Abstract
Regeneration of lost or injured tissues is very common in biology. Unfortunately, humans lack adequate regeneration in the heart and many other organs that are commonly ravaged by modern diseases. A revolution in stem cell biology has led to an explosion of interest in therapies that can awaken the regeneration potential in patients. In just the past decade, we have learned that any cell type from any patient, including cells from a blood sample or skin biopsies, can potentially be reprogrammed into a stem cell, and that patient’s stem cell can generate billions of new cells of a variety of differentiated cell types, including cardiomyocytes, endothelial cells and neurons. Stem cell biology is already changing how we approach human genetics and drug discovery, and clinical efforts to harness stem cell biology for heart failure are well underway. Here we predict that regenerative biology will initially struggle to hurdle economic and technical barriers for realistic treatments of heart diseases. However, regenerative biologists are beginning to design new approaches that were previously unimaginable, and ultimately regenerative medicine will have a profound impact for heart diseases.
Keywords: Heart Failure, Drugs, Genetics
Introduction
For centuries, biologists have studied salamanders that can regrow entire new limbs after amputation. Flatworms, when beheaded, grow a new head from the beheaded corpse (and the severed head grows a new body) 1. All species, from bacteria to plants to animals, can regenerate to some degree, and it has been said that without regeneration, there could be no life 2. In fact, humans regenerate many of their tissues, including skin, blood, liver and intestinal mucosa, routinely, efficiently, and often perfectly. Skeletal muscle regenerates remarkably well; every time we strain a muscle, muscle stem cells repair the injury and this is a key component of the conditioning associated with exercise training. But the adult human heart appears to have little of the regenerative ability of our skeletal muscles. The challenge of this limitation has triggered considerable enthusiasm in using different types of stem cells to repair failing human hearts. We will not review this substantial effort in human cardiac cell therapy, as expert reviews have been published recently 3–5. Here, we attempt to predict where regenerative biology and its translational discipline, regenerative medicine, will take us in the future, with a focus on cardiovascular medicine.
Regenerative biology has many definitions but a simple one is the study of how organisms replace lost or damaged tissue with new tissue. We are now entering an era where regenerative biology is turning from science fiction into science, with realistic regenerative medical applications beginning to emerge. This change is based on a broader and more fundamental understanding of cell biology, and this new view has laid a foundation for the potential repair of organs that were previously viewed as being incapable of regeneration or even modest repair. The explosion of interest in regenerating diseased hearts and other organs is being fueled by advances in many fields including nanotechnology, genetics, imaging and other fields. But, at the center of this new regeneration movement are recent advances in understanding the biology of stem cells (Figure 1). Thus, before making predictions for the future of regenerative medicine, it is important to review some of the recent and stunning events in the stem cell field. At a fundamental level, these events have changed how we view diseased organs, because we now see in every cell—even the most diseased cells—the potential to become transformed into a different cell type.
Figure 1. Stem cell biology and reprogramming.
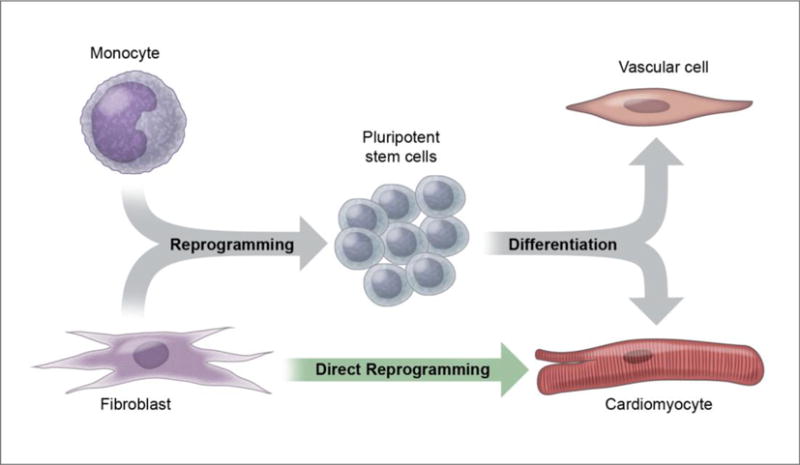
While embryonic stem cell advances have enabled the generation of any cell type in the human body, the recognition that cells can be reprogrammed to other cells has opened the door to more diverse regenerative medicine approaches. Notably, fibroblasts can be directly converted to cardiomyocytes, without passing through a stem cell state.
The Impact of Stem Cell Biology
Stem cells, by definition, are cells that can self-renew as well as form multiple types of differentiated cells. Embryonic Stem Cells, or ES cells, are cells derived from the blastocyst before implantation into the uterine wall, and ES cells are capable of giving rise to all cell types in the body. This property is referred to as pluripotency. Mouse embryonic stem cells were first derived in 1981 6, 7, and human embryonic stem cells were derived by Thomson and colleagues in 1988 8. Derivation of embryonic stem cells usually destroys the embryo, and, thus, research with embryonic stem cells has been controversial 9. Embryonic stem cells are derived from donated embryos; no human embryonic stem cell lines can be created without consent of the donor, and they are never made from aborted fetuses. Mouse and human embryonic stem cells can be easily directed into differentiating to cardiomyocytes through a series of sequential small molecule, protein, or nucleic acid signals 10, 11. Human cardiomyocytes derived from human embryonic stem cells have been shown to improve the function of infarcted rodent hearts 12, and have also been shown to improve the function of infarcted non-human primate hearts, although ventricular arrhythmias occurred 13.
Adult stem cells are cells that reside in adult tissues such as bone marrow, the hair follicle, and intestinal epithelium, and adult stem cells have the capacity of self-renewal and differentiation. Many putative adult stem cells have been reported to generate cardiomyocytes, with some of these candidate cells in clinical trials 14. Enthusiasm for adult cardiac stem cells has been justified to some extent, as this concept could lead to autologous cell transplantation for heart failure analogous to the widely used hematopoietic stem cell approach. However, in contrast to hematopoietic stem cells, cells proposed to be cardiac stem cells are less rigorously characterized and typically represent heterogeneous populations of cells that display a low level of differentiation into cardiac myocytes. Furthermore, the potential for adult cardiac stem cells may have been overstated, as recent data do not indicate that the adult mammalian heart has a highly regenerative stem cell population 15.
