Abstract
Wine was prepared from three varieties of Phyllanthus viz., P. emblica (wild and cultivated) and P. acidus. Among the wines prepared, cultivated Emblica wine had the highest total phenolic (11.02 μg gallic acid equivalent/ml) and flavonoid (59.46 μg quercetin equivalent/ml) content. Further gallic acid, syringic acid, protocatechuic acid and caffeic acid were present in higher amounts in wine from the cultivated variety compared to other wines. HPLC analysis showed that in juice of the cultivated variety, gallic acid and coumaric acid were found in higher amounts than that in the corresponding wine. Antioxidant assays, LDL oxidation prevention, foam cell prevention and nitrite scavenging activities (cell lines) were found to be highest in cultivated Emblica juice and wine with an activity of 15 μg/ml and 14 μg/ml (nitrite assay) and 108.649 μg ascorbic acid equivalent/mg and 321.622 μg ascorbic acid equivalent /mg (total antioxidant capacity) respectively. CD36 expression was reduced and ABCA1 expression was increased to the highest extent by the cultivated Emblica wine and juice. Further, antioxidant activity was seen to increase during the course of fermentation. Sensory analysis showed that cultivated Emblica wine was sweeter compared to the other wines.
Keywords: Phyllanthus wine, Phenolic acids, Foam cell, Nitrite radicals
Introduction
Previous epidemiological studies have shown that consumption of fruit juices and wines have a wide variety of health promoting effects, particularly in reducing the risk of cardiovascular diseases (Renaud and De Lorgeril 1992). Antioxidant phenolic compounds like phenolic acids, polyphenols and flavonoids have been shown to scavenge free radicals. This has an impact on the redox mechanisms that lead to inflammation based conditions such as atherosclerosis and certain types of cancer (Uttara et al. 2009). Atherosclerosis is a disease of the arteries, characterised by the deposition of plaques of fatty material on their inner walls.
This constitutes a major risk for cardiovascular disease, which is a leading cause of death worldwide. Atherosclerosis is characterised by the formation of foam cells, created due to the generation of free radicals like nitrite radicals which oxidize LDL (Libby et al. 2011).
Previous reports have shown that moderate consumption of fermented beverages such as wine prevent coronary heart disease by reducing atherosclerosis through beneficial effects on high- density lipoprotein-cholesterol. It has been shown that antioxidant activities of fruits and vegetables increased dramatically during fermentation. Hence we can presume that fermented alcoholic products from fruits may have higher beneficial properties (Rai et al. 2010). In order to maintain the substantial antioxidant properties of fermented products, determination of their principal antioxidant components is vital. During fermentation of foods, desirable biochemical changes and significant modifications in flavour and texture occur through the activity of microorganisms or enzymes. The composition of phenolic compounds is also modified by yeasts as a result of conversion of non-phenolic substances, solubilisation and extraction of phenolics by the ethanol produced during fermentation (Singleton and Noble 1976). The antioxidant activity of wine has been previously related to its phenolic content including compounds such as flavonols, procyanidins and phenolic acids. Grape phenolics have been shown to inhibit platelet aggregation and also possess anti-inflammatory activity (Manach et al., 2005). Even though there is well-documented evidence for the health promoting activity of wine made from grapes, not much work has been carried out with the fruits of Phyllanthus emblica L. (Indian gooseberry).
Phyllanthus emblica belongs to the family Euphorbiaceae and is an edible fruit indigenous to Southeast Asia (Barthakur and Arnold 1991). It is found chiefly in Bangladesh, China, India, Malaysia, Pakistan and Sri Lanka. This fruit has been used in India from ancient times due to its well-known antioxidant and medicinal properties. It has been used as a major ingredient in medicinal formulations of Ayurveda, the ancient system of traditional medicine mentioned in the texts of Vedas. The fruit has been shown to have anti-inflammatory, antiviral and hypolipidemic effects (Scartezzini and Speroni 2000). Although fresh fruit was shown to have the major activity, the fruit is hardly commercialised. There is possibility of commercialising the fermented fruit. There have been no reports either on the biological activity of products made from these fruits or on the effect of fermentation on the antioxidant and phenolic profile of these fruits. Hence, in this paper we investigated the antioxidant and anti-atherogenic activities of wines and juices prepared from three different species of Phyllanthus and investigated the change in composition of phenolic and flavonoid compounds during the course of fermentation.
Materials and methods
Fermentation of phyllanthus species
Fruits of three different species of Phyllanthus, i.e., wild variety of Phyllanthus emblica, Phyllanthus acidus (sweet variety) from Kodagu district of Karnataka, India and locally available Phyllanthus emblica (commonly available cultivated cultivar NA-10) were collected. Fruits were washed with distilled water and stored at −20 °C until further use. Juices of the fruits were prepared by squeezing the fruits after grinding followed by filtration and lyophilization. For preparation of wine, fruits were cut into pieces, deseeded and used for fermentation. Saccharomyces cerevisiae (S-101) from CFTRI culture collection centre was used for fermentation. Inoculum was prepared by using the method as mentioned in our earlier work (Rai et al. 2010). Wine was prepared using 20 % (w/v) pulp of Emblica fruits and refractive index was adjusted to 22°brix using sucrose and pH to 4.5 using 0.1 N KOH. The must was pasteurized at 62° for 20 min and incubated with 5 % S.cerevisiae. Fermentation was carried out in glass containers at 25 °C. Samples were drawn at intervals of 5 days, till 21 days. On the 10th day of fermentation, pulp was removed from the must and fermentation was carried out without pulp. A sample of must was used for experiments after lyophilization. Wine was filtered, pasteurized at 62 °C for 20 min and racked before bottling. The bottled wines were stored at room temperature. Samples were analyzed in triplicates for various physical, chemical and microbiological parameters (Rai et al. 2010). Alcohol content of wine samples was estimated by the method of Caputi et al. (1968).
Gas chromatography analysis of alcohol
A Shimadzu 2010 plus GC with a flame ionization detector was used for the analysis. A ZB-wax capillary column (30 m × 0.25 mm × 0.25 μm) was used with a temperature programming from 30 °C to 70 °C at 14 °C /min rise to 220 °C at 25 °C /min for 3 min. Injector and detector temperatures were 220 °C and 250 °C, respectively. Nitrogen carrier flow was 1 ml/min and 0.5 μl of the sample was injected.
Phytochemical screening
Total phenolic content of all the samples was estimated by the method of Gülçin et al. (2002) using Folin-Ciocalteu’s reagent. The amount of total phenolic compounds in the extracts was expressed as μg gallic acid equivalents/ml. The total flavonoid content in the extracts was determined by the method described by Basniwal et al. (2009) and results were expressed as μg quercetin equivalents/ml.
