Abstract
Purpose
The current standard of care for patients with locally advanced prostate cancer is a combination of androgen deprivation and radiation therapy. Radiation is typically given with androgen suppression when testosterone levels are at their nadir. Recent reports have shown that androgen stimulation of androgen deprived prostate cancer cells leads to formation of double strand breaks (DSBs). Here, we exploit this finding and investigate the extent and timing of androgen induced DSBs and their effect on tumor growth following androgen stimulation in combination with ionizing radiation (IR).
Experimental Design
Androgen induced DNA damage was assessed by comet assays and γH2A.X foci formation. Effects of androgen stimulation and radiation were determined in vitro and in vivo with xenograft models.
Results
We document that androgen treatment of androgen deprived prostate cancer cell lines resulted in a dose and time dependent induction of widespread DSBs. Generation of these breaks was dependent on androgen receptor (AR) and topoisomerase II beta (TOP2B) but not on cell cycle progression. In vitro models demonstrated a synergistic interaction between ionizing radiation and androgen stimulation when IR is given at a time point corresponding with high levels of androgen-induced DSB formation. Furthermore, in vivo studies showed a significant improvement in tumor growth delay when radiation was given shortly after androgen repletion in castrated mice.
Conclusions
These results suggest a potential cooperative effect and improved tumor growth delay with androgen-induced DSBs and radiation with implications for improving the therapeutic index of prostate cancer radiation therapy.
Keywords: Androgen receptor, prostate, radiation therapy, androgen induced DSBs
INTRODUCTION
Prostate cancer remains the second leading cause of all cancer related deaths in men in the United states (1). In particular, in patients diagnosed with high-risk PCa (usually defined as pretreatment PSA > 20 ng/mL, Gleason ≥ 8, or clinical stage ≥ T2c), the 10 year disease free survival rate even under modern therapy is only around 48% (2). Currently, the standard of care for high-risk or locally advanced PCa involves high dose fractionated radiation therapy in combination with neoadjuvant, concurrent, and long term (approximately 2 years) androgen deprivation therapy (ADT) (2). Radiation therapy is typically started 2 months after the initiation of ADT, when testosterone levels are at a nadir and the androgen receptor (AR) signaling axis is inactive. The combination of radiation and ADT has led to significant improvements in 10-year progression free survival (47.7% vs. 22.7%) and prostate cancer specific survival (89.7% vs. 69.6%) compared to radiation therapy alone (2). Unfortunately, a substantial fraction of men undergoing ADT in combination with radiation therapy experience disease recurrence. In addition, patients also can experience local side effects of dose-escalated radiation treatment, demonstrating the need for more refined therapy approaches that can improve the therapeutic index of radiation based treatment.
Ionizing radiation (IR) results in a large number of cellular insults of which the induction of DNA double strand breaks (DSBs) is the most cytotoxic. Therefore, agents that can induce DNA DSBs or inhibit repair of such breaks can lead to significant and potentially synergistic sensitization to the cytotoxic effects of ionizing radiation (3). If such DNA damaging agents or DSB repair inhibitors can be directed specifically at prostate cancer cells, then it may be possible to significantly enhance the therapeutic index of radiation based therapy for prostate cancer.
Interestingly, recent reports have shown that DSBs are transiently induced during transcriptional initiation by nuclear hormone receptors and other transcription factors through a mechanism that involves topoisomerase II beta (TOP2B) (4-7). In particular, induction of AR transcriptional programs by androgen stimulation, particularly at supraphysiological doses, has been shown to result in widespread, transient DSBs in prostate cancer cells in a TOP2B dependent manner (4). We hypothesize that these androgen induced DSBs can selectively sensitize AR-positive cells to DNA damaging agents including IR. Here we show that, under castrate conditions, acute activation of AR signaling by androgen stimulation combined with appropriately timed delivery of IR at the peak of DSB formation leads to synergistic growth inhibition of AR-positive prostate cancer cells in vitro and in vivo.
Materials and Methods
Cell culture, reagents, and qRT-PCR
LNCaP, VCaP and PC3 cells were purchased from the ATCC (Manassas, VA, originally received 2009). LAPC4 cells were obtained from Dr. Charles Sawyers’ laboratory (UCLA, originally received 2002). All cell lines were grown as described previously and maintained at low passage number (4). Cell line authenticity was routinely confirmed by STR genotyping in 6-10 month intervals (last tested 12/2014). To deplete cells of androgens, cells were washed with serum-free medium three times for 1 h and incubated in 5% charcoal-stripped FBS containing medium for at least 48 hrs before DHT treatments. Dihydrotestosterone (DHT) was purchased from Sigma-Aldrich (St. Louis, MO) and enzalutamide was purchased from Selleck (Selleck Chem, Houston, TX). Western blot analyses were performed with anti-AR (06-680, Millipore), anti-TOP2B (NB100-40842, Novus) and anti-β-actin (A5441, Sigma-Aldrich) primary antibodies. qRT-PCR of androgen regulated genes (TMPRSS2, KLK3), and DNA repair genes (PRKDC, POLE2, FANCI), were carried out as described previously (4) using the following primer pairs: GAPDH (F: GGAGCGAGATCCCTCCAAAAT, R: GGCTGTTGTCATACTTCTCATGG); TMPRSS2 (F: TACTCTGGAAGTTCATGGGC R: GTCATCCACTATTCCTTGGCT); KLK3 (F: TGAACCAGAGGAGTTCTTGAC R: TGACGTGATACCTTGAAGCA); PRKDC (F: AGCTGGCTTGCGCCTATTT R: GGGCACACCACTTTAACAAGA); POLE2 (F: CTGCTCCGTGGTGAAGCTAT, R: ATTCCTGGACTGCTGCTTCC); FANCI (F: TTCTCACTGCTCTTTTCAGGGAT, R: GCCCTGTTTCCTTTAGCTGC).
