Abstract
Background
Treatment options for infections with soil-transmitted helminths (STH) - Ascaris lumbricoides, Trichuris trichiura and the two hookworm species, Ancylostoma duodenale and Necator americanus - are limited despite their considerable global health burden. The aim of the present study was to test the activity of an openly available FDA library against laboratory models of human intestinal nematode infections.
Methods
All 1,600 drugs were first screened against Ancylostoma ceylanicum third-stage larvae (L3). Active compounds were scrutinized and toxic compounds, drugs indicated solely for topical use, and already well-studied anthelmintics were excluded. The remaining hit compounds were tested in parallel against Trichuris muris first-stage larvae (L1), Heligmosomoides polygyrus third-stage larvae (L3), and adult stages of the three species in vitro. In vivo studies were performed in the H. polygyrus and T. muris mice models.
Results
Fifty-four of the 1,600 compounds tested revealed an activity of > 60 % against A. ceylanicum L3 (hit rate of 3.4 %), following incubation at 200 μM for 72 h. Twelve compounds progressed into further screens. Adult A. ceylanicum were the least affected (1/12 compounds active at 50 μM), while eight of the 12 test compounds revealed activity against T. muris L1 (100 μM) and adults (50 μM), and H. polygyrus L3 (200 μM). Trichlorfon was the only compound active against all stages of A. ceylanicum, H. polygyrus and T. muris. In addition, trichlorfon achieved high worm burden reductions of 80.1 and 98.9 %, following a single oral dose of 200 mg/kg in the T. muris and H. polygyrus mouse model, respectively.
Conclusion
Drug screening on the larval stages of intestinal parasitic nematodes is feasible using small libraries and important given the empty drug discovery and development pipeline for STH infections. Differences and commonalities in drug activities across the different STH species and stages were confirmed. Hits identified might serve as a starting point for drug discovery for STH.
Electronic supplementary material
The online version of this article (doi:10.1186/s13071-016-1616-0) contains supplementary material, which is available to authorized users.
Keywords: FDA library, Soil-transmitted helminthiasis, Drug sensitivity assay, Trichuris muris, Ancylostoma ceylanicum, Heligmosomoides polygyrus
Background
Infections with soil-transmitted helminths (STH) - Ascaris lumbricoides, Trichuris trichiura and the two hookworm species, Ancylostoma duodenale and Necator americanus - are enormously prevalent and responsible for a main part of the global health burden associated with neglected tropical diseases. Approximately 1.45 billion people are infected globally with at least one STH species [1]. In 2013, 4.0 million disability adjusted life years (DALYs) were attributed to intestinal nematode infections [2]. Symptoms include malnutrition, iron deficiency, anemia, intestinal obstruction, chronic dysentery, rectal prolapse, and poor weight gain [3]. To date, the most cost-effective public health strategy is to control morbidity by periodically administering anthelmintic drugs of mainly albendazole and mebendazole, on a large-scale. In 2014, 138.2 and 258 million preschool-aged and school-aged children, respectively, were treated with the benzimidazoles in endemic countries [4]. This number will increase considerably over the next years, since more than 800 million children require annual treatment with anthelmintic drugs [4]. Drug resistance is a serious concern in light of the enormous drug pressure. In addition, the spectrum of activity of the benzimidazoles is sub-optimal; albendazole and mebendazole have only low efficacy against the intestinal nematode species T. trichiura when administered at single doses [5]. It is therefore imperative that the drug discovery pipeline produces viable alternatives.
Drug discovery for most of the STH is truly neglected. For example, in 2014 as little as 678,299 and 171,197 USD were invested in drug discovery research for hookworm and T. trichiura infections, respectively (https://gfinder.policycures.org/PublicSearchTool). To our knowledge there are no novel drug candidates for soil-transmitted helminthiasis in the drug development pipeline.
In order to contribute to research and development efforts for these neglected tropical diseases, a series of libraries were screened for activity against hookworm and/or whipworm in the framework of a Gates-funded project in collaboration with the Drugs for Neglected Disease initiative. Here we present our results with an openly available FDA library. All 1,600 drugs were first screened against the larval stages of A. ceylanicum. Active compounds were examined and toxic compounds, drugs indicated solely for topical use and already well-studied anthelmintics were excluded. The remaining hit compounds were tested in parallel against adult A. ceylanicum and against T. muris L1 and Heligmosomoides polygyrus L3 and adults in vitro. In vivo studies were performed in the H. polygyrus and T. muris mice models.
Methods
Animals
Three-week-old male Syrian Golden hamsters were purchased from Charles River (Sulzfeld, Germany). Four-week-old female NMRI mice and female C57BL/10ScSnOlaHsd mice (age 3 weeks) were purchased from Harlan Laboratories (Horst, The Netherlands and Blackthorn, United Kingdom, respectively). All animals were kept in macrolon cages under environmentally-controlled conditions (temperature: 25 °C, humidity: 70 %, light/dark cycle 12 h/12 h) and had free access to water (municipal tap water supply) and rodent food. Rodents were allowed to acclimatize for one week before infection. The current study was approved by the local veterinary agency, based on Swiss cantonal and national regulations (permission no. 2070).
Drugs
The FDA Pharmakon compound library was purchased from MicroSource Discovery Systems, Inc. (USA). Compounds (10 mM, dissolved in DMSO) were stored at -80 °C until use. Levamisole and ivermectin, used for the in vitro controls, were purchased from Fluka and Sigma-Aldrich, respectively (Buchs, Switzerland), dissolved at 10 mM stock solutions in DMSO, and stored at -20 °C until further use. For the in vivo studies, bitoscanate, chlorcyclizine HCl, clemastine, dicyclomine HCl, drofenine HCl, ethopropazine HCl, lansoprazole, metformin HCl, morantel, natamycin, trichlorfon, and trimipramine maleate were purchased from Sigma-Aldrich.
