Abstract
Analysis of repetitive DNA sequence content and divergence among the repetitive functional classes is a well-accepted approach for estimation of inter- and intra-generic differences in plant genomes. Among these elements, microsatellites, or Simple Sequence Repeats (SSRs), have been widely demonstrated as powerful genetic markers for species and varieties discrimination. We present PlantFuncSSRs platform having more than 364 plant species with more than 2 million functional SSRs. They are provided with detailed annotations for easy functional browsing of SSRs and with information on primer pairs and associated functional domains. PlantFuncSSRs can be leveraged to identify functional-based genic variability among the species of interest, which might be of particular interest in developing functional markers in plants. This comprehensive on-line portal unifies mining of SSRs from first and next generation sequencing datasets, corresponding primer pairs and associated in-depth functional annotation such as gene ontology annotation, gene interactions and its identification from reference protein databases. PlantFuncSSRs is freely accessible at: http://www.bioinfocabd.upo.es/plantssr.
Keywords: short tandem repeats (STRs), NGS, gene ontology (GO), inter-pro, functional domains markers
Introduction
Identification of repetitive patterns in genomic DNA has proved to be a powerful approach to reveal diversity and to discriminate plant populations and individuals within species. Microsatellites or Simple Sequence Repeats (SSRs) formed as a result of the strand-slippage mechanism (Schlötterer and Harr, 2001) have been used widely as functional genetic markers (Studer et al., 2010), for testing genetic fidelity, genetic variability (Rahman and Rajora, 2002; Schellenbaum et al., 2008) and for population genetic studies (Sim et al., 2009). However, the previously described approaches such as by screening the small insert genomic DNA libraries (Shokeen et al., 2007) are time consuming and not so cost effective. Furthermore, SSRs identified by such approaches have no certainty of association to the functional domains. Leveraging the computational advances, in silico mining approaches using transcriptomics have filled a major gap in the development of these functional classes of markers (Sablok and Shekhawat, 2008; Sablok et al., 2011), which could be potentially used for developing the markers harboring the functional domains for marker assisted gene selection, genotyping, and anchoring quantitative trait localization (QTL; Parida et al., 2010; Kujur et al., 2013) mainly due to the associative nature of the mined SSRs to the coding region variations and the associated functional variations.
Recently, several SSRs have been linked to putative functional domains; classifying them into a new class of functional markers called simple sequence repeats functional domains markers (SSR-FDMs) in model and non-model species (Yu et al., 2010; Bhattacharyya et al., 2014). Realizing the wide importance of SSRs, several online repositories and data mining tools have been developed to address the need for on-line mining of these markers in case of nuclear genomes such as PlantMarkers (Rudd et al., 2005), SSR Biome and SSR taxonomy (Jewell et al., 2006), UgMicroSatDb (Aishwarya and Sharma, 2008), MoccaDB (Plechakova et al., 2009), CicArMiSatDB (Doddamani et al., 2014), and for Coffee expressed sequence tags (ESTs) (Poncet et al., 2006) to assist the mining of the SSRs. However, there are some limitations to the previously developed tools that have restricted, in particular, the possibility to make comparisons across different datasets from different species as they either lack integration of the browsing platform with unified annotations or they are oriented toward specific species such as CicArMiSatDB (Doddamani et al., 2014), and FmMDb (B et al., 2013). In case of organelle genomes, we previously established ChloroMitoSSRDB (Sablok et al., 2013) and ChloroMitoSSRDB 2.00 (Sablok et al., 2015) to provide the large-scale access to the organelle derived markers.
Next generation sequencing (NGS) provides a cost-efficient way of transcript identification and facilitates the development of transcript based SSRs markers for model and non-model species, which has resulted in rapid increases in the data made available online. However, much of this data is scattered across numerous websites and has not been mined or annotated for the identification of functional SSRs. Recently, there have been some efforts to consolidate such data for example TropiTree1 is a repository displaying the mined SSRs from NGS transcript assemblies for 24 tropical plants (Russell et al., 2014). Taking into account the limitations mentioned, we were motivated to develop PlantFuncSSRs, available at http://www.bioinfocabd.upo.es/plantssr, which is a unified functional SSRs portal displaying mined functional SSRs from 274 ESTs based transcript assemblies, and more than 100 NGS transcripts assemblies. PlantFuncSSRs also provides detailed primer pair information, functional annotations, and putative homologs to the transcript assemblies in Uniprot and curated SSR-FDMs in a single unified platform. We believe that the availability of the above resource will aid the rapid development of functional SSRs in non-model plant species.
