et al. The mouse Dazla gene encodes a cytoplasmic protein essential for gametogenesis. Nature 389, 73–7 (1997). [DOI] [PubMed] [Google Scholar]
- Houston D. W., Zhang J., Maines J. Z., Wasserman S. A. & King M. L. A Xenopus DAZ-like gene encodes an RNA component of germ plasm and is a functional homologue of Drosophila boule. Development 125, 171–80 (1998). [DOI] [PubMed] [Google Scholar]
- Johnson A. D., Bachvarova R. F., Drum M. & Masi T. Expression of axolotl DAZL RNA, a marker of germ plasm: widespread maternal RNA and onset of expression in germ cells approaching the gonad. Dev Biol 234, 402–15 (2001). [DOI] [PubMed] [Google Scholar]
- Tamori Y., Iwai T., Mita K. & Wakahara M. Spatio-temporal expression of a DAZ-like gene in the Japanese newt Cynops pyrrhogaster that has no germ plasm. Dev Genes Evol 214, 615–27 (2004). [DOI] [PubMed] [Google Scholar]
- Maegawa S., Yasuda K. & Inoue K. Maternal mRNA localization of zebrafish DAZ-like gene. Mech Dev 81, 223–6 (1999). [DOI] [PubMed] [Google Scholar]
- Xu H., Li M., Gui J. & Hong Y. Cloning and expression of medaka dazl during embryogenesis and gametogenesis. Gene Expr Patterns 7, 332–8 (2007). [DOI] [PubMed] [Google Scholar]
- Eberhart C. G., Maines J. Z. & Wasserman S. A. Meiotic cell cycle requirement for a fly homologue of human Deleted in Azoospermia. Nature 381, 783–5 (1996). [DOI] [PubMed] [Google Scholar]
- Karashima T., Sugimoto A. & Yamamoto M. Caenorhabditis elegans homologue of the human azoospermia factor DAZ is required for oogenesis but not for spermatogenesis. Development 127, 1069–79 (2000). [DOI] [PubMed] [Google Scholar]
- Li M. et al. Germ cell sex prior to meiosis in the rainbow trout. Protein Cell 2, 48–54 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li M. et al. Differential conservation and divergence of fertility genes boule and dazl in the rainbow trout. PLoS One 6, e15910 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng M. H., Maines J. Z. & Wasserman S. A. Biphasic subcellular localization of the DAZL-related protein boule in Drosophila spermatogenesis. Dev Biol 204, 567–76 (1998). [DOI] [PubMed] [Google Scholar]
- Yu Z. et al. Dazl promotes germ cell differentiation from embryonic stem cells. J Mol Cell Biol 1, 93–103 (2009). [DOI] [PubMed] [Google Scholar]
- Kee K., Angeles V. T., Flores M., Nguyen H. N. & Reijo Pera R. A. Human DAZL, DAZ and BOULE genes modulate primordial germ-cell and haploid gamete formation. Nature 462, 222–5 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Extavour C.a.A., M. Mechanisms of germ cell specification across the metazoans: epigenesis and preformation. Development 130, 5869–84 (2003). [DOI] [PubMed] [Google Scholar]
- Nieuwkoop P.D.a.S., L. A. Primordium Germ Cells in the Invertebrates: from epigenesis to preformation . (Cambridge: Cambridge University Press, 1981). [Google Scholar]
- Ephrussi A. & Lehmann R. Induction of germ cell formation by oskar. Nature 358, 387–92 (1992). [DOI] [PubMed] [Google Scholar]
- Seydoux G. & Strome S. Launching the germline in Caenorhabditis elegans: regulation of gene expression in early germ cells. Development 126, 3275–83 (1999). [DOI] [PubMed] [Google Scholar]
- Tam P. P. & Zhou S. X. The allocation of epiblast cells to ectodermal and germ-line lineages is influenced by the position of the cells in the gastrulating mouse embryo. Dev Biol 178, 124–32 (1996). [DOI] [PubMed] [Google Scholar]
- Knaut H., Pelegri F., Bohmann K., Schwarz H. & Nusslein-Volhard C. Zebrafish vasa RNA but not its protein is a component of the germ plasm and segregates asymmetrically before germline specification. J Cell Biol 149, 875–88 (2000). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raz E. Primordial germ-cell development: the zebrafish perspective. Nat Rev Genet 4, 690–700 (2003). [DOI] [PubMed] [Google Scholar]
