Abstract
Background
Sleep disturbance is common among patients with heart failure (HF) who also experience symptom burden and poor functional performance.
Objective
We evaluated the extent to which sleep-related, daytime symptoms (fatigue, excessive daytime sleepiness, and depressive symptoms) mediate the relationship between sleep disturbance and functional performance among patients with stable HF.
Methods
We recruited patients with stable HF for this secondary analysis of data from a cross-sectional, observational study. Participants completed unattended ambulatory polysomnography from which the respiratory disturbance index (RDI) was calculated; along with a Six-Minute Walk Test; questionnaires to elicit sleep disturbance (Pittsburgh Sleep Quality Index; Insomnia Symptoms from the Sleep Habits Questionnaire); daytime symptoms (Center for Epidemiologic Studies Depression Scale; Global Fatigue Index; Epworth Sleepiness Scale); and self-reported functional performance (Medical Outcomes Study SF36 V2 Physical Function Scale). We used structural equation modeling with latent variables for the key analysis. Follow-up, exploratory regression analysis with bootstrapped samples was used to examine the extent to which individual daytime symptoms mediated effects of sleep disturbance on functional performance after controlling for clinical and demographic covariates.
Results
The sample included 173 New York Heart Association Class I-IV HF patients (N = 60/34.7% women; M = 60.7, SD = 16.07 years of age). Daytime symptoms mediated the relationship between sleep disturbance and functional performance. Fatigue and depression mediated the relationship between insomnia symptoms and self-reported functional performance, while fatigue and sleepiness mediated the relationship between sleep quality and functional performance. Sleepiness mediated the relationship between the respiratory index and self-reported functional performance only in people who did not report insomnia.
Conclusions
Daytime symptoms explain the relationships between sleep disturbance and functional performance in stable HF.
Keywords: depression, fatigue, heart failure, insomnia, mediation, sleep, sleep apnea, sleepiness
Almost 75% of heart failure (HF) patients—a population of over 5 million Americans (Go et al., 2014)—and many more patients worldwide, report sleep disturbance (Redeker, Jeon et al., 2010; Redeker & Stein, 2006; Riegel et al., 2012). Sleep disturbance is more common among HF patients than “healthy” controls (Redeker & Stein, 2006) and the general population (National Sleep Foundation, 2005). Acute sleep disturbance may be related to poorly controlled HF, but chronic sleep disturbance, including insomnia symptoms such as difficulty with initiating sleep, maintaining sleep, and/or awakening too early in the morning, or nonrestorative sleep, is also common in these patients (Redeker, Jeon et al., 2010). Sleep-disordered breathing, including obstructive sleep apnea and Cheyne-Stokes Breathing/Central sleep apnea, occurs in approximately half of HF patients, but is not consistently associated with self-reported sleep disturbance (Redeker, Jeon et al., 2010).
Among HF patients, sleep disturbance contributes to poor quality of life (Andrews, Coviello, Hurley, Rose, & Redeker, 2013; C. M. Baldwin et al., 2001; Bröstrom, Stromberg, Dalstrom & Fridlund, 2004) and functional performance (Redeker, Jeon et al., 2010) of “the day to day corporeal activities people do in the normal course of their lives to meet basic needs, fulfill usual roles, and maintain health and wellbeing,” (Leidy, 1994, p. 198), including objective measures of exercise capacity (Mayou, Blackwood, Bryant, & Garnham, 1991) and self-reported physical function (C. Baldwin et al., 2001; Redeker & Hilkert, 2005; Redeker, Jeon et al., 2010). HF patients think sleep disturbance is critical to daytime function, but clinicians fail to address it (Andrews et al., 2013).
Although the restorative functions of sleep are not completely understood, fatigue, depression, and sleepiness are common in HF and are often consequences of sleep disturbance in many populations (Blinderman, Homel, Billings, Portenoy, & Tennstedt, 2008; Johansson et al., 2012; Jurgens et al., 2009; Lee et al., 2010; Redeker, Jeon et al., 2010; Redeker & Stein, 2006; Song, Moser, Rayens, & Lennie, 2010). These sleep-related symptoms may occur singly or in clusters during everyday life with HF (Jurgens et al., 2009; Lee et al., 2010; Song et al., 2010) and may contribute to voluntary or involuntary decrements in functional performance. The purpose of this report was to evaluate the extent to which sleep-related daytime symptoms (fatigue, sleepiness, and depressive symptoms) mediate the relationship between sleep disturbance and functional performance among patients with stable HF.
Methods
Design
The study comprised a secondary analysis of data from a cross-sectional, observational study conducted between the years 2003–2006. The overall purpose of the parent study was to evaluate the extent to which sleep and sleep disorders were associated with daytime symptoms and functional performance among patients with stable HF who were recruited from evidence-based HF disease management settings. Previous reports of the overall study included the direct effects of insomnia (Redeker, Jeon et al., 2010) and sleep disordered breathing (Redeker, Muench et al., 2010) on symptoms and function. The investigators also reported the relationships between pain and fatigue (Conley, Feder, & Redeker, 2015); the contributions of comorbid diabetes to symptoms and function (Fritschi & Redeker, 2015); and the associations of nocturia with sleep disturbance and function (Redeker et al., 2012). There have not been substantial changes in care of HF patients since the time of the study which likely affected the sleep, symptom, and functional performance variables.
Human subjects’ approval was obtained at the University of Medicine & Dentistry of New Jersey where the original study took place, each of the recruitment sites, and Yale University, where this secondary analysis was conducted. All participants provided written informed consent.
Sample
Participants included adults who were 18 years of age or older and had stable chronic New York Heart Class I-IV HF. They were recruited from five HF disease management programs in the Northeastern United States. Exclusion criteria included pregnancy, alcohol and/or illicit drug use, Parkinson’s disease, obstructive valvular, hypertrophic, or surgically correctable heart disease, previously diagnosed sleep disorders, and end-stage renal failure. Participants who had unstable medical or psychiatric conditions, active titration of vasoactive medications, hospitalizations, or emergency department visits for HF within the past month were not excluded, but participation was delayed until participants had been stable on medications and not admitted to the hospital or emergency department for HF for at least one month (Redeker, Jeon et al., 2010).
