Abstract
PURPOSE
Broad, hybrid capture-based next-generation sequencing (NGS), as a clinical test, uses less tissue to identify more clinically relevant genomic alterations compared to profiling with multiple non-NGS tests. We set out to determine the frequency of such genomic alterations via this approach in tumors where previous extensive non-NGS testing had not yielded a targetable driver alteration.
METHODS
We enrolled lung adenocarcinoma patients with a ≤15 pack-year smoking history whose tumors previously tested “negative” for alterations in 11 genes (mutations in EGFR, ERBB2, KRAS, NRAS, BRAF, MAP2K1, PIK3CA, and AKT1, and fusions involving ALK, ROS1, and RET) via multiple non-NGS methods. We performed hybridization capture of the coding exons of 287 cancer-related genes and 47 introns of 19 frequently rearranged genes and sequenced these to deep, uniform coverage.
RESULTS
Actionable genomic alterations with a targeted agent based on NCCN guidelines were identified in 26% (8/31: EGFR G719A, BRAF V600E, SOCS5-ALK, CLIP4-ALK, CD74-ROS1, KIF5B-RET [n=2], CCDC6-RET). 7 of these patients either received or are candidates for targeted therapy. Comprehensive genomic profiling using this method also identified a genomic alteration with a targeted agent available on a clinical trial in an additional 39% (12/31).
CONCLUSION
Broad, hybrid capture-based NGS identified actionable genomic alterations in 65% (95% CI 48–82%) of tumors from never or light smokers with lung cancers deemed without targetable genomic alterations by earlier extensive non-NGS testing. These findings support first-line profiling of lung adenocarcinomas using this approach as a more comprehensive and efficient strategy compared to non-NGS testing.
Keywords: next-generation sequencing, lung, adenocarcinoma, molecular profiling
INTRODUCTION
Therapeutic approaches to lung cancers have quickly shifted towards an emphasis on molecularly targeted therapy in genotypic subsets of patients. The discovery of activating mutations in the epidermal growth factor receptor (EGFR) gene in 2003(1–3) and rearrangements involving the anaplastic lymphoma kinase (ALK) gene in 2007(4) ushered in an era where the identification of key oncogenic alterations emerged as the critical determinant of benefit to targeted therapy. Phase III trials have demonstrated that response rate and progression-free survival are improved with targeted therapy in comparison to chemotherapy.(5–7) In the United States, the EGFR tyrosine kinase inhibitors (TKIs) erlotinib and afatinib, and the ALK TKIs crizotinib and ceritinib are approved for patients with EGFR-mutant and ALK-rearranged advanced lung cancers, respectively.(8, 9)
Recent data from The Cancer Genome Atlas (TCGA) revealed that lung cancers rank among the most genomically-complex of tumors among the 12 cancer types studied by the TCGA Pan-Cancer effort.(10, 11) This genomic complexity allows the opportunity to exploit the presence of other molecular alterations as therapeutic targets in patients. Over the last decade alone, the number of lung cancer drivers for which active targeted therapeutics have been identified has steadily risen. In lung adenocarcinomas, these include, beyond EGFR mutations and ALK fusions, mutations in ERBB2 (HER2),(12) BRAF,(13) PIK3CA,(14) and AKT1,(15) recurrent gene fusions involving ROS1,(16) and RET,(17) and MET amplification,(18) with an ever-growing list of other potential candidates. Lung cancers from patients with a never smoking history have a unique molecular profile in comparison to lung cancers from smokers. Tumors from never smokers are characterized by lower overall mutation frequencies and are enriched for targetable drivers such as EGFR mutations, and ALK, ROS1, and RET fusions.(19)
The evolution of molecular diagnostic platforms that permit rapid identification of oncogenic alterations has played a central role in allowing continued expansion of this approach. In the face of few targetable oncogenes, molecular testing previously followed a one driver-one test approach, with the use of Sanger sequencing to detect EGFR mutations and break apart fluorescence in situ hybridization (FISH) to detect ALK fusions. With an ever-expanding number of drivers of interest, multiplex polymerase chain reaction (PCR)-based platforms such as Sequenom (Sequenom, San Diego, CA) and SNaPShot (Applied Biosystems, Foster City, CA) were developed to simultaneously interrogate mutation hotspots in multiple oncogenes.(20) In several larger centers, the pre-NGS approach to diagnostic testing commonly involved one of the latter methodologies in addition to multiplex sizing assays, FISH tests for recurrent gene fusions, and immunohistochemistry to determine overexpression or protein loss. However, from the perspective of the clinician, the clinical laboratories, and the patient, the amount of tissue, effort, and time required to complete such as an algorithm has become less and less feasible.
Next-generation sequencing (NGS) or massively-parallel DNA sequencing represents an important technologic advance in the evolution of molecular diagnostic tools. NGS allows for the simultaneous detection of multiple alterations in relevant cancer genes in a single test,(21) but this ability can depend on the type of target enrichment used, namely hybrid capture or PCR. Most PCR capture-based NGS assays are designed as “hot spot” tests, sequencing predefined areas of oncogenes with known associations with resistance or sensitivity to approved agents. These tests commonly detect base substitutions with high sensitivity, but small insertions and deletions with lower sensitivity. Although providing a substantial advance over single gene or marker analysis, PCR-based NGS assays do not routinely detect copy number changes (amplification or homozygous deletions) or gene fusions.
