This study shows that among 903 816 severe sepsis and 410 623 sepsis hospitalizations at academic medical center–affiliated hospitals during 2012–2014, prior receipt of a solid organ transplant (kidney, liver, or co-transplant) was associated with reduced inpatient mortality.
Keywords: sepsis, outcomes, transplant, infection, critical care
Abstract
Background. Solid organ transplant (SOT) recipients are at elevated risk of sepsis. The impact of SOT on outcomes following sepsis is unclear.
Methods. We performed a retrospective cohort study using data from University HealthSystem Consortium, a consortium of academic medical center affiliates. We examined the association between SOT and mortality among patients hospitalized with severe sepsis or explicitly coded sepsis in 2012–2014. We used International Classification of Diseases, Ninth Revision (ICD-9) codes to identify severe sepsis, explicitly coded sepsis, and SOT (kidney, liver, heart, lung, pancreas, or intestine transplants). We fit random-intercept logistic regression models to account for clustering by hospital.
Results. There were 903 816 severe sepsis hospitalizations (39 618 [4.4%] with SOT) and 410 623 sepsis hospitalizations (14 526 [3.9%] with SOT) in 250 hospitals. SOT recipients were younger and more likely to be insured by Medicare than those without SOT. Among hospitalizations for severe sepsis and sepsis, in-hospital mortality was lower among those with vs those without SOT (5.5% vs 9.4% for severe sepsis; 8.7% vs 12.7% for sepsis). After adjustment, the odds ratio for mortality comparing SOT patients vs non-SOT was 0.83 (95% confidence interval [CI], .79–.87) for severe sepsis and 0.78 (95% CI, .73–.84) for sepsis. Compared to non-SOT patients, kidney, liver, and co-transplant (kidney-pancreas/kidney-liver) recipients demonstrated lower mortality. No association was present for heart transplant, and lung transplant was associated with higher mortality.
Conclusions. Among patients hospitalized for severe sepsis or sepsis, those with SOT had lower inpatient mortality than those without SOT. Identifying the specific strategies employed for populations with improved mortality could inform best practices for sepsis among SOT and non-SOT populations.
In the United States, nearly 30 000 transplants were performed in 2014, and the number of solid organ transplant (SOT) recipients living with a functioning graft has been growing [1]. Although transplantation is a high-cost, intensive procedure, it is considered the best treatment for individuals experiencing organ failure, and has been shown to extend the life expectancy of recipients dramatically [2, 3]. For example, kidney transplant has been demonstrated to be the most cost-effective treatment for patients suffering from end-stage renal disease [4]. However, infection is the second leading cause of death for SOT recipients, a population that experiences increased risk of infectious complications posttransplant due to induced immunosuppression, prolonged hospitalization, and intensive surgery [5–8].
Severe sepsis, or infection with systemic inflammation complicated by organ dysfunction, poses a substantial burden on the US healthcare system, leading to >750 000 hospitalizations and 200 000 deaths annually [9, 10]. Severe sepsis remains a leading cause of death in the United States, with in-hospital mortality ranging from 12% to 26% [11, 12]. Sepsis represents a common complication of transplant procedures, and SOT recipients have been shown to be at increased risk for sepsis compared with the general population [5, 13, 14]. However, several studies have shown that the syndrome of sepsis has a wider range of causative organisms and differing presentation among immunosuppressed individuals compared with immunocompetent individuals [15, 16]. In addition, traditional markers of systemic inflammatory response syndrome may not be present among the immunosuppressed, despite active overwhelming infection [17, 18]. Consistent with a reduced inflammatory response, a single-center study comparing bacteremic sepsis patients who were prior SOT recipients with non-SOT patients matched on age, sex, and location at the day of blood culture collection demonstrated increased survival among those with prior SOT [19]. However, few national studies have been conducted to determine if these observations are consistent across the broader population of SOT recipients.
Further study of sepsis outcomes among SOT recipients could help to inform future strategies for prevention and management, and also allow for optimization of posttransplant care among SOT recipients. Therefore, we examined the association between prior receipt of SOT and inpatient mortality following severe sepsis and sepsis in a national consortium of academic medical center–affiliated hospitals.
METHODS
Study Design
We conducted a retrospective cohort study using hospital discharge data from the University HealthSystem Consortium (UHC) clinical database (CDB). All data were submitted to UHC from individual institutions and provided for research purposes through the CDB. The study received approval from the Institutional Review Board of the University of Alabama at Birmingham for retrospective analysis of existing data.
Data Source
The UHC is a consortium of academic medical centers and affiliated hospitals in the United States. With hospitals in 42 states, UHC aims to improve clinical, operational, and financial performance [20]. For quality improvement purposes, UHC maintains the CDB, which is a database that contains administrative data submitted by hospitals in the consortium. This data source captures the elements of the standard UB-04 reporting form and encompasses data pertaining to patient demographics, discharge diagnoses, procedures, and outcomes. We used available UHC CDB data for the period 1 January 2012 through 31 December 2014.
Cohort Selection
The cohorts examined in this study consisted of patients hospitalized for severe sepsis and sepsis from UHC hospitals submitting data to the CDB. We excluded patients <18 years old, prisoners or those discharged to law enforcement, patients with unknown or other disposition (ie, left against medical advice), and transfers. We identified severe sepsis using the Angus taxonomy of International Classification of Diseases, Ninth Revision (ICD-9) discharge diagnosis codes [9]. Severe sepsis hospitalizations were defined as those with codes for both infection and organ dysfunction, a strategy that has demonstrated modest sensitivity, high specificity, and high positive predictive value for the identification of events [21–23]. We additionally included explicit sepsis (ICD-9 code 995.91) and severe sepsis (995.92) codes in our definition (cases with code 995.91 must have had concurrent presence of organ dysfunction to be considered severe sepsis). For a sensitivity analysis with regard to coding practices for infection and organ dysfunction, we also defined a cohort of sepsis patients as hospitalizations with explicit codes for sepsis, severe sepsis, or septic shock (ICD-9 codes 995.91, 995.92, or 785.52).