Variables and Measures
Functional performance
Because of our interest in the multidimensional aspects of functional performance, we measured self-reported (10-item Medical Outcomes Study Short Form-36 Physical Functional Scale) (Ware, Snow, Kosinski, & Gandek, 1993; Ware & Sherbourne, 1992) and objective physical function (Six-Minute Walk Test: 6MWT) (Guyatt et al., 1985). The 6MWT (Guyatt et al., 1985) measures the distance walked in six minutes in the clinic setting and is correlated with exercise performance (Lipkin, Scriven, Crake, & Poole-Wilson, 1986), functional capacity, and self-reported functional status (Guyatt et al., 1985). It was conducted with standard methods (Guyatt, 1987).
The Medical Outcomes Study SF-36 Physical Function Scale is widely used, valid, reliable, and demonstrated a coefficient alpha of .90 in HF patients (Redeker, Jeon et al., 2010). Norm-based scoring was used. A higher score indicates better function.
Daytime symptoms
We measured daytime symptoms that are often associated with sleep disturbance and common among patients with HF (Redeker, Jeon et al., 2010), including fatigue [Global Fatigue Index from the Multi-Dimensional Assessment of Fatigue Scale, developed from the Piper Fatigue Scale (Piper et al., 1989)], excessive daytime sleepiness [Epworth Sleepiness Scale (Johns, 1991, 1992)] and depressive symptoms [Centers for the Epidemiological Studies of Depression Scale (Devins & Orme, 1985; Radloff & Teri, 1986)]. These measures are widely used, well-validated, and internally consistent among HF patients (Redeker, Jeon et al., 2010).
Sleep disturbance
We used three characteristics to capture the multidimensional nature of sleep disturbance among patients with heart failure: global sleep quality, insomnia symptoms, and sleep-disordered breathing. Insomnia symptoms were evaluated with three questions from the Sleep Habits Questionnaire used in the Sleep Heart Health Study (Baldwin et al., 2001); each item was rated on a 5-item scale with response options ranging from 0 = never to 5 = almost always to indicate the frequency with which respondents experienced difficulty initiating or maintaining sleep and/or awakening too early in the morning. We computed a total insomnia score (possible score = 0–15) from the sum of these responses—coefficient alpha was 0.83 (Redeker, Jeon et al., 2010). We also used the score in a dichotomous manner in which the presence of insomnia was defined as a response of “often” or “almost always” to one or more of the three insomnia symptoms.
We measured sleep quality with the Pittsburgh Sleep Quality Index (PSQI; Buysse, Reynolds, Monk, Berman, & Kupfer, 1989), a well-validated and widely used measure of global sleep quality. The PSQI measures seven domains: subjective sleep quality; sleep latency; sleep duration; habitual sleep efficiency; sleep disturbances; use of sleep medication; and daytime dysfunction over the last month. The PSQI has a range of 0–21; a score of 5 or higher indicates poor sleep quality.
We also conducted full, unattended polysomnography (PSG) in participants’ homes for one night, and calculated the respiratory disturbance index (RDI) (sum of apneas, hypopneas, and respiratory event-related arousals/hour of sleep) as the primary measure of sleep disordered breathing—according to standard criteria (American Academy of Sleep Medicine Task Force, 1999; Redeker, Muench et al., 2010). A higher RDI indicates more severe sleep-disordered breathing. We previously reported full details of the acquisition and scoring of the polysomnographic data (Redeker, Muench et al., 2010).
Procedures
Clinic personnel obtained informed consent, reviewed medical records, and administered the 6MWT. Certified referred potential participants to the study. A research assistant explained the study, sleep technicians attached the PSG sensors and set up the recording devices in participants’ homes in the evening; PSG was recorded for one night, and the technician returned to pick up the equipment in the morning. Participants completed the study questionnaires while at home. The 6MWT was completed in the clinic setting.
Data Analysis
Data were double entered and converted to SAS data format; all analyses were performed with SAS version 9.3. Descriptive analyses were conducted to evaluate the distribution of demographic and clinical data and the outcome variables. Data analyses were performed with standardized scores computed from the sleep disturbance (insomnia symptoms, sleep quality, RDI), daytime symptoms (fatigue, sleepiness, depression symptoms), and self-reported and objective functional performance variables. Bivariate correlations between the sleep disturbance, daytime symptom, and functional performance variables were computed with Pearson’s correlation coefficient.
Main analysis
To examine the extent to which the daytime symptoms mediated the relationships between sleep disturbance and functional performance, we performed structural equation modeling (SEM) with PROC CALIS to estimate the direct effects of sleep disturbance (latent variable indicated by PSQI and insomnia scores) and the indirect (mediation) effects via daytime symptoms (sleepiness [ESS], depression [CESD], fatigue [GFI]) on functional performance (latent variable with indicators PF score and 6MWT distance). This model did not include covariates due to lack of a sufficient sample size. The model estimation was performed using the full information maximum likelihood (FIML) method to handle missing data. The parameter estimation was constrained by fixing the latent variances to 1. Goodness of fit was examined using the comparative fit index (CFI) and the root mean square error of approximation (RMSEA).
Follow-up exploration
As a follow-up to the SEM, and to further explore the relationships among the variables, we computed two regression models. In the first model, we examined the extent to which sleep disturbance predicted daytime symptoms; in the second regression model we evaluated the extent to which sleep disturbance and daytime symptoms predicted functional performance. In each model, we controlled for age, gender, New York Heart Association Functional Classification, and comorbidity because these potential confounders were associated with the sleep disturbance, daytime symptom, and/or functional performance variables. The direct effects were evaluated with the coefficient reflecting the association of sleep disturbance with functional performance, adjusted for a mediating variable. The indirect (mediation) effect was estimated as the product of the effect of sleep disturbance on a daytime symptom and the effect of a daytime symptom on functional performance after adjusting for a sleep disturbance variable. We used a bootstrapping approach to estimate the direct and indirect effects and 95% confidence intervals. We calculated direct and indirect effects with 2,000 bootstrap samples with replacement from the entire sample using PROC SURVEYSELECT, SAS version 9.3. The expected direct and indirect effects were obtained from the mean of 2,000 estimates, and 95% confidence intervals were obtained from the 2.5th and 97.5th percentiles of the estimates.