Materials and Methods
Data Resources for PlantFuncSSRs
To integrate previously published plant EST data, all Putative Unique Transcripts (PUT) representing 273 transcript assemblies were downloaded from PlantGDB (Version release 187) available from http://www.plantgdb.org/ (Dong et al., 2004). Additionally, version control 74 NGS transcriptomes available at PhytoMetaSync2 (Facchini et al., 2012; Xiao et al., 2013), 14 medicinal plant transcriptomes available from medicinal plant genomics resource (MPGR)3 (Góngora-Castillo et al., 2012; Góngora-Castillo and Buell, 2013) and 3 Brachypodium sylvaticum transcriptomes available from http://jaiswallab.cgrb.oregonstate.edu/genomics (Fox et al., 2013) were downloaded, representing a total of 364 plant species.
SSRs Identification and Functional Assignments
For systematic identification of SSR, all the transcripts (ESTs as well as NGS) assemblies were first scanned for the presence of the homopolymer errors and sequence ambiguity was removed using the est_trimmer tool available at: http://pgrc.ipk-gatersleben.de/misa/download/est_trimmer.pl with the following settings: -amb = 2.50 -tr5 = T, 5.50 -tr3 = A, 5.50. Following the transcript ambiguity removal and trimming of the homopolymer runs, MISA (MIcroSAtellite identification tool) (Thiel et al., 2003) was deployed to identify the microsatellites. In the present version of the PlantFuncSSRs, we classified microsatellites as repetitive stretches of motifs of a minimum and 12-mer repetitive stretch as mono-, 6-mer repetitive stretches as di, 4-mer repetitive stretches of tri- and tetra-, and a minimum of 3-mer repetitive stretch as penta- and hexa-nucleotide. Additionally, the identified SSRs have been classified into perfect and compound repeats, with compound repeats interrupted by a minimum of 100 bp as previously described (Victoria et al., 2011). Primer pairs were designed for all of the identified SSRs using primer3 available from primer3.sourceforge.net (Untergasser et al., 2012) using the settings as described in MISA (Thiel et al., 2003).
Following SSRs identification, in-depth functional annotation of the identified SSRs was carried out using the standalone annotator Sma3s (Muñoz-Mérida et al., 2014), which uses the plant taxonomic division set in the Uniprot database4, including both Swiss-Prot and TrEMBL sections to enrich the final annotation. The annotations gave the found Gene Ontology (GO) terms which were subsequently linked to their GO_SLIM terms using the plant GO slim available from www.geneontology.org, in order to simplify the GO terms and allow cross-comparison. In this way, each SSRs sequence was identified with the more probable gene name and description, as well as both GO terms from the existing three categories and Swiss-Prot keywords, all of them for cataloging the SSRs and assigning functional domains. The IntAct annotations and Interactions were cross-linked using the IntAct resources available from EBI at: http://www.ebi.ac.uk/intact/. The functional SSRs annotation also includes putative InterPro domains (Quevillon et al., 2005, pathways from UniProt to have more details of the involved biological processes. PlantFuncSSRs presents only those SSRs, which have functional annotations appended to them and are thus termed as SSR-functional markers.