A landmark event in stem cell biology was the discovery of induced pluripotent stem cells (iPS cells) with properties of embryonic stem cells, as reported in 2006 by Shinya Yamanaka’s team at Kyoto University. In this study, it was shown that an iPS cell can be generated from differentiated cells using reprogramming factors 16. This monumental achievement meant that potentially any cell could be used to generate unlimited numbers of any differentiated cell type, without using embryonic stem cells (Figure 1). Yamanaka and John Gurdon, who had shown the potential of reprogramming in frogs in 1958 17, were awarded the Nobel Prize in 2012 for the discovery that mature cells can be reprogrammed to pluripotency. Since Yamanaka’s transformative discovery, iPS technology has allowed the routine generation of human cardiomyocytes from iPS cells derived from human fibroblasts 18, 19. Clinical trials for macular degeneration with retinal pigment epithelial cells derived from iPS cells are underway 20.
One of the consequences of iPS technology has been easing of the ethical tensions over the use of embryonic stem cells, as iPS cell generation requires only a skin fibroblast or another cell type from the donor. However, the impact of reprogramming of cells goes far beyond iPS cell generation for generating heart cells. We now recognize the potential to turn almost any cell into any other cell type, and this has opened the floodgates of new reprogramming strategies 21. Importantly, cells can now be directly reprogrammed to other types of cells without passing through a stem cell state, potentially limiting the generation of teratomas and cancers. For example, in the pancreas, exocrine cells that don’t produce insulin can be directly reprogrammed to form beta cells for diabetes without removing the cells from the animal 22, and in the brain, one type of neuron can be directly reprogrammed to another type of neuron 23. The direct conversion of adult skin fibroblasts to functional endothelial cells has also been documented 24. In the heart, direct reprogramming has allowed investigators to turn cardiac fibroblasts into cardiac myocytes25, 26. The concept that local fibroblasts, which generate the scar of the injured heart, can be directly converted into functional cardiac myocytes without returning to a pluripotent state, is a stunning example of the power of stem cell biology.
In concert with advances in many fields, particularly human genetics, stem cell and reprogramming technologies will revolutionize cardiovascular biology over the next generation. It is obvious that the ability to generate billions of cardiac myocytes, as well as other cell types, has the potential to replace damaged cardiovascular tissue by cell transplantation. Less obvious is the potential for stem cell science to transform virtually all areas of cardiovascular therapeutics, changing how diseases are studied and how therapies are developed. Even if injection of cells into damaged hearts is never a widely-adopted therapy for patients, and even if we can’t grow new myocardium that is routinely sewn into hearts, the principles of regenerative biology will have a profound impact on how we view new therapies for cardiovascular disease. For example, it may be possible for us to stimulate endogenous regeneration mechanisms within the myocardium through proteins or gene delivery, leading to new cardiomyocyte formation without exogenous delivery of cells. In fact, this “in situ” regeneration approach could be much more economically attractive than cell therapy.
We recognize that translating regenerative biology principles into real benefits for patients will take many years, and, in some cases, many decades. We know there will be disappointments and missteps along the way. Some of our predictions may seem as fanciful as science fiction, but we note that this is how implantable defibrillators and drug eluting stents were once viewed. So, while we are cautious about what therapies are “right around the corner”, we are very optimistic that regenerative biology will change cardiovascular medicine.
Prediction 1: Regenerative medicine will initially struggle to become practical reality
Stem cell biology has set off an explosion of efforts to generate virtually every type of cell. Although stem cells provide enormous potential for all of medicine, we envision that many cardiovascular diseases will not be direct therapeutic targets for regenerative medicine over the next decade. For example, human cardiomyocyte pacemaker cells have been made from the keratinocytes from a hair plucked from a patient—a truly fascinating achievement 27. However, while electronic pacemaker technology is far from perfect, the reliability of a stem cell-derived sino-atrial node may never exceed or even approach our current pacemaker technology. The bar for replacing electronic pacemaker technology is extremely high, and since pacemakers and leads will continue to improve, the bar will only move higher. Regenerative medicine therapies will need to be competitive in light of improvements in devices, surgical techniques and standard pharmacological approaches (Figure 2).
Figure 2. Needs unmet by non-regenerative therapies will define regenerative medicine opportunities.
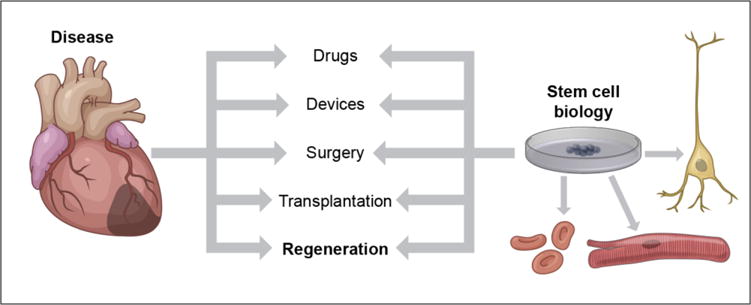
Regenerative treatments will emerge but will be most useful in areas where more traditional approaches leave voids of unmet needs; thus, progress in other fields will play a major role in determining where regenerative breakthroughs can occur. However, stem cell biology will influence all approaches, by allowing testing and validation with human tissue of diverse genetic backgrounds.
Thus, the excitement of stem cell biology, and the dream of replacing diseased cells with healthy ones, will be met with an economic and practical reality. In many human diseases, including in the cardiovascular system, progress in more conventional therapies will make regenerative medicine uncompetitive. For example, consider the changes in the treatment of acute myocardial infarction over the past few decades. Once, only a minority of patients with myocardial infarction were candidates for percutaneous intervention because most people did not live close enough to hospitals with catheterization laboratories. With the demonstration of the benefits of primary percutaneous coronary intervention, by the year 2000, almost 80% of United States residents lived within 60 minutes of a hospital capable of primary percutaneous coronary intervention 28. Because most patients with acute myocardial infarction currently do well if they make it to the catheterization laboratory and are successfully reperfused, the opportunity for regenerating myocardium immediately after myocardial infarction will be limited to patients with clear extensive damage or some subset of patients that can be predicted to have poor outcomes. In this manner, the success in treating acute coronary thrombosis has reduced the need for approaches to regenerate myocardium immediately after infarction. Of course, many patients still develop post-infarction heart failure, which continues to carry important short term and long term risk. 29 However, if we can identify and intervene in these patients at the right time, heart muscle regeneration after infarction could prove to be a transformative therapy. If the risks and costs of the regenerative therapy were small, then even patients with very modest infarctions could be candidates for therapy.