HPLC analysis
Phenolic and flavonoid analysis by HPLC was performed using the method described by Ruangchakpet and Tanaboon (2007). UV detectors at 280 nm and 370 nm were used for the analysis. Organic acids in the wine samples were analysed using the HPLC system as described by Jayaprakash and Sakariah (2002). Detection was carried out by a UV detector at 210 nm.
In vitro antioxidant assays
The free radical scavenging activity of the wines, 10th day must, 10th day solid (collected along with the 10th day must) and juices was measured in vitro by the 1,1-diphenyl-2-picrylhydrazyl (DPPH) assay (Brand-Williams et al. 1995) at different concentrations (1-1000 μg/ml) using ascorbic acid as standard. Nitrite radical scavenging activity was investigated by Griess Illosvoy reaction (Hyoung Lee 1992) using ascorbic acid and ibuprofen as standard.
Results were expressed as IC50 which indicates the concentration required to bring about 50 % inhibition. The total antioxidant capacity of the samples was examined by the phosphomolybdenum method according to Prieto et al. (1999). The antioxidant capacity of extract was evaluated as ascorbic acid equivalents (μg ascorbic acid/mg extract). Prevention of LDL oxidation was assessed by the method of Fairclough et al. (1978) using ascorbic acid as standard antioxidant. Isolation of Low Density Lipoprotein (LDL) was done according to the method of Orrego et al. (2009) using NaCl/KBr density gradient followed by ultracentrifugation.
Cell line antioxidant assays
RAW 264.7 cells were procured from National Centre for Cell Science (NCCS), Pune, India, and cultured in Dulbecco’s modified Eagle medium (DMEM) (Sigma-Aldrich) supplemented with 10 % fetal bovine serum (Himedia) and 1 % Penicillin-streptomycin (Sigma-Aldrich) antibiotic and maintained in a humidified atmosphere with 5 % CO2 in a carbon dioxide incubator. Lipopolysaccharide (LPS) induced nitrite radical scavenging activity was performed by the method described by Lee et al. (2012). Prevention of foam cell formation by incubating RAW 264.7 cells with oxidized LDL (ox-LDL) and extracts for 48 h was carried out using the method of Greenspan et al. (1997). Results were expressed as IC50 (μg/ml).
Western blot analysis
Protein expression of ABCA1 and CD36 was measured by western blot analysis. RAW 264.7 cells (3 × 107) were cultured in the presence Ox-LDL (50 μg/ml), with/without test samples (100 μg/ml) or ascorbic acid (10 μg/ml) for 48 h. Cell extracts were obtained by centrifugation at 14,000×g at 4 °C for 10 min. The proteins were electrophoresed using SDS PAGE and transferred on to PVDF membranes. The membranes were then blocked with 5 % (w/v) non-fat dry milk in PBS at room temperature for 1 h. Then the membranes were incubated with polyclonal anti-CD36 and anti-ABCA1 (1: 1000) and anti-β-actin antibodies at 4 °C overnight. The PVDF membranes were incubated with peroxidase-conjugated goat anti-rabbit IgG antibody at room temperature for 1 h and stained using diaminobenzidine and hydrogen peroxide and photographed using digital camera. Western blot quantification was performed using ImageJ software. The quantification of CD36 and ABCA1 was expressed as the ratio of the protein expression of CD36 or ABCA1 with that of β-actin (housekeeping gene).
Sensory analysis
Descriptive sensory analysis of wine samples were conducted using a standard method - Quantitative descriptive analysis (QDA) technique. A 15 cm structured line scale anchoring low and high on the extremes ends was used. A sensory panel consisting of 10 trained judges, who are familiar with wine quality aspects, were participated in the sensory analysis. The samples were served using a three digit coded sample containers. The judges analyzed the samples and given their verdict on the score card by marking across the line scale against each sensory descriptors. A sensory profilogram was generated using the sample mean values. Significant difference between mean scores was established using Duncan’s New Multiple Range Test (DMRT).
Statistical analysis
All experiments were performed as individual experiments with three trials in each (n = 3) and results were expressed as mean ± SD. Statistical analysis was carried out with (SPSS package version 10.0) using ANOVA followed by Tukey’s test (p < 0.05). Correlation and regression analysis were performed using Microsoft excel 2007.
Results and discussion
Effect of fermentation on the chemical profile of the phyllanthus fruits
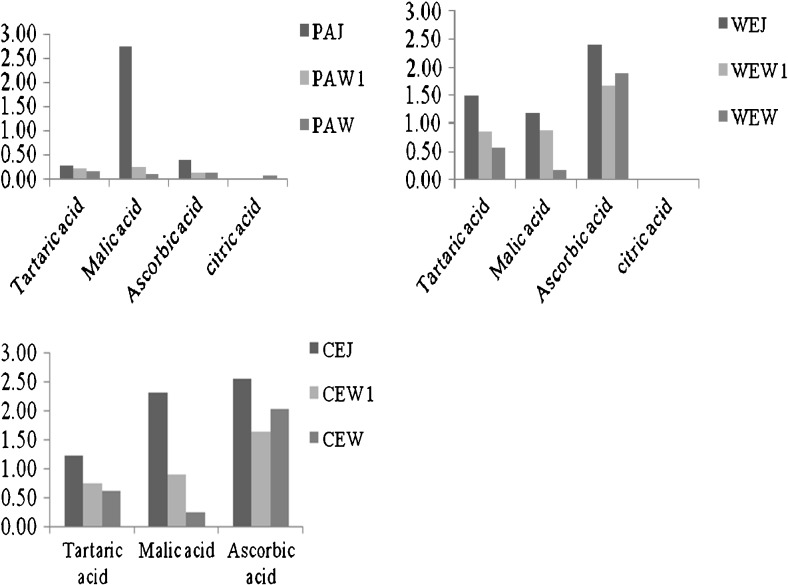
During fermentation, there was a decrease in tartaric and malic acid content in all Phyllanthus wine samples as seen in Fig. 1. In cultivated and wild variety samples, ascorbic acid content was found to increase significantly (Fig. 1). However, the antioxidant activities could not be attributed to it, due to the fact that ascorbic acid alone showed lower activities than the juices and wines of cultivated variety in all the antioxidant assays performed. Moreover, there are some in vivo studies indicating that antioxidant activities of the fruit cannot be attributed to ascorbic acid alone (Bhattacharya et al. 1999). Hence, the high antioxidant activities of wild and cultivated wine varieties could be due to the synergistic effect of the phenolic and flavonoid compounds, generated during the fermentation, with ascorbic acid. However, in case of PAW there was a decrease in ascorbic acid content and a threefold increase in the concentration of citric acid (Fig. 1). The increased activity is attributed to the transformation of phenolic acids during fermentation (Rai et al. 2010).
Fig. 1.
