Abstract
BACKGROUND
The authors investigated the prevalence of pretreatment urinary, sexual, hormonal, and bowel dysfunction in a contemporary, population-based prostate cancer cohort. They also explored the associations between baseline function and age, comorbidity, and timing of baseline survey completion with respect to treatment.
METHODS
The Comparative Effectiveness Analysis of Surgery and Radiation (CEASAR) study is a population-based, prospective cohort study that enrolled 3691 men with incident prostate cancer during 2011 and 2012. Pretreatment function was ascertained using the Expanded Prostate Cancer Index-26 (EPIC-26). Data were stratified by age, comorbidity, and timing of baseline survey completion with respect to treatment. Unadjusted and multivariable linear regression analyses were performed to evaluate the relations between exposures and pretreatment function.
RESULTS
After applying exclusion criteria, the study cohort comprised 3072 men. A strikingly high proportion of men reported inability to obtain erections satisfactory for intercourse (45%) and some degree of urinary incontinence (17%) at baseline. Sexual function was particularly age-sensitive, with patients aged ≤60 years reporting summary scores in excess of 30 points higher than patients aged ≥75 years (P <.001). Compared with the healthiest men, highly comorbid patients reported less favorable function in each domain, including urinary incontinence (summary score, 89.5 vs 74.1; P <.001) and sexual function (summary score, 70.8 vs 32.9; P <.001). Although statistically significant differences in summary scores were identified between patients who completed the baseline questionnaire before treatment (52%) versus after treatment (48%), the absolute differences were small (range, 1–3 points).
CONCLUSIONS
Patients with newly diagnosed prostate cancer exhibit a wide distribution of pretreatment function. The current data may be used to redefine the population “at risk” for treatment-related harms.
Keywords: prostate cancer, quality of life, urinary function, sexual function, bowel function
INTRODUCTION
Prostate cancer management is largely preference-sensitive and is strongly influenced by the perceived risk of treatment-related sexual, urinary, and bowel dysfunction.1–4 Numerous studies have revealed the considerable impact of prostate cancer treatments on post-treatment sexual, urinary, and bowel function.5–8 Regardless of treatment, baseline function is a well known predictor of post-treatment health-related quality of life.1–4,9 Over the past 20 years, there has been a consistent rise in both the incidence and the prevalence of obesity5–8,10 and diabetes mellitus,11 both known risk factors for erectile dysfunction.12,13 Furthermore, the past 2 decades have witnessed considerable increases in direct-to-consumer marketing for functional conditions, such as urinary irritation, urinary incontinence, irritable bowel syndrome, and erectile dysfunction. Whether stigma reduction through direct-to-consumer marketing for functional urologic conditions has changed the distribution of reporting remains unknown.
Given the critical importance of functional outcomes in the population-level evaluation of the comparative effectiveness and harms of various prostate cancer management strategies, a complete understanding of the baseline distribution of disease-specific function is essential. Furthermore, as we embark on an era of value-based health care, accurate assessment of treatment-related harms requires contemporary assessment of the population prevalence of pretreatment urinary, sexual, hormonal, and bowel dysfunction to most appropriately estimate treatment effects. To this end, the objective of the current study was to report the contemporary prevalence of urinary, sexual, hormonal, and bowel dysfunction, defined as the proportion of patients in a population suffering from disease-specific dysfunction. In addition, we sought to evaluate the relations between age, comorbidity, and pretreatment function reporting among participants in the Comparative Effectiveness Analysis of Surgery and Radiation (CEASAR) study. Finally, we aimed to ascertain the effect of timing of baseline survey completion on baseline function reporting.
MATERIALS AND METHODS
CEASAR is a population-based, hybrid, prospective cohort study designed to compare the effectiveness and harms of management strategies for localized prostate cancer (clinicaltrials.gov identifier NCT01326286), enrolling men aged <80 years with clinically localized (less than clinical T3aN0M0) adenocarcinoma of the prostate and a prostate-specific antigen level <50 ng/mL. Using a rapid case ascertainment system, the study identified all eligible men who were diagnosed with prostate cancer from 5 registries in the National Cancer Institute Surveillance, Epidemiology, and End Results Program: Atlanta/Rural Georgia, Los Angeles, Louisiana, New Jersey, and Utah. To enhance the complement of men receiving novel therapies, men were recruited from Cancer of the Prostate Strategic Urologic Research Endeavor (CaPSURE), an observational prostate cancer registry.14 Details surrounding the rationale, objectives, and methods of CEASAR have been reported previously.15 Institutional review board approval was obtained at all participating sites as well as at the Vanderbilt University coordinating site.
Participants completed a baseline survey comprising multiple scales, including the Expanded Prostate Cancer Index-26 (EPIC-26), a reliable and valid instrument that was developed to measure prostate cancer-specific function.16,17 The EPIC-26 measures function and bother in 5 domains: urinary incontinence, urinary irritation/obstruction, sexual function, bowel function, and hormonal function/vitality. Each domain comprises 4 to 7 items scaled to 100, with the domain summary score representing the average of scores on all items in the domain. Given the uniform scaling, effect sizes in linear regression models represent the point difference between groups.