In vitro tests on A. ceylanicum L3
A. ceylanicum larvae (L3) were obtained by filtering the feces of infected hamsters and cultivating the eggs for 9 days in the dark at 24 °C. L3 were washed in penicillin and streptomycin-supplemented tap water and kept under refrigeration until used. For the drug assay, 40 L3 were placed in each well of a 96-well plate for each compound, in duplicate. Worms were incubated with 200 μM of the test drug and culture medium, which was composed of 100 μl HBSS medium (Hanks’ Balanced Salt Solution Modified; GIBCO, Lucerne, Switzerland) supplemented with 25 μg/ml amphotericin B, 100 U/ml penicillin, and 100 μg/ml streptomycin (Sigma-Aldrich). Worms incubated with culture medium and 1 % DMSO served as negative controls. Wells containing larvae, medium, and 200 μM of levamisole served as positive controls. The plates were kept at room temperature and in the dark for up to 72 h, after which the drug effect was evaluated. To do so, first the total number of L3 per well was determined. Then, 100 μl of hot water (≈80 °C) was added to each well and the larvae that responded to this stimulus (the moving L3) were counted. The proportion of larval death was determined. A cut-off of 60 % was used to define activity, hence compounds achieving > 60 % larval death progressed to further testing.
In vitro tests on A. ceylanicum adult worms
Hamsters were infected per os with 150 A. ceylanicum L3. Three weeks post-infection, hamsters were euthanized with CO2 and the worms were collected from the intestine. For each compound, three A. ceylanicum adults were placed in each well of a 24-well plate, using 2 wells per compound. Worms were incubated in the presence of 50 μM of the drug to be tested, and culture medium, which was composed of HBSS medium supplemented with 10 % Fetal Calf Serum (FCS) (Connectorate AG, Dietikon, Switzerland), 25 μg/ml amphotericin B, 100 U/ml penicillin, and 100 μg/ml streptomycin. Worms incubated in 1 % DMSO in culture medium served as control. As a positive control, ivermectin was used at a concentration of 50 μM. Worms were kept in an incubator at 37 °C and 5 % CO2 for up to 72 h. Thereafter, the condition of the worms was microscopically evaluated using a viability scale from 3 (normal activity and no tegument alteration) to 0 (dead, completely granulated).
In vitro tests on T. muris L1 worms
Female C57BL/10ScSnOlaHsd mice were infected per os with 200–250 T. muris eggs showing around 90–95 % embryonation (checked microscopically). After 7 weeks, T. muris eggs were collected from the feces of the infected mice by the flotation method using saturated NaCl solution in Milli-Q water. T. muris eggs were stored in Milli-Q water in the dark for 3 months at 23–25 °C, until the eggs were embryonated. The hatching process to obtain T. muris L1 has been described elsewhere [6]. For the assay, 40 L1 larvae were placed in each well of a 96-well plate. Worms were incubated in the presence of 100 μl RPMI 1640 medium with 12.5 μg/ml amphotericin B, 500 U/ml penicillin, 500 μg/ml streptomycin, and 100 μM of the drug to be tested. Each drug was tested in duplicate. L1 larvae incubated with culture medium and 1 % DMSO served as a negative control and larvae incubated with levamisole at a concentration of 100 μM were included as a positive control. The assay was kept in an incubator at 37 °C and 5 % CO2 for 24 h. At 24 h, the total number of L1 larvae per well was counted. The larvae were then stimulated with 100 μl hot water (≈80 °C) and the moving L1 larvae were counted.
In vitro assay with T. muris adult worms
Seven weeks post-infection of female C57BL/10ScSnOlaHsd mice, T. muris adult worms were collected from the intestines. Three T. muris adult worms were placed in each well of a 24-well plate. Worms were incubated with culture medium and 50 μM of the drug to be tested. Each compound was tested in duplicate. Trichuris muris adult worms incubated with 1 % DMSO and culture medium served as control. As positive control, levamisole was used at a concentration of 50 μM. Worms were kept in an incubator at 37 °C and 5 % CO2 for 72 h and were subsequently microscopically evaluated using a viability scale from 3 (normal activity and no tegument alteration) to 0 (dead, completely granulated).
Heligmosomoides polygyrus L3 in vitro studies
Female NMRI mice were infected with 80 H. polygyrus L3 per os. H. polygyrus eggs were obtained from infected feces. The eggs were then placed on agar and, after 9 days in the dark at 24 °C, the L3 larvae hatched. For the drug assay, 40 L3 larvae were placed in each well of a 96-well plate. Worms were incubated in the presence of 100 μl RPMI 1640 medium, supplemented with 0.63 μg/ml amphotericin B, 500 U/ml penicillin, 500 μg/ml streptomycin, and 100 μM of the drug to be tested. Each drug was tested in duplicate. Worms incubated with 1 % DMSO and culture medium served as a negative control and wells containing larvae, medium and 100 μM levamisole served as a positive control. The plates were kept at room temperature for up to 72 h. To assess the effect of the compound, the total number of L3 larvae per well was counted, the larvae were stimulated with 100 μl hot water (≈80 °C), and the moving L3 were counted.
In vitro assay with H. polygyrus adults
Female NMRI mice were infected with 80 H. polygyrus L3 per os. Two weeks post-infection, mice were dissected and three adult worms were placed in each well of a 24-well plate. Worms were incubated with culture medium and 50 μM of the test drug. Each compound was tested in duplicate. Adult worms incubated with medium only and 50 μM levamisole and ivermectin served as negative and positive control, respectively. Worms were kept in an incubator at 37 °C and 5 % CO2 for 72 h and, subsequently, were microscopically evaluated using a viability scale from 3 to 0.