Results
We approached 324 patients, and 173 completed the study. Reasons for exclusion were previously reported (Redeker, Muench et al., 2010). Descriptive statistics for the demographic, clinical, sleep disturbance, daytime symptom, and functional performance variables are presented in Table 1. Some of these characteristics were previously reported (Redeker, Jeon et al., 2010; Redeker, Muench et al., 2010), but are repeated in this paper to facilitate understanding of the additional analyses presented here. Among the 61 participants who were minority group members, 50 were Black (Redeker, Jeon et al., 2010). There were no statistically significant differences in the primary study variables between study sites.
TABLE 1.
Sample Characteristics
| Characteristic | N | (%) |
|---|---|---|
| Demographic and clinical | ||
| Gender (male) | 113 | (65.3) |
| Race (White) | 110 | (64.3) |
| New York Heart Association class | ||
| I | 5 | (8.9) |
| II | 95 | (54.9) |
| III | 61 | (35.3) |
| IV | 12 | (6.9) |
| M | (SD) | |
| Age (years) | 60.4 | (16.07) |
| Left ventricular ejection fraction (%) | 32.6 | (15.20) |
| Body mass index | 30.7 | (8.03) |
| Charlson Comorbidity Index | 2.5 | (1.52) |
| Sleep disturbance | ||
| Insomnia symptoms (IS) | 6.0 | (3.10) |
| Global sleep quality (PSQI) | 8.7 | (4.20) |
| Respiratory disturbance index (RDI) | 24.3 | (19.00) |
| Daytime symptom | ||
| Fatigue (GFI) | 29.8 | (14.62) |
| Daytime sleepiness (ESS) | 8.3 | (4.34) |
| Depressive symptom (CESD) | 17.0 | (11.01) |
| Functional performance | ||
| Physical function (PF) | 22.7 | (2.17) |
| Six-minute walk (6MWT; feet) | 986 | (435) |
Note. N = 173. CESD = Center for Epidemiologic Studies—Depression Scale; ESS = Epworth Sleepiness Scale; GFI = Global Fatigue Index); IS = Insomnia Symptoms—Sleep Habits Questionnaire; PF = Physical Function Scale—Medical Outcome Studies Short Form-36; PSQI = Pittsburgh Sleep Quality Index.
The Pearson correlation coefficients for the associations between the sleep disturbance, daytime symptom, and functional performance variables are shown in Table 2. Insomnia symptoms and sleep quality were highly correlated with one another, and the RDI was negatively associated with insomnia, but not sleep quality. Insomnia symptoms and sleep quality were significantly associated with all of the daytime symptoms, while the RDI had a small, positive association with sleepiness. Sleep quality and insomnia symptoms were associated with self-reported physical function, but only sleep quality was associated with objective physical function (6MWT). The respiratory disturbance was not related to self-reported physical function in the bivariate analyses. Fatigue was significantly associated with self-reported and objective physical function, while sleepiness and depression were associated only with self-reported physical function. Self-reported and objective physical functions were moderately correlated with one another.
TABLE 2.
Sleep Disturbance, Daytime Symptoms, and Functional Performance: Pearson Correlations
| Variable | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | |
|---|---|---|---|---|---|---|---|---|---|
| Sleep disturbance | |||||||||
| 1. IS | Insomnia severity | 1.00 | |||||||
| 2. PSQI | Sleep quality | .69 | 1.00 | ||||||
| 3. RDI | Respiratory disturbance | −.18 | −.07 | 1.00 | |||||
| Daytime symptoms | |||||||||
| 4. GFI | Fatigue | .40 | .47 | −.05 | 1.00 | ||||
| 5. ESS | Daytime sleepiness | .17 | .24 | .17 | .27 | 1.00 | |||
| 6. CESD | Depressive symptoms | .45 | .49 | −.13 | .55 | .19 | 1.00 | ||
| Functional performance | |||||||||
| 7. PF (SF-36) | Physical function | −.27 | −.33 | −.09 | −.50 | −.28 | −.34 | 1.00 | |
| 8. 6MWT | Walk test | −.11 | −.22 | −.02 | −.26 | −.14 | −.13 | .47 | 1.00 |
Note. N = 173. 6MWT = Six-Minute Walk Test; CESD = Center for Epidemiologic Studies of Depression Scale; ESS = Epworth Sleepiness Scale; GFI = Global Fatigue Index; IS = Insomnia Symptoms—Sleep Habits Questionnaire; PF (SF-36) = Physical Function—Medical Outcomes Studies Short Form-36; PSQI = Pittsburgh Sleep Quality Index; RDI = Respiratory Disturbance Index.
