Abstract
The atypical antipsychotic risperidone (RSP) is often associated with weight gain and cardiometabolic side effects. The mechanisms for these adverse events are poorly understood and, undoubtedly, multifactorial in etiology. In light of growing evidence implicating the gut microbiome in the host's energy regulation and in xenobiotic metabolism, we hypothesized that RSP treatment would be associated with changes in the gut microbiome in children and adolescents. Thus, the impact of chronic (>12 months) and short-term use of RSP on the gut microbiome of pediatric psychiatrically ill male participants was examined in a cross-sectional and prospective (up to 10 months) design, respectively. Chronic treatment with RSP was associated with an increase in body mass index (BMI) and a significantly lower ratio of Bacteroidetes:Firmicutes as compared with antipsychotic-naïve psychiatric controls (ratio=0.15 vs 1.24, respectively; P<0.05). Furthermore, a longitudinal observation, beginning shortly after onset of RSP treatment, revealed a gradual decrease in the Bacteroidetes:Firmicutes ratio over the ensuing months of treatment, in association with BMI gain. Lastly, metagenomic analyses were performed based on extrapolation from 16S ribosomal RNA data using the software package, Phylogenetic Investigation of Communities by Reconstruction of Unobserved States (PICRUSt). Those data indicate that gut microbiota dominating the RSP-treated participants are enriched for pathways that have been implicated in weight gain, such as short-chain fatty acid production.
Introduction
Over the last two decades, the prescribing rate of second-generation antipsychotic medications (SGAs) to treat children has increased nearly eightfold, in light of evidence supporting their efficacy.1, 2, 3 However, significant weight gain and associated cardiometabolic disturbances are common side effects, placing patients at an elevated risk for type 2 diabetes and cardiovascular disease.1, 4, 5, 6 Alarmingly, the life expectancy of patients with severe mental illness is 20–25 years shorter than that of the general population, primarily due to increased cardiovascular disease.7, 8, 9
The mechanisms underlying SGA-induced weight gain are not well understood. The metabolic side effects are believed to be multifactorial in their etiology, including pharmacological, genetic, lifestyle and environmental contributors.10, 11, 12 Recent evidence also implicates the gut microbiome, which impacts various aspects of metabolism, including energy harvest and the development of obesity.3, 13, 14, 15, 16, 17 Davey et al. have shown that acute olanzapine treatment in rats resulted in significant weight gain and cardiometabolic dysfunction in association with changes in the gut microbiota.1, 2, 3 Importantly, mice lacking a microbiota do not gain as much weight compared with non-treated controls.18
The interaction between the gut microbiota and SGAs may well be bidirectional. In fact, gut bacteria utilize neurotransmitter signaling pathways targeted by antipsychotic medications.19, 20, 21 On the other hand, they have a substantial role in xenobiotic pharmacokinetics. For example, the cleavage of the benzisoxazole ring system of risperidone (RSP) is a process effected primarily by the intestinal microbiota, yielding alternative metabolites.7 Microbiota-mediated mechanisms such as these can contribute to the emergence of adverse drug events, including weight gain.7, 8, 9, 22
In contrast to olanzapine, whose use in adolescents has been discouraged by the Food and Drug Administration due to its propensity to cause significant weight gain,10, 11, 12 RSP is one of the most commonly prescribed SGAs, particularly in children and adolescents.2, 3 RSP is, nonetheless, associated with a moderately high potential to induce weight gain.4 Thus, we performed a cross-sectional examination of the impact of chronic RSP exposure in children and adolescents on gut bacterial composition and on the corresponding metabolic pathways. We further explored the longitudinal dynamics of the gut microbiota following the onset of RSP treatment. Our results indicate that RSP use is associated with alteration of the gut microbiome that is enriched for metabolic pathways that might cause or exacerbate weight gain.
Materials and methods
Sample collection
The cross-sectional study involved 18, 9–15-year-old, medically healthy males who had been on RSP for at least 1 year (chronic RSP group). The longitudinal study consisted of five males, aged 9–13 years, enrolled within 1 month of starting RSP. Ten 10–14-year-old participants, who were psychiatrically ill but not receiving any SGAs, served as controls. Height and weight were measured following standard guidelines during the research visits.23 Body mass index (BMI) was calculated and age–sex-specific BMI Z-scores were computed to account for natural growth.24 When available, anthropometric data were also extracted from the medical records to compute baseline BMI Z-score for RSP-treated participants within −31 to +3 days of RSP initiation.23 This allowed estimating the change in BMI Z-score between the time RSP was started and study visit. In order to do the same for the psychiatric control group, baseline BMI measurements were determined from the medical records to provide a comparable mean±s.d. interval between baseline BMI and BMI at time of stool sample collection across treatment groups. Stool samples were collected once in the cross-sectional study, and as frequently as every month in the longitudinal study. Stool samples were collected freshly and transported to the lab where they were aliquoted and frozen within 15–30 min. Alternatively, they were provided at home, placed on dry ice supplied by research staff and picked up within 24 h (while still frozen). Samples were stored at −80 °C until DNA extraction and were excluded from the study when antibiotic use occurred within 6 months of sample collection. The studies were approved by the Institutional Review Board at the University of Iowa.