Trichuris muris in vivo studies
C57BL/10ScSnOlaHsd mice were infected with 200 embryonated T. muris eggs per os. The drinking water for the mice contained dexamethasone (1 mg/ml) in order to suppress their immune system to avoid rejection of the infection. At 42 days post-infection, the stool was collected and filtered to check for the presence of eggs. Infected mice were placed in individual cages. The compounds were administered by gavage at dosages of 10–400 mg/kg (guided by the LD50 values of the drugs). The dose volume was adjusted according to the mouse’s body weight, which was assessed for each mouse directly prior to treatment. Four to six untreated mice served as controls. After 72 h, the stool was collected from each cage to check and count the dead worms released through the stool. Six days post-treatment, animals were killed by the CO2 method and the gastrointestinal tract was collected. The intestine was dissected and adult worms were collected and counted.
Heligmosomoides polygyrus in vivo studies
NMRI mice were infected with 80 H. polygyrus L3 per os. Fourteen days post-infection, mice were treated orally with the test drugs at dosages of 10–400 mg/kg. Four to six untreated mice served as controls. Ten days post-treatment, animals were killed by the CO2 method, and the gastrointestinal tract was collected. The intestine was dissected and adult worms were collected and counted.
Statistics
For the in vitro drug sensitivity assays, all viability scores/larval survival counts were averaged across replicates and normalized to the average viability scores/larval survival counts of the control wells (Microsoft Office Excel 2010) using the following formula: Drug effect (%) = 100 % – [100 % * (No. of live larvae alive/Total No. of larvae) drug/(No. of live larvae/Total No. of larvae) control]. To calculate the drug effect in in vivo studies, the worm burden (WB) of treated animals was calculated and compared with the worm burden of control mice, which were infected simultaneously but were not treated. The worm burden reduction (WBR) was calculated as follows: WBR (%) = 100 % – (100 % * WB treatment/WB control). The worm expulsion rates were calculated as follows: (c/d) × 100, where c is the total number of expelled worms in a treated group and d is the total worm count (expelled worms as well as worms present in the gut counted following dissection) of the same group. The Kruskal-Wallis test (Statsdirect, statistical software version 2.8.0) was used to determine statistical significance of WBRs at a level of 0.05.
Results
In vitro studies
The study flow is presented in Fig. 1. From 1,600 compounds tested, 54 compounds revealed an activity of > 60 % against A. ceylanicum L3 (hit rate of 3.4 %), following incubation at 200 μM for 72 h. Twenty of these compounds killed all L3. Results are summarized in Table 1. The activities of the standard drugs, levamisole and ivermectin, (also included in the library) are presented as reference. Both drugs resulted in death of all A. ceylanicum L3.
Fig. 1.
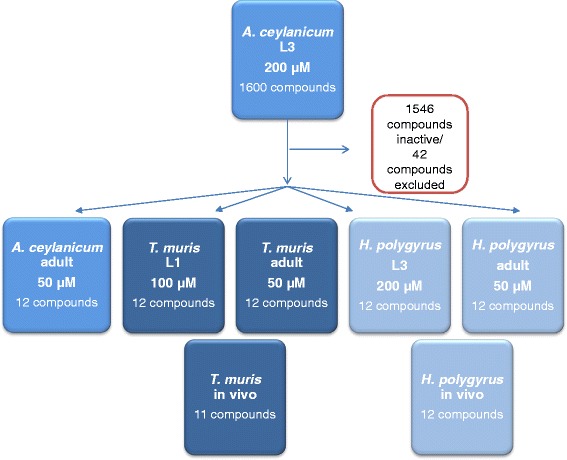
Screening flow applied testing a library of 1600 FDA approved compounds against A. ceylanicum, T. muris and H. polygyrus
Table 1.
In vitro activity profile of 56 FDA compounds (including 2 reference compounds) against A. ceylanicum and T. muris adult and larval stages
| Compound | Drug effect (%) | |||||
|---|---|---|---|---|---|---|
| A. ceylanicum L3 200 μM (72 h) | H. polygyrus L3 200 μM (72 h) | T. muris L1 100 μM (24 h) | A. ceylanicum adult 50 μM (72 h) | H. polyygrus adult 50 μM (72 h) | T. muris adult 50 μM (72 h) | |
| Levamisole | 100 | 100 | 100 | 0 | 56 | 100 |
| Ivermectin | 100 | 93 | 0 | 100 | 41 | 0 |
| Abamectin | 100 | ND, anthelmintic | ||||
| Amitriptyline HCl | 91 | ND, toxic | ||||
| Apomorphine HCl | 72 | ND, toxic | ||||
| Bifonazole | 67 | ND, topical application | ||||
| Bitoscanate | 100 | 20 | 100 | 28 | 34 | 100 |
| Carbachol | 89 | ND, topical application | ||||
| Chlorambucil | 95 | ND, toxic | ||||
| Chlorcyclizine HCl | 78 | 77 | 78 | 23 | 30 | 31 |
| Chlorhexidine dihydrochloride | 61 | ND, topical application | ||||
| Chloroxylenol | 100 | ND, topical application | ||||
| Chlorpromazine | 100 | ND, toxic | ||||
| Chlorprothixene HCl | 100 | ND, toxic | ||||
| Chlorpyrifos | 97 | ND, toxic | ||||
| Cisplatin | 87 | ND, toxic | ||||
| Clemastine fumarate | 76 | 91 | 2 | 47 | 30 | 100 |
| Clomiphene citrate | 80 | ND, toxic | ||||
| Coumophos | 100 | ND, toxic | ||||