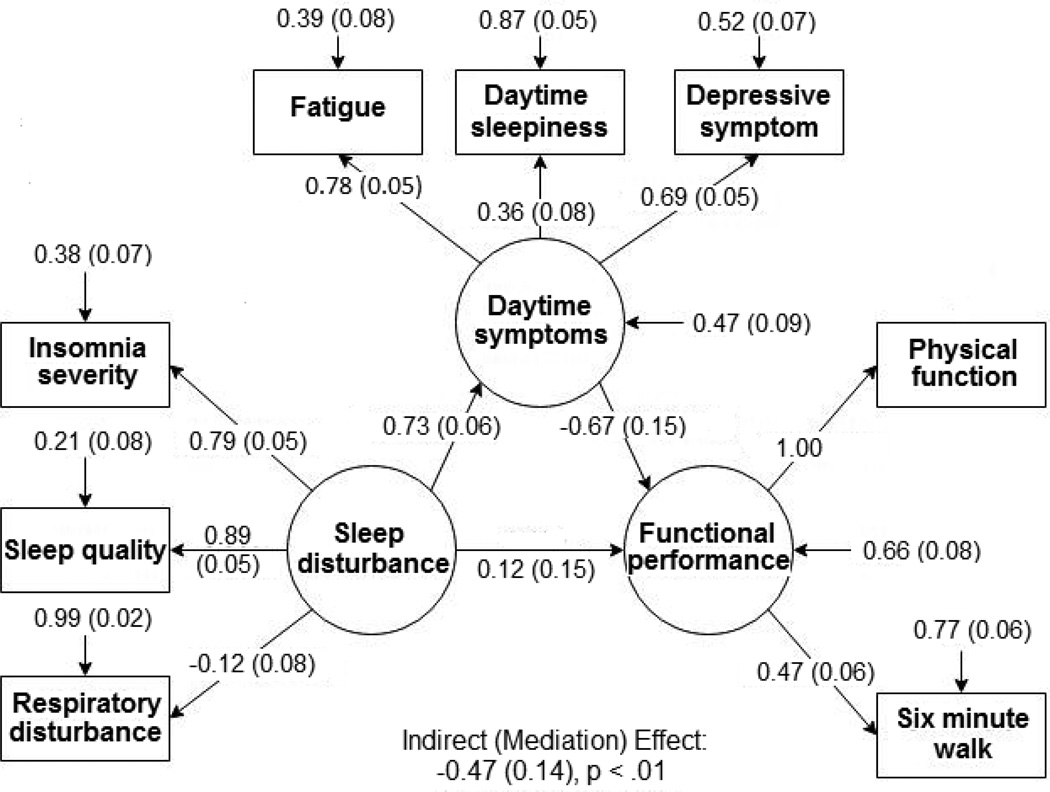
Figure 1 shows the results of the SEM in which we modeled the extent to which the latent variable of daytime symptoms (fatigue, sleepiness, and depression) mediated the relationship between the latent variable of sleep disturbance (insomnia severity, global sleep quality, respiratory disturbance) and the latent variable of functional performance. Due to a small error variance, an estimated error variance of physical function was negative. We addressed the problem by constraining its factor loading to 1. The model had an acceptable CFI of 0.96 and a RMSEA of 0.05 that indicates a moderate model fit (Bentler, 1990; Browne & Cudeck, 1993). The latent variable of sleep disturbance was well represented by the observed variables of insomnia severity and global sleep quality on sleep disturbance, but poorly represented by respiratory disturbance. The latent variables of daytime symptoms and functional performance were well represented by the observed self-reported variables. A higher level of sleep disturbance was significantly associated with more severe daytime symptoms (b* = 0.73, SE = 0.07, p < .0001). The overall indirect (mediation) effect through the daytime symptoms was statistically significant (mediation effect = −0.49, SE = 0.13, p =. 0002). The direct effect of sleep disturbance (direct effect = 0.12, SE = 0.15, p = .42) on functional performance was not statistically significant after adjusting for daytime symptoms. Thus, the association between sleep disturbance and functional performance was predominantly explained by the daytime symptoms.
FIGURE 1.
Standardized coefficients of structural equation model with daytime symptoms, sleep disturbance, and functional performance. The standardized coefficients are estimated by the full information maximum likelihood method.
Table 3 presents the estimated direct effects of each sleep disturbance variable on the functional performance variables and the indirect (mediation) effects of each daytime symptom between the individual sleep disturbance and functional performance variables. The coefficients and confidence intervals were estimated after adjusting for age, gender, New York Heart Association Functional Classification, and comorbidity from 2,000 bootstrap samples. Insomnia had significant indirect effects on self-reported physical function through fatigue and depressive symptoms on self-reported physical function; and sleep quality had significant indirect effects on self-reported physical function through fatigue and depressive symptoms. There were no statistically significant, indirect effects on objective physical function (6MWT) through the daytime symptoms.
TABLE 3.
Tests of Mediation for Daytime Symptoms between Sleep Disturbance and Functional Performance using Bootstrapping Methods
| DV: Physical functiona |
DV: 6MWT |
||||
|---|---|---|---|---|---|
| IV: Sleep disturbance | M: Daytime symptom | DE 95% CI |
IE 95% CI |
DE 95% CI |
IE 95%CI |
| Insomnia severity | Fatigueb | −.08 [−.20, .04] |
−.11** [−.18, −.06] |
−.02 [−.16, .12] |
−.05 [−.10, .003] |
| Daytime sleepinessc | −.15** [−.27, −.04] |
−.02 [−.05, .001] |
−.04 [−.05, .87] |
−.004 [−.03, .02] |
|
| Depressive symptomsd | −.11 [−.24, .02] |
−.06** [−.12, −.01] |
−.01 [−.16, .13] |
−.03 [−.09, .04] |
|
| Sleep quality | Fatigueb | −.15** [−.28, −.01] |
−.13** [−.20, −.06] |
−.15 [−.31, .02] |
−.04 [−.12, .02] |
| Daytime sleepinessc | −.19** [−.32, −.06] |
−.03** [−.07, −.001] |
−.13 [−.27, .01] |
−.002 [−.04, .03] |
|
| Respiratory disturbancee | Depressive symptomsd | −.17** [−.30, .03] |
−.06 [−.12, .001] |
−.12 [−.29, .05] |
−.01 [−.09, .05] |
| Fatigueb | −.11 [−.23, .07] |
−.01 [−.07, .04] |
.05 [−.09, .20] |
−.01 [−.04, .02] |
|
| Daytime sleepinessc | −.09 [−.21, .04] |
−.03** [−.06, −.003] |
.04 [−.10, .18] |
−.01 [−.03, .02] |
|
| Depressive symptomsd | −.13** [−.24, −.01] |
.03 [−.02, .05] |
.03 [−.11, .17] |
.01 [−.01, .03] |
|
Note. N = 173. The direct effects (effect of sleep disturbance on functional performance after controlling for daytime symptoms) and indirect effects (mediation effect through daytime symptoms) were estimated from 2000 bootstrap samples with replacement. The effects were adjusted for age, gender, New York Heart Association Functional Classification, and comorbidity. 6MWT = Six-Minute Walk Test; CESD = ; CI = confidence interval; DE = direct effect; DV = dependent variable; ESS = ; GFI = ; IE = indirect effect; IV = independent variable; M = mediator; RDI = Respiratory Disturbance Index.