Progress in other areas of cardiovascular therapeutics will also continue, and this is why we predict that regenerative medicine will begin to focus on major areas where devices, medications, and other approaches will likely leave major unmet needs. These may include genetic cardiomyopathies, where diagnosis may be made long before the emergence of disease. Cell-based solutions for major problems will be expensive to develop, and economics is one reason that a limited number of diseases will emerge as targets for regenerative therapies through cells. Chronic ischemic cardiomyopathy will remain an enormous clinical problem, and, therefore, replacing ventricular scar tissue with new regenerated myocardium continues to be a major challenge to regenerative biology. In 2015, Menasche and colleagues reported the first case of a human with severe heart failure treated with embryonic stem cell therapy, using a progenitor cell-seeded patch sewn to the left ventricle at the time of bypass surgery 30. The patient did well post-operatively, with an improvement in ejection fraction and no apparent complications, and this landmark event raises the very real prospect that cardiac regeneration through embryonic stem cells could help patients with chronic cardiomyopathy. But even chronic ischemic cardiomyopathy will be a moving target. For example, after decades of research, the combined therapy angiotensin receptor antagonist-neprilysin inhibitor has led to a breakthrough success for heart failure with reduced ejection fraction in humans 31. Regenerative therapies for chronic heart failure will eventually have to face the same efficacy and economic reality tests that all new therapies encounter.
Prediction 2: Stem cell biology, irrespective of therapy, will advance our understanding of diseases
Even if stem cells do not become a routine therapy for cardiovascular patients, stem cell biology will change how we study diseases in the next decade. Most notably, stem cell biology allows one to model a disease in diverse human backgrounds, since stem cells can be generated from any patient, and cardiovascular cells can then be created from those patients. For example, human cardiomyocytes have been generated from patients with long QT syndromes through reprogramming 32. This allows the study of a specific mutation in cells with the same genetic background as the patient, and potentially could allow one to identify drugs through in situ approaches that will have beneficial effects in that patient.
Human disease modeling by stem cell technology may be particularly useful for common diseases that have complex genetic causes, rather than monogenic diseases (Figure 3). Studies of the human genome have taught us that for many common diseases, like type II diabetes or obesity, there is more than one gene that contributes to the genetic risk of disease. Instead, many diseases have familial patterns that are linked to many distinct regions in the genome, and each of these regions has only a moderate or sometimes even a very minor influence on the trait 33. For example, there are at least 46 different validated gene loci for the inheritance of coronary artery disease in humans, but these combined loci explain less than 10% of the familial inheritance of the disease 34.
Figure 3. Disease in a dish specific to individual patients.
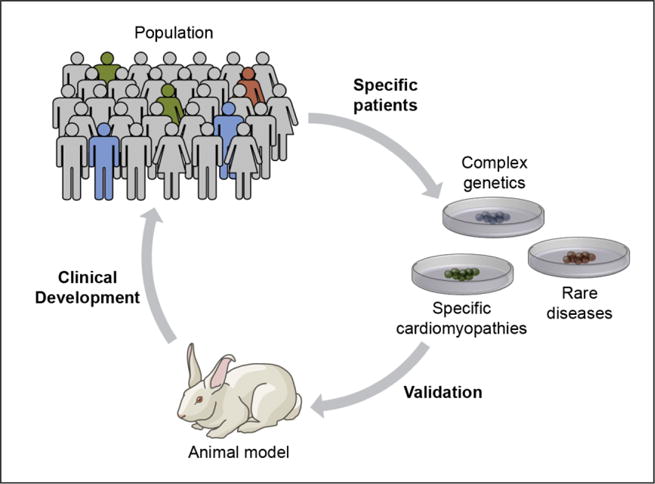
Even if most patients do not receive cellular therapies, regenerative biology will provide new testing platforms for therapy development. Generation of laboratory cells and tissues from specific patients provides the true genetic background of the disease. This approach is currently challenged by cost, time and reproducibility, but advances over the next decade will make this a routine approach for many diseases.
In view of this complexity, testing the interactions of many different genes is extremely challenging. Imagine all of the combinations of genetically modified mice that would need to be generated to explore the interactions between ten or more genes. Here, stem cell biology will have a major impact because it enables investigators to manipulate systematically many different genes, and it can be done within the identical genetic background of the patient. In this regard, creating stem cells from patients who have the disease will provide a more authentic system to study the human disease process than the laboratory mouse and other animals models. Furthermore, gene editing (discussed below) of those stem cells, will enable rigorous studies of complex disease mechanisms within the patient-specific tissues.
At the current time, the cost and efficiency of taking hair or skin cells, reprogramming them to stem cells, and then generating heart cells to study specific drugs prohibits widespread use of this approach. Other major challenges to developing “disease in a dish” models of cardiovascular diseases also exist, such as developing fully mature cardiomyocytes (most stem cell-derived heart cells are immature and do not develop into fully functional adult-like cardiac myocytes) and reducing the variability between different clones of iPS cells generated from the same cell 35. These barriers are sufficient to predict that modeling human diseases through iPS cells will be experimental for at least the next 5 years, and probably for the next decade. However, investigators are employing 3 dimensional culture systems and other engineering techniques such as molecularly designed cellular scaffolds and mechanically stimulated cultures that are increasingly making stem cell-derived cardiac myocytes increasingly more similar to bona fide adult human myocardium 36. It is thus likely that cardiovascular therapies will be discovered and developed by testing on laboratory-developed human tissues from the patients who actually carry the disease. In addition, it is quite possible that therapies will someday be individualized for patients through testing on their own cells, in the same way that breast cancer cells are now studied to determine if the patient will be hormone responsive. For example, Kass and colleagues have shown that the responses of stem cell-derived cardiomyocytes to anti-arrhythmia drugs are predictive of management outcomes in a family cohort with a long QT syndrome 37. Disease modeling based on iPS cells has largely been focused on cell autonomous (independent of other cells) features of myocardial disease such as QT length. The study of more complex multicellular genetic effects requires more tissue-like systems than individual cells 38.