| Cyproheptadine HCl | 88 | ND, toxic | ||||
| Dexlansoprazole | 88 | 35 | 1 | 47 | 17 | 55 |
| Diatrizoic acid | 81 | ND, toxic | ||||
| Dibenzothiophene | 100 | ND, topical application | ||||
| Dicyclomine HCl | 71 | 88 | 7 | 42 | 100 | 93 |
| Dimpylate | 100 | ND, toxic | ||||
| Doramectin | 100 | ND, anthelmintic | ||||
| Doxepin HCl | 64 | ND, toxic | ||||
| Drofenine HCl | 62 | 87 | 100 | 44 | 20 | 100 |
| Dyclonine HCl | 100 | ND, topical | ||||
| Eprinomectin | 100 | ND, anthelmintic | ||||
| Ethopropazine HCl | 100 | 89 | 100 | 33 | 100 | 100 |
| Fenbendazole | 66 | ND, anthelmintic | ||||
| Fenthion | 97 | ND, toxic | ||||
| Ftaxilide | 73 | ND, commercially not available | ||||
| Hexachlorophene | 100 | ND, topical | ||||
| Histamine dihydrochloride | 62 | ND, topical | ||||
| Mebendazole | 78 | ND, anthelmintic | ||||
| Metformin HCl | 100 | 11 | 100 | 50 | 27 | 75 |
| Metitepine mesylate | 70 | ND, toxic | ||||
| Morantel citrate | 60 | 100 | 100 | 47 | 80 | 94 |
| Moxidectin | 100 | ND, anthelmintic | ||||
| Natamycin | 95 | 14 | 100 | 53 | 10 | 61 |
| Phenothiazine | 98 | ND, toxic | ||||
| Phenylmercuric acetate | 100 | ND, topical | ||||
| Pramoxine HCl | 100 | ND, topical | ||||
| Pyrantel pamoate | 91 | ND, anthelmintic | ||||
| Selamectin | 79 | ND, anthelmintic, topical | ||||
| Tetramizole HCl | 100 | ND, anthelmintic | ||||
| Thiabendazole | 93 | ND, anthelmintic | ||||
| Thonzonium bromide | 98 | ND, topical | ||||
| Trichlorfon | 100 | 89 | 100 | 100 | 79 | 100 |
| Triclosan | 98 | ND, topical | ||||
| Triflupromazine HCl | 83 | ND, toxic | ||||
| Trimeprazine tartrate | 64 | ND, toxic | ||||
| Trimipramine maleate | 62 | 79 | 99 | 7 | 23 | 75 |
| Zoxazolamine | 98 | ND, toxic | ||||
Abbreviation: ND Not done
Of the 54 compounds, 12 compounds have a topical indication and 19 drugs are toxic or are associated with severe adverse events. Twelve drugs (including levamisole and ivermectin) are well-known anthelmintics and, with exception of bitoscanate and morantel (for which little information on the efficacy against these parasites is available), were therefore not considered for further testing in the framework of this work. One drug, ftaxilide was not commercially available. Hence, 12 compounds progressed into further screens. Against adult A. ceylanicum, only trichlorfon resulted in death of the worms, while the remaining 11 compounds revealed low activity (< 60 % activity). Eight of the 12 compounds affected H. polygyrus L3, with an activity of > 75 %. Adult H. polygyrus showed slightly higher sensitivity to the drugs as compared to adult A. ceylanicum, with dicyclomine HCl and ethopropazine HCl resulting in death of all worms, and trichlorfon and morantel revealing an activity of 79 and 80 %, respectively.
Against T. muris L1, nine of the 12 test compounds showed an activity ranging from 78–100 %, at a concentration of 100 μM after 24 h of incubation. Six of these compounds caused death of all worms. The parallel tests on adult T. muris yielded nine compounds with high activity (75–100 %) against T. muris adults, of which five compounds were lethal. There was a good correlation between larval and adult activity: two compounds were active against adult T. muris, but not active against L1. Conversely, one compound showed no activity against adult T. muris despite being active against L1 (Table 1).
Trichlorfon was the only compound active against all stages of A. ceylanicum, H. polygyrus and T. muris.
In vivo studies against H. polygyrus
Identified hits from in vitro studies (n = 12) were further evaluated for in vivo proof of principle. To maximize chances of success, compounds were evaluated at high doses. Single administrations were favored to mimic dosing regimens used in mass drug administration programs for treatment of STH infections with the current standard of care.
Low activity was observed for chlorcyclizine HCl, clemastine, dicyclomine HCl, drofenine HCl, ethopropazine HCl, lansoprazole, metformin HCl and trimipramine maleate, with WBRs below 41 %. In mice treated with a single oral dose of 400 mg/kg natamycin, a WBR of 59.7 % was observed. The highest activities were observed with bitoscanate and trichlorfon. Both dosages of bitoscanate (100 and 50 mg/kg) resulted in significant WBRs of 100 and 98.8 %, respectively. A WBR (98.9 %) was achieved with a single oral dose of 200 mg/kg trichlorfon, curing three of four treated mice (Table 2). At 100 mg/kg trichlorfon, the calculated WBR was still significant (81.9 %).
Table 2.
