Abstract
Background
While trauma patients are frequently cared for in the ICU, admission triage criteria are unclear and may vary among providers and institutions. The benefits of close monitoring must be weighed against the economic and opportunity costs of an ICU admission.
Materials and Methods
We conducted a retrospective cohort study of patients treated for blunt splenic injuries at 30 level I and II Pennsylvania trauma centers, 2011–2014. We used multivariable logistic regression to assess the relationship between ICU admission and mortality, adjusting for patient characteristics, injury characteristics, and physiology. We calculated center-level observed-to-expected ratios for ICU utilization and mortality and evaluated correlations with Spearman’s rho. We compared the proportion of patients receiving critical care procedures, such as mechanical ventilation or central line placement, between high- and low-ICU-utilization centers.
Results
Of 2,587 patients with blunt splenic injuries, 63.9% (1,654) were admitted to the ICU. Median injury severity score (ISS) was 17 overall, 13 for non-ICU patients and 17 for ICU patients (p < 0.001). In multivariable logistic regression, ICU admission was not significantly associated with mortality. Center-level risk-adjusted ICU admission rates ranged from 17.9% to 87.3%. Risk-adjusted mortality rates ranged from 1.2% to 9.6%. There was no correlation between O:E ratios for ICU utilization and mortality (rs = −0.2595, p=0.2103). Proportionately fewer ICU patients at high-utilization centers received critical care procedures than at low-utilization centers.
Conclusions
Risk-adjusted ICU utilization rates for splenic trauma varied widely among trauma centers, with no clear relationship to mortality. Standardizing ICU admission criteria could improve resource utilization without increasing mortality.
Keywords: blunt splenic injury, ICU utilization, trauma outcomes
1. Background
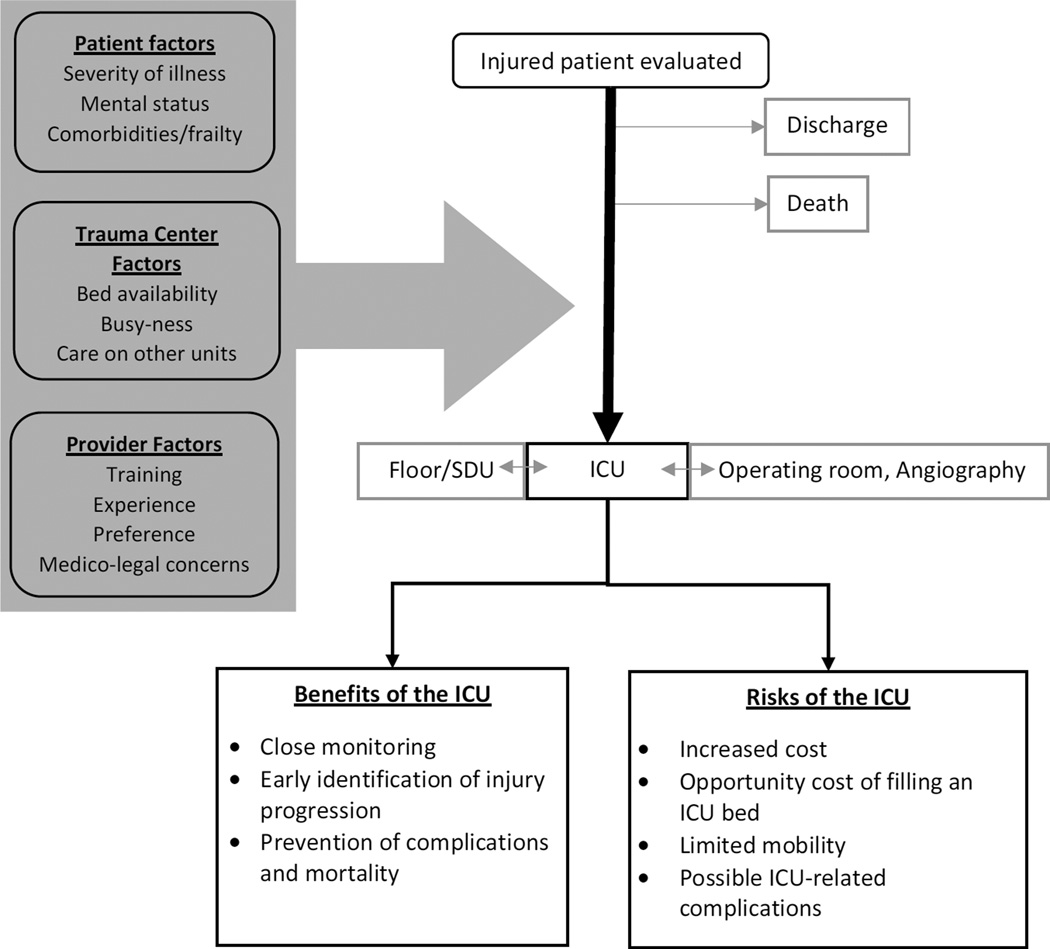
After traumatic injury, patients are commonly admitted to the ICU for post-operative observation or for a trial of non-operative management. Many of these patients do not require any ICU-specific care such as mechanical ventilation or pressor support. The goal of ICU admission in these patients is to facilitate close observation, with early identification of clinical deterioration and swift intervention to prevent adverse outcomes. However, an ICU stay in itself comes with increased cost to the patient and their insurer, and carries an opportunity cost to the trauma center by filling an ICU bed that could otherwise have gone to another patient.1 An ICU stay may also contribute to increased risk of complications such as delirium and hospital-acquired infection. Figure 1 shows a conceptual model of the role of ICU admission in trauma. Appropriate use of ICU resources is an important target for performance improvement not only in the context of increasing cost-containment pressure in the US health care system, as ICU care accounts for up to 1/3 of national hospital costs,2 but also as part of ongoing quality improvement efforts in trauma.