Comet Assay
Comet assay experiments were performed under neutral pH conditions in TBE buffer using a commercial protocol (Trevigen, Gaithersburg, MD). DNA was stained with SYBR Green and the comets were imaged by Zeiss Imager.Z1 fluorescent microscope (Carl Zeiss AG, Oberkochen, Germany) and analyzed using the software CometScore (Autocomet.com). The tail moments were calculated for at least 50 nuclei. Experiments involving siRNA-mediated knock down required an extended time frame for androgen deprivation due to the transfection protocol used. Due to slight variations in the androgen deprivation protocol, the magnitude of changes in comet tail moment differed in these experiments, and comparisons were made within experimental batches to measure the relative effect of androgen stimulation.
siRNA mediated knockdown
3×105 LNCaP and VCaP cells were seeded per well in 6 well plates in RPMI supplemented with 10% FBS. 16 hours following plating, siRNAs targeting TOP2B (Hs_TOP2B_6 FlexiTube, Qiagen) or AR (SMARTpool: ON-TARGETplus, Dharmacon) or non-targeting control siRNA (SMARTpool, Dharmacon) were transfected using RNAiMax transfection reagent (Life Technologies). The following day, cells were washed and medium was replaced with RPMI containing 5% charcoal-stripped FBS. 72 hours after transfection, cells in androgen-depleted conditions were exposed to DHT or vehicle control for 6 hours and analyzed by western blotting and neutral comet assays.
Clonogenic survival and cell viability assays
Androgen deprived cells were diluted to the appropriate density in IMDM media containing B27 supplement (Invitrogen) and placed in triplicate into 60-mm culture dishes. DHT was added and 6 h later cells were irradiated with 4 Gy of radiation (Gammacell 40 137Cs irradiator, Atomic Energy of Canada, LTD., Ottawa, Canada). After the treatments the media was supplemented with the 10% FBS (final concentration) and cells were cultured for 4 weeks. Resulting cell colonies were then stained with a solution of crystal violet in 50% methanol and colonies consisting of ≥ 30 cells were counted. Cell viability was measured using the CellTiter-Blue assay (Promega) according to the manufacturer’s protocol. In brief, androgen-deprived LNCaP cells were harvested and exposed to 0 or 4Gy ionizing radiation, plated in 96-well plates in triplicate at 1×103 cells per well, and incubated in standard cell culture conditions for 10 days. CellTiter reagent was added to each well and cells were incubated at 37°C for 4 hours. Reduction of resazurin (560Ex) to resorufin (590Em) was measured using a fluorescent plate reader, and data are presented as viability relative to non-irradiated conditions.
Xenograft experiments
All the animal experiments were performed according to protocols approved by the Animal Care and Use Committee at Johns Hopkins University. Athymic male nude mice (nu/nu, 8 weeks old) were obtained from the Animal Center Isolation Facility at Johns Hopkins University and maintained in a sterile environment. Mice were castrated and 1-cm long polydimethylsiloxane (Silastic) implants packed with testosterone (Sigma-Aldrich) were implanted subcutaneously as described previously (8). LAPC4 cells (3× 106 cells in 50% Matrigel, 50% PBS) or LNCaP (6×106 cells in 50% Matrigel, 50% PBS) were then injected in the mouse flank. When the tumor volume reached ~ 0.1 cm3, silastic implants were removed and mice were kept at castrate androgen levels for 16 days. For tumor growth delay studies, silastic implants were reimplanted at indicated time points. All xenograft radiation experiments were performed with a single dose of 8 Gy (JL Shepherd Mark 137Cs irradiator, JL Shepherd & Associates, San Fernando, CA) delivered to the tumor (with the rest of the body shielded) 4 days prior to reimplantation (traditional group) or 12 hours after reimplantation (experimental group). The control group received the same regimen of testosterone withdrawal and replacement, but did not receive any radiation. Tumors were measured every other day to calculate tumor volume (width × length × height × 0.52). Time to 4× tumor growth was determined as the time required for the tumor to reach 4 times its volume prior to radiation treatment. Using an independent cohort of mice for pharmacokinetic and pharmacodynamic studies, castrate, LAPC4 tumor-bearing animals were injected subcutaneously with 10 μg/g DHT dissolved in sesame oil. Serial biopsies were obtained from established tumors immediately before as well as 6 h, 12 h, 24 h, 48h and 72 h after DHT injection using an automatic biopsy system (18G × 9cm, Achieve, Ref A189, CareFusion, San Diego, CA). Biopsy specimens were then either fixed in formalin embedded in paraffin or snap-frozen. Serial serum samples from individual mice were collected by submandibular bleeding.
Measurement of DHT concentration in serum and xenograft tumor tissue
Serum and tumor DHT levels were measured using a competitive immunoassay kit from Alpha Diagnostic International Inc. (#1490, San Antonio, Texas) according to the manufacturer’s protocol. Tumor tissue was first homogenized prior to suspension in Passive Lysis Buffer (E1941, Promega Corporation, Madison, WI).
Cell cycle analyses
Cells were harvested using 0.25% trypsin-EDTA and resuspended in 300 μl of PBS at 4 °C. Ice-cold methanol:acetone (1:1) was added dropwise with intermittent vortexing and cells were stored overnight at −20 °C. Cells were washed with cold PBS and incubated with 0.25 ml of 5 μg/ml RNase A for 15 minutes at 37 °C. 0.25 ml of 100 μg/ml propidium iodide was added for 1 hour at room temperature and protected from light. DNA content was measured using a FACSCalibur flow cytometer and analyzed using Cell Quest software (BD Biosciences). Doublet discrimination was accomplished by gating on forward scatter-H v. FL3A and FL3A v. FL3-H. At least 10,000 events were analyzed in each sample and samples were run in triplicate. EdU (5-ethynyl-2’-deoxyuridine) incorporation was measured according to the manufacturer’s specifications (Life Technologies). EdU was visualized using a FACSCalibur flow cytometer (BD Biosciences) and propidium iodide was included as a DNA counterstain. At least 10,000 events were analyzed in each sample and samples were run in triplicate. For pharmacological cell cycle arrest experiments, cells were treated with 50 μM Lovastatin (Selleck Chem, Houston, TX) for 36h prior the androgen stimulation experiments.
Immunohistochemistry and immunofluorescence microscopy
DSBs induced by DHT were measured by immunofluorescent staining of phosphorylated H2A.X (γH2A.X). Cells were washed, fixed in 4% paraformaldehyde and permeabilized with 0.2% Triton-X100. Anti-γH2A.X (Ser139, 07-164, Millipore, Billerica, MA) antibodies were applied for 1 h at 1:1000 dilution. Immunocomplexes were labeled with Alexa Fluor 568 conjugated goat anti-mouse IgG (Invitrogen) and slides were mounted with DAPI containing mounting media (Prolong® Gold, Invitrogen). Fluorescent images were captured using a Zeiss Imager.Z1 fluorescence microscope (Carl Zeiss AG, Oberkochen, Germany). The number of γH2A.X foci per cell was quantitated for at least 50 cells per data point using ImageJ (National Institute of Health, Bethesada, USA) and the telometer plugin. For immunohistochemical labeling of γH2A.X, slides were steamed in HTTR buffer (Dako, Carpinteria, CA) and primary antibodies (07-164, Millipore) were applied at 1:4000 dilution at room temperature for 1 h. Immunostaining for caspase 3 was performed after pretreatment in citrate buffer followed by overnight incubation with anti-cleaved caspase 3 specific antibodies at 1:1000 dilution at 4° C (5A1E, Cell Signaling Technology, Beverly, MA). Immunocomplexes were visualized using PowerVison + Poly HRP from ImmunoVision Technologies Co (Norwell, MA, USA) with DAB as the chromogen.