As noted above, a powerful driving force in the development of stem cell-based laboratory models of human diseases will be economics. Many modern therapies are developed through careful mechanistic laboratory investigations in cells, followed by extensive testing in rodents and then in large animals. Although the cost increases with every step of this process, this course of investigation is employed to reduce the risk that a therapy will fail in humans. Currently, the mouse remains the mainstay of laboratory investigation due to ease of genetic engineering and widespread use of mouse experimental models. However, testing in mice is expensive relative to most cell culture experiments, and testing in large animals is much more expensive. Furthermore, mouse experimentation is severely limited by the inability to mimic the complexity of most human diseases, and most laboratory tests on mice are performed in a single genetically-inbred background. Thus, exploring the effects of a drug or therapy on human tissue will likely become common when the cost, efficiency and reproducibility of “disease in a dish” models are all improved. Of course, many human diseases, including most cardiovascular disorders, like hypertension and heart failure, require study in a system with intact physiology. For this reason, animal models will continue to be essential, and stem cell biology is going to complement rather than replace animal research.
Prediction 3: Stem cell therapy will provide an avenue for the application of genome editing
For serious genetic diseases, the ultimate goal is to precisely correct the defect through gene manipulation without harming the patient. The ability to manipulate the genome with ease has been an enormous challenge in molecular medicine, but recent stunning breakthroughs have opened genetic engineering to many possibilities. One of the most revolutionary discoveries has been the development of a gene editing technology called CRISPR/Cas9 39, 40. This technology is the adaptation of a bacterial defense system that allows the organism to cut the DNA of an invading virus. Through an understanding of the basic mechanisms that regulate this process, scientists have devised strategies that can be used to edit genes in potentially any organism and any type of cell in a rapid and accurate manner. The CRISPR/Cas9 system is sufficiently easy that, even though the technology was described in 2013, it has been simplified in a short time period and is now widely accessible. CRISPR/Cas9 gene editing can also be accomplished in the intact adult organism, and this proof of principle has been shown by reducing PCSK9 gene expression in the liver of the mouse, leading to dramatic reduction in cholesterol 41.
Gene editing combined with stem cell technology has the potential to revolutionize the care of monogenic diseases like sickle cell anemia in the near future. With generation of iPS cells from a patient, genetic defects can be corrected and then differentiated cells or tissues can then be transplanted into the patient (Figure 4). The first human application of genome editing, using the zinc finger technology that preceded CRISPR/Cas9, was used to inactivate CCR5, a co-receptor for the Human Immunodeficiency Virus, on human T cells. The treatment was relatively safe, and the blood level of HIV DNA decreased in most of the 12 patients enrolled in the study 42.
Figure 4. Gene editing to correct diseased cells.
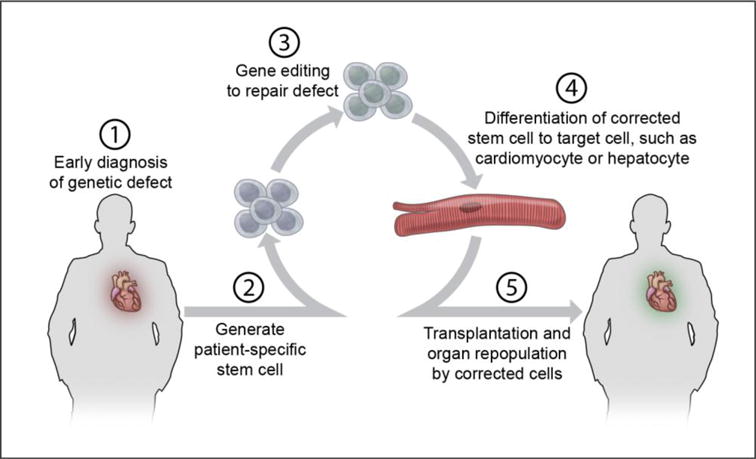
New technologies allow simple gene editing in stem cells, and this could enable unique approaches to many diseases. Stem cells from individual patients can be generated, with repair of one or even more genetic defects. The stem cells can then be directed to form cardiomyocytes or other types of cells, such as a hepatocyte with improved cholesterol metabolism. The cells with corrected function can be used to generate tissues for transplantation.
Genome editing is so efficient and powerful that inventors of the technology as well as other prominent scientists have called for a moratorium on human use until more consideration of the ethics of human gene editing can occur 43, 44. There will have to be extensive discussion and research to evaluate the safety and ethical concerns of changing human DNA in a manner that goes beyond currently accepted gene therapy. No one wants to see gene editing for cosmetic reasons or to bestow traits that lead to superiority in sporting events to our children. On the other hand, no one wants children to suffer from the debilitating effects of genetic diseases, such as Duchene’s muscular dystrophy. In this regard, Olson and colleagues have used CRISPR/Cas9 genome editing to reverse Duchene’s muscular dystrophy in mice, with improvements in both skeletal muscle and myocardium 45. Thus, it appears inevitable that genome editing will become a clinical approach for many very serious human diseases, but defining guidelines for gene editing is very prudent as the technology rapidly advances. Gene editing of differentiated somatic cells such as cardiomyocytes and fibroblasts will be more acceptable than gene editing of the germline, which has potential for harmful effects for not only that embryo, but also for future offspring of that embryo.
Genetic causes of cardiomyopathy are major contributors to heart failure, and thus it is attractive to consider gene editing for correction of lethal genetic cardiomyopathies. However, the myocardium is a much more challenging organ for this approach compared to, for example, the hematopoietic system, where stem cells are in routine clinical use. Extensive controversy arose over the past decade on whether the heart, like skeletal muscle, is continuously regenerated by endogenous stem cells that became cardiomyocytes. The recent advances in gene editing make the issue of the origins of our heart cells into much more than an academic question. If the myocardium were continuously being refreshed by stem cells, then the concept of gene-editing those stem cells and repairing the lethal mutation would be viable, as those repaired stem cells would then replace the diseased cardiomyocytes. Unfortunately, it appears that mammalian myocardium does not have a stem cell pool driving constant replacement of cardiomyocytes at a clinically significant rate 15. It now appears that the stem cell contribution to myocardial homeostasis in the adult may be very modest, and that most cardiomyocytes arise from pre-existing cardiomyocytes. Thus, replacing most of the diseased cardiomyocytes with stem-cell derived cardiomyocytes with genetic correction will not be an easy task. However, the concept should certainly not be abandoned, because it appears that, slowly, about half of the cardiomyocytes are replaced throughout a human lifetime 46. Thus, if a cardiomyopathy can be diagnosed at a very early age, one could envision approaches where transplant gene-corrected cardiomyocytes could slowly replace a substantial portion of the myocardium and thereby circumvent the disease that would develop later in life. There remain important questions about cell therapy, including how to maximize true long-term integration of transplanted cells into the complex environment of the myocardium.
