Abstract
Emergency departments (ED) in the United States see over eight million cases of chest pain annually. While a cardinal symptom of acute coronary syndrome (ACS), multiple emergent and non-emergent causes can attribute to chest pain. This case-based perspective describes the different sex-specific causes of angina seen in ED patients. Once coronary artery disease (CAD) is ruled out with standard protocols, microvascular dysfunction is perhaps the most prevalent but under-diagnosed cause of non-CAD related angina in ED patients. Additional causes include coronary artery spasm, coronary artery dissection, coronary artery endothelial dysfunction and myocardial bridging. Non-CAD related angina is associated with persistent chest pain causing poor function, quality of life, and recidivism. Clinicians should consider additional diagnostics to routinely screen for non-CAD related causes of angina in patients with recurrent chest pain. Future work is needed to better define the epidemiological, clinical, biological, and genetic correlates of microvascular dysfunction in these patients.
Keywords: chest pain, women, sex, gender, microvascular disease
Case Presentation of Chest Pain:
History: A 37-year old female with history of migraine and recent diagnosis of hypertension presented to the ED complaining of chest pressure and dyspnea on exertion for two weeks prior to presentation. She described pressure in the sternal area of her chest, constant, 7/10, radiating to her back, her jaw and her right arm. Patient reported being admitted to a nearby hospital with similar symptoms a month earlier. She had a transient left bundle branch block (LBBB) on her electrocardiogram and was worked up for acute coronary syndrome. Her cardiac biomarkers were negative, and she underwent a stress echocardiogram during which she exercised for 6 minutes on a Bruce protocol, achieving 7.1 metabolic equivalents (METS), and had normal left ventricular chamber size diminution and a normal baseline ejection fraction (~65 percent). Exercise was stopped secondary to fatigue, and the only concerning finding was that she had septal hypokinesis that was read as possible ischemia versus a consequence of her LBBB.
On the basis of her abnormal stress echocardiogram she also underwent stress perfusion imaging that showed possible anterior ischemia – again in the setting of her LBBB. On this basis she underwent a coronary angiography at the local hospital that revealed no obstructive coronary disease. She was discharged home, with a diagnosis of non-cardiac chest pain.
Over the next week, patient’s symptoms worsened with chest pain radiating down right arm, associated with profound fatigue, dizziness, lightheadedness, tingling in the jaw. These symptoms disrupted her work as a postal worker and brought her to the second emergency department for evaluation.
Past Surgical History: Right lower leg surgery for fracture, left ear surgery, laparoscopy, tonsillectomy.
Medications: Aspirin 81 mg, multivitamins.
Allergies: Bactrim, cephalosporins.
Family History: Mother had angioplasty at age 39, maternal grandfather with MI at 38, deceased from myocardial infarction (MI) at 42. Second cousin passed from massive MI at age 45.
Social History: Denies any current/past use/abuse of tobacco, alcohol, or illicit drugs. She was married with one child and worked full time.
Height/Weight/BMI: 5'4", 160 pounds, body mass index of 27.5.
Physical Exam: Patient appeared well in no distress. Vitals: Blood pressure 113/63 | Pulse 85 | Temperature 98.9 °Fahrenheit (Oral) | Respiration 18 | Oxygen saturation 98%. She had no jugular venous distention. Cardiovascular exam showed normal rate, regular rhythm, normal heart sounds and intact distal pulses. Chest exam showed normal effort and breath sounds normal. She exhibited no chest wall tenderness. Her neurological exam showed alert woman with normal speech and no focal findings.
Patient was admitted to the ED chest pain observation unit for further evaluation.
Introduction
Chest pain is the second most common presenting complaint of US emergency department patients over 15 years of age, accounting for over 8 million annual visits [1]. While a cardinal symptom of acute coronary syndrome and MI, multiple other emergent and non-emergent causes can attribute to chest pain. Women are more likely to present with the complaint of chest pain than men [1]. However CAD occurs more commonly and about a decade earlier in men as compared to women [2]. Identifying patients with ACS early is critical to initiate timely interventions and to prevent unnecessary morbidity and mortality. Thus, EDs often have extensive evidence-based policies and procedures to diagnose and rule out ACS. We now know that sex-specific differences exist in all aspects of ACS ranging from the pathophysiology of disease, presentation, treatment and outcomes. Though improvements have occurred in recent years, women with MI are found to have worse prognosis as compared to men. Independent of age, more women than men (26 percent versus 19 percent respectively) die within first year of a myocardial infarction [3]. This difference is accentuated among young women as compared to age-matched men [4]. Women are more vulnerable to slower diagnosis and inadequate treatment [5]. Also there has been a lag in identifying sex-specific evidence for diagnostics and therapeutics for women. This is partly due to the fact that women were excluded from research for a long time in an attempt to protect them from inadvertent adverse events. As a result, much of the evidence for treating heart disease has been tested primarily on men. Only a fifth of participants enrolled in clinical trials are women, while half of the participants in registries of myocardial infarction are usually women [6]. Even when included, sex-specific analysis is not routinely conducted to understand the differences in results based on biology. The National Institute of Health (NIH) introduced a policy to include women in clinical research in 1986. This was later mandated by law through the NIH Revitalization Act 1993 entitled Women and Minorities as Subjects in Clinical Research. The law reinforced the previous policy but included call for active outreach efforts to enroll women and minorities. Finally in 2014, NIH further addressed this gap by mandating all basic science research to include both male and female cells [7]. Since then, the number of women enrolled in clinical trials has increased but has stalled around 20 percent despite good efforts [8].
While mortality has decreased over the past four decades for both men and women, mortality for women aged 45 to 54 years has actually increased over the past several years [9]. Black and Hispanic women are at even greater risk for heart attacks and have poorer outcomes [3]. Other than mortality, women more often than men have recurrent chest pain that brings them back to the emergency departments. These persistent symptoms have been associated with high rates of recidivism, costs and poor functional outcomes [10-17]. It is important to understand these sex-specific variations in presentation and pathophysiology of angina in chest pain patients to adequately address the discrepancies in outcomes.
The Common Face of Angina in Men and Women
Of the nearly eight million visits to the ED, 15 to 20 percent visits are attributed to angina from ACS. These include ST-elevation myocardial infarction (STEMI) (5 percent), Non-ST elevation MI (NSTEMI) (10 to 15 percent) and unstable angina (5 percent). The remaining 80 percent of the patients have a range of causes (Table 1). The sex-distribution of these causes vary, and are important considerations in the evaluation of these patients in the ED.
Table 1. Sex-specific distribution for causes of chest pain in the ED.
| Women | Men | |
| Cardiac CAD related | ||
| Myocardial infarction | Yes | |
| Unstable angina | Yes | |
| Cardiac non-CAD related | ||
| Coronary spasm | Yes | |
| Coronary artery dissection | Yes | |
| Myocardial bridging | Yes | |
| Microvascular dysfunction | Yes | |
| Cardiac non ischemic | ||
| Myopericarditis | Yes | |
| Aortic dissection | Yes | |
| Valvular conditions | Yes | |
| Heart failure | Yes | |
| Takotsubo | Yes | |
| Non cardiac | ||
| Gastrointestinal | ||
| Reflux | Yes | Yes |
| Esophageal spasm | Yes | Yes |
| Esophageal perforation | Yes | |
| PUD | Yes | |
| Pulmonary | ||
| Pneumonia | Yes | |
| Pulmonary embolism | Yes | |
| Pleuritis | Yes | |
| Neoplasm | Yes | |
| Bronchitis | Yes | Yes |
| Pneumothorax | Yes | |
| Musculoskeletal | Yes | |
| Herpes Zoster | Yes | |
| Psychosomatic | Yes |
The classic symptoms of ACS were described by Samuel Levine in 1962 [18]. These symptoms described a man with clenched fist clutching his chest to signify crushing squeezing substernal chest discomfort that radiated to the arm, neck or jaw and was accompanied with diaphoresis, nausea and shortness of breath. These symptoms were dubbed as ‘typical’ presentation of myocardial infarction.