We used present-on-admission flags available in the data set to restrict the analysis to hospitalizations with infection and organ dysfunction present at the time of admission (or an explicit sepsis code present at the time of admission for the sepsis definition) [24]. In a sensitivity analysis, hospitalizations for severe sepsis were additionally classified as community-acquired or potentially healthcare-associated, based on whether or not the case was admitted from a skilled nursing facility or home health, was on dialysis, or represented a readmission within 30 days of a prior discharge from the same institution.
We used several control populations to determine if our findings were unique to sepsis. Specifically, we examined heart failure (ICD-9 codes 402.01, 402.11, 402.91, 404.01, 404.03, 404.11, 404.13, 404.91, 404.93, 428), acute myocardial infarction (AMI) (ICD-9 codes 410.x0, 410.x1 [where x = 0–9]), and chronic obstructive pulmonary disease (COPD) (ICD-9 codes 491.21, 491.22, 491.8, 491.9, 492.8, 493.20, 493.21, 493.22, 496, 518.81, 518.82, 518.84, 799.1) [25]. Discharge diagnoses had to be listed in the primary position for a hospitalization to be included in a given control population.
Exposure, Hospitalization Characteristics, and Outcome
The primary exposure was presence of an SOT. To identify patients with a history of SOT, we used ICD-9 discharge codes specifying status as an organ transplant recipient and codes specifying hospitalization for a complication related to a previously transplanted organ. Organ transplant status was obtained for kidney (ICD-9 code V42.0), heart (V42.1), lung (V42.6), liver (V42.7), pancreas (V42.83), and intestines (V42.84) [26]. We also identified organ complications present on admission for transplants of the kidney (996.81), heart (996.83), lung (996.84), liver (996.82), pancreas (996.86), and intestines (996.87) [26]. SOT recipients were categorized based on transplant organ, with the categories kidney, liver, heart, lung, co-transplant (kidney-pancreas or kidney-liver), and other (pancreas only or intestine).
Demographic factors obtained for the current study included age, sex, race, and insurance type. We categorized hospitalizations based on risk of mortality at admission using the 3M (St Paul, Minnesota) All Patient Refined diagnosis-related group (APR-DRG) measure with standard categories (minor, moderate, major, or extreme) [27]. We also determined discharge physician specialty, admission to the intensive care unit (ICU), and Elixhauser comorbidities [28]. This approach for risk adjustment included building a comprehensive model with factors that could be conceptualized as confounding the association between SOT and mortality, instead of using DRG-specific risk models [20]. We also calculated the total number of eligible, severe sepsis, and SOT hospitalizations per hospital.
We identified inpatient mortality using discharge status codes available in the data set. Hospitalized patients with any status code other than death were considered alive at the time of discharge. Discharge status codes in the UHC database have demonstrated high concordance with results obtained via patient-level case audits [29].
Statistical Analysis
We calculated the percentage of hospitalizations with severe sepsis and sepsis by SOT status. We also calculated the distributions of sepsis by transplanted organ. Characteristics of patients hospitalized for severe sepsis and, separately, sepsis were calculated and compared by SOT status using Pearson χ2 tests for categorical variables and nonparametric Wilcoxon rank-sum tests of equal distribution for continuous variables. We calculated the percentage of hospitalizations experiencing in-hospital mortality. The associations between SOT and inpatient mortality were estimated using mixed-effects logistic regression models specifying hospital identifiers as a random intercept. Odds ratios were calculated unadjusted and after adjustment for age, sex, race, insurance type, ICU admission, APR-DRG risk of mortality, discharge physician specialty (dichotomized as general internal medicine vs other), and Elixhauser comorbidities. We examined co-transplants (kidney-pancreas and kidney-liver) and classified pancreas-only or intestinal transplants as “Other.” We did not assess mortality among those with pancreas-only or intestinal transplants, as the total number of deaths was insufficient. We performed sensitivity analyses restricting the population to severe sepsis cases admitted to the ICU at any point during hospitalization, classifying severe sepsis cases as community-acquired or healthcare-associated, and examining the association between SOT and in-hospital mortality in control populations (heart failure, AMI, and COPD). We performed an additional sensitivity analysis among severe sepsis and sepsis hospitalizations presenting to institutions with at least 100 and 500 SOT hospitalizations per study year. All analyses were performed using Stata software version 13.1 (StataCorp, College Station, Texas).
RESULTS
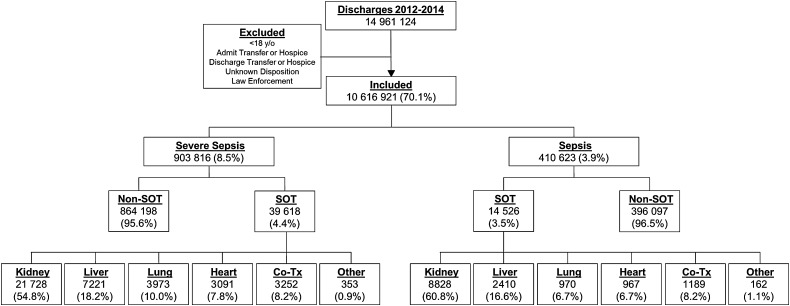
Among a total of 14 961 124 hospitalizations between 2012 and 2014, 10 616 921 hospitalizations at 250 UHC hospitals met the inclusion criteria for the current analysis. There were 903 816 hospitalizations for severe sepsis and 410 623 hospitalizations for sepsis. Kidney transplants were the most common SOT for both severe sepsis and sepsis cases (Figure 1). The median number of eligible hospitalizations per hospital over 2012–2014 was 36 544 (interquartile range [IQR], 6745–60 521), with a median of 3117 (IQR, 1565–5083) severe sepsis hospitalizations and 173 (IQR, 52–1409) SOT hospitalizations.