Physical Function—Medical Outcomes SF-36 score.
GFI score.
ESS score.
CESD score.
RDI score.
p < .05.
The RDI was not associated with self-reported or objective physical function through any of the daytime symptoms. There was a statistically significant, indirect effect of daytime sleepiness between the RDI and self-reported physical function (Table 3). The weak associations of the RDI with sleepiness and self-reported physical function (Table 2) resulted in failure to detect a mediation effect. In the overall sample, the RDI was not associated with sleepiness (b* = 0.007, SE = .028, p = .81) or self-reported physical function (b = −.001, SE = .014, p = .96).
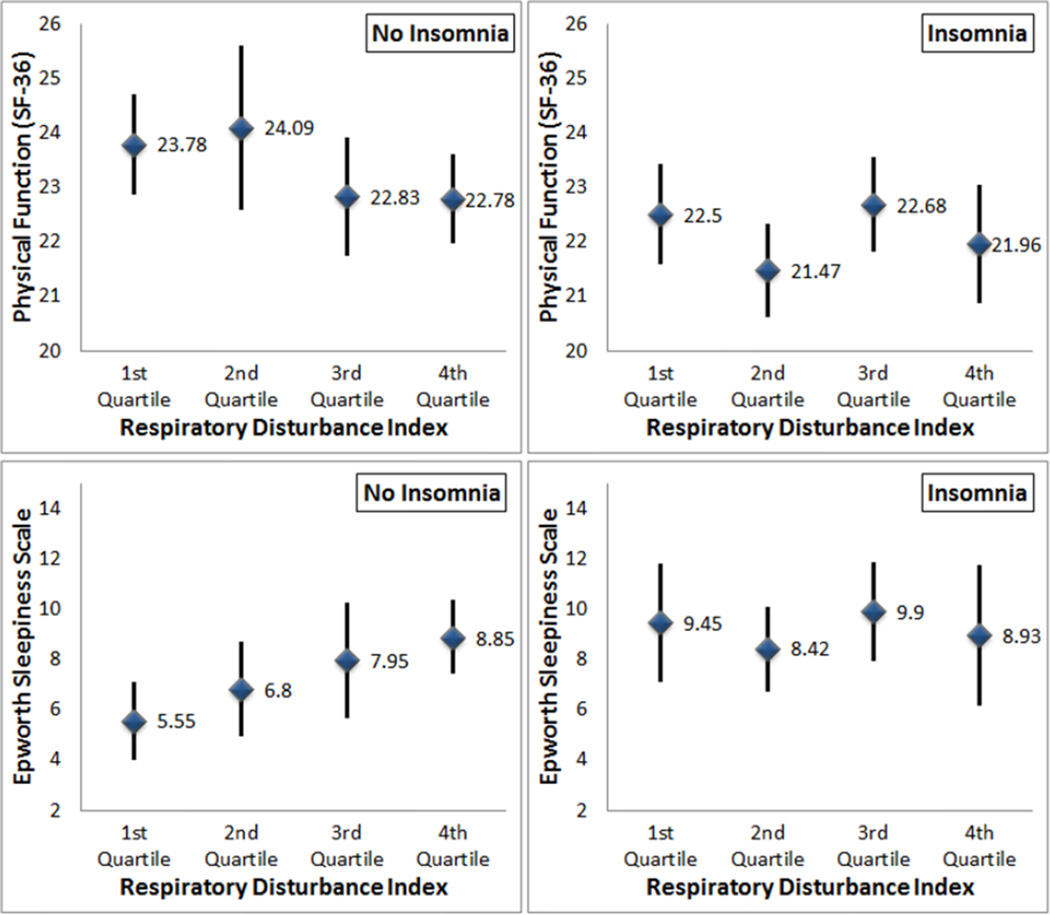
Participants with insomnia (half of the sample) (Redeker, Jeon et al., 2010) reported more severe sleepiness and poorer functional performance, compared to those without insomnia across all RDI levels. Therefore, we examined the associations between the RDI, sleepiness, and self-reported physical function separately in patients with and without insomnia. Figure 2 shows the means and 95% confidence intervals of self-reported physical function and sleepiness across quartiles of the RDI. Among patients without insomnia, the RDI was positively and significantly associated with sleepiness (r = .34) and negatively associated with self-reported physical function (r = −.20), but there were no relationships among these variables in people with insomnia (sleepiness: r = .03; self-reported physical function: r = −.01]. We found a stronger, indirect effect of sleepiness (indirect effect = −.08, 95% CI = [−.16, −.01]) between the RDI and self-reported physical function in the subset of patients without insomnia using the bootstrap samples.
FIGURE 2.
Self-reported physical function and daytime sleepiness by RDI quartiles in patients with and without insomnia.
Discussion
To our knowledge, this study is the first to examine the extent to which symptoms explain the relationships between sleep disturbance and functional performance in people with stable HF, and extends previous studies that focused only on the direct effects of insomnia and other attributes of sleep disturbance on individual and clustered symptoms and functional performance (Bröstrom et al., 2004; Lee et al., 2010; Redeker, 2006; Redeker, Jeon et al., 2010; Song et al., 2010). Our findings suggest that daytime symptoms, including depression, fatigue, and excessive daytime sleepiness, explained the relationships between self-reported sleep disturbance and functional performance. The mediation effects support the idea that daytime symptoms may be a causal pathway through which sleep disturbance contributes to functional performance. That is, people who have more severe sleep-related daytime symptoms have lower levels of functional performance.