Prediction 4: Regenerative medicine will promote the convergence of diverse fields
An interesting social phenomenon is the separation of scientific and clinical disciplines into silos that provide stable, albeit insulated, environments for collaboration and advancement within a given field. However, new technology often forces integration of those fields, in the same way that Transcatheter Aortic Valve Replacement is forcing convergence of cardiac surgery and interventional cardiology. Similarly, the regenerative medicine movement will force collaborations between many fields (Figure 5). Developmental biologists can define factors necessary to guide a critical biological step in regeneration—but they will need bioengineers and material scientists to help them implement a practical method of delivery. Geneticists will discover genes that may cause a disease, but they will need stem cell biologists to help them determine how the gene contributes to the disease mechanism.
Figure 5. Regenerative medicine will drive integration.
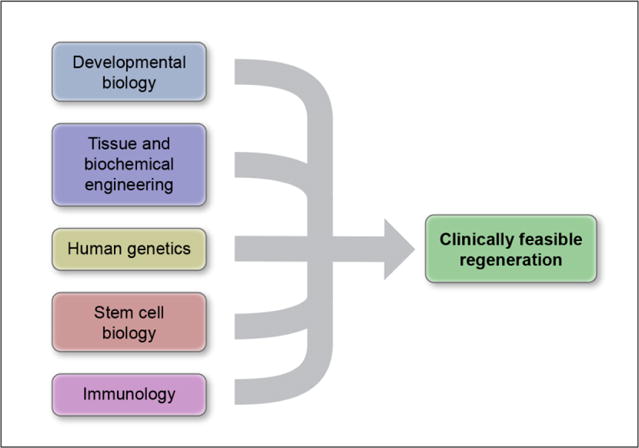
Achieving regeneration in patients will require integration of many skills. Most of these are obvious, but one particularly important integration step will be merging immunology with regenerative approaches.
This convergence of fields toward regeneration will stimulate innovations. Engineers will improve catheter technology to provide cell and regenerative proteins not only to any specific region of the heart, but to other organs as well. Molecular imaging will advance such that localizing and monitoring individual cells in humans may become feasible. And, cells themselves may be engineered to serve as biological monitors of their own health and the status of the environment.
One crucial integration event in the next decade will be the merging of immunology with regenerative medicine approaches. Clearly, the innate response is an essential component of a regenerative response to injury, and recent studies have provided a molecular framework by which immune modulators promote cellular reprogramming 47, 48. However, inflammation can be viewed as a double-edged sword, and all cell-based regenerative therapies must consider the immunological rejection of transplanted stem cells. Just as transplantation immunology arose as an essential component to life-saving organ transplantation, we will begin to see immunological responses as a limiting factor in regenerative medicine 49. Differentiated cells obtained through stem cell technology may elicit unanticipated immunological responses even if the cells were derived from the same patient 50. However, stem cell technology allows the potential of eliminating counterproductive immunological responses by using gene editing to delete antigens that provoke rejection from transplanted cells. In this manner, “universal donor” cells can potentially be generated for diverse recipients 51, conceptually similar to using O negative blood as a universal blood type in emergency situations. Thus, lessons from transplantation immunology will be critical for regenerative medicine.
We predict that advances in regenerative medicine over the next decade will arise from innovations from diverse fields, and these will mostly arise from farsighted investigators who are willing to take on new challenges that are beyond their areas of formal training. As with other competitive areas in biology, collaborative advances will become the norm, rather than individually driven advances. Young trainees who see this convergence of fields and disciplines will be the most successful in pushing regenerative approaches toward reality. Training programs that promote a cross-fertilization between fields—and encourage some “lack of focus” that we commonly advise against in academic medicine—may find that they are more likely to give rise to the next generation’s innovators.
Prediction 5: Therapy with combinations of cells will become necessary
Currently, the overwhelming focus in cardiovascular regenerative biology is on the cardiomyocyte. Regenerating damaged heart tissue will obviously require new cardiomyocyte generation, and thus getting new cardiomyocytes into myocardium, or driving intrinsic generation of new cardiomyocytes, is a central goal for the regenerative medicine approach to systolic heart failure. However, over the next decade, we will see much more attention to non-cardiomyocytes in the heart regeneration field. It has long been known that interactions with fibroblast and endothelial cells are important for normal cardiomyocyte function 52–54. Just as vascular smooth muscle and endothelium participate in an ongoing regulation of vascular tone, cardiac function relies on normal function of diverse cell types and a more holistic approach will undoubtedly be required.
Prediction 6: Cardiovascular diseases that currently seem beyond regenerative medicine will become viable targets
We began with the prediction that non-regenerative treatments like devices and drugs for some cardiovascular conditions will be sufficiently successful that regenerative medicine approaches will not be commercially viable or will not be needed at all. Here we predict the converse: Many diseases that currently seem beyond regenerative biology will become regenerative medicine targets, because there will be major needs that cannot be met by conventional pharmacological or surgical approaches. For example, myocarditis treatments developed from stem cell-based models, complex congenital heart diseases repaired by a combination of surgery and gene editing, engineered microvessels for intractable peripheral vascular disease and pulmonary hypertension treated by regrowth of intact pulmonary units—these are just a few of the areas where regenerative biology approaches could have an impact. As we develop cell-based models of disease that come closer to human tissue generation, new approaches to problems that now seem daunting will emerge.
Conclusion
Here we have attempted to make predictions based on our current understanding of where the disciplines of regenerative biology and stem cell biology are taking us. But what will be the first cardiovascular regenerative breakthrough therapy, and when will it occur? Can we predict which cell therapy strategy will improve heart function in a large number of patients?