Multiplex DNA sequencing and assembly
Extracted genomic DNA was subjected to multiplex paired-end Illumina (Illumina, San Diego, CA, USA) sequencing of the V1-2 region of bacterial 16S ribosomal RNA (rRNA) genes with a MiSeq instrument (Chapel Hill, NC, USA) in the UNC Microbiome Core Facility. See Supplemental Methods for further details about the analysis.
Results
Eighteen male participants, mean age 12.2 (s.d.=2.5) years, provided stool samples for the cross-sectional study, after a mean of 3.6 (s.d.=2.4) years of RSP treatment. In addition to RSP, participants also used psychostimulants (n=18, 100%), α-2 agonists (n=12, 66%) and selective serotonin reuptake inhibitors (n=2, 11%). Over the course of treatment, BMI Z-score increased by a mean of 0.31 (s.d.=1.11) points. Ten boys who had not received RSP served as psychiatric controls. Their mean age was 12.0 (s.d.=1.8) years. They were taking psychostimulants (n=7, 70%), α-2 agonists (n=3, 30%) and selective serotonin reuptake inhibitors (n=2, 20%). Over a comparable time interval (see Methods section), their BMI Z-score remained virtually unchanged (mean ΔBMI Z-score=0.09, s.d.=0.61). Psychiatric diagnoses and gastrointestinal symptoms were not different between the two groups, nor was food intake (Supplementary Table 1).
The longitudinal study consisted of five participants aged between 9 and 13 years (mean=11.7, s.d.=1.1), with a mean BMI Z-score at study entry of 0.12 (s.d.=0.84). Participants provided a stool sample within a few days (mean=3.2, s.d.=5.2) of starting RSP treatment and then up to monthly, for 10 months. Over the course of the treatment, BMI Z-scores increased by a mean of 0.28 (s.d.=0.23) units. All five participants were additionally taking psychostimulants and three of them (60%) were also taking α-2 agonists.
Chronic RSP treatment is associated with a distinct gut microbiome
To evaluate whether microbial diversity is altered during chronic treatment with RSP, we surveyed fecal bacterial populations using 16S rRNA sequencing. The chronic RSP group displayed a significantly higher Shannon diversity when compared with psychiatric control participants (5.9 vs 5.2, respectively, P<0.05; Supplementary Figures 1A and B). There was a higher phylogenetic diversity (P=0.05; Supplementary Figures 1C and D) as well as a numerically, but not statistically, increased number of observed species in psychiatric control participants (Supplementary Figures 1E and F).
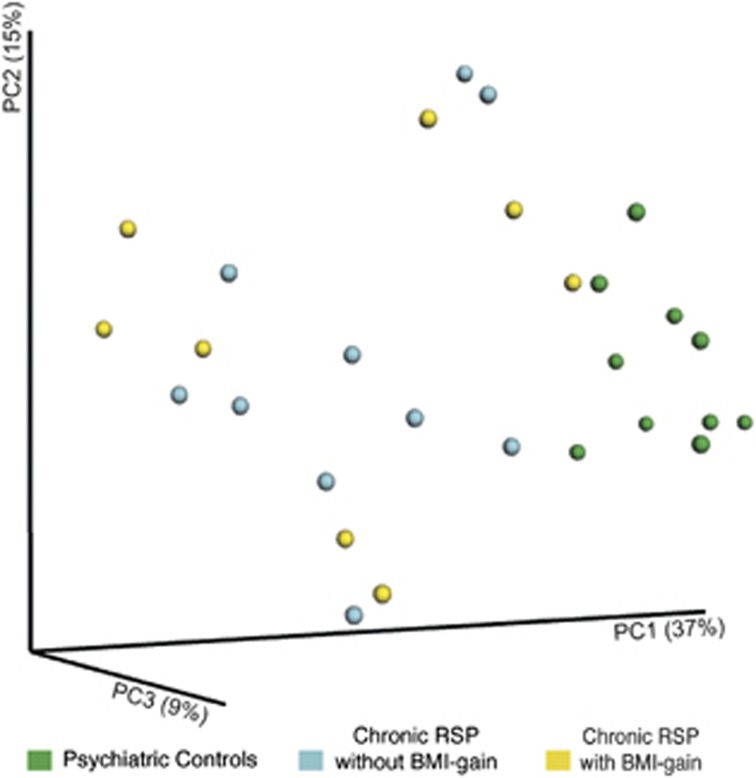
Using UniFrac, a phylogenetic distance metric, participants in the chronic RSP group were significantly different from psychiatric controls (analysis of similarity (ANOSIM) R=0.516, P<0.05). This indicates that a robust difference exists between the overall gut microbial profiles of the two groups (Figure 1). However, using this same metric among participants taking RSP, there was no significant difference (ANOSIM R=0.001, P=0.41) in microbial profiles between those who had a significant BMI gain (n=10), defined as a BMI Z-score increase by ⩾0.5 units, and those who did not (n=8; Supplementary Figure 2B).