In vivo studies against H. polygyrus and T. muris
| Treatment | Dose (mg/kg) | T. muris | H. polygyrus | |||||
|---|---|---|---|---|---|---|---|---|
| No. of mice cured/investigated | Average worm number | Worm burden reduction (%) | Worm expulsion rate (%) | No. of mice cured/investigated | Average worm number | Worm burden reduction (%) | ||
| Control a | – | 0/6 | 170.95 | – | – | – | --- | --- |
| Control b | – | 0/4 | 163.5 | – | – | – | --- | --- |
| Control c | – | 0/4 | 197.3 | – | – | – | --- | --- |
| Control 1 | 0/8 | 61.3 | --- | |||||
| Control 2 | 0/8 | 21.1 | ||||||
| Control 3 | 0/12 | 47.0 | ||||||
| Bitoscanatea,3 | 100 | 0/3* | 70.5 | 58.8 | 0.3 | 4/4 | 0 | 100 |
| Bitoscanate2 | 50 | ND | – | – | – | 3/4 | 0.25 | 98.8¶ |
| Chlorcyclizine HClc,1 | 100 | 0/3 | 115.0 | 41.7 | 0/4 | 39.5 | 35.6 | |
| Clemastineb,1 | 100 | 0/3 | 130.7 | 20.1 | 2.5 | 0/4 | 84.8 | 0 |
| Dicyclomine HClb,1 | 400 | 0/3 | 245.5 | 0 | 39.1 | 0/4 | 36.8 | 40.1 |
| Drofenine HCla,1 | 400 | 0/3 | 267.6 | 0 | 3.1 | 0/4 | 54.0 | 12.0 |
| Ethoproprazine HClb,1 | 200 | 0/3 | 368.0 | 0 | 6.7 | 0/4 | 45.5 | 25.8 |
| Lansoprazole1 | 400 | ND | – | – | – | 0/4 | 43.0 | 29.9 |
| Metformin HCla,1 | 400 | 0/3 | 140.6 | 17.7 | 0.6 | 0/4 | 80.0 | 0 |
| Morantelc,1 | 400 | 0/3** | 367.0 | 0 | 0 | 0/2** | ||
| Natamycina,1 | 400 | 0/3*** | – | – | – | 2/4 | 24.8 | 59.7 |
| Trichlorfonc,1 | 200 | 1/3 | 34.0 | 80.1¶ | 67.6 | 3/4 | 0.5 | 98.9¶ |
| Trichlorfona | 100 | 0/3 | 121.0 | 28.8 | 3.3 | 2/4 | 8.5 | 81.9¶ |
| Trimipramine maleatec,1 | 10 | 0/3 | 113.3 | 42.6 | 0.1 | 0/4 | 71.5 | 0 |
Each * indicates a mouse that died. Superscripts 1–3 and a-c refer to the respective control groups ¶ P < 0.05
In vivo studies against T. muris
Nine of the 11 compounds tested in vivo revealed a low activity against T. muris (WER < 7 % and WBRs < 45 %). Natamycin and morantel were not tolerated by the mice and five out of six mice died within 24 h post-treatment. Treatment of infected mice with bitoscanate achieved a WBR of 42 %, however, one of the mice died two days after treatment. The highest activity was observed with trichlorfon administered at 200 mg/kg, which resulted in a WBR of 80 % and a WER of 67 %. Halving the dose to 100 mg/kg resulted in a low WBR and WER (28.8 and 3.3 %, respectively) (Table 2).
Discussion
Drug repurposing is the key drug discovery strategy for human helminth infections, since anthelmintic drug discovery and development has languished [7]. Veterinary anthelmintics have obviously been the most attractive candidates for cross-over development for the treatment of human STH infections, since the regulatory standards for veterinary drugs are compatible with those for human drugs and a track-record of their use in animals exists [8]. Indeed, all of the few anthelmintics currently used for humans stem from veterinary medicine. In the present work, we applied a broader repurposing strategy, by evaluating 1,600 compounds from the FDA library, which contains approved drugs over a range of indications.
While a handful of studies employing target screening approaches have been conducted to identify new pharmacophores for the treatment of human STH infections [9], phenotypic screening of selected libraries on parasitic nematodes is basically non-existent. However, whole-organism screens have several advantages over target-based screening, since they are not constrained to single targets (which are not well characterized for helminths) [10]. Indeed, phenotypic approaches are more successful for small-molecule, first-in-class medicines than target based approaches [11]. High throughput assays with Caenorhabditis elegans often serve as a substitute for helminth phenotypic assays, because most assays for parasitic nematodes are laborious and low throughput. However, the correlation of activity against C. elegans to parasitic nematodes is not universal, with for example albendazole lacking activity against C. elegans [12].
We observed a hit rate of 3.4 % against A. ceylanicum larvae, our primary screen, with hits ranging across a large spectrum of indications. For comparison, a hit rate of ~7.6 % was observed against S. mansoni schistosomula using the same library and also encompassing a wide range of compound indications [13]. As highlighted already for the S. mansoni screen [13], it was disappointing that many of the hits were unsuitable for further testing, notably due to documented toxicity or restriction to topical use. In addition, we excluded the common anthelmintics (e.g. mebendazole, pyrantel). Selected compounds were next tested against a panel of helminths, namely A. ceylanicum adults, H. polygyrus larvae and adults, as well as T. muris larvae and adults. A few observations are offered for discussion. First, A. ceylanicum L3 were selected for the primary screen since, compared to adult stages, their use offers many advantages, mainly in terms of ethical considerations, numbers available, and ease of provision [14]. However, larval stages may not always be equally as sensitive as the target parasite stage, the adult worms. For example, the veterinary anthelmintic, monepantel, lacks activity against A. ceylanicum L3, while it is active against the adult worm [15]. Hence, larval-based assays should be validated in terms of how larval sensitivity compares to the sensitivity of adult worms. Additional file 1: Table S1 summarizes the activity of all 54 A. ceylanicum L3 active drugs (including topical and toxic drugs) against adult A. ceylanicum, T. muris L1 and adult T. muris. In general, A. ceylanicum L3 were more sensitive in our assay to the test compounds than the adult stages. Increased sensitivities of larval forms over adult worms have been reported earlier including for S. mansoni schistosomula [16]. Though a high false positive rate (larval activity does not always translate to adult activity) is not optimally cost-effective, the risk of losing interesting compounds is minimal, and in any case, larval pre-screens are still more cost-effective and more ethical than conducting the entire screen on adult stage worms. However, for T. muris this trend could not be confirmed (Additional file 1: Table S1). Adult T. muris were equally or even more sensitive than T. muris L1. Second, to our knowledge, we have for the first time compared the sensitivities of a small series of test drugs on larval and adult stages of the two hookworm species. The H. polygyrus model is widely used to study intestinal nematode infections, as it is easy to maintain in the laboratory and far more cost-effective than other laboratory hookworm rodent models [17]. The activity of compounds on A. ceylanicum L3 and H. polygyrus L3 was similar (only bitoscanate, dexlansoprazole, metformin and natamycin lacked activity against H. polygyrus L3). Similarly, the A. ceylanicum adult screen mirrored the adult H. polygyrus screen (except for dicyclomine HCl and ethopropazine, which showed activity against adult H. polygyrus). Hence, our results with a small panel of compounds support the use of H. polygyrus in the framework of anthelmintic drug discovery.
