Abstract
Cytokine-induced killer (CIK) cells are T cell derived ex vivo expanded cells with both NK and T cell properties. They exhibit potent anti-tumor efficacy against various malignancies in preclinical models and have proven safe and effective in clinical studies. We combined CIK cell adoptive immunotherapy with IL-12 cytokine immunotherapy in an immunocompetent preclinical breast cancer model. Combining CIK cells with IL-12 increased anti-tumor efficacy in vivo compared to either therapy alone. Combination led to full tumor remission and long-term protection in 75% of animals. IL-12 treatment sharply increased the anti-tumor efficacy of short-term cultured CIK cells that exhibited no therapeutic effect alone. Bioluminescence imaging based in vitro cytotoxicity and in vivo homing assays revealed that short-term cultured CIK cells exhibit full cytotoxicity in vitro, but display different tumor homing properties than fully expanded CIK cells in vivo. Our data suggest that short-term cultured CIK cells can be “educated” in vivo, producing fully expanded CIK cells upon IL-12 administration with anti-tumor efficacy in a mouse model. Our findings demonstrate the potential to improve current CIK cell-based immunotherapy by increasing efficacy and shortening ex vivo expansion time. This holds promise for a highly efficacious cancer therapy utilizing synergistic effects of cytokine and cellular immunotherapy.
Electronic supplementary material
The online version of this article (doi:10.1007/s00262-010-0860-y) contains supplementary material, which is available to authorized users.
Keywords: Cytokine-induced killer cells, IL12, In vivo imaging, Bioluminescence imaging
Introduction
Several adoptive cell transfer regimens have emerged as potential therapeutic options to treat a wide range of malignancies. Since the initial promise of lymphokine-activated killer cells (LAK), and tumor-infiltrating lymphocytes (TIL), cytokine-induced killer cells (CIK) have emerged as a foundation of a more potent cellular therapy and have demonstrated safety and efficacy in clinical trials [12, 20, 25, 29–34, 42]. CIK cells can be derived from peripheral blood mononuclear cells from healthy donors and cancer patients as well as cord blood [1, 18]. They exhibit a stronger anti-tumor effect than LAK cells in vivo and overcome the problem of TILs where recovery of sufficient numbers of cells for therapeutic use is difficult. After ex vivo expansion with IFN-gamma, anti-CD3, and IL-2, the enriched human CIK population exhibits NK-like T cell functions and markers (CD3+ CD56+ in humans, CD3+ DX5+ in mice), and these markers appear to correlate with cytotoxicity. The cytokines used in ex vivo expansion of CIK cells have long been considered as therapeutic options themselves. Particularly IL-12 was long thought to be the most promising anti-tumor cytokine due to its broad and potent anti-tumor effects in preclinical models [9, 10]. It was shown that for ex vivo expansion of CIK cells IL-2 or IL-12 show similar utility, while having different physiological roles [43, 44].
IL-12 stimulates a TH1 immune response and presentation of tumor antigens through upregulation of MHC molecules. It moreover facilitates IFN-gamma production and was shown to be anti-angiogenic. However, the clinical use of IL-12 was hampered by toxicities associated with cytokine delivery in humans and limited efficacy at tolerable doses [2, 3, 8, 28, 40]. While alternative strategies of local IL-12 delivery or expression have been pursued [17, 21, 24, 40], it has been suggested that IL-12 may be most effective in combination therapy where lower doses can be applied. Thus, efforts have been reported to combine IL-12 therapy with chemotherapy, vaccination strategies and additional cytokines [11, 16, 19, 36, 41]. Similarly, cytokine therapy may offer benefits to CIK cell therapy. This combination may serve to reduce the time required for CIK expansion, and to enhance the CIK cell activity. The ex vivo expansion protocol for CIK cells is both labor- and cost-intensive, and is a concern for routine clinical application of CIK cell-based therapies. Moreover, combination therapies offer the possibility of overcoming the problems of cancer heterogeneity and resistance to therapy. This has been shown for CIK cells in combination with oncolytic viruses where the CIK cells were effectively used to deliver virus particles into the tumor site, exploiting the tumor specific homing abilities of CIK cells for intratumoral delivery [35].
Here, we describe a novel approach of combining IL-12 cytokine therapy with CIK based adoptive immunotherapy, and demonstrate, in living animals, that this combination results in a synergistic, anti-tumor effect, and that there are differences in CIK cell homing properties in the presence or absence of IL-12. Short-term CIK (stCIK) cell cultures, and fully expanded CIK cell cultures were tested and the results suggested that in vivo delivery of the cytokines could be used for in vivo CIK expansion. We determined that the stCIK cultures have different activities from those described for CIK cells, and these differences may complement activity of CIK cells.
Materials and methods
Animals
FVB/N mice were obtained from Charles River Laboratories (Wilmington, MA, USA). Generation of the luciferase transgenic mouse line (FVB.luc+) was described previously [6, 7]. All mice used were littermates or age-matched (6–12 weeks of age), provided access to water and food ad libitum, and housed in the Research Animal Facility at Stanford University. All procedures were approved by the Animal Care and Use Committee of Stanford University.
Construction of retroviral vector
Previously described L2G expression cassette [6, 7] was inserted into the EcoRI site of pMSCVpuro vector (Clontech, Palo Alto, CA, USA), the resulting construct was named pMSCVpuro.L2G and used to generate amphotropic retrovirus with help of the Phoenix system (kind gift of G. Nolan, Stanford).
Cell isolation and culture
DB7 tumor cells were originally derived from FVB/NTg(MMTV-PyVmT Y315F/Y322F) mice and are described elsewhere [5]. To increase tumor take, the cells were passaged through FVB/N mice before implantation. For imaging purposes DB7 cells were transduced with a retroviral vector expressing a dual function reporter gene (L2G) comprised of firefly luciferase (fLuc) and green fluorescent protein (GFP) driven by a hybrid promoter consisting of the beta-Actin promoter and the CMV enhancer (CAGS). Transduced cells were screened for fLuc expression with an IVIS 50 system (Caliper Life Sciences, Hopkinton, MA, USA) and 25 positive clones selected and combined to obtain a population representative of the parental population (DB7luc+). CIK cells were obtained by ex vivo expansion of splenocytes derived from donor mice as described in detail previously [4]. Briefly, splenocytes were treated with IFN-γ (R&D Systems, Minneapolis, MN, USA) for 24 h before transfer to a flask coated with anti-CD3 antibody (clone 145-2C11, BD Biosciences, San Jose, CA, USA) and addition of IL-2 (Proleukin, Novartis, East Hanover, NJ, USA). Fresh medium was added every 2–3 days and cells maintained at a density of >106/mL for 6 (stCIK) and 14 days (CIK), respectively.