- Yoon C., Kawakami K. & Hopkins N. Zebrafish vasa homologue RNA is localized to the cleavage planes of 2- and 4-cell-stage embryos and is expressed in the primordial germ cells. Development 124, 3157–65 (1997). [DOI] [PubMed] [Google Scholar]
- Herpin A. et al. Specification of primordial germ cells in medaka (Oryzias latipes). BMC Dev Biol 7, 3 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li M. et al. Medaka vasa is required for migration but not survival of primordial germ cells. Mech Dev 126, 366–81 (2009). [DOI] [PubMed] [Google Scholar]
- Shinomiya A., Tanaka M., Kobayashi T., Nagahama Y. & Hamaguchi S. The vasa-like gene, olvas, identifies the migration path of primordial germ cells during embryonic body formation stage in the medaka, Oryzias latipes. Dev Growth Differ 42, 317–26 (2000). [DOI] [PubMed] [Google Scholar]
- Tanaka M., Kinoshita M., Kobayashi D. & Nagahama Y. Establishment of medaka (Oryzias latipes) transgenic lines with the expression of green fluorescent protein fluorescence exclusively in germ cells: a useful model to monitor germ cells in a live vertebrate. Proc Natl Acad Sci USA 98, 2544–9 (2001). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kurokawa H. et al. Time-lapse analysis reveals different modes of primordial germ cell migration in the medaka Oryzias latipes. Dev Growth Differ 48, 209–21 (2006). [DOI] [PubMed] [Google Scholar]
- Li Z., Li M., Hong N., Yi M. & Hong Y. Formation and cultivation of medaka primordial germ cells. Cell Tissue Res 357, 71–81 (2014). [DOI] [PubMed] [Google Scholar]
- Hong N. et al. Dnd Is a critical specifier of primordial germ cells in the medaka fish. Stem Cell Reports 6, 411–21 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Braat A. K., van de Water S., Korving J. & Zivkovic D. A zebrafish vasa morphant abolishes vasa protein but does not affect the establishment of the germline. Genesis 30, 183–5 (2001). [DOI] [PubMed] [Google Scholar]
- Koprunner M., Thisse C., Thisse B. & Raz E. A zebrafish nanos-related gene is essential for the development of primordial germ cells. Genes Dev 15, 2877–85 (2001). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weidinger G. et al. dead end, a novel vertebrate germ plasm component, is required for zebrafish primordial germ cell migration and survival. Curr Biol 13, 1429–34 (2003). [DOI] [PubMed] [Google Scholar]
- Liu L. et al. Medaka dead end encodes a cytoplasmic protein and identifies embryonic and adult germ cells. Gene Expr Patterns 9, 541–8 (2009). [DOI] [PubMed] [Google Scholar]
- Lasko P. F. & Ashburner M. The product of the Drosophila gene vasa is very similar to eukaryotic initiation factor-4A. Nature 335, 611–7 (1988). [DOI] [PubMed] [Google Scholar]
- Houwing S., Berezikov E. & Ketting R. F. Zili is required for germ cell differentiation and meiosis in zebrafish. EMBO J 27, 2702–11 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Houwing S. et al. A role for Piwi and piRNAs in germ cell maintenance and transposon silencing in Zebrafish. Cell 129, 69–82 (2007). [DOI] [PubMed] [Google Scholar]
- Lin Y. M. et al. Presence of DAZL transcript and protein in mature human spermatozoa. Fertil Steril 77, 626–9 (2002). [DOI] [PubMed] [Google Scholar]
- Hong N. et al. Interordinal chimera formation between medaka and zebrafish for analyzing stem cell differentiation. Stem Cells Dev 21, 2333–41 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hong N. et al. Accessibility of host cell lineages to medaka stem cells depends on genetic background and irradiation of recipient embryos. Cell Mol Life Sci 67, 1189–1202 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hong Y., Winkler C. & Schartl M. Pluripotency and differentiation of embryonic stem cell lines from the medakafish (Oryzias latipes). Mech Dev 60, 33–44 (1996). [DOI] [PubMed] [Google Scholar]
- Yi M., Hong N. & Hong Y. Generation of medaka fish haploid embryonic stem cells. Science 326, 430–3 (2009). [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.