Figure 1.
University HealthSystem Consortium study population flowchart, 2012–2014. Severe sepsis defined using the Angus criteria (International Classification of Diseases, Ninth Revision [ICD-9] codes for infection and organ dysfunction), while sepsis was defined as any hospitalization with an explicit ICD-9 code for sepsis (995.91), severe sepsis (995.92), or septic shock (785.52). All definitions required that ICD-9 codes were present at the time of admission. Severe sepsis and explicit sepsis are not mutually exclusive definitions; cases can belong to both groups. All percentages relative to category above. Abbreviations: Co-Tx, kidney-pancreas or kidney-liver co-transplant; Other, pancreas-only transplant or intestine transplant; SOT, solid organ transplant.
Among severe sepsis and sepsis hospitalizations, those with prior SOT were more likely to be younger, male, and insured by Medicare or private insurance (Table 1). Patients with SOT were also more likely to have hypertension, diabetes, renal failure, liver disease, and deficiency anemia, and to have been discharged by a specialist. In contrast, SOT patients were less likely to be admitted to the ICU and have comorbid heart failure, paralysis, neurological disorder, chronic pulmonary disease, metastatic cancer, solid tumor, obesity, alcohol or drug abuse, and psychoses. Differences in discharge characteristics were similar when severe sepsis was classified as community-acquired or healthcare-associated (Supplementary Table 1).
Table 1.
Characteristics of Individuals Hospitalized for Severe Sepsis and Sepsis by Solid Organ Transplant Status, 2012–2014
| Characteristic | Severe Sepsis Hospitalizations |
Sepsis Hospitalizations |
||||
|---|---|---|---|---|---|---|
| Non-SOT (n = 864 198) | Any SOT (n = 39 618) | P Value | Non-SOT (n = 396 097) | Any SOT (n = 14 526) | P Value | |
| Age, y, median (IQR) | 66 (55–79) | 58 (47–66) | <.001 | 63 (50–76) | 57 (45–65) | <.001 |
| Age, y | <.001 | <.001 | ||||
| ≤30 | 3.9 | 7.9 | 6.8 | 8.5 | ||
| 31–45 | 8.3 | 15.5 | 12.0 | 17.1 | ||
| 46–60 | 24.9 | 35.3 | 25.7 | 34.8 | ||
| ≥61 | 63.0 | 41.3 | 55.5 | 39.6 | ||
| Sex | <.001 | <.001 | ||||
| Male | 49.7 | 54.9 | 50.8 | 54.7 | ||
| Female | 50.3 | 45.1 | 49.2 | 45.3 | ||
| Race | <.001 | <.001 | ||||
| White | 67.2 | 63.1 | 65.1 | 61.9 | ||
| Black/African American | 22.6 | 24.0 | 22.9 | 24.2 | ||
| Other | 10.2 | 12.9 | 12.0 | 13.9 | ||
| Pay type | <.001 | <.001 | ||||
| Medicare | 64.8 | 69.8 | 58.9 | 70.7 | ||
| Medicaid | 15.3 | 9.0 | 17.4 | 9.0 | ||
| Private | 13.4 | 19.5 | 15.7 | 18.6 | ||
| Self-pay | 4.2 | 1.0 | 5.2 | 0.9 | ||
| Other | 2.4 | 0.8 | 2.9 | 0.8 | ||
| APR-DRG ROM (on admission) | <.001 | <.001 | ||||
| Minor | 5.0 | 6.5 | 9.2 | 6.5 | ||
| Moderate | 22.5 | 32.0 | 17.6 | 19.2 | ||
| Major | 48.9 | 44.4 | 38.8 | 44.9 | ||
| Extreme | 23.6 | 17.1 | 34.3 | 29.4 | ||
| Admitted to ICU (at any time during hospitalization) | 33.7 | 27.1 | <.001 | 39.8 | 38.5 | .002 |
| Elixhauser comorbidity group | ||||||
| Congestive heart failure | 22.0 | 14.7 | <.001 | 19.7 | 16.0 | <.001 |
| Valvular disease | 7.8 | 6.7 | <.001 | 7.9 | 8.5 | .009 |
| Pulmonary circulation disease | 7.0 | 6.4 | <.001 | 6.6 | 6.8 | .29 |
| Peripheral vascular disease | 9.1 | 8.5 | <.001 | 8.3 | 9.6 | <.001 |
| Hypertension | 57.8 | 60.8 | <.001 | 58.1 | 71.6 | <.001 |
| Paralysis | 5.0 | 1.7 | <.001 | 7.7 | 2.3 | <.001 |
| Other neurological disorders | 13.6 | 7.6 | <.001 | 15.4 | 8.5 | <.001 |
| Chronic pulmonary disease | 28.5 | 13.8 | <.001 | 24.5 | 12.7 | <.001 |
| Diabetes without complications | 26.0 | 31.5 | <.001 | 24.8 | 29.7 | <.001 |
| Diabetes with complications | 9.2 | 18.6 | <.001 | 8.9 | 20.3 | <.001 |
| Hypothyroidism | 15.2 | 14.5 | <.001 | 13.5 | 14.6 | <.001 |
| Renal failure | 27.3 | 60.8 | <.001 | 23.2 | 70.2 | <.001 |
