Abstract
Patient presented with swelling over right temporal region causing the dilemma over diagnosis. Sonography and CT scan confirmed the presence of cysticercus in right temporal region. Pharmacological management was instituted and the cyst resolved eventually. Ultrasonography and CT imaging play an important role in establishing the diagnosis of rare case of cysticercosis of temporalis muscle and can be treated successfully by pharmacological modality.
Keywords: Temporalis cysticercosis, Ultrasonography, Pharmacological management
Introduction
Taenia solium infestation is one of the ‘neglected tropical diseases’ of worldwide public-health importance. As per declaration by 2003 World Health Assembly it is eradicable [1].
Infection with the larval (cysticercus) stage of T. solium leads to cysticercosis commonly involving the brain, meninges, and eyes, which together constitute 86 % of the cases. The remainder are located in the muscles, subcutaneous layers, liver, heart, lungs, and peritoneum [2]. Cases in the maxillofacial region, including the tongue and cheek muscles, are rarely reported [3]. We report a case of cysticercosis involving the temporalis muscle diagnosed on the basis of high-resolution sonography.
Case Report
A 16 year old boy reported to the department of Oral and Maxillofacial Surgery with the chief complaint of pain and swelling over right temporal region and a limited/restricted mouth opening subsequent to the swelling.
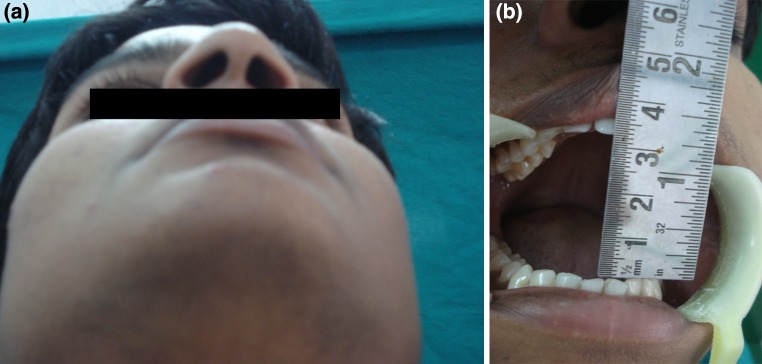
The inspection revealed a solitary diffuse swelling with blended, imperceptible margins along the lateral aspect of the face on the right side above the zygomatic arch and extending over the right temple and upper eyelid (Fig. 1a, b). Palpation was confirmatory of the inspection findings and revealed a pitting type edema, elevated local temperature, tender swelling which was fixed to the underlying subcutaneous tissue structures. Mouth opening of the patient was recorded as 12 mm (Fig. 2).
Fig. 1.
a Diffused swelling extending to lateral part of right upper eyelid. b Worm’s eye view showing swelling in right upper eyelid
Fig. 2.
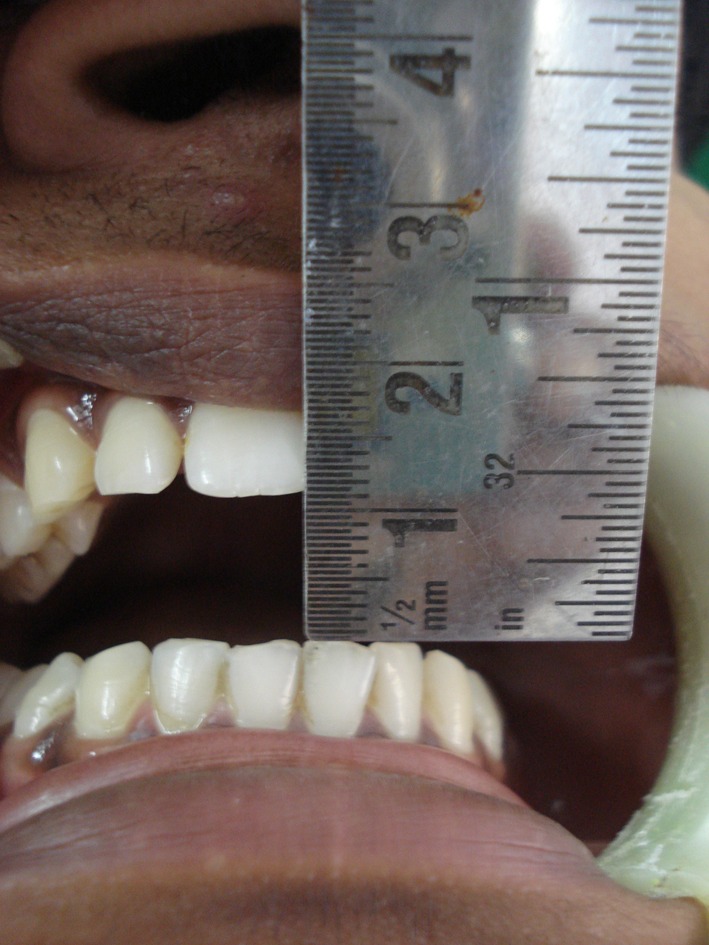
Pre treatment mouth opening of 12 mm
Upon further investigations, the orthopantomogram (OPG) was found to be non-remarkable. Ultrasonography revealed hypoechoic area in right temporalis muscle and small well defined cystic lesion with nodular hyperechoic (nodule) scolex seen within the cystic lesion (Fig. 3). Computed Tomography confirmed the presence of cysticercosis in the right temporalis muscle (Fig. 4).