The baseline survey asked patients to document whether they had initiated treatment for prostate cancer at the time of the survey completion and, if so, patients were to recall their pretreatment function. Six months after completing the baseline survey, patients were asked to complete a second survey that measured the EPIC-26 and numerous other scales, including the Total Illness Burden Index-Prostate Cancer (TIBI-CaP), a self-reported comorbidity scale that has been validated in men with prostate cancer.18 TIBI-CaP scores were categorized as previously reported (score categories: 0–2, 3–5, 6–8, and 9+), with higher scores representing a greater burden of comorbid illness.18
The current study was limited to eligible participants who responded to the baseline survey within 180 days of diagnosis and responded to the 6-month survey. Responses to individual items were summarized as binary measures to facilitate clinical interpretation. Data were stratified based on timing of response to the baseline survey (before or after self-reported initiation of prostate cancer treatment) to evaluate for the presence of bias in reporting bias in reporting pre-treatment function. In addition, data were stratified by age group (ages ≤60 years, 61–65 years, 66–70 years, 71–75 years, and ≥76 years) and comorbidity. Between-group comparison of baseline function were performed using the Pearson chi-square test, the Wilcoxon rank-sum test, the 2-sample t test, and the Kruskal-Wallis test. The choice of statistical test was based on data distribution. Multivariable linear regression analysis was performed, adjusting for age, race, and time from diagnosis to baseline survey completion, to identify the effect of differential timing of baseline survey completion on domain summary scores. In addition, given the colinearity of age and comorbidity, multivariable linear regression was used to determine the effect of comorbidity on baseline function while adjusting for age.
All P values were 2-sided, and P values <.05 were considered statistically significant. The statistical software packages R (version 2.13.0; R Foundation for Statistical Computing, Vienna, Austria) and STATA (version 12.1; StatCorp, College Station, Tex) were used for all statistical analyses.
RESULTS
From January 2011 to February 2012, 8625 men were invited to participate in CEASAR, and 7343 with incident prostate cancer were deemed eligible for participation. Of these, 3691 men (50%) completed a baseline survey, comprising 3429 men enrolled from the Surveillance, Epidemiology, and End Results sites and 262 men enrolled from CaPSURE. Our final study cohort consisted of 3072 men (83%) who completed the baseline survey within 180 days of diagnosis and also completed the 6-month follow-up survey. Complete demographic data are presented in Table 1.
TABLE 1.
Baseline Characteristics of the Study Cohort Stratified by the Initiation of Treatment at Baseline Survey
| Characteristic | Overall Study Sample
|
Initiated Treatment at Time of Baseline Survey: Percentage of Patients (No.)
|
||||
|---|---|---|---|---|---|---|
| Total No. | Percentage of Patients (No.) | Total No. | No | Yes | P | |
| Age: Median [IQR], y | 3063 | 65 [59–70] | 3037 | 65 [59–70] | 65 [59–70] | .229 |
| TIBI-CaP: Median score [IQR] | 3072 | 3 [2–5] | 3046 | 3 [2–5] | 3 [2–5] | .366 |
| Time from enrollment to baseline survey: Mean ±SD, d | 2750 | 70.8 ±47.6 | 2727 | 60.6 ±46.7 | 80.1 ±46.1 | <.001 |
| Race | 3050 | 3024 | ||||
| White/Caucasian | 75 (2273) | 73 (1164) | 76 (1093) | .009 | ||
| Black/African American | 14 (431) | 16 (255) | 12 (172) | |||
| Latino/Hispanic | 7 (222) | 7 (110) | 8 (108) | |||
| Other | 4 (124) | 4 (57) | 5 (65) | |||
| Annual income | 2866 | 2902 | .429 | |||
| <$30,000 | 23 (649) | 21 (291) | 23 (350) | |||
| $30,001 to $50,000 | 20 (575) | 20 (276) | 20 (308) | |||
| $50,001 to $100,000 | 31 (879) | 31 (433) | 30 (457) | |||
| >$100,000 | 27 (763) | 28 (390) | 26 (397) | |||
| Education | 3050 | 3025 | .555 | |||
| Some high school or less | 10 (320) | 10 (155) | 11 (157) | |||
| High school graduate | 21 (646) | 22 (353) | 20 (288) | |||
| Some college | 22 (681) | 22 (353) | 22 (323) | |||
| College graduate | 23 (692) | 22 (354) | 23 (334) | |||
| Graduate or professional school | 23 (711) | 23 (370) | 23% (338) | |||
| Insurance | 3072 | 2977 | .355 | |||
| Private or HMO | 44 (1356) | 45 (710) | 44 (636) | |||
| Medicare | 40 (1221) | 38 (606) | 41 (601) | |||
| Medicaid | 6 (178) | 6 (95) | 6 (82) | |||
| VA or Military | 7 (204) | 7 (116) | 6 (87) | |||
| No insurance | 1 (36) | 1 (21) | 1 (15) | |||
| Unknown | 0 (8) | 0 (5) | 0 (3) | |||
| Employment | 3072 | 3046 | .542 | |||
| Works full time | 39 (1195) | 38 (605) | 40 (582) | |||
| Works part time | 8 (247) | 8 (126) | 8 (120) | |||
| Retired | 48 (1460) | 48 (771) | 46 (673) | |||
| Unemployed | 6 (170) | 6 (93) | 5 (76) | |||
Abbreviations: HMO, health maintenance organization; IQR, interquartile range; SD, standard deviation; TIBI-CaP, Total Illness Burden Index-Prostate Cancer; VA, Veterans Administration.