Only two compounds revealed activity in vivo, namely trichlorfon and bitoscanate. In addition, a moderate activity was observed with natamycin against H. polygyrus (two out of four mice cured), while the drug was not tolerated by T. muris infected mice. The low in vivo activity of many of the in vivo tested drugs might not be surprising, as bioavailable drugs from the FDA library of 1,600 approved drugs may not have the ideal profile for in vivo activity. Though there are exceptions (e.g. levamisole, ivermectin or albendazole) an ideal treatment for STH, particularly Trichuris spp. infections should be only poorly absorbed in order to target worms sitting in the gastrointestinal tract.
In vivo, trichlorfon revealed high activity against H. polygyrus and T. muris, while bitoscanate showed high activity against hookworm but was moderately active against T. muris. The organophosphate trichlorfon is mainly used as an ectoparasiticide [18]. However, it is also known for its antischistosomal properties [trichlorfon (metrifonate) was marketed for the treatment of S. haematobium prior to the advent of praziquantel] [19] and is applied for the control of intestinal nematode parasites of cattle and sheep. Trichlorfon is particularly used for nematodes that have developed resistance to other commonly-used anthelmintics [20]. In the past years, it was investigated for the treatment of Alzheimer’s disease. Metrifonate, at various fixed and loading doses, was associated with significant cognitive improvement compared to placebo, where the slow-release break-down byproduct, 2,2-dichlorovinyl dimethyl phosphate (DDVP), is supposed to be the active component [21, 22]. The broad activity of metrifonate is perhaps not surprising considering its mechanism of action; metrifonate is a cholinergic drug, acting as an irreversible non-selective acetylcholinesterase and butyrylcholinesterase inhibitor [21]. There is a history of active cholinergic anthelmintics including pyrantel and levamisole, which are both selective nAChR agonists, and ivermectin and moxidectin, which are modulators of glutamate-gated ion channels and nAChRs [23]. Emerging cholinergic anthelmintics, monepantel, tribendimidine and derquantel [24–26] have different nAChR subtype selectivities.
Bitoscanate was widely used prior to the advent of the benzimidazoles for the treatment of hookworm infections [27, 28]. Despite its history of use, the literature is very scarce. Bitoscanate belongs to the broad isothiocyanate class of anthelmintic compounds, which occur widely in nature, particularly in plants of the mustard (Brassicacae) family [29]. In India, clinical trials with thousands of hookworm-infected patients were conducted, which revealed a high efficacy of the drug. Conflicting results were observed with regard to Trichuris spp.; while a good trichuricidal activity was observed in some settings [30], the high efficacy was not consistently seen [28]. Mild and transient adverse events were reported in the large number of studies done with bitoscanate [28]. However, in the US bitoscanate is listed as an extremely hazardous substance (http://nj.gov/health/eoh/rtkweb/documents/fs/2172.pdf) which, when released in certain amounts in the environment, may be of immediate concern to the community. A related compound, amoscanate, was found to be effective against hookworm and schistosomes in humans, but was accompanied with severe liver toxicity in laboratory animals at high doses [31, 32]. However, another related compound, nitroscanate, is widely employed in veterinary medicine to treat roundworm, hookworm and tapeworms [33]. Currently, the isothiocyanates are researched for activity against intestinal bacteria and cancer [34, 35].
The same library was screened against S. mansoni larval stage and adult worms [13] with a very different set of hits. When compared, 15 (27.8 %) of the compounds identified as hits in this screen were also hits against S. mansoni newly-transformed schistosomula (NTS) (abamectin, chlorprothixene hydrochloride, clomiphene citrate, doramectin, eprinomectin, hexachlorophene, metformin hydrochloride, metitepine mesylate, moxidectin, natamycin, phenylmercuric acetate, selamectin, thonzonium bromide, trichlorfon and triflupromazine hydrochloride). The vast majority of these common hits were either toxic or for topical use only. However, it is also noted that the macrocyclic lactones, a group that acts on glutamate-gated ion channels [36], in general appear to be active against both S. mansoni and hookworm.
Conclusions
In conclusion, we tested the FDA library of 1,600 approved drugs using a screening cascade with the larval stages of the parasitic nematode, A. ceylanicum, as a primary model. Our work has demonstrated that drug screening on the larval stages of intestinal parasitic nematodes is feasible using small libraries and shows differences and commonalities in drug activities across the different STH species and stages. A moderate hit rate of 3.4 % was observed with the FDA library of approved drugs, however many of the active drugs consisted of toxic compounds, compounds for topical use, or anthelmintics. Trichlorfon and bitoscanate were the only active compounds in in vivo studies; however both drugs have clear limitations. Nonetheless, structurally related pharmacophores might serve as a starting point for drug discovery for STH infections.