As shown in the SEM, and further demonstrated in the regression models, symptoms primarily mediated the effects of self-reported sleep disturbance on self-reported, rather than objectively measured physical function. Several explanations for these discrepancies are plausible and may be related to differences in the physical function/functional performance constructs or the situations under which they were measured. For example, the SF-36 Physical function scale elicits self-reports of the frequencies with which activities are habitually performed during everyday life, while the 6MWT is a more specific measure of functional capacity (what the person is able to do), conducted “on demand” in the clinical setting at a single point in time. The negative influence of sleep-related symptoms on functional performance may be voluntary (i.e., people choose not to perform activities due to daytime symptoms) or involuntary (i.e., people are unable to perform activities). It is also possible that negative affectivity explains the stronger associations among the self-reported sleep disturbance, symptoms, and physical function variables compared with the associations with objective functional performance (6MWT). That is, people who perceive symptoms may be more likely to perceive poorer function. Sleep disturbance may be associated with or influence biological and motivational factors that contribute to both daytime symptoms and functional performance. Further studies are needed of the biobehavioral causal mechanisms underpinning the indirect effects observed in this study.
Excessive daytime sleepiness, in contrast to fatigue, is considered a specific symptom of sleep loss (Pigeon, Sateia, & Ferguson, 2003), and is a common outcome of sleep-disordered breathing (obstructive sleep apnea and/or Cheyne-Stokes Breathing/Central Sleep apnea), prevalent among the patients in this study (Redeker, Muench et al., 2010) and among HF patients in general. However, sleepiness had a smaller, indirect effect on the relationship between sleep disturbance and physical function than fatigue, and the RDI—an indicator of sleep disordered breathing—was negatively associated with insomnia and associated with sleepiness and physical function only in people without insomnia. The reasons for these relationships are not known, but may be explained by our earlier finding that the overall level of sleepiness was higher, and self-reported functional performance was lower in people with insomnia (Redeker, Jeon et al., 2010) compared to those without insomnia. Given that patients often do not fully recognize the extent of their sleepiness, additional study of the role of daytime sleepiness with objective measures, such as the psychomotor vigilance test, is needed to further explain these relationships.
Although insomnia and sleep-disordered breathing are frequently comorbid, especially in HF patients, the relative contributions (or interactive contributions) of these conditions to patient outcomes should be considered in future studies—especially given the unexpected, small, negative association between insomnia severity and RDI in this study.
Differences in the magnitudes and directions of the correlations among the individual sleep disturbance variables and with the individual symptom and functional performance variables likely reflect the multidimensional nature of objective and subjective sleep characteristics, and the different dimensions of sleep/sleep disturbance elicited by the respective sleep disturbance variables. For example, the RDI is a measure of sleep-disordered breathing (not sleep itself), while insomnia symptoms are self-reported phenomena that are symptoms or indicators of insomnia—a sleep disorder defined by self-report and self-reported sleep quality is a more general indicator. As with other symptoms, self-reports (e.g., insomnia severity and sleep disturbance) are frequently not closely associated with objective indicators (e.g., RDI).
The effects of sleep disturbance on functional performance, after controlling for clinical and demographic covariates, underscore the importance of sleep disturbance among HF patients—a group for whom daytime symptoms and functional performance are usually attributed primarily to the negative impact of HF itself, and suggest the potential benefits of sleep-promoting interventions to symptoms and functional performance in these vulnerable patients. For example, cognitive behavioral therapy for insomnia produced large effects on fatigue among HF patients (Redeker et al., 2015), and improved biomarkers of cardiac risk (Carroll et al., 2015; Irwin et al., 2015; Irwin et al., 2014) among a sample of older adults of whom the majority had cardiovascular disorders. Meditation and guided imagery may also be efficacious in improving sleep and sleep-related symptoms among these patients (Kwekkeboom & Bratzke, 2015). Further studies are needed of the extent to which behavioral interventions for insomnia improve symptoms (Conley & Redeker, 2015) and how, in turn, these improvements lead to improved functional performance. Recent evidence that insomnia predicts incident HF and death (Laugsand, Strand, Platou, Vatten, & Janszky, 2014) further underscore the importance of understanding these relationships.
Limitations
Although the findings of this study suggest the meditational role of symptoms, the cross-sectional nature of the study precludes full understanding of the causal nature of the observed relationships among the study variables. Future longitudinal studies and randomized controlled trials are needed to confirm the causal nature these findings. Cross-sectional studies could produce biased estimates of parameters for longitudinal mediation especially when there is a perfect mediation (e.g., no direct effect of sleep disturbance on functional performance). Therefore, future studies could use methods such as a cross-lagged panel model for longitudinal data to reduce the biased parameters of mediation in future study (MacKinnon, 2008; Maxwell & Cole, 2007).
A limitation of the regression analysis, in which we tested multiple models, is the possibility of type I error and small sample size that may have limited the ability to detect statistically significant effects. This may have also explained the discrepancies between the specific symptoms that mediated the effects of insomnia (fatigue and depressive symptoms) versus sleep quality (fatigue and excessive daytime sleepiness) on physical function. However, taken together, the findings of these analyses are consistent with the results of the SEM in which type I error is not a concern due to the simultaneous entry of the variables into the model. Nevertheless, future studies should be conducted with larger samples that may have more power to detect significant effects.
Conclusions and Recommendations
Taken together, the findings of this study suggest that daytime symptoms explain the relationships between sleep disturbance and functional performance among patients with stable HF. Future studies are needed to improve understanding of the biobehavioral mechanisms for these relationships, confirm the suggested causal effects, and examine the effects of sleep promoting interventions on sleep disturbance, daytime symptoms, and functional performance.
Acknowledgments
The authors acknowledge that this project was funded by NIH R01NR08022 & NIH P20 NR014126 (Nancy S. Redeker, PI).
Footnotes
The authors have no conflicts of interest to report.
