Abstract
Aim:
Diabetes mellitus is a chronic condition characterized by hyperglycemia. The objective of the study was to estimate the prevalence of cardiac autonomic neuropathy in a rural area of South India, among the known diabetics after comparing them with the age-matched healthy controls, utilizing noninvasive cardiac autonomic neuropathy reflex tests.
Materials and Methods:
A case–control study was conducted for 4 months (October 2014 to January 2015) at an Urban Health and Training Center (UHTC) of a Medical College located in Kancheepuram district, Tamil Nadu. The study was conducted among 126 diagnosed Type 2 diabetes patients and in 152 age- and sex-matched healthy controls to ensure comparability between the cases and controls and, thus, reduce variability due to demographic variables. All the study subjects (cases and controls) were selected from the patients attending UHTC during the study duration, provided they satisfied the inclusion and exclusion criteria. Study participants were subjected to undergo noninvasive cardiac autonomic neuropathy reflex tests. The associations were tested using paired t-test for the continuous (mean ± standard deviation) variables.
Results:
The overall prevalence of cardiac autonomic neuropathy among diabetic patients was found to be as 53.2% (67/126). On further classification, positive (abnormal) results were obtained in 56 (sympathetic – 44.4%) and 51 (parasympathetic – 40.5%) diabetic cases. Overall, heart rate variation during deep breathing was found to be the most sensitive test to detect parasympathetic autonomic neuropathy while the diastolic blood pressure response to sustained handgrip exercise was the most sensitive method to detect sympathetic neuropathy dysfunction.
Conclusion:
The overall prevalence of cardiac autonomic neuropathy among diabetic patients was found to be as 53.2%. Even though cardiac autonomic neuropathy can be detected by various invasive tests, noninvasive tests remain a key tool to detect it in the remote settings in a cost-effective and user-friendly manner without making people visit higher centers.
Keywords: Cardiac autonomic neuropathy, diabetes, India
INTRODUCTION
Diabetes mellitus (DM) is a chronic condition characterized by hyperglycemia precipitating because of the complete (Type 1 DM) or partial (Type 2 DM) absence of the insulin hormone.[1] The recent estimates suggest that worldwide almost 350 million people suffer from diabetes, and these estimates are expected to be doubled by the year 2030 if no active measures are taken.[2,3] The epidemiological studies done across different settings have revealed that a significant increase in both the incidence and onset of disease has been recorded.[4,5,6] India has been labeled as the “diabetes capital” of the world, owing to the share of highest number of people with diabetes.[7,8]
DM has been often associated with a wide range of complications (viz., cardiovascular diseases, nephropathy, retinopathy, and neuropathy), all of which can result in morbidity, disability, and even mortality.[9,10] The autonomic nervous system plays a crucial role in the maintenance of normal body homeostasis.[11] Autonomic neuropathy is one of the least recognized/understood, dreaded and troublesome complications of diabetes, which can cast a serious negative impact on the quality of life of diabetes patients.[12,13] The diabetic autonomic neuropathy tends to affect the functioning of multiple systems-cardiovascular, urogenital, gastrointestinal, pupillomotor, thermoregulatory, and sudomotor-resulting in a significant proportion of the mortality and morbidity associated with the disease.[11,12] Poor glycemic control, long duration of diabetes, increasing age, female gender, and smoking have been identified as the potential risk factors for diabetic autonomic neuropathy and subsequent cardiac autonomic neuropathy.[12,14] Findings of an epidemiological study suggested that almost 50–77% of the diabetic patients had evidence of cardiovascular autonomic neuropathy.[15]
Although symptomatic autonomic neuropathy generally does not result long after the onset of diabetes, nevertheless subclinical autonomic dysfunction can occur within 1 year (Type 2 DM) and within 2 years (Type 1 DM) of initiation of DM.[11] In-fact, conclusive evidence is available to reveal that almost 25–50% of patients with diabetic autonomic neuropathy dies within 5–10 years of diagnosis and is a significant cause for augmenting the direct and indirect medical costs for the patient and their family members.[12]
The present study has been conducted with an objective to estimate the prevalence of cardiac autonomic neuropathy in a rural area of South India, among the known diabetics after comparing them with the age-matched healthy controls, utilizing noninvasive cardiac autonomic neuropathy reflex tests.
