Abstract
Tumor lymphatic vessels (LV) serve as a conduit of tumor cell dissemination, due to their leaky nature and secretion of tumor-recruiting factors. Though lymphatic endothelial cells (LEC) lining the LV express distinct factors (also called lymphangiocrine factors), these factors and their roles in the tumor microenvironment are not well understood. Here we employ LEC, microvascular endothelial cells (MEC), and human umbilical vein endothelial cells (HUVEC) cultured in triple-negative MDA-MB-231 tumor-conditioned media (TCM) to determine the factors that may be secreted by various EC in the MDA-MB-231 breast tumor. These factors will serve as endothelium derived signaling molecules in the tumor microenvironment. We co-injected these EC with MDA-MB-231 breast cancer cells into animals and showed that LEC support tumor growth, HUVEC have no significant effect on tumor growth, whereas MEC suppress it. Focusing on LEC-mediated tumor growth, we discovered that TCM-treated LEC (‘tumor-educated LEC') secrete high amounts of EGF and PDGF-BB, compared to normal LEC. LEC-secreted EGF promotes tumor cell proliferation. LEC-secreted PDGF-BB induces pericyte infiltration and angiogenesis. These lymphangiocrine factors may support tumor growth in the tumor microenvironment. This study shows that LV serve a novel role in the tumor microenvironment apart from their classical role as conduits of metastasis.
The tumor microenvironment is an emerging target to treat tumor growth and metastasis1. The tumor microenvironment comprises cancer cells, extracellular matrix (ECM), and non-cancer stromal cells. The non-cancer stromal cells are more stable and targetable, compared to cancer cells, as the cancer cells experience genetic mutations and epigenetic alterations, resulting in heterogeneity and plasticity2. Importantly, these stromal cells contribute to cancer progression through their complex crosstalk with cancer cells. For example, certain types of immune cells, such as tumor-associated macrophages (TAM) accelerate tumor angiogenesis and tumor growth by overexpressing angiogenic and tumor promoting factors in the tumor microenvironment3. Some mesenchymal cell types, such as cancer-associated fibroblasts (CAF) also induce tumor growth, epithelial to mesenchymal transition (EMT), and metastasis through their secreted factors4,5.
The other important component in the tumor microenvironment is the blood and lymphatic vasculature. Blood and lymphatic vessels are formed in and around the tumor via the angiogenic and lymphangiogenic cues derived from cancer cells and stromal cells6,7. Tumor blood vessels are well recognized as key players in tumor growth, because growing tumor cells require oxygen and nutrients via the blood supply into the tumor8. More recently studies detailing the crosstalk between cancer cells and blood endothelial cells (BEC) via the BEC-secreted proteins (also called angiocrine factors)9,10,11,12,13,14,15,16 have helped to change the perspective on the role of the blood vasculature from that of a passive conduit to a more inductive role.
Lymphatic vessels (LV) in the tumor microenvironment are primarily considered as a route of tumor dissemination17. LV are leaky compared to blood vessels, because they are only sparsely covered with pericytes and smooth muscle cells18. Thus in certain types of cancers such as in breast cancer, metastasis occurs preferentially through the lymphatic vasculature compared to the blood vessel mediated spreading19,20. LEC show distinct gene expression profiles from BEC21. However, LEC-secreted proteins (also called lymphangiocrine factors) in tumor microenvironment and their crosstalk with cancer cells are poorly understood compared to BEC-derived angiocrine factors. Thus an understanding of the lymphangiocrine factors would add to the current understanding of the role of the LV in lymphatic dissemination of tumor cells22.
We previously reported on pro-metastatic crosstalk between breast cancer cells and LEC. We showed that LEC within pre-metastatic lungs and lymph nodes are enhanced and conditioned by triple-negative breast cancer (TNBC) cell secretions and promote spontaneous metastases within one month23. Here we investigate roles of LEC in the tumor microenvironment in primary tumor growth and pericyte recruitment by employing in vitro tumor-educated LEC models and in vivo LEC-included breast tumor xenograft and matrigel plug models. This study shows for the first time that LEC can support tumor growth through lymphangiocrine factors.
Results
LEC within tumor microenvironment promote tumor growth
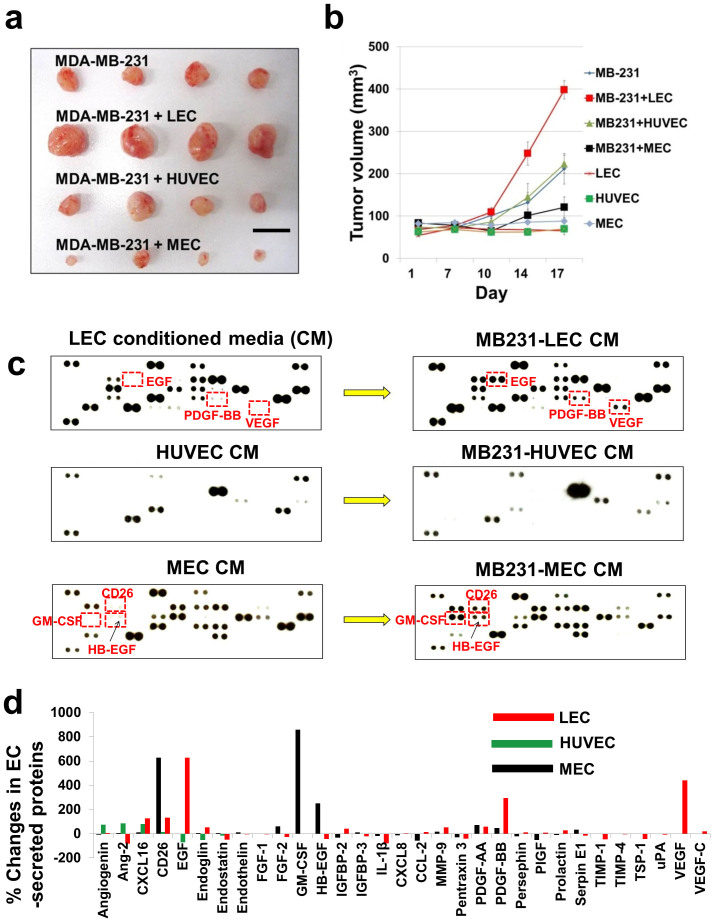
We co-injected EC (LEC, MEC, and HUVEC) with MDA-MB-231 breast cancer cells into animals. In a conventional MDA-MB-231 (MB231) breast tumor xenograft model, two million MB231 cancer cells are orthotopically inoculated with 50% matrigel supplement. In this study, however, the conventional xenograft model was modified as follows: the number of cancer cells was decreased to 300,000 MB231 cells; a large number of LEC or MEC or HUVEC (2,000,000 cells) were included with the cancer cells, consistent with the procedures employed in several previous studies24,25,26,27. Matrigel was not added in the tumor/EC mixture to remove matrigel effects: in general, matrigel significantly promotes primary tumor establishment and growth28. This new xenograft model slows down tumor growth rates, compared to the normal xenograft model; at the same time it gives a better understanding of roles of EC in tumor growth. There were seven experimental arms: 300,000 MB231 only (N = 8); 300,000 MB231 + 2,000,000 LEC (N = 8); 300,000 MB231 + 2,000,000 MEC (N = 8); 300,000 MB231 + 2,000,000 HUVEC (N = 8); 2,000,000 LEC only (N = 2); 2,000,000 MEC only (N = 2); and 2,000,000 HUVEC only (N = 2). Total volume of each injection was identical. LEC-included MB231 tumors showed dramatic increases in the tumor growth rate, compared to tumors containing only MB231 cells. Interestingly, MEC-included MB231 tumor showed suppression of tumor growth. HUVEC-included MB231 tumors did not show any difference compared to MB231 tumors (Fig. 1a,b).
Figure 1. LEC promote tumor growth and show distinctive secretomes under TCM condition.