In contrast, hybrid capture-based NGS assays allow the identification of not only hotspot mutations, but also interrogates the entire coding sequence of oncogenes and tumor suppressor genes and the introns of selected genes involved in gene fusions, and allow assessment of copy number alterations, all from a single formalin fixed paraffin embedded specimen.(22)
With this in mind, we set out to perform a broad, hybrid capture-based NGS assay (FoundationOne) on tumor specimens from patients with lung adenocarcinomas who tested negative for a panel of 11 known drivers via a standard molecular diagnostic algorithm previously used at our institution. Our intent was to define the incremental potential benefit of such an approach to detect previously undiagnosed genomic alterations amenable to targeted therapy in this defined patient population.
MATERIALS AND METHODS
Patient identification and selection
Patients with lung adenocarcinomas harboring no evidence of a genomic alteration based on a focused panel of non-NGS testing who were treated at the Memorial Sloan Kettering Cancer Center (MSK) between 2006 and 2013 were identified. Testing for these alterations was performed under a separate, ongoing, prospective program (the MSK Lung Cancer Mutational Analysis Program or LC-MAP) in patients with pathologically-confirmed lung adenocarcinomas. Tissue and clinical data were collected under a protocol approved by the institutional review board or waiver of authorization.
Non-NGS testing was comprised of a number of tests for known lung cancer alterations in 11 genes (EGFR, ERBB2, KRAS, NRAS, BRAF, MAP2K1, PIK3CA, AKT1, ALK, ROS1, and RET).(14) A multiplex mass-spectrometry-based system (Sequenom) was used to study 91 point mutations in EGFR, ERBB2, KRAS, NRAS, BRAF, MAP2K1, PIK3CA, and AKT1 (Supplementary Table 1). Multiplex sizing assays tested for insertions or deletions in EGFR exons 19 and 20, and ERBB2 exon 20. Three FISH break apart assays were used to screen for gene rearrangements involving ALK, ROS1, and RET.
Patients whose tumors tested negative for the above alterations were eligible if they were never smokers or smoked ≤15 pack-years of cigarettes, had an Eastern Cooperative Oncology Group (ECOG) performance status of 0–1, and stage IIIB/IV disease or early-stage disease with radiographic findings suspicious for recurrence not amenable to local therapy. Sufficient tumor tissue for broad, hybrid capture-based NGS was required. Our intent was to identify patients who were candidates for targeted therapy should an actionable genomic alteration be identified.
Broad, hybrid capture-based NGS assay
Tumor samples were pre-screened at MSK for adequacy as defined by an initial requirement of 10–15 unstained slides of formalin-fixed paraffin-embedded (FFPE) tissue. Tissue was sent to a Clinical Laboratory Improvement Amendments (CLIA)-certified and College of American Pathologists (CAP)-accredited laboratory (Foundation Medicine, Cambridge, MA) for NGS. Additional pathologic review of submitted specimens was performed to determine tissue adequacy (defined as ≥20% tumor nuclei and ≥50 ng of DNA) prior to testing. Macro-dissection to enrich specimens of ≤20% tumor content was performed as warranted. DNA was extracted from unstained FFPE sections and quantified by a Picogreen fluorescence assay.
In samples deemed adequate, 50–100 ng of DNA was used for whole-genome shotgun library construction. Hybridization capture of 4,557 exons of 287 cancer-related genes and 47 introns of 19 genes frequently rearranged in solid tumors was performed. Hybrid-capture libraries were then sequenced to >500x average unique coverage with >100x at >99% of exons using Illumina HiSeq2000 or 2500 sequencers (Supplementary Figure 1 and 2). Sequencing data were processed using a customized analysis pipeline designed to detect multiple classes of genomic alterations including base substitutions, short insertions and deletions, copy number alterations, and genomic rearrangements. Once available, results were released to the treating physician to allow identification of appropriate targeted therapeutics for patients whose tumors harbored an actionable genomic alteration.
RESULTS
Patients
We identified 47 patients with lung adenocarcinomas that harbored no evidence of a genomic alteration via focused non-NGS testing. Of these patients, non-NGS testing with multiple assays resulted in tissue exhaustion in 34% (n=16/47) of cases and a repeat biopsy was either not feasible or declined by the patient.
Testing was successfully performed on tumor from the remaining 31 cases. Clinical and pathologic features are summarized in Table 1. The median age at diagnosis was 60 years (range 29–78) and the majority of patients were never smokers (71%, n=22/31). In 71% of patients (n=22/31), tumor tested was obtained from the same procedure as tumor used for non-NGS testing.
Table 1. Clinicopathologic Features.
The clinicopathologic profile of 31 patients whose tumors successfully underwent broad, hybrid capture-based next-generation sequencing is shown. These tumors previously tested “negative” for alterations in 11 genes (mutations in EGFR, ERBB2, KRAS, NRAS, BRAF, MAP2K1, PIK3CA, and AKT1, and fusions involving ALK, ROS1, and RET) via non-NGS methods
| Clinicopathologic Features (n=31) | |
|---|---|
| Age at diagnosis (years) | median 60 (range 29–78) |
|
| |
| Sex | |
| M | 42% (n=13) |
| F | 58% (n=18) |
|
| |
| Cigarette smoking history | |
| Never | 71% (n=22) |
| ≤15 pack-years | 29% (n=9) |
|
| |
| Pathology | |
| Lung Adenocarcinomas | 100% (n=31) |
|
| |
| Tumor sample source | |
| Primary | 52% (n=16) |
| Metastatic | 48% (n=15) |
| Pleura | 16% (n=5) |
| Lymph node | 16% (n=5) |
| Other | 16% (n=5) |
|
| |
| Procedure to obtain tumor | |
| Core needle | 29% (n=9) |
| Surgical | 71% (n=22) |
| Lobectomy | 19% (n=6) |
| Wedge | 19% (n=6) |
| VATS pleural biopsy | 16% (n=5) |
| Excision of metastatic focus | 16% (n=5) |
|
| |
| Number of procedures to complete both non-NGS and NGS testing | |
| 1 | 16% (n=5) |
| 2 | 36% (n=11) |
| ≥3 | 48% (n=15) |
Most patients (84%, n=26/31) required ≥ 2 tumor biopsies (median 3, range 2–6) to complete testing. Of these patients, 69% (n=18/26) underwent multiple biopsies in order to complete non-NGS testing alone and sufficient tissue remained for the NGS assay. In 31% of cases (n=8/26), non-NGS testing resulted in tissue exhaustion and an additional biopsy was required to complete NGS testing. For each new biopsy, an attempt to run all non-NGS tests was made, including tests which were previously performed on prior tissue. Only 29% (n=5/31) of samples were derived from the initial procedure performed to diagnose lung cancer. Most samples (71%, n=22/31) were obtained from a surgical procedure such as a lobectomy, wedge resection, VATS pleural biopsy, or excision of a metastatic focus.