MATERIALS AND METHODS
Settings and design
A case–control study was conducted for 4 months (October 2014 to January 2015) at an Urban Health and Training Center (UHTC) of a Medical College located in Kancheepuram district, Tamil Nadu. The study was conducted among 126 diagnosed Type 2 diabetes patients and in 152 age-sex matched healthy controls to ensure comparability between the cases and controls and thus, reduce variability due to demographic variables.
All the healthy individuals (not a known case of diabetes or hypertension or other chronic diseases were selected as controls), in the ratio of 1:1/2 (case:control). For diabetes, the controls were screened using fasting and postprandial blood sugar estimation. For hypertension, the controls were subjected to three readings of blood pressure (BP) measurement, and the lowest one was recorded. For other chronic diseases, self-reported history was considered as the evidence. A total of 19 controls were excluded due to being recently diagnosed with diabetes (n = 8), hypertension (n = 5), and a positive history of coronary heart disease (n = 6). All the study subjects (cases and controls) were selected from the patients attending the UHTC during the study duration, provided they satisfied the inclusion and exclusion criteria.
Inclusion criteria
All Type 2 diabetes patients with history of onset of disease before 1 year (cases) and healthy age- and sex-matched controls attending the UHTC and were willing to participate in the study were enrolled in the study.
Exclusion criteria
All those who were not willing to participate or were suffering from comorbid conditions such as severe chronic obstructive pulmonary disease, acute severe bronchial asthma, severe hypertension, and marked electrocardiogram (ECG) changes suggestive of myocardial ischemia/infarction were excluded from the study.
Methodology
A semi-structured questionnaire was used for eliciting the sociodemographic details of the study participants. Each of the study participants was subjected to thorough clinical examination (pulse rate, BP, screening for medical/surgical complications) and anthropometric examination (measurement of body weight) using standardized procedure with the aid of standard equipment. In addition, routine ECG was conducted to detect any potential cardiac complication so that high-risk subjects can be excluded from the study. Further, study participants were subjected to undergo noninvasive cardiac autonomic neuropathy reflex tests, namely.
Tests for cardiac parasympathetic action:
Valsalva ratio: The participants were instructed to lie on bed and then instructed to blow into a mouthpiece connected to a mercury sphygmomanometer and holding it at a pressure of 40 mmHg for 15 s while continuous ECG was recorded. ECG taken after this maneuver in lead II and V1 is looked, and the ratio of longest R-R interval to shortest R-R interval was calculated (valsalva ratio). Normal valsalva ratio was >1.21, and values <1.2 were considered abnormal.[16]
Heart rate (HR) variation during deep breathing: The patients were asked to sit quietly and take six deep breaths (10 s/breath) for total 1 min. Continuous ECG was recorded throughout the period, and onset of each inspiration and expiration was marked. The maximum and minimum R-R intervals during each breathing cycle were measured and converted to beats/min. Further, the estimates were expressed in terms of mean to indicate difference between maximum and minimum HRs for the six measured cycles in beats/min; (normal response >15 beats/min, borderline 11–14 beats/min; abnormal response <10 beats/min).[16]
Immediate HR response to standing: While performing this test patient lies comfortable in the bed and HR was recorded continuously on an ECG machine. Then, patients were asked to stand up unaided and a marking was done on ECG at the point of starting to stand-up. Subsequently, the shortest R-R interval at or around the 15th beat and largest R-R interval at or around the 30th beat after starting were measured with a ruler. The characteristic HR response was expressed as 30:15 ratio (normal if >1.04; borderline between 1.01 and 1.03; and abnormal if <1.00).[16]
Tests for cardiac sympathetic action:
BP response to sudden standing: The test was performed by measuring the patient's BP while he was lying down quietly and again when he stood up. The postural fall after 2 min in BP was taken as the difference between systolic BP lying and the systolic BP standing (normal response 10 mmHg; borderline 11–29 mmHg; abnormal response > 30 mmHg).[16]
BP response to sustained handgrip: Study subjects were asked to hold an inflated BP cuff using the handgrip of the dominant arm for a few seconds, and it was repeated thrice. Highest of the three readings is called maximum voluntary contraction. Subsequently, participants were instructed to simply maintain handgrip, and results were expressed as the difference between the highest diastolic BP (DBP) during handgrip exercise and the mean of three DBP readings before handgrip began (normal response >16 mmHg; borderline 11–15 mmHg; abnormal <10 mmHg). However, all borderline tests from above mentioned five tests were interpreted as abnormal in the present study.[16]
Ethical considerations
Institutional ethical clearance was obtained before the start of the study. Written informed consent was obtained from the study participants, and utmost care was taken to ensure confidentiality.