(a) Two million of LEC, HUVEC, or MEC were pre-mixed with 300,000 MB231 cells respectively, after which the cell mixtures were orthotopically inoculated into the thoracic part (2nd mammary site from the top) of athymic nude mice without supplementing with matrigel (N = 8). For control, just 300,000 MB231 (N = 8) or just 2,000,000 EC (LEC; MEC; HUVEC, N = 2 each EC) were inoculated. Representative tumors in each group, scale bar represents 10 mm. (b) Tumor growth curve. (c) Fifty five angiogenesis-related factors from tumor-educated LEC/MEC/HUVEC (MB231-LEC/MB231-MEC/MB231-HUVEC) were examined by using reverse western arrays. Factors secreted from LEC, MEC, and HUVEC before and after incubating them in 30% TCM were analyzed and compared. MB231-LEC shows high expression of EGF, VEGF and PDGF-BB, compared to normal LEC. MB231-MEC shows high expression in CD26, HB-EGF and GM-CSF, compared to normal MEC. HUVEC does not show dramatic changes. (d) % Changes in EC secreted proteins. Average pixel density of the pair of duplicate spots was determined by using the ImageJ. Expression level changes more than 200% after TCM treatment were considered as significant. Data (b) are reported as mean ± s.e.m.
LEC show distinctive secretomes after tumor education with TCM
Having the results shown in Figure 1a and 1b, we hypothesized that LEC, MEC and HUVEC would secrete different factors in response to MB231 tumor-conditioned media (TCM) and these factors could differentially affect tumor growth. To test this hypothesis, tumor-educated LEC (LEC cultured in TCM, also called MB231-LEC), tumor-educated MEC (MB231-MEC), and tumor-educated HUVEC (MB231-HUVEC) were prepared by the process described in the Supplementary Fig. S1 online. The tumor-educated conditioned media from these tumor-educated EC were compared with normal conditioned media obtained from normal LEC, MEC, and HUVEC to show how the TCM affects the various endothelial secretomes (Fig. 1c,d). A Proteome Profiler Antibody Arrays Kit for Human Angiogenesis (R&D systems, Minneapolis, MN) was used to determine 55 angiogenesis-related factors in these conditioned media (Fig. 1c). Average pixel number of the pair of duplicate spots was determined and the % changes in protein levels were plotted. Changes in protein levels more than 200% between normal and tumor-induced groups were considered to be significant. After TCM education, LEC showed large increases in EGF (>608%), PDGF-BB (>281%), and VEGF (>600%); MEC showed large increases in GM-CSF (>810%), CD26 (>580%), and HB-EGF (210%); however HUVEC did not show any dramatic changes (<200%) (Fig. 1d). As LEC have shown enhanced tumor growth (Fig. 1a,b), we focused on LEC secretome (EGF, PDGF-BB, and VEGF). A previous experiment with the same antibody array showed that MB231 TCM contains Angiogenin, CXCL16, Endothelin-1, HB-EGF, IL-8, MMP-8, MMP-9, PDGF-AA, PlGF, and VEGF29. Hence, we excluded VEGF in the LEC secretome and further studied EGF and PDGF-BB, the factors that are not expressed by MB231 cells.
MB231-LEC express high amounts of EGF which induces cancer cell proliferation
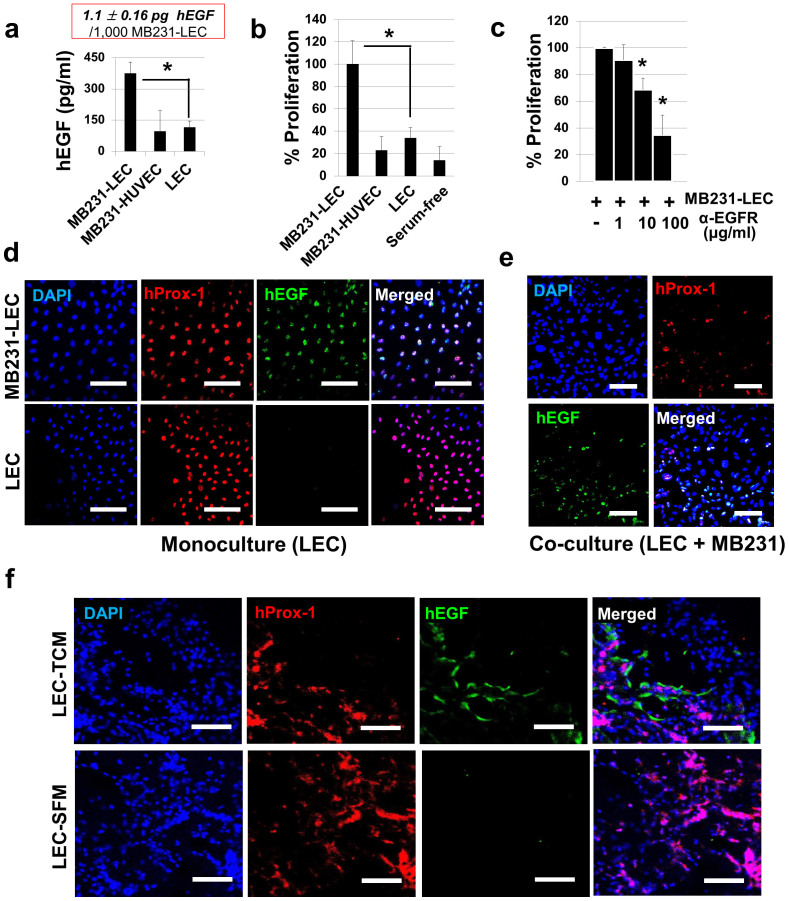
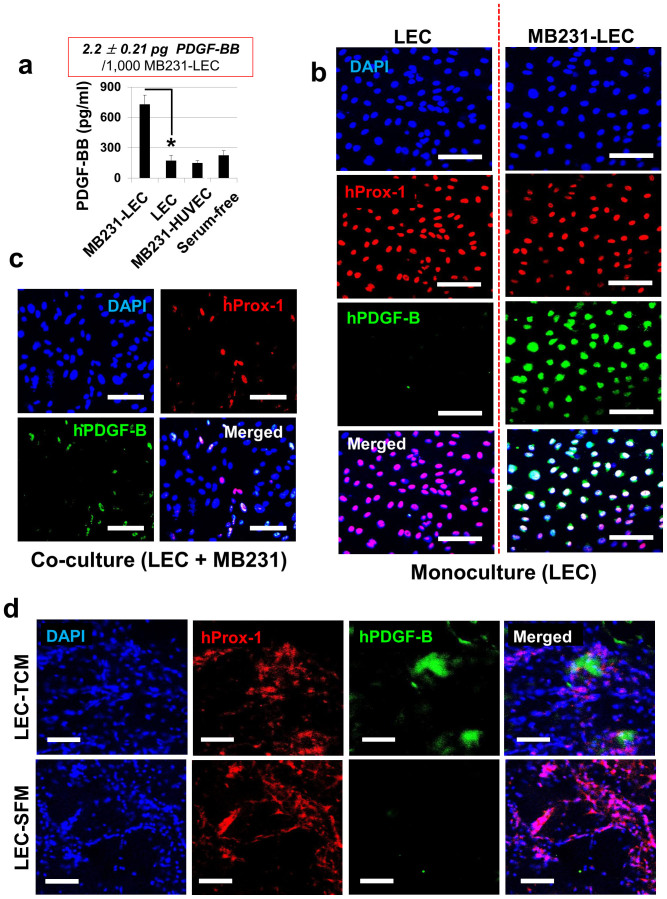
An ELISA for human EGF was performed with MB231-LEC, MB231-HUVEC, and normal LEC conditioned media (Fig. 2a). hEGF was highly expressed in MB231-LEC compared to other groups (Fig. 2a). We estimated that 1,000 MB231-LEC cells expressed 1.1 ± 0.16 pg hEGF. MB231 cell proliferation was assessed in MB231-LEC conditioned media (MB231-LEC CM), MB231-HUVEC CM, normal LEC CM, and serum-free media (Fig. 2b). Only the MB231-LEC CM potently promoted MB231 cell proliferation (Fig. 2b). Next, MB231 cell proliferation in the MB231-LEC CM was assessed with anti-EGFR neutralizing antibodies (Fig. 2c). Anti-EGFR antibodies blocked MB231-LEC induced MB231 cell proliferation (Fig. 2c). Confluent LEC were treated with control media (30% SFM + 70% EGM) or 30% TCM media (30% TCM + 70% EGM) overnight (Fig. 2d). When LEC were incubated with 30% TCM, LEC expressed hEGF, however, normal LEC in the normal media did not express hEGF (Fig. 2d). We further studied hEGF expression in LEC in a LEC/MB231 co-culture model (Fig. 2e). We mixed LEC and MB231 (1:1 ratio) in 50% EGM and 50% RPMI-1640 media and make the cell adhere on the bottom of plates overnight. We then replaced the co-media with serum-free media and incubated cells overnight. We fixed the cells and stained them with antibodies for hProx-1 (LEC marker) and hEGF (Fig. 2e). In the co-culture condition, the Prox-1 positive LEC were colocalized with hEGF, demonstrating lymphatic expression of hEGF in tumor/LEC co-culture models (Fig. 2e). Mice with LEC-included matrigel plugs were treated with TCM or SFM for 10 days as described in Supplementary Fig. S2 online. The excised gel plugs were analyzed with anti-hProx-1 and anti-hEGF antibodies. Colocalization of hProx-1 and hEGF only in the TCM-treated LEC/matrigel plugs, not in the SFM-treated group, suggests that LEC secrete EGF when exposed to tumor-secreted factors (Fig. 2f).