Figure 1.
Differences in the fecal microbial communities in chronic risperidone (RSP)-treated participants vs psychiatric controls. PcoA of unweighted UniFrac distances between psychiatric control participants (green), chronic RSP-treated participants with significant body mass index (BMI) gain (blue, i.e., an increase in age−sex-specific BMI Z-score⩾0.5 units since starting RSP), and RSP-treated participants without significant BMI gain (yellow). Each point shows the average distance between individuals. Results are derived from bacterial V1−V2 16S rRNA data sets. ANOSIM R=0.5169, P=0.0001. PCoA, principal component analysis.
Chronic and acute treatment with RSP are associated with weight gain and alterations in gut bacterial composition
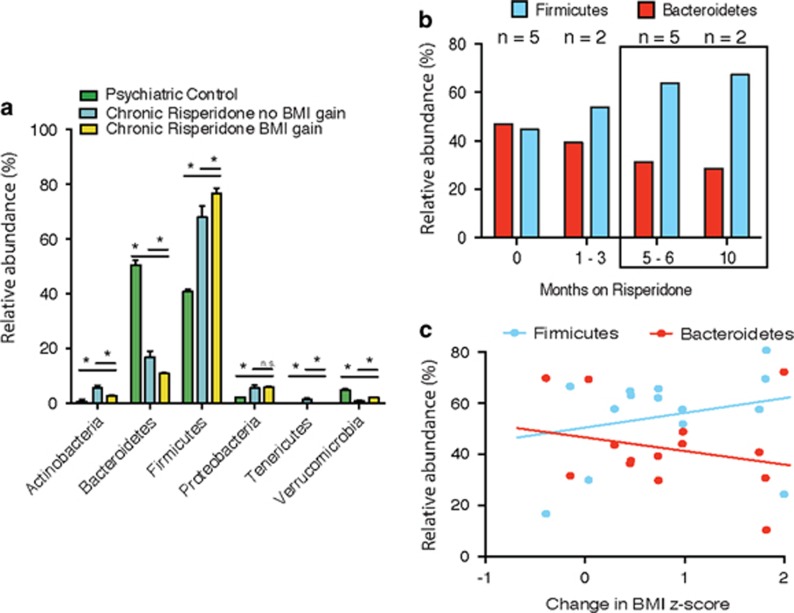
The most common bacterial phyla in healthy adults are Firmicutes and Bacteroidetes, with significant representation of Actinobacteria and Proteobacteria. The relative proportions of these phyla sometimes diverge widely, reflecting not only interpersonal, geographical and lifestyle variations but also perturbations caused by disease.25 In our study, the psychiatric control participants displayed a distribution of the two major gut phyla, Firmicutes and Bacteroidetes, comparable to that observed in healthy populations26 (Figure 2a and Supplementary Table 2). In contrast, participants treated long-term with RSP and who also displayed a significant BMI gain had significantly less Bacteroidetes than Firmicutes relative to controls (ratio=0.20 vs 1.24, respectively, P<0.05). Importantly, significant differences in bacterial composition also differentiated RSP-treated participants who exhibited significant BMI gain from those who did not (Figure 2a and Supplementary Table 2). Finally, the bacterial composition within each of the two RSP treatment groups (regardless of changes in BMI Z-score) was different from that found in psychiatric controls (Figure 2a and Supplementary Table 2). Thus, both long-term RSP treatment as well as significant RSP-related BMI gain are independently associated with an altered distribution of the gut microbiota.
Figure 2.
Phyla-level abundances. (a) Phyla-level abundances in psychiatric controls (green), chronic risperidone (RSP)-treated participants with significant body mass index (BMI) gain (blue, that is, an increase in age–sex-specific BMI Z-score ⩾0.5 units since starting RSP) and chronic RSP-treated participants without significant BMI gain (yellow). (b) Trajectory of change over time in the two major gut bacterial phyla, Bacteroidetes and Firmicutes, following the initiation of RSP. (c) Correlation of change in percent abundance of Bacteroidetes and Firmicutes, and change in BMI Z-score following the initiation of RSP.
Data from sequential stool sampling revealed an increase in Firmicutes relative to Bacteroidetes, apparent within 1–3 months after initiating RSP treatment, that is, with the first follow-up stool sample (Figure 2b). The trend became more prominent over a longer period of exposure. Importantly, the percent abundance of Firmicutes and Bacteroidetes appeared to correlate with the RSP-induced increase in BMI Z-score, albeit not significantly, likely owing to the small sample size (for Bacteroidetes: Spearman's r=−0.29, P=0.30; for Firmicutes: r=0.32, P=0.26; Figure 2c).