Since the Levine’s sign, we have documented other presentations of MI. These range from sharp pleuritic, burning or reproducible chest pain, to epigastric pain, unusual fatigue, dizziness, feeling of doom and generalized weakness that are considered ‘atypical’ symptoms of MI.
Most studies indicate that both men and women present mostly commonly with chest discomfort [19]. This was also seen in young men and women with MI (< 55 years of age) [20]. However the description of chest pain can differ and may not be the classic Levine’s sign that was originally described in a man. [21]. We prefer the term ‘chest discomfort’ as some patients (typically women) decline calling it ‘pain’ when presenting to the ED and prefer the word ‘discomfort’ in their chest. Women in general complain of atypical symptoms more frequently and also describe a larger number of symptoms than men [22]. In fact, a segment of women especially < 45 years of age with MI may not have chest pain at all. In a registry based review of about a million patients with MI, a third were found to have no chest pain. This occurs more commonly in women as compared to men (42.0 percent vs 30.7 percent) and more often with NSTEMI [23]. Such patients are important to recognize as they have higher mortality as compared to patients who present with chest pain [23]. Variation in clinical presentation or pathophysiology could explain some of the sex-specific difference in mortality. It could explain delay in presentation to a hospital as well as higher threshold for further tests such as coronary angiography in women because of underestimation of their risk. In addition, gender might also play a role as ‘feminine traits’ have been associated with higher rates of recurrent ACS, particularly in young patients with MI [24]. The case presentation described above narrates the ‘typical atypical’ symptoms that are seen in women presenting with chest pain to the ED.
Typical angina occurs when blood flow through the coronary artery is restricted due to an anatomical restriction. When the focal narrowing of the main coronary artery exceeds 50 percent in the left main coronary or >70 percent of any epicardial artery or its branch, it is labeled as obstructive coronary artery disease [25]. Myocardial infarction in these patients occurs as a result of plaque rupture or plaque erosion. Plaque rupture with a culprit lesion is seen more commonly in men and in postmenopausal women, especially those with fatal MI. However plaque erosion is more commonly seen in younger women with MI and those with non-obstructive CAD (< 50 percent stenosis) [26]. This heterogeneity in pathophysiology of classic infarction is seen more often in women as compared to men. A systematic study of young men and women with MI revealed that 1 in 8 women did not fit the classic pathophysiology of MI. More sex-specific work is needed to better define the underlying mechanisms for these differences [27].
The Uncommon Faces of Angina Seen in the Emergency Department
The current focus of ED management is to correctly identify angina representing obstructive CAD in patients presenting with chest pain. This is because missing patients with ACS has been linked with high short-term mortality [28]. As seen with our case presentation, some patients present again with recurrent symptoms after a comprehensive work up for acute ischemia. Our experience indicates that up to a third of chest pain patients discharged from an ED after a negative work up have recurrent chest pain within a month and about 10 to 15 percent return for further testing [29-30]. These patients represent a heterogeneous group of causes (Table 1). An astute clinician has to consider these diagnoses while evaluating a patient in the ED with chest pain. This is where a careful history and exam is very helpful. When whittled down to a few, laboratory markers and diagnosis focused imaging can help further differentiate the various causes of chest pain.
Once obstructive CAD has been ruled out, an ED physician should consider other sex-specific causes of angina through invasive or non-invasive imaging. Figure 1 provides a quick overview of the causes of angina. While obstructive CAD is seen more commonly in men, alternate forms of angina are more commonly seen in women. These sex-specific mechanisms are summarized below:
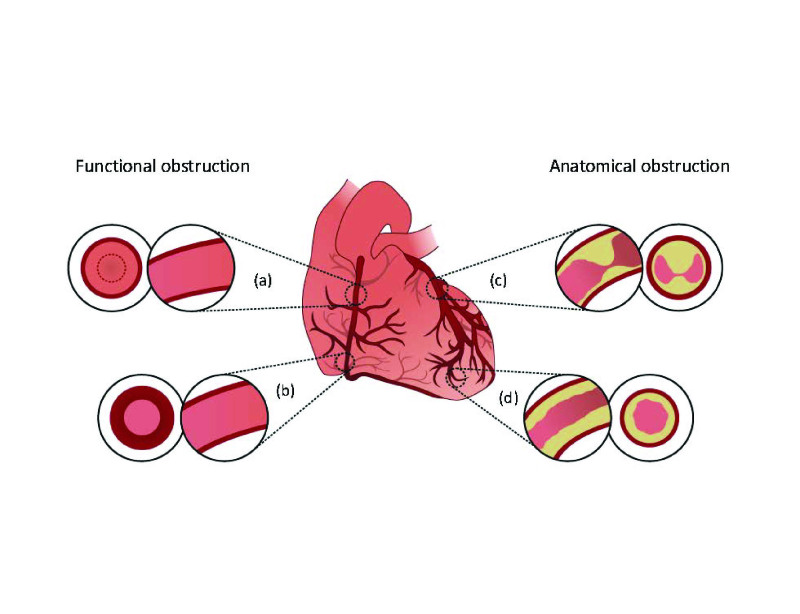
Figure 1.
Simple pictorial representation of sex-specific pathophysiology of angina. Varied mechanisms include: (a) Endothelial dysfunction of coronary artery; (b) coronary artery spasm; (c) focal luminal narrowing of coronary artery (obstructive coronary artery disease, coronary artery dissection, myocardial bridging); and (d) diffuse narrowing of microvessels (< 300 μm) causing microvascular dysfunction (structural, functional or autonomic mismatch).
Coronary artery spasm can sometimes cause angina and rarely infarction. It is thought to occur more commonly in women, in smokers and in cocaine users. When presenting as ACS, 25 percent of these patients may not have culprit lesion at the site of the spasm [31]. The underlying pathophysiology is thought to be autonomic imbalance and hence this is often triggered by a hyperdynamic state, exercise or emotional stress [32]. Diagnosis is often suspected on the basis of positive biomarker or electrocardiogram as ACS and definitive diagnosis is made on coronary angiography when coronaries are found to be clean. During angiography, these spasms can be induced by administering intracoronary acetylcholine and can be relieved by nitroglycerin. Treatment is usually calcium channel blockers.
Coronary artery dissection is a rare cause of MI (< 1 percent of all patients undergoing angiography). However it predominantly occurs in female, typically in < 50 years of age, in peripartum period and is associated with connective tissue disorders. The left anterior descending artery (LAD) is most often involved. Coronary artery dissection should be suspected in young females presenting with MI who lack classic factors for MI, especially in the peripartum period [5]. Mortality is low in these patients however 17 percent are associated with recurrent dissection [33]. Evidence for best treatment is controversial due to rarity of this condition.
Myocardial bridging: This uncommon anomaly is more often seen in women and approximates < 5 percent of all patients undergoing angiography. This is an anatomic variant created when the coronary artery (typically LAD) tunnels through a segment of myocardium. As a result, it can get compressed during systolic compression. External compression along with delayed diastolic relaxation can limit blood flow to the distal myocardium during times of increased demand causing angina. Diagnosis is suspected on the basis of symptoms, low positive biomarker or abnormal stress test that shows regional defect without calcification. Angina is classically worsened by intracoronary nitroglycerin. A computed tomography angiography (CTA), coronary MRI or angiography helps make the definitive diagnosis. Initial treatment is beta-blockers or calcium channel blockers. If it fails, coronary artery bypass surgery or myomectomy is preferred over coronary stents [34].
Coronary artery endothelial dysfunction: In the absence of anatomical obstruction, some patients can have abnormal vasoreactivity of the coronary artery (conduit artery) due to abnormal endothelial function. This results in vasoconstrictive response to stress as opposed to vasodilation. This functional obstruction can impede blood flow and cause angina in a segment of these patients. Presenting as ACS, these patients are typically diagnosed by administering intracoronary acetylcholine. Treatment is typically geared towards underlying risk factors for atherosclerosis and symptom management with calcium channel blockers.