HPLC Profiles of organic acids. Organic acid content in P. acidus juice (PAJ), P. acidus wine 1st day sample (PAW 1) and P. acidus wine sample (PAW), wild emblica juice (WEJ), wild emblica wine 1st day sample (WEW 1) and wild emblica wine (WEW), cultivated emblica juice (CEJ), cultivated emblica variety wine 1st day sample (CEW 1) and cultivated emblica wine (CEW). Each bar graph value indicates value mean ± SD (standard deviation) from triplicate measurements (n = 3) and significance was found out at P ≤ 0.05 using Tukey’s test
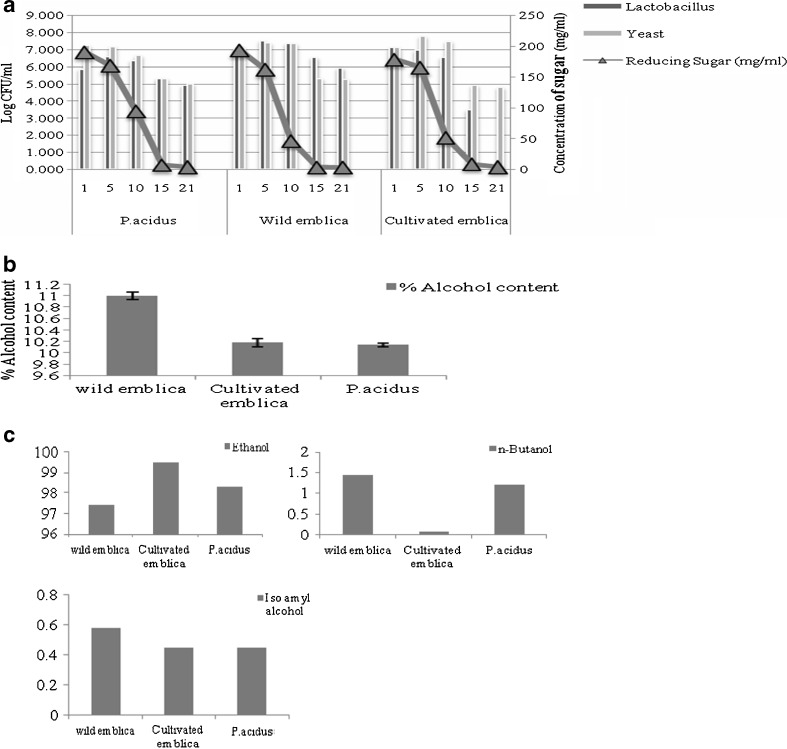
Kinetics of fermentation indicated that in all the 3 wines, growth of yeast was exponential till the 5th day of the fermentation, after which it slowly decreased (Fig. 2a). Reducing sugars were completely utilized by 15 days of fermentation in cultivated Emblica wine with yeast being the dominant microorganism (Fig. 2a). The alcohol content of the three different wines was in the range of 10 to 11 % (Fig.2b). Further, wine samples were analyzed for alcohol composition i.e., relative percentage of alcohol by gas chromatography (Fig. 2c). Results showed absence of methanol and the content of ethanol to be higher in all the wine samples. There was reduction in n-butanol and isoamyl alcohol in CEW.
Fig. 2.
a Lactobacillus count, yeast count and reducing sugar content in the three wines. b Percentage alcohol content in the three wines. Each bar graph value indicates value mean ± SD (standard deviation) from triplicate measurements (n = 3) and significance was found out at P ≤ 0.05 using Tukey’s test. c Relative percentage of ethanol, butanol and iso amyl alcohol in the three wines. Each bar graph value indicates value mean ± SD (standard deviation) from triplicate measurements (n = 3) and significance was found out at P ≤ 0.05 using Tukey’s test
Phytochemical screening
CEJ had higher phenolic content compared to its corresponding wine. Similar results were observed in case of pomegranate where the mixture of juice from arils, mesocarp and epicarp showed higher phenolic content and antioxidant activity than its corresponding wine (Wasila et al. 2013). As seen in Table 1, when compared to other wines, the total phenolic content (TPC) was found to be highest in the CEW. The TPC was found to increase from 9.63 μg GAE/ml on the 10th day in CEM to 11.02 μg GAE/ml during the wine stage (CEW) thus showing an increasing trend during fermentation in all the three wines. Similar pattern of increase of phenolic content during the course of fermentation was observed in another study (Gordillo et al. 2013). The wines also showed highest TFC compared to their respective juices (Table 1). Of the wines, the highest TFC was seen in the case of CEW followed by WEW.
Table 1.
Antioxidant, anti-inflammatory activities and Total phenolic and total flavonoid content in the wines, juices and 10th day must of the three Phyllanthus varieties
| Samples | DPPH IC50(μg/ml) | Nitrite assay IC50 (μg/ml) | TPC(μg GAE /ml) | TFC(μg QE/ml) | TAC (μg AAE/mg dry wt. |
|---|---|---|---|---|---|
| CEJ | 1.7 ± 1.6a | 14.0 ± 0.0a | 14.4 ± 0.00a | 55.7 ± 0.0a | 321.6 ± 0.0b |
| WEJ | 6.0 ± 0.0a | 21.0 ± 1.4ab | 5.4 ± 0.0b | 55.4 ± 0.0a | 205.7 ± 0.0c |
| PAJ | 520.0 ± 3.4c | 500.0 ± 6.2d | 0.1 ± 0.0d | 24.4 ± 0.0c | 47.3 ± 0.0e |
| CEM | 25.8 ± 2.3b | 30.0 ± 0.0ab | 9.6 ± 0.0e | 55.4 ± 0.0a | 81.1 ± 0.0d |
| WEM | 10.0 ± 2.4ab | 140.0 ± 1.7c | 1.1 ± 0.0f | 54.7 ± 0.0a | 51.9 ± 0.0e |
| PAM | 950.0 ± 0.0e | 800.0 ± 16.9f | 0.1 ± 0.0d | 28.3 ± 0.0bc | 26.8 ± 0.0f |
| CEW | 5.0 ± 0.7a | 15.0 ± 1.4a | 11.0 ± 0.0a | 59.5 ± 0.0a | 108.7 ± 0.0a |
| WEW | 8.0 ± 0.3ab | 38.00 ± 2.6b | 2.3 ± 0.0b | 58.8 ± 0.0a | 105.9 ± 0.0a |
| PAW | 750.0 ± 0.0d | 730.0 ± 7.1e | 0.1 ± 0.0c | 30.8 ± 0.0b | 23.5 ± 0.0f |
| Ascorbic acid | 5.3 ± 0.1a | 25.0 ± 0.0ab | - | - | - |
| Ibuprofen | - | 20.0 ± 0.0ab | - | - | - |
CEJ Cultivated Emblica Juice, WEJ Wild Emblica Juice, PEJ Phyllanthus acidus juice, CEM Cultivated Emblica Must, WEM Wild Emblica Must, PEM Phyllanthus acidus Must, CEW Cultivated Emblica Wine, WEW Wild Emblica Wine, PEW Phyllanthus acidus Wine, GAE gallic acid equivalent, QE Quercetin Equivalent, AAE Ascorbic Acid Equivalent, TPC Total Phenolic Content, TFC Total Flavonoid Content, TAC Total Antioxidant Capacity. Results are expressed as means ± SD, n = 3. Lower IC50 indicates higher activity. Values within a column followed by different letters in superscripts are significantly different by Tukey’s test after ANOVA (P < 0.05)
Even here, there was an increasing pattern of TFC during the course of fermentation, although the increase was not statistically significant. Previously a change in flavonoid content was reported by Wu et al. (2011), to be a contributing factor to the antioxidant activity of the fermented fruit juice. HPLC analysis (Table 2) for phenolics and flavonoids showed that myricetin content was very high in CEW i.e., 56.25 μg/ml extract (Table 2). Quercetin, kaempferol and luteolin were present in CEJ but found to be absent in the CEW. In CEJ, gallic acid and coumaric acid were found in higher amounts than that found in the CEW.