| Liver disease | 9.8 | 18.9 | <.001 | 7.7 | 17.8 | <.001 |
| AIDS | 0.6 | 0.2 | <.001 | 0.3 | 0.1 | .007 |
| Lymphoma | 2.4 | 1.5 | <.001 | 3.0 | 1.8 | <.001 |
| Metastatic cancer | 4.9 | 1.4 | <.001 | 6.5 | 1.7 | <.001 |
| Solid tumor without metastases | 4.6 | 1.7 | <.001 | 5.1 | 2.2 | <.001 |
| Rheumatoid arthritis | 4.4 | 4.9 | <.001 | 4.7 | 5.6 | <.001 |
| Coagulopathy | 19.6 | 16.5 | <.001 | 13.3 | 14.2 | .002 |
| Obesity | 15.6 | 11.2 | <.001 | 14.6 | 11.9 | <.001 |
| Weight loss | 11.3 | 11.3 | .64 | 12.7 | 10.9 | <.001 |
| Fluid and electrolyte disorders | 50.1 | 52.5 | <.001 | 50.7 | 51.2 | .24 |
| Chronic blood loss anemia | 1.5 | 1.0 | <.001 | 1.2 | 0.8 | <.001 |
| Deficiency anemias | 35.2 | 47.9 | <.001 | 34.7 | 47.7 | <.001 |
| Alcohol abuse | 8.2 | 2.5 | <.001 | 6.0 | 2.0 | <.001 |
| Drug abuse | 5.4 | 1.8 | <.001 | 5.5 | 1.7 | <.001 |
| Psychoses | 6.7 | 3.6 | <.001 | 6.4 | 3.3 | <.001 |
| Depression | 16.0 | 15.6 | .04 | 14.7 | 14.5 | .61 |
| Discharge physician specialty | <.001 | <.001 | ||||
| Family practice | 5.7 | 1.6 | 5.4 | 2.0 | ||
| General internal medicine | 46.8 | 23.0 | 46.8 | 28.3 | ||
| Pulmonary/critical care | 7.0 | 9.3 | 8.0 | 9.0 | ||
| Hospitalist | 16.6 | 9.4 | 17.3 | 12.1 | ||
| Cardiology | 3.3 | 6.2 | 1.9 | 5.1 | ||
| Internal medicine hematology/oncology | 4.2 | 1.3 | 4.9 | 1.5 | ||
| Gastroenterology | 1.4 | 4.9 | 0.7 | 3.8 | ||
| Infectious diseases | 1.5 | 0.8 | 1.7 | 1.2 | ||
| Nephrology | 1.3 | 19.6 | 1.4 | 17.2 | ||
| Hepatology | 0.2 | 1.0 | 0.1 | 0.8 | ||
| Other | 12.1 | 23.0 | 11.8 | 18.9 | ||
Data are presented as column percentage. Total of 903 816 severe sepsis and 410 623 sepsis hospitalizations, 2012–2014. Severe sepsis defined using the Angus criteria (International Classification of Diseases, Ninth Revision [ICD-9] codes for infection and organ dysfunction), while sepsis was defined as any hospitalization with an explicit ICD-9 code for sepsis (995.91), severe sepsis (995.92), or septic shock (785.52). All definitions required that ICD-9 codes were present at the time of admission. P values from Pearson χ2 tests of association for categorical variables and Wilcoxon rank-sum test of equal distribution for continuous variables.
Abbreviations: APR-DRG, All Patient Refined diagnosis-related group; ICU, intensive care unit; IQR, interquartile range; ROM, risk of mortality; SOT, solid organ transplant.
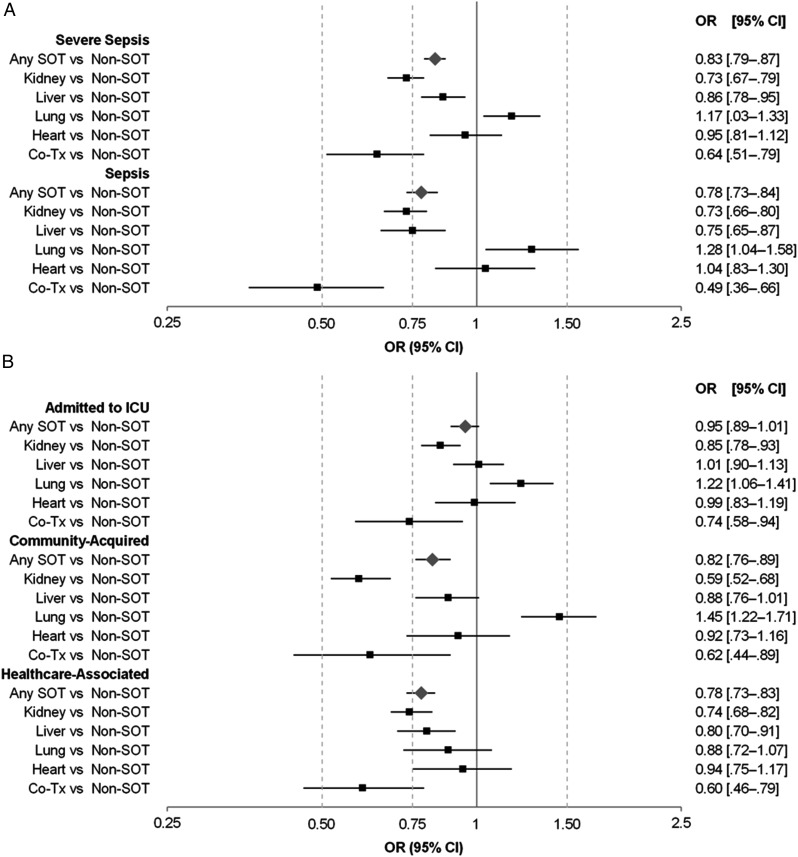
Among hospitalizations for severe sepsis and sepsis, SOT recipients had lower mortality than non-SOT (severe sepsis non-SOT, 9.4% [n = 81 300 deaths]; SOT, 5.5% [n = 2170 deaths]), and after multivariable adjustment, hospitalizations with prior SOT had statistically significantly reduced odds of inpatient mortality compared with non-SOT hospitalizations (Table 2; Figure 2A). Recipients with prior transplant of the kidney, liver, or co-transplant (kidney/liver or kidney/pancreas) had lower mortality, whereas lung transplant recipients had higher mortality. When the analyses were restricted to severe sepsis cases admitted to the ICU, kidney and co-transplants were associated with lower mortality, lung transplants were associated with higher mortality, and no difference was present for liver transplants (Table 3; Figure 2B). For severe sepsis cases classified as community-acquired, recipients with prior kidney, liver, or co-transplants had lower mortality and lung transplant recipients had higher mortality (Figure 2B; Supplementary Table 2). For healthcare-associated severe sepsis, kidney, liver, or co-transplant recipients had lower mortality compared with non-SOT, while a history of lung transplant was not associated with mortality (Figure 2B; Supplementary Table 2). Measures of association were similar to the primary analyses when restricted to hospitals caring for at least 100 and 500 SOT recipients per year (data not shown).