Microvascular dysfunction: This includes a group of syndromes that affect smaller arterioles in the myocardium (< 350 um) as opposed to the conduit coronary artery. Our experience shows that microvascular dysfunction may be the cause of angina in up to 40 percent of ED patients with recurrent symptoms, particularly in women [30]. The syndrome was long thought to be unique to women. This is primarily because the role of this disease in chest pain was first investigated in women only [35]. However we have noted it to be present in both women and in men while remaining a female predominant condition (5:1 ratio) [36-38]. A recent study measured coronary flow reserve (CFR) and microcirculatory resistance in 139 patients without obstructive coronary artery disease established through coronary angiography and found microvascular dysfunction to be present in 21 percent of the patients [39]. The NIH sponsored Women’s Ischemic Syndrome Evaluation (WISE) group showed that microvascular angina is a source of persistent chest pain in almost half of the women with clean coronaries on angiography [10,40]. In a 20-year old cohort undergoing coronary angiography, Sara et al found an even higher percentage of microvascular abnormalities (63 percent)in patients (predominantly women) with non-obstructive CAD using additional provocative testing [41]. We have found high rates of this condition in the low-moderate risk ED population (38 percent) and recommend routine screening of patients with recurrent chest pain for microvascular angina.
Angina in patients with microvascular dysfunction occurs due to reduced blood flow that could be from diffuse thickening of the arteriole, endothelial dysfunction or from increased resistance in the microcirculation. One of the challenges of advancing the field of microvascular angina has been lack of a uniform classification [21]. It is distinct from syndrome X. The classic syndrome X or Prinzmetal angina was attributed to spasm of the epicardial disease as opposed to disease of the small vessels. This is an important distinction as the diagnosis, therapy and prognosis of the two conditions differ.
The pathophysiology of angina is these patients is attributed to a response to increased myocardial demand from structural, functional and autonomic abnormalities. Small vessels are the major determinant of coronary vascular resistance. Structural causes include either altered remodeling of the microvasculature or luminal obstruction from microembolization. Functional causes include increased vasomotor resistance, endothelial dysfunction, altered vascular tone, coagulation abnormalities, and altered metabolic switch [42-46]. Autonomic causes includes enhanced adrenergic drive or impaired parasympathetic system, a reason thought to explain high rates seen in women [47-48]. Enhanced pain sensitivity is thought to be another cause of recurrent symptoms through attenuation of cardiac afferent fibers [49].
At a molecular level, microvascular angina is thought to occur due to an imbalance of vasodilators (such as reduced availability of nitric oxide) and abnormal vasoreactivity (due to increase in inflammatory markers such as endothelin factor, interleukin 1 and 6, vascular endothelial growth factor). In this context, it has been proposed that metabolic disturbances such as obesity even without diabetes can trigger oxidative stress, release circulating free fatty acids and create a proinflammatory milieu that causes insulin resistance and endothelial dysfunction leading to microvascular dysfunction [50-52]. Metalloproteinases (MMP) have also been implicated through their role in remodeling of small vessel disease in the brain [53]. The role of MMP in cardiac population has been studied in the context of hypertension, diabetic microvascular disease and in heart failure but not specifically in patients with cardiac microvascular angina. Future work evaluating the role of inflammatory markers and metalloproteinases in patients with microvascular angina is needed to better understand the pathophysiology of this disease as well as to develop screening markers for the ED.
How Patient Sex can Affect Evaluation of Chest Pain:
When a clinician is faced with evaluating chest pain, the first goal should be to rule out life threatening conditions such as ACS, pulmonary embolism or aortic dissection as was done in our case. A careful clinical assessment helps tease out the wider differential as listed in Table 1. Most patients with ACS present with chest discomfort, regardless of the type of infarction. However women more often than men, particularly those younger than 55 years of age, may present with atypical symptoms or without any chest pain [23]. It is therefore important to maintain a low index of suspicion in younger females who have Framingham risk factors for both typical and atypical presentations [19].
Patients with microvascular angina may present with atypical symptoms. Our institutional experience shows these patients describe a variety of symptoms ranging from exertional chest pain to resting chest pain with or without radiation. Emotional stress is a common trigger. Unlike obstructive CAD that is well predicted by Framingham risk factors, patients with microvascular dysfunction are less clearly defined by the same risk factors [41,54,55]. These patients also do not always have exertional symptoms and do not respond to nitroglycerin which dilates larger vessels [56]. As a result, commonly used angina evaluation scores may not apply to this population [57]. Systematic sex-specific qualitative work is needed to better measure and monitor chest pain symptoms from microvascular angina as well its predictors.
Once ruled out for obstructive CAD, if patients continue to have chest pain then a clinician should consider alternative forms of angina particularly in women where these alternate pathophysiological are more commonly seen. Epidemiologically, women tend to have lower prevalence of obstructive CAD despite having classic angina symptoms [58,59]. The gold standard is to assess these forms with invasive coronary angiography. In the absence of CAD, alternate forms of ischemia can be elicited through comprehensive testing for endothelial dysfunction (using intracoronary acetylcholine), coronary microvascular dysfunction (with index of microcirculatory resistance) and CFR (using intracoronary adenosine), occult diffuse epicardial coronary disease (using fractional flow reserve) and myocardial bridging (using intravascular ultrasound) [58]. An abnormal FFR is considered < .80. While there is good safety data for these testing during angiography, universal screening for alternate forms of ischemia has not gained popularity.
In low risk emergency department patients however invasive investigation is rarely an option. A suitable alternative would be to assess CFR in these patients by non-invasive techniques such as cardiac positron emission tomography (PET), Doppler echocardiography or magnetic resonance imaging (MRI) [60,62]. CFR is the ratio of maximal myocardial blood flow (MBF) in response to increased oxygen demand to basal MBF (in ml/min/g). The increase in flow (normal: 2 to 4 folds) is regulated by a decrease in resistance. CFR is an integrated measure of flow through the microvessels (95 percent) and larger epicardial vessels (< 5 percent) [63]. Both global and regional CFR are calculated by dividing stress MBF by rest MBF. These are then corrected for the respective heart rate and blood pressure product.
This approach corrects for differences in resting hemodynamics and metabolic conditions between subjects, and the significant changes in the hemodynamics associated with regadenoson stress. The correction is for change in heart rate and blood pressure. Depressed CFR (< 2.5 corrected or uncorrected) [64] is indicative of reduced flow from a variety of causes such as obstructive coronary artery disease, coronary artery dissection or spasm. All these conditions would produce a regional defect and/or would be associated with coronary calcification as measured by PET/CT. Even among individuals with CAD, noninvasive imaging has documented abnormal perfusion in myocardial regions supplied by vessels without apparent obstructive CAD [65]. Without a more proximal flow-limiting stenosis, coronary microvasculature modulates 95 percent of the myocardial blood flow. Thus in the absence of regional defect or calcification, reduced coronary flow would typically signify microvascular dysfunction.
Advances in spatial and temporal resolution in cardiac MRI technology as well as lack of radiation offers a significant advantage of this modality over other non invasive techniques. However, dark rim image artifacts that are typically seen in the submyocardium by MRI can be confused with real perfusion defects [42,66]. Doppler echo and myocardial contrast echo are good alternatives. However, even in experienced hands detection of Doppler flow for LAD is possible only in 90 percent of the cases and obesity poses a technical limitation [42]. Computer Tomography-derived CFR is an upcoming technology but is currently limited by high radiation dose (11 mSv), increased contrast medium dose, beam artifacts and insufficient coverage of the heart [42].
The main advantage of 3D PET/CT is its high sensitivity, which allows precise reproducible quantification of blood flow, enables improved intrinsic spatial resolution and attenuation correction especially in heavier females, and has been linked with clinical outcomes in relation to coronary flow reserve [42,61-67]. The disadvantage is related to radiation exposure. This risk can be minimized by a) using Rubidium with a short half-life (76 seconds) that allows rapid imaging, and b) using 3D technology that reduces dose 3 fold to under 3mSv (equivalent to one year of background radiation) [72]. However, the 3D acquisition mode suffers from high random events and scatter, including scatter from activity outside the field of view, and may lead to detector saturation problems paralyzing the system. Recently, deKemp et al. demonstrated that quantitative 3D cardiac imaging appears to be accurate with low dose 82Rb activity administered [73].