Anti-tumor efficacy of CIK and IL-12 in vivo
To study the anti-tumor efficacy of CIK cells and IL-12, FVB/N mice were inoculated with 2.5 × 106 DB7luc+ cells subcutaneously. Tumors were allowed to establish for 2 weeks before 2 × 5 × 106 CIK cells derived from FVB/N donor mice were injected intravenously (IV) into the tail vein.
Starting 1 day prior to CIK cell injection tumors were measured by caliper measurement and in vivo bioluminescence imaging. Subsequent measurements were carried out daily for the first week and twice weekly thereafter. To compare tumor growth and remission in response to IL-12, the mice were given a daily bolus of murine IL-12 [200 ng (full dose) or 20 ng (1/10) in 500 μL PBS supplemented with 1% mouse serum] (Peprotech) for 5 days, or five mock daily intraperitoneal (IP) injections; these treatments start on the day of CIK cell injection. Mice whose tumors were no longer palpable and could not be detected by bioluminescence imaging were classified as rescued and monitored for relapse for 4 months. After this time period, the rescued mice were re-challenged by injection of DB7luc+ tumor cells into the flank opposite to the original site of inoculation and monitored by in vivo bioluminescence imaging.
Survival data was statistically evaluated by log-rank analysis. Differences in tumor sizes were compared by Student’s t test. Full results of statistical analysis can be found in supplemental data.
For imaging, the mice were injected with luciferin [potassium salt, IP at a dose of 150 μg/kg] 10 min prior to image acquisition on an IVIS SPECTRUM (Caliper) for 1 min. Analysis of signal intensity was performed using Living Image software (Caliper).
Flow cytometry
Cells were pelleted (300×g, 5 min) and suspended in Dulbecco’s PBS containing 1% FBS (FACS buffer). The cell suspensions were placed on ice and treated with FITC-α-mouse CD3 (clone 145-2C11, BD Pharmingen), PE-α-mouse CD49b (clone DX5, BD Biosciences), or PE-α-mouse NKG2D/CD314 (clone CX5, eBioscience). Separate samples were stained with FITC-conjugated hamster IgG1 κ, PE-conjugated rat IgM, or PE-conjugated rat IgG1, κ isotype control antibodies to quantitate non-specific antibody binding. All samples were stained with 1 μg of antibody per 106 cells and incubated on ice for 30 min. The cells were then pelleted, rinsed with FACS buffer, and analyzed on a BD FACSCalibur flow cytometer equipped with a 488-nm argon laser. Data analysis was performed using FlowJo software (Tree Star, Ashland, OR, USA). At least 10,000 cells were analyzed for each sample, and cell viability was ascertained by gating the samples on the basis of forward scatter (to sort by size) and side scatter (to sort by granularity).
In vitro cytotoxicity assay
Cytotoxicity assays were performed by incubation of CIK cells with fLuc expressing tumor cells and monitoring luminescence intensity from surviving cells [35]. The cytotoxic effect is reflected by a loss of fLuc activity as the expressing tumor cells are killed. DB7luc+ target cells (104 cells/well) were plated in 96-well plates. Effector cells were added at different effector-to-target ratios and medium added to total 200 μL. All ratios of cells (including control wells with tumor only or tumor only pretreated with 1% Triton X-100) were plated in triplicate and incubated for 6 h in a tissue culture incubator. Luciferin (2 μL, 30 mg/ml, Caliper) was then added to each well and light output (photons/s/well) measured on an IVIS 50 imaging system (Caliper). All images were analyzed with LivingImage analysis software (Caliper) and percent survival calculated relative to control wells.
In vivo imaging of CIK cell homing and proliferation
To monitor CIK cell proliferation and localization, FVB/N mice were inoculated with 2.5 × 106 DB7 cells subcutaneously. Tumors were allowed to establish for 2 weeks before 2 × 5 × 106 CIK cells derived from FVB.luc+ donor mice were injected IV. For imaging, the mice were injected with luciferin and imaged as described above for 5 min; dorsal and ventral views were obtained. Analysis of signal intensity was performed using Living Image software (Caliper). For quantitative comparison of tumor homing dynamics the light emission [photons/s/region of interest (ROI)] was measured for the tumor area and an ROI of the same size in the neck area. The latter was subtracted from the tumor signal for plotting purposes. To compare homing and proliferation in response to IL-12, mice were given daily doses of mIL-12 [200 ng in 500 μL in PBS supplemented with 1% mouse serum] (Peprotech, Rocky Hill, NJ, USA), or a mock IP injection, for a period of 5 day starting on the day of CIK injection.
Results
CIK cell–IL-12 combination therapy leads to complete tumor remission in 75% of treated animals
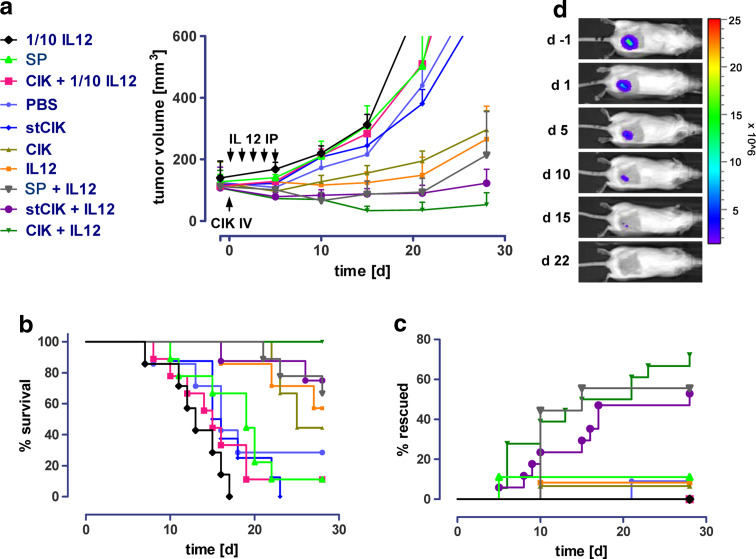
We compared the anti-tumor efficacy of CIK cells that were cultured for the full term of enrichment (14 days) to CIK cells from short-term cultures (stCIK, enrichment for 6 days) and to freshly isolated splenocytes, both alone and in combination with IL-12. The highest overall efficacy was achieved after administration of a combination of CIK cells with 200 ng of IL-12 for 5 days, as assessed by tumor regression, animal survival and percentage of mice with complete remission (Fig. 1a–d). Thirty days post-treatment, none of the mice treated with the IL-12 and CIK combination, exhibited a tumor size of more than 300 mm3. For comparison, only 70% of animals receiving stCIK cells or splenocytes in combination with full dose IL-12 remained under 300 mm3. Mice treated with low dose IL-12, or splenocytes and stCIK cells alone appeared to have a slightly accelerated tumor growth as compared to the PBS treated control group (Fig. 1a, b).