Figure 2. MB231-LEC express EGF and induce cancer cell proliferation.
(a) ELISA for human EGF on MB231-LEC, MB231-HUVEC, and normal LEC conditioned media. MB231-LEC express high amount of EGF compared to normal LEC (*P = 0.042). We estimate that 1,000 MB231-LEC cells expressed 1.1 ± 0.16 pg hEGF. (b) MB231 cell proliferation was assessed in MB231-LEC CM, MB231-HUVEC CM, normal LEC CM, and serum-free media. MB231-LEC CM significantly promotes MB231 cell proliferation, compared to normal LEC CM (*P = 0.040). (c) MB231 cell proliferation induced by MB231-LEC is blocked by using anti-EGFR antibodies (*P < 0.05). (d) Normal LEC in normal EGM media (‘LEC') or LEC in TCM (‘MB231-LEC') show distinct hEGF expression. Normal LEC do not express hEGF, but TCM-conditioned LEC express hEGF. Scale bars represent 100 μm. (e) LEC and MB231 were cultured in the same plate, showing the LEC/MB231 co-culture effects. In the co-culture condition, LEC start to express hEGF, however MB231 do not show hEGF expression. Scale bars represent 100 μm. (f) LEC-included matrigel plugs from TCM or SFM-treated athymic nude mice were analyzed 10 days after matrigel implantation. Human Prox-1 positive LEC in the matrigel plugs express hEGF when the matrigel-implanted animals are treated with TCM (‘LEC-TCM' group). LEC from SFM-treated animals do not express hEGF (‘LEC-SFM' group). Scale bars represent 100 μm. Data (a,b,c) are reported as mean ± s.e.m.
LEC are associated with tumor cell proliferation
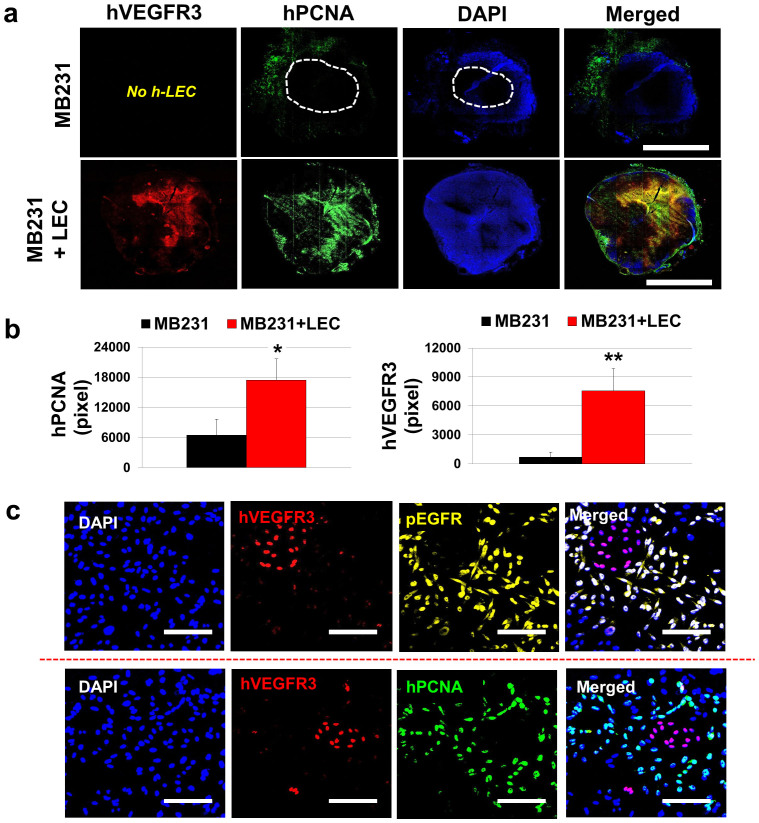
Upon the discovery that EGF expressed by MB231-LEC induces MB231 cancer cell proliferation (Fig. 2), we analyzed LEC-included MB231 tumors and normal MB231 tumors by immunostaining them with anti-human VEGFR3 (a lymphatic marker) and anti-human PCNA (proliferating-cell nuclear antigen) antibodies to detect human LEC and proliferating tumor cells (Fig. 3a). We observed that LEC-included tumors had more prevalent human PCNA-positive cells around hVEGFR3-positive LEC areas, compared to normal MB231 tumors. However, normal MB231 tumors showed less PCNA-positive cells in the intratumoral area, and the proliferating cells were mostly detected in the peritumoral areas (Fig. 3a,b). The results suggest that LEC may facilitate tumor cell proliferation in the tumor microenvironment. To confirm the proliferating human cells are not LEC, but MB231 cells, we further observed the LEC-included MB231 tumor sample in a high resolution (Fig. 3c). Human VEGFR3-positive LEC (red) were not colocalized with pEGFR (yellow, pseudo-colored) and hPCNA (green), but other human cells adjacent to LEC (supposed to be MB231 cancer cells) showed pEGFR and hPCNA signals (Fig. 3c). This demonstrates that LEC-secreted EGF (Fig. 2) can phosphorylate EGFR in the LEC-adjacent MB231 cells and induce MB231 cell proliferation however autocrine effects of the LEC-secreted EGF in LEC proliferation was not detected (Fig. 3c).
Figure 3. LEC support MB231 cell proliferation in tumor xenografts.
(a) Two million LEC were pre-mixed with 300,000 MB231 cells, then, the cell mixtures were orthotopically inoculated into the thoracic mammary fat pad. For control, just 300,000 MB231 were inoculated. Human PCNA positive area representing proliferating human cells were colocalized with LEC in the LEC-included tumors, suggesting that LEC may support tumor cell proliferation. Scale bars represent 4 mm. (b) Quantification of hPCNA (*P = 0.038) and hVEGFR3 (**P = 0.008) in the tumor samples. Twelve randomly selected 10× images (per group) were obtained and the labeled antigens were quantified by using the ImageJ. (c) Proliferating human cells might be either human MB231 or human LEC. Further observation of the LEC-included tumor in a high resolution confirms that the proliferating human cells are human MB231 cells, not the human LEC. Human VEGFR3-positive LEC (red) were not colocalized with pEGFR (yellow, pseudo-colored) and hPCNA (green), but other human cells adjacent to LEC that are supposed to be human MB231 cells, show pEGFR and PCNA signals. Scale bars represent 100 μm. Data (b) are reported as mean ± s.e.m.