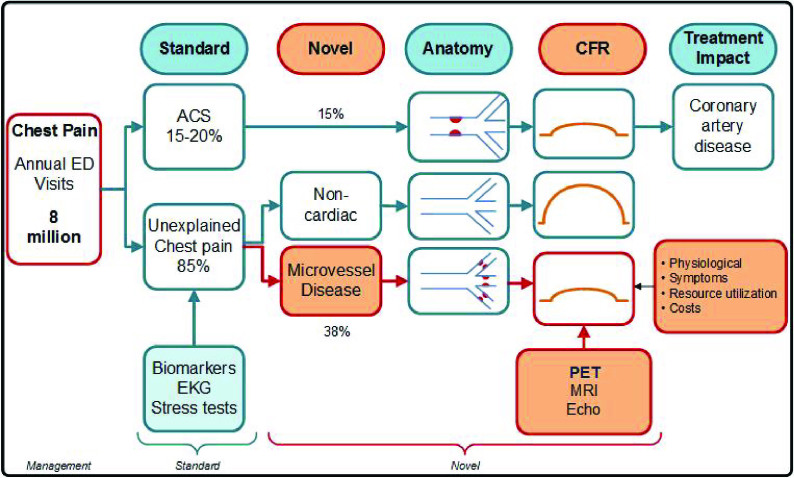
Lack of diagnosis in the face of ongoing angina symptoms can result in repeat ED visits and hospitalizations, and often cumulative unnecessary work ups [74-77]. Up to three quarters of patients with angina and non-CAD could have alternate causes of angina that should be explored further [39]. We recommend routine screening for microvascular dysfunction in the ED to help identify patients with ongoing ischemia with an aim to curtail their symptoms, related costs and recidivism through early and appropriate interventions [Figure 2].
Figure 2.
Role of screening and treatment for microvascular angina in emergency department chest pain patients.
Back to the Case — ED Course
In the ED observation unit, our 37-year old patient underwent serial cardiac biomarkers that remained negative. Her thyroid profile was normal. Her C-reactive protein was within normal limits. Lipid profile was normal with total cholesterol of 199, high-density cholesterol at 45, low-density cholesterol at 94 and triglycerides at 100. She had traveled by car about 800 miles two weeks prior to ED presentation. Given profound dyspnea she had a d-dimer checked that was positive and a CT was done that ruled out pulmonary embolism.
As we excluded the various etiologies of her chest pain, we suspected microvascular angina in our patient. She underwent a 3D 82Rb PET/CT imaging after vasodilation with Regadenoson and at rest. It showed normal global and regional left ventricular function and no calcification. This corroborated the findings from her recent angiography. Analysis of coronary flow demonstrated impaired coronary flow reserve with correction for rate pressure product of 1.8 (uncorrected CFR < 2.5). No pertinent non-cardiac findings were found.
Case Follow Up
Patient was discharged with a diagnosis of microvascular angina. On follow up, she continued to have episodes of chest discomfort, and these episodes are somewhat variable in characteristic. They sometimes occur with exertion, but not always. In addition, she has marked fatigue, and feels as though it is a chore to get through the day at work.
Given her history of migraine headache, preeclampsia, and also gives a history suggestive of Raynaud’s type symptoms involving her fingers and toes, she underwent an extensive work up for definitive clinical evidence of a systemic vasculitis, or a classic auto-immune / rheumatologic disease associated classically with vasculitis. This was negative. She had rate-related LBBB on ECG. Her non-LBBB ECG showed low-voltage in the limb leads, and non-specific ST changes anteriorily. A possibility of infiltrative process was considered but not pursued at the time given resolution of her symptoms. An outpatient echo showed normal left ventricle function and size and normal valves. She was continued on daily aspirin. She was also given a trial of Ranolazine that improved her symptoms considerably. She was discharged to her primary physician’s care. She has remained symptom free for two years since her ED visit.
Treating the Different Faces of Angina
The American Heart Association has published recommendations for obstructive CAD including STEMI reperfusion strategies including indications and time expectations for use of thrombolytics and primary Percutaneous Coronary Intervention (PCI), and for the treatment of NSTEMI including medical management and indications for early PCI [5]. Regardless of these well-disseminated guidelines, women are often undertreated and have significant delays in reperfusion, pronounced when transferred from outlying hospitals to primary PCI sites [19]. There is total agreement on aggressive management of cardiac risk factors to maximize health regardless of the underlying pathophysiological mechanism. This includes interventions for smoking cessation, reducing or maintaining a healthy weight, stress reduction, exercise, and monitoring and control of hypertension, diabetes and lipid profiles. The primary goal is to reduce morbidity, mortality and to improve the quality of life.
Evidence is sparse to guide practitioners in providing optimal treatment for microvascular angina. Recommendations for treatment of angina without CAD were last issued in 2002, as mostly level B and C evidence [78]. Importantly, current treatment strategies for obstructive CAD, such as angioplasty and stenting, are not helpful in microvascular disease. The convention is to control cardiac risk factors as they are linked with endothelial dysfunction and CFR, and determine severity of microvascular angina [79-82]. However, there is limited data to support this. As a result, despite a good prognosis, these patients return with chest pain, have poor physical function and stamina [83].
While structural and functional changes underlie microvascular angina, the first-line treatments are geared towards altering hemodynamics. They act by producing systemic vasodilation, lowering left ventricular pressure, myocardial wall tension, and reducing oxygen consumption. None have been found to be universally effective. Treatment is therefore through trial and error. The median number of subjects enrolled in studies of microvascular angina evaluating CFR is 11 [84]. Nitrates are primary venodilators and do not work on the microvessels [85]. Calcium channel blockers have some benefit although the effects have been controversial [86]. Beta-blockers may help in patients with high sympathetic tone. But their side effects including fatigue, and reduced exercise capacity is a limitation in young patients. Also their benefit in microvascular disease is limited by a vasoconstricting effect on the peripheral arteries [87-92]. Angiotension converting enzyme inhibitors have shown some benefit by reducing small vessel resistance and by vascular remodeling [93]. There is also a potential role of newer drugs such as Ranolazine and Ivabradine in these patients [94,95].
Prognosis
Over the past decade remarkable improvement in mortality has occurred for women. This is partly attributed to improved awareness and application of evidence-based therapies for typical angina. Further work is however needed to better define and manage the different faces of angina, that occur more commonly in women than in men [5].
Applying the current standard of care, most patients with microvascular angina remain undiagnosed and receive no therapy [96]. This is problematic as prognosis of patients with microvascular angina is not as benign as previously thought [77]. Symptomatic patients are at higher risk for adverse outcomes (defined as death, MI, nonfatal stroke, heart failure requiring hospitalization) compared with asymptomatic cohorts with a 2.5 percent annual event rate [35,77,97,101]. Murthy et al showed that microvascular dysfunction is a powerful and continuous predictor of major adverse cardiac events (hazard ratio, 0.80 per 10 percent increase in coronary flow reserve), even without subclinical atherosclerosis [82]. It is associated with recurrent angina leading to high costs and resource utilization [75,102,103]. The WISE study revealed that half of the patients with microvascular angina have persistent symptoms causing functional disability, repeat angiography rate as 13.2 percent and repeated hospitalization rate x 1.8 fold higher than patients with single vessel CAD [10-17]. Characterizing and defining sex-specific optimal treatment options for these patients will therefore address an important gap in current clinical practice.
Conclusion and Outlook
Chest pain in men and women can occur from a multitude of causes. These include life-threatening conditions such as acute coronary syndrome to less critical causes of angina such as microvascular dysfunction. Identifying the correct diagnosis is an essential element in providing optimal therapy to these patients. New American Heart Association guidelines provide optimal treatment guidelines for treating MI from obstructive CAD in women. It also recommends prioritizing more sex-specific work to improve recognition of alternate faces of angina particularly microvascular angina [5]. Emergency Medicine researchers have also prioritized further research in the area of microvascular angina [21]. These recommendations are in line with the NIH symposium, “Small Vessel Disease, Big Health Problems” held in September 2014. With a focus to turn discovery into health, workgroups prioritized trans-disciplinary mechanism-based treatment trials around three themes: a) identifying who/when to treat, b) development of in-vitro or in-vivo models including human cohorts and c) conducting patient driven research [104]. Routine screening of microvascular angina in our current practice is impeded by insufficient epidemiological, biological, clinical and genetic data on these patients. Future work focusing on improved diagnostics and therapeutics would help improve our understanding of some of the sex-specific mechanisms of chest pain seen in the emergency department. Any clinician who routinely evaluates patients with chest pain, has to keep abreast of the advances in sex biology order to correctly identify and treat all faces of angina in their patients.