We reluctantly predict that most rigorously performed and adequately powered clinical trials of cardiac cell therapy will yield null results in the upcoming decade. We are doubtful about early attempts to regenerate the heart not because we feel the pioneers in human cardiac cell therapy are being hasty, but because early failure is very often the outcome for initial attempts of a new approach. Cardiac transplantation, coronary stents and the initial attempts at human gene therapy are just a few examples of new technologies with sobering initial outcomes that were then followed by dramatic success. To the credit of investigators who have been experimenting with human cardiac cell therapy, adverse events have been impressively rare, demonstrating their attention to safety in these early years. Over the next decade, we will learn from human attempts at cardiac cell therapy—either that it is beneficial in some form, or that we must know much more about cardiac biology before we can successfully regenerate myocardium in humans.
While no one truly knows if human cardiac cell therapy may benefit patients in the next decade, we can comfortably predict that stem cell biology will shape regenerative medicine not only for the next ten years, but also far beyond that. The ability to control cells in ways we previously couldn’t imagine has changed our view of biology itself. There is enormous work to be done, in order to control cell fate as precisely as we can target the beta adrenergic receptor with beta blockers, to generate cells as reliably as we can generate small molecule drugs like statins, and to learn to edit genomes as adroitly as we reprogram pacemakers—to name just a few of the challenges ahead. For cardiovascular patients, regenerative biology may not turn the corner into regenerative bedside medicine in the next decade, but it will do this during the first half of this century. For those of us fortunate enough to witness the breakthroughs, it will be as amazing as watching a child grow.
Acknowledgments
We thank Brock Reeve of the Harvard Stem Cell Institute for his helpful comments.
Funding Sources
This work was supported in part by NIH grants (HL117986, HL122987, AG047131 to RTL) and (HL120160, HL116591 and P01HL081587 to KW) and Fondation Leducq (to RTL).
Footnotes
Journal Codes: [16] Myocardial cardiomyopathy disease; [137] Cell biology/structural biology
Conflict of Interest Disclosures
Dr. Lee is a consultant to Merck, Pfizer, Dyax and Mesoblast. Dr. Lee is founder and consultant to ProteoThera, Inc. Dr. Walsh has sponsored research from Takeda Pharmaceuticals Company.
References
- 1.Rink JC. Stem cell systems and regeneration in planaria. Development genes and evolution. 2013;223:67–84. doi: 10.1007/s00427-012-0426-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Goss RJ. Principles of regeneration. New York: Academic Press; 1969. [Google Scholar]
- 3.Behfar A, Crespo-Diaz R, Terzic A, Gersh BJ. Cell therapy for cardiac repair–lessons from clinical trials. Nature reviews Cardiology. 2014;11:232–46. doi: 10.1038/nrcardio.2014.9. [DOI] [PubMed] [Google Scholar]
- 4.Rosen MR, Myerburg RJ, Francis DP, Cole GD, Marban E. Translating stem cell research to cardiac disease therapies: pitfalls and prospects for improvement. Journal of the American College of Cardiology. 2014;64:922–37. doi: 10.1016/j.jacc.2014.06.1175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Telukuntla KS, Suncion VY, Schulman IH, Hare JM. The advancing field of cell-based therapy: insights and lessons from clinical trials. Journal of the American Heart Association. 2013;2:e000338. doi: 10.1161/JAHA.113.000338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Evans MJ, Kaufman MH. Establishment in culture of pluripotential cells from mouse embryos. Nature. 1981;292:154–6. doi: 10.1038/292154a0. [DOI] [PubMed] [Google Scholar]
- 7.Martin GR. Isolation of a pluripotent cell line from early mouse embryos cultured in medium conditioned by teratocarcinoma stem cells. Proceedings of the National Academy of Sciences of the United States of America. 1981;78:7634–8. doi: 10.1073/pnas.78.12.7634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Thomson JA, Itskovitz-Eldor J, Shapiro SS, Waknitz MA, Swiergiel JJ, Marshall VS, Jones JM. Embryonic stem cell lines derived from human blastocysts. Science. 1998;282:1145–7. doi: 10.1126/science.282.5391.1145. [DOI] [PubMed] [Google Scholar]
- 9.McCarthy M. US researchers push past stem-cell restrictions. States and private sources fund research denied federal support by Bush ban. Lancet. 2004;363:868–9. doi: 10.1016/s0140-6736(04)15775-9. [DOI] [PubMed] [Google Scholar]
- 10.He JQ, Ma Y, Lee Y, Thomson JA, Kamp TJ. Human embryonic stem cells develop into multiple types of cardiac myocytes: action potential characterization. CircRes. 2003;93:32–39. doi: 10.1161/01.RES.0000080317.92718.99. [DOI] [PubMed] [Google Scholar]
- 11.Murry CE, Keller G. Differentiation of embryonic stem cells to clinically relevant populations: lessons from embryonic development. Cell. 2008;132:661–80. doi: 10.1016/j.cell.2008.02.008. [DOI] [PubMed] [Google Scholar]
- 12.Laflamme MA, Chen KY, Naumova AV, Muskheli V, Fugate JA, Dupras SK, Reinecke H, Xu C, Hassanipour M, Police S, O’Sullivan C, Collins L, Chen Y, Minami E, Gill EA, Ueno S, Yuan C, Gold J, Murry CE. Cardiomyocytes derived from human embryonic stem cells in pro-survival factors enhance function of infarcted rat hearts. Nature biotechnology. 2007;25:1015–24. doi: 10.1038/nbt1327. [DOI] [PubMed] [Google Scholar]