Fig. 1.
Anti-tumor efficacy of IL-12 and CIK cell therapies and their combinations in vivo. Mice were inoculated with DB7luc+ tumor cells subcutaneously. After tumors had established, CIK cells of different age were injected intravenously (IV). Tumors were measured by caliper measurement and in vivo bioluminescence imaging. To compare tumor growth and remission in response to IL-12, the mice were given a daily bolus of murine IL-12 [200 ng (full dose) or 20 ng (1/10)] or mock injections for 5 days (IP) starting on the day of CIK cell injection. The upper left legend applies to a–c SC splenocytes, CIK cytokine-induced killer cells (full 14 days culture), stCIK short-term CIK cells (6 days culture). a Tumor growth as determined by caliper measurement in response to treatments. Error bars represent standard error of the mean (SEM), b Kaplan–Meier plots of animals receiving different treatments. Tumor volume of 300 mm3 was used as a surrogate endpoint. c Inversed Kaplan–Meier plots of animals receiving different treatments. Tumor remission was followed by BLI and animals with no detectable tumor were classified as rescued (cf. d, where color bar represents light emission as photons/s/cm2/steradian. Bar maximum is 1.5 × 106 and bar minimum is 2.5 × 107). Rescued animals were monitored for several months and relapse was never observed. Group sizes (N) were: PBS (7); IL12 (7); CIK (9); CIK + IL12 (9); stCIK (8); stCIK + IL12 (8); CIK + 10% IL12 (9); SP (9); SP + IL12 (9); 10% IL12 (7)
Furthermore, the combination of CIK cells and a full dose of IL-12 led to complete tumor remission in 75% of the mice treated compared to a 55% remission rate after combining IL-12 with stCIK cells or splenocytes. Neither one of the cell preparations nor IL-12 alone led to full remission in more then 10% of the mice tested, matching the controls (Fig. 1c, d). Mice with full tumor remission were followed for 4 months, and no relapses observed. Moreover, re-challenging these mice by injection of a second dose of the original inoculum of tumor cells did not lead to tumor formation and 3 weeks after the injection bioluminescence signals had disappeared completely (Data not shown), indicating the mice had developed immunological memory.
IL-12 treatment promotes anti-tumor efficacy of stCIK populations
While CIK cells in combination with full dose IL-12 exhibited the highest anti-tumor efficacy by multiple criteria (Fig. 1), it was striking that the administration of IL-12 to mice that received stCIK cells led to efficacy that was well beyond that observed for IL-12 alone. Since the stCIK cells exhibited no efficacy in vivo when administered alone, the data indicates a synergistic effect of the two therapies beyond a mere additive effect of IL-12 and CIK cells. The stCIK cells therefore benefited more from the combination with IL-12 than CIK cells, suggesting that an in vivo expansion could be achieved.
Characteristics of CIK cell populations
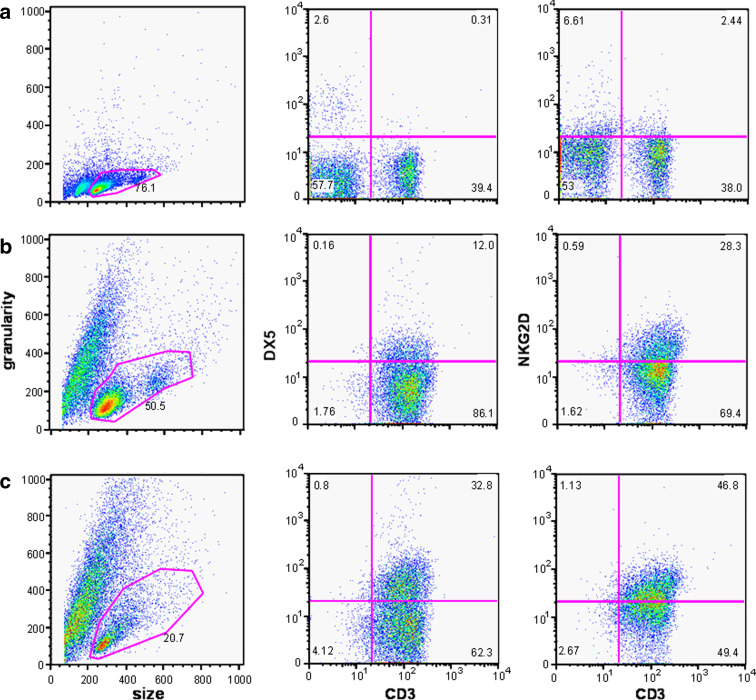
Flow cytometry analyses of fresh splenocytes, stCIK cells (day 6 of the ex vivo expansion) and CIK cells (day 14 of the ex vivo expansion) revealed a maturation gradation. Granularity and size distribution of the cells (Fig. 3a–c) indicated decreasing numbers of viable cells (pink region of interest). Viable cells in full term CIK cells demonstrate strongly decreased proliferation (Suppl. Fig. 1). Furthermore, compared to splenocytes and stCIK cells, no large numbers of dividing cells were present in the fully enriched cultures of CIK cells. While freshly isolated splenocytes were approximately 55% CD3+, the CIK cell populations were >95% CD3+ after 6 days. The percentages of CD3+/DX5+ and CD3+/NKG2D+ cells increased from <1 and 2.5%, respectively, in splenocytes to 12 and 28% after 6 days of enrichment and eventually reached 33 and 47% in the fully enriched CIK population after 14 days (Fig. 3a–c, Suppl. Fig. 2).
Fig. 3.