Discriminatory OTUs are detectable within treatment groups associated with weight gain
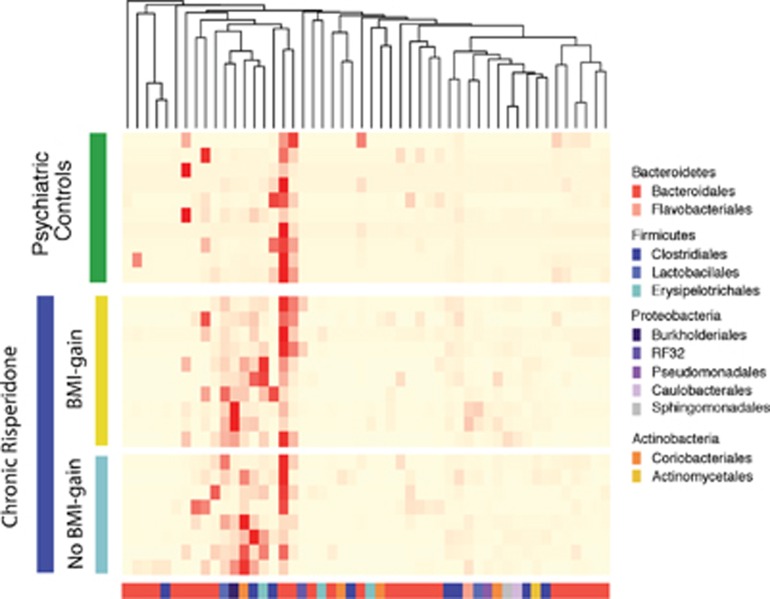
Fifty key operational taxonomic units (OTUs) were identified as sufficient to discriminate between psychiatric controls and chronic RSP participants (Figure 3). Only three genera, belonging to the phylum Bacteroidetes, were more abundant in the psychiatric controls. In contrast, 47 OTUs were significantly more prevalent in the RSP participants, the most abundant being Clostridium sp., Collinsella aerofaciens, Lactobacillus sp., Ralstonia sp. and the Erysipelotrichaceae family.
Figure 3.
Relative abundance of unique genus-level operational taxonomic units (OTUs) of the gut microbiota of chronic risperidone (RSP)-treated participants and psychiatric controls. Fifty unique OTUs were defined by three methods (Metastats comparison, random forests algorithm and linear discriminant analysis effect size analysis). Genus-level OTUs are colored by phyla (bottom, x-axis) and individual biological replicates (left, green or blue), where red hues denote increased relative abundance of a unique OTU. BMI, body mass index.
Importantly, Clostridium sp., Lactobacillus sp., Ralstonia sp. and Erysipelotrichaceae family members were more abundant in participants in the chronic RSP group who also displayed a significant BMI gain. In contrast, the order Coriobacteriales, specifically C. aerofaciens, was more abundant in participants within the chronic RSP group who did not show a significant BMI gain. These results suggest that specific Firmicutes and Burkholderiales species may have a role in RSP-induced weight gain while Coriobacteriales may be protective. In addition, Ruminococcaceae family members were most relatively abundant in RSP participants with significant BMI gain compared with psychiatric controls, whereas Bacteroidetes spp. were most relatively abundant in psychiatric controls compared with RSP participants regardless of BMI gain (Supplementary Figure 3).
Gene frequency reveals altered microbial metabolic functionality following chronic treatment with RSP
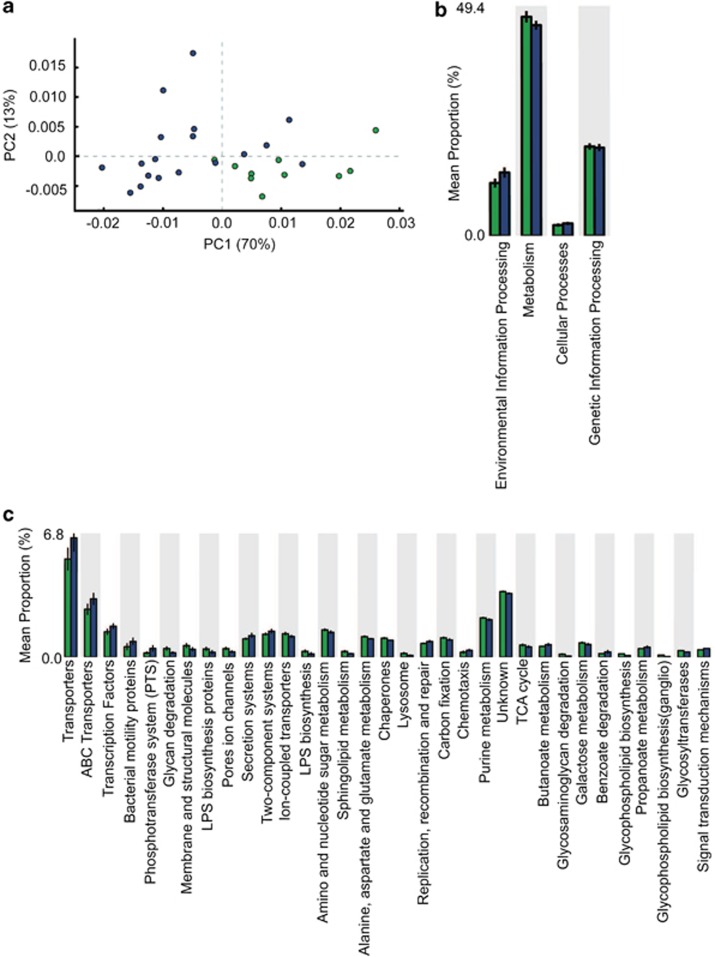
To predict the potential consequences of altering microbial gut composition in response to RSP treatment, we assessed the metabolic capacity of the microbiota based on the presence of organisms discerned by 16S rRNA sequences utilizing the algorithm, Phylogenetic Investigation of Communities by Reconstruction of Unobserved States (PICRUSt).27 Overall, out of 6909 potential metabolic features, we found 1212 KEGG (Kyoto Encyclopedia of Genes and Gene Systems) orthologs that were significantly different between the two groups (P<0.05; Figure 4). Families of KEGG orthologs were found to vary based on treatment group using principal component analysis (Figure 4a). By assigning KEGG orthologs to specific bacterial metabolic pathways, it was revealed that participants in the chronic RSP treatment group had microorganisms that were enriched for environmental information processing pathways (for example, response to antibiotics) and cellular processes (Figure 4b). In contrast, psychiatric control participants had significantly more orthologs classified in bacterial metabolism pathways (Figure 4b). A more detailed analysis revealed that those pathways prominent in the chronic RSP treatment group included membrane transport, cell motility, xenobiotics biodegradation and metabolism, transcription