Figure 1.
Conceptual Model of ICU Utilization in Trauma
The spleen is the most commonly injured solid organ in blunt trauma, with mortality rates of approximately 13%.3 Since the 1970s, preservation of the spleen has been an increasingly prominent goal of post-injury management, with the aim of sparing patients both operative morbidity and the lifelong increased susceptibility to infection associated with splenectomy.4,5 Non-operative management strategies for blunt splenic injuries range from angiography with embolization of the spleen to observation in or out of the hospital. Observation, with or without angiography, has been advocated for children and adults, as well as for low- and high-grade injuries in stable patients.6 Non-operative management has been shown to be safe, with approximately 10% of patients proceeding to operation for deteriorating clinical status within 24 hours, and approximately 3% requiring later splenectomy.5–8 However, there is considerable variation in practice among surgeons and between centers with respect both to criteria for nonoperative management, and for the details of this practice.9
ICU utilization for blunt splenic injuries appears to vary among centers. In one pediatric study, ICU admission ranged from 9–73% for grade 1 and 2 abdominal injuries, and from 18–82% for grade 3–5.10 Physician preferences vary similarly. In a survey of AAST members, Zarzaur et al. found substantial variation in ICU utilization, with the percentage of respondents recommending ICU admission ranging from 8.1% for grade 1 injuries to 26.5% for grade 2 injuries, and 70% preferring ICU for grade 3.9 Even so, the 2012 EAST Practice Management Guidelines for non-operative management of blunt splenic injury note that despite a growing body of literature, many questions remain unanswered, including “optimum length of stay for both the ICU and hospital.”11 To evaluate the role of ICU admission in patients with splenic injuries, we conducted a retrospective cohort study of prospectively-collected data from the Pennsylvania Trauma Outcomes Study. We hypothesized that ICU utilization would vary significantly among centers; that centers with higher-than-expected ICU utilization would have correspondingly lower mortality; and that patients admitted to the ICU would have lower risk-adjusted mortality than those admitted elsewhere.