Flow cytometry analysis revealed phenotypic differences between CIK cell populations of different ages. CD3, DX5 and NKG2D surface expression was analyzed by flow cytometry. At least 10,000 cells were analyzed for each sample, and cell viability was ascertained by gating the samples on the basis of forward scatter (to sort by size) and side scatter (to sort by granularity). a Freshly isolated splenocytes. b Short-term (day 6) CIK population. c CIK cells at day 14
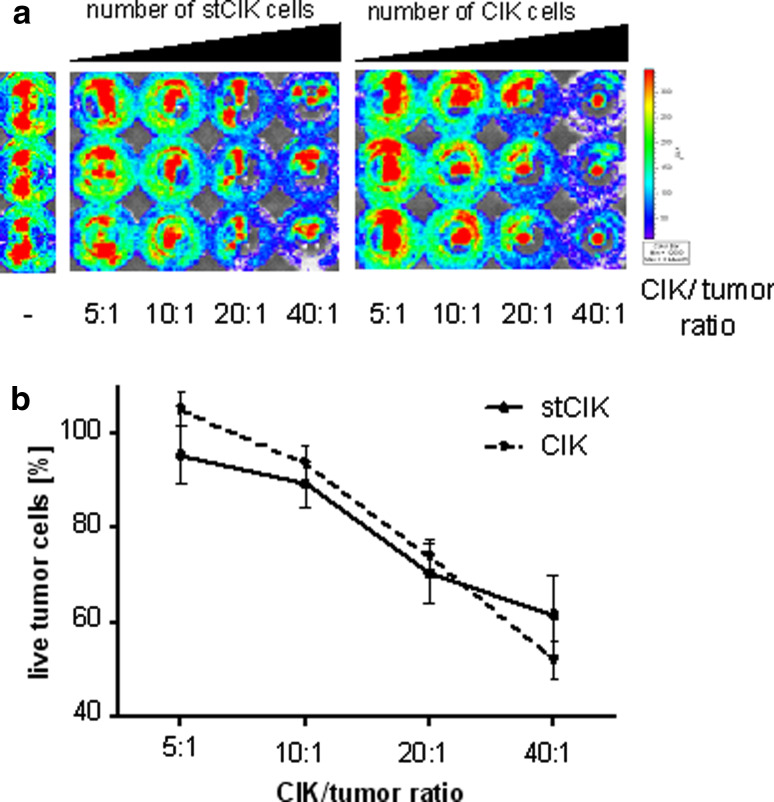
stCIK cell populations display full in vitro cytotoxicity after only 6 days of ex vivo expansion
stCIK cells did not exhibit notable anti-tumor efficacy in vivo (Fig. 1). We were therefore interested in whether this was due to lacking cytotoxicity or tumor homing abilities. To test this, we set up an in vitro cytotoxicity assay, where no homing is necessary to gain close contact to the tumor cells. In this assay both populations of cells exhibited dose dependent killing of fLuc labeled DB7 cells reaching about 40% after 16 h at a CIK:tumor cell ratio of 40:1. This assay demonstrated that the tumor cell killing ability of stCIK cells matched that of CIK cells as soon as cell–cell contact is established (Fig. 4). This suggests that different tumor homing abilities play a role for the different anti-tumor efficacy of stCIK compared to CIK cells in vivo.
Fig. 4.
Comparison of cytotoxicity of CIK populations of different maturity by luminescence based in vitro cytotoxicity assay. The cytotoxic effect results in a loss of fLuc activity as the expressing tumor cells are killed. Percent survival was calculated relative to control wells. a False color bioluminescence readout reflecting the number of live tumor cells after 6 h incubation with respective doses of CIK cells in a representative experiment. b Graphical representation of data obtained from the experiment in Fig. 4a. Control wells are shown on far left. Error bars represent standard deviation of replicates
Homing of stCIK cell populations to the tumor site is delayed and not exclusive
In order to gain insight into the behavior of CIK cells that exhibit different anti-tumor efficacies in vivo, but not in vitro we applied a modified experimental in vivo setup compared to the efficacy study. In these experiments, mice were inoculated with parental DB7 tumor cells instead of DB7luc+. CIK cells were expanded from a fLuc expressing donor mouse providing us with a visible tag that enabled noninvasive studies of homing and proliferation of these cells after transfer to tumor-bearing mice.
After injection of labeled CIK cells, we observed a specific accumulation of cells at the tumor site 1–2 days after injection (Fig. 2). This signal decreased to background level by day 4. By contrast, after injection of stCIK into mice, the tumor-associated signal was observed about 1 day after that of the CIK population and the signals from mice that received stCIK cells had up to four times the intensity of that from CIK cells at the tumor site. In addition, we observed other sites of homing and proliferation after injection of stCIK cells. The strongest signal, beside the tumor site, was seen in a region corresponding to the spleen. None of the control experiments showed an increased signal from either the tumor or other organ sites.
Fig. 2.
Tumor homing and expansion of stCIK cells compared to CIK cells in vivo as assessed by in vivo bioluminescence imaging (BLI) over a 5-day period after cell injection. Light emission was measured at the shaved tumor site and normalized by subtracting a background signal from a reference area in the neck area. Selected BLI images are shown for illustration purposes for day 2 and day 4. Error bars represent standard deviation
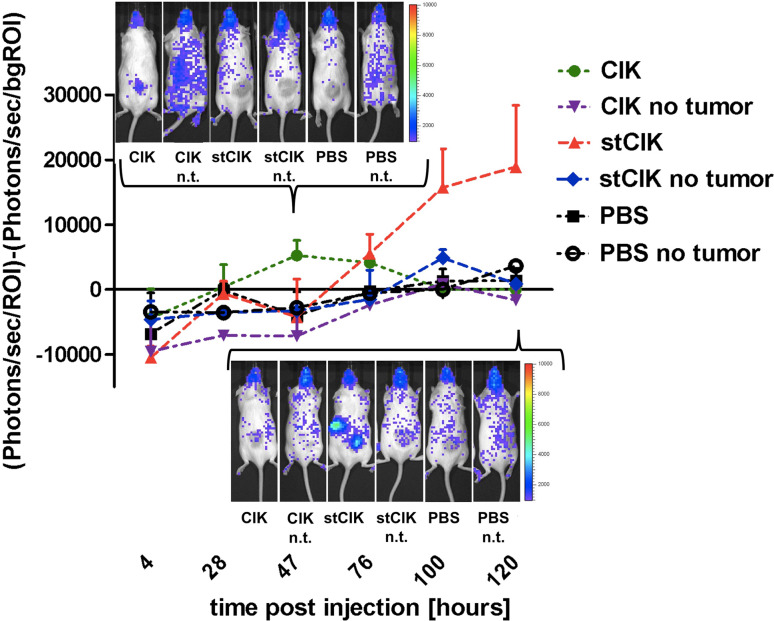
IL-12 treatment enhances stCIK cell proliferation and persistence at the tumor site, in spleen and lymph nodes in vivo
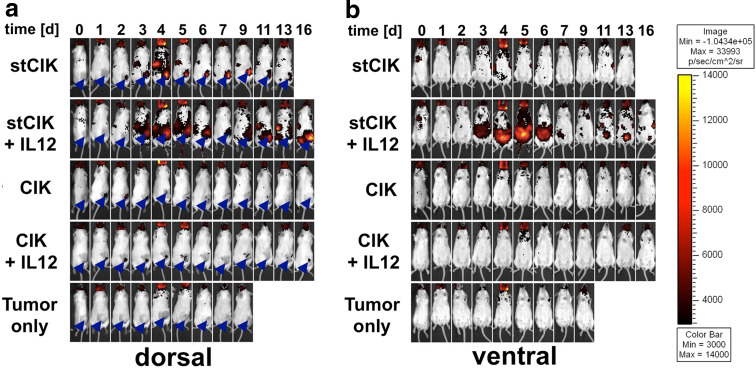
Additional experiments demonstrated that intraperitoneally injected IL-12 did not drastically alter CIK cell homing or proliferation (Fig. 5). For example, the ventral view (Fig. 5b) did not show any specific accumulation of labeled cells while the dorsal view showed tumor-associated homing not unlike that of CIK cells that did not receive IL-12. In the latter case the fLuc signals were visible at the tumor site 1 day post injection and disappeared after 5–6 days. A faint signal seemed to persist at these sites in mice that received IL-12 (Fig. 5a).