and signal transduction (Figure 4c). Within the xenobiotics biodegradation and metabolism category, major pathways included butanoate, propanoate, fatty acid and tryptophan metabolism (Figure 4c), all of which have been implicated in weight gain.28, 29 Conversely, pathways in greater abundance in psychiatric controls included glycan biosynthesis and metabolism, as well as metabolism of cofactors and vitamins. Here, glycan biosynthesis comprises mostly lipopolysaccharide biosynthesis, which is found primarily in Gram-negative organisms such as Bacteroidetes spp. The latter were vastly decreased in the RSP participants. Thus, PICRUSt reveals a potential mechanistic link between the observed shifts in bacterial taxa and their corresponding metabolic potential in response to treatment with RSP.
Figure 4.
Phylogenetic Investigation of Communities by Reconstruction of Unobserved States (PICRUSt) predicted KEGG (Kyoto Encyclopedia of Genes and Gene Systems) orthologs. 1212 Significant KEGG orthologs were predicted with PICRUSt software. (a) PCoA of KEGG orthologs in chronic risperidone (RSP)-treated participants vs psychiatric controls. (b, c) Global and individual KEGG pathways of chronic RSP-treated participants (blue) vs psychiatric controls (green). PCoA, principal component analysis.
Discussion
To our knowledge, this is the first description of a shift in the human gut microbiota following the use of SGAs. We found a significant shift in Bacteroidetes and Firmicutes in participants exposed chronically to RSP (76 and 11%, respectively) when compared with psychiatric controls (51 and 41%, respectively). The magnitude of these differences was most prominent in those participants who had gained significant weight. These differences in the gut microbiome composition following chronic treatment are consistent with findings from the prospective study where similar changes appeared to correlate with the magnitude of change in BMI Z-score.
The long-term health sequelae of obesity are significant and include cardiovascular disease and type 2 diabetes,30 particularly when obesity develops in childhood. Pediatric patients suffering from psychiatric disorders are at an elevated risk for obesity due to disease-related factors; this was a primary reason for selecting as controls participants with psychiatric illness but who are SGA naive. Although pharmacotherapy is often unavoidable to address serious symptoms and improve functioning, it is concerning that SGAs induce weight gain, compounding the health risks associated with obesity. SGA-induced weight gain is multifactorial and it is likely that the gut microbiome is a key contributor. In fact, a 3-week treatment with olanzapine induced weight gain in rats and led to a 4.3% decrease in Bacteroidetes.1 Despite the differences between the rat and human microbiomes, it is notable that, similar to olanzapine-treated rats, our participants treated chronically with RSP exhibited significantly lower relative prevalence of Bacteroidetes. Notably, a decrease in Bacteroidetes of a comparable magnitude has been reported in obese individuals, although the evidence is not without inconsistency.13, 31 Importantly, our results from the longitudinal study provide preliminary confirmation that a change in gut bacterial composition (including a decrease in Bacteroidetes) follows the initiation of RSP treatment and correlates with weight gain during the initial treatment phase.