2. Materials and Methods
2. 1 Setting and Population
The Pennsylvania Trauma Outcomes Survey (PTOS) registry was queried to identify patients treated at all 30 level I and II trauma centers in Pennsylvania for splenic injury from 2011–2014. This state trauma registry includes all patients admitted for at least 48 hours; all patients admitted for at least 36 hours with injury severity score ≥9; all ICU and step down admissions regardless of duration; all deaths; and all transfers. Patients are excluded from the registry if they have an isolated hip fracture, or injuries due to asphyxiation, drownings, poisonings, or in-hospital injury.12
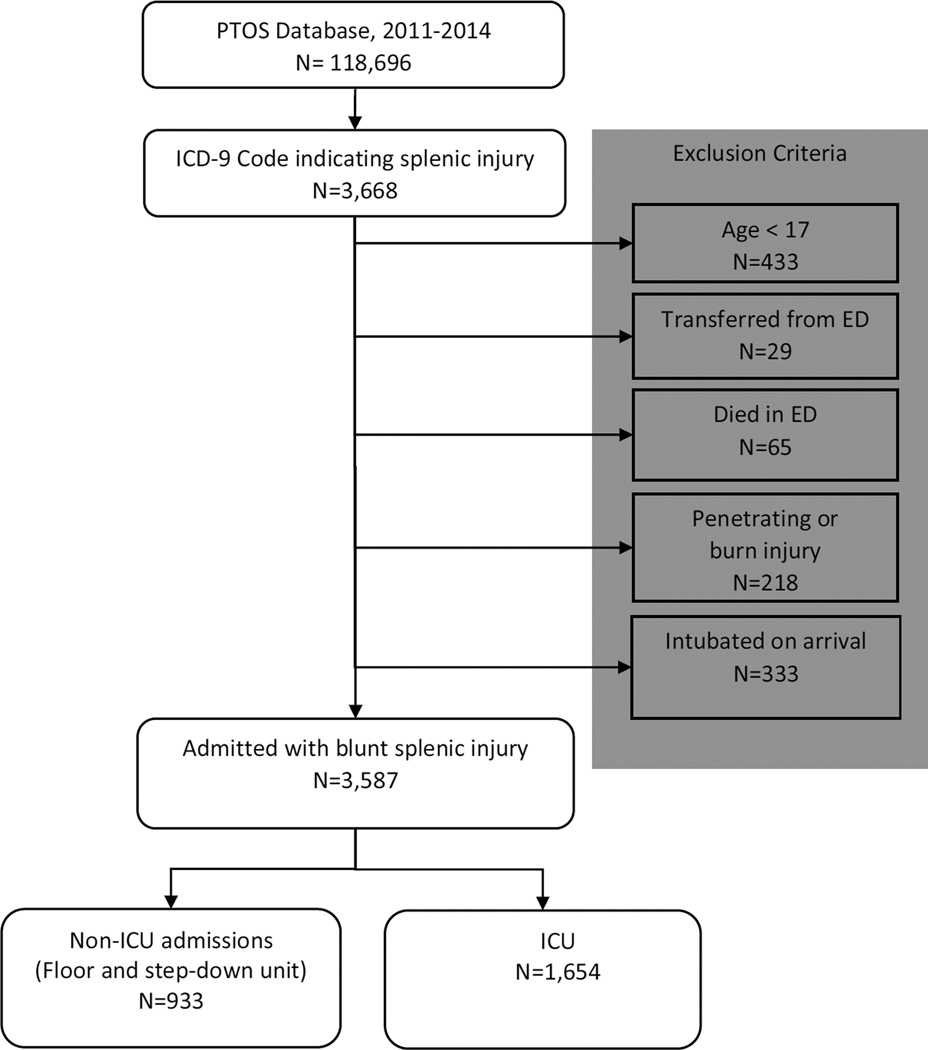
As shown in Figure 2, Patients were included if they had an ICD-9 code indicating splenic injury (Codes 865.0–865.19). Patients were excluded if their age was <17, if the mechanism of injury was penetrating, or if they died in or were discharged from the trauma bay. Patients who were transferred out of the trauma center were excluded, as their outcomes could not be determined. Patients transferred into the ED from another institution were included. Patients who were intubated on arrival were also excluded, as this would mandate admission to the ICU. The center-level analysis excluded centers with < 20 eligible patients, and centers with zero mortalities. ICU utilization was defined as ICU admission from the trauma bay or after an immediate operation. Patients first admitted to another ward may have been transferred to the ICU later in their clinical course, but were categorized as non-ICU admissions in this analysis.