Statistical analysis
Data entry and statistical analysis were performed using SPSS version 18 (IBM, America). Frequency distributions and percentages were computed for all the variables. The statistical significance was set at P < 0.05. The associations were tested using paired t-test for the continuous (mean ± standard deviation) variables. Multiple linear regression analysis was used to predict the values of fasting blood sugar.
RESULTS
Table 1 depicts the distribution of study subjects on the basis of their sex. A total of 126 diabetic patients and 152 age-matched controls were included in the study-group after using proper inclusion and exclusion criteria. There was male (case – 62.7%; controls – 55.3%) predominance in contrast to the female participants (case – 37.3%; controls – 44.7%) in both of the groups. Maximum number of patients (45.2%) was reported in the age group 41–60 years, followed by the age group of 61–80 years (43.7%). However, younger patients (0–30 year age group) constituted an insignificant portion (4%) of the total patients in the study.
Table 1.
Sex-wise break-up of “study-subjects”

Table 2 represents the distribution of basic anthropometric and blood sugar levels among the diabetics and age-matched controls. A highly significant difference in mean fetal bovine serum (P < 0.01) and mean Post-prandial blood sugar (PPBS) (P < 0.01) between the two groups was observed. However, no such association was found when the two groups were compared with regard to body weight.
Table 2.
Distribution of basic parameters among diabetic study-subjects and age-matched controls
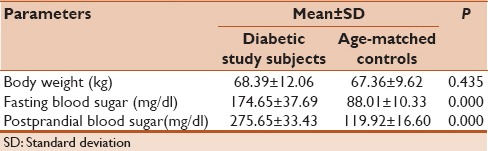
Table 3 depicts the results of different tests performed to assess cardiac para-sympathetic neuropathy dysfunction among the cases and age-matched controls. However, in both the groups the heart response to deep breathing (P > 0.05), immediate HR response to lying followed by ECG monitoring (P > 0.05) and HR response to valsalva maneuver (P > 0.05) were significantly associated with all the different age groups except 0–20 years.
Table 3.
Cardiac autonomic (para-sympathetic) neuropathy dysfunction among cases and age-matched controls
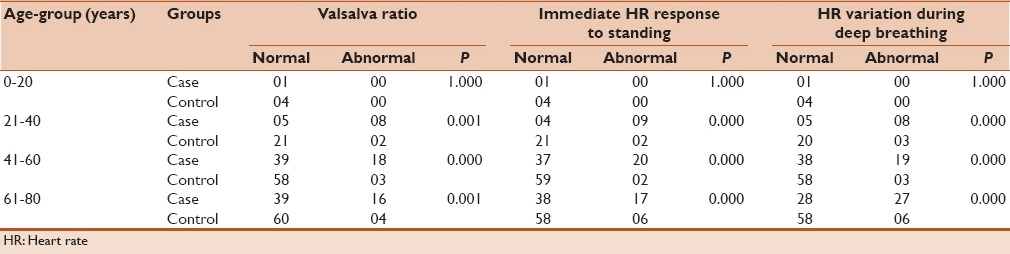
Table 4 depicts the results of different tests performed to assess cardiac sympathetic neuropathy dysfunction among the cases and age-matched controls. It was observed that the BP response to sudden standing (P < 0.05) and DBP response to sustained handgrip was significantly associated with different age groups in both cases and controls.
Table 4.