Fig. 5.
Tumor homing and proliferation of fLuc expressing CIK cell populations of different ages in vivo in response to IL-12. After tumors had established, CIK cells were injected intravenously (IV). Mice were given daily doses of mIL-12 or a mock IP injection for a period of 5 day starting on the day of CIK injection. Each group consisted of four animals. Images from the same representative animals are shown over the time course for dorsal and ventral views. Arrow heads indicate tumor locations. a Dorsal view highlighting differences in cell homing between stCIK and full term CIK cell populations. b Ventral view highlighting differences in proliferation capacity between stCIK and full term CIK cell populations
Sharp differences were observed in mice that received a combination of IL-12 with stCIK cells compared to mice that received cells only. While in both cases there was a delay in signal at the tumor site compared to CIK cells (1 day later without, and 2 day later with IL-12; Fig. 5a), this signal was stronger and more persistent with IL-12. Furthermore, signal from lymph nodes and spleen appeared similarly stronger and prolonged. The most obvious effect of IL-12 on stCIK populations in vivo however was apparent in the ventral view (Fig. 5b): Starting on day 3 a strong signal indicative of cell proliferation was observed in the abdominal region. This signal was only apparent in mice that received IL-12 and appeared in close proximity to the intraperitoneal injection site. The signal peaked on day 5 and was not detectable after day 7. Similarly to the dorsal view we observed signals from presumed lymph nodes that appeared around day 9. Based on theses observations IL-12 influences the homing and expansion patterns of stCIK cells in vivo.
Discussion
CIK cells alone and in combination with other therapies have proven both useful and safe [20, 32, 35, 42]. IL-12 showed great initial promise in preclinical models but exhibited toxicity problems in humans [2, 3, 8–10, 28, 40]. We combined IL-12-based cytokine therapy with CIK cell therapy to achieve synergistic effects that increase the anti-tumor efficacy of CIK cells and enable the use of low doses of IL-12. In patients, this combination therapy might potentially overcome the issue of high dose toxicity of IL-12. We furthermore aimed to shorten the ex vivo expansion process for CIK cells by in vivo education and expansion of CIK cells to save time and cost for the clinical translation of CIK cell therapy. We therefore tested this combination therapy in an immunocompetent mouse model using subcutaneously implanted mammary carcinoma cells (DB7) engineered to express firefly luciferase (fLuc) and CIK cells expanded from syngeneic FVB donor mice. In this model we tested the anti-tumor efficacy of splenocytes and CIK cells after different lengths of enrichment time, both with and without IP delivered IL-12. We further examined whether we could lower the concentration of IL-12 in the combination therapy with the goal of clinical translatability. We followed the average tumor size in mice of the same groups but also tracked individual responses using Kaplan–Meier analysis and inverse Kaplan–Meier analysis (Fig. 1).
Our studies demonstrated that a combination of IL-12 with CIK cells outperformed any other tested treatment regimen including IL-12 and CIK as single therapies in terms of tumor regression, complete remissions, and animal survival rates. Our data do not conclusively demonstrate whether this effect is additive or synergistic. However, the two treatments alone did not lead to full remission in more mice than the control treatment, while the combination therapy rescued almost 75% of the animals pointing to a synergistic effect. We observed a clearly synergistic effect when combining IL-12 with either splenocytes or stCIK cells: neither of the latter alone showed any anti-tumor effect, while the combination provided an enhanced anti-tumor effect beyond that of IL-12 only. While we expected this synergy for stCIK cells, the results were surprising for the combination of IL-12 and splenocytes which was only slightly lower (within the margin of error) than that of stCIK cells plus IL-12. This can probably be attributed to the broad immunostimulatory effects of IL-12 including IFN-γ release, increased cytotoxicity and T cell proliferation. However, it is unlikely that this observation from the animal model could be exploited in a clinical setting using freshly isolated PBMCs because a substantial number of cells needed to be collected from a patient prior to cell transfer.
CIK cell populations undergo a maturation process during ex vivo expansion, which is evident in the flow cytometry data presented in Fig. 3 and was studied earlier [4, 38]. During this process, a heterogeneous population of splenocytes develops into a more homogenous CIK population with the peak of anti-tumor activity at about 14 days of enrichment. Despite the apparent enrichment of CD3+ DX5+ and NKG2D+ cells early in this process (Fig. 3) coinciding with a cytotoxic effect in the co-culture assay (Fig. 4), there was no measurable anti-tumor effect observed in mice that received stCIK cells (Fig. 1). Later in the process (days 6–14) the amount of viable cells decreases from 50% to about 20% implying that stCIK cells exhibit considerably more proliferation compared to more mature CIK cell cultures. This was confirmed by the proliferative capacity of these cells observed in vivo (Fig. 5), and is probably crucial for the increased proliferative response and efficacy that we observed after delivery of IL-12 to mice that received stCIK injections. We were also interested to find out why stCIK cells showed no anti-tumor efficacy in vivo yet. Generally, two important functional aspects underlie anti-tumor potential in vivo: first, the ability of CIK cells to kill a tumor cell upon direct contact, as measured by an in vitro cytotoxicity assay; second, the ability of CIK cells to home to the tumor site to exert their cytotoxic effect. We therefore assayed the cytotoxic potential of stCIK and CIK cells in co-culture with tumor cells and reproducibly noted similar tumor cell killing abilities for the two cell types (Fig. 4). To assess the tumor homing of CIK cells we used CIK cells derived from transgenic donor mice expressing fLuc (CIKluc+). These cells could be followed in vivo over a period of several days via bioluminescence imaging [13, 26]. These in vivo homing experiments showed a trafficking and proliferation pattern of stCIK cells that is markedly distinct from CIK cells. The stCIK cells still accumulated at the tumor site, but with a delay (Fig. 2). These observations indicate that stCIK populations contain heterogeneous subpopulations with separated tumor homing, cytotoxic and proliferative properties, while mature cells appear more homogenous with respect to trafficking and cytotoxic abilities. In terms of the propagation of CIK cell cultures, it thus appears that the second week of ex vivo expansion and cytokine stimulation is crucial for differentiation and/or proliferation of the CIK effector population which can both home to tumor sites and kill cancer cells.