RESULTS
Androgen induced transient DSBs in AR positive prostate cancer cells
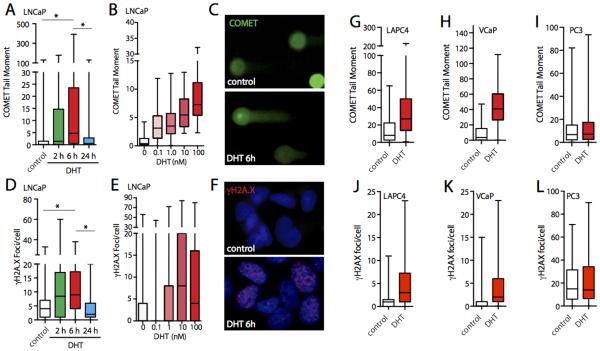
To evaluate the kinetics and extent of androgen induced DSBs in androgen responsive cells (LNCaP, LAPC4, VCaP), we treated androgen deprived cells with dihydrotestosterone (DHT). The induction of DSBs was then assessed by either neutral comet assay or immunofluorescence staining for γH2A.X. Comet tail moments as well as number of γH2A.X foci were strongly increased 6 h after DHT treatment indicating that androgen triggered induction of DSBs (Figure 1). Androgen induced DSBs appeared to be transient, since after 24 h of DHT exposure DSB levels returned to near baseline levels. DHT induced DNA damage and damage response followed a dose dependent increase and showed highest levels at supraphysiologic DHT concentrations (100 nM). Importantly, short-term androgen treatment induced both DSBs and γH2A.X foci in three AR positive cell lines (LNCaP, LAPC4 and VCaP) (Figure 1 A,D and G,J and H,K) but not in AR negative PC3 cells (Figure 1I,L) suggesting that the AR is required for mediating androgen induced DNA damage. At acute time points between 0 – 12 hours after androgen stimulation where we observed increasing DSB formation, there was no evident upregulation of DNA repair genes that were found to be induced by androgens at later time points > 24 hours here and in previous studies (9,10)(Supplementary Figure 1).
Figure 1. Kinetics, dose dependence and extent of androgen induced DSB in prostate cancer cell lines.
Box plot graphs show distribution of tail moments from neutral comet assay (A, B, G, H, I) in LNCaP (A, B), LAPC4, VCaP, and PC3 cells (G, H, I respectively), and number of γH2AX foci (D, E, J, K, L), after DHT stimulation at indicated time points. For time course studies, a final concentration of 100 nM DHT was used. All dose escalation studies were analyzed 6 h post stimulation. (C) and (F) show representative images of comet assay and γH2A.X immunostaining of cells treated with DHT or solvent control for 6 h. Androgen-induced breaks can also be observed in other AR-positive cell lines (LAPC4, VCaP) treated with 100 nM DHT for 6 h (G, H), but not in similarly treated AR-negative PC3 cells (I). * P < 0.01, *** P < 0.001.
Development of androgen induced DSB is dependent on AR and TOP2B but not on cell cycle progression
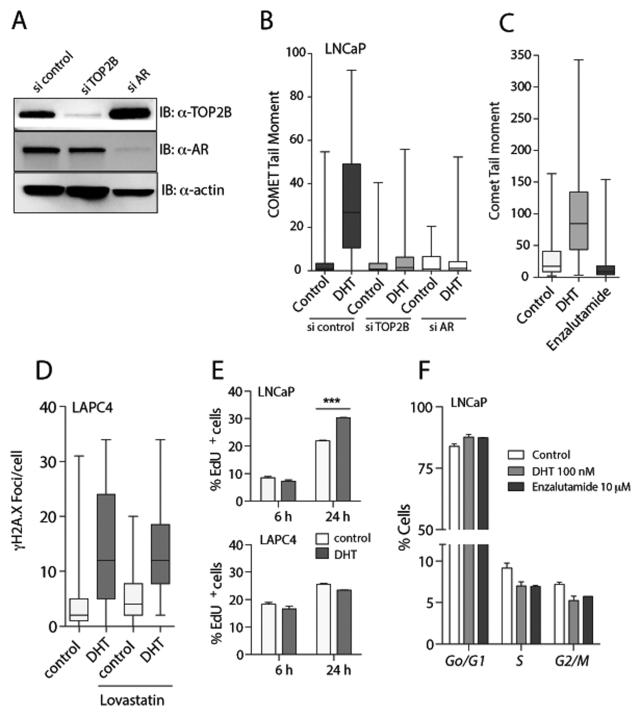
Based on previous results from Haffner and colleagues implicating AR and TOP2B activity in the formation of androgen induced double strand breaks near AR target sites (4), we hypothesized that the global androgen induced DNA damage observed here may be dependent on AR and TOP2B. To formally evaluate the role of AR and TOP2B in androgen induced DSBs we carried out transient small RNA interference (RNAi) mediated depletion of TOP2B and AR in LNCaP cells and performed neutral comet assays to survey global DNA damage (Figure 2A/B). RNAi mediated depletion of either AR or TOP2B greatly reduced the global level of androgen induced DNA DSB (Figure 2B). Similarly, TOP2B- and AR-dependence of androgen induced DNA damage was observed in VCaP cells (Supplementary Figure 2). In addition, induction of DSBs required the agonistic action of DHT on the AR, since treatment with a potent AR-antagonist (enzalutamide) did not result in an increase in comet tail moments (Figure 2C, Supplementary Figure 2C).. These observations strongly suggest that androgen induced DSBs are functionally dependent on androgen receptor mediated signaling and TOP2B activity. Since androgen stimulation can modulate cell cycle kinetics in prostate cancer cells, we investigated the possibility that cell cycle progression, particularly to S-phase or mitosis which can each be associated with replicative stress, could contribute to androgen induced DSB. We pretreated LAPC4 cells with lovastatin to induce a pharmacological G1 arrest. Lovastatin and control vehicle pre-treated cells were then stimulated with DHT and androgen induced DSBs were determined by γH2A.X staining (Figure 2D). Pharmacological cell cycle arrest did not change the levels of androgen induced γH2A.X foci, suggesting that cell cycle progression is not required for androgen mediated DSB formation. Furthermore, 6 h after DHT treatment, when highest levels of DSBs were observed, both LNCaP and LAPC4 cells did not show a significant increase in cell proliferation as measured by EdU incorporation (Figure 2E) and LNCaP did not show a change in S-phase fractions (Figure 2F) as compared to control treated cells. These results strongly suggest that androgen exposure of prostate cancer cells leads to a dose dependent induction of DSBs, which is independent of cell cycle progression but dependent on AR and TOP2B.