Table 2.
Odds Ratios for Inpatient Mortality Associated With Solid Organ Transplant Among Hospitalizations for Severe Sepsis and Sepsis, 2012–2014
| Population | Total No. | Inpatient Mortality Events, No. (%) | Crude | Adjusteda |
|---|---|---|---|---|
| OR (95% CI) | OR (95% CI) | |||
| Severe sepsis | ||||
| Any SOT vs non-SOT | ||||
| Non-SOT | 864 198 | 81 300 (9.4) | Ref | Ref |
| SOT | 39 618 | 2170 (5.5) | 0.54 (.51–.56) | 0.83 (.79–.87) |
| Specific organ vs non-SOT | ||||
| Non-SOT | 864 198 | 81 300 (9.4) | Ref | Ref |
| Kidney | 21 728 | 876 (4.0) | 0.39 (.36–.42) | 0.73 (.67–.79) |
| Liver | 7221 | 620 (8.6) | 0.87 (.80–.95) | 0.86 (.78–.95) |
| Lung | 3973 | 340 (8.6) | 0.85 (.76–.95) | 1.17 (1.03–1.33) |
| Heart | 3091 | 210 (6.8) | 0.66 (.58–.76) | 0.95 (.81–1.12) |
| Co-transplantb | 3252 | 106 (3.3) | 0.33 (.27–.40) | 0.64 (.51–.79) |
| Sepsis | ||||
| Any SOT vs non-SOT | ||||
| Non-SOT | 396 097 | 50 187 (12.7) | Ref | Ref |
| SOT | 14 526 | 1207 (8.3) | 0.62 (.58–.66) | 0.78 (.73–.84) |
| Specific organ vs non-SOT | ||||
| Non-SOT | 396 097 | 50 187 (12.7) | Ref | Ref |
| Kidney | 8828 | 576 (6.5) | 0.48 (.44–.52) | 0.73 (.66–.80) |
| Liver | 2410 | 311 (12.9) | 1.02 (.90–1.15) | 0.75 (.65–.87) |
| Lung | 970 | 140 (14.4) | 1.14 (.95–1.15) | 1.28 (1.04–1.58) |
| Heart | 967 | 114 (11.8) | 0.89 (.73–1.08) | 1.04 (.83–1.30) |
| Co-transplantb | 1189 | 56 (4.7) | 0.36 (.28–.47) | 0.49 (.36–.66) |
Severe sepsis defined using the Angus criteria (International Classification of Diseases, Ninth Revision [ICD-9] codes for infection and organ dysfunction), while sepsis was defined as any hospitalization with an explicit ICD-9 code for sepsis (995.91), severe sepsis (995.92), or septic shock (785.52). All definitions required that ICD-9 codes were present at the time of admission.
Abbreviations: CI, confidence interval; OR, odds ratio; Ref, reference; SOT, solid organ transplant.
a Adjusted for age, sex, race, insurance type, intensive care unit admission, discharge physician specialty (internal medicine vs not), All Patient Refined risk of mortality, and Elixhauser comorbidities. ORs estimated using random-intercept logistic regression.
b Co-transplants include hospitalizations with a prior kidney transplant in addition to a pancreas or liver transplant. Subtotals do not sum to total SOT, as pancreas-only and intestine transplants were included in the any SOT group, but not examined in transplant-specific analyses.
Figure 2.
Odds ratios for inpatient mortality among severe sepsis and sepsis hospitalizations, 2012–2014. A, Primary analysis of the association between solid organ transplant (SOT) and inpatient mortality among severe sepsis and sepsis hospitalizations. B, Sensitivity analyses of the association between SOT and inpatient mortality among select subgroups of severe sepsis hospitalizations. Severe sepsis was defined using the Angus criteria (International Classification of Diseases, Ninth Revision [ICD-9] codes for infection and organ dysfunction), while sepsis was defined as any hospitalization with an explicit ICD-9 code for sepsis (995.91), severe sepsis (995.92), or septic shock (785.52). All definitions required that ICD-9 codes were present at the time of admission. Healthcare-associated severe sepsis was defined as any case that was admitted from a skilled nursing facility or home health, was on dialysis, or represented a readmission within 30 days of a prior discharge from the same institution. All models adjusted for age, sex, race, insurance type, intensive care unit (ICU) admission, discharge physician specialty (internal medicine vs not), All Patient Refined risk of mortality, and Elixhauser comorbidities. Odds ratios (ORs) were estimated using random-intercept logistic regression. Abbreviations: CI, confidence interval; Co-Tx, kidney-pancreas or kidney-liver co-transplant.
Table 3.