Because we observed increased anti-tumor efficacy in mice that received a combination of IL-12 and stCIK cells, we were interested whether this is associated with CIK proliferation or a different homing pattern in vivo. CIK cells did not show an altered proliferation and homing pattern when combined with IL-12 injections, while stCIK cells reacted strongly to IL-12. Most notably, we observed a strong proliferation of stCIK cells in the peritoneal cavity, a 24 h delayed arrival of cells at the tumor site, and longer persistence at the tumor site, spleen and lymph nodes. These data indicate that IL-12 is able to increase the proliferative capacity of stCIK cells in vivo compared to unstimulated stCIK cells, which is also supported by the stimulating effect of IL12 in vitro (Suppl. Fig. 1). Recognizing that stCIK cells lack anti-tumor efficacy in vivo and that the combination of IL-12 and stCIK leads to vastly increased efficacy compared to IL-12 alone, we conclude that our strategy of an education of CIK cells in vivo is viable. It seems to result in an effector cell population that combines tumor homing with cytotoxic capacities. However, it is clear from in vivo imaging of stCIK cells with and without IL-12 stimulation that abundance of cells in the mouse and particularly at the tumor site does not generally correlate with anti-tumor efficacy. In this context, it is striking that we observed a delayed arrival or expansion of cells from stCIK populations after IL-12 stimulation. A possible explanation is that those cells differentiate elsewhere after IL-12 stimulation and get to the tumor site later in a more mature state with cytotoxic abilities.
The exact phenotype and role of different cell types in the enriched CIK population remains to be fully described. In humans it was demonstrated that CD3+ CD56+ double positive populations (corresponding to CD3+ DX5+ in mice) are associated with anti-tumor efficacy in vivo [4, 38]. It has been reported that the majority of these cells in CIK populations are not derived from the initial CD3+ CD56+ subpopulation, which actually expands poorly under CIK culture conditions, but rather from CD4+ CD8+ T cells which acquire CD3+ CD56+ expression only after activation [22]. Terminally differentiated effector CD3+ CD56+ T cell populations are known to possess potent cytotoxicity but have low proliferative capacity [27, 39]. On the other hand, CD3+ CD56- cells, representing early effector T cells, exhibit reduced cytotoxicity but a higher capacity to proliferate, home to lymph nodes and persist in vivo, which is what we observed after injection of stCIK cells (Figs. 2, 5) [14]. Based on these findings, an effective strategy may be to inject combinations of CIK and stCIK cells in combination with IL-12 in order to achieve a longer lasting and thus more robust anti-tumor effect. Additional studies are needed to further characterize surface marker expression and functionality of the one, or several, effector cell types in CIK populations. For the work presented here, it seems of particular relevance to characterize the CIK cell subpopulation that possess cytotoxic and tumor homing abilities, along with the subpopulation of stCIK cells that gives rise to these cells in vitro and in vivo after IL-12 stimulation. Comparison of immune cells recovered from the tumor site and peritoneal cavity after IL-12 stimulation may help refine the cell surface markers that define the effector population. While it has been shown that the cytotoxicity of CIK cells is largely mediated by recognition of stress-related ligands through the NKG2D receptor and is dependent on perforin, less details are known about the mechanisms and molecules involved in tumor homing [37, 38]. Several chemokine receptors and selectins have been shown to play a role in homing of CIK cells to the tumor site [23, 26]. Moreover, the secretion of chemokines to attract more immune-effector cells like granulocytes, monocytes, and NK-cells could be a mechanism of action of CIK cells beside direct interaction and lysis of tumor targets [15, 23].
In summary, we report that combination of IL-12 with CIK cells led to a strongly increased anti-tumor efficacy in a mouse model compared to either therapy alone and holds promise for a highly efficacious cancer therapy utilizing synergistic effects. Even otherwise ineffective stCIK cells benefited from the IL-12 treatment and exhibited strong anti-tumor effects. The data presented here indicate that the combination of adoptive cellular immunotherapy with cytokine therapy can be exploited to shorten ex vivo cultivation procedures through education and expansion of therapeutically active cells in vivo. In the future, the addition of IL-2 to this combination or engineering CIK cells to express their cytokines (as attempted for IL-2 [32]) might hold promise for further increased efficacy and utility since it could allow a reduction of the doses of the individual cytokines.
Electronic supplementary material
Below is the link to the electronic supplementary material.
References
- 1.Alvarnas JC, Linn YC, Hope EG, Negrin RS. Expansion of cytotoxic CD3+ CD56+ cells from peripheral blood progenitor cells of patients undergoing autologous hematopoietic cell transplantation. Biol Blood Marrow Transplant. 2001;7:216–222. doi: 10.1053/bbmt.2001.v7.pm11349808. [DOI] [PubMed] [Google Scholar]
- 2.Atkins MB, Robertson MJ, Gordon M, Lotze MT, DeCoste M, DuBois JS, Ritz J, Sandler AB, Edington HD, Garzone PD, Mier JW, Canning CM, Battiato L, Tahara H, Sherman ML. Phase I evaluation of intravenous recombinant human interleukin 12 in patients with advanced malignancies. Clin Cancer Res. 1997;3:409–417. [PubMed] [Google Scholar]
- 3.Bajetta E, Del Vecchio M, Mortarini R, Nadeau R, Rakhit A, Rimassa L, Fowst C, Borri A, Anichini A, Parmiani G. Pilot study of subcutaneous recombinant human interleukin 12 in metastatic melanoma. Clin Cancer Res. 1998;4:75–85. [PubMed] [Google Scholar]