ᅟ
ᅟ
Funding
JK is grateful to the European Research Council (ERC-2013-CoG 614739-A_HERO) for financial support. The funders had no role in design, in the collection, analysis, and interpretation of data; in the writing of the manuscript; and in the decision to submit the manuscript for publication.
Availability of data and materials
The datasets supporting the conclusion of this article are included within the article and its additional file.
Authors’ contributions
RA and JK designed the studies. RA, MV and NC carried out the experiments. JK wrote the first draft of the manuscript. GP, NC and IS revised the manuscript. All authors read and approved the final version of the manuscript.
Competing interests
The authors declare that they have no competing interests.
Consent for publication
Not applicable.
Ethics approval
The current study was approved by the local veterinary agency based on Swiss cantonal and national regulations (permission no. 2070).
Additional file
In vitro activity profile of 56 FDA compounds against A. ceylanicum and T. muris larval and adult stages (including topical and toxic drugs). (DOCX 52 kb)
References
- 1.Pullan RL, Brooker SJ. The global limits and population at risk of soil-transmitted helminth infections in 2010. Parasit Vectors. 2012;5:81. doi: 10.1186/1756-3305-5-81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Murray CJ, Barber RM, Foreman KJ, Abbasoglu Ozgoren A, Abd-Allah F, Abera SF, et al. Global, regional, and national disability-adjusted life years (DALYs) for 306 diseases and injuries and healthy life expectancy (HALE) for 188 countries, 1990–2013: quantifying the epidemiological transition. Lancet. 2015;386:2145–91. doi: 10.1016/S0140-6736(15)61340-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ojha SC, Jaide C, Jinawath N, Rotjanapan P, Baral P. Geohelminths: public health significance. J Infect Dev Ctries. 2014;8:5–16. doi: 10.3855/jidc.3183. [DOI] [PubMed] [Google Scholar]
- 4.World Health Organisation Soil-transmitted helminthiases: number of children treated in 2014. Wkly Epidemiol Rec. 2015;90:705–11. [PubMed] [Google Scholar]
- 5.Keiser J, Utzinger J. The drugs we have and the drugs we need against major helminth infections. Adv Parasitol. 2010;73:197–230. doi: 10.1016/S0065-308X(10)73008-6. [DOI] [PubMed] [Google Scholar]
- 6.Wimmersberger D, Tritten L, Keiser J. Development of an in vitro drug sensitivity assay for Trichuris muris first-stage larvae. Parasit Vectors. 2013;6:42. doi: 10.1186/1756-3305-6-42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Panic G, Duthaler U, Speich B, Keiser J. Repurposing drugs for the treatment and control of helminth infections. Int J Parasitol Drugs Drug Resist. 2014;4:185–200. doi: 10.1016/j.ijpddr.2014.07.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.de Hostos EL, Nguyen T. Anthelmintic drugs: tools and shortcuts for the long road from discovery to product. In: Parasitic Helminths: Wiley-VCH Verlag GmbH & Co. KGaA; 2012: 217–232.
- 9.Cho Y, Vermeire JJ, Merkel JS, Leng L, Du X, Bucala R, et al. Drug repositioning and pharmacophore identification in the discovery of hookworm MIF inhibitors. Chem Biol. 2011;18:1089–101. doi: 10.1016/j.chembiol.2011.07.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Paveley RA, Bickle QD. Automated imaging and other developments in whole-organism anthelmintic screening. Parasite Immunol. 2013;35:302–13. doi: 10.1111/pim.12037. [DOI] [PubMed] [Google Scholar]
- 11.Swinney DC, Anthony J. How were new medicines discovered? Nat Rev Drug Discov. 2011;10:507–19. doi: 10.1038/nrd3480. [DOI] [PubMed] [Google Scholar]
- 12.Keiser J. Is Caenorhabditis elegans the magic bullet for anthelminthic drug discovery? Trends Parasitol. 2015;31:455–6. doi: 10.1016/j.pt.2015.08.004. [DOI] [PubMed] [Google Scholar]
- 13.Panic G, Vargas M, Scandale I, Keiser J. Activity profile of an FDA-approved compound library against Schistosoma mansoni. PLoS Negl Trop Dis. 2015;9:e0003962. doi: 10.1371/journal.pntd.0003962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Paveley RA, Mansour NR, Hallyburton I, Bleicher LS, Benn AE, Mikic I, et al. Whole organism high-content screening by label-free, image-based Bayesian classification for parasitic diseases. PLoS Negl Trop Dis. 2012;6:e1762. doi: 10.1371/journal.pntd.0001762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Tritten L, Silbereisen A, Keiser J. In vitro and in vivo efficacy of monepantel (AAD 1566) against laboratory models of human intestinal nematode infections. PLoS Negl Trop Dis. 2011;5:e1457. doi: 10.1371/journal.pntd.0001457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ingram-Sieber K, Cowan N, Panic G, Vargas M, Mansour NR, Bickle QD, et al. Orally active antischistosomal early leads identified from the open access malaria box. PLoS Negl Trop Dis. 2014;8:e2610. doi: 10.1371/journal.pntd.0002610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Harris NL, Pleass R, Behnke JM. Understanding the role of antibodies in murine infections with Heligmosomoides (polygyrus) bakeri: 35 years ago, now and 35 years ahead. Parasite Immunol. 2014;36:115–24. doi: 10.1111/pim.12057. [DOI] [PubMed] [Google Scholar]