Timing of Baseline Survey Completion
Of the 3072 men included in the current analysis, 1451 (48%) had initiated prostate cancer treatment at the time of the baseline survey. We observed a racial difference in the proportion of men that had initiated treatment before completing the baseline survey (48% of Caucasian men vs 40% of African American men; P =.009). Not surprisingly, the interval between diagnosis and baseline survey completion was shorter in the subgroup that had not initiated treatment. Complete data stratified by timing of the baseline survey are presented in Table 1.
Baseline summary scores stratified by treatment initiation are presented in Table 2. Despite statistically significant differences in each of the domain summary scores between patients who had and who had not initiated treatment, the absolute differences in scores were small (range, 1–3 points) relative to the overall standard deviation of each summary score (range, 12.2–31.2 points). Multivariable linear regression analysis was performed to evaluate the independent effect of recall on domain summary scores, adjusting for age, race, and time from diagnosis. Again, the absolute between-group differences remained small (range, 1.0–3.7 points).
TABLE 2.
Mean Domain Summary Scores Stratified by the Initiation of Treatment at Baseline Survey
| Summary Score | Initiated Treatment: Mean ±SEM
|
Multivariable Modela
|
|||
|---|---|---|---|---|---|
| No | Yes | Pb | Coefficientc | 95% CI | |
| Sexual function | 62.5 ±0.82 | 59.1 ±0.89 | .004 | −3.7 | −6.1 to −1.27 |
| Urinary irritation | 84.3 ±0.42 | 81.3 ±0.48 | <.001 | −3.1 | −4.4 to −1.7 |
| Urinary incontinence | 88.7 ±0.47 | 86.6 ±0.56 | .025 | −1.7 | −3.3 to −0.2 |
| Hormonal function | 89.2 ±0.37 | 88.0 ±0.41 | .036 | −1.0 | −2.1 to 0.2 |
| Bowel function | 94.0 ±0.31 | 92.9 ±0.35 | .012 | −1.0 | −1.9 to −0.1 |
Abbreviations: CI, confidence interval; SEM, standard error of the mean.
Model adjusted for age, race, and time from diagnosis to enrollment.
P values were determined using a 2-sample t test.
No treatment at the time of the baseline survey served as the reference category.
The proportion of men reporting pretreatment inability to achieve erection sufficient for intercourse in treated and untreated patients was 47% and 43%, respectively (P =.02). Similar findings were observed in the urinary incontinence domain, with 9% and 5% of treated and untreated patients reporting pretreatment severe urinary incontinence, respectively (P =.001). Patients who had initiated treatment were more likely to report moderate or severe pretreatment bother secondary to both weak urinary stream (18% vs 14%; P =.001) and urinary frequency (24% vs 19%; P =.001). Patients who had initiated prostate cancer treatment were more likely to report hot flashes at baseline (5% vs 3%; P =.001); however, no other statistically significant differences were identified in the hormonal or bowel function domains.
Age
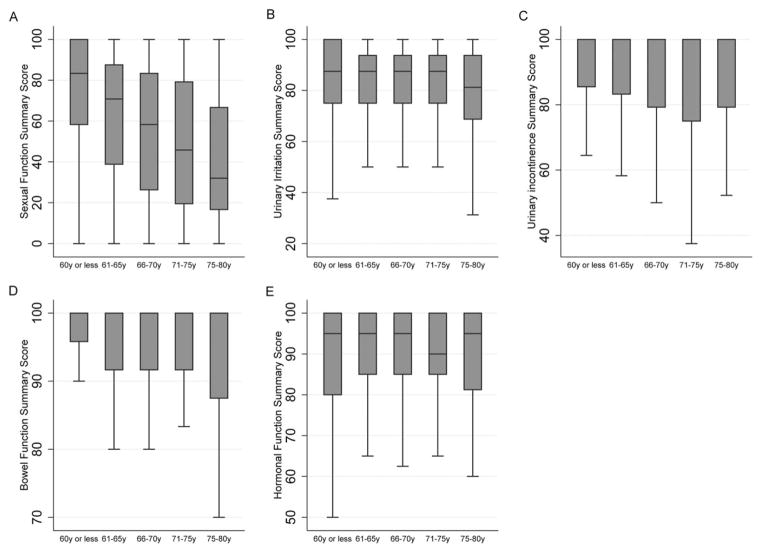
Pretreatment disease-specific function was inversely related to age at diagnosis, as demonstrated in Figure 1. Pretreatment sexual function was particularly sensitive to age, with men aged ≤60 years reporting mean sexual function summary scores in excess of 30 points higher than participants aged ≥75 years. Despite statistically significant correlations between age and urinary incontinence, urinary irritation, and bowel function summary scores, the absolute between-strata differences in summary scores were small (range, 2.7–4.4 points). There were no between-strata differences in the hormonal function domain. We performed sensitivity analyses by fitting domain-specific linear regression models in which age was treated as a continuous variable, and the results were consistent with our principal findings (Supporting Fig. 1 [see online supporting information]).