Results and Discussion
PlantFuncSSRs Architecture and Visualization
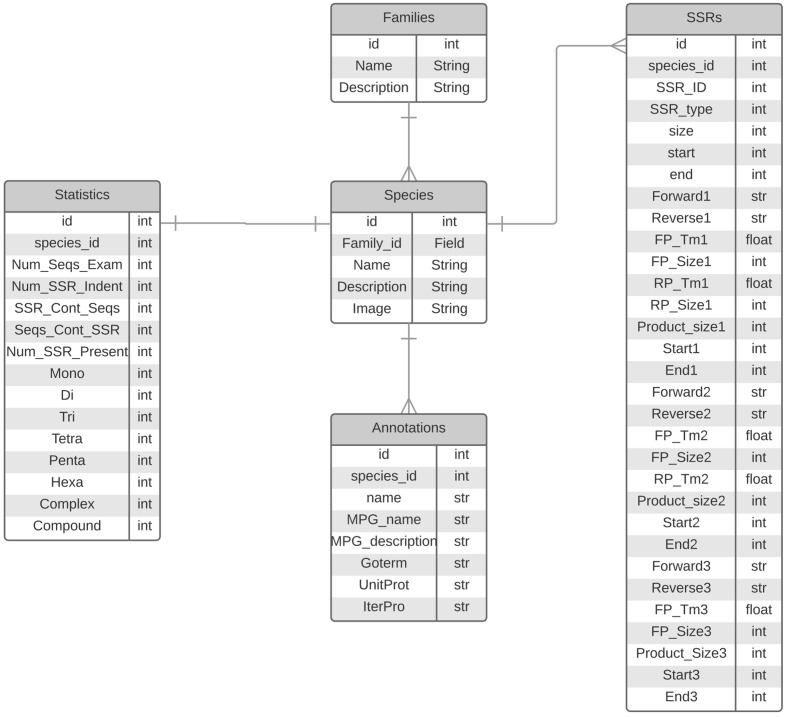
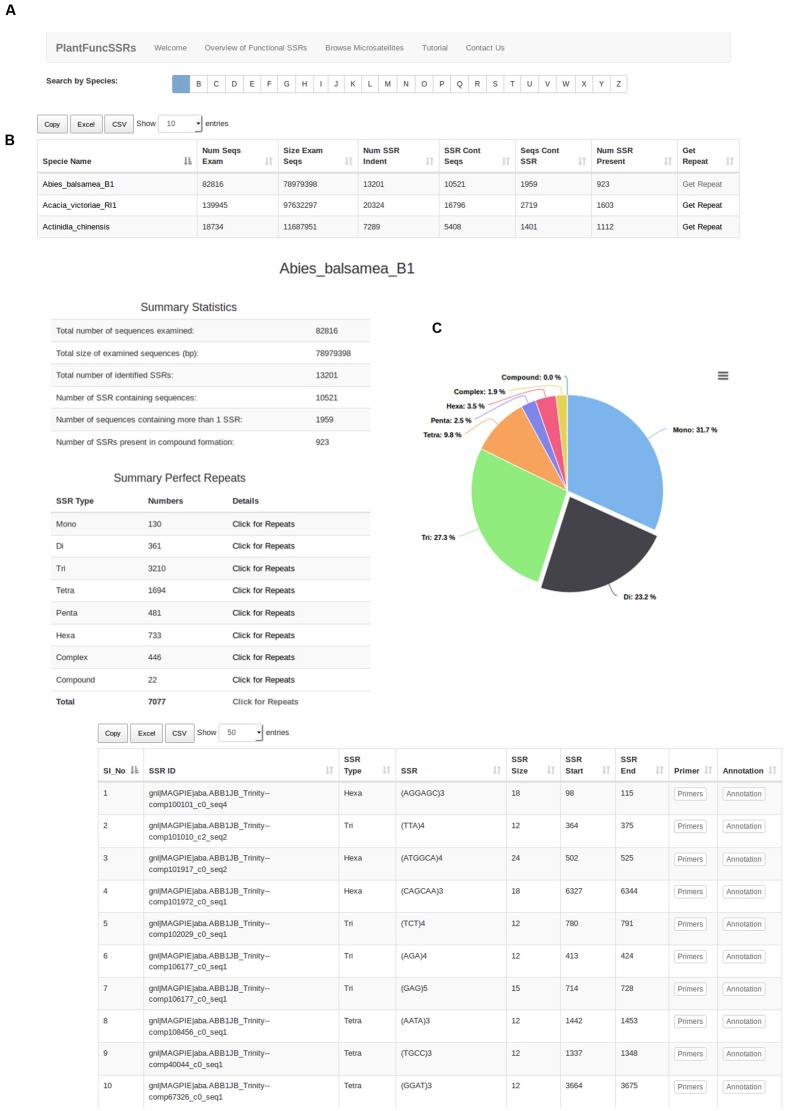
Expressed sequence tags and NGS based Transcriptome reconstruction represent the functional portion of the genome and have been widely used as resources to mine and develop functional markers. Developing an efficient browsing system for the mining of repeats is an important task, as this can be widely applied to a wide range of on-going plant breeding and crop improvement research. To develop an efficient browsing system, PlantFuncSSRs architecture has been developed using Ruby Rails and MySQL, which provides faster integration and query based searches to the users. The current version of the PlantFuncSSRs presents more than 2 million SSRs and SSR-FDMs from 364 species for easy access and browsing of transcript derived plant SSRs across the plant kingdom (Table 1). These species are ranging from important crops to wild species, from mono- to di-cots, from annual to polyannual and wood species. Integration of visualization features with the rapid mining of the data is a key central feature that has been implemented in the PlantFuncSSRs. A schema of the database architecture in the form of entity-relationship is given in Figure 1. For the visualization of the SSRs and the associated information, several hierarchal levels of classified information have been inter-linked in PlantFuncSSRs (Figure 2). The front-end portal is user-friendly and allows the end-users to search SSRs as “species-wise”, “family wise”, or “advanced search menu” (Figure 2). A quick search implementation pattern displays the embedded species information in quick select “species” and “families”, which are hyperlinked pages to the respective species and provide a quick view of the functional SSRs present in each species. Figure 3 shows the webpage browsing of PlantFuncSSRs with detailed classification of the identified SSRs for user-selected species of interest. Alphabetical classification of the species provides an additional advantage for the users to quickly look for their species of interest (Figure 3).