Table 2.
HPLC Profiles of phenolics and flavonoids
| Fractions | CE W | CEM (μg/ml) | CEJ (μg/ml) | WEW (μg/ml) | WEM (μg/ml) | WEJ (μg/ml) | PAW (μg/ml) | PAM (μg/ml) | PAJ (μg/ml) |
|---|---|---|---|---|---|---|---|---|---|
| Gallic acid | 4.26 | 5.39 | 6.03 | 0.77 | 0.10 | 1.68 | 0.05 | 0.33 | 0.02 |
| Tannic acid | ND | 0.69 | ND | 0.10 | 0.01 | ND | 0.01 | 0.10 | 0.01 |
| Protocatechui c acid | 0.05 | ND | 0.12 | 0.01 | 0.0003 | 0.04 | 0.001 | 0.01 | 0.0022 |
| Syringic acid | 3.76 | 2.08 | 1.11 | 0.15 | 0.001 | 1.04 | 0.03 | 0.38 | 0.01 |
| Vanillin | ND | ND | ND | 0.54 | ND | ND | ND | ND | ND |
| Coumaric acid | 0.20 | ND | 3.75 | 0.46 | 0.00110 | 1.74 | 0.02 | 0.22 | 0.01 |
| Caffeic acid | 1.58 | 0.47 | ND | ND | 0.00204 | ND | 0.0199 | ND | ND |
| Rutin | 3.22 | 1.57 | ND | ND | ND | ND | 4.86 | 3.85 | 3.95 |
| Myricetin | 56.25 | 52.67 | 49.07 | 57.94 | 54.60 | 55.62 | 24.27 | 17.04 | 20.46 |
| Apigenin | ND | ND | ND | ND | ND | ND | 0.828 | 7.3470 | 0.1902 |
| Quercetin | ND | 0.59 | 1.19 | 0.72 | ND | ND | ND | 0.16 | 0.0004 |
| Hydroxybenz oic acid | ND | 0.53 | ND | ND | 0.00032 | 0.04 | 0.01 | 0.03 | - |
| Luteolin | ND | ND | 0.94 | ND | ND | ND | ND | ND | ND |
| Kaempferol | ND | ND | 3.57 | ND | ND | ND | ND | ND | 0.01 |
| Transcinnami c acid | ND | ND | ND | ND | ND | 0.45 | ND | ND | 0.0014 |
| Vanillic acid | 1.35 | ND | 3.11 | ND | ND | 0.09 | ND | ND | ND |
| Ferulic acid | ND | ND | ND | ND | ND | ND | ND | ND | 0.004 |
ND not detected
Gallic acid, syringic acid, protocatechuic acid and caffeic acid were present in high amounts in CEW compared to other wines, which could be responsible for the higher antioxidant activity of these wines. Gallic acid was earlier found to decrease the secretion of inflammatory mediators in endothelial cells (Hidalgo et al. 2012). Phenolic and flavonoid compounds like syringic acid, protocatechuic acid, caffeic acid etc. have been shown to have antioxidant, anti-inflammatory and anti-atherosclerotic activities (Hidalgo et al. 2012; Eliana et al. 2014). Hydroxycinnamic and hydroxybenzoic acids like coumaric acid, vanillic acid and protocatechuic acid are not found in free form in fruits and occurs in free form only in processed foods like fermented foods (Claudine et al. 2004). Hence these compounds are not detected in the solid (data not shown) and 10th day must stage but found only in wine stage in the cultivated Emblica variety as free unconjugated forms were used as HPLC standards to authenticate the peaks. Thus it can be deducted that these compounds are hydrolyzed to the free form only after the 10th day of fermentation in the case of cultivated Emblica variety.
However, in the case of wild emblica and P. acidus, the hydrolysis could be taking place in the first 10 days of fermentation as evident from their detection in the solid (data not shown) and 10th day must stage. In juice, the presence of vanillic acid could be due to the presence of both free and conjugated forms of hydroxybenzoic acids in juices (De et al. 1999).
Comparison of antioxidant assays of different species of phyllanthus juices and wines
The 1, 1-diphenyl-2-picrylhydrazyl (DPPH) assay (DPPH) free radical-scavenging activities, total antioxidant activities and nitrite radical scavenging activities of the three Phyllanthus varieties are presented in Table 1. Lower IC50 indicates higher activity. The activities of the samples were in the following order CEW > WEW > PAW. The activities were found to be the lowest in PAW. The activities of the juices were higher than their corresponding wines.
Similar results were seen earlier in the case of pomegranate juice and wine (Maria et al. 2000) where the antioxidant activity of the juice was found to be higher than that of the wine.
The free radical scavenging activity of wines increased during the course of fermentation (from an IC50 of 25.80 μg/ml in CEM to 5 μg/ml in the case of CEW and from an IC50 of 10 μg/ml in WEM to 8 μg/ml in WEW in case of DPPH). This pattern of increase in antioxidant activity during the fermentation process was seen in a previous study in red wine (No et al. 2008). The activities of the solids which were left over after the collection of must were also tested for the antioxidant activity. However, these solids showed the least activity in all of the species selected and hence, the data have not been presented.