Figure 1.
Domain summary scores stratified by age are illustrated for (A) sexual function, (B) urinary irritation, (C) urinary incontinence, (D) hormonal function, and (E) bowel function. Boxes represent the median scores/interquartile range (whiskers, ± 1.5*(IQR)).
Responses to individual items mirrored the correlations between age and domain summary scores. There was a significant increase in the proportion of patients reporting erection insufficient for intercourse with age. Specifically, 29% of men aged ≤60 years reported inability to achieve an erection sufficient for intercourse compared with 71% of men aged >75 years (P <0.001). It is noteworthy that men aged ≤60 years were more likely to report the use of phosphodiesterase-5 (PDE-5) inhibitors than men aged ≥75 years (25% vs 20%; P =.016). With respect to hormonal function age appeared to be protective against depressive symptoms, with men aged ≥75 years less commonly reporting moderate-to-severe bother secondary to feeling depressed than men aged ≤60 years (4% vs 10%; P <.001). We identified no other statistically significant, age-dependent differences in the hormonal domain. It is also worth noting that younger men were more likely to report rectal pain, with 5% of men aged ≤60 years reporting moderate-to-severe bother secondary to rectal pain compared with 2% of men aged ≥75 years (P =.002). Responses to selected individual items stratified by age are presented in Supporting Table 1 (see online supporting information).
Comorbidity
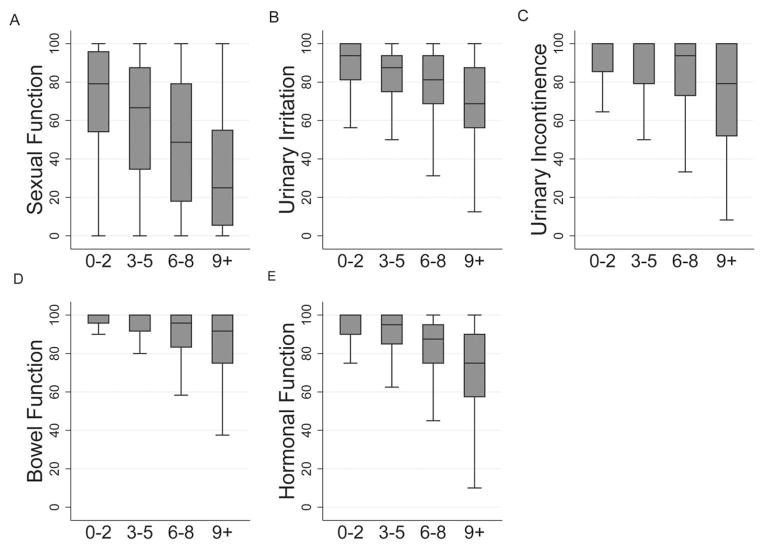
In addition to evaluating the effect of age, we sought to characterize the relationship between comorbidity and pretreatment disease-specific function. The relationship between comorbidity and domain summary scores are displayed in Figure 2. We identified considerable variation in the magnitude of between strata-differences. Whereas a 37.9-point difference in sexual function summary score was identified between patients in the lowest and highest comorbidity strata, only an 11.3-point difference was identified in the bowel function domain. We performed sensitivity analyses by fitting domain-specific linear regression models in which TIBI-CaP was treated as a continuous variable, and the results were consistent with our principal findings (Supporting Fig. 2 [see online supporting information]).
Figure 2.
Domain summary scores stratified by the Total Illness Burden Index-Prostate Cancer (TIBI-CaP) scale are illustrated, including summary scores for (A) sexual function, (B) urinary irritation, (C) urinary incontinence, (D) bowel function, and (E) hormonal function. Boxes represent the median scores/interquartile range (whiskers, ± 1.5*(IQR)).
Investigation of individual items revealed significant between-strata differences. Whereas 31% of patients with TIBI-CaP scores of from 0 to 2 (minimal to no comorbidity) reported erections insufficient for intercourse, 76% of patients with TIBI-CaP scores of 9+ (significant comorbidity) reported unsatisfactory pretreatment erectile function (P <.001). The risk of sexual bother increased with increasing comorbidity, with 21% of patients who had TIBI-CaP scores from 0 to 2 and 57% of those who had TIBI-CaP scores of 9+ reporting moderate-to-severe bother secondary to sexual dysfunction (P <.001). The risk of suffering from urinary incontinence was directly related to comorbidity, with 6% of patients who had TIBI-CaP scores from 0 to 2 and 21% of those who had TIBI-CaP scores of 9+ reporting frequent dribbling or no urinary control (P <.001). Irritative urinary symptoms also were associated with comorbidity, and highly comorbid patients more frequently reported bother secondary to weak stream (P <.001) and urinary frequency (P <.001). Increasing comorbidity was associated with increasing risk of depressive symptoms, with 4% of patients who had TIBI-CaP scores from 0 to 2 reporting moderate or severe bother because of feeling depressed compared with 19% of those who had TIBI-CaP scores of 9+ (P <.001). Increasing comorbidity was associated with increasing risk of bowel dysfunction. Specifically, 10% of patients in the highest comorbidity stratum suffered from increased frequency of bowel movements compared with 2% of patients who had minimal or no comorbidity (P <.001). In addition, 6% of patients who had TIBI-CaP scores of 9+ reported a moderate-to-severe problem because of fecal incontinence compared with 1% of those who had TIBI-CaP scores from 0 to 2 (P <.001). Comorbidity also was associated with increasing risk of rectal pain (TIBI-CaP scores of 9+ vs 0–2, 10% vs 3%, respectively; P <.001) and bowel urgency (TIBI-CaP scores of 9+ vs 0–2, 11% vs 3%, respectively; P <.001). Responses to selected individual items stratified by TIBI-CaP score are presented in Supporting Table 2 (see online supporting information).