Using Unifrac, which is an index of overall bacterial profile, we found no significant association between those who exhibited significant BMI gain and those who did not, following RSP treatment (Figure 1). However, when individual phyla, genera and species were compared, chronic RSP participants with a significant BMI gain were found to harbor a unique set of organisms, which distinguishes them from the chronic RSP participants without BMI gain (Figure 2). Therefore, it would be critical to examine how baseline microbial profile may shape one's propensity to gain weight following the initiation of SGAs.
To identify specific taxa that correlate with significant weight gain following RSP treatment, we described discriminatory OTUs associated with each treatment group (Figure 3). For psychiatric controls, the discriminatory OTUs were attributable to the class Bacteroidales. In contrast, for participants in the chronic RSP group with significant BMI gain, Ralstonia sp., Clostridium sp. and members of the Erysipelotrichaceae family were the most abundant OTUs. In contrast, C. aerofaciens was the most abundant discriminatory OTU in the chronic RSP group without significant BMI gain. Taken together, these results suggest that alterations in the membership of Firmicutes and Actinobacteria phyla may drive overall major changes in the gut microbiota in relation to long-term RSP treatment. In contrast, alterations in the membership of Firmicutes and Proteobacteria may be linked to weight gain following long-term RSP treatment. This raises the possibility for Actinobacteria to be used for the development of novel therapeutics (prebiotics or probiotics) to prevent or reverse weight gain following RSP treatment. Indeed, it has been shown that Bacteriodales species not only ameliorate gut abnormalities (often found in individuals with psychiatric disorders) but may also restore social and behavioral impairments.32, 33, 34
Gut bacterial metabolic pathways have also been linked to obesity.13, 35 In fact, bacterial fermentation of complex carbohydrates and proteins produce short-chain fatty acids in the proximal colon, optimizing caloric harvest from the diet.13 Short-chain fatty acids directly provide energy to colonocytes, and absorption into the portal circulation stimulates adipogenesis.19, 28 In addition, short-chain fatty acids are believed to control anorectic hormone (peptide YY and glucagon-like peptide-1) release or production through the free fatty acid receptor 2 and free fatty acid receptor 3.36 In our study, higher levels of KEGG-associated pathways for butyrate and propionate metabolism were found within the RSP treatment group compared with psychiatric controls (Figure 4), suggesting that the gut microbiome of these individuals may have higher levels of short-chain fatty acids production, potentially leading to weight gain.
Our analysis using PICRUSt also found that the microbiota of participants treated chronically with RSP were enriched for KEGG orthologs affecting tryptophan metabolism (Figure 4). Tryptophan and its metabolite, 5-hydroxytryptamine or serotonin, are both critical to a number of physiological processes, including affective regulation and energy homeostasis. Growing evidence has also implicated neurotransmitters in bacterial signaling, further supporting the vast connectivity of the microbiome–gut–brain axis.20, 37 Specifically, gut bacteria are essential to the normal development of peripheral and central serotonin signaling pathways in the host.38 Moreover, probiotic administration has been shown to influence the availability of tryptophan, suggesting that microbiota manipulation could be a valid therapeutic strategy to modulate central nervous system signaling.39 In light of our findings, we speculate that alterations in tryptophan metabolism in the gut may mediate, at least in part, SGA-induced weight gain. Altered tryptophan metabolism may also affect the therapeutic benefit of RSP treatment.
This study has several limitations that must be acknowledged. First, the sample size of both studies was relatively small. This may have led to false-positive results. However, in light of the preclinical findings reviewed earlier and the apparent consistency in the pattern of change in bacterial composition across both human studies (that is, decreased Bacteroidetes:Firmicutes ratio), it is likely that our findings reflect a true effect of RSP treatment. It may be possible that changes in bacterial composition reflected an earlier change in dietary intake induced by RSP. However, the dietary data collected identified no differences between the groups. Polypharmacy, which is often observed in SGA-treated children, may additionally have a role in altering the microbiome. However, neither psychostimulant nor selective serotonin reuptake inhibitor use was associated with changes in gut bacterial composition in our participants (Supplementary Figure 2). Nonetheless, our findings require replication in a larger cohort, including females and in the absence of polypharmacy. Moreover, as diet has a pivotal role in shaping the gut microbiome, an evaluation of the impact of the western diet on RSP-induced weight gain will be critical for future studies to undertake.
Taken together, our findings offer preliminary evidence that the human gut microbiome is altered in patients treated chronically with RSP and may be associated with weight gain. Significant alterations in the microbiome were seen at the phyla level following both acute and chronic exposure to RSP. In addition, discriminatory genera (OTUs) were identified, which may help inform efforts to develop probiotics as a therapeutic. Prospective studies in both humans and mice are underway to further investigate the basis of these findings.
Acknowledgments
This work was funded in part by a pilot research award from the Fraternal Order of Eagles Diabetes Research Center at the University of Iowa, Carver College of Medicine and by the National Institute of Mental Health (K23MH085005) and the National Center for Research Resources (2UL1TR000442-06).