- 13.Chong JJ, Yang X, Don CW, Minami E, Liu YW, Weyers JJ, Mahoney WM, Van Biber B, Cook SM, Palpant NJ, Gantz JA, Fugate JA, Muskheli V, Gough GM, Vogel KW, Astley CA, Hotchkiss CE, Baldessari A, Pabon L, Reinecke H, Gill EA, Nelson V, Kiem HP, Laflamme MA, Murry CE. Human embryonic-stem-cell-derived cardiomyocytes regenerate non-human primate hearts. Nature. 2014;510:273–7. doi: 10.1038/nature13233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Gerbin KA, Murry CE. The winding road to regenerating the human heart. Cardiovascular pathology : the official journal of the Society for Cardiovascular Pathology. 2015;24:133–140. doi: 10.1016/j.carpath.2015.02.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.van Berlo JH, Molkentin JD. An emerging consensus on cardiac regeneration. Nature medicine. 2014;20:1386–93. doi: 10.1038/nm.3764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Takahashi K, Yamanaka S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell. 2006;126:663–76. doi: 10.1016/j.cell.2006.07.024. [DOI] [PubMed] [Google Scholar]
- 17.Gurdon JB, Elsdale TR, Fischberg M. Sexually mature individuals of Xenopus laevis from the transplantation of single somatic nuclei. Nature. 1958;182:64–5. doi: 10.1038/182064a0. [DOI] [PubMed] [Google Scholar]
- 18.Braam SR, Passier R, Mummery CL. Cardiomyocytes from human pluripotent stem cells in regenerative medicine and drug discovery. Trends Pharmacol Sci. 2009;30:536–45. doi: 10.1016/j.tips.2009.07.001. [DOI] [PubMed] [Google Scholar]
- 19.Zhang J, Wilson GF, Soerens AG, Koonce CH, Yu J, Palecek SP, Thomson JA, Kamp TJ. Functional cardiomyocytes derived from human induced pluripotent stem cells. Circulation research. 2009;104:e30–41. doi: 10.1161/CIRCRESAHA.108.192237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kamao H, Mandai M, Okamoto S, Sakai N, Suga A, Sugita S, Kiryu J, Takahashi M. Characterization of human induced pluripotent stem cell-derived retinal pigment epithelium cell sheets aiming for clinical application. Stem Cell Reports. 2014;2:205–18. doi: 10.1016/j.stemcr.2013.12.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Addis RC, Epstein JA. Induced regeneration–the progress and promise of direct reprogramming for heart repair. Nature medicine. 2013;19:829–36. doi: 10.1038/nm.3225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Zhou Q, Brown J, Kanarek A, Rajagopal J, Melton DA. In vivo reprogramming of adult pancreatic exocrine cells to beta-cells. Nature. 2008;455:627–32. doi: 10.1038/nature07314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Amamoto R, Arlotta P. Development-inspired reprogramming of the mammalian central nervous system. Science. 2014;343:1239882. doi: 10.1126/science.1239882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Han JK, Chang SH, Cho HJ, Choi SB, Ahn HS, Lee J, Jeong H, Youn SW, Lee HJ, Kwon YW, Cho HJ, Oh BH, Oettgen P, Park YB, Kim HS. Direct conversion of adult skin fibroblasts to endothelial cells by defined factors. Circulation. 2014;130:1168–78. doi: 10.1161/CIRCULATIONAHA.113.007727. [DOI] [PubMed] [Google Scholar]
- 25.Qian L, Huang Y, Spencer CI, Foley A, Vedantham V, Liu L, Conway SJ, Fu JD, Srivastava D. In vivo reprogramming of murine cardiac fibroblasts into induced cardiomyocytes. Nature. 2012;485:593–8. doi: 10.1038/nature11044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Song K, Nam YJ, Luo X, Qi X, Tan W, Huang GN, Acharya A, Smith CL, Tallquist MD, Neilson EG, Hill JA, Bassel-Duby R, Olson EN. Heart repair by reprogramming non-myocytes with cardiac transcription factors. Nature. 2012;485:599–604. doi: 10.1038/nature11139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Mandel Y, Weissman A, Schick R, Barad L, Novak A, Meiry G, Goldberg S, Lorber A, Rosen MR, Itskovitz-Eldor J, Binah O. Human embryonic and induced pluripotent stem cell-derived cardiomyocytes exhibit beat rate variability and power-law behavior. Circulation. 2012;125:883–93. doi: 10.1161/CIRCULATIONAHA.111.045146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Nallamothu BK, Bates ER, Wang Y, Bradley EH, Krumholz HM. Driving times and distances to hospitals with percutaneous coronary intervention in the United States: implications for prehospital triage of patients with ST-elevation myocardial infarction. Circulation. 2006;113:1189–95. doi: 10.1161/CIRCULATIONAHA.105.596346. [DOI] [PubMed] [Google Scholar]
- 29.Desta L, Jernberg T, Lofman I, Hofman-Bang C, Hagerman I, Spaak J, Persson H. Incidence, Temporal Trends, and Prognostic Impact of Heart Failure Complicating Acute Myocardial Infarction: The SWEDEHEART Registry (Swedish Web-System for Enhancement and Development of Evidence-Based Care in Heart Disease Evaluated According to Recommended Therapies): A Study of 199,851 Patients Admitted With Index Acute Myocardial Infarctions, 1996 to 2008. JACC Heart failure. 2015;3:234–42. doi: 10.1016/j.jchf.2014.10.007. [DOI] [PubMed] [Google Scholar]
- 30.Menasche P, Vanneaux V, Hagege A, Bel A, Cholley B, Cacciapuoti I, Parouchev A, Benhamouda N, Tachdjian G, Tosca L, Trouvin JH, Fabreguettes JR, Bellamy V, Guillemain R, Suberbielle Boissel C, Tartour E, Desnos M, Larghero J. Human embryonic stem cell-derived cardiac progenitors for severe heart failure treatment: first clinical case report. Eur Heart J. 2015;36:2011–2017. doi: 10.1093/eurheartj/ehv189. [DOI] [PubMed] [Google Scholar]
- 31.Braunwald E. The Path to an Angiotensin Receptor Antagonist-Neprilysin Inhibitor in the Treatment of Heart Failure. Journal of the American College of Cardiology. 2015;65:1029–1041. doi: 10.1016/j.jacc.2015.01.033. [DOI] [PubMed] [Google Scholar]
- 32.Itzhaki I, Maizels L, Huber I, Zwi-Dantsis L, Caspi O, Winterstern A, Feldman O, Gepstein A, Arbel G, Hammerman H, Boulos M, Gepstein L. Modelling the long QT syndrome with induced pluripotent stem cells. Nature. 2011;471:225–9. doi: 10.1038/nature09747. [DOI] [PubMed] [Google Scholar]
- 33.Lander ES. Initial impact of the sequencing of the human genome. Nature. 2011;470:187–97. doi: 10.1038/nature09792. [DOI] [PubMed] [Google Scholar]