Genomic Alterations
One or more genomic alterations were uncovered by hybrid capture-based NGS in tumors from 94% (n=29/31) of patients. Across 31 patient samples tested, a total of 96 individual genomic alterations were found, with a median of 3 alterations (range 0–7) per sample. These alterations are summarized by patient sample in Table 2 (including median exon coverage, mutation allele frequency, and copy number) and most commonly involved TP53 (14%, n=13/96), EGFR (7%, n=7/96), MDM2 (5%, n=5/96), KRAS (4%, n=4/96), CDK4 (4%, n=4/96), and SETD2 (4%, n=4/96).
Table 2. Next-generation sequencing (NGS) results.
The results of hybrid capture-based clinical NGS assay in tumors from 31 patients are presented. In cases where the NGS assay identified a genomic alteration that previous non-NGS testing interrogated but did not detect, the potential reasons for non-detection are detailed.
| Stage at Diagnosis |
Age | Sex | Smoking history (pack years) |
Number of Genomic Alterations Detected |
Med exon coverage |
Mutation (allele frequency) |
Fusion | Amplification (copy number) |
Loss | Genomic alteration uncovered by NGS that prior non- NGS testing was poised to detect |
|
|---|---|---|---|---|---|---|---|---|---|---|---|
| Alteration | Potential reason not detected by non- NGS testing |
||||||||||
| IV | 59 | F | 0 | 7 | 754 |
BRCA1 E1011K (5%) DNMT3A R882H (2%) ATR H2277fs*38 (7%) SETD2 R1598* (7%) NF2 splice (8%) |
none |
NFKBIA (10) NKX2-1 (10) |
None | none | N/A |
| IIIA | 57 | F | 4 | 0 | 652 | none | none | none | none | none | N/A |
| IV | 61 | M | 0 | 3 | 815 | none | SOCS5-ALK |
CDK4 (>20) MDM2 (>20) |
none | SOCS5-ALK | ALK FISH clearly negative, possible complex rearrangement |
| IV | 48 | M | 0 | 2 | 695 | SETD2 K1486*fs (29%) | CCDC6-RET | none | none | CCDC6-RET | RET FISH failure, after fusion detected by NGS, alternate FISH protocol performed and RET fusion was confirmed |
| IV | 53 | M | 0 | 3 | 1077 | TP53 V197G (10%) | none |
NFKBIA (10) NKX2-1 (10) |
none | none | N/A |
| IV | 51 | M | 0 | 1 | 600 | KRAS Q61H (13%) | none | none | none | KRAS Q61H | potentially secondary to lower sensitivity of Sequenom |
| IB | 34 | F | 0 | 3 | 860 | none | SHC1-ERBB2 | MCL1 (9) | none | none | N/A |
| IIB | 57 | F | 0 | 1 | 962 | LRP1B R790* (50%) | none | none | none | none | N/A |
| IA | 60 | M | 0 | 3 | 917 |
TP53 Y220C (33%) EGFR exon20 ins V769_D770insGGTR (33%) |
none | MYC (8) | none |
EGFR exon20 ins V769_D770 insGGTR |
potentially secondary to lower sensitivity of EGFR sizing assay |
| IIIA | 52 | F | 0 | 6 | 809 | none | BRCA2 rearrangement |
MDM2 (11) MDM4 (8) AKT3 (8) IKBKE (8) |
CDKN2A | none | N/A |
| IIA | 66 | F | 2 | 6 | 802 |
BCORL1 P380L (59%) CTNNB1 T41A (1%) FGFR1 T141R (52%) TP53 E198* (15%) |
none |
NFKBIA (8) NKX2-1 (8) |
none | none | N/A |
| IV | 48 | F | 0 | 4 | 1017 |
APC E1309fs*4 (46%) EGFR L747P (50%) |
none | None |
PTEN SMAD4 |
none | Note: EGFR L747P was a double mutation (c.2239_2240TT>CC) that was not interrogated by Sequenom |
| IV | 78 | F | 0 | 1 | 842 | ARID1A S303fs*50 (10%) | none | None | none | none | N/A |
| IV | 63 | F | 10 | 1 | 616 | TP53 P64fs*84 (5%) | none | none | none | none | N/A |
| IIIA | 64 | F | 0 | 1 | 672 |
EGFR exon 18 E709_T710>D (17%) |
none | none | none | none | N/A |
| IB | 77 | F | 0 | 1 | 760 | none | CD74-ROS1 | none | none | CD74-ROS1 | negative via ROS1 FISH, poor hybridization noted in some areas |
| IIIA | 45 | F | 4 | 0 | 488 | none | none | none | none | none | N/A |
| IV | 75 | M | 0 | 1 | 562 | BRAF V600E (1%) | none | none | none | BRAF V600E | potentially secondary to lower sensitivity of Sequenom: a low frequency of the alteration was detected via NGS |