Association of Solid Organ Transplant Status With Inpatient Mortality Among Severe Sepsis Cases Admitted to the Intensive Care Unit at Any Point During Hospitalization, 2012–2014
| Population | Total No. | Inpatient Mortality Events, No. (%) |
Crude | Adjusteda |
|---|---|---|---|---|
| OR (95% CI) | OR (95% CI) | |||
| Any SOT vs non-SOT | ||||
| Non-SOT | 287 150 | 56 883 (19.8) | Ref | Ref |
| SOT | 10 600 | 1765 (16.7) | 0.80 (.76–.84) | 0.95 (.89–1.01) |
| Specific organ vs non-SOT | ||||
| Non-SOT | 287 150 | 56 883 (19.8) | Ref | Ref |
| Kidney | 4933 | 701 (14.2) | 0.68 (.63–.74) | 0.85 (.78–.93) |
| Liver | 2154 | 507 (23.5) | 1.19 (1.07–1.32) | 1.01 (.90–1.13) |
| Lung | 1577 | 279 (17.7) | 0.83 (.73–.95) | 1.22 (1.06–1.41) |
| Heart | 1098 | 175 (15.9) | 0.75 (.64–.88) | 0.99 (.83–1.19) |
| Co-transplantb | 740 | 88 (11.9) | 0.57 (.45–.71) | 0.74 (.58–.94) |
Severe sepsis defined using the Angus criteria (International Classification of Diseases, Ninth Revision [ICD-9] codes for infection and organ dysfunction), requiring that all ICD-9 codes were present at the time of admission.
Abbreviations: CI, confidence interval; OR, odds ratio; Ref, reference; SOT, solid organ transplant.
a Adjusted for age, sex, race, insurance type, discharge physician specialty (internal medicine vs not), All Patient Refined risk of mortality, and Elixhauser comorbidities. ORs estimated using random-intercept logistic regression.
b Co-transplants include hospitalizations with a prior kidney transplant in addition to a pancreas or liver transplant.
For the nonsepsis control populations, there were no statistically significant differences in mortality between SOT and non-SOT in crude analyses for heart failure and AMI patients, whereas mortality was higher for COPD patients with vs without SOT (Supplementary Table 3). After adjustment, prior SOT was not significantly associated with in-hospital mortality among any of the control populations (heart failure, AMI, or COPD). However, the odds ratio for mortality among heart failure patients was similar in magnitude and direction to the odds ratio for mortality observed among patients hospitalized for severe sepsis and sepsis.
DISCUSSION
In the current study, SOT recipients hospitalized with severe sepsis or sepsis exhibited lower mortality compared with the non-SOT population. While patients with kidney, liver transplants, or co-transplants experienced lower severe sepsis and sepsis mortality, lung transplant recipients demonstrated increased mortality. We did not identify similar mortality differences among SOT recipients hospitalized for AMI or COPD, but after adjustment for confounding factors, there was a non–statistically significant association indicating lower odds of mortality for SOT recipients hospitalized with heart failure.
A number of prior studies have addressed infection among transplant recipients, showing that individuals with prior SOT are at increased risk of infection and sepsis following transplant [5–7, 13, 30]. Kalil et al previously conducted a matched, propensity score–adjusted analysis of bacteremic sepsis patients presenting to a single tertiary care academic medical center in the United States [19]. Time-to-event analyses showed that SOT recipients had lower mortality following sepsis. In a single-center study of Staphylococcus aureus bacteremia, Malinis et al also observed lower 30-day mortality among SOT recipients [31]. We similarly observed reduced mortality for kidney, liver, and co-transplant recipients. Also consistent with prior studies, and in contrast to other transplant types, we found that lung transplant recipients had increased odds of mortality [32, 33]. However, our study expands on prior work in that we used data from a national sample of hospitals, we compared mortality across several relevant control populations, and we made use of robust risk adjustment measures.
The findings of the current study contradict the common perception that SOT recipients are at increased risk of death following sepsis. There are several potential explanations for reduced sepsis and severe sepsis mortality among SOT recipients with prior kidney, liver, and co-transplants. This population receives a number of immunosuppressive drugs including induction and maintenance therapies to prevent host rejection of the transplanted organ, and it is possible that these medications could provide some protection from the most damaging aspects of sepsis pathophysiology. Attenuation of the early hyperinflammatory phase of sepsis could protect SOT recipients by preventing early death as well as by provoking a lessened reciprocal anti-inflammatory response in the late phase of sepsis [15, 18]. This could alter the course of the sepsis event for these patients. Specifically, this could lead to improved survival by stopping progression toward major inflammatory/coagulation responses or preventing the massive loss of CD4 and CD8 T, B, and dendritic cells observed in late sepsis, ultimately reducing the risk of secondary nosocomial infection and helping to facilitate resolution of active septic foci [18]. Alternatively, it is possible that kidney, liver, and co-transplant recipients presented with more easily treatable infections (eg, urinary tract infection) or milder forms of organ dysfunction (eg, acute kidney injury) compared with the non-SOT or other transplant populations, leading to improved outcomes [34]. In contrast to kidney, liver, and co-transplant recipients, increased odds of mortality among lung recipients could be explained by presentation with more severe infections, such as pneumonia, or inability to compensate for increased oxygen demand during critical illness [32, 33, 35, 36].
Our observation of lower mortality among recipients of kidney, liver, or co-transplants could also be due to care-related factors. SOT recipients hospitalized for sepsis may benefit from attentive clinical care, including convenient access to on-call medical advice, expedited access to specialty transplant outpatient clinics, and low thresholds for hospital admission. However, when examining outcomes for hospitalizations with other time-sensitive conditions (AMI and COPD), we did not observe any differences in the odds of mortality, suggesting that increased access to specialty care would not in and of itself explain the lower mortality for sepsis. Although not statistically significant, a protective effect of SOT was present among those hospitalized for heart failure. This finding could be explained by underlying infection among these patients (with resultant care similar to those hospitalized for sepsis) or potential differences in the level of care received between SOT and non-SOT populations. Our results do not exclude earlier, more aggressive infection treatment with antibiotics and intravenous fluids—and prevention of sepsis progression—as a plausible explanation for the observed associations for kidney, liver, and co-transplant recipients. Further efforts must evaluate these important aspects of sepsis care in greater detail.