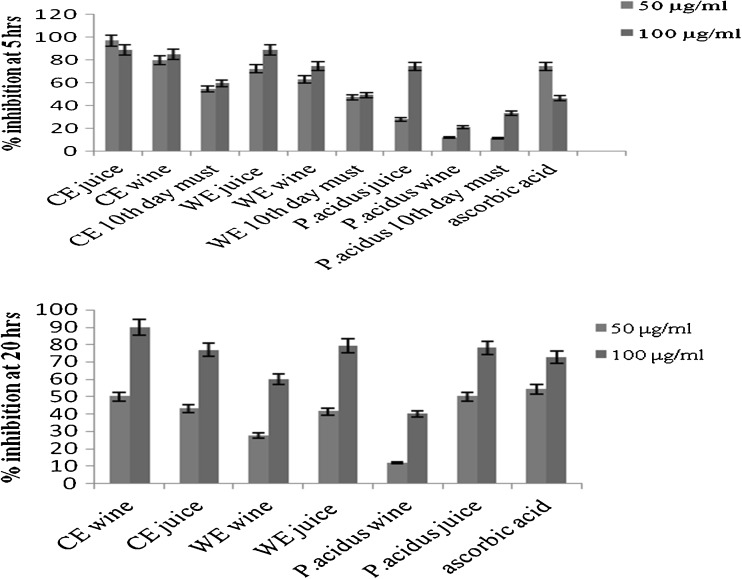
In the LDL oxidation prevention assay, among the wines tested, CEW showed the highest activity at both 5 h (50 μg/ml = 80 %, 100 μg/ml = 85 %) and 20 h (50 μg/ml = 50 %, 100 μg/ml = 90 %) and also at both the doses used. However, at 5 h, the activity of CEW was found to be lower than CEJ, as seen in Fig. 3. This situation was seen to change at 20 h where the activity of CEW was found to be higher than CEJ, showing that wine provided a sustained protection from LDL oxidation. The WEJ had higher activity than WEW at both doses and time intervals tested, although the difference was not statistically significant (i.e, 72.22 % for a dose of 50 μg/ml and 88.89 % at a dose of 100 μg/ml at 5 h and 41.46 % for 50 μg/ml and 79.27 % for 100 μg/ml at 20 h). The activity of P. acidus wine was found to be the lowest. Here again the activity was found to increase during the course of fermentation for all the three wine samples (increases being statistically significant only in the case of 20 h incubation time). The activity of CEW was found to be higher than that of ascorbic acid. Previously, LDL oxidation preventing ability of red wine was found to be greater than that of α-tocopherol (Landrault et al. 2001). The activity of solids collected on the 10th day of fermentation was the lowest in all varieties (data not shown).
Fig. 3.
LDL oxidation prevention assay after 5 h and 20 h incubation with oxidized LDL and the wines and juices of the three Phyllanthus varieties. CE Cultivated Emblica variety, WE Wild Emblica variety. Each bar graph value indicates value mean ± SD (standard deviation) from triplicate measurements (n = 3) and significance was found out at P ≤ 0.05 using Tukey’s test
Cell line antioxidant assays
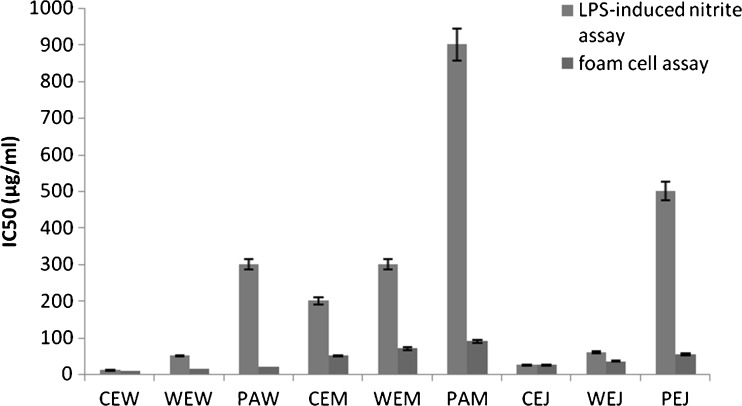
The process of LDL oxidation, due to inflammatory conditions, causes the macrophages to consume the oxidized LDL in an uncontrolled manner which then leads to formation of lipid-laden foam cells. This forms the basis of atherosclerotic lesions (Fermont et al. 1999). LPS-induced nitrite radical production activates macrophages and thus imitates the inflammatory conditions that cause foam cell formation (Guy et al. 1999). LPS-induced nitrite radical scavenging assay and oxidized LDL induced foam cell prevention assay (Fig. 4) showed the activity in the order CEW > WEW > PAW. These activities were found to be significantly higher than the corresponding juices –CEJ, WEJ and PAJ. Even here the activity increased significantly during the course of fermentation for all wines. The activity of the solids collected on the 10th day of fermentation was the lowest in all varieties (data not shown).
Fig. 4.
Nitrite assay in LPS stimulated and foam cell formation assay in ox-LDL induced RAW 264.7 cell lines incubated with wines and juices of the three Phyllanthus varieties. CEJ Cultivated Emblica Juice, WEJ Wild Emblica Juice, PEJ Phyllanthus acidus juice, CEM Cultivated Emblica Must, WEM Wild Emblica Must, PEM Phyllanthus acidus Must, CEW Cultivated Emblica Wine, WEW Wild Emblica Wine, PEW Phyllanthus acidus Wine. Lower IC50 indicates higher activity. Each bar graph value indicates value mean ± SD (standard deviation) from triplicate measurements (n = 3) and significance was found out at P ≤ 0.05 using Tukey’s test
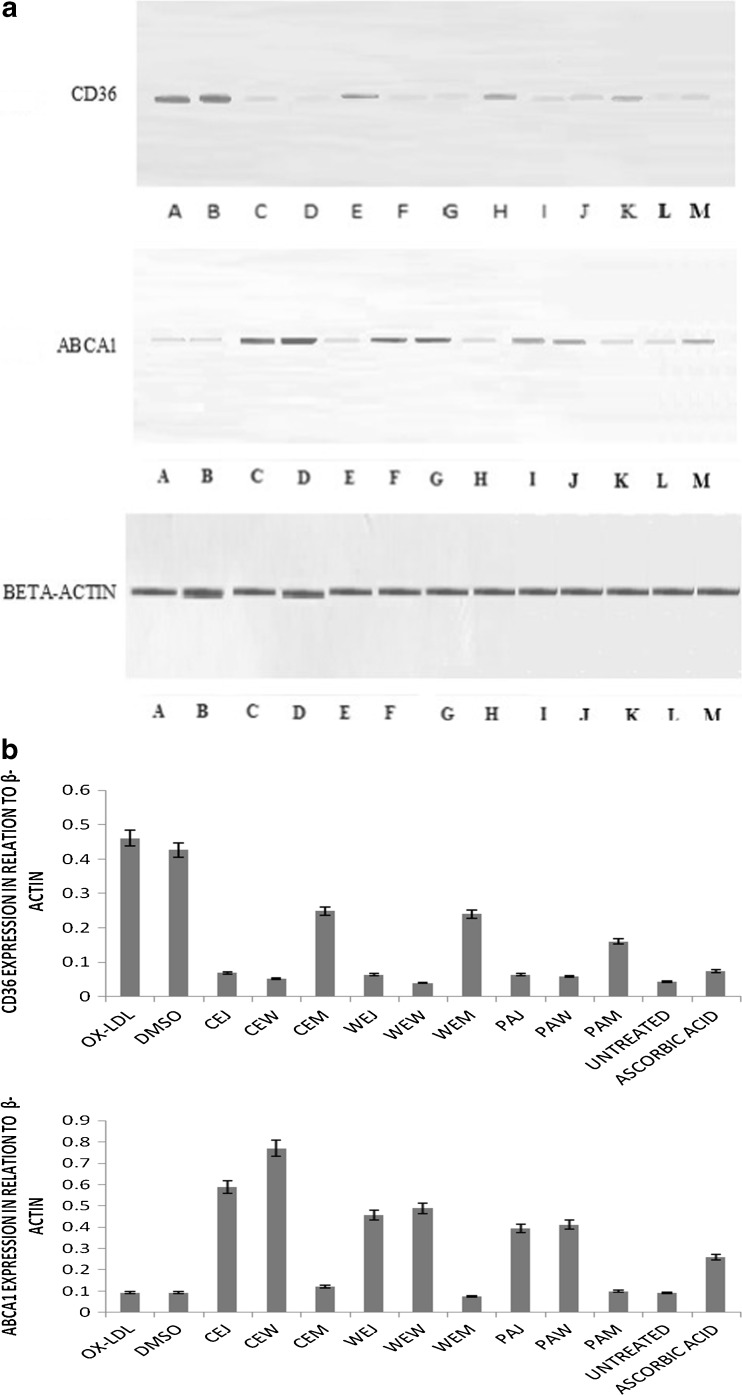