Glossary
- ED
emergency department
- CAD
coronary artery disease
- ACS
acute coronary syndrome
- MI
Myocardial infarction
- LBBB
Left bundle branch block
- METS
Metabolic equivalent
- BP
blood pressure
- NIH
national institute of health
- STEMI
ST-evaluation myocardial infarction
- NSTEMI
non ST-elevation myocardial infarction
- CTA
computed tomography angiography
- MRI
magnetic resonance imaging
- WISE
women’s ischemic syndrome evaluation
- MBF
myocardial blood flow
- MMP
metalloproteinases
- CFR
coronary flow reserve
- PET
positron emission tomography
- LAD
left anterior descending artery
- mSv
millisievert
References
- CDC. National Hospital Ambulatory Medical Care Survey 2011. Available from: http://www.cdc.gov/nchs/data/ahcd/nhamcs_emergency/2010_ed_web_tables.pdf .
- Jousilahti P, Vartiainen E, Tuomilehto J. et al. Sex, age, cardiovascular risk factors, and coronary heart disease: a prospective follow-up study of 14 786 middle-aged men and women in Finland. Circulation. 1999;99(9):1165–1172. doi: 10.1161/01.cir.99.9.1165. [DOI] [PubMed] [Google Scholar]
- Mozaffarian D, Benjamin EJ, Go AS. et al. Heart disease and stroke statistics--2015 update: a report from the American Heart Association. Circulation. 2015;131(4):e29–e322. doi: 10.1161/CIR.0000000000000152. [DOI] [PubMed] [Google Scholar]
- Vaccarino V, Parsons L, Every NR. et al. Sex-based differences in early mortality after myocardial infarction. National Registry of Myocardial Infarction 2 Participants. N Engl J Med. 1999;341(4):217–225. doi: 10.1056/NEJM199907223410401. [DOI] [PubMed] [Google Scholar]
- Mehta LS, Beckie TM, DeVon HA. et al. Acute Myocardial Infarction in Women: A Scientific Statement From the American Heart Association. Circulation. 2016;133(9):916–947. doi: 10.1161/CIR.0000000000000351. [DOI] [PubMed] [Google Scholar]
- Wenger NK. Are we there yet? Closing the gender gap in coronary heart disease recognition, management and outcomes. Expert Rev Cardiovasc Ther. 2013;11(11):1447–1450. doi: 10.1586/14779072.2013.845526. [DOI] [PubMed] [Google Scholar]
- Clayton JA, Collins FS. Policy: NIH to balance sex in cell and animal studies. Nature. 2014;509(7500):282–283. doi: 10.1038/509282a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Melloni C, Berger JS, Wang TY. et al. Representation of women in randomized clinical trials of cardiovascular disease prevention. Circulation Cardiovascular quality and outcomes. 2010;3(2):135–142. doi: 10.1161/CIRCOUTCOMES.110.868307. [DOI] [PubMed] [Google Scholar]
- National Center for Health Statistics. Health US. With Special Feature on Socioeconomic Status and Health. 2011. Available from: http://www.cdc.gov/nchs/data/hus/hus11.pdf . [PubMed]
- Bairey Merz CN, Shaw LJ, Reis SE. et al. Insights from the NHLBI-Sponsored Women's Ischemia Syndrome Evaluation (WISE) Study: Part II: gender differences in presentation, diagnosis, and outcome with regard to gender-based pathophysiology of atherosclerosis and macrovascular and microvascular coronary disease. J Am Coll Cardiol. 2006;47(3):S21–S29. doi: 10.1016/j.jacc.2004.12.084. [DOI] [PubMed] [Google Scholar]
- Bugiardini R, Manfrini O, Pizzi C. et al. Endothelial function predicts future development of coronary artery disease: a study of women with chest pain and normal coronary angiograms. Circulation. 2004;109(21):2518–2583. doi: 10.1161/01.CIR.0000128208.22378.E3. [DOI] [PubMed] [Google Scholar]
- Shaw LJ, Heller GV, Travin MI. et al. Cost analysis of diagnostic testing for coronary artery disease in women with stable chest pain. Economics of Noninvasive Diagnosis (END) Study Group. J Nucl Cardiol. 1999;6(6):559–569. doi: 10.1016/s1071-3581(99)90091-0. [DOI] [PubMed] [Google Scholar]
- Shaw LJ, Merz CNB, Pepine CJ. et al. The Economic Burden of Angina in Women With Suspected Ischemic Heart Disease: Results From the National Institutes of Health-National Heart, Lung, and Blood Institute-Sponsored Women's Ischemia Syndrome Evaluation. . Circulation. 2006;114(9):894–904. doi: 10.1161/CIRCULATIONAHA.105.609990. [DOI] [PubMed] [Google Scholar]
- von Mering GO, Arant CB, Wessel TR. et al. Abnormal coronary vasomotion as a prognostic indicator of cardiovascular events in women: results from the National Heart, Lung, and Blood Institute-Sponsored Women's Ischemia Syndrome Evaluation (WISE). Circulation. 2004;109(6):722–725. doi: 10.1161/01.CIR.0000115525.92645.16. [DOI] [PubMed] [Google Scholar]
- Hochman JS, Tamis JE, Thompson TD. et al. Sex, clinical presentation, and outcome in patients with acute coronary syndromes. Global Use of Strategies to Open Occluded Coronary Arteries in Acute Coronary Syndromes IIb Investigators. N Engl J Med. 1999;341(4):226–232. doi: 10.1056/NEJM199907223410402. [DOI] [PubMed] [Google Scholar]
- Delcour KS, Khaja A, Chockalingam A. et al. Outcomes in Patients With Abnormal Myocardial Perfusion Imaging and Normal Coronary Angiogram. Angiology. 2009;60(3):318–321. doi: 10.1177/0003319708319938. [DOI] [PubMed] [Google Scholar]
- Gulati M, Cooper-DeHoff RM, McClure C. et al. Adverse Cardiovascular Outcomes in Women With Nonobstructive Coronary Artery Disease: A Report From the Women's Ischemia Syndrome Evaluation Study and the St James Women Take Heart Project. . Arch Intern Med. 2009;169(9):843–850. doi: 10.1001/archinternmed.2009.50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levine SA. Clinical Heart Disease. 3rd ed. Philadelphia: W. B. Saunders Co.; 1945. p. 96. [Google Scholar]
- D'Onofrio G, Safdar B. Sex differences in reperfusion in young patients with ST-segment-elevation myocardial infarction: results from the VIRGO study. Circulation. 2015;131(15):1324–1332. doi: 10.1161/CIRCULATIONAHA.114.012293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khan NA, Daskalopoulou SS, Karp I. et al. Sex differences in acute coronary syndrome symptom presentation in young patients. JAMA Internal Medicine. 2013;173(20):1863–1871. doi: 10.1001/jamainternmed.2013.10149. [DOI] [PubMed] [Google Scholar]
- Safdar B, Nagurney JT, Anise A. et al. Gender-specific research for emergency diagnosis and management of ischemic heart disease: proceedings from the 2014 Academic Emergency Medicine Consensus Conference Cardiovascular Research Workgroup. . Acad Emerg Med. 2014;21(12):1350–1360. doi: 10.1111/acem.12527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McSweeney JC, Cleves MA, Zhao W. et al. Cluster analysis of women's prodromal and acute myocardial infarction symptoms by race and other characteristics. J Cardiovasc Nurs. 2010;25(4):311–322. doi: 10.1097/JCN.0b013e3181cfba15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Canto JG, Rogers WJ, Goldberg RJ. et al. Association of age and sex with myocardial infarction symptom presentation and in-hospital mortality. JAMA. 2012;307(8):813–822. doi: 10.1001/jama.2012.199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pelletier R, Khan NA, Cox J. et al. Sex Versus Gender-Related Characteristics: Which Predicts Outcome After Acute Coronary Syndrome in the Young? J Am Coll Cardiol. 2016;67(2):127–135. doi: 10.1016/j.jacc.2015.10.067. [DOI] [PubMed] [Google Scholar]