Cardiac autonomic “sympathetic” neuropathy dysfunction among cases and age-matched controls
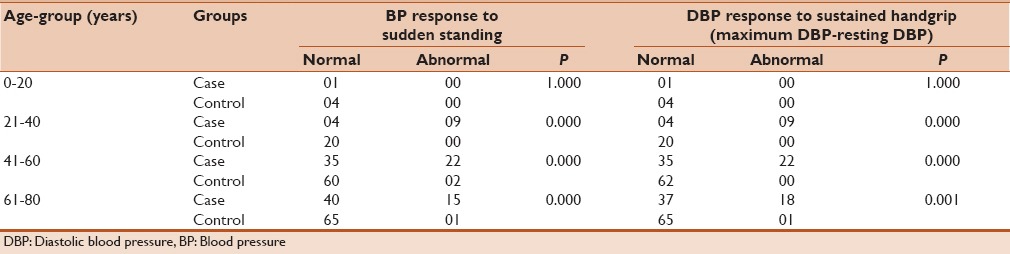
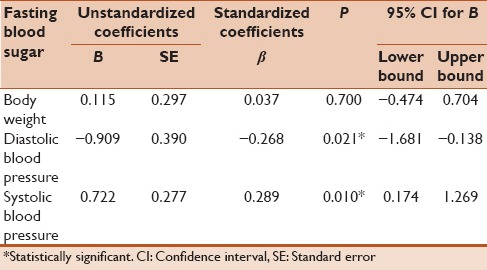
Table 5 represents the results obtained from a multiple linear regression analysis using backward model to predict the estimates of fasting blood sugar from body weight, DBP, and systolic BP. The DBP and systolic BP statistically significantly predicted the values of fasting blood sugar. For each 1 mm increase in DBP, there was a decline in the fasting blood sugar level by 0.909, while for each 1 mm increase in systolic BP, there was an increase in the fasting blood sugar level by 0.722.
Table 5.
Multiple linear regression analyses to predict the fasting blood sugar
DISCUSSION
In this study, the overall prevalence of cardiac autonomic neuropathy among diabetic patients was found to be as 53.2% (67/126). The findings of the study were almost similar with the results obtained from a study conducted in an institutional setting of Eastern India.[16] Another study from the western part of India revealed parasympathetic dysfunction in 58% subjects and sympathetic dysfunction in 20% of the diabetic patients.[17] On further classification, positive (abnormal) results were obtained in 56 (sympathetic – 44.4%) and 51 (parasympathetic – 40.5%) diabetic cases in response to the noninvasive cardiac autonomic neuropathy reflex tests. These findings suggested that sympathetic tests were more sensitive in detecting autonomic dysfunctions than the parasympathetic tests. Contrasting findings were obtained in a hospital-based study from Kolkata.[16]
Overall, HR variation during deep breathing was found to be the most sensitive test to detect parasympathetic autonomic neuropathy. Identical number of parasympathetic dysfunction was detected among the known diabetics using valsalva ratio and on applying immediate HR response to standing. Quite similar results were obtained in another cross-sectional study done with an objective to estimate prevalence of cardiac autonomic neuropathy.[16] Among the controls, the parasympathetic tests were found positive in 12 subjects. Even though none of the three methods were found to be statistically significant, the utility of the noninvasive tests in detecting autonomic dysfunction cannot be undermined.
As far as cardiac autonomic sympathetic neuropathy dysfunction is concerned, the DBP response to sustained handgrip exercise was the most sensitive method by which 49 patients were identified. However, even the BP response to sudden standing was almost identical in detecting cases of sympathetic dysfunction. These findings were in conformity with another study done in the Sindh province of Pakistan.[18]
The strength of the study was its setting as it was conducted in an outreach center where all those people who cannot go to a tertiary institute can avail the services. The study had its limitation also as the sample size was less, and not all noninvasive tests were employed, and no complications were assessed to ascertain the prognosis.