Figure 2.
Study Inclusion and Exclusion Criteria
The PTOS registry is maintained by the Pennsylvania Trauma Systems Foundation, Mechanicsburg, Pennsylvania, (PTSF) which is the trauma center accrediting body for the commonwealth of Pennsylvania. Data was received with no identifying patient information. Patients were linked to trauma centers, but these centers were also only labeled by a de-identified trauma center number. This study was performed with the permission of PTSF, which specifically disclaims responsibility for any analyses, interpretations or conclusions. The study was deemed exempt by the University of Pennsylvania Institutional Review Board.
2.2 Statistical analysis
The primary outcome for the patient-level analysis was in-hospital mortality. The primary exposure was ICU admission. Additional variables included demographics; injury diagnoses; mechanism of injury; injury severity score (ISS); admission Glasgow Coma Score (GCS); admission systolic blood pressure (SBP); and pre-existing conditions. Comorbidities evaluated included: cardiac disease, hypertension, ulcer disease, history of bariatric surgery, use of anticoagulant or antiplatelet medication, psychiatric disease, attention deficit disorder, HIV/AIDS, use of steroids, cirrhosis, arthritis, dementia, Parkinson’s disease, stroke, chronic renal insufficiency, alcohol abuse, drug abuse, prior traumatic brain injury, thyroid disease, ascites, functional dependency, peripheral vascular disease, chronic lung disease, diabetes, cancer, and smoking. Splenic injuries were classified as AAST Grade 1–2 or Grade 3–5. Grade was estimated from ICD-9 codes using validated methodology.13 Trauma centers report up to 3 levels of alert, as well as trauma consults, with requirements for each level determined by each institution. Categorical variables were compared using Chi2 tests, and continuous variables were compared using the Wilcoxon rank-sum test. We conducted bivariate logistic regression analyses on candidate predictors with both ICU admission and mortality as outcomes. For each outcome, predictors with a p-value of <0.2 in bivariate analysis were included in multivariable logistic regression models. Standard errors of the final models were adjusted for clustering at the center level.
To estimate risk-adjusted center-level ICU utilization rates and mortality rates, we added center fixed effects to each final model, and compared model fit with and without the center predictor using the likelihood ratio test. We used the risk adjustment models from the patient level analysis to calculate observed-to-expected (O:E) center-level ICU utilization and mortality ratios with 95% confidence intervals and evaluated correlations between ICU utilization and mortality with Spearman’s rho. Centers were classified as outliers if their 95% confidence interval did not cross 1. Caterpillar plots were constructed demonstrating center rankings and identifying outliers. We divided centers into quartiles based on ICU utilization O:E ratios. We tabulated the proportion of ICU patients receiving each of 4 critical care procedures at each center, as well as the mean number of procedures received, and the proportion with no specific critical care procedure documented. We identified procedures associated with clear ICD-9 procedure codes that we expected to be common across centers: intubation or mechanical ventilation, central line placement, arterial line placement or monitoring, and tracheostomy. For all except tracheostomy, we limited consideration to procedures documented to occur in the ICU, to exclude, for example, intubation and ventilation limited to the OR. Use of hemodialysis was included in the total number of procedures received, but was not examined separately, as a total of only 18 patients were documented as receiving dialysis. No patient in the dataset had a procedure code associated with vasopressor infusion. We compared the proportion of ICU patients receiving each procedure between centers in the highest quartile and centers in the lowest quartile of ICU O:E using Chi2 tests.
2.3 Sensitivity Analyses
To assess the robustness of our model, we assessed included predictors for multicollinearity, and analyzed the performance of two limited models for mortality, one using only ICU admission and a propensity score for ICU admission as predictors, and another using only ICU admission, injury severity, age, and admitting physiology as predictors.