- 34.Bjorkegren JL, Kovacic JC, Dudley JT, Schadt EE. Genome-Wide Significant Loci: How Important Are They?: Systems Genetics to Understand Heritability of Coronary Artery Disease and Other Common Complex Disorders. Journal of the American College of Cardiology. 2015;65:830–845. doi: 10.1016/j.jacc.2014.12.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Eschenhagen T, Mummery C, Knollmann BC. Modelling sarcomeric cardiomyopathies in the dish: from human heart samples to iPSC cardiomyocytes. Cardiovascular research. 2015;105:424–38. doi: 10.1093/cvr/cvv017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Masumoto H, Ikuno T, Takeda M, Fukushima H, Marui A, Katayama S, Shimizu T, Ikeda T, Okano T, Sakata R, Yamashita JK. Human iPS cell-engineered cardiac tissue sheets with cardiomyocytes and vascular cells for cardiac regeneration. Scientific reports. 2014;4:6716. doi: 10.1038/srep06716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Terrenoire C, Wang K, Tung KW, Chung WK, Pass RH, Lu JT, Jean JC, Omari A, Sampson KJ, Kotton DN, Keller G, Kass RS. Induced pluripotent stem cells used to reveal drug actions in a long QT syndrome family with complex genetics. The Journal of general physiology. 2013;141:61–72. doi: 10.1085/jgp.201210899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Hinson JT, Chopra A, Nafissi N, Polacheck WJ, Benson CC, Swist S, Gorham J, Yang L, Schafer S, Sheng CC, Haghighi A, Homsy J, Hubner N, Church G, Cook SA, Linke WA, Chen CS, Seidman JG, Seidman CE. HEART DISEASE. Titin mutations in iPS cells define sarcomere insufficiency as a cause of dilated cardiomyopathy. Science. 2015;349:982–6. doi: 10.1126/science.aaa5458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Doudna JA, Charpentier E. Genome editing. The new frontier of genome engineering with CRISPR-Cas9. Science. 2014;346:1258096. doi: 10.1126/science.1258096. [DOI] [PubMed] [Google Scholar]
- 40.Doudna JA. Genomic engineering and the future of medicine. Jama. 2015;313:791–2. doi: 10.1001/jama.2015.287. [DOI] [PubMed] [Google Scholar]
- 41.Ding Q, Strong A, Patel KM, Ng SL, Gosis BS, Regan SN, Cowan CA, Rader DJ, Musunuru K. Permanent alteration of PCSK9 with in vivo CRISPR-Cas9 genome editing. Circulation research. 2014;115:488–92. doi: 10.1161/CIRCRESAHA.115.304351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Tebas P, Stein D, Tang WW, Frank I, Wang SQ, Lee G, Spratt SK, Surosky RT, Giedlin MA, Nichol G, Holmes MC, Gregory PD, Ando DG, Kalos M, Collman RG, Binder-Scholl G, Plesa G, Hwang WT, Levine BL, June CH. Gene editing of CCR5 in autologous CD4 T cells of persons infected with HIV. The New England journal of medicine. 2014;370:901–10. doi: 10.1056/NEJMoa1300662. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Baltimore D, Berg P, Botchan M, Carroll D, Charo RA, Church G, Corn JE, Daley GQ, Doudna JA, Fenner M, Greely HT, Jinek M, Martin GS, Penhoet E, Puck J, Sternberg SH, Weissman JS, Yamamoto KR. Biotechnology. A prudent path forward for genomic engineering and germline gene modification. Science. 2015;348:36–8. doi: 10.1126/science.aab1028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Lanphier E, Urnov F, Haecker SE, Werner M, Smolenski J. Don’t edit the human germ line. Nature. 2015;519:410–1. doi: 10.1038/519410a. [DOI] [PubMed] [Google Scholar]
- 45.Long C, McAnally JR, Shelton JM, Mireault AA, Bassel-Duby R, Olson EN. Prevention of muscular dystrophy in mice by CRISPR/Cas9-mediated editing of germline DNA. Science. 2014;345:1184–8. doi: 10.1126/science.1254445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Bergmann O, Bhardwaj RD, Bernard S, Zdunek S, Barnabe-Heider F, Walsh S, Zupicich J, Alkass K, Buchholz BA, Druid H, Jovinge S, Frisen J. Evidence for cardiomyocyte renewal in humans. Science. 2009;324:98–102. doi: 10.1126/science.1164680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Lee J, Sayed N, Hunter A, Au KF, Wong WH, Mocarski ES, Pera RR, Yakubov E, Cooke JP. Activation of innate immunity is required for efficient nuclear reprogramming. Cell. 2012;151:547–58. doi: 10.1016/j.cell.2012.09.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Sayed N, Wong WT, Ospino F, Meng S, Lee J, Jha A, Dexheimer P, Aronow BJ, Cooke JP. Transdifferentiation of human fibroblasts to endothelial cells: role of innate immunity. Circulation. 2015;131:300–9. doi: 10.1161/CIRCULATIONAHA.113.007394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Zakrzewski JL, van den Brink MR, Hubbell JA. Overcoming immunological barriers in regenerative medicine. Nature biotechnology. 2014;32:786–94. doi: 10.1038/nbt.2960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Zhao T, Zhang ZN, Rong Z, Xu Y. Immunogenicity of induced pluripotent stem cells. Nature. 2011;474:212–5. doi: 10.1038/nature10135. [DOI] [PubMed] [Google Scholar]
- 51.Riolobos L, Hirata RK, Turtle CJ, Wang PR, Gornalusse GG, Zavajlevski M, Riddell SR, Russell DW. HLA engineering of human pluripotent stem cells. Molecular therapy : the journal of the American Society of Gene Therapy. 2013;21:1232–41. doi: 10.1038/mt.2013.59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Hsieh PC, Davis ME, Lisowski LK, Lee RT. Endothelial-cardiomyocyte interactions in cardiac development and repair. Annual review of physiology. 2006;68:51–66. doi: 10.1146/annurev.physiol.68.040104.124629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Kakkar R, Lee RT. Intramyocardial fibroblast myocyte communication. Circulation research. 2010;106:47–57. doi: 10.1161/CIRCRESAHA.109.207456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Walsh K, Shiojima I. Cardiac growth and angiogenesis coordinated by intertissue interactions. The Journal of clinical investigation. 2007;117:3176–9. doi: 10.1172/JCI34126. [DOI] [PMC free article] [PubMed] [Google Scholar]