Fig. 3.

Ultrasonography revealing cystic lesion with nodular hyperechoic scolex
Fig. 4.
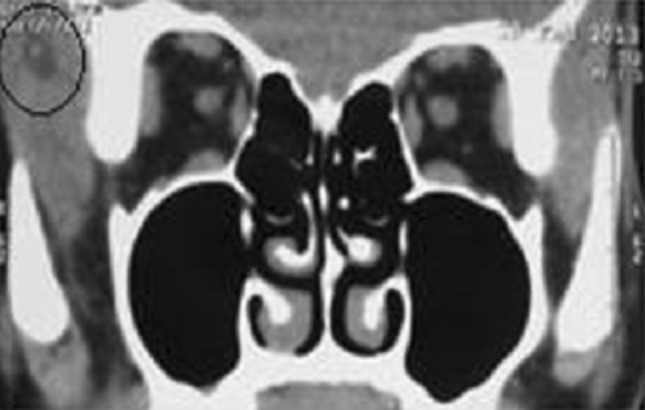
CT scan confirming the presence of cysticercosis in the right temporalis muscle
Conservative treatment was instituted which included Tablet Albendazole 400 mg twice daily for 14 days, tablet prednisolone 30 mg once daily for first 3 days, 20 mg once daily for next 7 days and subsequent tapering dose for next 4 days along with tablet diclofenac and paracetamol combination if needed by the patient.
The post treatment follow-up showed improvement in the mouth opening and swelling (Fig. 5a, b). Ultrasonography revealed no hypoechoic area which was seen earlier confirming complete resolution of the cysticercosis lesion (Fig. 6).
Fig. 5.
a Worm’s eye view showing the absence of swelling. b Improved post treatment mouth opening
Fig. 6.
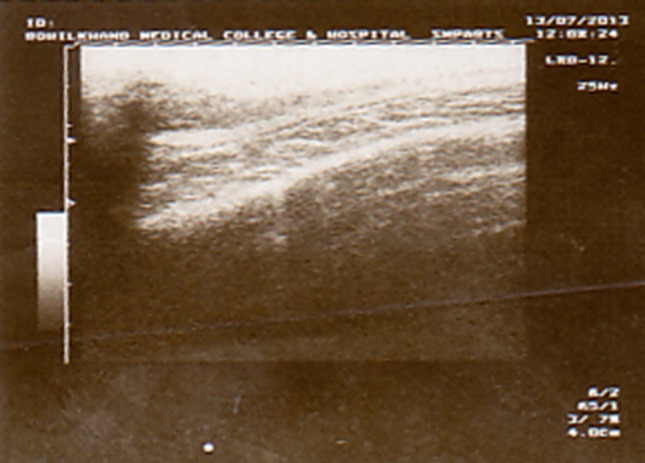
Post treatment ultrasonograph of the right temporalis muscle confirming the resolution of the hypoechoic area seen earlier
Discussion
Cysticercosis as a disease has been reported since the times of Hippocrates. However the works of Kuchenmiester (1855) and later Leucart (1956) had first identified the life cycle of T. solium and differentiated it from T. saginata.