Figure 2. Formation of androgen induced DSB requires AR and TOP2B but not cell cycle progression.
(A) Representative western blot shows robust depletion of TOP2B and AR in si TOP2B and AR transfected cells. (B) LNCaP cells were transiently transfected with siRNAs targeting TOP2B, AR or scrambled control. Seventy-two hours after transfection and concomitant androgen deprivation, cells were treated with 100 nM DHT or vehicle control for 6 h and subjected to neutral comet assay. Shown is the distribution of COMET tail moments across 50 randomly selected cells for each condition. (C) Stimulation of LNCaP cells with the androgen receptor agonist DHT (100 nM for 6 h) induced DSBs as measured by neutral comet assay, whereas treatment with the androgen receptor antagonist enzalutamide (10 μM for 6 h) did not induce DSBs. (D) To evaluate the role of cell cycle progression in androgen induced DSB formation, LAPC4 cells were either treated with solvent control or 50 μM lovastatin for 36 h prior to stimulation with 100 nM DHT for 6 h. Note that androgen induced γH2A.X foci formation was not different between control and lovastatin pre-treated cells. (E) S-phase cell fraction, as measured by the percentage of EdU positive cells, was not different during the early phase of androgen stimulation. (F) Treatment with DHT and enzalutamide for 6 h did not significantly change cell cycle distribution of LNCaP cells grown in charcoal stripped FBS.
Short-term androgen stimulation sensitizes AR positive prostate cancer cells to IR
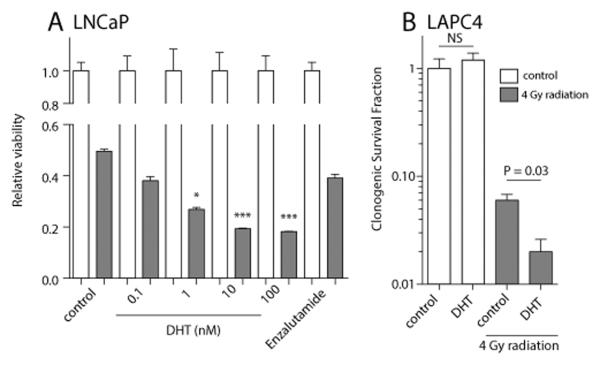
IR is known to cause a diverse array of cellular damage. The majority of the lethal effects are the consequence of IR induced DSBs, which are generated in a dose dependent manner (11). Since androgen stimulation could induce DNA damage in androgen-deprived prostate cancer cells (Figure 1 and 2) (4), we aimed to determine if exposure of AR-positive cells to supraphysiological levels of androgens for 6 hours (the point of maximal induction of DSBs, but absence of significant androgen mediated cell cycle progression as shown in Figure 1 and 2) could synergize with IR. We used two androgen dependent prostate cancer cell line models, LNCaP and LAPC4. Cells were deprived of androgens and cell viability and clonogenic survival assays were performed after short-term androgen exposure followed by IR (Figure 3). Whereas DHT treatment alone did not alter clonogenicity and cell viability, pre-stimulation of cells with 100 nM DHT for 6 h followed by 4 Gy radiation significantly decreased cell survival compared to IR alone (Figure 3). Similar to the induction of DSBs, a dose dependency was observed and only high doses of DHT (> 1 nM) resulted in significant reduction of cell viability in combination with IR. Importantly, pretreatment with the AR antagonist enzalutamide prior to IR did not decrease cell viability. Furthermore, this DHT-mediated effect was only observed in AR-positive cells (LNCaP and LAPC4) and not in AR-negative PC3 cells (Supplementary Figure 3). These in vitro data demonstrate that androgen deprivation followed by short term androgen exposure prior to IR shows a synergistic effect on reducing tumor cell viability, suggesting that altered timing of radiation to a point at which androgen induced DNA breaks are greatest could improve therapeutic outcomes. Furthermore, the androgen induced radiosensitization effect is AR dependent and occurs with supraphysiological doses of DHT.
Figure 3. Viability and survival of AR positive prostate cancer cells following IR and DHT treatments.
(A) Relative viability of LNCaP cells exposed to vehicle control, 0.1-100 nM DHT, or enzalutamide (10 μM) without IR (white bars) or in combination with 4 Gy of IR (red bars). (B) Clonogenic survival of LAPC4 cells pretreated with DHT (100 nM) for 6 h followed by 4 Gy radiation. NS, not significant.
Rapid increase of androgens to supraphysiological levels triggers DSBs and apoptosis in vivo
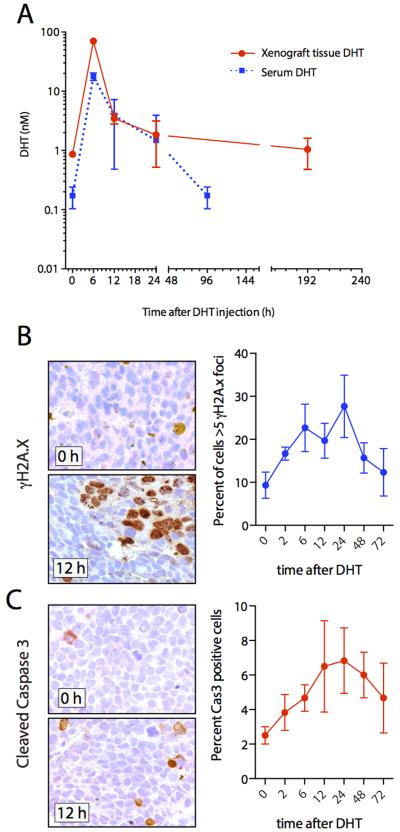
To assess whether androgen stimulation of androgen deprived prostate cancer cells would produce DSBs in vivo, we castrated mice that were bearing established LAPC4 xenograft tumors in their flanks. Two weeks after castration, animals received subcutaneous injection of 10 μg/g DHT. DHT levels were then serially determined in serum and in tumor xenograft tissues obtained from tumor biopsies (Figure 4). DHT injection resulted in a sharp increase in both tumor and serum DHT levels and peaked around 6 h post injection. Importantly, supraphysiological levels of DHT were reached in both xenograft tissues and serum (peak levels, 17.8 nM and 70.1 nM for tissue and serum respectively) (Figure 4A). Xenograft tumor biopsies were analyzed for evidence of DHT induced DSB and induction of apoptosis using immunohistochemical detection of γH2A.X and cleaved caspase 3 respectively (Figure 4A and B). In line with our previous in vitro findings, we observed an increase in γH2A.X foci over time, with highest levels detected after 24 h (Figure 4B). Importantly, γH2A.X foci returned to near baseline levels 72 h after treatment, supporting the transient nature of the androgen induced DSBs also in vivo, similar to our previous observations in vitro (Figure 1). The in vivo kinetics appeared to be different compared to the kinetics observed in vitro, likely due to the more complex pharmacokinetics of dihydrotestosterone in vivo. It is worth noting that in this model androgen stimulation alone resulted in a measurable increase in apoptosis in tumor cells, suggesting that androgen induced breaks can potentially be sufficient to induce cell death in at least a subset of cells (Figure 4C). This result might provide an explanation for previous observations showing that in vivo growth rates of several prostate cancer cell line models are inhibited by high dose androgens (12-14).