Table 1.
Table describing the classified repeats types and embedded functional categories in PlantFuncSSRs.
| Type of SSRs | Number of SSRs |
|---|---|
| P1 SSRs-FDMs | 221008 |
| P2 SSRs-FDMs | 200702 |
| P3 SSRs-FDMs | 1067949 |
| P4 SSRs-FDMs | 358245 |
| P5 SSRs-FDMs | 102593 |
| P6 SSRs-FDMs | 142452 |
| Compound SSRs-FDMs (C and C∗) | 292472 |
| Functionally embedded SSRs annotations | |
| Gene names | 2278574 |
| Descriptions | 2332906 |
| Gene ontologies | 1986736 |
| Uniprot (keywords) | 2122976 |
| InterPro domains | 2172553 |
FIGURE 1.
Entry–Entity relationship diagram of PlantFuncSSRs.
FIGURE 2.
PlantFuncSSRs: Schematic view of the PlantFuncSSRs and the browsing options implemented in PlantFuncSSRs. The structure of PlantFuncSSRs allows for the browsing of the functional SSRs either according to the species or according to the family.
FIGURE 3.
Alphabet sorting of the species names and search patterns (A); Species specific page showing the information on the identified Simple Sequence Repeats (SSRs) and also the functional SSRs. “Click for repeats” pages are directly hyperlinked to the functional SSRs (B); Weblayout describing the functional repeats identified in the respective plant species with information on type of repeat, classification of repeat, size, motif, start, and end coordinates and associated primers and functional annotation (C).
Each record in the species displays the Species_Name, Num_Seqs_Exam, Size_Exam_Seqs, Num_SSR_Ident, SSR_Cont_Seqs, Seqs_Cont_SSR, and Num_SSR_Present providing summarized information on the number of the identified SSRs for that particular species of interest lined to the primer pair information and high throughput functional annotation (Figure 3). In PlantFuncSSRs, each species page has been hyperlinked to the corresponding repeat information pages that present detailed information on several statistics such as total number of sequences examined, total size of examined sequences (bp), total number of identified SSRs, number of SSR containing sequences, number of sequences containing more than one SSR and compound SSRs (Figure 3). In addition, to this summary information, each species classified page also details the types and distribution of the repeats in tabular format, which can be sorted “on the fly”. An integral part of PlantFuncSSRs is to describe the associated primer pair information for each species to facilitate the development of functional SSRs for diversity analysis. To augment such capacity, each functional SSR has been associated with primer pages and detailed functional annotations, which describes the set of the “ready to use” primers for the functional validation of the corresponding SSRs (Figure 4).
FIGURE 4.
Pop-up Primer display window for the user selected functional SSRs (A); Pop-up window showing high throughput functional annotation for the user selected functional SSRs (B).