| IIA | 55 | M | 2 | 4 | 729 |
TP53 S127F (7%) TBX3 S615A (47%) RB1 Y321* (9%) |
KIF5B-RET | none | none | KIF5B-RET | negative via RET FISH at MSK and outside institution, initially identified as complex RET fusion by NGS and later determined to be KIF5B-RET after RNAseq testing |
| IV | 70 | M | 0 | 6 | 580 | PMS2 V415M (41%) | ADAM2-PRKDC |
CDK4 (8) MDM2 (16) IGF1R (10) EGFR (16) |
none | none | N/A |
| IV | 43 | M | 2 | 2 | 659 |
CSF1R V32G (53%) FAT3 R3910L (2%) |
none | none | none | none | N/A |
| IV | 76 | M | 0 | 5 | 638 |
NF1 Q83fs*2 (7%) SETD2 S2117FS*10 (23%) SETD2 S918fs*1 (18%) |
none |
KRAS (6) MDM2 (7) |
none | none | N/A |
| IV | 73 | F | 0 | 5 | 685 |
EP300 P925T (62%) TP53 K132N (20%) FLT3 E765* (7%) |
none |
EPHA3 (7) MYC (7) |
none | none | N/A |
| IB | 29 | F | 0 | 6 | 312 |
CDKN2A R80* (39%) ARID2 R80fs*10 (12%) |
CLIP4-ALK |
CCND1 (>20) FGF19 (>20) FGF4 (>20) |
None | CLIP4-ALK | ALK FISH clearly negative, however, CLIP4 and ALK are only 35kb apart on chromosome 2p, rearrangement may have been formed by cryptic inversion that FISH would be unable to detect |
| IIIA | 62 | F | 0 | 3 | 914 |
KIT R956Q (46%) TP53 A159fs*19 (9%) |
KIF5B-RET | none | none | KIF5B-RET | RET FISH clearly negative; however, performed on tumor sample from a separate biopsy from that subjected to NGS testing |
| IIA | 71 | F | 0 | 4 | 536 |
EGFR exon 18 E709_T710>D (31%) TP53 P219fs*10 (13%) |
none |
KRAS (6) CCNE1 (14) |
none | none | N/A |
| IV | 41 | F | 2 | 6 | 851 |
EGFR G719A (36%) EGFR L707W (35%) PTCH1 T1064M (51%) TP53 splice (25%) |
none | none |
CDKN2A CDKN2B |
EGFR G719A | found on the same allele as concurrent EGFR L707W mutation, likely interfering with hybridization of Sequenom extension primer |
| IV | 44 | F | 12 | 3 | 706 |
STK11 H168R (10%) TP53 Y234C (9%) TP53 splice (6%) |
none | none | none | none | N/A |
| IV | 62 | M | 0 | 2 | 747 | none | none |
CDK4 (14) MDM2 (>20) |
none | none | N/A |
| IV | 78 | M | 15 | 3 | 745 |
KRAS G12C (22%) TP53 G245R (15%) ATRX P2478fs*2 (18%) |
none | none | none | KRAS G12C | potentially secondary to lower sensitivity of Sequenom |
| IV | 75 | M | 0 | 4 | 778 |
ERBB2 L755F EZH2 R213H TP53 R248Q |
none | CDK4 | none | none | N/A |
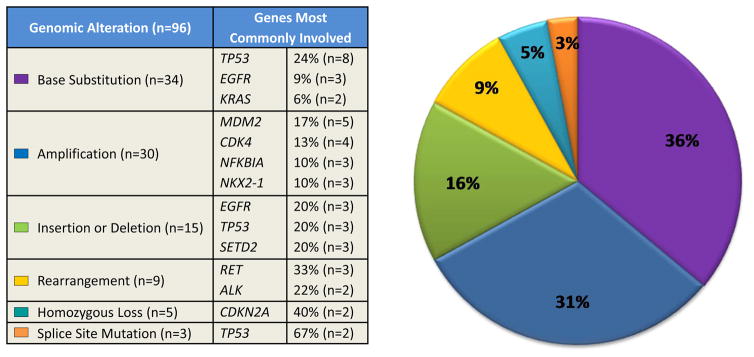
Small mutations comprised 55% of the detected abnormalities (Figure 1): 36% (n=34/96) were non-synonymous base substitutions, 16% (n=15/96) insertion or deletion, and 3% (n=3/96) splice site mutation. For non-synonymous point mutations, TP53 was the most commonly mutated gene (24%, n=8/34), followed by EGFR (9%, n=3/34) and KRAS (6%, n=2/34). Insertion or deletion most commonly involved EGFR, TP53, and SETD2 (20%, n=3/15 for each). The majority of splice site mutations involved TP53 (67%, n=2/3).
Figure 1. Frequency of genomic alterations (GAs) identified via next-generation sequencing.
A total of 96 GAs were identified in 31 lung adenocarcinomas. GAs were divided into six categories: base substitution, amplification, insertion/deletion, rearrangement, homozygous loss, and splice site mutation. The frequency of each of these categories is depicted in the pie chart on the right. The frequency of each of the most commonly involved genes under each category is detailed on the left.
Gene amplification comprised 31% (n=30/96) of genomic alterations. MDM2 was the most frequently amplified gene (17%, n=5/30). Gene loss comprised 9% (n=9/96) of all genomic alterations and was most commonly observed with CDKN2A (40%, n=2/5). Fusion genes were found in 9% (n=9/96). These most commonly involved RET (33%, n=3/9) and ALK (22%, n=2/9).