Figure 4. Androgen treatment induces DSB and apoptosis in prostate cancer cells in vivo.
(A) DHT concentration in tumor xenograft tissue (red solid line) and serum (blue dotted line) from LAPC4 xenograft bearing mice (n=3 for each time point) injected with 10 μg/g DHT. (B, C) Representative images of serial biopsies from LAPC4 xenogaft tumors immunolabeled for γH2A.X and cleaved caspase 3. Corresponding graphs show the percentage of cells with > 5 γH2A.X foci and percent of cleaved caspase 3 positive cells in 6 high power fields for each time point.
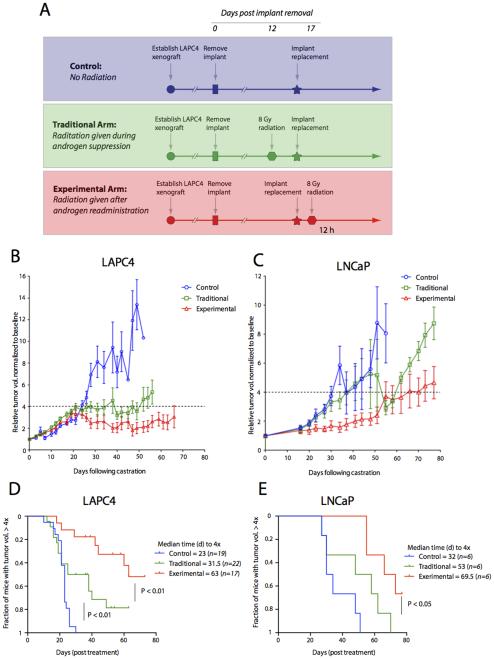
Androgen and IR synergize in vivo in controlling prostate cancer xenograft growth
A combination of ADT together with fractionated IR is currently the standard of care for locally advanced prostate cancer. IR is usually given when testosterone levels are at a nadir and ADT is continued for a period of time after radiation. To mimic a clinically relevant treatment scenario in an in vivo model of human prostate cancer we used the two androgen responsive cell lines LAPC4 and LNCaP, which show robust response to IR (Supplementary Figure 3). Xenografts of LNCaP and LAPC4 cells were established in castrate nude mice in which testosterone levels were controlled by a silastic testosterone containing implant. When tumors reached a consistent size, the implant was removed to achieve castrate testosterone levels. After 16 days of castration, xenograft tumors received either no IR (control) or a single dose of 8 Gy IR during castration (traditional) or 12 h after (experimental) re-administration of the testosterone implant producing an acute peak of testosterone levels (Figure 5A). Tumor size was then evaluated longitudinally over a period of 8 weeks and time to progression (defined as > 4x tumor volume) was calculated. Tumors that were not irradiated showed a rapid progression (Figure 5D and E), as did tumors that were irradiated without androgen manipulation (Supplementary Figure 4). Most interestingly, mice that received testosterone implants 12 h prior to IR showed smaller tumor sizes (Figure 5B and C) and a significantly smaller fraction reached the progression benchmark (Figure 5D and E) compared to mice that received IR during castration when testosterone levels were at a nadir (Figure 5B, p < 0.01 and C, p < 0.05). These results suggest that, in the setting of ADT, stimulation with supraphysiological androgen levels prior to IR may improve treatment outcomes for radiation therapy of prostate cancer.
Figure 5. Androgen repletion and IR result in increased tumor growth delay in vivo.
(A) Schema of study design. A single dose of 8 Gy of radiation is given 4 days prior to implant replacement (traditional group) or 12 hours after implant replacement (experimental groups). The control group did not receive any IR. Relative tumor growth is shown in LAPC4 (B) and LNCaP (C). Kaplan Meier curves indicate time to tumor size progression to > 4× of the initial volume in LAPC4 (D) and LNCaP (E).
DISCUSSION
Given that androgens are known to fuel prostate cancer growth and survival, androgen suppression has been a mainstay of therapy for advanced prostate cancer. For locally advanced prostate cancer, several large clinical trials have demonstrated the supremacy of radiation therapy combined with ADT compared to radiation therapy alone or ADT alone (2,15). A number of recent studies have demonstrated a reciprocal link wherein DNA damage/IR can upregulate androgen signaling, and androgen signaling can upregulate DNA repair genes (9-11,16-18) These studies have suggested that these mechanisms may underlie the known clinical benefit of ADT combined with IR for treatment of high-risk prostate cancer.