- Comparison of coronary bypass surgery with angioplasty in patients with multivessel disease. The Bypass Angioplasty Revascularization Investigation (BARI) Investigators. N Engl J Med. 1996;335(4):217–225. doi: 10.1056/NEJM199607253350401. [DOI] [PubMed] [Google Scholar]
- Farb A, Burke AP, Tang AL. et al. Coronary plaque erosion without rupture into a lipid core. A frequent cause of coronary thrombosis in sudden coronary death. Circulation. 1996;93(7):1354–1363. doi: 10.1161/01.cir.93.7.1354. [DOI] [PubMed] [Google Scholar]
- Spatz ES, Curry LA, Masoudi FA. et al. The VIRGO Classification System: A Taxonomy for Young Women with Acute Myocardial Infarction. Circulation. 2015;132(18):1710–1718. doi: 10.1161/CIRCULATIONAHA.115.016502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pope JH, Aufderheide TP, Ruthazer R. et al. Missed diagnoses of acute cardiac ischemia in the emergency department. N Engl J Med. 2000;342(16):1163–1170. doi: 10.1056/NEJM200004203421603. [DOI] [PubMed] [Google Scholar]
- Safdar B, Bezek SK, Sinusas AJ. et al. Elevated CK-MB with a normal troponin does not predict 30-day adverse cardiac events in emergency department chest pain observation unit patients. Crit Pathw Cardiol. 2014;13(1):14–19. doi: 10.1097/HPC.0000000000000001. [DOI] [PubMed] [Google Scholar]
- Safdar B, Ali A, D'Onofrio G. et al. Microvascular Dysfunction as Opposed to Conduit Artery Disease Explains Sex-Specific Chest Pain in Emergency Department Patients With Low to Moderate Cardiac Risk. Clin Ther. 2016;38(2):240–255. doi: 10.1016/j.clinthera.2015.12.010. [DOI] [PubMed] [Google Scholar]
- Ong P, Athanasiadis A, Hill S. et al. Coronary artery spasm as a frequent cause of acute coronary syndrome: The CASPAR (Coronary Artery Spasm in Patients With Acute Coronary Syndrome) Study. J Am Coll Cardiol. 2008;52(7):523–527. doi: 10.1016/j.jacc.2008.04.050. [DOI] [PubMed] [Google Scholar]
- Lanza GA, Careri G, Crea F. et al. Mechanisms of coronary artery spasm. Circulation. 2011;124(16):1774–1782. doi: 10.1161/CIRCULATIONAHA.111.037283. [DOI] [PubMed] [Google Scholar]
- Tweet MS, Hayes SN, Pitta SR. et al. Clinical features, management, and prognosis of spontaneous coronary artery dissection. Circulation. 2012;126(5):579–588. doi: 10.1161/CIRCULATIONAHA.112.105718. [DOI] [PubMed] [Google Scholar]
- Mohlenkamp S, Hort W, Ge J. et al. Update on myocardial bridging. Circulation. 2002;106(20):2616–2622. doi: 10.1161/01.cir.0000038420.14867.7a. [DOI] [PubMed] [Google Scholar]
- Reis SE, Holubkov R, Conrad Smith AJ. et al. Coronary microvascular dysfunction is highly prevalent in women with chest pain in the absence of coronary artery disease: results from the NHLBI WISE study. Am Heart J. 2001;141(5):735–741. doi: 10.1067/mhj.2001.114198. [DOI] [PubMed] [Google Scholar]
- Kaski JC, Rosano GM, Collins P. et al. Cardiac syndrome X: clinical characteristics and left ventricular function. Long-term follow-up study. J Am Coll Cardiol. 1995;25(4):807–814. doi: 10.1016/0735-1097(94)00507-M. [DOI] [PubMed] [Google Scholar]
- Sullivan AK, Holdright DR, Wright CA. et al. Chest pain in women: clinical, investigative, and prognostic features. BMJ. 1994;308(6933):883–886. doi: 10.1136/bmj.308.6933.883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Humphries KH, Pu A, Gao M. et al. Angina with "normal" coronary arteries: sex differences in outcomes. Am Heart J. 2008;155(2):375–381. doi: 10.1016/j.ahj.2007.10.019. [DOI] [PubMed] [Google Scholar]
- Lee BK, Lim HS, Fearon WF. et al. Invasive evaluation of patients with angina in the absence of obstructive coronary artery disease. Circulation. 2015;131(12):1054–1060. doi: 10.1161/CIRCULATIONAHA.114.012636. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reis Se, Holubkov R, Conrad Smith AJ. et al. Coronary microvascular dysfunction is highly prevalent in women with chest pain in the absence of coronary artery disease: results from the NHLBI WISE study. Am Heart J. 2001;141(5):735–741. doi: 10.1067/mhj.2001.114198. [DOI] [PubMed] [Google Scholar]
- Sara JD, Widmer RJ, Matsuzawa Y. et al. Prevalence of Coronary Microvascular Dysfunction Among Patients With Chest Pain and Nonobstructive Coronary Artery Disease. JACC Cardiovasc Interv. 2015;8(11):1445–1453. doi: 10.1016/j.jcin.2015.06.017. [DOI] [PubMed] [Google Scholar]
- Camici PG, d'Amati G, Rimoldi O. Coronary microvascular dysfunction: mechanisms and functional assessment. Nat Rev Cardiol. 2015;12(1):48–62. doi: 10.1038/nrcardio.2014.160. [DOI] [PubMed] [Google Scholar]
- Cannon RO 3rd, Epstein SE. "Microvascular angina" as a cause of chest pain with angiographically normal coronary arteries. . Am J Cardiol. 1988;61(15):1338–1343. doi: 10.1016/0002-9149(88)91180-0. [DOI] [PubMed] [Google Scholar]
- Bugiardini R, Bairey M. Angina with "normal" coronary arteries: a changing philosophy. JAMA. 2005;293(4):477–484. doi: 10.1001/jama.293.4.477. [DOI] [PubMed] [Google Scholar]
- Pepine CJ, Kerensky RA, Lambert CR. et al. Some thoughts on the vasculopathy of women with ischemic heart disease. J Am Coll Cardiol. 2006;47(3):S30–S35. doi: 10.1016/j.jacc.2005.09.023. [DOI] [PubMed] [Google Scholar]
- Suwaidi JA, Hamasaki S, Higano ST. et al. Long-term follow-up of patients with mild coronary artery disease and endothelial dysfunction. Circulation. 2000;101(9):948–954. doi: 10.1161/01.cir.101.9.948. [DOI] [PubMed] [Google Scholar]
- Lanza GA, Giordano A, Pristipino C. et al. Abnormal cardiac adrenergic nerve function in patients with syndrome X detected by [123I]metaiodobenzylguanidine myocardial scintigraphy. Circulation. 1997;96(3):821–826. doi: 10.1161/01.cir.96.3.821. [DOI] [PubMed] [Google Scholar]
- Gulli G, Cemin R, Pancera P. et al. Evidence of parasympathetic impairment in some patients with cardiac syndrome X. Cardiovasc Res. 2001;52(2):208–216. doi: 10.1016/s0008-6363(01)00369-8. [DOI] [PubMed] [Google Scholar]
- Rosen SD, Paulesu E, Wise RJ. et al. Central neural contribution to the perception of chest pain in cardiac syndrome X. Heart. 2002;87(6):513–519. doi: 10.1136/heart.87.6.513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steinberg HO, Tarshoby M, Monestel R. et al. Elevated circulating free fatty acid levels impair endothelium-dependent vasodilation. J Clin Invest. 1997;100(5):1230–1239. doi: 10.1172/JCI119636. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steinberg HO, Chaker H, Leaming R. et al. Obesity/insulin resistance is associated with endothelial dysfunction. Implications for the syndrome of insulin resistance. J Clin Invest. 1996;97(11):2601–2610. doi: 10.1172/JCI118709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perticone F, Ceravolo R, Candigliota M. et al. Obesity and body fat distribution induce endothelial dysfunction by oxidative stress: protective effect of vitamin C. Diabetes. 2001;50(1):159–165. doi: 10.2337/diabetes.50.1.159. [DOI] [PubMed] [Google Scholar]