Previously red wine has been shown to prevent foam cell formation (Aviram and Fuhrman 2002). Under atherosclerotic conditions, increased expression of scavenger receptor CD36 causes the maximum uptake of ox-LDL in macrophages. This when complemented with reduced cholesterol efflux due to comparatively reduced ABCA1 expression causes cholesterol loading within macrophages eventually leading to foam cell formation. These foam cells are the building blocks of atherosclerotic plaque (Kuo-Yun et al. 2010). Western blot analysis (Fig.5) showed that highest ABCA1 expression was caused by CEW which was 8.17 fold and a 6.34 fold increase by CEJ compared to control (without extracts). The expression caused by all musts was very less. The lowest CD36 expression was again shown by CEW showing 8.85 fold decrease followed by CEJ showing 6.7 fold decrease compared to control thus explaining their high foam cell preventing abilities. In cell line assays, the higher activities of wines over their respective juices could be explained by the fact that alcohols increase the cellular permeability of wines and therefore help in carrying the important nutrients into the cells thus increasing the bioavailability of important nutrients (phenolics, flavonoids, organic acids) in the wines (Conceicao et al. 2013).
Fig. 5.
a Protein expression by western blot analysis (as described in text). All lanes except L lane has been treated with ox-LDL. A- ox-LDL treated, B-vehicle (DMSO) with ox-LDL, C- Cultivated Emblica Juice, D-Cultivated Emblica Wine, E- Cultivated Emblica Must, F- Wild Emblica Juice, G-Wild Emblica Wine, H- Wild Emblica Must, I- P.acidus juice, J- P.acidus wine, K- P.acidus must, L-cells treated with native LDL, M-ascorbic acid. b Quantification of western blots by Image J software. CEJ Cultivated Emblica Juice, WEJ Wild Emblica Juice, PEJ Phyllanthus acidus juice, CEM Cultivated Emblica Must, WEM Wild Emblica Must, PEM Phyllanthus acidus Must, CEW Cultivated Emblica Wine, WEW Wild Emblica Wine, PEW Phyllanthus acidus Wine. Each bar graph value indicates value mean ± SD (standard deviation) from triplicate measurements (n = 3) and significance was found out at P ≤ 0.05 using Tukey’s test
There was a statistically significant positive correlation (p < 0.05) between total phenolic content and total antioxidant assay (r2 = 0.80). Regression analysis showed a positive relationship between the two, supported by the fact that the phenolic compounds contributed 57.76 % of total antioxidant activity (r2 = 0.5776) at p < 0.05.
Sensory analysis
Initially many sensory descriptors were generated for wine samples and among those only few - selected descriptors were used in the score card. These descriptors were chosen based on their potential to bring out the flavour differences between the samples. The impact making sensory attributes were color (yellowish to brown), suspended particles, body, cloudiness, alcoholic aroma, fruity, fermented, molasses, sweet, sour, bitter, astringent. A `overall quality score’ was also included in the study to get a `single quality score or index’ for wine samples.
Colour
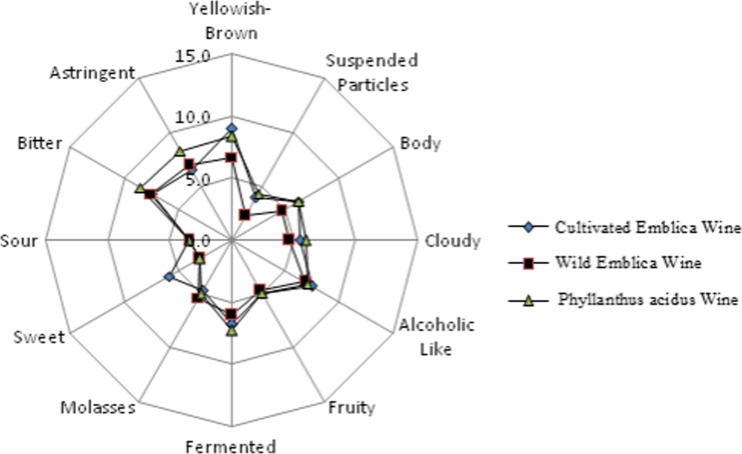
The primary sensory attributes is the colour. The colour of the wine samples was varied from yellowish to brown. Among the samples P. acidus was yellowish in colour, while the other samples were brownish in colour. Suspended tiny particles were observed in cultivated Emblica wine and also in wild Emblica wine samples, which can cause the settling problems while storage. However, P. acidus wine sample did not have any particles in it. The body of the wine samples was relatively thicker in cultivated and wild samples than Mysore wine sample. Cloudiness was also was found more in cultivated and wild Emblica wine samples. Sweetness perception was observed more in cultivated samples, then the other two wines. Undesirable astringency and bitterness sensory notes were highly associated with Wild Emblica wine samples. A sensory profilogram generated with different sensory descriptors is presented as Fig. 6 for comparison. Hedonic rating of wine samples (out of 9 point hedonic scale) was performed and was found to be 7.6 for cultivated Emblica wine, 7.1 for wild Emblica wine and 6.9 for P. acidus wine.
