Abstract
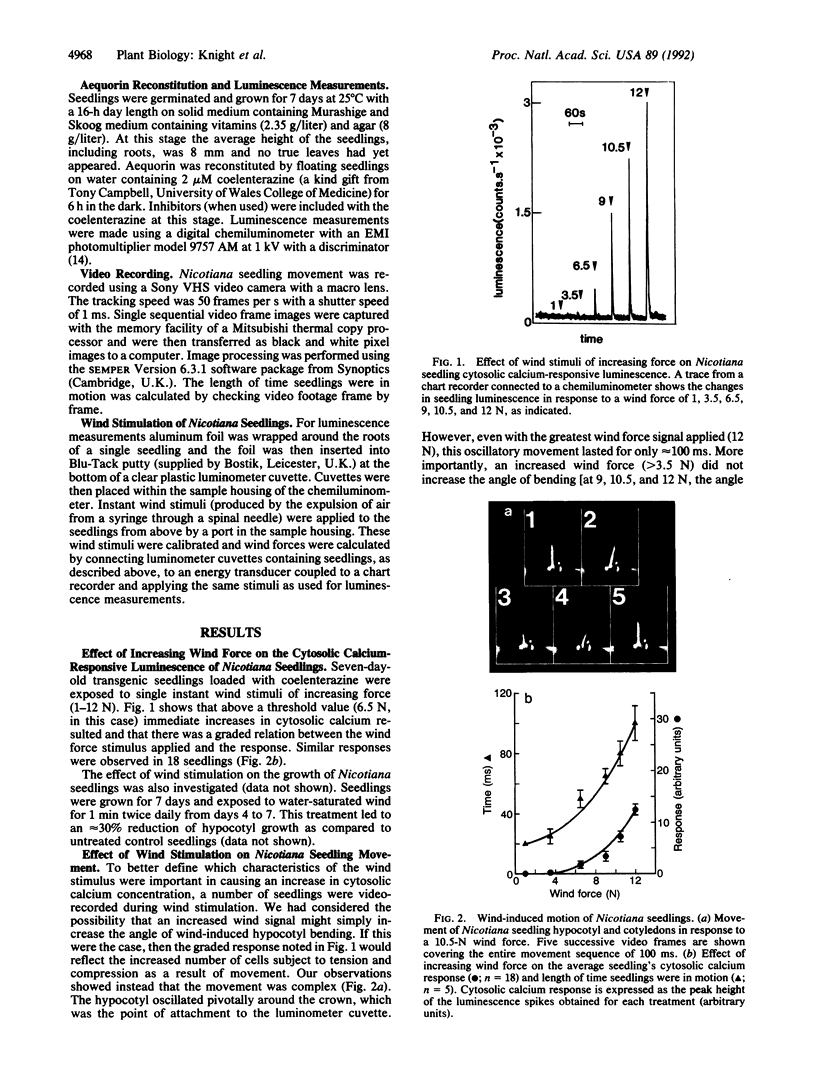
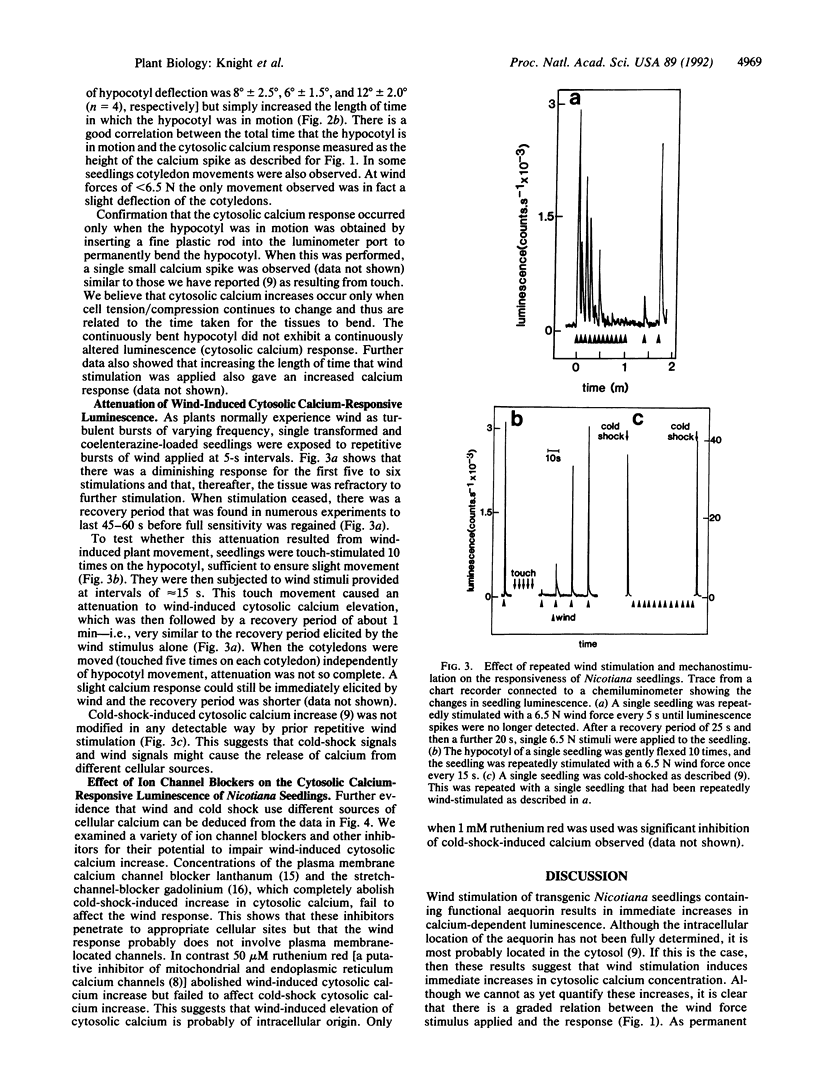
Wind is one of the most unusual and more dramatic of the environmental signals to modify plant development. Wind-stimulated crops are also known to experience considerable reductions in growth and subsequent yield. There is at present no experimental data to suggest how wind signals are perceived and transduced by plant cells. We have genetically transformed Nicotiana plumbaginifolia to express aequorin and thus produced luminous plants that directly report cytosolic calcium by emitting blue light. With these plants we have found wind stimulation to cause immediate increases in cytosolic calcium and our evidence, based on the use of specific inhibitors, suggests that this calcium is mobilized from organelle sources. Our data further suggest that wind-induced movement of tissues, by mechanically stimulating and stressing constituent plant cells, is responsible for the immediate elevation of cytosolic calcium; increases occur only when the plant tissue is actually in motion. Repeated wind stimulation renders the cells refractory to further calcium signaling but responsiveness is rapidly recovered when stimulation is subsequently diminished. Our data suggest that mechanoperception in plant cells may possibly be transduced through intracellular calcium. Since mechanoperception and transduction are considered crucial to plant morphogenesis, our observations suggest that calcium could be central in the control and generation of plant form.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bevan M. Binary Agrobacterium vectors for plant transformation. Nucleic Acids Res. 1984 Nov 26;12(22):8711–8721. doi: 10.1093/nar/12.22.8711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Braam J., Davis R. W. Rain-, wind-, and touch-induced expression of calmodulin and calmodulin-related genes in Arabidopsis. Cell. 1990 Feb 9;60(3):357–364. doi: 10.1016/0092-8674(90)90587-5. [DOI] [PubMed] [Google Scholar]
- Campbell A. K., Hallett M. B., Weeks I. Chemiluminescence as an analytical tool in cell biology and medicine. Methods Biochem Anal. 1985;31:317–416. doi: 10.1002/9780470110522.ch7. [DOI] [PubMed] [Google Scholar]