- Rosenberg GA. Matrix metalloproteinases and their multiple roles in neurodegenerative diseases. Lancet neurology. 2009;8(2):205–216. doi: 10.1016/S1474-4422(09)70016-X. [DOI] [PubMed] [Google Scholar]
- Ong P, Athanasiadis A, Hill S. et al. Coronary microvascular dysfunction assessed by intracoronary acetylcholine provocation testing is a frequent cause of ischemia and angina in patients with exercise-induced electrocardiographic changes and unobstructed coronary arteries. Clin Cardiol. 2014;37(8):462–467. doi: 10.1002/clc.22282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matsuzawa Y, Sugiyama S, Sugamura K. et al. Digital assessment of endothelial function and ischemic heart disease in women. J Am Coll Cardiol. 2010;55(16):1688–1696. doi: 10.1016/j.jacc.2009.10.073. [DOI] [PubMed] [Google Scholar]
- Kanatsuka H, Eastham CL, Marcus ML. et al. Effects of nitroglycerin on the coronary microcirculation in normal and ischemic myocardium. J Cardiovasc Pharmacol. 1992;19(5):755–763. [PubMed] [Google Scholar]
- Spertus JA, Winder JA, Dewhurst TA. et al. Development and evaluation of the Seattle Angina Questionnaire: a new functional status measure for coronary artery disease. J Am Coll Cardiol. 1995;25(2):333–341. doi: 10.1016/0735-1097(94)00397-9. [DOI] [PubMed] [Google Scholar]
- Pepine CJ, Ferdinand KC, Shaw LJ. et al. Emergence of Nonobstructive Coronary Artery Disease: A Woman's Problem and Need for Change in Definition on Angiography. J Am Coll Cardiol. 2015;66(17):1918–1933. doi: 10.1016/j.jacc.2015.08.876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Patel MR, Peterson ED, Dai D. et al. Low diagnostic yield of elective coronary angiography. N Engl J Med. 2010;362(10):886–895. doi: 10.1056/NEJMoa0907272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ludmer PL, Selwyn AP, Shook TL. et al. Paradoxical vasoconstriction induced by acetylcholine in atherosclerotic coronary arteries. New Engl J Med. 1986;315(17):1046–1051. doi: 10.1056/NEJM198610233151702. [DOI] [PubMed] [Google Scholar]
- Campisi R. Noninvasive assessment of coronary microvascular function in women at risk for ischaemic heart disease. . Int J Clin Pract. 2008;62(2):300–307. doi: 10.1111/j.1742-1241.2007.01391.x. [DOI] [PubMed] [Google Scholar]
- Kaufmann PA, Camici PG. Myocardial Blood Flow Measurement by PET: Technical Aspects and Clinical Applications. J Nucl Med. 2005;46(1):75–88. [PubMed] [Google Scholar]
- Chareonthaitawee P, Kaufmann PA, Rimoldi O. et al. Heterogeneity of resting and hyperemic myocardial blood flow in healthy humans. Cardiovasc Res. 2001;50(1):151–161. doi: 10.1016/s0008-6363(01)00202-4. [DOI] [PubMed] [Google Scholar]
- Geltman EM, Henes CG, Senneff MJ. et al. Increased myocardial perfusion at rest and diminished perfusion reserve in patients with angina and angiographically normal coronary arteries. J Am Coll Cardiol. 1990;16(3):586–595. doi: 10.1016/0735-1097(90)90347-r. [DOI] [PubMed] [Google Scholar]
- Uren NG, Marraccini P, Gistri R. et al. Altered coronary vasodilator reserve and metabolism in myocardium subtended by normal arteries in patients with coronary artery disease. J Am Coll Cardiol. 1993;22(3):650–658. doi: 10.1016/0735-1097(93)90172-w. [DOI] [PubMed] [Google Scholar]
- Di Bella EV, Parker DL, Sinusas AJ. On the dark rim artifact in dynamic contrast-enhanced MRI myocardial perfusion studies. Magn Reson Med. 2005;54(5):1295–1299. doi: 10.1002/mrm.20666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taqueti VR, Everett BM, Murthy VL. et al. Interaction of impaired coronary flow reserve and cardiomyocyte injury on adverse cardiovascular outcomes in patients without overt coronary artery disease. Circulation. 2015;131(6):528–535. doi: 10.1161/CIRCULATIONAHA.114.009716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murthy VL, Naya M, Foster CR. et al. Improved cardiac risk assessment with noninvasive measures of coronary flow reserve. Circulation. 2011;124(20):2215–2224. doi: 10.1161/CIRCULATIONAHA.111.050427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nagamachi S, Czernin J, Kim AS. et al. Reproducibility of measurements of regional resting and hyperemic myocardial blood flow assessed with PET. J Nucl Med. 1996;37(10):1626–1631. [PubMed] [Google Scholar]
- Herzog BA, Husmann L, Valenta I. et al. Long-term prognostic value of 13N-ammonia myocardial perfusion positron emission tomography added value of coronary flow reserve. J Am Coll Cardiol. 2009;54(2):150–156. doi: 10.1016/j.jacc.2009.02.069. [DOI] [PubMed] [Google Scholar]
- Schindler TH, Schelbert HR, Quercioli A. et al. Cardiac PET imaging for the detection and monitoring of coronary artery disease and microvascular health. JACC Cardiovascular imaging. 2010;3(6):6223–640. doi: 10.1016/j.jcmg.2010.04.007. [DOI] [PubMed] [Google Scholar]
- Einstein AJ. Effects of radiation exposure from cardiac imaging: how good are the data? J Am Coll Cardiol. 2012;59(6):553–565. doi: 10.1016/j.jacc.2011.08.079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Renaud JM, Mylonas I, McArdle B. et al. Clinical interpretation standards and quality assurance for the multicenter PET/CT trial rubidium-ARMI. J Nucl Med. 2014;55(1):58–64. doi: 10.2967/jnumed.112.117515. [DOI] [PubMed] [Google Scholar]
- Ockene IS, Shay MJ, Alpert JS. et al. Unexplained chest pain in patients with normal coronary arteriograms: a follow-up study of functional status. N Engl J Med. 1980;303(22):1249–1252. doi: 10.1056/NEJM198011273032201. [DOI] [PubMed] [Google Scholar]
- Safdar B, Dziura J, Bathulapalli H. et al. Chest pain syndromes are associated with high rates of recidivism and costs in young United States Veterans. BMC Fam Pract. 2015;16:88. doi: 10.1186/s12875-015-0287-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shaw LJ, Merz CN, Pepine CJ. et al. The economic burden of angina in women with suspected ischemic heart disease: results from the National Institutes of Health--National Heart, Lung, and Blood Institute--sponsored Women's Ischemia Syndrome Evaluation. Circulation. 2006;114(9):894–904. doi: 10.1161/CIRCULATIONAHA.105.609990. [DOI] [PubMed] [Google Scholar]
- Johnson BD, Shaw LJ, Pepine CJ. et al. Persistent chest pain predicts cardiovascular events in women without obstructive coronary artery disease: results from the NIH-NHLBI-sponsored Women's Ischaemia Syndrome Evaluation (WISE) stu. Eur Heart J. 2006;27(12):1408–1415. doi: 10.1093/eurheartj/ehl040. [DOI] [PubMed] [Google Scholar]
- Braunwald E, Antman EM, Beasley JW. et al. ACC/AHA guideline update for the management of patients with unstable angina and non-ST-segment elevation myocardial infarction--2002: summary article: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Committee on the Management of Patients With Unstable Angina). Circulation. 2002;106(14):1893–1900. doi: 10.1161/01.cir.0000037106.76139.53. [DOI] [PubMed] [Google Scholar]
- Flavahan NA. Atherosclerosis or lipoprotein-induced endothelial dysfunction. Potential mechanisms underlying reduction in EDRF/nitric oxide activity. Circulation. 1992;85(5):1927–1938. doi: 10.1161/01.cir.85.5.1927. [DOI] [PubMed] [Google Scholar]