MB231-LEC express high amounts of PDGF-BB and recruit pericytes in vivo
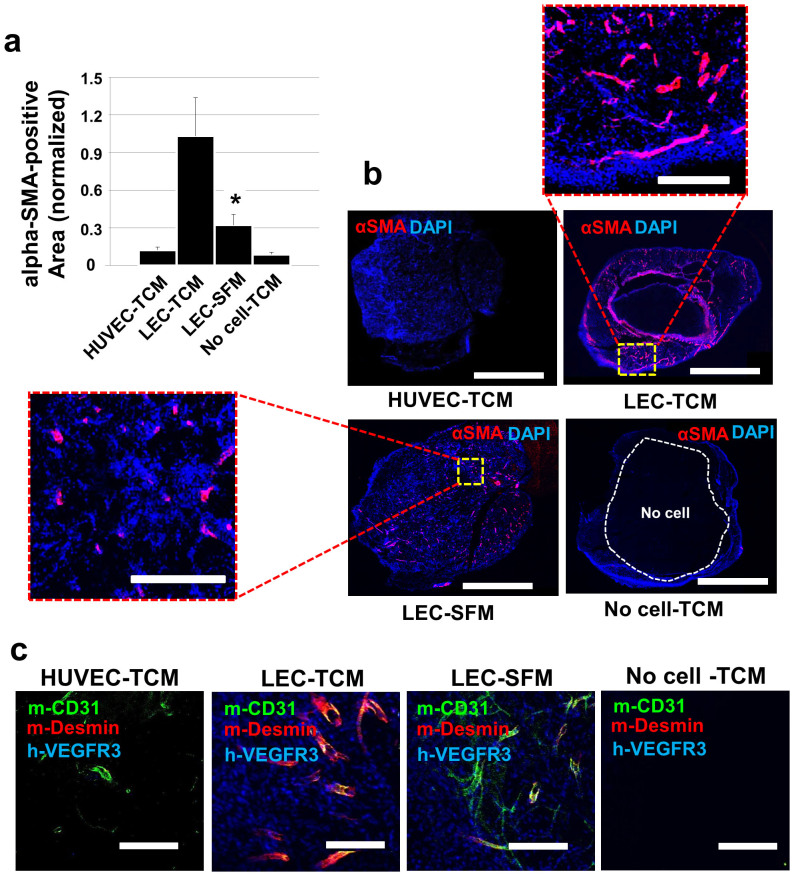
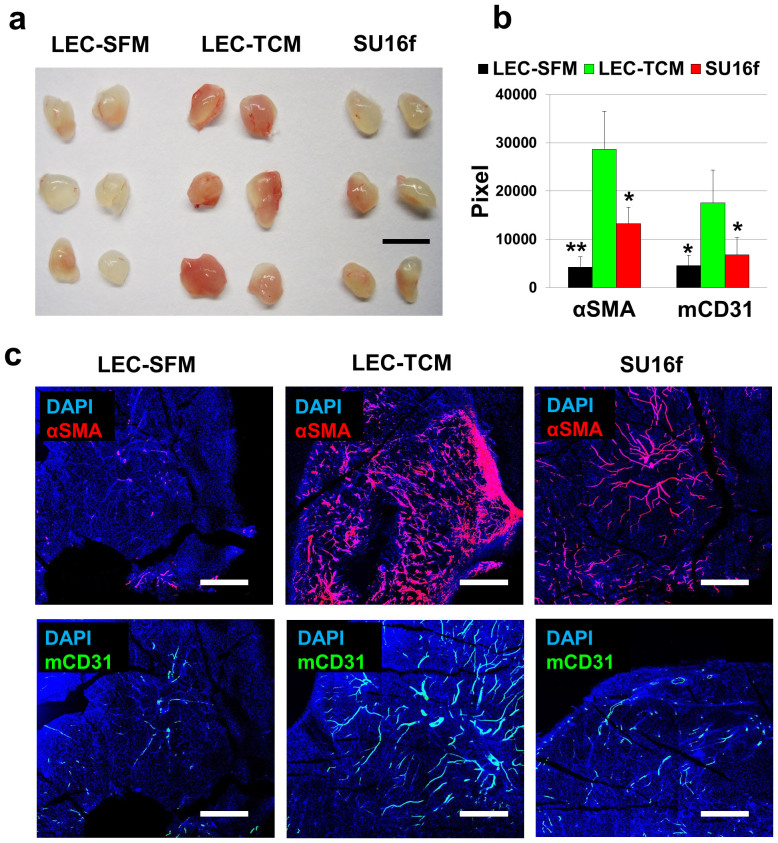
Two million LEC or HUVEC included matrigels were implanted in athymic nude mice and the mice were subcutaneously treated with 50 μl TCM or SFM for 10 days. A no-cell group was included as a control group. Ten days after the matrigel implantation, the plugs were excised and analyzed with anti-αSMA (alpha smooth muscle actin) and anti-desmin antibodies, as αSMA and desmin are markers for pericytes30,31. LEC-included matrigel from TCM treated animals (‘LEC-TCM' group) showed dramatic recruitment of αSMA-positive cells whereas HUVEC-TCM, LEC-SFM and No cell-TCM groups showed fewer αSMA-positive cells (Fig. 4a,b). Anti-desmin (red) and anti-mCD31 (green) antibodies were employed to assess the pericyte coverage on the blood vessels (Fig. 4c). Anti-hVEGFR3 antibodies (blue) were also used to detect human LEC initially loaded in the gel plugs. The pericyte coverage was more frequently observed in LEC-matrigel plugs from mice that had been treated with TCM than other groups, including ‘HUVEC-TCM', ‘LEC-SFM', and ‘No cell-TCM' groups (Fig. 4c). We also demonstrated that anti-αSMA and anti-desmin antibodies are not immunoreactive with human VEGFR3-positive LEC (blue) or mouse CD31-positive BEC (green) in the LEC-SFM control, suggesting that the anti-αSMA and anti-desmin antibodies are pericyte specific (Supplementary Fig. S3 online).
Figure 4. MB231-LEC recruit pericytes in vivo.
High-concentrate matrigel containing LEC or HUVEC (2 × 106/gel) and heparin (10 units/gel) was injected subcutaneously. TCM or SFM (50 μl/injection) was subcutaneously dosed daily for 10 days, the mice were euthanized, and the gel plugs were excised and analyzed. (a) Gel plugs were sectioned and probed with anti-alpha-SMA (αSMA) antibodies. αSMA positive cells per 12 randomly selected areas were quantified by using ImageJ. The LEC-TCM group shows significant infiltration of the αSMA positive cells, compared to the LEC-SFM group (*P = 0.031). (b) Representative confocal images for αSMA-positive area in four different groups of plugs (HUVEC-TCM, LEC-TCM, LEC-SFM, No cell-TCM). Scale bars in the whole images and in the enlarged images represent 5,000 μm and 1,000 μm, respectively. (c) To confirm the pericyte infiltration and blood vessel coverages by the pericytes, four different plug samples were co-stained with anti-desmin and anti-mCD31 antibodies. LEC-included matrigel plugs in animals treated with TCM showed higher coverage of the blood vessels by pericytes, compared to TCM-treated HUVEC, SFM-treated LEC, and TCM-treated no cell groups. Anti-hVEGFR3 antibodies (blue) were used to detect human LEC that are included in the gel plugs. Only LEC-TCM and LEC-SFM group showed LEC. Scale bars represent 200 μm. Data (a) are reported as mean ± s.e.m.
PDGF-BB in MB231-LEC conditioned media (MB231-LEC CM) was measured by ELISAs, comparing it with normal LEC CM, MB231-HUVEC CM, and serum-free media (Fig. 5a). MB231-LEC express high amounts of PDGF-BB compared to the other groups (Fig. 5a). We estimated that 1,000 MB231-LEC cells expressed 2.2 ± 0.21 pg PDGF-BB. Angiogenesis reverse array data (Fig. 1) showed that EGF increased around 600%, PDGF-BB increased around 280% in MB231-LEC, compared to normal LEC. As the change in PDGF-BB was relatively smaller than EGF, we additionally performed quantitative RT-PCR for gene expression of hPDGFB in LEC to account for the protein expression data. Approximately 3.4 times more enhanced hPDGFB gene expression was observed in MB231-LEC (Supplementary Fig. S4 online). Confluent LEC were treated with control media (30% SFM + 70% EGM) or 30% TCM media (30% TCM + 70% EGM) overnight (Fig. 5b). When only LEC were treated with TCM, LEC expressed hPDGF-B (Fig. 5b). In LEC/MB231 co-culture model, we also showed that hProx-1 positive LEC (red) express hPDGF-B (green) (Fig. 5c). Mice with LEC-included matrigel plugs were treated with TCM or SFM. LEC-included matrigel from TCM treated animals (‘LEC-TCM') showed human PDGF-B expression around human LEC (hProx-1), compared to ‘LEC-SFM' matrigel plugs, demonstrating that tumor-educated LEC express hPDGF-B in vivo (Fig. 5d).
Figure 5. MB231-LEC express PDGF-BB.