The overall sepsis mortality rates observed in this study were lower than previously reported in some studies, but they were similar to figures reported by Kalil et al [19]. This observation could be due to the characteristics of our study population. We excluded all transfers to a given hospital, a subgroup that may have included cases of higher severity. We believe this exclusion is important, as we were unable to ascertain any information pertaining to care received prior to arrival at the hospital of study. In addition, all cases were treated at academic medical center–affiliated hospitals, which may have been more attuned to current sepsis care guidelines, and could have been better poised to care for complex cases. Our mortality rate could also reflect temporal trends in hospital sepsis mortality [12].
Strengths of the current study include a large sample size, robust risk adjustment, and validated definitions. However, our results must be interpreted in light of several limitations. We used ICD-9 discharge diagnosis codes to define sepsis as well as prior organ transplant, and misclassification may be present. However, the definitions we used have been widely employed in prior studies, with the Angus definition of severe sepsis previously shown to have modest sensitivity, high specificity, and a high positive predictive value [21–23]. Although we adjusted for a number of potential confounding factors, we were not able to obtain granular clinical information related to the inflammatory response during sepsis hospitalization or factors relevant to prior transplantation. Thus, unmeasured confounding could have affected our estimates. Because we used administrative data, we were also unable to determine certain aspects of infection (eg, the source of sepsis or presence of bloodstream infection) or the appropriateness of antibiotic regimens provided, in addition to other relevant aspects of disease presentation and management.
In conclusion, using data from a national database of academic medical center–affiliated hospitals, we observed that hospitalized severe sepsis and sepsis patients with prior transplant had lower in-hospital mortality compared with those without SOT. Identifying the specific mechanisms contributing to lower mortality in this population could help to inform best practices for posttransplant care and sepsis prevention and treatment.
Supplementary Data
Supplementary materials are available at http://cid.oxfordjournals.org. Consisting of data provided by the author to benefit the reader, the posted materials are not copyedited and are the sole responsibility of the author, so questions or comments should be addressed to the author.
Notes
Acknowledgments. We thank Samuel F. Hohmann, PhD, MS-HSM, at University HealthSystem Consortium for his assistance with reviewing the article.
Author contributions. J. P. D., H. E. W., and J. W. B. conceived the study. H. E. W. and J. P. D. obtained the dataset. J. P. D. and H. E. W. carried out the analysis. J. P. D. drafted the article, and all authors contributed to its critical review and revision. H. E. W. and J. P. D. had full access to the data for this study and take responsibility for the integrity and accuracy of the data analysis.
Disclaimer. The funding sources had no role in the study's design, conduct, or reporting.
Financial support. J. P. D. received support from the Agency for Healthcare Research and Quality, Rockville, Maryland (award number T32-HS013852). H. E. W. received grant support from the National Institute for Nursing Research (award number R01-NR012726). J. E. L. received grant support from the National Institute of Diabetes and Digestive and Kidney Diseases (award number K23-DK103918). M. M. S. received grant support from the National Heart, Lung, and Blood Institute (award number K24-HL111154).
Potential conflicts of interest. R. B. M. has been a consultant for Alexion and Astellas; has received grant support from Alexion, Astellas, Bristol-Myers Squibb, and Amgen. J. W. B. reports consulting for Merck, Astellas, and Pfizer. M. M. S. and P. M. report investigator-initiated research from Amgen. All other authors report no potential conflicts. All authors have submitted the ICMJE Form for Disclosure of Potential Conflicts of Interest. Conflicts that the editors consider relevant to the content of the manuscript have been disclosed.
References
- 1.United Network for Organ Sharing. Donation and transplantation data. Available at: http://www.unos.org/donation/index.php?topic=data Accessed 16 May 2015.
- 2.Wolfe RA, Ashby VB, Milford EL et al. Comparison of mortality in all patients on dialysis, patients on dialysis awaiting transplantation, and recipients of a first cadaveric transplant. N Engl J Med 1999; 341:1725–30. [DOI] [PubMed] [Google Scholar]
- 3.Wolfe RA, McCullough KP, Schaubel DE et al. Calculating life years from transplant (LYFT): methods for kidney and kidney-pancreas candidates. Am J Transplant 2008; 8:997–1011. [DOI] [PubMed] [Google Scholar]
- 4.Abecassis M, Bartlett ST, Collins AJ et al. Kidney transplantation as primary therapy for end-stage renal disease: a National Kidney Foundation/Kidney Disease Outcomes Quality Initiative (NKF/KDOQITM) conference. Clin J Am Soc Nephrol 2008; 3:471–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Abbott KC, Oliver JD 3rd, Hypolite I et al. Hospitalizations for bacterial septicemia after renal transplantation in the United States. Am J Nephrol 2001; 21:120–7. [DOI] [PubMed] [Google Scholar]
- 6.Karuthu S, Blumberg EA. Common infections in kidney transplant recipients. Clin J Am Soc Nephrol 2012; 7:2058–70. [DOI] [PubMed] [Google Scholar]
- 7.Weikert BC, Blumberg EA. Viral infection after renal transplantation: surveillance and management. Clin J Am Soc Nephrol 2008; 3(suppl 2):S76–86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Fishman JA, Issa NC. Infection in organ transplantation: risk factors and evolving patterns of infection. Infect Dis Clin North Am 2010; 24:273–83. [DOI] [PubMed] [Google Scholar]