While these data support a potential mechanism by which today's standard use of neo-adjuvant and concomitant androgen suppression and radiation may work, here, we explore a new paradigm to exploit the androgen receptor axis through the effects of high dose, short term androgen treatment during ADT in combination with IR. The data presented here demonstrates that androgen treatment of androgen-deprived prostate cancer cells generates transient DSBs in a dose dependent manner that can synergize with IR-induced DNA damage in substantially reducing tumor growth. What initially appears to be a counter-intuitive strategy for the treatment of advanced prostate cancer was in part motivated by compelling preclinical evidence from in vitro and in vivo prostate cancer models demonstrating that, high-dose androgens can decrease tumor cell growth and viability (12-14). Furthermore, several proof-of principle clinical trials have established that supraphysiologic androgens can be safely given to patients with recurrent or metastatic prostate cancer (14,19,20). More encouragingly, a number of case reports and a recent proof-of-principle trial have shown dramatic reduction of disease burden after androgen treatment in patients with castration resistant metastatic disease (13,14,21-23). Taken together, these observations support a view that androgen stimulation can generate vulnerabilities for AR-positive prostate cancer cells (21). Our results show that supraphysiological concentrations of DHT (≥10 nM) robustly induce DNA breaks, as measured by neutral comet assay, and result in the activation of DNA damage response pathways, as evident by increased H2A.x phosphorylation. Importantly, we show that the induction of androgen induced DSBs is independent of androgen induced cell cycle progression since pharmacological cell cycle arrest at G1 does not decrease the level of androgen induced DSBs (Figure 2). Furthermore, at time points where maximal induction of DSBs is observed (in vitro after 6 h) no significant cell cycle progression and DNA synthesis is present (Figure 2). These observations suggest that the generation of androgen induced DSBs is not associated with replicative stress in S-phase or mitosis, and more likely involves processes involved in transcriptional initiation. Mechanistically, we show that the induction of DSBs is dependent on AR and TOP2B since transient RNAi mediated depletion of AR and TOP2B blocked the induction of androgen induced DSBs. This is the first evidence that the nucleus-wide hormone induced DSBs are mediated by TOP2B in prostate cancer cells. DSBs mediated by TOP2B have been shown to occur during initiation of transcriptional programs and are thought to be transient in nature and are likely repaired by TDP2 followed by non-homologous end joining (4,21,24,25). Since the nature of topoisomerase (26) and IR induced DNA lesions (27) differ, both DNA insults occurring at the same time could overwhelm the repair machinery and therefore result in increased cell death. Furthermore, given that induction of DSBs has also been shown to occur during transcriptional initiation of other nuclear hormone receptors such as the estrogen receptor, it will be interesting to assess whether estrogen stimulation of ER-positive cells (or generally hormone stimulation of cells signaling through the cognate hormone receptor) can also result in radiosensitization in future studies (5,6).
Several potential alternative or complementary mechanisms for the observed synergy of androgen stimulation and IR may also be at play. It is well established that androgen receptor signaling can influence the cell cycle. In particular, androgen stimulation of androgen depleted cells induces proliferation and cell cycle entry (28); therefore modulation of cell cycle kinetics may be responsible for differential radiosensitization. Here we show that short-term androgen exposure does not result in significant changes in cell cycle distribution of prostate cancer cells. Furthermore, androgen induced breaks can also be induced in cells which have been pharmacologically arrested in G1. Therefore androgen-induced changes in cell cycle kinetics is an unlikely explanation for the observed increased sensitization at early time points after androgen stimulation. In previous preclinical and clinical studies, hormonal treatment was used to synchronize cells in vulnerable cell cycle phases for subsequent radiation and chemotherapies (29). The approach outlined here is fundamentally different from these studies. Activation of hormonal signaling is not used for cell cycle synchronization; in fact at the time points used in the present study no changes in cell cycle progression are detected, but a different mechanism that likely relies on a cell-cycle independent induction of DSBs is exploited. It is important to highlight that the timing of hormone exposure, induction of DSBs and resulting radiosensitization needs to be carefully examined in further pre-clinical and clinical studies in order to investigate the potential to apply this paradigm clinically for radiosensitization of human prostate cancers.
Additionally, the in vitro and in vivo studies (performed in athymic nude mice) carried out here are largely focused on studying the effect of high-dose androgens and IR on cancer cell intrinsic DNA damage, response, and growth/survival. Interestingly, since it has been observed in previous studies that both androgen treatment and IR can have important effects on tumor microenvironmental, and host immune responses (30-34), it will be important to examine these cancer cell extrinsic mechanisms during further pre-clinical and clinical studies investigating the effectiveness of acute androgen stimulation to sensitize to radiation therapies.
An important aspect of the above described androgen induced breaks is that this mechanism relies on the induction of androgen signaling. It is therefore possible that multiple cycles of androgen depletion followed by androgen stimulation would lead to DSB induction and radiosensitization at each cycle, and thus better control. Such an androgen cycling schema, in which a patient undergoing ADT would receive multiple pulses of androgen boosts followed by IR could be implemented into the currently used fractionated radiation treatment regimens. More generally, the observation that androgen stimulation of androgen deprived cells selectively sensitizes AR-positive cells to IR has important implications for prostate cancer radiation therapy. Many normal pelvic tissues that are exposed to IR during external beam radiation therapy of the prostate do not express AR. Therefore, the above described approach should improve therapeutic index by selectively sensitizing prostate cancer cells to IR without sensitizing normal pelvic tissues and enhancing toxicity. One important consequence of this unique tissue specific sensitization could be to allow for a reduction of the IR total dose, improving direct radiation related quality of life outcomes. Furthermore, as noted in previous studies, intermittent high-dose androgen therapy can significantly improve the overall quality of life of patients undergoing ADT (14).
The data presented here could be viewed as contradictory to two previously published studies that suggest that anti-androgen treatment results in a reduced expression of androgen regulated DNA repair genes and decreased classical non-homologous end-joining capacity (9,10). These observations suggest that androgen regulated DNA repair genes are involved in the immediate repair of DNA damage inflicted by IR. It is however very important to note that the androgen-induced transcriptional up-regulation of DNA repair proteins observed in those studies was not apparent in the acute time scales where we observe maximal DSB formation (0-12 hours after stimulation), but appears to be a later response occurring after 24 hours, concomitant with the resolution of DSBs (see Figure 1 and Supplementary Figure 1). We speculate that such upregulation of DNA repair genes by AR signaling at later time points may represent a programmed mechanism occurring as a response to the propensity for androgen signaling to induce DNA damage. Therefore, the timing of androgen treatment and IR is critical. Here we show that administration of IR in this vulnerable phase shortly after DHT treatment results in reduced cancer cell growth both in vivo and in vitro. In this phase, when androgen induced DSBs are at a high level and induction of androgen responsive repair pathways has not fully developed, a window for a true synergistic effect between androgen and IR induced DSBs seems to exist. Furthermore, here we show that supraphysiologic levels of DHT are required to robustly induce DSBs. In both studies by Goodwin et al. and Polkinghorn et al. lower concentration of androgens are used, which might have different effects on cancer cell survival (9,10). Additionally, in the paradigm proposed here, high-dose androgen pulses would be given during IR therapy in the context of long-term ADT; patients would return to castrate hormone levels in between pulses of high-dose androgen, and during adjuvant hormonal therapy after fractionated IR treatment. Thus, the paradigm presented here would still take advantage of the known benefit of neoadjuvant and adjuvant ADT, the mechanistic basis of which has been attributed to inhibition of the reciprocal link between IR induced AR signaling, and androgen induced DNA damage response (9-11,16-18). Therefore, the present study represents an important extension of our previous knowledge on androgen receptor signaling and DNA damage. The observations in this study and these previous studies are mutually supportive and suggest a time and dose dependent differential response of androgen signaling in prostate cancer cells exposed to IR.