Clinically-Relevant Genomic Alterations
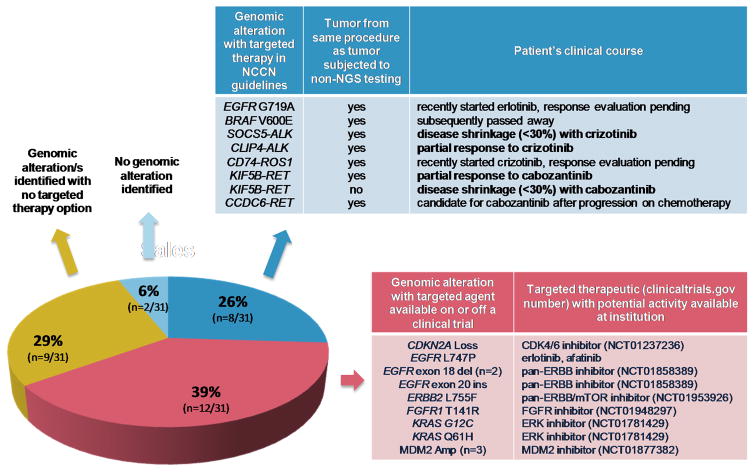
A genomic alteration with a corresponding targeted therapeutic based on the National Comprehensive Cancer Network (NCCN) guidelines for non-small cell lung cancer was identified in 26% (n=8/31) of patients. The drivers identified in tumors from these eight patients are as follows: EGFR G719A, BRAF V600E, SOCS5-ALK, CLIP4-ALK, CD74-ROS1, KIF5B-RET (n=2) and CCDC6-RET (Figure 2).
Figure 2. Clinical next-generation sequencing (NGS) and targeted therapy use.
The results of NGS of lung adenocarcinomas that harbored no genomic alterations (GAs) in 11 genes (EGFR, ERBB2, KRAS, NRAS, BRAF, MAP2K1, PIK3CA, AKT1, ALK, ROS1, and RET) via a focused panel of non-NGS testing in never or ≤15 pack-year smokers are shown. The percentage of patients with results that fall into one of four categories is depicted in the pie chart.
In these 8 patients, mass spectrometry genotyping (Sequenom) and break apart FISH testing had not detected these alterations, for a variety of possible reasons detailed in Table 2. Of note, in all but 1 of 8 patients, tumor samples used for non-NGS and NGS testing were obtained from the same biopsy or procedure.
In an additional 39% (n=12/31) of patients, an actionable genomic alteration was discovered for which targeted therapy was available either on an ongoing trial at the institution or off-protocol. These include the following alterations and the corresponding therapy available at detection: CDKN2A loss (CDK4/6 inhibitor, NCT01237236), EGFR L747P (erlotinib, afatinib), EGFR exon 18 deletion (n=2, pan-ERBB inhibitor, NCT01858389), EGFR exon 20 insertion (pan-ERBB inhibitor, NCT01858389), ERBB2 L755F (ERBB1/2/3 inhibitor, NCT01953926), FGFR1 T141R (FGFR inhibitor, NCT01948297), KRAS G12C (ERK inhibitor, NCT01781429), KRAS Q61H (ERK inhibitor, NCT01781429), and MDM2 amplification (n=3, MDM2 inhibitor, NCT01877382).
Outcomes of Targeted Therapy Use
Of the eight patients in whose tumors an alteration with a corresponding targeted agent based on the NCCN non-small cell lung cancer guidelines was identified by the NGS assay, six (with tumors harboring CLIP4-ALK, SOCS5-ALK, CD74-ROS1, KIF5B-RET, KIF5B-RET, EGFR G719A) went on to receive targeted therapy.
Two patients demonstrated a partial response (RECIST v1.1) to targeted therapy: CLIP4-ALK with crizotinib, and KIF5B-RET with cabozantinib (NCT01639508). Both patients remain on therapy and are progression-free at 5 and 7 months, respectively. Disease shrinkage <30% (stable disease by RECIST v1.1) and a clinical response to therapy were noted in two others: SOCS5-ALK with crizotinib, and KIF5B-RET with cabozantinib (NCT01639508). The former patient died from disease progression while the latter remains progression-free on cabozantinib at 3 months. Two additional patients have begun targeted therapy but are pending a response evaluation: erlotinib for EGFR G719A, and crizotinib for CD74-ROS1. Unless otherwise specified, targeted therapeutic agents were prescribed as standard of care and were acquired commercially.
One of the remaining two cases, the patient whose tumor harbors a CCDC6-RET fusion, is currently receiving chemotherapy and will receive cabozantinib on disease progression. The patient whose tumor harbored a BRAF V600E mutation died before targeted therapy could be considered.
Of the 12 additional patients in whose tumors an actionable genomic alteration was discovered for which targeted therapy was available either on an ongoing trial at the institution or off-protocol, 11 remain on alternate systemic therapy and are candidates for targeted therapy on progression. The patient whose tumor harbors EGFR L747P recently started erlotinib and a response evaluation is pending.
DISCUSSION
While some composite algorithms which use multiple non-NGS tests, such as previously employed at our center, identify a driver in most patients with lung adenocarcinomas, more comprehensive hybrid capture-based NGS assays afford the possibility of driver alteration detection in patients where no such genomic alteration was found initially on tumor analysis.(23) In a report from the Lung Cancer Mutation Consortium (LCMC), comprehensive non-NGS genomic profiling of 1,007 lung adenocarcinoma specimens across multiple institutions was performed. A driver was identified in 64% of tumors, resulting in the use of targeted therapy in 28% of patients.(24) However, largely because of limited tissue resources, only 70% of cases could have the full complement of LCMC-mandated testing. Despite this, patients whose genomic alteration was matched to a targeted therapy appeared to live substantially longer than those with a similar driver who received non-targeted therapies.