(a) ELISAs for PDGF-BB on MB231-LEC conditioned media, normal LEC CM, MB231-HUVEC CM, and serum-free media (SFM). MB231-LEC express high amount of PDGF-BB, compared to normal LEC (*P = 0.026). We estimate that 1,000 MB231-LEC cells expressed 2.2 ± 0.21 pg hPDGF-BB. (b) Normal LEC in normal EGM media (‘LEC') or LEC in TCM (‘MB231-LEC') show distinct hPDGF-B expression. Normal LEC do not express PDGF-B, but TCM conditioned LEC express PDGF-B. Scale bars represent 100 μm. (c) LEC and MB231 were co-cultured in a same plate. In the co-culture condition, LEC start to express PDGF-B, however MB231 do not show PDGF-B expression. Scale bars represent 100 μm. (d) LEC-included matrigel plug assay. Matrigel containing LEC (2 × 106/gel) and heparin (10 units/gel) was injected subcutaneously. TCM or SFM (50 μl/injection) was subcutaneously dosed daily for 10 days, the mice were euthanized, and the gel plugs were excised and analyzed. Human Prox-1 positive hLEC in the matrigel plugs express human PDGF-B (hPDGF-B) when the animals are treated with TCM (‘LEC-TCM' group). hLEC from SFM-treated animals do not express PDGF-B (‘LEC-SFM' group). Scale bars represent 100 μm. Data (a) are reported as mean ± s.e.m.
SU16f, a PDGFRβ inhibitor blocks MB231-LEC induced pericyte recruitment and angiogenesis
We further performed LEC-included matrigel plug assays with a PDGFRβ inhibitor to address the role of LEC-derived PDGF-BB in the model (Fig. 6). We employed SU16f, a PDGFRβ inhibitor. SU16f selectively inhibits PDGFRβ over VEGFR2, FGFR1, and EGFR more than 14-fold, 229-fold and 10000-fold, respectively. This information was provided by Tocris Biosciences, a provider of the SU16f (Catalog No, 3304). Nine animals with LEC-included matrigel plugs (two plugs per mouse) were treated with SFM (3 mice) or TCM (3 mice) or TCM + SU16f (3 mice). Fifty microliter of SFM or TCM were administered subcutaneously every day; 10 mg/kg SU16f were additionally administered orally every day for the TCM + SU16f group. After 10 days, matrigel plugs were excised and analyzed. Macroscopic images for the gel plugs indicated that the LEC-TCM group experienced more intra-gel angiogenesis compared to other groups (Fig. 6a). We stained the plugs with anti-αSMA and anti-mCD31 antibodies to detect pericytes and blood vessels (Fig. 6b,c). The LEC-TCM group showed enhanced pericyte recruitment, and angiogenesis, compared to LEC-SFM (Fig. 6b). SU16f treatment significantly inhibits LEC-TCM induced pericyte infiltration and angiogenesis (Fig. 6b,c). The inhibition of PDGFRβ upon TCM induction by using SU16f and the decreases in pericyte recruitment and angiogenesis demonstrate that MB231-LEC secreted PDGF-BB induce pericyte recruitment and angiogenesis via the PDGFRβ signaling pathway.
Figure 6. SU16f, a PDGFRβ inhibitor blocks MB231-LEC induced pericyte recruitment and angiogenesis in vivo.
Evaluating roles of LEC-expressed PDGF-BB in pericyte recruitment and angiogenesis, SU16f, a selective PDGFRβ inhibitor was employed. There were three groups: ‘LEC-SFM' group (LEC-matrigel injection and SFM treatment for 10 days); ‘LEC-TCM' group (LEC-matrigel injection and TCM treatment for 10 days); and ‘LEC-TCM-SU16f' group (LEC-matrigel injection and TCM/SU16f co-treatment for 10 days). TCM and SFM were injected subcutaneously. SU16f was dissolved in 10% DMSO solution (PBS base) and administered orally (10 mg/kg/day). Two control groups (‘LEC-TCM' and ‘LEC-SFM' groups) were also orally treated with vehicle (10% DMSO in PBS). (a) Macroscopic images of the gel plugs. Scale bar represents 10 mm. (b) Pericyte recruitment (αSMA positive cells, *P = 0.046; **P = 0.0067) and angiogenesis (mCD31, *P < 0.05) in the plugs. Six randomly selected 10× images from each gel plug were analyzed. Four gels from each group, giving total 24 images from each group, were used for the quantification. (c) Representative confocal images for αSMA- or mCD31-positive areas in the plugs. Scale bars represent 1,000 μm. Data (b) are reported as mean ± s.e.m.
Discussion
The presence of LEC, a component of lymphatic endothelium in tumor microenvironment is a result of tumor lymphangiogenesis. Tumor lymphangiogenesis is driven by tumor cell-secreted lymphangiogenic growth factors such as VEGFC and VEGFD32. It is well established that lymphatic vasculatures in the tumor microenvironment serve as routes of tumor dissemination because of that lymphatic vessels are much more permeable compared to blood vessels33. We add a new concept of the important role of LEC-secreted proteins (also called lymphangiocrine factors) and their signals in tumor microenvironment to the classical understanding of tumor lymphangiogenesis. We discovered that tumor-associated (or tumor-educated) LEC express EGF and PDGF-BB and influence the tumor microenvironment. We employed LEC-included MB231 tumor xenograft models with 2,000,000 LEC and 300,000 MB231 cells (Fig. 1a,b). We have designed our in vivo model based on previous studies24,25,26,27. As primary human endothelial cells do not survive for a long time after subcutaneous injection in a mouse model, many of previous studies have employed a high number of endothelial cells. For example, Franses et al. injected 1,000,000 EC adjacent to 12 day old primary lung tumors24. Neiva et al. co-injected EC and cancer cells with the 900,000:100,000 ratios into mice25. Other stromal cells have also been co-injected with cancer cells. Suzuki et al co-injected mesenchymal stem cells (MSC) and cancer cells with the 2,500,000:500,000 (5:1) ratios26. Fridman et al. summarized very diverse ratios between cancer cell (250,000 to 4,000,000) and stromal cells, including EC, fibroblasts, MSC, bone marrow derived stromal cells (74,000 to 5,000,000) for the co-injection models27. Using our breast tumor xenograft model, we showed that LEC-included MB231 tumors exhibit a rapid tumor growth rate compared to normal MB231 tumors or MEC/HUVEC-included MB231 tumors (Fig. 1a,b). This might be related to LEC-secreted EGF (Fig. 2) and its enhancement of tumor cell proliferation (Fig. 3). The role of EGF in tumor cell proliferation has been well studied in many types of cancer using experimental and computational approaches34,35. The role of the host (mouse) LEC in EGF-driven tumor growth versus the injected hLEC could be assessed by the application of anti-human EGF or anti-mouse EGF antibodies which would specifically block hEGF or mEGF. These experiments would address the possible clinical relevance of LEC-expressed EGF in patients. Moreover, the mechanisms of EGF expression in tumor-educated LEC need to be further elucidated. Although many of studies have previously focused on the mechanism of EGF expression in cancer cells36, studies to determine the mechanism of EGF secretion by LEC have not been done.
MB231-LEC also express PDGF-BB and may influence tumor microenvironment (Fig. 4,5,6). PDGF-BB is involved in tumor growth, pericyte coverage, angiogenesis, and tumor drug resistance37,38,39. Increased pericyte infiltration and angiogenesis by MB231-LEC (Fig. 4) was observed, and this can be explained by LEC-secreted PDGF-BB (Fig. 5), and the inhibition of the PDGFRβ signaling pathway with SU16f (a PDGFRβ inhibitor) (Fig. 6). Animals implanted with LEC-included matrigel were treated with ‘SFM' or ‘TCM' or ‘TCM + SU16f'. The results demonstrated that LEC-secreted PDGF-BB mediates pericyte infiltration in vivo through the PDGFRβ signaling pathway (Fig. 6b). Interestingly, MB231-LEC also induced angiogenesis, and this was blocked by SU16f treatment (Fig. 6). mCD31 positive blood endothelial cells were less detected in SU16f treated group, compared to LEC-TCM group (Fig. 6c). Though it is known that the PDGF-BB/PDGFRβ signaling pathway can induce tumor angiogenesis39, it needs to be further investigated whether PDGF-BB and other PDGF receptors (PDGFRαα, PDGFRαβ, and PDGFRββ) can induce angiogenesis. Among all the PDGF family, including PDGF-AA, PDGF-BB, PDGF-AB, PDGF-CC, and PDGF-DD, PDGF-BB is the only ligand can activates all three PDGF receptors, including PDGFRαα, PDGFRαβ, and PDGFRββ40. In applications other than cancer, in the field of regenerative medicine it is important to make mature blood vessels in transplanted tissues41. Pericyte coverage is one criterion that determines whether the transplanted tissues are successfully grafted in the host42. In the present study, LEC-included matrigel plugs in animals that were treated with TCM showed pericyte infiltration and coverage on the blood vessels (Fig. 4c). In this sense, PDGF-BB expressing LEC could be an attractive source of growth factors in tissue engineering models, if the key factors in TCM inducing PDGF-BB are determined.