In conclusion we provide the first evidence that high dose androgens can create a vulnerable time period for sensitization to IR in AR-positive prostate cancer cells. An appropriately timed combination of high dose androgens and IR can increase tumor cell death and reduce prostate cancer cell growth. Taken together, our data support further investigation of using androgen stimulation of androgen deprived prostate cancer cells to selectively sensitize AR-positive prostate cancer cells to genotoxic agents such as ionizing radiation.
Supplementary Material
STATEMENT OF TRANSLATIONAL RELEVANCE.
Radiation therapy combined with pharmacological androgen suppression is standard of care for locally advanced and high risk prostate cancer. Paradoxically, recent evidence suggests that acute androgen stimulation of prostate cancer cells results in substantial DNA damage. Here we explore the therapeutic utility of this paradigm and show that in vitro and in vivo short-term treatment with androgens under castrate conditions improves therapeutic responses to subsequent radiation therapy. These data suggest that androgen and ionizing radiation induced DSBs might function synergistically and that high-dose androgen given in the appropriate time window before ionizing radiation could have radiosensitizing effects. This paradigm could allow selective radiosensitization of AR-positive prostate cancer cells while sparing nearby normal tissues, potentially improving therapeutic index of ionizing radiation, thus warranting further clinical investigation.
ACKNOWLEDGMENTS
The authors would like to acknowledge Marcella Sutherland and Bonnie Gambichler from the Johns Hopkins TMA core facility for their help with tissue processing and Jessica Hicks for advice on immunostaining.
Funding: This study was supported in part by grants from the National Institutes of Health/National Cancer Institute grants R01CA183965 (S.Y.), P50CA058236 (T.L.D., W.G.N., S.Y.), R01CA070196 (W.G.N., S.Y.), P30CA006973, the Prostate Cancer Foundation (S.Y. and W.G.N.), and the Ellen B. Masenhimer Fellowship (S.Y.). M.C.H. is supported by a Young Investigator Award from the Prostate Cancer Foundation. A generous gift from Michael Armstrong helps support M.H.
Footnotes
Conflict of interest: None.
References
- 1.Siegel RL, Miller KD, Jemal A. Cancer statistics, 2015. CA A Cancer Journal for Clinicians. 2015;65:5–29. doi: 10.3322/caac.21254. [DOI] [PubMed] [Google Scholar]
- 2.Bolla M, Van Tienhoven G, Warde P, Dubois JB, Mirimanoff R-O, Storme G, et al. External irradiation with or without long-term androgen suppression for prostate cancer with high metastatic risk: 10-year results of an EORTC randomised study. Lancet Oncol. 2010;11:1066–73. doi: 10.1016/S1470-2045(10)70223-0. [DOI] [PubMed] [Google Scholar]
- 3.Palacios DA, Miyake M, Rosser CJ. Radiosensitization in prostate cancer: mechanisms and targets. BMC Urol. BioMed Central Ltd. 2013;13:4. doi: 10.1186/1471-2490-13-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Haffner MC, Aryee MJ, Toubaji A, Esopi DM, Albadine R, Gurel B, et al. Androgen-induced TOP2B-mediated double-strand breaks and prostate cancer gene rearrangements. Nat Genet. 2010;42:668–75. doi: 10.1038/ng.613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Ju B-G, Lunyak VV, Perissi V, Garcia-Bassets I, Rose DW, Glass CK, et al. A topoisomerase IIbeta-mediated dsDNA break required for regulated transcription. Science. American Association for the Advancement of Science; 2006;312:1798–802. doi: 10.1126/science.1127196. [DOI] [PubMed] [Google Scholar]
- 6.Williamson LM, Lees-Miller SP. Carcinogenesis. Vol. 32. Oxford University Press; 2011. Estrogen receptor α-mediated transcription induces cell cycle-dependent DNA double-strand breaks; pp. 279–85. [DOI] [PubMed] [Google Scholar]
- 7.Wong RHF, Chang I, Hudak CSS, Hyun S, Kwan H-Y, Sul HS. A role of DNA-PK for the metabolic gene regulation in response to insulin. Cell. 2009;136:1056–72. doi: 10.1016/j.cell.2008.12.040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kyprianou N, Isaacs JT. Quantal relationship between prostatic dihydrotestosterone and prostatic cell content: critical threshold concept. Prostate. 1987;11:41–50. doi: 10.1002/pros.2990110106. [DOI] [PubMed] [Google Scholar]
- 9.Goodwin JF, Schiewer MJ, Dean JL, Schrecengost RS, de Leeuw R, Han S, et al. A hormone-DNA repair circuit governs the response to genotoxic insult. Cancer Discovery. American Association for Cancer Research. 2013;3:1254–71. doi: 10.1158/2159-8290.CD-13-0108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Polkinghorn WR, Parker JS, Lee MX, Kass EM, Spratt DE, Iaquinta PJ, et al. Androgen Receptor Signaling Regulates DNA Repair in Prostate Cancers. Cancer Discovery. 2013;3:1245–53. doi: 10.1158/2159-8290.CD-13-0172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Jackson SP, Bartek J. The DNA-damage response in human biology and disease. Nature. 2009;461:1071–8. doi: 10.1038/nature08467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Umekita Y, Hiipakka RA, Kokontis JM, Liao S. Human prostate tumor growth in athymic mice: inhibition by androgens and stimulation by finasteride. Proc Natl Acad Sci USA. 1996;93:11802–7. doi: 10.1073/pnas.93.21.11802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Isaacs JT, D'Antonio JM, Chen S, Antony L, Dalrymple SP, Ndikuyeze GH, et al. Prostate. Vol. 72. Wiley Subscription Services, Inc., A Wiley Company; 2012. Adaptive auto-regulation of androgen receptor provides a paradigm shifting rationale for bipolar androgen therapy (BAT) for castrate resistant human prostate cancer; pp. 1491–505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Schweizer MT, Antonarakis ES, Wang H, Ajiboye AS, Spitz A, Cao H, et al. Effect of bipolar androgen therapy for asymptomatic men with castration-resistant prostate cancer: Results from a pilot clinical study. Sci Transl Med. American Association for the Advancement of Science. 2015;7:269ra2–269ra2. doi: 10.1126/scitranslmed.3010563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Widmark A, Klepp O, Solberg A, Damber J-E, Angelsen A, Fransson P, et al. Endocrine treatment, with or without radiotherapy, in locally advanced prostate cancer (SPCG-7/SFUO-3): an open randomised phase III trial. Lancet. 2009;373:301–8. doi: 10.1016/S0140-6736(08)61815-2. [DOI] [PubMed] [Google Scholar]