In this series, we demonstrate that, despite extensive prior conventional non-NGS testing, 26% of apparently “driver-negative” never or light smokers with lung adenocarcinoma may have tumors that harbor a genomic alteration uncovered only by a more comprehensive NGS approach. Of the 8 patients for whom such an alteration was detected in this study, 6 went on to receive targeted therapy. All 6 of these patients derived clinical benefit from targeted therapy initiation, and a partial response or evidence of disease shrinkage was noted in all 4 patients who have undergone a radiographic evaluation for disease response. Interestingly, the majority of these alterations were recurrent gene rearrangements involving ALK, ROS1, and RET.
Reasons for non-detection of these genomic alterations via non-NGS testing are varied. These include lower sensitivity, complex rearrangements undetectable by standard FISH, and, possibly, heterogeneity between different tumor biopsies or sites. Clinical samples sometimes contain biologically-relevant genomic alterations at low allele frequencies due to excess non-neoplastic cells, leading to false negative results on some forms of non-NGS testing. For such samples, deep and uniform unique coverage (median >500x) is often necessary for a thorough analysis due to low tumor purity common in many metastatic tumor types.(21) Regarding recurrent gene rearrangements, at least one large series has previously demonstrated the capability of this type of NGS assay to detect ALK fusions in patients whose tumors previously tested negative for an ALK rearrangement via FISH.(25)
It is important to point out that this effort focused on a select population of patients with pathologically-confirmed adenocarcinomas who were never smokers or smoked cigarettes ≤15 pack-years. This strategy was undertaken in an attempt to enrich the diagnostic yield of NGS for potential drivers. However, while many clinically actionable lung cancer drivers are more commonly found in tumors of never smokers, these drivers have been identified in tumors from smokers as well, and patients treated with the associated therapy appear to fare as well as the never or light former smoker population.(26, 27) In addition, other actionable drivers such as some BRAF (13, 28) and KRAS mutations(29, 30) are enriched in tumors from patients with a significant history of smoking. No clinical characteristics can be used to select NSCLC patients whose tumors should be tested, and current guidelines recommend routine ALK and EGFR testing of tumors from all patients (preferably as part of a multiplex panel) with adenocarcinomas, large cell carcinomas, NSCLC NOS (not otherwise specified), and squamous lung cancers from never smokers and small diagnostic biopsies.(31)
Molecular diagnostic algorithms that employ multiple non-NGS tests are becoming less tenable due to their relatively large tissue requirements. The majority of patients in this series required one or more procedures to complete testing. Specifically, more than two thirds of these patients required multiple biopsies to complete non-NGS testing alone. In addition, those who had sufficient tissue for analysis were largely patients who had undergone a surgical procedure for diagnosis and/or molecular testing. The type of NGS assay used in the present study offers the advantage of more comprehensive genomic characterization using as low as 50 ng of DNA which can be obtained from lung cancer specimens with sufficient tumor content using core biopsy needles on average or a carefully prepared fine needle aspiration.(32, 33)
Lastly, the use of broad, hybrid capture-based NGS for more comprehensive molecular genotyping expands the scope molecular alterations interrogated and permits new driver alteration discovery. Highly specialized types of massively-parallel sequencing, after rigorous analytic validation such as that described here, allow for the simultaneous interrogation of a broader spectrum of genes, including mutations in all exons of oncogenes and tumor suppressor genes, copy number gains and losses, and recurrent gene rearrangements.(34) Partly to address the types of testing concerns revealed in the present study, our center (MSKCC) has recently implemented a similar broad, hybrid capture-based NGS assay.(35)
With the advent of this approach, the process of matching the range of genomic alterations discovered with potential targeted therapeutics will undoubtedly represent an enormous challenge and an unprecedented opportunity for clinicians. If functional data are available, that should help in discriminating strong potential drivers from passenger alterations. In the absence of such data, however, for patients with limited systemic therapy choices, targeted therapy that inhibits the oncogenic protein of interest or a downstream pathway remains a valid option.(36)
CONCLUSIONS
Broad, hybrid capture-based NGS assays have the potential to uncover clinically-actionable genomic alterations in never smokers or ≤15 pack year smokers whose lung adenocarcinomas do not harbor a potential driver via non-NGS testing. This approach provides a comprehensive and rapid interrogation of the cancer genome using potentially less tumor tissue than standard algorithms. In this series, the majority of patients either received or became eligible for targeted therapy due to the discovery of a clinically-actionable genomic alteration via NGS. When available, we recommend the use of such NGS-based assays as the optimal molecular diagnostic platform for patients with lung cancers. Our center (MSKCC) has since adopted this type of NGS assay as our primary testing method of choice for patients with advanced lung cancers and other tumor types.(35)
Supplementary Material
STATEMENT OF TRANSLATIONAL RELEVANCE.
Selecting the most optimal platform for molecular diagnostic profiling represents a crucial step in the management of patients with advanced lung adenocarcinomas. The use of broad, hybrid capture-based next-generation sequencing (NGS) resulted in the identification of actionable genomic alterations in close to two-thirds of lung adenocarcinomas that previously tested “negative” for known alterations by multiple conventional non-NGS tests including multiplex mass spectrometry, sizing assays, and FISH. Upon driver identification via NGS, several patients whose tumors would have otherwise been deemed “driver-negative” via non-NGS testing subsequently received and responded to targeted therapy. These results underscore the important role of broad, hybrid capture-based NGS in the clinic as a single test that interrogates a wide range of genomic alterations using potentially less tissue than standard non-NGS testing.
Acknowledgments
NIH P01 CA129243 (M.G. Kris and M. Ladanyi) for partial support of non-NGS testing at MSKCC. Ryma Benayed, Catherine O’Reilly, and Ahmet Zehir (MSKCC) for RNA-based confirmation of the HIP1-ALK fusion.