Contributor Information
Sangchoon Jeon, Research Scientist, Yale School of Nursing, West Haven, CT.
Nancy S. Redeker, Beatrice Renfield Term Professor of Nursing, Yale School of Nursing, West Haven, CT.
References
- American Academy of Sleep Medicine Task Force. Sleep-related breathing disorders in adults: Recommendations for syndrome definition and measurement techniques in clinical research. Sleep. 1999;22:662–689. [PubMed] [Google Scholar]
- Andrews LK, Coviello J, Hurley E, Rose L, Redeker NS. “I’d eat a bucket of nails if you told me it would help me sleep:” Perceptions of insomnia and its treatment in patients with stable heart failure. Heart & Lung. 2013;42:339–345. doi: 10.1016/j.hrtlng.2013.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baldwin C, Griffith KA, Nieto FJ, O’Connor GT, Walsleben JA, Redline S. The association of sleep-disordered breathing and sleep symptoms with quality of life in the Sleep Heart Health Study. Sleep. 2001;24:96–105. doi: 10.1093/sleep/24.1.96. [DOI] [PubMed] [Google Scholar]
- Bentler PM. Comparative fit indexes in structural models. Psychological Bulletin. 1990;107:238–246. doi: 10.1037/0033-2909.107.2.238. [DOI] [PubMed] [Google Scholar]
- Blinderman CD, Homel P, Billings JA, Portenoy RK, Tennstedt SL. Symptom distress and quality of life in patients with advanced congestive heart failure. Journal of Pain and Symptom Management. 2008;35:594–603. doi: 10.1016/j.jpainsymman.2007.06.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bröstrom A, Strömberg A, Dahlström U, Fridlund B. Sleep difficulties, daytime sleepiness, and health-related quality of life in patients with chronic heart failure. Journal of Cardiovascular Nursing. 2004;19:234–242. doi: 10.1097/00005082-200407000-00003. [DOI] [PubMed] [Google Scholar]
- Browne MW, Cudeck R. Alternative ways of assessing model fit. In: Bollen K, Long JS, editors. Testing structural equation models. Newbury Park, NJ: Sage; 1993. pp. 136–162. [Google Scholar]
- Buysse DJ, Reynolds CF, III, Monk TH, Berman SR, Kupfer DJ. The Pittsburgh Sleep Quality Index: A new instrument for psychiatric practice and research. Psychiatry Research. 1989;28:193–213. doi: 10.1016/0165-1781(89)90047-4. [DOI] [PubMed] [Google Scholar]
- Carroll JE, Seeman TE, Olmstead R, Melendez G, Sadakane R, Bootzin R, Irwin MR. Improved sleep quality in older adults with insomnia reduces biomarkers of disease risk: Pilot results from a randomized controlled comparative efficacy trial. Psychoneuroendocrinology. 2015;55:184–192. doi: 10.1016/j.psyneuen.2015.02.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Conley S, Feder S, Redeker NS. The relationship between pain, fatigue, depression and functional performance in stable heart failure. Heart & Lung. 2015;44:107–112. doi: 10.1016/j.hrtlng.2014.07.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Conley S, Redeker NS. Cognitive behavioral therapy for insomnia in the context of cardiovascular conditions. Current Sleep Medicine Reports. 2015;1:157–165. doi: 10.1007/s40675-015-0019-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Devins G, Orme C. Center for Epidemiologic Studies Depression Scale. In: Keyser DJ, Sweetland RC, editors. Test critiques. II. Kansas City, MO: Test Corporation of American; 1985. pp. 144–160. [Google Scholar]
- Fritschi C, Redeker NS. Contributions of comorbid diabetes to sleep characteristics, daytime symptoms, and physical function among patients with stable heart failure. Journal of Cardiovascular Nursing. 2015;30:411–419. doi: 10.1097/JCN.0000000000000183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Go AS, Mozaffarian D, Roger VL, Benjamin EJ, Berry JD, Blaha MJ, Fullerton HJ. Heart disease and stroke statistics—2014 update: A report from the American Heart Association. Circulation. 2014;129:399–410. doi: 10.1161/01.cir.0000442015.53336.12. [DOI] [PubMed] [Google Scholar]
- Guyatt GH, Thompson PJ, Berman LB, Sullivan MJ, Townsend M, Jones NL, Pugsley SO. How should we measure function in patients with chronic heart and lung disease? Journal of Chronic Diseases. 1985;38:517–524. doi: 10.1016/0021-9681(85)90035-9. [DOI] [PubMed] [Google Scholar]
- Irwin MR, Olmstead R, Breen EC, Witarama T, Carrillo C, Sadeghi N, Cole S. Cognitive behavioral therapy and tai chi reverse cellular and genomic markers of inflammation in late-life insomnia: A randomized controlled trial. Biological Psychiatry. 2015;78:721–729. doi: 10.1016/j.biopsych.2015.01.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Irwin MR, Olmstead R, Carrillo C, Sadeghi N, Breen EC, Witarama T, Nicassio P. Cognitive behavioral therapy vs. tai chi for late life insomnia and inflammatory risk: A randomized controlled comparative efficacy trial. Sleep. 2014;37:1543–1552. doi: 10.5665/sleep.4008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johansson P, Riegel B, Svensson E, Bröstrom A, Alehagen U, Dahlström U, Jaarsma T. The contribution of heart failure to sleep disturbances and depressive symptoms in older adults. Journal of Geriatric Psychiatry and Neurology. 2012;25:179–187. doi: 10.1177/0891988712458366. [DOI] [PubMed] [Google Scholar]
- Johns MW. A new method for measuring daytime sleepiness: The Epworth sleepiness scale. Sleep. 1991;14:540–545. doi: 10.1093/sleep/14.6.540. [DOI] [PubMed] [Google Scholar]
- Johns MW. Reliability and factor analysis of the Epworth Sleepiness Scale. Sleep. 1992;15:376–381. doi: 10.1093/sleep/15.4.376. [DOI] [PubMed] [Google Scholar]