- 16.Tarish FL, Schultz N, Tanoglidi A, Hamberg H, Letocha H, Karaszi K, et al. Castration radiosensitizes prostate cancer tissue by impairing DNA double-strand break repair. Sci Transl Med. 2015;7:312re11–1. doi: 10.1126/scitranslmed.aac5671. [DOI] [PubMed] [Google Scholar]
- 17.Spratt DE, Evans MJ, Davis BJ, Doran MG, Lee MX, Shah N, et al. Androgen Receptor Upregulation Mediates Radioresistance after Ionizing Radiation. Cancer Res. American Association for Cancer Research; 2015;75:4688–96. doi: 10.1158/0008-5472.CAN-15-0892. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Al-Ubaidi FLT, Schultz N, Loseva O, Egevad L, Granfors T, Helleday T. Castration therapy results in decreased Ku70 levels in prostate cancer. Clin Cancer Res. American Association for Cancer Research; 2013;19:1547–56. doi: 10.1158/1078-0432.CCR-12-2795. [DOI] [PubMed] [Google Scholar]
- 19.Feltquate D, Nordquist L, Eicher C, Morris M, Smaletz O, Slovin S, et al. Rapid androgen cycling as treatment for patients with prostate cancer. Clin Cancer Res. American Association for Cancer Research; 2006;12:7414–21. doi: 10.1158/1078-0432.CCR-06-1496. [DOI] [PubMed] [Google Scholar]
- 20.Morris MJ, Huang D, Kelly WK, Slovin SF, Stephenson RD, Eicher C, et al. Phase 1 trial of high-dose exogenous testosterone in patients with castration-resistant metastatic prostate cancer. Eur Urol. 2009;56:237–44. doi: 10.1016/j.eururo.2009.03.073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Haffner MC, De Marzo AM, Meeker AK, Nelson WG, Yegnasubramanian S. Transcription-induced DNA double strand breaks: both oncogenic force and potential therapeutic target? Clin Cancer Res. American Association for Cancer Research; 2011;17:3858–64. doi: 10.1158/1078-0432.CCR-10-2044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Mathew P. Prolonged control of progressive castration-resistant metastatic prostate cancer with testosterone replacement therapy: the case for a prospective trial. Ann Oncol. Oxford University Press; 2008;19:395–6. doi: 10.1093/annonc/mdm568. [DOI] [PubMed] [Google Scholar]
- 23.Prout GR, Brewer WR. Response of men with advanced prostatic carcinoma to exogenous administration of testosterone. Cancer. 1967;20:1871–8. doi: 10.1002/1097-0142(196711)20:11<1871::aid-cncr2820201112>3.0.co;2-d. [DOI] [PubMed] [Google Scholar]
- 24.Ashour ME, Atteya R, El-Khamisy SF. Topoisomerase-mediated chromosomal break repair: an emerging player in many games. Nat Rev Cancer. 2015;15:137–51. doi: 10.1038/nrc3892. [DOI] [PubMed] [Google Scholar]
- 25.mez-Herreros FGO, Schuurs-Hoeijmakers JHM, McCormack M, Greally MT, Rulten S, Romero-Granados RIO, et al. TDP2 protects transcription from abortive topoisomerase activity and is required for normal neural function. Nat Genet. Nature Publishing Group. 2014:1–9. doi: 10.1038/ng.2929. [DOI] [PubMed] [Google Scholar]
- 26.Schellenberg MJ, Appel CD, Adhikari S, Robertson PD, Ramsden DA, Williams RS. Mechanism of repair of 5'-topoisomerase II-DNA adducts by mammalian tyrosyl-DNA phosphodiesterase 2. Nature Publishing Group. Nature Publishing Group; 2012;19:1363–71. doi: 10.1038/nsmb.2418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Jeggo P, Löbrich M. Radiation-induced DNA damage responses. Radiat Prot Dosimetry. 2006;122:124–7. doi: 10.1093/rpd/ncl495. [DOI] [PubMed] [Google Scholar]
- 28.Schiewer MJ, Augello MA, Knudsen KE. The AR dependent cell cycle: mechanisms and cancer relevance. Mol Cell Endocrinol. 2012;352:34–45. doi: 10.1016/j.mce.2011.06.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Lippman ME, Sorace RA, Bagley CS, Danforth DW, Lichter A, Wesley MN. Treatment of locally advanced breast cancer using primary induction chemotherapy with hormonal synchronization followed by radiation therapy with or without debulking surgery. NCI Monogr. 1986:153–9. [PubMed] [Google Scholar]
- 30.Kissick HT, Sanda MG, Dunn LK, Pellegrini KL, On ST, Noel JK, et al. Androgens alter T-cell immunity by inhibiting T-helper 1 differentiation. Proc Natl Acad Sci USA. National Acad Sciences; 2014;111:9887–92. doi: 10.1073/pnas.1402468111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ludgate CM. Optimizing cancer treatments to induce an acute immune response: radiation Abscopal effects, PAMPs, and DAMPs. Clin Cancer Res. American Association for Cancer Research; 2012;18:4522–5. doi: 10.1158/1078-0432.CCR-12-1175. [DOI] [PubMed] [Google Scholar]
- 32.Leach DA, Need EF, Toivanen R, Trotta AP, Palenthorpe HM, Tamblyn DJ, et al. Stromal androgen receptor regulates the composition of the microenvironment to influence prostate cancer outcome. Oncotarget. 2015;6:16135–50. doi: 10.18632/oncotarget.3873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Yang W, Levine AC. Androgens and prostate cancer bone metastases: effects on both the seed and the soil. Endocrinol Metab Clin North Am. 2011;40:643–53–x. doi: 10.1016/j.ecl.2011.05.001. [DOI] [PubMed] [Google Scholar]
- 34.Morse MD, McNeel DG. Prostate cancer patients on androgen deprivation therapy develop persistent changes in adaptive immune responses. Hum Immunol. 2010;71:496–504. doi: 10.1016/j.humimm.2010.02.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.