Functional SSRs and Functional Importance of PlantFuncSSRs
Microsatellites (SSRs) have been shown to be regulators of a number of plant genes demonstrating their importance as key players in regulating plant function (Faville et al., 2004). FuncPlantSSRs offers a wide variety of functional annotations for the identified SSRs such as GO terms, GO slim categories, pathways, descriptions to identify the sequences and comparing with putative homologues, and motif and domain modules to offer the domain architecture for the sequences. Recently, increasing interest toward the functional linkage of the markers to the domain association and function can be seen from several recent reports in plants such as Ocimum basilicum (Gupta et al., 2010), Seasmum indicum (Bhattacharyya et al., 2014), Elaeis guineensis (Tranbarger et al., 2012), and Camellia sinensis (Sahu et al., 2012) suggesting the role of the functional SSRs as important markers for developing the functional genic approaches for marker enrichment in plants. Nonetheless, established reports of the functional association of the repeats with the catalytic domains (Parida et al., 2010; Yu et al., 2010) has been widely developed. For quick advanced searches, PlantFuncSSRs offer several functionalities, such as searches customized and optimized on various hierarchal levels i.e., Family, Species, Type of Repeat, Number of Repeat, Functional annotation, GO annotation, and IPR annotations (Figure 2). Availability of the curated information provides end users with the flexibility to narrow their searches to functional SSRs linked to specific categories, motif types or functional annotations. Taking into account the vast amount of the species coverage and associated functional SSRs present in the PlantFuncSSRs, we believe that the PlantFuncSSRs provides access to the most comprehensive catalog available for the functional SSRs from plant transcriptomes.
Conclusion
In the present version of the PlantFuncSSRs, we bring together under a unified portal the mining of the SSRs from the publically available first and second generation datasets. PlantFunctSSRs has been designed with an aim to serve as a stand-alone single access platform for the analysis of functional SSRs from first and NGS datasets for a large number of sequenced plant transcriptomes. In addition to providing the most comprehensive available resource for exploring and validating plant functional SSRs, the built in annotation platform will allow the users to have wide access to the functional relevance of the validated SSRs thus provides a valuable functional SSRs resource to support plant diversity, population and functional marker research.
Author Contributions
GS conceived and designed the research, identified SSRs and linked the SSRs to functions, AP and AM-M provided the annotation, TD build the database and the web-interface, TYS helped in the data integration, CSCS hosted the database, GS wrote the manuscript, NP, AS, PR, and JAH provided revisions. All authors have read and approved the manuscript.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
Gaurav Sablok thanks Plant Functional Biology and Climate Change Cluster (C3), University of Technology Sydney, PO Box 123, Broadway, NSW 2007, Australia, for providing the computational facilities. An internal grant number to GS (2226018) supported this work. JAH and TYS were partially supported by High Impact Research Chancellory Grant UM.C/625/1/HIR/MOHE/SCI/19 from the University of Malaya.
Footnotes
References
- Aishwarya V., Sharma P. C. (2008). UgMicroSatdb: database for mining microsatellites from unigenes. Nucleic Acids Res. 36 D53–D56. 10.1093/nar/gkm811 [DOI] [PMC free article] [PubMed] [Google Scholar]
- B V. S., Muthamilarasan M., Misra G., Prasad M. (2013). FmMDb: a versatile database of foxtail millet markers for millets and bioenergy grasses research. PLoS ONE 8:e71418 10.1371/journal.pone.0071418 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bhattacharyya U., Pandey S. K., Dasgupta T. (2014). Identification of EST-SSRs and FDM in sesame (Sesamum indicum L.) through data mining. Sch. J. Agric. Sci. 4 60–69. [Google Scholar]