Footnotes
Disclosure Statement
SB, JRG, JSR, PS, DL, and VAM are employed by and own stock of Foundation Medicine. ML owns stock of Foundation Medicine.
Financial Support N/A
References
- 1.Pao W, Miller V, Zakowski M, Doherty J, Politi K, Sarkaria I, et al. EGF receptor gene mutations are common in lung cancers from “never smokers” and are associated with sensitivity of tumors to gefitinib and erlotinib. Proc Natl Acad Sci USA. 2004;101:13306–11. doi: 10.1073/pnas.0405220101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Lynch TJ, Bell DW, Sordella R, Gurubhagavatula S, Okimoto RA, Brannigan BW, et al. Activating mutations in the epidermal growth factor receptor underlying responsiveness of non-small-cell lung cancer to gefitinib. N Engl J Med. 2004;350:2129–39. doi: 10.1056/NEJMoa040938. [DOI] [PubMed] [Google Scholar]
- 3.Paez JG, Janne PA, Lee JC, Tracy S, Greulich H, Gabriel S, et al. EGFR mutations in lung cancer: correlation with clinical response to gefitinib therapy. Science. 2004;304:1497–500. doi: 10.1126/science.1099314. [DOI] [PubMed] [Google Scholar]
- 4.Soda M, Choi YL, Enomoto M, Takada S, Yamashita Y, Ishikawa S, et al. Identification of the transforming EML4-ALK fusion gene in non-small-cell lung cancer. Nature. 2007;448:561–6. doi: 10.1038/nature05945. [DOI] [PubMed] [Google Scholar]
- 5.Mok TS, Wu YL, Thongprasert S, Yang CH, Chu DT, Saijo N, et al. Gefitinib or carboplatin-paclitaxel in pulmonary adenocarcinoma. N Engl J Med. 2009;361:947–57. doi: 10.1056/NEJMoa0810699. [DOI] [PubMed] [Google Scholar]
- 6.Rosell R, Carcereny E, Gervais R, Vergnenegre A, Massuti B, Felip E, et al. Erlotinib versus standard chemotherapy as first-line treatment for European patients with advanced EGFR mutation-positive non-small-cell lung cancer (EURTAC): a multicentre, open-label, randomised phase 3 trial. Lancet Oncol. 2012;13:239–46. doi: 10.1016/S1470-2045(11)70393-X. [DOI] [PubMed] [Google Scholar]
- 7.Shaw AT, Kim DW, Nakagawa K, Seto T, Crino L, Ahn MJ, et al. Crizotinib versus chemotherapy in advanced ALK-positive lung cancer. N Engl J Med. 2013;368:2385–94. doi: 10.1056/NEJMoa1214886. [DOI] [PubMed] [Google Scholar]
- 8.Sequist LV, Yang JC, Yamamoto N, O’Byrne K, Hirsh V, Mok T, et al. Phase III study of afatinib or cisplatin plus pemetrexed in patients with metastatic lung adenocarcinoma with EGFR mutations. J Clin Oncol. 2013;31:3327–34. doi: 10.1200/JCO.2012.44.2806. [DOI] [PubMed] [Google Scholar]
- 9.Shaw AT, Kim DW, Mehra R, Tan DS, Felip E, Chow LQ, et al. Ceritinib in ALK-rearranged non-small-cell lung cancer. N Engl J Med. 2014;370:1189–97. doi: 10.1056/NEJMoa1311107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kandoth C, McLellan MD, Vandin F, Ye K, Niu B, Lu C, et al. Mutational landscape and significance across 12 major cancer types. Nature. 2013;502:333–9. doi: 10.1038/nature12634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lawrence MS, Stojanov P, Polak P, Kryukov GV, Cibulskis K, Sivachenko A, et al. Mutational heterogeneity in cancer and the search for new cancer-associated genes. Nature. 2013;499:214–8. doi: 10.1038/nature12213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Stephens P, Hunter C, Bignell G, Edkins S, Davies H, Teague J, et al. Lung cancer: intragenic ERBB2 kinase mutations in tumours. Nature. 2004;431:525–6. doi: 10.1038/431525b. [DOI] [PubMed] [Google Scholar]
- 13.Paik PK, Arcila ME, Fara M, Sima CS, Miller VA, Kris MG, et al. Clinical characteristics of patients with lung adenocarcinomas harboring BRAF mutations. J Clin Oncol. 2011;29:2046–51. doi: 10.1200/JCO.2010.33.1280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Chaft JE, Arcila ME, Paik PK, Lau C, Riely GJ, Pietanza MC, et al. Coexistence of PIK3CA and other oncogene mutations in lung adenocarcinoma-rationale for comprehensive mutation profiling. Mol Cancer Ther. 2012;11:485–91. doi: 10.1158/1535-7163.MCT-11-0692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Bleeker FE, Felicioni L, Buttitta F, Lamba S, Cardone L, Rodolfo M, et al. AKT1(E17K) in human solid tumours. Oncogene. 2008;27:5648–50. doi: 10.1038/onc.2008.170. [DOI] [PubMed] [Google Scholar]
- 16.Bergethon K, Shaw AT, Ou SH, Katayama R, Lovly CM, McDonald NT, et al. ROS1 rearrangements define a unique molecular class of lung cancers. J Clin Oncol. 2012;30:863–70. doi: 10.1200/JCO.2011.35.6345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Drilon A, Wang L, Hasanovic A, Suehara Y, Lipson D, Stephens P, et al. Response to Cabozantinib in patients with RET fusion-positive lung adenocarcinomas. Cancer Discov. 2013;3:630–5. doi: 10.1158/2159-8290.CD-13-0035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Camidge DR, Ou SH, Shapiro G, Otterson GA, Villaruz LC, Villalona-Calero MA, et al. Efficacy and safety of crizotinib in patients with advanced c-MET-amplified non-small cell lung cancer (NSCLC) J Clin Oncol. 2014;32:5s. (suppl; abstr 8001) [Google Scholar]