- Panza JA, Quyyumi AA, Brush JE Jr.. et al. Abnormal endothelium-dependent vascular relaxation in patients with essential hypertension. N Engl J Med. 1990;323(1):22–27. doi: 10.1056/NEJM199007053230105. [DOI] [PubMed] [Google Scholar]
- Cai H, Harrison DG. Endothelial dysfunction in cardiovascular diseases: the role of oxidant stress. Circ Res. 2000;87(10):840–844. doi: 10.1161/01.res.87.10.840. [DOI] [PubMed] [Google Scholar]
- Murthy VL, Naya M, Taqueti VR. et al. Effects of Gender on Coronary Microvascular Dysfunction and Cardiac Outcomes. Circulation. 2014;129(24):2518–2527. doi: 10.1161/CIRCULATIONAHA.113.008507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lamendola P, Lanza GA, Spinelli A. et al. Long-term prognosis of patients with cardiac syndrome X. Int J Cardiol. 2010;140(2):197–199. doi: 10.1016/j.ijcard.2008.11.026. [DOI] [PubMed] [Google Scholar]
- Marinescu MA, Loffler AI, Ouellette M. et al. Coronary microvascular dysfunction, microvascular angina, and treatment strategies. JACC Cardiovascular imaging. 2015;8(2):210–220. doi: 10.1016/j.jcmg.2014.12.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Russo G, Di Franco A, Lamendola P. et al. Lack of effect of nitrates on exercise stress test results in patients with microvascular angina. Cardiovasc Drugs Ther. 2013;27(3):229–234. doi: 10.1007/s10557-013-6439-z. [DOI] [PubMed] [Google Scholar]
- Sutsch G, Oechslin E, Mayer I. et al. Effect of diltiazem on coronary flow reserve in patients with microvascular angina. Int J Cardiol. 1995;52(2):135–143. doi: 10.1016/0167-5273(95)02458-9. [DOI] [PubMed] [Google Scholar]
- Togni M, Vigorito F, Windecker S. et al. Does the beta-blocker nebivolol increase coronary flow reserve? Cardiovasc Drugs Ther. 2007;21(2):99–108. doi: 10.1007/s10557-006-0494-7. [DOI] [PubMed] [Google Scholar]
- Schiffrin EL. Circulatory therapeutics: use of antihypertensive agents and their effects on the vasculature. J Cell Mol Med. 2010;14(5):1018–1029. doi: 10.1111/j.1582-4934.2010.01056.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lund-Johansen P, Omvik P. Acute and chronic hemodynamic effects of drugs with different actions on adrenergic receptors: a comparison between alpha blockers and different types of beta blockers with and without vasodilating effect. . Cardiovasc Drugs Ther. 1991;5(3):605–615. doi: 10.1007/BF03029729. [DOI] [PubMed] [Google Scholar]
- Schiffrin EL, Deng LY. Effects of a beta-blocker or a converting enzyme inhibitor on resistance arteries in essential hypertension. Hypertension. 1994;23(1):83–91. doi: 10.1161/01.hyp.23.1.83. [DOI] [PubMed] [Google Scholar]
- Schiffrin EL, Park JB, Intengan HD. et al. Correction of arterial structure and endothelial dysfunction in human essential hypertension by the angiotensin receptor antagonist losartan. Circulation. 2000;101(4):1653–1659. doi: 10.1161/01.cir.101.14.1653. [DOI] [PubMed] [Google Scholar]
- Schiffrin EL, Deng LY. Structure and function of resistance arteries of hypertensive patients treated with a beta-blocker or a calcium channel antagonist. J Hypertens. 1996;14(10):1247–1255. doi: 10.1097/00004872-199610000-00014. [DOI] [PubMed] [Google Scholar]
- Pauly DF, Johnson BD, Anderson RD. et al. In women with symptoms of cardiac ischemia, nonobstructive coronary arteries, and microvascular dysfunction, angiotensin-converting enzyme inhibition is associated with improved microvascular function: A double-blind randomized study from the National Heart, Lung and Blood Institute Women's Ischemia Syndrome Evaluation (WISE). Am Heart J. 2011;162(4):678–684. doi: 10.1016/j.ahj.2011.07.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tagliamonte E, Rigo F, Cirillo T. et al. Effects of ranolazine on noninvasive coronary flow reserve in patients with myocardial ischemia but without obstructive coronary artery disease. Echocardiography. 2015;32(3):516–521. doi: 10.1111/echo.12674. [DOI] [PubMed] [Google Scholar]
- Villano A, Di Franco A, Nerla R. et al. Effects of ivabradine and ranolazine in patients with microvascular angina pectoris. Am J Cardiol. 2013;112(1):8–13. doi: 10.1016/j.amjcard.2013.02.045. [DOI] [PubMed] [Google Scholar]
- Phan A, Shufelt C, Merz CN. et al. Persistent chest pain and no obstructive coronary artery disease. JAMA. 2009;301(14):1468–1474. doi: 10.1001/jama.2009.425. [DOI] [PubMed] [Google Scholar]
- Jespersen L, Hvelplund A, Abildstrom SZ. et al. Stable angina pectoris with no obstructive coronary artery disease is associated with increased risks of major adverse cardiovascular events. Eur Heart J. 2012;33(6):734–744. doi: 10.1093/eurheartj/ehr331. [DOI] [PubMed] [Google Scholar]
- Pepine CJ, Anderson RD, Sharaf BL. et al. Coronary microvascular reactivity to adenosine predicts adverse outcome in women evaluated for suspected ischemia results from the National Heart, Lung and Blood Institute WISE (Women's Ischemia Syndrome Evaluation) study. J Am Coll Cardiol. 2010;55(25):2825–2832. doi: 10.1016/j.jacc.2010.01.054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- von Mering GO, Arant CB, Wessel TR. et al. Abnormal coronary vasomotion as a prognostic indicator of cardiovascular events in women: results from the National Heart, Lung, and Blood Institute-Sponsored Women's Ischemia Syndrome Evaluation (WISE). Circulation. 2004;109(6):722–725. doi: 10.1161/01.CIR.0000115525.92645.16. [DOI] [PubMed] [Google Scholar]
- van de Hoef TP, van Lavieren MA, Damman P. et al. Physiological basis and long-term clinical outcome of discordance between fractional flow reserve and coronary flow velocity reserve in coronary stenoses of intermediate severity. Circ Cardiovasc Interv. 2014;7(3):301–311. doi: 10.1161/CIRCINTERVENTIONS.113.001049. [DOI] [PubMed] [Google Scholar]
- Gulati M, Cooper-DeHoff RM, McClure C. et al. Adverse cardiovascular outcomes in women with nonobstructive coronary artery disease: a report from the Women's Ischemia Syndrome Evaluation Study and the St James Women Take Heart Project. Arch Intern Med. 2009;169(9):843–850. doi: 10.1001/archinternmed.2009.50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bugiardini R, Manfrini O, Pizzi C. et al. Endothelial function predicts future development of coronary artery disease: a study of women with chest pain and normal coronary angiograms. Circulation. 2004;109(21):2518–2523. doi: 10.1161/01.CIR.0000128208.22378.E3. [DOI] [PubMed] [Google Scholar]
- Johnson BD, Shaw LJ, Pepine CJ. et al. Persistent chest pain predicts cardiovascular events in women without obstructive coronary artery disease: results from the NIH-NHLBI-sponsored Women's Ischaemia Syndrome Evaluation (WISE) study. Eur Heart J. 2006;27(12):1408–1415. doi: 10.1093/eurheartj/ehl040. [DOI] [PubMed] [Google Scholar]
- NIH. Report on Small Blood Vessels: Big Health Problems? Workshop. 2014. Available from: http://www.ninds.nih.gov/news_and_events/proceedings/Report-for-Small-Blood-Vessels.htm .