- Gilroy S., Read N. D., Trewavas A. J. Elevation of cytoplasmic calcium by caged calcium or caged inositol triphosphate initiates stomatal closure. Nature. 1990 Aug 23;346(6286):769–771. doi: 10.1038/346769a0. [DOI] [PubMed] [Google Scholar]
- Jones R. S., Mitchell C. A. Calcium ion involvement in growth inhibition of mechanically stressed soybean (Glycine max) seedlings. Physiol Plant. 1989;76:598–602. doi: 10.1111/j.1399-3054.1989.tb05485.x. [DOI] [PubMed] [Google Scholar]
- Knight M. R., Campbell A. K., Smith S. M., Trewavas A. J. Transgenic plant aequorin reports the effects of touch and cold-shock and elicitors on cytoplasmic calcium. Nature. 1991 Aug 8;352(6335):524–526. doi: 10.1038/352524a0. [DOI] [PubMed] [Google Scholar]
- Neel P. L., Harris R. W. Motion-induced inhibition of elongation and induction of dormancy in liquidambar. Science. 1971 Jul 2;173(3991):58–59. doi: 10.1126/science.173.3991.58. [DOI] [PubMed] [Google Scholar]
- Parkhurst D. F., Pearman G. I., Neel P. L., Harris R. W. Tree seedling growth: effects of shaking. Science. 1972 Feb 25;175(4024):918–919. doi: 10.1126/science.175.4024.918. [DOI] [PubMed] [Google Scholar]
- Pietrzak M., Shillito R. D., Hohn T., Potrykus I. Expression in plants of two bacterial antibiotic resistance genes after protoplast transformation with a new plant expression vector. Nucleic Acids Res. 1986 Jul 25;14(14):5857–5868. doi: 10.1093/nar/14.14.5857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poovaiah B. W., Leopold A. C. Deferral of leaf senescence with calcium. Plant Physiol. 1973 Sep;52(3):236–239. doi: 10.1104/pp.52.3.236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poovaiah B. W., Leopold A. C. Inhibition of abscission by calcium. Plant Physiol. 1973 May;51(5):848–851. doi: 10.1104/pp.51.5.848. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Turgeon R., Webb J. A. Growth inhibition by mechanical stress. Science. 1971 Nov 26;174(4012):961–962. doi: 10.1126/science.174.4012.961. [DOI] [PubMed] [Google Scholar]
- Yang X. C., Sachs F. Block of stretch-activated ion channels in Xenopus oocytes by gadolinium and calcium ions. Science. 1989 Feb 24;243(4894 Pt 1):1068–1071. doi: 10.1126/science.2466333. [DOI] [PubMed] [Google Scholar]