In normal MB231 tumors, the stromal pericytes around blood vessels appear to undergo natural apoptosis in vivo (Supplementary Fig. S5 online). Normal MB231 tumor xenografts were established and the excised tumors were analyzed using antibodies for cleaved caspase 3 (CC-3), mLYVE-1, mCD31, and lectin. Interestingly, most apoptosis-experiencing cells were located around blood vessels and not around lymphatic vessels (Supplementary Fig. S5 online). More precisely, antibodies for CC-3, alpha-smooth muscle actin (αSMA), and lectin were used to detect apoptosis-experiencing cells, pericytes, and blood vessels. The apoptosis-experiencing cells were αSMA positive pericytes residing around blood vessels (Supplementary Fig. S5 online). One possible explanation could be that tumor angiogenesis would be so active because of the over-expressed levels of angiogenic factors such as VEGFA that the rate of blood vessel sprouting would be too high for pericytes to keep up and cover the blood vessels so as to allow them to mature. This hypothesis would also require that the pericytes that fail to cover blood vessels be more vulnerable to apoptotic signals, compared to pericytes fully associated with the blood endothelium. Another possible explanation for the apoptosis of pericytes is that the tumor microenvironment contains apoptotic molecules. This idea is quite attractive because death of pericytes is closely related to blood vessel mediated metastasis43,44 and tumors may set the balance between adequate perfusion and metastasis by differentially regulating pericyte apoptosis. These hypotheses need to be further tested. However, regardless of mechanisms, the data showing apoptosis-experiencing pericytes suggest that PDGF-BB secreted by tumor-educated LEC (MB231-LEC) may influence pericyte recruitment and maintenance in the tumor microenvironment.
Interestingly, MEC-included MB231 tumors showed suppression of tumor growth. Currently, the role of blood endothelial cells (BEC) in tumor growth is quite controversial. It has been reported that BEC block tumor growth24. In that study, perlecan from BEC muted the proliferative and invasive phenotype of lung and breast cancer cells in vitro. However, perlecan silencing in BEC significantly increased IL6 secretion in BEC, eliminating the ability of the BEC to inhibit cancer cell invasiveness. In contrast, other studies have reported that BEC induce tumor growth via cancer stem cell related mechanisms and tumorigenic crosstalk with tumor microenvironment14,45,46. In the present MB231 model, HUVEC did not affect tumor growth, but MEC suppressed tumor growth, suggesting that different BEC lines could result in different phenotypes. Interestingly, MEC has a distinct secretome; CD26, GM-CSF, and HB-EGF are highly expressed. HB-EGF is also highly expressed by MB231 cells as described in our previous study29, so the contribution of the HB-EGF secreted by MEC to tumor growth is likely to be minimal. CD26 and GM-CSF have been linked to tumor inhibitory mechanisms. CD26 (also called DPPIV) inhibitors extended GLP-2 mediated tumor promoting effects on intestinal cancer cells47. CD26 inhibited the malignant phenotype of prostate cancer cells by blocking the basic fibroblast growth factor signaling pathway48. GM-CSF inhibited breast cancer growth and metastasis by invoking an anti-angiogenic program in tumor-educated macrophages49. GM-CSF also involved in anti-tumor immunity50,51. Therefore, the role of MEC secreted factors in tumor growth needs to be further investigated.
In summary, lymphangiocrine factors derived from LEC may support tumor growth by promoting tumor cell proliferation, pericyte infiltration, and angiogenesis whereas MEC promote tumor suppression. This study shows for the first time the diverse roles that different EC can play in the breast tumor microenvironment.
Methods
Cell culture
Human umbilical vein endothelial cells (HUVEC), lymphatic endothelial cells (LEC), and microvascular endothelial cells (MEC) were purchased from Lonza, and grown in EGM-2 (HUVEC) and EGM-2MV (LEC and MEC). MDA-MB-231 breast cancer cells were provided by Dr. Zaver Bhujwalla (JHMI, Radiology and Oncology). MDA-MB-231 cells were propagated in RPMI-1640 medium supplemented with 10% FBS and 1% penicillin/streptomycin (Sigma).
Conditioned media
When MDA-MB-231 cells reached confluence in T175 tissue culture flasks, the normal growth media was replaced with 8 ml of serum-free media (SFM). After 24 h incubation, the supernatant was centrifuged and filtered through 0.2 μm syringe filters (Corning). The resulting tumor-conditioned media (TCM) was stored in aliquots at −80°C. When LEC/HUVEC/MEC reached 30% confluence in T75 tissue culture flasks, the growth media (EGM) was replaced with 30% TCM (TCM:EGM = 3:7) to allow the TCM to educate the LEC/HUVEC/MEC. LEC/HUVEC/MEC were allowed to grow in the media for 4 days then the media was replaced with 3 ml SFM containing 2% FBS (not supplemented with bullet kit). After 48 h, the supernatant was centrifuged and filtered. The resulting tumor-educated LEC/HUVEC/MEC conditioned media (MB231-LEC or MB231-HUVEC or MB231-MEC) was stored in aliquots at −80°C to avoid multiple freeze thaws.
LEC-included tumor xenograft models
To evaluate roles of the various endothelial cells (EC) in the tumor microenvironment, a relatively small number of MDA-MB-231 cells (3 × 105/mouse, 100 μl of RPMI-1640 complete media with no matrigel) were orthotopically inoculated into the thoracic mammary fat pad of the athymic nude mice (female, 5–6 weeks) with or without two million LEC, MEC, or HUVEC. This system was used to maximize the contribution of EC and to evaluate the roles of these EC in tumor growth and the tumor microenvironment. The tumor size was measured by using a caliper, and the volume was calculated, using the formula: V = 0.52 × (tumor length) × (tumor width)2.
Reverse western blot assays
For reverse western blot, Proteome Profiler Antibody Array Kits for human angiogenesis factors (R&D Systems) was used, according to the manufacturer's instructions. Factors present in LEC, MEC, or HUVEC conditioned media were compared with those in conditioned media from tumor-educated LEC (MB231-LEC), MB231-MEC, or MB231-HUVEC. Average signal (pixel density) of the pair of duplicate spots was determined using ImageJ. Expression level changes more than 200% after TCM treatment were considered as a significant perturbation. We further studied these highly changed target molecules (e.g., EGF and PDGF-BB in LEC).
LEC-included matrigel plug assay
High-concentrate matrigel (500 μl, BD Biosciences) containing LEC or HUVEC (2 × 106/gel) and heparin (10 units/gel) was injected subcutaneously on the ventral side of both flanks of athymic nude mice. TCM or SFM (50 μl/injection) was subcutaneously administered daily for 10 days, the mice were euthanized, and the gel plugs were excised and analyzed. Evaluating role of LEC-expressed PDGF-BB in pericyte recruitment and angiogenesis, SU16f, a selective PDGFRβ inhibitor was employed in the same LEC-included matrigel plug assays. There were three groups: ‘LEC-SFM' group (LEC-matrigel injection and SFM treatment for 10 days); ‘LEC-TCM' group (LEC-matrigel injection and TCM treatment for 10 days); and ‘SU16f' group (LEC-matrigel injection and TCM/SU16f co-treatment for 10 days). TCM and SFM were injected subcutaneously. SU16f was dissolved in 10% DMSO solution (PBS base) and orally administered using an oral gavage for mouse feeding (10 mg/kg/day). Two control groups (‘LEC-TCM' and ‘LEC-SFM' groups) were also orally treated with vehicle (10% DMSO in PBS).
Immunofluorescence
We performed immunofluorescence with tumor tissues, matrigel plugs, and mono- or co-cultured cells. Tumor tissues and matrigel plugs, fixed in 10% neutral buffered formalin (Sigma) overnight, were placed in 30% sucrose (Sigma) in PBS, incubated overnight at 4°C, and frozen in the O.C.T. compound (Sakura). Sections of 10-μm thickness were cut at −20°C. To perform immunostaining with monocultured LEC, 25,000 LEC in normal media were plated in an 8-well Tissue-Tek and the cells were incubated overnight. The normal media was replaced with control media (30% SFM + 70% EGM) or 30% TCM media (30% TCM + 70% EGM), then the plate was incubated overnight. In LEC/MB231 co-culture model, we mixed 10,000 LEC and 10,000 MB231 (1:1) in 50% EGM and 50% RPMI-1640 media and make the cell adhere on the bottom for overnight. We then replaced the co-media with serum-free media and incubated the cells overnight again. Mono- or co-cultured cells were fixed in 10% neutral buffered formalin for 1 h at room temperature (RT) before immunostaining.