Not surprisingly, there was a statistically significant positive correlation between the TIBI-CaP score and age (Spearman correlation =0.30; P <.001). Given the colinearity of age and comorbidity, multivariable linear regression analysis was performed to evaluate the main effects of age and TIBI-CaP score on each domain summary score (Table 3). Controlling for age, the TIBI-CaP score was negatively correlated with each domain summary score. Considering the magnitude of the adjusted model coefficients, it is likely that the relations between comorbidity and disease-specific function are clinically significant.
TABLE 3.
Multivariable Linear Regression Model of Baseline Summary Scores for Sexual Function, Urinary Irritation, Urinary Incontinence, Hormonal Function, and Bowel Function
| Summary Score | Coefficient | P | 95% CI |
|---|---|---|---|
| Sexual function | |||
| Age (continuous) | −1.24 | <.001 | −1.39 to −1.09 |
| TIBI-CaP 3–5 (vs 0–2) | −5.95 | <.001 | −8.53 to −3.38 |
| TIBI-CaP 6–8 (vs 0–2) | −14.74 | <.001 | −18.34 to −11.15 |
| TIBI-CaP 9+ (vs 0–2) | −30.04 | <.001 | −36.07 to −24.00 |
| Urinary irritation | |||
| Age (continuous) | −0.10 | .037 | −0.17 to −0.01 |
| TIBI−CaP 3–5 (vs 0–2) | −3.63 | <.001 | −5.08 to −2.20 |
| TIBI-CaP 6–8 (vs 0–2) | −8.88 | <.001 | −10.90 to −6.86 |
| TIBI-CaP 9+ (vs 0–2) | −16.86 | <.001 | −20.30 to −13.42 |
| Urinary incontinence | |||
| Age (continuous) | −0.09 | .056 | −0.19 to 0.002 |
| TIBI-CaP 3–5 (vs 0–2) | −0.56 | .516 | −2.23 to 1.12 |
| TIBI-CaP 6–8 (vs 0–2) | −4.95 | <.001 | −7.29 to −2.60 |
| TIBI-CaP 9+ (vs 0–2) | −14.68 | <.001 | −18.63 to −10.73 |
| Hormonal function | |||
| Age (continuous) | 0.21 | <.001 | 0.14 to 0.28 |
| TIBI-CaP 3–5 (vs 0–2) | −4.19 | <.001 | −5.40 to −2.99 |
| TIBI-CaP 6–8 (vs 0–2) | −11.22 | <.001 | −12.91 to −9.53 |
| TIBI-CaP 9+ (vs 0–2) | −23.36 | <.001 | −26.21 to −20.50 |
| Bowel function | |||
| Age (continuous) | −0.01 | .678 | −0.07 to 0.48 |
| TIBI-CaP 3–5 (vs 0–2) | −1.57 | .003 | −2.63 to −0.52 |
| TIBI-CaP 6–8 (vs 0–2) | −6.54 | <.001 | −8.01 to −5.07 |
| TIBI-CaP 9+ (vs 0–2) | −11.38 | <.001 | −13.88 to −8.89 |
Abbreviations: CI, confidence interval; TIBI-CaP, Total Illness Burden Index-Prostate Cancer.
DISCUSSION
This description of the contemporary prevalence of pre-treatment urinary, sexual, bowel, and hormonal function from the CEASAR study characterizes the distribution of disease-specific health-related quality of life in a contemporary, population-based cohort. The study revealed a considerable burden of pretreatment urinary and sexual dysfunction among men with newly diagnosed prostate cancer. Accurate assessment of the prevalence of pretreatment urinary, sexual, hormonal, and bowel dysfunction is required to properly estimate the harms attributed to prostate cancer screening and/or treatment. Failure to incorporate contemporary prevalence of pretreatment dysfunction into the population-level evaluation of risk and benefit of prostate cancer screening and/or treatment may result in over-estimation of treatment-related harms and thereby inappropriately tilt the balance of benefits and harms away from prostate cancer screening and/or treatment.
Our analysis revealed several notable findings, most importantly the wide distribution of pretreatment disease-specific function. Indeed, 45% of men reported erectile function insufficient for intercourse at the time of study enrollment, 7% of patients reported “frequent dribbling or no urinary control,” and 17% reported urinary leakage “at least once per day or more.” These data underscore the significant burden of pretreatment dysfunction among men with newly diagnosed prostate cancer. In addition, the study revealed a significant, independent association of pretreatment function in multiple domains, age, and burden of comorbid illness.