- Doddamani D., Katta M. A., Khan A. W., Agarwal G., Shah T. M., Varshney R. K. (2014). CicArMiSatDB: the chickpea microsatellite database. BMC Bioinformatics 15:212 10.1186/1471-2105-15-212 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dong Q., Schlueter S. D., Brendel V. (2004). PlantGDB, plant genome database and analysis tools. Nucleic Acids Res. 32 D354–D359. 10.1093/nar/gkh046 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Facchini P. J., Bohlmann J., Covello P. S., De Luca V., Mahadevan R., Page J. E., et al. (2012). Synthetic biosystems for the production of high-value plant metabolites. Trends Biotechnol. 30 127–131. 10.1016/j.tibtech.2011.10.001 [DOI] [PubMed] [Google Scholar]
- Faville M. J., Vecchies A. C., Schreiber M., Drayton M. C., Hughes L. J., Jones E. S., et al. (2004). Functionally associated molecular genetic marker map construction in perennial ryegrass (Lolium perenne L.). Theor. Appl. Genet. 110 12–32. 10.1007/s00122-005-1959-y [DOI] [PubMed] [Google Scholar]
- Fox S. E., Preece J., Kimbrel J. A., Marchini G. L., Sage A., Youens-Clark K., et al. (2013). Sequencing and de novo transcriptome assembly of Brachypodium sylvaticum (Poaceae). Appl. Plant Sci. 1:1200011 10.3732/apps.1200011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Góngora-Castillo E., Buell C. R. (2013). Bioinformatics challenges in de novo transcriptome assembly using short read sequences in the absence of a reference genome sequence. Nat. Prod. Rep. 30 490–500. 10.1039/c3np20099j [DOI] [PubMed] [Google Scholar]
- Góngora-Castillo E., Childs K. L., Fedewa G., Hamilton J. P., Liscombe D. K., Magallanes-Lundback M., et al. (2012). Development of transcriptomic resources for interrogating the biosynthesis of monoterpene indole alkaloids in medicinal plant species. PLoS ONE 7:e52506 10.1371/journal.pone.0052506 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gupta S., Shukla R., Roy S., Naresh S., Sharma A. (2010). In silico SSR and FDM analysis through EST sequences in Ocimum basilicum. Plant Omics J. 3 121–128. [Google Scholar]
- Jewell E., Robinson A., Savage D., Erwin T., Love C. G., Lim G. A. C., et al. (2006). SSRPrimer and SSR taxonomy tree: biome SSR discovery. Nucleic Acids Res. 34 W656–W659. 10.1093/nar/gkl083 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kujur A., Bajaj D., Saxena M. S., Tripathi S., Upadhyaya H. D., Gowda C. L. L., et al. (2013). Functionally relevant microsatellite markers from chickpea transcription factor genes for efficient genotyping applications and trait association mapping. DNA Res. 20 355–373. 10.1093/dnares/dst015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muñoz-Mérida A., Viguera E., Claros M. G., Trelles O., Pérez-Pulido A. J. (2014). Sma3s: a three-step modular annotator for large sequence datasets. DNA Res. 21 341–353. 10.1093/dnares/dsu001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parida S. K., Pandit A., Gaikwad K., Sharma T. R., Srivastava P. S., Singh N. K., et al. (2010). Functionally relevant microsatellites in sugarcane unigenes. BMC Plant Biol. 10:251 10.1186/1471-2229-10-251 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Plechakova O., Tranchant-Dubreuil C., Benedet F., Couderc M., Tinaut A., Viader V., et al. (2009). MoccaDB - an integrative database for functional, comparative and diversity studies in the Rubiaceae family. BMC Plant Biol. 9:123 10.1186/1471-2229-9-123 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poncet V., Rondeau M., Tranchant C., Cayrel A., Hamon S., de Kochko A., et al. (2006). SSR mining in coffee tree EST databases: potential use of EST-SSRs as markers for the Coffea genus. Mol. Genet. Genom. 6 436–449. 10.1007/s00438-006-0153-5 [DOI] [PubMed] [Google Scholar]
- Quevillon E., Silventoinen V., Pillai S., Harte N., Mulder N., Apweiler R., et al. (2005). InterProScan: protein domains identifier. Nucleic Acids Res. 33 116–120. 10.1093/nar/gki442 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rahman M. H., Rajora O. P. (2002). Microsatellite DNA fingerprinting, differentiation, and genetic relationships of clones, cultivars, and varieties of six poplar species from three sections of the genus Populus. Genome 45 1083–1094. 10.1139/g02-077 [DOI] [PubMed] [Google Scholar]