After blocking with 5% normal goat or normal chicken serum (Jackson Immunoresearch) in PBST (0.3% Triton) for 1 h at room temperature (RT), the tissue sections or cells were treated with one or more of the following primary antibodies overnight at 4°C: goat anti-human VEGFR3 (1:200, R & D systems), rabbit anti-Prox-1 antibody (1:500, AngioBio), rat anti-mouse CD31 (1:100, BD Pharmingen), mouse anti-human EGF (1:200, Santa Cruz), mouse anti-human PDGF-B (1:200, Santa Cruz), rat anti-human PCNA (1:1000, BD Pharmingen), goat anti-mouse desmin (Santa-Cruz), mouse anti-smooth muscle actin Cy-3 (SMA, 1:500, Sigma), rabbit anti-pEGFR (Cell Signaling), rabbit anti-cleaved caspase 3 (1:500, Cell Signaling), and goat anti-mouse lectin FITC (1:100, Sigma). After 3 rinses with PBST, sections were incubated for 1 hour at RT with one or more of the following secondary antibodies (1:500): FITC-conjugated goat anti-rat, FITC-conjugated chicken anti-goat, rhodamine-conjugated goat anti-rat, Cy3-conjugated goat anti-rabbit, Alexa Fluor 488 goat anti-mouse, FITC goat anti-rabbit, DyLight405 goat anti-rabbit, and DyLight405 goat anti-mouse antibodies (all from Jackson Immunoresearch). After 3 rinses with PBST, the samples were counterstained with DAPI (1:10,000, Roche) (5 min at RT). The samples were washed with PBST once and mounted with the ProLong Gold anti-fade reagent (Invitrogen) in the dark. Fluorescent signals were visualized and digital images were obtained using the LSM-510 confocal microscope (Carl Zeiss).
Real Time PCR
LEC (700,000 cells per flask) were plated in 100 mm tissue culture flasks in normal EGM media. After one day when they are attached on the bottom of the flask, the normal media was replaced with experimental control media (70% EGM + 30% SFM) or TCM media (70% EGM + 30% TCM), and cells were further incubated for overnight. Total RNA was extracted from normal LEC or tumor-educated LEC by the JHU Core using Qiagen's RNeasy Plus Mini Kit. Briefly, LEC were lysed, and disrupted with a QIAshredder spin column prior to putting over a special column for removal of genomic DNA. RNA was bound to second spin column for RNA extraction, washed several times, then RNA was eluted with RNase-free water. To prepare cDNA, normal LEC or tumor-educated LEC derived RNA (0.5 μg per group) was mixed with 4 μl of 5× iScript reaction mix (BioRad), 1 μl of iScript reverse transcriptase (BioRad), and nuclease-free water making total volume of 20 μl. cDNA synthesis was performed by the iQ5 multicolor RT-PCR Detection System (BioRad). Gathered 20 μl cDNA was 5 times more diluted with nuclease-free water for next RT-PCR. 5 μl of SYBR, 1 μl of primer (described below), 1 μl of cDNA (5 times diluted), and 3 μl nuclease-free water were mixed, and the RT-PCR was carried out using the CFX96 Real-Time System and C1000 Touch Thermal Cycler. We used BioRad Prime PCR assays for given target genes, human PDGFB, and human GAPDH.
The amplicon context sequence of the human PDGFB primers is AGCTCGCCTCCAGAGTGGGAGCGGGTCATGTTCAGGTCCAACTCGGCCCCATC TTCCTCTCCGGGGTCTCCGTGCAGCAGGCGTTGGAGATCATCAAAGGAGCGGA TCGAGTGGTC (assay ID: qHsaCID0016004, BioRad).
The amplicon context sequence of the human GAPDH primer is GTATGACAACGAATTTGGCTACAGCAACAGGGTGGTGGACCTCATGGCCCACAT GGCCTCCAAGGAGTAAGACCCCTGGACCACCAGCCCCAGCAAGAGCACAAGAG GAAGAGAGAGACCCTCACTGCTGGGGAGTCCCTGCCACAC (assay ID: qHsaCED0038674, BioRad).
Cell proliferation assays
Proliferation assays were performed by using the WST-1 reagent (Roche) as previously described52. WST-1 is reduced by mitochondrial dehydrogenases and transformed into dark yellow colored Formazan. MDA-MB-231 cells were seeded in 96-well plates at a density of 1,500 cells/well in 100 μl RPMI-1640 media. The plates were incubated overnight, then the RPMI-1640 media was replaced with LEC or HUVEC conditioned media (LEC-TCM, LEC-SFM, HUVEC-TCM) or serum-free media (SFM). Anti-EGFR antibodies (R&D systems) were employed in the LEC conditioned media to inhibit LEC-derived EGF effects in cancer cell proliferation. The plates were incubated for 72 h, after which the conditioned media (with or without anti-EGFR antibodies) was removed and 100 μl of WST-1 solution (1:10 dilution of WST-1 in SFM) was added. After incubation for 4 h, the absorbance at 450 nm was measured using the Victor V plate reader (Perkin Elmer).
Statistical Analysis
Error bars correspond to s.e.m, unless otherwise stated. Differences between two groups are regarded as significant when P is less than 0.05 using the Student's t-test.
Supplementary Material
Supplementary Information
Acknowledgments
We thank Dr. Zaver Bhujwalla for providing MDA-MB-231 breast cancer cells. We are grateful to Dr. Farhad Vesuna and Ashley Irving for their help in RT-PCR experiments. This work was supported by the grants from the National Institutes of Health R01 CA138264 and the Safeway Foundation.
Footnotes
Author Contributions E.L., N.B.P. and A.S.P. conceived the work. E.L. performed most of the experiments. E.L., N.B.P. and A.S.P. interpreted the experimental data. E.L. wrote the manuscript, and E.L., N.B.P. and A.S.P. edited the manuscript.