Fig. 6.
Sensory profilogram of wines of the three Phyllanthus varieties
Conclusion
Of the three wines studied, CEW was found to have the highest antioxidant activity as shown by DPPH assay, nitrite scavenging assay and total antioxidant activity. It also showed the highest ability to prevent LDL oxidation, macrophage foam cell formation (by the ingestion of oxidized LDL) and LPS –stimulated nitric oxide production in macrophages. Juices were shown to have high activities compared to their respective wines in in vitro assays. However in cell line assays like foam cell prevention assay, prevention of LPS-induced nitrite formation assay and CD36 and ABCA1 protein expression, the activities of CEW and WEW seemed to be higher than their corresponding juices. This was probably due to the alcohol content which increased the bioavailability of phenolic compounds in the fruit. Thus wine production can be seen as an important food formulation that can improve the bioavailability of essential nutrients in food. The antioxidant activity was found to increase during the course of fermentation. Phenolic content was also found to increase during the course of fermentation. HPLC analysis showed the presence of phenolic compounds with antioxidant activity like gallic acid, syringic acid, protocatechuic acid and caffeic acid and vanillic acid in cultivated Emblica wine. Further regression analysis showed that phenolic compounds are responsible for the antioxidant activity. Moreover cultivated Emblica wine was also sweeter compared to other wines.
Acknowledgments
The research was funded by the Council of Scientific and Industrial Research (CSIR), India. The authors thank Dr. Ravi R. from the department of Traditional Food & Sensory Science, CFTRI, Mysore for his help in sensory analysis of the wines.
Abbreviations
- CEW
Cultivated Emblica Wine
- CEM
must collected on the 10th day of fermentation of cultivated Emblica
- CES
fruit solids left after removal of CEM
- CEJ
Cultivated Emblica Juice
- WEW
Wild Emblica Wine
- WEM
must collected on the 10th day of fermentation of Wild Emblica
- WES
fruit solids left after removal of WEM
- WEJ
Wild Emblica Juice
- PAW
Phyllanthus acidus Wine
- PAM
must collected on the 10th day of fermentation of Phyllanthus acidus
- PAS
fruit solids left after removal of PAM
- PAJ
Phyllanthus acidus Juice
Compliance with ethical standards
Conflict of interest
The authors declare no conflict of interest.
Footnotes
Highlights
• Phenolic compounds were found to increase during the course of fermentation.
• Highest phenolic content seen in cultivated Emblica wine and juice.
• Antioxidant and anti-atherosclerotic activities increased during fermentation.
• Highest anti-atherosclerotic effect seen in cultivated Emblica wine and juice.
• The alcohols found in the wines were ethanol, n-butanol and isoamyl alcohol.
References
- Aviram M, Fuhrman B. Wine flavonoids protect against LDL oxidation and atherosclerosis. Ann N Y Acad Sci. 2002;957:146–161. doi: 10.1111/j.1749-6632.2002.tb02913.x. [DOI] [PubMed] [Google Scholar]
- Barthakur NN, Arnold NP. Chemical analysis of the emblic (Phyllanthus emblica L.) and its potential as a food source. Sci Hortic. 1991;47:99–105. doi: 10.1016/0304-4238(91)90031-S. [DOI] [Google Scholar]
- Basniwal PK, Suthar M, Rathore GS, Gupta R, Kumar V, Pareek A, Jain D. In-vitro antioxidant activity of hot aqueous extract of Helicteres isora Linn. fruits. Nat Prod Rad. 2009;8:483–487. [Google Scholar]
- Bhattacharya A, Chatterjee A, Ghosal S, Bhattacharya SK. Antioxidant activity of active tannoid principles of Emblica officinalis (amla) Indian J Exp Biol. 1999;37:676–680. [PubMed] [Google Scholar]
- Brand-Williams W, Cuvelier ME, Berset C. Use of a free radical method to evaluate antioxidant activity. LWT Food Sci Technol. 1995;28:25–30. doi: 10.1016/S0023-6438(95)80008-5. [DOI] [Google Scholar]
- Caputi A, Ueda M, Brown T. Spectrophotometric determination of ethanol in wine. Am J Enol. 1968;19:160–165. [Google Scholar]
- Claudine M, Augustin S, Christine M, Christian R, Liliana J. Polyphenols: food sources and bioavailability. Am J Clin Nutr. 2004;79:727–747. doi: 10.1093/ajcn/79.5.727. [DOI] [PubMed] [Google Scholar]
- Conceicao C, Ana F, Elisa K, Fatima M. Interaction of polyphenols with the intestinal and placental absorption of some nutrients and other compounds. In: Ronald RW, Victor RP, Sherma Z, editors. Polyphenols in human health and disease. USA: Academic Press; 2013. pp. 523–536. [Google Scholar]
- De AR, Martinez M, Vicente C, Legaz ME. Free and conjugated polyamines and phenols in raw and alkaline-clarified sugarcane juices. J Agric Food Chem. 1999;47:3086–3092. doi: 10.1021/jf980715+. [DOI] [PubMed] [Google Scholar]
- Eliana S, Mari J, Tiina T, Maija L, Vesa V, Ioanna C, Eva K, Sofia K, Paraskevi M. Phenolic acid composition, antiatherogenic and anticancer potential of honeys derived from various regions in Greece. PLoS One. 2014;9 doi: 10.1371/journal.pone.0094860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fairclough GF, Schuh J, Jr, Haschemeyer RH. Oxygen-mediated heterogeneity of apo-low-density lipoprotein. Proc Natl Acad Sci U S A. 1978;75:3173–3177. doi: 10.1073/pnas.75.7.3173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fermont L, Belguendouz L, Delpal S. Antioxidant activity of resveratrol and alcohol-free wine polyphenols related to LDL oxidation and polyunsatured fatty acids. Life Sci. 1999;64:2511–2521. doi: 10.1016/S0024-3205(99)00209-X. [DOI] [PubMed] [Google Scholar]
- Gordillo B, Cejudo-Bastante MJ, Rodriguez-Pulido FJ, Gonzalez-Miret LM, Heredia FJ. Application of the differential colorimetry and polyphenolic profile to the evaluation of the chromatic quality of Tempranillo red wines elaborated in warm climate. influence of the presence of oak wood chips during fermentation. Food Chem. 2013;141:2184–2190. doi: 10.1016/j.foodchem.2013.05.014. [DOI] [PubMed] [Google Scholar]