Disclaimer
The content is solely the responsibility of the authors and does not necessarily represent the official views of the funding agencies.
The authors declare no conflict of interest.
Footnotes
Supplementary Information accompanies the paper on the Translational Psychiatry website (http://www.nature.com/tp)
Supplementary Material
References
- Davey KJ, Cotter PD, O'Sullivan O, Crispie F, Dinan TG, Cryan JF et al. Antipsychotics and the gut microbiome-olanzapint-induced metabolic dysfunction is attenuated by antibiotic administration in the rat. Transl Psychiatry 2013; 3: e309–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Olfson M, Blanco C, Liu S-M, Wang S, Correll CU. National trends in the office-based treatment of children, adolescents, and adults with antipsychotics. Arch Gen Psychiatry 2012; 69: 1247. [DOI] [PubMed] [Google Scholar]
- Findling RL, Steiner H, Weller EB. Use of antipsychotics in children and adolescents. J Clin Psychiatry 2005; 66(Suppl 7): 29–40. [PubMed] [Google Scholar]
- De Hert M, Dobbelaere M, Sheridan EM, Cohen D, Correll CU. Metabolic and endocrine adverse effects of second-generation antipsychotics in children and adolescents: a systematic review of randomized, placebo controlled trials and guidelines for clinical practice. Eur Psychiatry 2011; 26: 144–158. [DOI] [PubMed] [Google Scholar]
- Correll CU. Cardiometabolic risk of second-generation antipsychotic medications during first-time use in children and adolescents. JAMA 2009; 302: 1765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Newcomer JW. Metabolic risk during antipsychotic treatment. Clin Ther 2004; 26: 1936–1946. [DOI] [PubMed] [Google Scholar]
- Sousa T, Paterson R, Moore V, Carlsson A, Abrahamsson B, Basit AW. The gastrointestinal microbiota as a site for the biotransformation of drugs. Int J Pharm 2008; 363: 1–25. [DOI] [PubMed] [Google Scholar]
- Olfson M, Crystal S, Huang C, Gerhard T. Trends in antipsychotic drug use by very young, privately insured children. J Am Acad Child Adolesc Psychiatry 2010; 49: 13–23. [DOI] [PubMed] [Google Scholar]
- Allison DB, Mentore JL, Heo M, Chandler LP, Cappelleri JC, Infante MC et al. Antipsychotic-induced weight gain: a comprehensive research synthesis. Am J Psychiatry 1999; 156: 1686–1696. [DOI] [PubMed] [Google Scholar]
- Lilly E.. Important Safety Information on ZYPREXA® (olanzapine) and Use in Adolescents. Eli Lilly and Co.: Indianapolis, IN, USA, 2010, p 4.
- Malhotra AK, Lencz T, Correll CU, Kane JM. Genomics and the future of pharmacotherapy in psychiatry. Int Rev Psychiatry 2007; 19: 523–530. [DOI] [PubMed] [Google Scholar]
- Gohlke JM, Dhurandhar EJ, Correll CU, Morrato EH, Newcomer JW, Remington G et al. Recent advances in understanding and mitigating adipogenic and metabolic effects of antipsychotic drugs. Front Psychiatry 2012; 3: 62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Turnbaugh PJ, Ley RE, Mahowald MA, Magrini V, Mardis ER, Gordon JI. An obesity-associated gut microbiome with increased capacity for energy harvest. Nature 2006; 444: 1027–1131. [DOI] [PubMed] [Google Scholar]
- Turnbaugh PJ, Hamady M, Yatsunenko T, Cantarel BL, Duncan A, Ley RE et al. A core gut microbiome in obese and lean twins. Nature 2008; 457: 480–484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ridaura VK, Faith JJ, Rey FE, Cheng J, Duncan AE, Kau AL et al. Gut microbiota from twins discordant for obesity modulate metabolism in mice. Science 2013; 341: 1241214–1241214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Severance EG, Yolken RH, Eaton WW. Autoimmune diseases, gastrointestinal disorders and the microbiome in schizophrenia: more than a gut feeling. Schizophr Res 2014; 6: 1–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dinan TG, Borre YE, Cryan JF. Genomics of schizophrenia: time to consider the gut microbiome? Mol Psychiatry 2014; 19: 1252–1257. [DOI] [PubMed] [Google Scholar]
- Morgan AP, Crowley JJ, Nonneman RJ, Quackenbush CR, Miller CN, Ryan AK et al. The antipsychotic olanzapine interacts with the gut microbiome to cause weight gain in mouse. PLoS One 2014; 9: e115225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wong JMW, de Souza R, Kendall CWC, Emam A, Jenkins DJA. Colonic health: fermentation and short chain fatty acids. J Clin Gastroenterol 2006; 40: 235–243. [DOI] [PubMed] [Google Scholar]
- Collins SM, Surette M, Bercik P. The interplay between the intestinal microbiota and the brain. Nat Rev Microbiol 2012; 10: 735–742. [DOI] [PubMed] [Google Scholar]