References
- Kim Y., Stolarska M. A. & Othmer H. G. The role of the microenvironment in tumor growth and invasion. Prog Biophys Mol Biol 106, 353–379 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Polyak K., Haviv I. & Campbell I. G. Co-evolution of tumor cells and their microenvironment. Trends Genet 25, 30–38 (2009). [DOI] [PubMed] [Google Scholar]
- Mukhtar R. A., Nseyo O., Campbell M. J. & Esserman L. J. Tumor-associated macrophages in breast cancer as potential biomarkers for new treatments and diagnostics. Expert Rev Mol Diagn 11, 91–100 (2011). [DOI] [PubMed] [Google Scholar]
- Yu Y. et al. Cancer-associated fibroblasts induce epithelial-mesenchymal transition of breast cancer cells through paracrine TGF-beta signalling. Br J Cancer 110, 724–732 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cirri P. & Chiarugi P. Cancer-associated-fibroblasts and tumour cells: a diabolic liaison driving cancer progression. Cancer Metastasis Rev 31, 195–208 (2012). [DOI] [PubMed] [Google Scholar]
- Jain R. K. & Carmeliet P. SnapShot: Tumor angiogenesis. Cell 149, 1408–1408 e1401 (2012). [DOI] [PubMed] [Google Scholar]
- Raica M. & Ribatti D. Targeting tumor lymphangiogenesis: an update. Curr Med Chem 17, 698–708 (2010). [DOI] [PubMed] [Google Scholar]
- Kerbel R. S. Tumor angiogenesis. N Engl J Med 358, 2039–2049 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Butler J. M., Kobayashi H. & Rafii S. Instructive role of the vascular niche in promoting tumour growth and tissue repair by angiocrine factors. Nat Rev Cancer 10, 138–146 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lu J. et al. Endothelial cells promote the colorectal cancer stem cell phenotype through a soluble form of Jagged-1. Cancer Cell 23, 171–185 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Franses J. W. & Edelman E. R. The evolution of endothelial regulatory paradigms in cancer biology and vascular repair. Cancer Res 71, 7339–7344 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krishnamurthy S. et al. Endothelial cell-initiated signaling promotes the survival and self-renewal of cancer stem cells. Cancer Res 70, 9969–9978 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- St Croix B. et al. Genes expressed in human tumor endothelium. Science 289, 1197–1202 (2000). [DOI] [PubMed] [Google Scholar]
- Warner K. A. et al. Endothelial cells enhance tumor cell invasion through a crosstalk mediated by CXC chemokine signaling. Neoplasia 10, 131–139 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zeng Q. et al. Crosstalk between tumor and endothelial cells promotes tumor angiogenesis by MAPK activation of Notch signaling. Cancer Cell 8, 13–23 (2005). [DOI] [PubMed] [Google Scholar]
- Cols M. et al. Stromal endothelial cells establish a bidirectional crosstalk with chronic lymphocytic leukemia cells through the TNF-related factors BAFF, APRIL, and CD40L. J Immunol 188, 6071–6083 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alitalo A. & Detmar M. Interaction of tumor cells and lymphatic vessels in cancer progression. Oncogene 31, 4499–4508 (2012). [DOI] [PubMed] [Google Scholar]
- Kim U., Park H. C. & Choi K. H. Differential permeability of lymphatic and blood vessels in determining the route of metastasis as demonstrated by indirect lymphography. Clin Exp Metastasis 6, 291–299 (1988). [DOI] [PubMed] [Google Scholar]
- Moskowitz M. et al. Breast cancer screening. Preliminary report of 207 biopsies performed in 4, 128 volunteer screenees. Cancer 36, 2245–2250 (1975). [DOI] [PubMed] [Google Scholar]
- Ran S., Volk L., Hall K. & Flister M. J. Lymphangiogenesis and lymphatic metastasis in breast cancer. Pathophysiology 17, 229–251 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hirakawa S. et al. Identification of vascular lineage-specific genes by transcriptional profiling of isolated blood vascular and lymphatic endothelial cells. Am J Pathol 162, 575–586 (2003). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alitalo K. The lymphatic vasculature in disease. Nat Med 17, 1371–1380 (2011). [DOI] [PubMed] [Google Scholar]
- Lee E., Pandey N. B. & Popel A. S. Pre-treatment of mice with tumor-conditioned media accelerates metastasis to lymph nodes and lungs: a new spontaneous breast cancer metastasis model. Clin Exp Metastasis 31, 67–79 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Franses J. W., Baker A. B., Chitalia V. C. & Edelman E. R. Stromal endothelial cells directly influence cancer progression. Sci Transl Med 3, 66ra65 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neiva K. G. et al. Endothelial cell-derived interleukin-6 regulates tumor growth. BMC Cancer 14, 99 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suzuki K. et al. Mesenchymal stromal cells promote tumor growth through the enhancement of neovascularization. Mol Med 17, 579–587 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fridman R., Benton G., Aranoutova I., Kleinman H. K. & Bonfil R. D. Increased initiation and growth of tumor cell lines, cancer stem cells and biopsy material in mice using basement membrane matrix protein (Cultrex or Matrigel) co-injection. Nat Protoc 7, 1138–1144 (2012). [DOI] [PubMed] [Google Scholar]
- Fridman R. et al. Enhanced tumor growth of both primary and established human and murine tumor cells in athymic mice after coinjection with Matrigel. J Natl Cancer Inst 83, 769–774 (1991). [DOI] [PubMed] [Google Scholar]
- Lee E., Koskimaki J. E., Pandey N. B. & Popel A. S. Inhibition of lymphangiogenesis and angiogenesis in breast tumor xenografts and lymph nodes by a peptide derived from transmembrane protein 45A. Neoplasia 15, 112–124 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nico B. et al. Desmin-positive pericytes in the chick embryo chorioallantoic membrane in response to fibroblast growth factor-2. Microvasc Res 68, 13–19 (2004). [DOI] [PubMed] [Google Scholar]
- Skalli O. et al. Alpha-smooth muscle actin, a differentiation marker of smooth muscle cells, is present in microfilamentous bundles of pericytes. J Histochem Cytochem 37, 315–321 (1989). [DOI] [PubMed] [Google Scholar]
- Skobe M. et al. Induction of tumor lymphangiogenesis by VEGF-C promotes breast cancer metastasis. Nat Med 7, 192–198 (2001). [DOI] [PubMed] [Google Scholar]
- Mayerson H. S., Patterson R. M., Mc K. A., Lebrie S. J. & Mayerson P. Permeability of lymphatic vessels. Am J Physiol 203, 98–106 (1962). [DOI] [PubMed] [Google Scholar]
- Ozawa S. et al. Stimulation by EGF of the growth of EGF receptor-hyperproducing tumor cells in athymic mice. Int J Cancer 40, 706–710 (1987). [DOI] [PubMed] [Google Scholar]
- Athale C. A. & Deisboeck T. S. The effects of EGF-receptor density on multiscale tumor growth patterns. J Theor Biol 238, 771–779 (2006). [DOI] [PubMed] [Google Scholar]
- Shima I. et al. Expression of epidermal growth-factor (EGF), matrix metalloproteinase-9 (mmp-9) and proliferating cell nuclear antigen (pcna) in esophageal cancer. Int J Oncol 6, 833–839 (1995). [DOI] [PubMed] [Google Scholar]
- Greco C. et al. c-MYC deregulation is involved in melphalan resistance of multiple myeloma: role of PDGF-BB. Int J Immunopathol Pharmacol 19, 67–79 (2006). [PubMed] [Google Scholar]
- Xue Y. et al. PDGF-BB modulates hematopoiesis and tumor angiogenesis by inducing erythropoietin production in stromal cells. Nat Med 18, 100–110 (2012). [DOI] [PubMed] [Google Scholar]
- Nissen L. J. et al. Angiogenic factors FGF2 and PDGF-BB synergistically promote murine tumor neovascularization and metastasis. J Clin Invest 117, 2766–2777 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cao Y. Opinion: emerging mechanisms of tumour lymphangiogenesis and lymphatic metastasis. Nat Rev Cancer 5, 735–743 (2005). [DOI] [PubMed] [Google Scholar]
- Baranski J. D. et al. Geometric control of vascular networks to enhance engineered tissue integration and function. Proc Natl Acad Sci U S A 110, 7586–7591 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- He W. et al. Pericyte-based human tissue engineered vascular grafts. Biomaterials 31, 8235–8244 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garcia-Roman J. & Zentella-Dehesa A. Vascular permeability changes involved in tumor metastasis. Cancer Lett 335, 259–269 (2013). [DOI] [PubMed] [Google Scholar]
- Xian X. et al. Pericytes limit tumor cell metastasis. J Clin Invest 116, 642–651 (2006). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kamenova B. et al. Effective treatment of the brachial plexus syndrome in breast cancer patients by early detection and control of loco-regional metastases with radiation or systemic therapy. Int J Clin Oncol 14, 219–224 (2009). [DOI] [PubMed] [Google Scholar]
- Karl E. et al. Unidirectional crosstalk between Bcl-xL and Bcl-2 enhances the angiogenic phenotype of endothelial cells. Cell Death Differ 14, 1657–1666 (2007). [DOI] [PubMed] [Google Scholar]
- Masur K., Schwartz F., Entschladen F., Niggemann B. & Zaenker K. S. DPPIV inhibitors extend GLP-2 mediated tumour promoting effects on intestinal cancer cells. Regul Pept 137, 147–155 (2006). [DOI] [PubMed] [Google Scholar]
- Wesley U. V., McGroarty M. & Homoyouni A. Dipeptidyl peptidase inhibits malignant phenotype of prostate cancer cells by blocking basic fibroblast growth factor signaling pathway. Cancer Res 65, 1325–1334 (2005). [DOI] [PubMed] [Google Scholar]
- Eubank T. D. et al. Granulocyte macrophage colony-stimulating factor inhibits breast cancer growth and metastasis by invoking an anti-angiogenic program in tumor-educated macrophages. Cancer Res 69, 2133–2140 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Borrello I. M. et al. Granulocyte-macrophage colony-stimulating factor (GM-CSF)-secreting cellular immunotherapy in combination with autologous stem cell transplantation (ASCT) as postremission therapy for acute myeloid leukemia (AML). Blood 114, 1736–1745 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Emens L. A. GM-CSF-secreting vaccines for solid tumors. Curr Opin Investig Drugs 10, 1315–1324 (2009). [PubMed] [Google Scholar]
- Lee E., Rosca E. V., Pandey N. B. & Popel A. S. Small peptides derived from somatotropin domain-containing proteins inhibit blood and lymphatic endothelial cell proliferation, migration, adhesion and tube formation. Int J Biochem Cell Biol 43, 1812–1821 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary Information