To maximize our sample size and to limit the risk of ascertainment bias, we include patients with heterogeneous injury patterns. We conducted subgroup analyses excluding patients with moderate-to-severe head injuries (AIS ≥ 3 for body region 1), and excluding patients with moderate-to-severe injuries (AIS ≥ 3) to any body region except the abdomen.
All analyses were conducted using Stata (Version 14.1, 2015, StataCorp, College Station TX).
3. Results
3.1 Patient-level analysis
A total of 2,587 patients were included in the analysis, of whom 63.9% (1,654) were admitted to the ICU. This ranged from 59.9% of patients with grade 1–2 splenic injuries to 72.1% of those with grade ≥3. Median ISS was 17 overall, 13 for non-ICU patients and 17 for ICU patients (p < 0.001). Trauma with the highest alert level were more likely to result in ICU admission. All additional injuries were more common among ICU patients, including head injury, skeletal injury, thoracic injury, and other abdominal injuries. ICU patients had lower admission GCS and SBP, and were more likely to have received ≥2L of fluid during pre-hospital transport or blood transfusions in the emergency department. A history of cancer was slightly more common in ICU than non-ICU patients (1.4% vs. 0.5%, p=0.04), as was a history of drug abuse (8.0% vs. 5.9%, p=0.04). No other comorbidity was significantly more common in one or the other group. Pre-existing DNR documentation was present in < 2% of both groups, with no significant difference. Characteristics of ICU and non-ICU patients are reported in Table 1.
Table 1.
Characteristics of patients admitted to the ICU vs. elsewhere
| Non-ICU N=933 | ICU N=1,654 | p-Value | |
|---|---|---|---|
| Demographics | |||
| Male | 588 (63.0) | 1,083 (65.5) | 0.21 |
| Age | 42 (24–57) | 42 (24–59) | 0.47 |
| Race/Ethnicity | 0.01 | ||
| White (non-Hispanic) | 832 (90.4) | 1,420 (87.1) | |
| Black (non-Hispanic) | 50 (5.4) | 127 (7.8) | |
| Hispanic | 20 (2.2) | 62 (3.8) | |
| Asian (non-Hispanic) | 10 (1.1) | 9 (0.6) | |
| Other | 8 (0.9) | 13 (0.8) | |
| Insurance | 0.22 | ||
| Medicare | 130 (13.9) | 205 (12.4) | |
| Medicaid | 108 (11.6) | 233 (14.1) | |
| Private | 247 (26.5) | 414 (25.0) | |
| Uninsured/other | 448 (48.0) | 802 (48.5) | |
| Injury Characteristics | |||
| Trauma alert level | <0.001 | ||
| 1 | 109 (12.6) | 484 (30.7) | |
| 2 | 394 (45.4 | 738 (46.9) | |
| 3 | 144 (16.6) | 174 (11.1) | |
| Consult | 220 (25.4) | 179 (11.4) | |
| Mechanism of injury | 0.09 | ||
| Fall | 236 (25.3) | 356 (21.6) | |
| Traffic | 603 (64.7) | 1,128 (68.5) | |
| Other | 93 (10.0) | 163 (9.9) | |
| Injury severity score | 13 (9–17) | 17 (12–27) | <0.001 |
| Spleen injury grade | <0.001 | ||
| 1–2 | 680 (72.9) | 1,015 (61.4) | |
| 3–5 | 242 (25.9) | 626 (37.9) | |
| Head injury | <0.001 | ||
| Head AIS 1–2 | 205 (22.0) | 371 (22.4) | <0.001 |
| Head AIS ≥ 3 | 72 (7.7) | 272 (16.4) | |
| Skeletal injury | 325 (34.8) | 822 (49.7) | <0.001 |
| Additional abdominal injury | 200 (21.4) | 558 (33.7) | <0.001 |
| Thoracic injury | 455 (48.8) | 1,026 (62.0) | <0.001 |
| Transfer patient | 304 (32.6) | 388 (23.5) | |
| Physiological indicators | |||
| GCS on admission | 15 (15–15) | 15 (13–15) | <0.001 |
| SBP on admission | 131 (117–146) | 125 (108–142) | <0.001 |
| Transfused in ED | 48 (5.1) | 226 (13.7) | <0.001 |
| Received >2L of fluid pre-hospital | 150 (16.1) | 393 (23.8) | <0.001 |
Continuous variables presented as mean (IQR). Categorical variables presented as N(%)
Among non-ICU patients, 16.1% (150) underwent angiography, compared to 10.9% (181) of ICU patients, p < 0.001. Of non-ICU patients, 8.9% (83) underwent splenectomy, compared to 25.5% (421) of ICU patients (p < 0.001). Overall, more ICU patients underwent laparotomy (27.1% vs. 10.9% of non-ICU patients, p < 0.001). Of non-ICU patients who did require a laparotomy, 45.1% were performed more than 2 hours after arrival, compared to 30.4% for ICU patients, p = 0.004.