- Haiser HJ, Turnbaugh PJ. Developing a metagenomic view of xenobiotic metabolism. Pharmacol Res 2013; 69: 21–31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Patterson AD, Turnbaugh PJ. Microbial determinants of biochemical individuality and their impact on toxicology and pharmacology. Cell Metab 2014; 20: 761–768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Calarge CA, Xie D, Fiedorowicz JG, Burns TL, Haynes WG. Rate of weight gain and cardiometabolic abnormalities in children and adolescents. J Pediatr 2012; 161: 1010–1015.e1011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ogden CL, Kuczmarski RJ, Flegal KM, Mei Z, Guo S, Wei R et al. Centers for Disease Control and Prevention 2000 Growth Charts for the United States: improvements to the 1977 National Center for Health Statistics Version. Pediatrics 2002; 109: 45–60. [DOI] [PubMed] [Google Scholar]
- Ley RE, Bäckhed F, Turnbaugh P, Lozupone CA, Knight RD, Gordon JI. Obesity alters gut microbial ecology. Proc Natl Acad Sci USA 2005; 102: 11070–11075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huttenhower C, Gevers D, Knight R, Abubucker S, Badger JH, Chinwalla AT et al. Structure, function and diversity of the healthy human microbiome. Nature 2012; 486: 207–214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Langille MGI, Zaneveld J, Caporaso JG, McDonald D, Knights D, Reyes JA et al. Predictive functional profiling of microbial communities using 16S rRNA marker gene sequences. Nat Biotechnol 2013; 31: 814–821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hong Y-H, Nishimura Y, Hishikawa D, Tsuzuki H, Miyahara H, Gotoh C et al. Acetate and Propionate Short Chain Fatty Acids Stimulate Adipogenesis via GPCR43. Endocrinology 2005; 146: 5092–5099. [DOI] [PubMed] [Google Scholar]
- Clarke G, Stilling RM, Kennedy PJ, Stanton C, Cryan JF, Dinan TG., Minireview. Gut microbiota: the neglected endocrine organ. Mol Endocrinol 2014; 28: 1221–1238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Expert Panel on the Identification, Evaluation, and Treatment of Overweight in Adults.. Clinical guidelines on the identification, evaluation, and treatment of overweight and obesity in adults: executive summary. expert panel on the identification, evaluation, and treatment of overweight in adults. Am J Clin Nutr 1998; 68: 899–917. [DOI] [PubMed] [Google Scholar]
- Ley RE, Turnbaugh PJ, Klein S, Gordon JI. Microbial ecology: human gut microbes associated with obesity. Nature 2006; 444: 1022–1023. [DOI] [PubMed] [Google Scholar]
- Hsiao EY, McBride SW, Hsien S, Sharon G, Hyde ER, McCue T et al. Microbiota modulate behavioral and physiological abnormalities associated with neurodevelopmental disorders. Cell 2013; 155: 1451–1463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buie T, Campbell DB, Fuchs GJ, Furuta GT, Levy J, VandeWater J et al. Evaluation, diagnosis, and treatment of gastrointestinal disorders in individuals with ASDs: a consensus report. Pediatrics 2010; 125: S1–S18. [DOI] [PubMed] [Google Scholar]
- Mazmanian SK, Round JL, Kasper DL. A microbial symbiosis factor prevents intestinal inflammatory disease. Nature 2008; 453: 620–625. [DOI] [PubMed] [Google Scholar]
- Bäckhed F, Ding H, Wang T, Hooper LV, Koh GY, Nagy A et al. The gut microbiota as an environmental factor that regulates fat storage. Proc Natl Acad Sci USA 2004; 101: 15718–15723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sleeth ML, Thompson EL, Ford HE, Zac-Varghese SEK, Frost G. Free fatty acid receptor 2 and nutrient sensing: a proposed role for fibre, fermentable carbohydrates and short-chain fatty acids in appetite regulation. Nutr Res Rev 2010; 23: 135–145. [DOI] [PubMed] [Google Scholar]
- Schwarcz R, Bruno JP, Muchowski PJ, Wu H-Q. Kynurenines in the mammalian brain: when physiology meets pathology. Nat Rev Neurosci 2012; 13: 465–477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clarke G, Grenham S, Scully P, Fitzgerald P, Moloney RD, Shanahan F et al. The microbiome-gut-brain axis during early life regulates the hippocampal serotonergic system in a sex-dependent manner. Mol Psychiatry 2012; 18: 666–673. [DOI] [PubMed] [Google Scholar]
- Desbonnet L, Garrett L, Clarke G, Bienenstock J, Dinan TG. The probiotic Bifidobacteria infantis: an assessment of potential antidepressant properties in the rat. J Psychiatr Res 2008; 43: 164–174. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.