- 4.Baker J, Verneris MR, Ito M, Shizuru JA, Negrin RS. Expansion of cytolytic CD8(+) natural killer T cells with limited capacity for graft-versus-host disease induction due to interferon gamma production. Blood. 2001;97:2923–2931. doi: 10.1182/blood.V97.10.2923. [DOI] [PubMed] [Google Scholar]
- 5.Borowsky AD, Namba R, Young LJT, Hunter KW, Hodgson JG, Tepper CG, McGoldrick ET, Muller WJ, Cardiff R, Gregg JP. Syngeneic mouse mammary carcinoma cell lines: Two closely related cell lines with divergent metastatic behavior. Clin Exp Metastasis. 2005;22:47–58. doi: 10.1007/s10585-005-2908-5. [DOI] [PubMed] [Google Scholar]
- 6.Cao YA, Bachmann MH, Beilhack A, Yang Y, Tanaka M, Swijnenburg RJ, Reeves R, Taylor-Edwards C, Schulz S, Doyle TC, Fathman CG, Robbins RC, Herzenberg LA, Negrin RS, Contag CH. Molecular imaging using labeled donor tissues reveals patterns of engraftment, rejection, and survival in transplantation. Transplantation. 2005;80:134–139. doi: 10.1097/01.TP.0000164347.50559.A3. [DOI] [PubMed] [Google Scholar]
- 7.Cao YA, Wagers AJ, Beilhack A, Dusich J, Bachmann MH, Negrin RS, Weissman IL, Contag CH. Shifting foci of hematopoiesis during reconstitution from single stem cells. Proc Natl Acad Sci USA. 2004;101:221–226. doi: 10.1073/pnas.2637010100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Car BD, Eng VM, Lipman JM, Anderson TD. The toxicology of interleukin-12: a review. Toxicol Pathol. 1999;27:58–63. doi: 10.1177/019262339902700112. [DOI] [PubMed] [Google Scholar]
- 9.Cavallo F, Signorelli P, Giovarelli M, Musiani P, Modesti A, Brunda MJ, Colombo MP, Forni G. Antitumor efficacy of adenocarcinoma cells engineered to produce interleukin 12 (IL-12) or other cytokines compared with exogenous IL-12. J Natl Cancer Inst. 1997;89:1049–1058. doi: 10.1093/jnci/89.14.1049. [DOI] [PubMed] [Google Scholar]
- 10.Colombo MP, Trinchieri G. Interleukin-12 in anti-tumor immunity and immunotherapy. Cytokine Growth Factor Rev. 2002;13:155–168. doi: 10.1016/S1359-6101(01)00032-6. [DOI] [PubMed] [Google Scholar]
- 11.Del Vecchio M, Bajetta E, Canova S, Lotze MT, Wesa A, Parmiani G, Anichini A. Interleukin-12: biological properties and clinical application. Clin Cancer Res. 2007;13:4677–4685. doi: 10.1158/1078-0432.CCR-07-0776. [DOI] [PubMed] [Google Scholar]
- 12.Dudley ME, Rosenberg SA. Adoptive cell transfer therapy. Semin Oncol. 2007;34:524–531. doi: 10.1053/j.seminoncol.2007.09.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Edinger M, Cao YA, Verneris MR, Bachmann MH, Contag CH, Negrin RS. Revealing lymphoma growth and the efficacy of immune cell therapies using in vivo bioluminescence imaging. Blood. 2003;101:640–648. doi: 10.1182/blood-2002-06-1751. [DOI] [PubMed] [Google Scholar]
- 14.Gattinoni L, Klebanoff CA, Palmer DC, Wrzesinski C, Kerstann K, Yu Z, Finkelstein SE, Theoret MR, Rosenberg SA, Restifo NP. Acquisition of full effector function in vitro paradoxically impairs the in vivo antitumor efficacy of adoptively transferred CD8+ T cells. J Clin Invest. 2005;115:1616–1626. doi: 10.1172/JCI24480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Giroux M, Denis F. CD1d-unrestricted human NKT cells release chemokines upon Fas engagement. Blood. 2005;105:703–710. doi: 10.1182/blood-2004-04-1537. [DOI] [PubMed] [Google Scholar]
- 16.Gollob JA, Veenstra KG, Parker RA, Mier JW, McDermott DF, Clancy D, Tutin L, Koon H, Atkins MB. Phase I trial of concurrent twice-weekly recombinant human interleukin-12 plus low-dose IL-2 in patients with melanoma or renal cell carcinoma. J Clin Oncol. 2003;21:2564–2573. doi: 10.1200/JCO.2003.12.119. [DOI] [PubMed] [Google Scholar]
- 17.Heinzerling L, Dummer R, Pavlovic J, Schultz J, Burg G, Moelling K. Tumor regression of human and murine melanoma after intratumoral injection of IL-12-encoding plasmid DNA in mice. Exp Dermatol. 2002;11:232–240. doi: 10.1034/j.1600-0625.2001.110306.x. [DOI] [PubMed] [Google Scholar]
- 18.Introna M, Franceschetti M, Ciocca A, Borleri G, Conti E, Golay J, Rambaldi A. Rapid and massive expansion of cord blood-derived cytokine-induced killer cells: an innovative proposal for the treatment of leukemia relapse after cord blood transplantation. Bone Marrow Transplant. 2006;38:621–627. doi: 10.1038/sj.bmt.1705503. [DOI] [PubMed] [Google Scholar]
- 19.Lee P, Wang F, Kuniyoshi J, Rubio V, Stuges T, Groshen S, Gee C, Lau R, Jeffery G, Margolin K, Marty V, Weber J. Effects of interleukin-12 on the immune response to a multipeptide vaccine for resected metastatic melanoma. J Clin Oncol. 2001;19:3836–3847. doi: 10.1200/JCO.2001.19.18.3836. [DOI] [PubMed] [Google Scholar]
- 20.Leemhuis T, Wells S, Scheffold C, Edinger M, Negrin RS. A phase I trial of autologous cytokine-induced killer cells for the treatment of relapsed Hodgkin disease and non-Hodgkin lymphoma. Biol Blood Marrow Transplant. 2005;11:181–187. doi: 10.1016/j.bbmt.2004.11.019. [DOI] [PubMed] [Google Scholar]
- 21.Lenzi R, Rosenblum M, Verschraegen C, Kudelka AP, Kavanagh JJ, Hicks ME, Lang EA, Nash MA, Levy LB, Garcia ME, Platsoucas CD, Abbruzzese JL, Freedman RS. Phase I study of intraperitoneal recombinant human interleukin 12 in patients with Mullerian carcinoma, gastrointestinal primary malignancies, and mesothelioma. Clin Cancer Res. 2002;8:3686–3695. [PubMed] [Google Scholar]
- 22.Lu PH, Negrin RS. A novel population of expanded human CD3+ CD56+ cells derived from T cells with potent in vivo antitumor activity in mice with severe combined immunodeficiency. J Immunol. 1994;153:1687–1696. [PubMed] [Google Scholar]
- 23.Marin V, Dander E, Biagi E, Introna M, Fazio G, Biondi A, D’Amico G. Characterization of in vitro migratory properties of anti-CD19 chimeric receptor-redirected CIK cells for their potential use in B-ALL immunotherapy. Exp Hematol. 2006;34:1219–1229. doi: 10.1016/j.exphem.2006.05.004. [DOI] [PubMed] [Google Scholar]