- Rudd S., Schoof H., Mayer K. (2005). PlantMarkers–a database of predicted molecular markers from plants. Nucleic Acids Res. 33 D628–D632. 10.1093/nar/gki074 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Russell J. R., Hedley P. E., Cardle L., Dancey S., Morris J., Booth A., et al. (2014). TropiTree: an NGS-based EST-SSR resource for 24 tropical tree species. PLoS ONE 9:e102502 10.1371/journal.pone.0102502 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sablok G., Luo C., Lee W. S., Rahman F., Tatarinova T. V., Harikrishna J. A., et al. (2011). Bioinformatic analysis of fruit-specific expressed sequence tag libraries of Diospyros kaki Thunb: view at the transcriptome at different developmental stages. Biotech 1 35–45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sablok G., Mudunuri S. B., Patnana S., Popova M., Fares M. A., Porta N. L. (2013). ChloroMitoSSRDB: open source repository of perfect and imperfect repeats in organelle genomes for evolutionary genomics. DNA Res. 20 127–133. 10.1093/dnares/dss038 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sablok G., Padma Raju G. V., Mudunuri S. B., Prabha R., Singh D. P., Baev V., et al. (2015). ChloroMitoSSRDB 2.00: more genomes, more repeats, unifying SSRs search patterns and on-the-fly repeat detection. Database (Oxford) 2015:bav084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sablok G., Shekhawat N. S. (2008). Bioinformatics analysis of distribution of microsatellite markers (SSRs)/single nucleotide polymorphism (SNPs) in expressed transcripts of Prosopis juliflora: frequency and distribution. J. Comput. Sci. Syst. Biol. 1 87–91. [Google Scholar]
- Sahu J., Sarmah R., Dehury B., Sarma K., Sahoo S., Sahu M., et al. (2012). Mining for SSRs and FDMs from expressed sequence tags of Camellia sinensis. Bioinformation 8 260–266. 10.6026/97320630008260 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schellenbaum P., Mohler V., Wenzel G., Walter B. (2008). Variation in DNA methylation patterns of grapevine somaclones (Vitis vinifera L.). BMC Plant Biol. 8:78 10.1186/1471-2229-8-78 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schlötterer C., Harr B. (2001). “Microsatellite instability,” in Encyclopedia of Life Sciences (New York, NY: John Wiley & Sons, Ltd; ), 1–4. [Google Scholar]
- Shokeen B., Sethy N. K., Kumar S., Bhatia S. (2007). Isolation and characterization of microsatellite markers for analysis of molecular variation in the medicinal plant Madagascar periwinkle (Catharanthus roseus (L.) G. Don). Plant Sci. 172 441–451. 10.1016/j.plantsci.2006.10.010 [DOI] [Google Scholar]
- Sim S. C., Yu J. K., Jo Y. K., Sorrells M. E., Jung G. (2009). Transferability of cereal EST-SSR markers to ryegrass. Genome 52 431–437. 10.1139/g09-019 [DOI] [PubMed] [Google Scholar]
- Studer B., Kölliker R., Muylle H., Asp T., Frei U., Roldán-Ruiz I. (2010). EST-derived SSR markers used as anchor loci for the construction of a consensus linkage map in ryegrass (Lolium spp.). BMC Plant Biol. 10:177 10.1186/1471-2229-10-177 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thiel T., Michalek W., Varshney R., Graner A. (2003). Exploiting EST databases for the development and characterization of gene-derived SSR-markers in barley (Hordeum vulgare L.). Theor. Appl. Genet. 106 411–422. 10.1007/s00122-002-1031-0 [DOI] [PubMed] [Google Scholar]
- Tranbarger T. J., Kluabmongkol W., Sangsrakru D., Morcillo F., Tregear J. W., Tragoonrung S., et al. (2012). SSR markers in transcripts of genes linked to post-transcriptional and transcriptional regulatory functions during vegetative and reproductive development of Elaeis guineensis. BMC Plant Biol. 12:1 10.1186/1471-2229-12-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Untergasser A., Cutcutache I., Koressaar T., Ye J., Faircloth B. C., Remm M., et al. (2012). Primer3-new capabilities and interfaces. Nucleic Acids Res. 40 e115 10.1093/nar/gks596 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Victoria F. C., da Maia L. C., de Oliveira A. C. (2011). In silico comparative analysis of SSR markers in plants. BMC Plant Biol. 11:15 10.1186/1471-2229-11-15 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xiao M., Zhang Y., Chen X., Lee E. J., Barber C., Chakrabarty R., et al. (2013). Tanscriptome analysis based on next-generation sequencing of non-model plants producing specialized metabolites of biotechnological interest. J. Biotechnol. 166 122–134. 10.1016/j.jbiotec.2013.04.004 [DOI] [PubMed] [Google Scholar]
- Yu J. K., Paik H., Choi J. P., Han J. H., Choe J. K., Hur C. G. (2010). Functional domain marker (FDM): an in silico demonstration in Solanaceae using simple sequence repeats (SSRs). Plant Mol. Biol. Rep. 28 352–356. 10.1007/s11105-009-0154-8 [DOI] [Google Scholar]