It is a common parasitic disease affecting most of the developing countries, especially Latin-America, Asia and Africa. These countries have high prevalence of cysticercosis because of their close contact between humans and pigs, and poor hygiene [4].
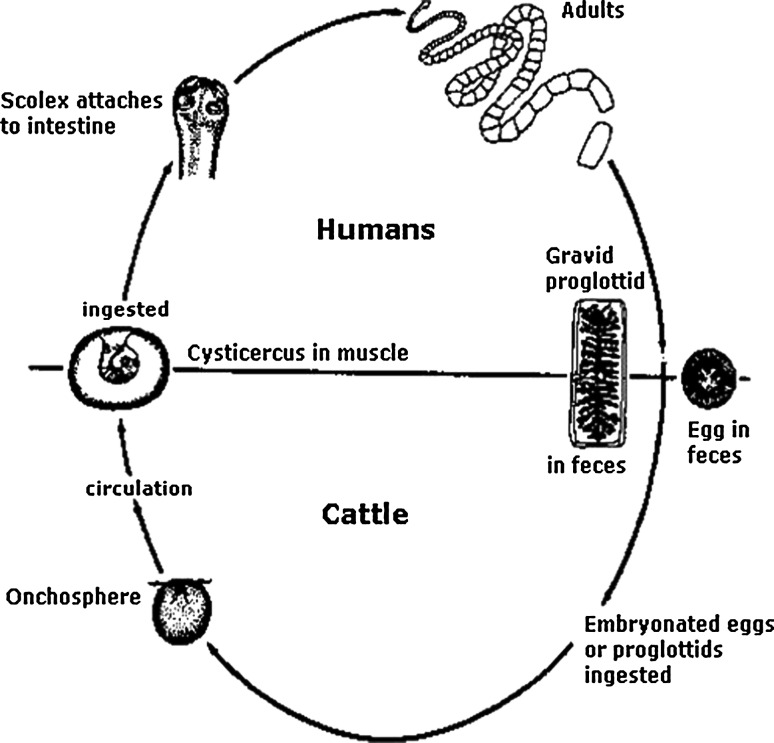
Taenia Solium uses two hosts in its life cycle with human being as a definite host that harbours the adult tape worm (Fig. 7). This infestation may present clinically as ‘intestinal taeniasis’ due to ingestion of undercooked pork contaminated with cysticerci, or/and as ‘tissue cysticercosis’ in subcutaneous tissue, muscle, or other tissues/organs due to ingestion of viable eggs released from the worm carriers [1].
Fig. 7.
Life cycle of Taenia solium
Three sets of variables determine the clinical presentation, course and prognosis: (1) location, growth, size and number of cysts; (2) viability of lesions, stage of cyst degeneration and presence of calcifications; and (3) type and degree of host response. Commonly, patients present with multiple lesions such as cysts of different sizes associated with varying degrees of host response, nodular enhancing lesions and residual calcifications sometimes accompanied by perilesional edema [5]. The cysts are ovoid and white to opalescent, rarely exceeding 1.5 cm, and contain an invaginated scolex with hooklets that are bathed in clear cyst fluid [6].
In our case there was solitary muscular involvement which is rare (Table 1) and caused diagnostic dilemma to the clinician.
Table 1.
| Author/year | Muscles affected | No. of cases | |
|---|---|---|---|
| 1 | Reddi et al. 2001 [7] | Masseter | 1 |
| 2 | Sidhu et al. 2002 [8] | Masseter | 1 |
| 3 | Sethi et al. 2007 [9] | Temporalis | 1 |
| 4 | Mittal et al. 2008 [10] | Masseter | 1 |
| 5 | Gokarn et al. 2011 [11] | Masseter | 1 |
| 6 | Kumar et al. 2011 [12] | Temporalis | 1 |
| 7 | Kumar et al. 2011 [13] | Masseter | 1 |
| 8 | Sarabjeet Singh et al. 2013 [14] | Temporalis | 2 |
Vijayaraghavan described four different patterns of muscular cysticercosis. The first type is cysticercus cyst with an inflammatory mass around it, as a result of the death of the larva. The second type is an irregular cyst with very minimal fluid on one side, indicating a leakage of fluid. The eccentric echogenic protrusion from the wall due to the scolex is not seen within the cyst. The third appearance is a large irregular collection of exudative fluid within the muscle with the typical cysticercus cyst containing the scolex, situated eccentrically within the collection. This appearance is similar to an intramuscular abscess. In our case the sonographic appearance is similar to this type as hypoechoic area was seen in the right temporal muscle and a small well defined cystic lesion with nodular hyperechoic (nodule) scolex was seen within the cystic lesion while clinical appearance was similar to intramuscular abscess causing diagnostic dilemma. The fourth type of sonographic appearance is that of calcified cysticercosis [15].