- 24.Mazzolini G, Alfaro C, Sangro B, Feijoo E, Ruiz J, Benito A, Tirapu I, Arina A, Sola J, Herraiz M, Lucena F, Olague C, Subtil J, Quiroga J, Herrero I, Sadaba B, Bendandi M, Qian C, Prieto J, Melero I. Intratumoral injection of dendritic cells engineered to secrete interleukin-12 by recombinant adenovirus in patients with metastatic gastrointestinal carcinomas. J Clin Oncol. 2005;23:999–1010. doi: 10.1200/JCO.2005.00.463. [DOI] [PubMed] [Google Scholar]
- 25.Mule JJ, Shu S, Schwarz SL, Rosenberg SA. Adoptive immunotherapy of established pulmonary metastases with LAK cells and recombinant interleukin-2. Science. 1984;225:1487–1489. doi: 10.1126/science.6332379. [DOI] [PubMed] [Google Scholar]
- 26.Nishimura R, Baker J, Beilhack A, Zeiser R, Olson JA, Sega EI, Karimi M, Negrin RS. In vivo trafficking and survival of cytokine-induced killer cells resulting in minimal GVHD with retention of antitumor activity. Blood. 2008;112:2563–2574. doi: 10.1182/blood-2007-06-092817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Powell DJ, Jr, Dudley ME, Robbins PF, Rosenberg SA. Transition of late-stage effector T cells to CD27+ CD28+ tumor-reactive effector memory T cells in humans after adoptive cell transfer therapy. Blood. 2005;105:241–250. doi: 10.1182/blood-2004-06-2482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Rook AH, Wood GS, Yoo EK, Elenitsas R, Kao DM, Sherman ML, Witmer WK, Rockwell KA, Shane RB, Lessin SR, Vonderheid EC. Interleukin-12 therapy of cutaneous T-cell lymphoma induces lesion regression and cytotoxic T-cell responses. Blood. 1999;94:902–908. [PubMed] [Google Scholar]
- 29.Rosenberg SA, Restifo NP, Yang JC, Morgan RA, Dudley ME. Adoptive cell transfer: a clinical path to effective cancer immunotherapy. Nat Rev Cancer. 2008;8:299–308. doi: 10.1038/nrc2355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Rosenberg SA, Spiess P, Lafreniere R. A new approach to the adoptive immunotherapy of cancer with tumor-infiltrating lymphocytes. Science. 1986;233:1318–1321. doi: 10.1126/science.3489291. [DOI] [PubMed] [Google Scholar]
- 31.Rosenberg SA, Yannelli JR, Yang JC, Topalian SL, Schwartzentruber DJ, Weber JS, Parkinson DR, Seipp CA, Einhorn JH, White DE. Treatment of patients with metastatic melanoma with autologous tumor-infiltrating lymphocytes and interleukin 2. J Natl Cancer Inst. 1994;86:1159–1166. doi: 10.1093/jnci/86.15.1159. [DOI] [PubMed] [Google Scholar]
- 32.Schmidt-Wolf IG, Finke S, Trojaneck B, Denkena A, Lefterova P, Schwella N, Heuft HG, Prange G, Korte M, Takeya M, Dorbic T, Neubauer A, Wittig B, Huhn D. Phase I clinical study applying autologous immunological effector cells transfected with the interleukin-2 gene in patients with metastatic renal cancer, colorectal cancer and lymphoma. Br J Cancer. 1999;81:1009–1016. doi: 10.1038/sj.bjc.6690800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Schmidt-Wolf IG, Lefterova P, Mehta BA, Fernandez LP, Huhn D, Blume KG, Weissman IL, Negrin RS. Phenotypic characterization and identification of effector cells involved in tumor cell recognition of cytokine-induced killer cells. Exp Hematol. 1993;21:1673–1679. [PubMed] [Google Scholar]
- 34.Schmidt-Wolf IG, Negrin RS, Kiem HP, Blume KG, Weissman IL. Use of a SCID mouse/human lymphoma model to evaluate cytokine-induced killer cells with potent antitumor cell activity. J Exp Med. 1991;174:139–149. doi: 10.1084/jem.174.1.139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Thorne SH, Negrin RS, Contag CH. Synergistic antitumor effects of immune cell-viral biotherapy. Science. 2006;311:1780–1784. doi: 10.1126/science.1121411. [DOI] [PubMed] [Google Scholar]
- 36.Tsung K, Meko JB, Tsung YL, Peplinski GR, Norton JA. Immune response against large tumors eradicated by treatment with cyclophosphamide and IL-12. J Immunol. 1998;160:1369–1377. [PubMed] [Google Scholar]
- 37.Verneris MR, Ito M, Baker J, Arshi A, Negrin RS, Shizuru JA. Engineering hematopoietic grafts: purified allogeneic hematopoietic stem cells plus expanded CD8 + NK-T cells in the treatment of lymphoma. Biol Blood Marrow Transplant. 2001;7:532–542. doi: 10.1016/S1083-8791(01)70014-6. [DOI] [PubMed] [Google Scholar]
- 38.Verneris MR, Karami M, Baker J, Jayaswal A, Negrin RS. Role of NKG2D signaling in the cytotoxicity of activated and expanded CD8+ T cells. Blood. 2004;103:3065–3072. doi: 10.1182/blood-2003-06-2125. [DOI] [PubMed] [Google Scholar]
- 39.Wajchman HJ, Pierce CW, Varma VA, Issa MM, Petros J, Dombrowski KE. Ex vivo expansion of CD8+ CD56+ and CD8+ Cancer Res. 2004;64:1171–1180. doi: 10.1158/0008-5472.CAN-3254-2. [DOI] [PubMed] [Google Scholar]
- 40.Weiss GR, O’Donnell MA, Loughlin K, Zonno K, Laliberte RJ, Sherman ML. Phase 1 study of the intravesical administration of recombinant human interleukin-12 in patients with recurrent superficial transitional cell carcinoma of the bladder. J Immunother. 2003;26:343–348. doi: 10.1097/00002371-200307000-00006. [DOI] [PubMed] [Google Scholar]
- 41.Weiss JM, Subleski JJ, Wigginton JM, Wiltrout RH. Immunotherapy of cancer by IL-12-based cytokine combinations. Expert Opin Biol Ther. 2007;7:1705–1721. doi: 10.1517/14712598.7.11.1705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Weng DS, Zhou J, Zhou QM, Zhao M, Wang QJ, Huang LX, Li YQ, Chen SP, Wu PH, Xia JC. Minimally invasive treatment combined with cytokine-induced killer cells therapy lower the short-term recurrence rates of hepatocellular carcinomas. J Immunother. 2008;31:63–71. doi: 10.1097/CJI.0b013e31815a121b. [DOI] [PubMed] [Google Scholar]
- 43.Zoll B, Lefterova P, Csipai M, Finke S, Trojaneck B, Ebert O, Micka B, Roigk K, Fehlinger M, Schmidt-Wolf GD, Huhn D, Schmidt-Wolf IG. Generation of cytokine-induced killer cells using exogenous interleukin-2, -7 or -12. Cancer Immunol Immunother. 1998;47:221–226. doi: 10.1007/s002620050524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Zoll B, Lefterova P, Ebert O, Huhn D, Von Ruecker A, Schmidt-Wolf IG. Modulation of cell surface markers on NK-like T lymphocytes by using IL-2, IL-7 or IL-12 in vitro stimulation. Cytokine. 2000;12:1385–1390. doi: 10.1006/cyto.2000.0733. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.