- Jurgens CY, Moser DK, Armola R, Carlson B, Sethares K, Riegel B. Symptom clusters of heart failure. Research in Nursing Health. 2009;32:551–560. doi: 10.1002/nur.20343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kwekkeboom KL, Bratzke LC. A systematic review of relaxation, meditation, and guided imagery strategies for symptom management in heart failure. Journal of Cardiovascular Nursing. 2015 doi: 10.1097/JCN.0000000000000274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laugsand LE, Strand LB, Platou C, Vatten LJ, Janszky I. Insomnia and the risk of incident heart failure: A population study. European Heart Journal. 2014;35:eht019. doi: 10.1093/eurheartj/eht019. [DOI] [PubMed] [Google Scholar]
- Lee KS, Song EK, Lennie TA, Frazier SK, Chung ML, Heo S, Moser DK. Symptom clusters in men and women with heart failure and their impact on cardiac event-free survival. Journal of Cardiovasc Nursing. 2010;25:263–272. doi: 10.1097/JCN.0b013e3181cfbb88. [DOI] [PubMed] [Google Scholar]
- Leidy NK. Functional status and the forward progress of merry-go-rounds: Toward a coherent analytical framework. Nursing Research. 1994;43:196–202. [PubMed] [Google Scholar]
- Lipkin DP, Scriven AJ, Crake T, Poole-Wilson PA. Six minute walking test for assessing exercise capacity in chronic heart failure. British Medical Journal (Clinical Research Edition) 1986;292:653–655. doi: 10.1136/bmj.292.6521.653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MacKinnon DP. Introduction to statistical mediation models. New York, NY: Erlbaum; 2008. pp. 193–236. [Google Scholar]
- Maxwell SE, Cole DA. Bias in cross-sectional analyses of longitudinal mediation. Psychological Methods. 2007;12:23–44. doi: 10.1037/1082-989X.12.1.23. [DOI] [PubMed] [Google Scholar]
- Mayou R, Blackwood R, Bryant B, Garnham I. Cardiac failure: Symptoms and functional status. Journal of Psychosomatic Research. 1991;35:399–407. doi: 10.1016/0022-3999(91)90035-m. [DOI] [PubMed] [Google Scholar]
- Pigeon WR, Sateia MJ, Ferguson RJ. Distinguishing between excessive daytime sleepiness and fatigue: Toward improved detection and treatment. Journal of Psychosomatic Research. 2003;54:61–69. doi: 10.1016/s0022-3999(02)00542-1. [DOI] [PubMed] [Google Scholar]
- Piper BF, Lindsey AM, Dodd MJ, Ferketich S, Paul SM, Wellver S. The development of an instrument to measure the subjective dimensions of fatigue. In: Funk SG, Tornquist E, Champagne M, Wiese R, editors. Key aspects of comfort: Management of pain, fatigue, and nausea, Part III. New York, NY: Springer; 1989. pp. 199–208. [Google Scholar]
- Radloff LS, Teri L. 6/Use of the Center for Epidemiological Studies-Depression Scale with older adults. Clinical Gerontology. 1986;5:119–136. [Google Scholar]
- Redeker NS. Somatic symptoms explain differences in psychological distress in heart failure patients vs a comparison group. Progress in Cardiovascular Nursing. 2006;21:182–189. doi: 10.1111/j.0889-7204.2006.05643.x. [DOI] [PubMed] [Google Scholar]
- Redeker NS, Adams L, Berkowitz R, Blank L, Freudenberger R, Gilbert M, Rapoport D. Nocturia, sleep and daytime function in stable heart failure. Journal of Cardiac Failure. 2012;18:569–575. doi: 10.1016/j.cardfail.2012.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Redeker NS, Hilkert R. Sleep and quality of life in stable heart failure. Journal of Cardiac Failure. 2005;11:700–704. doi: 10.1016/j.cardfail.2005.07.003. [DOI] [PubMed] [Google Scholar]
- Redeker NS, Jeon S, Andrews L, Cline J, Jacoby D, Mohsenin V. Feasibility and efficacy of a self-management intervention for insomnia in stable heart failure. Journal of Clinical Sleep Medicine. 2015;11:1109–1119. doi: 10.5664/jcsm.5082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Redeker NS, Jeon S, Muench U, Campbell D, Walsleben J, Rapoport DM. Insomnia symptoms and daytime function in stable heart failure. Sleep. 2010;33:1210–1216. doi: 10.1093/sleep/33.9.1210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Redeker NS, Muench U, Zucker MJ, Walsleben J, Gilbert M, Freudenberger R, Rapoport DM. Sleep disordered breathing, daytime symptoms, and functional performance in stable heart failure. Sleep. 2010;33:551–560. doi: 10.1093/sleep/33.4.551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Redeker NS, Stein S. Characteristics of sleep in patients with stable heart failure versus a comparison group. Heart & Lung. 2006;35:252–261. doi: 10.1016/j.hrtlng.2005.10.007. [DOI] [PubMed] [Google Scholar]
- Riegel B, Glaser D, Richards K, Sayers SL, Marzolf A, Weintraub WS, Goldberg LR. Modifiable factors associated with sleep dysfunction in adults with heart failure. European Journal of Cardiovascular Nursing. 2012;11:402–409. doi: 10.1016/j.ejcnurse.2011.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Song EK, Moser DK, Rayens MK, Lennie TA. Symptom clusters predict event-free survival in patients with heart failure. Journal of Cardiovascular Nursing. 2010;25:284–291. doi: 10.1097/JCN.0b013e3181cfbcbb. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ware JE, Snow KK, Kosinski M, Gandek B. SF-36 Health survey: Manual and interpretation guide. Boston, MA: Health Institute, New England Medical Center; 1993. [Google Scholar]
- Ware JE, Jr, Sherbourne CD. The MOS 36-item short-form health survey (SF-36): I. Conceptual framework and item selection. Medical Care. 1992;30:473–483. http://www.jstor.org/stable/3765916. [PubMed] [Google Scholar]