- 18.Chang CC, Rahmawaty A, Chang ZW. Molecular and immunological responses of the giant freshwater prawn, Macrobrachium rosenbergii, to the organophosphorus insecticide, trichlorfon. Aquat Toxicol. 2013;130–131:18–26. doi: 10.1016/j.aquatox.2012.12.024. [DOI] [PubMed] [Google Scholar]
- 19.Utzinger J, Keiser J. Schistosomiasis and soil-transmitted helminthiasis: common drugs for treatment and control. Expert Opin Pharmacother. 2004;5:263–85. doi: 10.1517/14656566.5.2.263. [DOI] [PubMed] [Google Scholar]
- 20.Fiel C, Guzman M, Steffan P, Rodriguez E, Prieto O, Bhushan C. The efficacy of trichlorphon and naphthalophos against multiple anthelmintic-resistant nematodes of naturally infected sheep in Argentina. Parasitol Res. 2011;109(Suppl 1):S139–48. doi: 10.1007/s00436-011-2410-8. [DOI] [PubMed] [Google Scholar]
- 21.Schneider LS, Giacobini E. Metrifonate. A cholinesterase inhibitor for Alzheimer’s disease therapy. CNS Drug Rev. 1999;5:13–26. doi: 10.1111/j.1527-3458.1999.tb00083.x. [DOI] [Google Scholar]
- 22.Lopez-Arrieta JM, Schneider L. Metrifonate for Alzheimer's disease. Cochrane Database Syst Rev. 2006;2:Cd003155. doi: 10.1002/14651858.CD003155.pub3. [DOI] [PubMed] [Google Scholar]
- 23.Martin RJ, Puttachary S, Buxton SK, Verma S, Robertson AP. The Conqueror Worm: recent advances with cholinergic anthelmintics and techniques excite research for better therapeutic drugs. J Helminthol. 2015;89:387–97. doi: 10.1017/S0022149X1400039X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kaminsky R, Ducray P, Jung M, Clover R, Rufener L, Bouvier J, et al. A new class of anthelmintics effective against drug-resistant nematodes. Nature. 2008;452:176–80. doi: 10.1038/nature06722. [DOI] [PubMed] [Google Scholar]
- 25.Rufener L, Maser P, Roditi I, Kaminsky R. Haemonchus contortus acetylcholine receptors of the DEG-3 subfamily and their role in sensitivity to monepantel. PLoS Pathog. 2009;5:e1000380. doi: 10.1371/journal.ppat.1000380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Buxton SK, Charvet CL, Neveu C, Cabaret J, Cortet J, Peineau N, et al. Investigation of acetylcholine receptor diversity in a nematode parasite leads to characterization of tribendimidine- and derquantel-sensitive nAChRs. PLoS Pathog. 2014;10:e1003870. doi: 10.1371/journal.ppat.1003870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Harinasuta T, Bunnag D. Clinical field trial of bitoscanate in Necator americanus infection, South Thailand. Prog Drug Res. 1975;19:64–9. doi: 10.1007/978-3-0348-7090-0_8. [DOI] [PubMed] [Google Scholar]
- 28.Samuel MR. Clinical experience with bitoscanate. Prog Drug Res. 1975;19:96–107. doi: 10.1007/978-3-0348-7090-0_14. [DOI] [PubMed] [Google Scholar]
- 29.Pedras MS, Zheng QA, Gadagi RS. The first naturally occurring aromatic isothiocyanates, rapalexins A and B, are cruciferous phytoalexins. Chem Commun (Camb) 2007;4:368–70. doi: 10.1039/B615424G. [DOI] [PubMed] [Google Scholar]
- 30.Biagi F. Experience with bitoscanate in hookworm disease and trichuriasis in Mexico. Prog Drug Res. 1975: 23–27 [DOI] [PubMed]
- 31.Shapiro TA, Were JB, Talalay P, Bueding E, Rocco L, Danso K, et al. Clinical evaluation of amoscanate in healthy male volunteers. Am J Trop Med Hyg. 1986;35:945–53. doi: 10.4269/ajtmh.1986.35.945. [DOI] [PubMed] [Google Scholar]
- 32.Doshi JC, Vaidya AB, Sen HG, Mankodi NA, Nair CN, Grewal RS. Clinical trials of a new anthelmintic, 4-isothiocyanato-4′-nitrodiphenylamine (C.9333-Go/CGP 4540), for the cure of hookworm infection. Am J Trop Med Hyg. 1977;26:636–9. doi: 10.4269/ajtmh.1977.26.636. [DOI] [PubMed] [Google Scholar]
- 33.Craig TM, Mercer SH, Wade CG, Lynn RC. Efficacy of nitroscanate against naturally acquired infection with Ancylostoma caninum, Dipylidium caninum, and Trichuris vulpis in dogs. Am J Vet Res. 1991;52:574–5. [PubMed] [Google Scholar]
- 34.Nowicki D, Rodzik O, Herman-Antosiewicz A, Szalewska-Palasz A. Isothiocyanates as effective agents against enterohemorrhagic Escherichia coli: insight to the mode of action. Sci Rep. 2016;6:22263. doi: 10.1038/srep22263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Wu X, Zhou QH, Xu K. Are isothiocyanates potential anti-cancer drugs? Acta Pharmacol Sin. 2009;30:501–12. doi: 10.1038/aps.2009.50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Cully DF, Vassilatis DK, Liu KK, Paress PS, Van der Ploeg LH, Schaeffer JM, Arena JP. Cloning of an avermectin-sensitive glutamate-gated chloride channel from Caenorhabditis elegans. Nature. 1994;371:707–11. doi: 10.1038/371707a0. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets supporting the conclusion of this article are included within the article and its additional file.