Although laboratory investigations are inferior to the radiological investigations in diagnosing the cysticercosis, Serology is the most useful laboratory test of which ELISA has 74 % of sensitivity and is highly specific for the appropriate antigens while enzyme linked immunoelectrotransfer blot has 95 % sensitivity and 100 % specificity. However they are more useful in cases with multiple cysts. Beside these, stool test can be carried out for ova and parasite [16].
Ultrasonography, computer tomography (CT) and magnetic resonance imaging (MRI) play an important and essential role in understanding of cysticercosis. The histopathological changes resulting from cysticercosis are well known and have been extensively described in the Handbook of Neurology in 1912 [5].
MRI is the investigation that best reflects the pathological state of the cyst and host response. As with most of the infections of long duration, there are distinct pathological stages and in cysticercosis, these are reflected in the radiological examination. The radiological techniques have allowed easy, non-invasive way to visualize lesions, establish the diagnosis in most cases, determine the treatments likely to be worthwhile and gauge the effectiveness of therapy. Cysticercosis can be staged according to the MRI findings:
Stage I—Shows enhancement around cysts implying disruption of the blood brain barrier and initial recognition of the cyst and reaction to it by the host. The parasite is still viable and appears on the T1 image to be a hypodense cystic structure. Some degree of associated inflammation is present.
Stage II—The next stage is distinguished by the presence of both enhancement and surrounding edema indicative of inflammation. At this stage an intense inflammatory response surrounds the cyst forming a host inflammatory pseudocapsule. At this stage, the basic molecular make-up of the cyst has changed, probably irreversibly damaged and over a period, the cyst becomes smaller with decreased edema but usually with enhancement.
Stage III—Eventually, the cyst resolves into a small, characteristically round lesion with or without calcification and accompanying enhancement. Microscopically, these are granulomas containing varying amounts of parasite remnants but, sometimes, only the suggestion of the previous presence of the T. solium larva is detection of calcareous corpuscles, characteristic microscopic calcium containing deposits characteristic of cestodes and hooklets.
There are five treatment modalities that can be offered to patients: (1) larvicidal agents to kill the cystic larvae and/or tapeworm; (2) corticosteroids or other immunosuppressive agents to decrease or prevent inflammation; (3) anti-seizure medication to prevent or decrease the severity and number of seizures; (4) surgical-based therapies to decrease the mass effect of cysts with or without accompanying inflammation; and (5) general supportive measures in impaired individuals or symptomatic treatments [5].
We instituted the conservative management with Albandazole and Prednisolone. Pharmacological management of cysticercosis with the cysticidal drugs like praziquantel and albendazole help by reducing the parasite burden by facilitating the death of the cysts. However, it may be associated with severe reactions, like local tissue swelling and generalized anaphylactic reaction as a result of massive release of antigens. The corticosteroids would help in decreasing the incidence of such complications if used before starting the cysticidal drug [4].
In cases of disseminated cysticercosis, albendazole is used in a dose of 15 mg/kg/day for 30 days and praziquantel in a dose of 50–75 mg/kg/day in three divided dosage for 15–20 days. The advantage of albendazole over praziquantel is that the former also destroys subarachnoid and ventricular cysts because of its better penetration in CSF, as well as the fact that it can be administered jointly with corticosteroid agents for anti-inflammatory therapy [4]. Serum levels of praziquantel are increased by ingestion with meals, high carbohydrate diet, co-administration with and cimetidine and ingestion with grapefruit juice while that of albendazole is increased by fatty food [5].
Prognosis in the maxillofacial region is excellent with no recurrence. In contrast to the gravity of the disease in the cerebral, ocular, or cardiac sites, it is often well-tolerated. When associated with other localizations, the prognosis depends upon the site and number of other larval localizations and may be very severe [17].