The current study leveraged the timing of baseline survey administration to investigate the presence of differential function reporting among men who had and had not initiated treatment at the time of the baseline survey. It is noteworthy that men who self-identified as African American were less likely to have initiated treatment at the time of the baseline survey than men of other racial groups. After adjustment for relevant factors, we observed small but statistically significant differences in each of the functional domain summary scores between men who had and had not initiated treatment. In each domain, men who had initiated treatment reported lower pretreatment function than men who had not initiated treatment. However, the magnitude of these differences was uniformly small and was unlikely to achieve clinical significance.
The population prevalence of sexual dysfunction has been reported in numerous settings. Saigal et al, using data from the National Health and Nutrition Examination Survey (NHANES), reported that 4%, 17%, 22%, and 48% of all men ages 50 to 59 years, 60 to 69 years, 70 to 74 years, and ≥75 years, respectively, were unable to achieve erection sufficient for intercourse.13 The age-specific rates of erectile dysfunction in CEASAR were higher (26%, 46%, 60%, and 72%, respectively) than those reported in NHANES. However, both the instruments and the techniques used to elicit patient-reported sexual function were different between NHANES and CEASAR, and this may account for the observed differences. Furthermore, whereas NHANES participants were recruited from the general population, the CEASAR cohort included only men with newly diagnosed prostate cancer. Certainly, a new prostate cancer diagnosis may result in cancer anxiety that translates into acute change in sexual function; however, it is also possible that the higher observed prevalence of erectile dysfunction in CEASAR reflects changes in risk factors such as diabetes, hypertension, and obesity, or that reporting of male sexual dysfunction has increased because of shifting cultural norms.
Although ample data address the population prevalence of male sexual dysfunction, fewer data address the prevalence of male urinary incontinence. Markland et al, again using NHANES, observed that the overall risk of urinary incontinence was 14% in American men aged >20 years with time-dependent and race-dependent variation.19 Litwin et al administered the University of California-Los Angeles Prostate Cancer Index to 268 elderly men without prostate cancer and observed that 6% reported severe urinary incontinence, defined as no urinary control or frequent leakage.20 This finding is similar to the observed rate of pretreatment severe urinary incontinence in CEASAR (7%). Irritative lower urinary tract symptoms (LUTS) are more common in the general population, with 22% of male participants aged ≥40 years reporting at least 1 symptom suggestive of LUTS in the 2000 to 2001 NHANES.21 Similarly, 22% and 17% of CEASAR participants reported moderate-to-severe bother secondary to urinary frequency and weak urinary stream, respectively. Despite between-study differences, the similar findings in the pretreatment CEASAR cohort and the NHANES cohort suggest that there is less population-level variation in urinary symptoms than in sexual function.
The issue of timing of survey response with respect to treatment has important implications in population-based quality-of-life research. Given the limitations in case ascertainment when conducting prospective data collection, the ability to measure baseline function before the initiation of cancer treatment remains a challenge. There is conflicting evidence surrounding the effect of recall on estimates of baseline function. Litwin and McGuigan observed that men with prostate cancer uniformly overestimated baseline function compared with data ascertained at the true baseline.23 Fransson et al reported that men were more likely to underestimate baseline urinary and bowel function but to overestimate sexual function.23 This is in contrast to our study, in which patients who were asked to recall their pretreatment function uniformly reported worse pretreatment function in each domain than those reporting pretreatment function at their true baseline. Legler et al performed a similar study in which men were asked to recall pretreatment urinary, sexual, and bowel function 6 months after treatment. The study demonstrated greater intraobserver agreement between pretreatment assessment and recall. Nonetheless, investigation of individual items revealed that men were more likely to overestimate their baseline function when asked about erectile function and urinary frequency.24 Whether there are patient-specific or treatment-specific factors that impact either the direction or the magnitude of recall bias remains unknown. Despite the identification of statistical differences between patients who had and had not initiated prostate cancer treatment, the magnitude of these differences was uniformly small. This is an indicator that timing of the baseline survey completion, whether before or after treatment initiation, is unlikely to result in meaningful between-group differences.
Although high-quality randomized trials have demonstrated improvements in survival with definitive treatment in certain subgroups,25,26 there remains appropriate concern and skepticism surrounding the balance of benefit and harm with prostate cancer screening and treatment. The risks of treatment, although considerable, must be considered in the context of a population with significant pre-existing disease-specific dysfunction, rendering a smaller and smaller proportion of men with newly diagnosed prostate cancer “at risk” for treatment-related harms. However, significant gaps in knowledge remain surrounding the balance of benefits and harms of population-based prostate cancer screening and/or treatment. It remains unclear how these findings will translate into individual-level and population-level decision-making, particularly in the context of a limited-resource environment. Furthermore, as prostate cancer remains highly preference-sensitive, it will become increasingly important to systematically incorporate patient preferences into assessment of the adequacy and appropriateness of shared decision-making. Doing so, however, will be predicated on the development of simple point-of-care methods to ascertain patient preferences.