- 19.Govindan R, Ding L, Griffith M, Subramanian J, Dees ND, Kanchi KL, et al. Genomic landscape of non-small cell lung cancer in smokers and never-smokers. Cell. 2012;150:1121–34. doi: 10.1016/j.cell.2012.08.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Li T, Kung HJ, Mack PC, Gandara DR. Genotyping and genomic profiling of non-small-cell lung cancer: implications for current and future therapies. J Clin Oncol. 2013;31:1039–49. doi: 10.1200/JCO.2012.45.3753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Meyerson M, Gabriel S, Getz G. Advances in understanding cancer genomes through second-generation sequencing. Nat Rev Genet. 2010;11:685–96. doi: 10.1038/nrg2841. [DOI] [PubMed] [Google Scholar]
- 22.Shendure J, Ji H. Next-generation DNA sequencing. Nat Biotechnol. 2008;26:1135–45. doi: 10.1038/nbt1486. [DOI] [PubMed] [Google Scholar]
- 23.Querings S, Altmuller J, Ansen S, Zander T, Seidel D, Gabler F, et al. Benchmarking of mutation diagnostics in clinical lung cancer specimens. PLoS One. 2011;6:e19601. doi: 10.1371/journal.pone.0019601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kris MG, Johnson BE, Berry LD, Kwiatkowski DJ, Iafrate AJ, Wistuba II, et al. Using multiplexed assays of oncogenic drivers in lung cancers to select targeted drugs. JAMA. 2014;311:1998–2006. doi: 10.1001/jama.2014.3741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ali SM, Ou SH, Peled N, Chmielecki J, Pinder MC, Palma NA, et al. Identifying ALK rearrangements that are not detected by FISH with targeted next-generation sequencing of lung carcinoma. J Clin Oncol. 2014;32:5s. (suppl; abstr 8049) [Google Scholar]
- 26.Dogan S, Shen R, Ang DC, Johnson ML, D’Angelo SP, Paik PK, et al. Molecular epidemiology of EGFR and KRAS mutations in 3,026 lung adenocarcinomas: higher susceptibility of women to smoking-related KRAS-mutant cancers. Clin Cancer Res. 2012;18:6169–77. doi: 10.1158/1078-0432.CCR-11-3265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.D’Angelo SP, Pietanza MC, Johnson ML, Riely GJ, Miller VA, Sima CS, et al. Incidence of EGFR exon 19 deletions and L858R in tumor specimens from men and cigarette smokers with lung adenocarcinomas. J Clin Oncol. 2011;29:2066–70. doi: 10.1200/JCO.2010.32.6181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Planchard D, Mazieres J, Riely GJ, Rudin CM, Barlesi F, Quoix EA, et al. Interim results of phase II study BRF113928 of dabrafenib in BRAF V600E mutation–positive non-small cell lung cancer (NSCLC) patients. J Clin Oncol. 2013:31. (suppl; abstr 8009) [Google Scholar]
- 29.Riely GJ, Marks J, Pao W. KRAS mutations in non-small cell lung cancer. Proc Am Thorac Soc. 2009;6:201–5. doi: 10.1513/pats.200809-107LC. [DOI] [PubMed] [Google Scholar]
- 30.Janne PA, Shaw AT, Pereira JR, Jeannin G, Vansteenkiste J, Barrios C, et al. Selumetinib plus docetaxel for KRAS-mutant advanced non-small-cell lung cancer: a randomised, multicentre, placebo-controlled, phase 2 study. Lancet Oncol. 2013;14:38–47. doi: 10.1016/S1470-2045(12)70489-8. [DOI] [PubMed] [Google Scholar]
- 31.NCCN. Clinical Practice Guidelines in Oncology: Non-Small Cell Lung Cancer version 1.2014. http://www.nccn.org/professionals/physician_gls/pdf/nscl.pdf.
- 32.Frampton GM, Fichtenholtz A, Otto GA, Wang K, Downing SR, He J, et al. Development and validation of a clinical cancer genomic profiling test based on massively parallel DNA sequencing. Nat Biotechnol. 2013;31:1023–31. doi: 10.1038/nbt.2696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Young G, Wang K, He J, Otto G, Hawryluk M, Zwirco Z, et al. Clinical next-generation sequencing successfully applied to fine-needle aspirations of pulmonary and pancreatic neoplasms. Cancer Cytopathol. 2013;121:688–94. doi: 10.1002/cncy.21338. [DOI] [PubMed] [Google Scholar]
- 34.Pritchard CC, Salipante SJ, Koehler K, Smith C, Scroggins S, Wood B, et al. Validation and implementation of targeted capture and sequencing for the detection of actionable mutation, copy number variation, and gene rearrangement in clinical cancer specimens. J Mol Diagn. 2014;16:56–67. doi: 10.1016/j.jmoldx.2013.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Cheng DT, Mitchell T, Zehir A, Shah RH, Benayed R, Syed A, et al. MSK-IMPACT: A hybridization capture-based next generation sequencing clinical assay for solid tumor molecular oncology. J Mol Diagn. doi: 10.1016/j.jmoldx.2014.12.006. (in press) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Tsimberidou AM, Iskander NG, Hong DS, Wheler JJ, Falchook GS, Fu S, et al. Personalized medicine in a phase I clinical trials program: the MD Anderson Cancer Center initiative. Clin Cancer Res. 2012;18:6373–83. doi: 10.1158/1078-0432.CCR-12-1627. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.