- Greenspan P, Yu H, Mao F, Gutman RL. Cholesterol deposition in macrophages: foam cell formation mediated by cholesterol-enriched oxidized low density lipoprotein. J Lipid Res. 1997;38:101–109. [PubMed] [Google Scholar]
- Gulcin I, Oktay M, Kufrevioglu OI, Aslan A. Determination of antioxidant activity of lichen Cetraria islandica (L) Ach J Ethnopharmacol. 2002;79:325–329. doi: 10.1016/S0378-8741(01)00396-8. [DOI] [PubMed] [Google Scholar]
- Guy MC, III, Stanley LH, Paul LF, Martha KC. The oxidation of lipoproteins by monocytes-macrophages- biochemical and biological mechanisms. J Biol Chem. 1999;274:25959–25962. doi: 10.1074/jbc.274.37.25959. [DOI] [PubMed] [Google Scholar]
- Hidalgo M, Martin-Santamaria S, Isidra R, Sanchez-Moreno C, Beatriz DPT, Gerald R, Sonia DPT. Potential anti-inflammatory, anti-adhesive, anti/estrogenic, and angiotensin-converting enzyme inhibitory activities of anthocyanins and their gut metabolites. Genes Nutr. 2012;7:295–306. doi: 10.1007/s12263-011-0263-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hyoung Lee S. Antioxidative activity of browning reaction products isolated from storage-aged Orange juice. J Agric Food Chem. 1992;40:550–552. doi: 10.1021/jf00016a004. [DOI] [Google Scholar]
- Libby P, Ridker PM, Hansson GK. Progress and challenges in translating the biology of atherosclerosis. Nature. 2011;473:317–325. doi: 10.1038/nature10146. [DOI] [PubMed] [Google Scholar]
- Jayaprakasha GK, Sakariah KK. Determination of organic acids in leaves and rinds of Garcinia indica (desr.) by LC. J Pharm Biomed Anal. 2002;28:379–384. doi: 10.1016/S0731-7085(01)00623-9. [DOI] [PubMed] [Google Scholar]
- Kuo-Yun L, Li-Chieh C, Kuo-Hui S, Yuan-Bin Y, Yu Ru K, Sheng-Huang H, Yu-Chu H, Chien-Yu C, Li-Ching C, Ching-Chian P, Tzong-Shyuan L. Erythropoietin suppresses the formation of macrophage foam cells role of liver X receptor α. Circulation. 2010;121:1828–1837. doi: 10.1161/CIRCULATIONAHA.109.876839. [DOI] [PubMed] [Google Scholar]
- Landrault N, Poucheret N, Ravel P, Gasc F, Cros G, Teissedre PL. Antioxidant capacities and phenolics levels of French wines from different varieties and vintages. J Agric Food Chem. 2001;49:3341–3348. doi: 10.1021/jf010128f. [DOI] [PubMed] [Google Scholar]
- Lee MY, Yuk JE, Kwon OK, Oh SR, Lee HK, Ahn KS. Zuonin B inhibits lipopolysaccharide-induced inflammation via downregulation of the ERK1/2 and JNK pathways in RAW 264.7 macrophages. Evid Based Complement Alternat Med. 2012;2012:1–14. doi: 10.1155/2012/728196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Manach C, Mazur A, Scalbert A. Polyphenols and prevention of cardiovascular diseases. Curr Opin Lipidol. 2005;16:77–84. doi: 10.1097/00041433-200502000-00013. [DOI] [PubMed] [Google Scholar]
- Maria IG, Francisco ATB, Betty HP, Deirdre MH, Adel AK. Antioxidant activity of pomegranate juice and its relationship with phenolic composition and processing. J Agric Food Chem. 2000;48:4581–4589. doi: 10.1021/jf000404a. [DOI] [PubMed] [Google Scholar]
- No JD, Dae-Hyoung L, Young-Soo H, Sang-Han L, Dae-Hyung L, Jong-Soo L. Changes of physicochemical properties and antioxidant activities of red wines during fermentation and post-fermentation. Kor J Microbiol Biotechnol. 2008;36:67–71. [Google Scholar]
- Orrego R, Leiva E, Cheel J. Inhibitory effect of three C-glycosylflavonoids from Cymbopogon citratus (lemongrass) on human low density lipoprotein oxidation. Molecules. 2009;14:3906–3913. doi: 10.3390/molecules14103906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prieto P, Pineda M, Aguilar M. Spectrophotometric quantitation of antioxidant capacity through the formation of a phosphomolybdenum complex: specific application to the determination of vitamin E. Anal Biochem. 1999;269:337–341. doi: 10.1006/abio.1999.4019. [DOI] [PubMed] [Google Scholar]
- Rai AK, Maya P, Anu AKA. Production of Garcinia wine: changes in biochemical parameters, organic acids and free sugars during fermentation of Garcinia must. Int J Food Sci Technol. 2010;45:1330–1336. doi: 10.1111/j.1365-2621.2010.02181.x. [DOI] [Google Scholar]
- Renaud S, De Lorgeril M. Wine, alcohol, platelets, and the French paradox for coronary heart disease. Lancet. 1992;339:1523–1526. doi: 10.1016/0140-6736(92)91277-F. [DOI] [PubMed] [Google Scholar]
- Ruangchakpet A, Tanaboon S. Effect of browning on total phenolic, flavonoid content and antioxidant activity in Indian gooseberry (Phyllanthus emblica Linn.) Kasetsart J (Nat Sci) 2007;41:331–337. [Google Scholar]
- Scartezzini P, Speroni E. Review on some plants of Indian traditional medicine with antioxidant activity. J Ethnopharmacol. 2000;71:23–43. doi: 10.1016/S0378-8741(00)00213-0. [DOI] [PubMed] [Google Scholar]
- Singleton VL, Noble AC (1976) Wine flavor and phenolic substances. In: Charalambous G, Katz I (eds), Phenolic, Sulfur, and Nitrogen Compounds in Food Flavors, ACS publications, Washington D.C., pp 47–70
- Uttara B, Singh AV, Zamboni P, Mahajan RT. Oxidative stress and neurodegenerative diseases: a review of upstream and downstream antioxidant therapeutic options. Curr Neuropharmacol. 2009;7:65–74. doi: 10.2174/157015909787602823. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wasila H, Li X, Liu L, Ahmad I, Ahmad S. Peel effects on phenolic composition, antioxidant activity, and making of pomegranate juice and wine. J Food Sci. 2013;78:C1166–C1172. doi: 10.1111/1750-3841.12204. [DOI] [PubMed] [Google Scholar]
- Wu SC, Yi-Shun S, Huang-Yu C. Antioxidant properties of lactobacillus- fermented and non-fermented Graptopetalum paraguayense E.Walther at different stages of maturity. Food Chem. 2011;129:804–809. doi: 10.1016/j.foodchem.2011.05.025. [DOI] [PubMed] [Google Scholar]