This study has several limitations that necessitate consideration. Comorbidity data were collected in the 6-month survey. Therefore, it is possible that a participant’s burden of comorbid illness might have changed between the baseline and 6-month assessments. In addition, given the patient-reported nature of the data in this analysis, the accuracy of treatment and comorbidity data is unknown. Nonetheless, given the contribution of the patient’s own perception of his disease and/or treatment to pretreatment function, elaborating the relation(s) between patient-reported factors and health-related quality of life is essential. Finally, it remains a challenge to define the magnitude of difference in patient-reported outcomes that constitutes clinically meaningful change. Approaches to determine the minimal important difference include the “within-patient” method,27 the “between-patient” method,28 and the half standard deviation rule proposed by Norman and colleagues.29 Regardless of the method chosen, it is essential to begin interpreting differences in patient-reported outcomes through the same lens that we use to determine clinical significance.
Conclusions
Pretreatment sexual and urinary complaints are common among men with newly diagnosed prostate cancer, whereas baseline bowel and hormonal dysfunction is uncommon. Burden of comorbid illness is strongly associated with pretreatment sexual, urinary, bowel, and hormonal dysfunction; whereas meaningful age-related differences are evident in the sexual function and urinary irritation domains. Furthermore, in our contemporary cohort, the effect of differential timing of baseline survey completion with respect to the timing of treatment is small. These data may be used to redefine the population “at risk” for treatment-related functional declines.
Supplementary Material
Acknowledgments
FUNDING SUPPORT
This study was supported by grant 1R01HS019356 from the US Agency for Healthcare Research and Quality (AHRQ) (to D.F.P.). Dr. Resnick was supported in part by a grant from the Urology Care Foundation Research Scholars Program and by the American Urological Association Southeastern Section Research Scholar Fund.
Footnotes
CONFLICT OF INTEREST DISCLOSURES
Dr. Resnick reports personal fees from Denreon and Photocure. Dr. Barocas reports personal fees from Janssen, GE Healthcare, and Dendreon. Dr. Morgans reports personal fees from Myriad. Dr. Cooperberg reports personal fees from Amgen, Dendreon, Genomic Health, Myriad, Genomedx, Abbott Laboratories, Astellas, and Janssen.
Additional Supporting Information may be found in the online version of this article.
References
- 1.Zeliadt SB, Moinpour CM, Blough DK, et al. Preliminary treatment considerations among men with newly diagnosed prostate cancer [serial online] Am J Manag Care. 2010;16:e121–e130. [PubMed] [Google Scholar]
- 2.Zeliadt SB, Ramsey SD, Penson DF, et al. Why do men choose 1 treatment over another? A review of patient decision making for localized prostate cancer. Cancer. 2006;106:1865–1874. doi: 10.1002/cncr.21822. [DOI] [PubMed] [Google Scholar]
- 3.Holmboe ES, Concato J. Treatment decisions for localized prostate cancer: asking men what’s important. J Gen Intern Med. 2000;15:694–701. doi: 10.1046/j.1525-1497.2000.90842.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Feldman-Stewart D, Brundage MD, Van Manen L, Svenson O. Patient-focused decision-making in early-stage prostate cancer: insights from a cognitively based decision aid. Health Expect. 2004;7:126–141. doi: 10.1111/j.1369-7625.2004.00271.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Sanda MG, Dunn RL, Michalski J, et al. Quality of life and satisfaction with outcome among prostate-cancer survivors. N Engl J Med. 2008;358:1250–1261. doi: 10.1056/NEJMoa074311. [DOI] [PubMed] [Google Scholar]
- 6.Gore JL, Kwan L, Lee SP, Reiter RE, Litwin MS. Survivorship beyond convalescence: 48-month quality-of-life outcomes after treatment for localized prostate cancer. J Natl Cancer Inst. 2009;101:888–892. doi: 10.1093/jnci/djp114. [DOI] [PubMed] [Google Scholar]
- 7.Resnick MJ, Koyama T, Fan KH, et al. Long-term functional outcomes after treatment for localized prostate cancer. N Engl J Med. 2013;368:436–445. doi: 10.1056/NEJMoa1209978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Johansson E, Steineck G, Holmberg L, et al. for the SPCG-4 Investigators. Long-term quality-of-life outcomes after radical prostatectomy or watchful waiting: the Scandinavian Prostate Cancer Group-4 randomised trial. Lancet Oncol. 2011;12:891–899. doi: 10.1016/S1470-2045(11)70162-0. [DOI] [PubMed] [Google Scholar]
- 9.Alemozaffar M, Regan MM, Cooperberg MR, et al. Prediction of erectile function following treatment for prostate cancer. JAMA. 2011;306:1205–1214. doi: 10.1001/jama.2011.1333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Freedman DS Centers for Disease Control and Prevention (CDC) Obesity—United States, 1988–2008. MMWR Surveill Summ. 2011;60(suppl):73–77. [PubMed] [Google Scholar]