- 9.Angus DC, Linde-Zwirble WT, Lidicker J et al. Epidemiology of severe sepsis in the United States: analysis of incidence, outcome, and associated costs of care. Crit Care Med 2001; 29:1303–10. [DOI] [PubMed] [Google Scholar]
- 10.Wang HE, Szychowski JM, Griffin R et al. Long-term mortality after community-acquired sepsis: a longitudinal population-based cohort study. BMJ Open 2014; 4:e004283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Gaieski DF, Edwards JM, Kallan MJ, Carr BG. Benchmarking the incidence and mortality of severe sepsis in the United States. Crit Care Med 2013; 41:1167–74. [DOI] [PubMed] [Google Scholar]
- 12.Iwashyna TJ, Angus DC. Declining case fatality rates for severe sepsis: good data bring good news with ambiguous implications. JAMA 2014; 311:1295–7. [DOI] [PubMed] [Google Scholar]
- 13.Kutinova A, Woodward RS, Ricci JF, Brennan DC. The incidence and costs of sepsis and pneumonia before and after renal transplantation in the United States. Am J Transplant 2006; 6:129–39. [DOI] [PubMed] [Google Scholar]
- 14.Sarnak MJ, Jaber BL. Mortality caused by sepsis in patients with end-stage renal disease compared with the general population. Kidney Int 2000; 58:1758–64. [DOI] [PubMed] [Google Scholar]
- 15.Kalil AC, Opal SM. Sepsis in the severely immunocompromised patient. Curr Infect Dis Rep 2015; 17:487. [DOI] [PubMed] [Google Scholar]
- 16.Gea-Banacloche JC, Opal SM, Jorgensen J et al. Sepsis associated with immunosuppressive medications: an evidence-based review. Crit Care Med 2004; 32:S578–90. [DOI] [PubMed] [Google Scholar]
- 17.Opal SM, Girard TD, Ely EW. The immunopathogenesis of sepsis in elderly patients. Clin Infect Dis 2005; 41(suppl 7):S504–12. [DOI] [PubMed] [Google Scholar]
- 18.Hotchkiss RS, Monneret G, Payen D. Immunosuppression in sepsis: a novel understanding of the disorder and a new therapeutic approach. Lancet Infect Dis 2013; 13:260–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kalil AC, Syed A, Rupp ME et al. Is bacteremic sepsis associated with higher mortality in transplant recipients than in nontransplant patients? A matched case-control propensity-adjusted study. Clin Infect Dis 2015; 60:216–22. [DOI] [PubMed] [Google Scholar]
- 20.University HealthSystem Consortium. Vizient Home Page. University HealthSystem Consortium CDB. Available at: https://www.vizientinc.com/. Accessed 16 May 2015.
- 21.Wang HE, Addis DR, Donnelly JP et al. Discharge diagnoses versus medical record review in the identification of community-acquired sepsis. Crit Care 2015; 19:42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Iwashyna TJ, Odden A, Rohde J et al. Identifying patients with severe sepsis using administrative claims: patient-level validation of the Angus implementation of the international consensus conference definition of severe sepsis. Med Care 2014; 52:e39–43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Jolley RJ, Sawka KJ, Yergens DW et al. Validity of administrative data in recording sepsis: a systematic review. Crit Care 2015; 19:139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Page DB, Donnelly JP, Wang HE. Community-, healthcare-, and hospital-acquired severe sepsis hospitalizations in the University HealthSystem Consortium. Crit Care Med 2015; 43:1945–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Yale New Haven Health Services Corporation/Center for Outcomes Research and Evaluation (YNHHSC/CORE). 2015 condition-specific measures updates and specifications report hospital-level 30-day risk-standardized readmission measures. Available at: http://www.qualitynet.org/dcs/ContentServer?c=Page&pagename=QnetPublic%2FPage%2FQnetTier4&cid=1219069855841 Accessed 24 June 2015.
- 26.Pant C, Anderson MP, O'Connor JA et al. Association of Clostridium difficile infection with outcomes of hospitalized solid organ transplant recipients: results from the 2009 Nationwide Inpatient Sample database. Transpl Infect Dis 2012; 14:540–7. [DOI] [PubMed] [Google Scholar]
- 27.3M Health Information Systems 2015. Available at: http://solutions.3m.com/wps/portal/3M/en_US/Health-Information-Systems/HIS/ Accessed 16 May 2015.
- 28.Elixhauser A, Steiner C, Harris DR, Coffey RM. Comorbidity measures for use with administrative data. Med Care 1998; 36:8–27. [DOI] [PubMed] [Google Scholar]
- 29.Sutton JM, Hayes AJ, Wilson GC et al. Validation of the University HealthSystem Consortium administrative dataset: concordance and discordance with patient-level institutional data. J Surg Res 2014; 190:484–90. [DOI] [PubMed] [Google Scholar]
- 30.Tveit DJ, Hypolite IO, Poropatich RK et al. Hospitalizations for bacterial pneumonia after renal transplantation in the United States. J Nephrol 2002; 15:255–62. [PubMed] [Google Scholar]
- 31.Malinis MF, Mawhorter SD, Jain A et al. Staphylococcus aureus bacteremia in solid organ transplant recipients: evidence for improved survival when compared with nontransplant patients. Transplantation 2012; 93:1045–50. [DOI] [PubMed] [Google Scholar]
- 32.Nunley DR, Bauldoff GS, Mangino JE, Pope-Harman AL. Mortality associated with Acinetobacter baumannii infections experienced by lung transplant recipients. Lung 2010; 188:381–5. [DOI] [PubMed] [Google Scholar]
- 33.Palmer SM, Alexander BD, Sanders LL et al. Significance of blood stream infection after lung transplantation: analysis in 176 consecutive patients. Transplantation 2000; 69:2360–6. [DOI] [PubMed] [Google Scholar]
- 34.Camargo LF, Marra AR, Pignatari AC et al. Nosocomial bloodstream infections in a nationwide study: comparison between solid organ transplant patients and the general population. Transpl Infect Dis 2015; 17:308–13. [DOI] [PubMed] [Google Scholar]
- 35.Aguilar-Guisado M, Givalda J, Ussetti P et al. Pneumonia after lung transplantation in the RESITRA Cohort: a multicenter prospective study. Am J Transplant 2007; 7:1989–96. [DOI] [PubMed] [Google Scholar]
- 36.Chan KM, Allen SA. Infectious pulmonary complications in lung transplant recipients. Semin Respir Infect 2002; 17:291–302. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.