CONCLUSION
The overall prevalence of cardiac autonomic neuropathy among diabetic patients was found to be as 53.2%. Even though cardiac autonomic neuropathy can be detected by various invasive tests, noninvasive tests remain a key tool to detect it in the remote settings in a cost-effective and user-friendly manner without making people visit higher centers.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
REFERENCES
- 1.Geneva: WHO Press; 1999. World Health Organization. Definition, Diagnosis and Classification of Diabetes Mellitus and Its Complications. [Google Scholar]
- 2.Danaei G, Finucane MM, Lu Y, Singh GM, Cowan MJ, Paciorek CJ, et al. National, regional, and global trends in fasting plasma glucose and diabetes prevalence since 1980: Systematic analysis of health examination surveys and epidemiological studies with 370 country-years and 2·7 million participants. Lancet. 2011;378:31–40. doi: 10.1016/S0140-6736(11)60679-X. [DOI] [PubMed] [Google Scholar]
- 3.Mathers CD, Loncar D. Projections of global mortality and burden of disease from 2002 to 2030. PLoS Med. 2006;3:e442. doi: 10.1371/journal.pmed.0030442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kinra S, Bowen LJ, Lyngdoh T, Prabhakaran D, Reddy KS, Ramakrishnan L, et al. Sociodemographic patterning of non-communicable disease risk factors in rural India: A cross sectional study. BMJ. 2010;341:c4974. doi: 10.1136/bmj.c4974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Chuang LM, Tsai ST, Huang BY, Tai TY Diabcare-Asia Study Group. The status of diabetes control in Asia – A cross-sectional survey of 24 317 patients with diabetes mellitus in 1998. Diabet Med. 2002;19:978–85. doi: 10.1046/j.1464-5491.2002.00833.x. [DOI] [PubMed] [Google Scholar]
- 6.Narayanappa D, Rajani HS, Mahendrappa KB, Prabhakar AK. Prevalence of prediabetes in school-going children. Indian Pediatr. 2011;48:295–9. doi: 10.1007/s13312-011-0061-6. [DOI] [PubMed] [Google Scholar]
- 7.Geneva: WHO Press; 2011. World Health Organization. Global Status Report on Non-communicable Diseases-2010. [Google Scholar]
- 8.Wild S, Roglic G, Green A, Sicree R, King H. Global prevalence of diabetes: Estimates for the year 2000 and projections for 2030. Diabetes Care. 2004;27:1047–53. doi: 10.2337/diacare.27.5.1047. [DOI] [PubMed] [Google Scholar]
- 9.American Diabetes Association. Implications of the United Kingdom prospective diabetes study. Diabetes Care. 2004;27:28–32. doi: 10.2337/diacare.21.12.2180. [DOI] [PubMed] [Google Scholar]
- 10.Zucchi P, Ferrari P, Spina ML. Diabetic foot: From diagnosis to therapy. G Ital Nefrol. 2005;22(Suppl 31):S20–2. [PubMed] [Google Scholar]
- 11.Jeyaraman K. Autonomic neuropathy. In: Thomas N, editor. A Practical Guide to Diabetes Mellitus. 6th ed. Vellor: Jaypee; 2012. pp. 234–9. [Google Scholar]
- 12.Rao MS. Noncoronary cardiac complications in diabetes. RSSDI Textbook of Diabetes Mellitus. In: Tripathy BB, editor. 2nd ed. New Delhi: Jaypee; 2012. pp. 798–801. [Google Scholar]
- 13.Freeman R. Diabetic autonomic neuropathy. Handb Clin Neurol. 2014;126:63–79. doi: 10.1016/B978-0-444-53480-4.00006-0. [DOI] [PubMed] [Google Scholar]
- 14.Maser RE, Lenhard MJ. Cardiovascular autonomic neuropathy due to diabetes mellitus: Clinical manifestations, consequences, and treatment. J Clin Endocrinol Metab. 2005;90:5896–903. doi: 10.1210/jc.2005-0754. [DOI] [PubMed] [Google Scholar]
- 15.Muhopadyaya J, Ray S. Cardiac autonomic neuropathy in diabetes. Med Update (API) 2004;14:35–8. [Google Scholar]
- 16.Basu AK, Bandyopadhyay R, Chakrabarti S, Paul R, Santra S. A study on the prevalence of cardiac autonomic neuropathy in type-2 diabetes in Eastern India. J Indian Acad Clin Med. 2010;11:190–4. [Google Scholar]
- 17.Mehta S, Mathur D, Chaturvedi M, Verma K. Incidence of cardiac autonomic neuropathy and its correlation with retinopathy, micro-albuminuria and glycated haemoglobin in non-insulin dependent diabetes mellitus. J Indian Med Assoc. 2002;100:141-3–152. [PubMed] [Google Scholar]
- 18.Khoharo HK, Qureshi F. Frequency of cardiac autonomic neuropathy in patients with type 2 diabetes mellitus reporting at a teaching hospital of Sindh. J Coll Physicians Surg Pak. 2008;18:751–4. [PubMed] [Google Scholar]