- 11.Geiss LS, Li Y, Kirtland K, et al. Centers for Disease Control and Prevention (CDC) Increasing prevalence of diagnosed diabetes–United States and Puerto Rico, 1995–2010. MMWR Morbidity and Mortality Weekly Reports. 2012;61:918–921. [PubMed] [Google Scholar]
- 12.Seftel AD, Sun P, Swindle R. The prevalence of hypertension, hyperlipidemia, diabetes mellitus and depression in men with erectile dysfunction. J Urol. 2004;171:2341–2345. doi: 10.1097/01.ju.0000125198.32936.38. [DOI] [PubMed] [Google Scholar]
- 13.Saigal CS, Wessells H, Pace J, Schonlau M, Wilt TJ Urologic Diseases in America Project. Predictors and prevalence of erectile dysfunction in a racially diverse population. Arch Intern Med. 2006;166:207–212. doi: 10.1001/archinte.166.2.207. [DOI] [PubMed] [Google Scholar]
- 14.Lubeck DP, Litwin MS, Henning JM, et al. The CaPSURE database: a methodology for clinical practice and research in prostate cancer. CaPSURE Research Panel Cancer of the Prostate Strategic Urologic Research Endeavor. Urology. 1996;48:773–777. doi: 10.1016/s0090-4295(96)00226-9. [DOI] [PubMed] [Google Scholar]
- 15.Barocas DA, Chen V, Cooperberg M, et al. Using a population-based observational cohort study to address difficult comparative effectiveness research questions: the CEASAR study. J Comp Eff Res. 2013;2:445–460. doi: 10.2217/cer.13.34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Wei JT, Dunn RL, Litwin MS, Sandler HM, Sanda MG. Development and validation of the Expanded Prostate Cancer Index Composite (EPIC) for comprehensive assessment of health-related quality of life in men with prostate cancer. Urology. 2000;56:899–905. doi: 10.1016/s0090-4295(00)00858-x. [DOI] [PubMed] [Google Scholar]
- 17.Szymanski KM, Wei JT, Dunn RL, Sanda MG. Development and validation of an abbreviated version of the Expanded Prostate Cancer Index Composite instrument for measuring health-related quality of life among prostate cancer survivors. Urology. 2010;76:1245–1250. doi: 10.1016/j.urology.2010.01.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Litwin MS, Greenfield S, Elkin EP, Lubeck DP, Broering JM, Kaplan SH. Assessment of prognosis with the Total Illness Burden Index for Prostate Cancer: aiding clinicians in treatment choice. Cancer. 2007;109:1777–1783. doi: 10.1002/cncr.22615. [DOI] [PubMed] [Google Scholar]
- 19.Markland AD, Richter HE, Fwu C-W, Eggers P, Kusek JW. Prevalence and trends of urinary incontinence in adults in the United States, 2001 to 2008. J Urol. 2011;186:589–593. doi: 10.1016/j.juro.2011.03.114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Litwin MS. Health related quality of life in older men without prostate cancer. J Urol. 1999;161:1180–1184. [PubMed] [Google Scholar]
- 21.Erickson BA, Vaughan-Sarrazin M, Liu X, Breyer BN, Kreder KJ, Cram P. Lower urinary tract symptoms and diet quality: findings from the 2000–2001 National Health and Nutrition Examination Survey. Urology. 2012;79:1262–1267. doi: 10.1016/j.urology.2012.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Litwin MS, McGuigan KA. Accuracy of recall in health-related quality-of-life assessment among men treated for prostate cancer. J Clin Oncol. 1999;17:2882–2888. doi: 10.1200/JCO.1999.17.9.2882. [DOI] [PubMed] [Google Scholar]
- 23.Fransson P, Damber J-E, Widmark A. Health-related quality of life 10 years after external beam radiotherapy or watchful waiting in patients with localized prostate cancer. Scand J Urol Nephrol. 2009;43:119–126. doi: 10.1080/00365590802519396. [DOI] [PubMed] [Google Scholar]
- 24.Legler J, Potosky AL, Gilliland FD, Eley JW, Stanford JL. Validation study of retrospective recall of disease-targeted function: results from the Prostate Cancer Outcomes Study. Med Care. 2000;38:847–857. doi: 10.1097/00005650-200008000-00008. [DOI] [PubMed] [Google Scholar]
- 25.Wilt TJ, Brawer MK, Jones KM, et al. Prostate Cancer Intervention Versus Observation Trial (PIVOT) Study Group. Radical prostatectomy versus observation for localized prostate cancer. N Engl J Med. 2012;367:203–213. doi: 10.1056/NEJMoa1113162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Bill-Axelson A, Holmberg L, Ruutu M, et al. SPCG-4 Investigators. Radical prostatectomy versus watchful waiting in early prostate cancer. N Engl J Med. 2011;364:1708–1717. doi: 10.1056/NEJMoa1011967. [DOI] [PubMed] [Google Scholar]
- 27.Juniper EF, Guyatt GH, Willan A, Griffith LE. Determining a minimal important change in a disease-specific quality of life questionnaire. J Clin Epidemiol. 1994;47:81–87. doi: 10.1016/0895-4356(94)90036-1. [DOI] [PubMed] [Google Scholar]
- 28.Wells GA, Tugwell P, Kraag GR, Baker PR, Groh J, Redelmeier DA. Minimum important difference between patients with rheumatoid arthritis: the patient’s perspective. J Rheumatol. 1993;20:557–560. [PubMed] [Google Scholar]
- 29.Norman GR, Sloan JA, Wyrwich KW. Interpretation of changes in health-related quality of life: the remarkable universality of half a standard deviation. Med Care. 2003;41:582–592. doi: 10.1097/01.MLR.0000062554.74615.4C. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.