Conclusion
Although rare, but cysticercosis of tempralis muscle should be considered as differential diagnosis of swelling over temporal region. Ultrasonography and CT scan play an important role in the diagnosis of cysticercosis.
Conflict of interest
None.
References
- 1.Akhtar MN, Agarwal S (2012) Disseminated cysticercosis incidentally diagnosed in a patient of low backache: a case report and concise review of literature. Asian Pac J Trop Med 5(7):582–586 [DOI] [PubMed]
- 2.Rao SC, Sharma H, Vinay KN, Vidya KC (2011) Oral cysticercosis a case report. Int J Clin Dent Sci 2(4):36–39
- 3.Timosca G, Gavrileta L. Cysticercosis of maxillofacial region. Oral Surg Oral Med Oral Pathol. 1974;37:390–400. doi: 10.1016/0030-4220(74)90112-1. [DOI] [PubMed] [Google Scholar]
- 4.Park SY, Kong MH, Kim JH, Song KY. Disseminated cysticercosis. J Korean Neurosurg Soc. 2011;9:190–193. doi: 10.3340/jkns.2011.49.3.190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Nash TE. Human case management and treatment of cysticercosis. Acta Trop. 2003;87:61–69. doi: 10.1016/S0001-706X(03)00056-1. [DOI] [PubMed] [Google Scholar]
- 6.Sharma P, Neupane S, Shrestha M, Dwivedi R, Paudel K. An ultrasonographic evaluation of solitary muscular and soft tissue cysticercosis. Kathmandu Univ Med J. 2010;8(30):257–260. doi: 10.3126/kumj.v8i2.3571. [DOI] [PubMed] [Google Scholar]
- 7.Reddi SP, Morales MJ, Addante RR. Solitary lesion in the masseter muscle. J Oral Maxillofac Surg. 2011;59(1):71–75. doi: 10.1053/joms.2001.19294. [DOI] [PubMed] [Google Scholar]
- 8.Sidhu R, Nada R, Palta A, Mohan H, Suri S. Maxillofacial cysticercosis: uncommon appearance of a common disease. J Oral Maxillofac Surg. 2002;21(2):199–202. doi: 10.7863/jum.2002.21.2.199. [DOI] [PubMed] [Google Scholar]
- 9.Sethi Pk, Sethi NK, Torgovnick J, Arsura E. Cysticercosis of temporalis muscle: an unusual cause of temporal headaches. A case report. J Headache Pain. 2007;8(5):315–316. doi: 10.1007/s10194-007-0410-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Mittal A, Das D, Iyer N, Nagaraj J, Gupta M. Masseter cysticercosis—a rare case diagnosed on ultrasound. Dentomaxillofacial Radiol. 2008;37(2):113–116. doi: 10.1259/dmfr/31885135. [DOI] [PubMed] [Google Scholar]
- 11.Gokarn A, Gokarn S, Rathod V. Ultrasonography for massete muscle cysticercosis. Indian Pediatr. 2011;48(2):141–143. [PubMed] [Google Scholar]
- 12.Kumar V, Gulati A, Mehra B. Cysticercosis of the temporalis muscle causing temporal headache in a pregnant woman. Int J Gynaecol Obstet. 2011;114(1):79. doi: 10.1016/j.ijgo.2011.01.022. [DOI] [PubMed] [Google Scholar]
- 13.Kumar BD, Dave B, Meghana SM. Cysticercosis of masseter. Indian J Dent Res. 2011;22(4):617. doi: 10.4103/0970-9290.90334. [DOI] [PubMed] [Google Scholar]
- 14.Singh S, et al. Cysticercosis involving muscle of mastication: a review and report of two cases. Case Rep Dent. 2013 doi: 10.1155/2013/814126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Vijayaraghavan SB (2004) Sonographic appearances in cysticercosis. J Ultrasound Med 23:423–427 [DOI] [PubMed]
- 16.Shivu ME, Jayanthi K. Primary sonographic diagnosis of subcutaneous cysticercosis. J Indian Acad Oral Med Radiol. 2011;23(3):241–244. [Google Scholar]
- 17.Deshmukh A, Avadhani A, Tupkari JV, Sardar M. Cysticercosis of the upper lip. J Oral Maxillofac Pathol. 2011;15(2):219–222. doi: 10.4103/0973-029X.84509. [DOI] [PMC free article] [PubMed] [Google Scholar]