In multivariable logistic regression, significant predictors of ICU admission included ISS (OR 1.02, 95% CI 1.01–1.05), receiving >2L of fluid pre-hospital (OR 1.3, 95% CI 1.1–1.6), grade ≥ 3 splenic injury (OR 1.4, 95% CI 1.1–1.8), presence of additional abdominal injuries (OR 1.3, 95% CI 1.1–1.5) and higher level of trauma alert. Presence of severe head injury and skeletal injury were also associated with higher odds of ICU admission, as was a history of alcohol abuse. Traffic-related mechanism was associated with reduced odds of ICU admission compared to falls. Hispanic patients were few in the data set, but had an increased odds of ICU admission. Model fit was good (AUC = 0.7211). Significant predictors (p<0.05) are presented in Table 2. There was no significant association between ICU admission and insurance status, thoracic injury, receiving blood in the ED, admission SBP or GCS, or transfer status. Additional comorbidities included in the model had no significant association with ICU admission (cancer, smoking status, stroke, peripheral vascular disease, attention deficit disorder, and functional dependence). Adding a center fixed effects to the model yielded significantly improved model fit, with AUC = 0.7956 and p-value for the likelihood ratio test <0.001.
Table 2.
Multivariable Logistic Regression Results: Factors Predicting ICU admission After Blunt Splenic Injury
| Odds ratio | 95% CI | p-Value | |
|---|---|---|---|
| Race/Ethnicity | |||
| White (non-Hispanic) | Ref | ||
| Black (non-Hispanic) | 1.2 | 0.6–2.3 | 0.636 |
| Hispanic | 1.9 | 1.1–3.3 | 0.027 |
| Asian (non-Hispanic) | 0.4 | 0.2–1.2 | 0.104 |
| Other | 1.0 | 0.6–1.7 | 0.996 |
| Trauma alert level | |||
| 1 | Ref | ||
| 2 | 0.5 | 0.3–0.8 | 0.001 |
| 3 | 0.4 | 0.1–1.0 | 0.049 |
| Consult | 0.3 | 0.1–0.5 | <0.001 |
| Mechanism of injury | |||
| Fall | Ref | ||
| Traffic | 0.7 | 0.5–1.0 | 0.044 |
| Other | 1.1 | 0.7–1.5 | 0.787 |
| Injury severity score | 1.02 | 1.01–1.05 | 0.011 |
| Spleen injury grade | |||
| Grade 1–2 or undetermined | Ref | ||
| Grade ≥ 3 | 1.4 | 1.1–1.8 | 0.016 |
| Head injury | |||
| AIS 0 | Ref | ||
| AIS 1–2 | 1.0 | 0.8–1.2 | 0.690 |
| AIS ≥ 3 | 1.5 | 1.0–2.1 | 0.039 |
| Skeletal injury | 1.2 | 1.0–1.4 | 0.014 |
| Additional abdominal injury | 1.3 | 1.1–1.5 | 0.002 |
| Alcohol abuse | 1.3 | 1.0–1.7 | 0.036 |
| Received >2L of fluid pre-hospital | 1.3 | 1.1–1.6 | 0.011 |
Crude mortality was 4.2% overall; 5.0% for ICU patients and 2.8% for non-ICU patients. The final regression model with mortality as the outcome incorporated ICU admission, patient characteristics, injury characteristics, admission physiology, and comorbidities; with excellent model fit (AUC 0.9490). ICU admission was not associated with any significant difference in mortality. Risk-adjusted mortality rates were 4.4% (95% CI 3.2%–5.5%) for non-ICU patients and 4.4% (95%CI 3.6%–5.2%) for ICU patients (p-value for the difference = 0.955). Significant predictors of mortality included age (OR 1.1, 95% CI 1.0–1.1), traffic injury (OR 2.4, 95% CI 1.1–5.5), presence of additional abdominal injuries (OR 2.4, 95% CI 1.6–3.6), ISS (OR 1.1, 95% CI 1.0–1.1), and receiving blood transfusion in the ED (OR 3.0, 95% CI 1.4–6.4). Cirrhosis was associated with substantially increased odds of death (OR 8.7, 95% CI 2.8–27.6), and history of stroke was associated with a 3.1-times increased odds of death (95% CI 1.0–9.8). Higher admission GCS and SBP were associated with lower odds of mortality. The remainder of the variables that met criteria (p < 0.2 in bivariate analysis) and were included in the final model had no independent, significant association with mortality. These were: insurance status; trauma alert level; injury grade; head, skeletal, or thoracic injury; and fluid received in the ED. Additional comorbidities that were included but showed no significant independent association with mortality were cardiac disease, hypertension, ulcer disease, anticoagulation, psychiatric illness, arthritis, drug abuse, smoking status, and pre-existing DNR documentation. Significant predictors identified in multivariable logistic regression analysis are presented in Table 3. Addition of the center fixed effects did not add to model fit (p-value for the likelihood ratio test = 0.4241). For all predictors included in the mortality model, the mean variance inflation factor was 1.15. The variance inflation factor for our variable indicating ICU admission was 1.14, indicating that very little of the standard error associated with this variable is due to multicollinearity. In a limited model including only injury severity, patient age, and admission physiology as covariates, ICU admission had no significant association with mortality (OR 0.9, 95% confidence interval 0.5–1.4). In a model including only the propensity score for ICU admission as a covariate, we again found no significant association between ICU admission and mortality (OR 0.7, 95% CI 0.4–1.2).
Table 3.
Multivariable Logistic Regression Results: Factors Predicting Mortality After Blunt Splenic Injury
| Odds ratio | 95% CI | p-Value | |
|---|---|---|---|
| ICU admission | 1.0 | 0.6–1.7 | 0.955 |
| Age (per each additional year) | 1.1 | 1.0–1.1 | <0.001 |
| Mechanism of injury | |||
| Fall | Ref | ||
| Traffic | 2.4 | 1.1–5.5 | 0.029 |
| Other | 0.8 | 0.2–4.5 | 0.842 |
| Additional abdominal injury | 2.4 | 1.6–3.6 | <0.001 |
| Transfused blood in ED | 3.0 | 1.4–6.4 | 0.004 |
| GCS on admission | 0.8 | 0.7–0.8 | <0.001 |
| Admission SBP (per 10 mmHg) | 0.9 | 0.8–1.0 | 0.017 |
| Injury severity score | 1.1 | 1.0–1.1 | <0.001 |
| Cirrhosis | 8.7 | 2.8–27.2 | <0.001 |
| History of stroke | 3.1 | 1.0–9.8 | 0.048 |
In a subgroup analysis limited to the 1,213 patients whose abbreviated injury score was <3 in all body regions other than the abdomen, 671 (55.3%) were admitted to the ICU. In this group, ICU admission was not significantly associated with mortality (odds ratio 0.8, 95% confidence interval 0.2–2.6). Among the 2,243 patients who did not have a moderate-to-severe head injury (AIS < 3 for body region 1), 1,382 (61.6%) were admitted to the ICU. There was no significant association between ICU admission and mortality (OR 1.1, 95% CI 0.5–2.4).
3.2 Center-level analysis
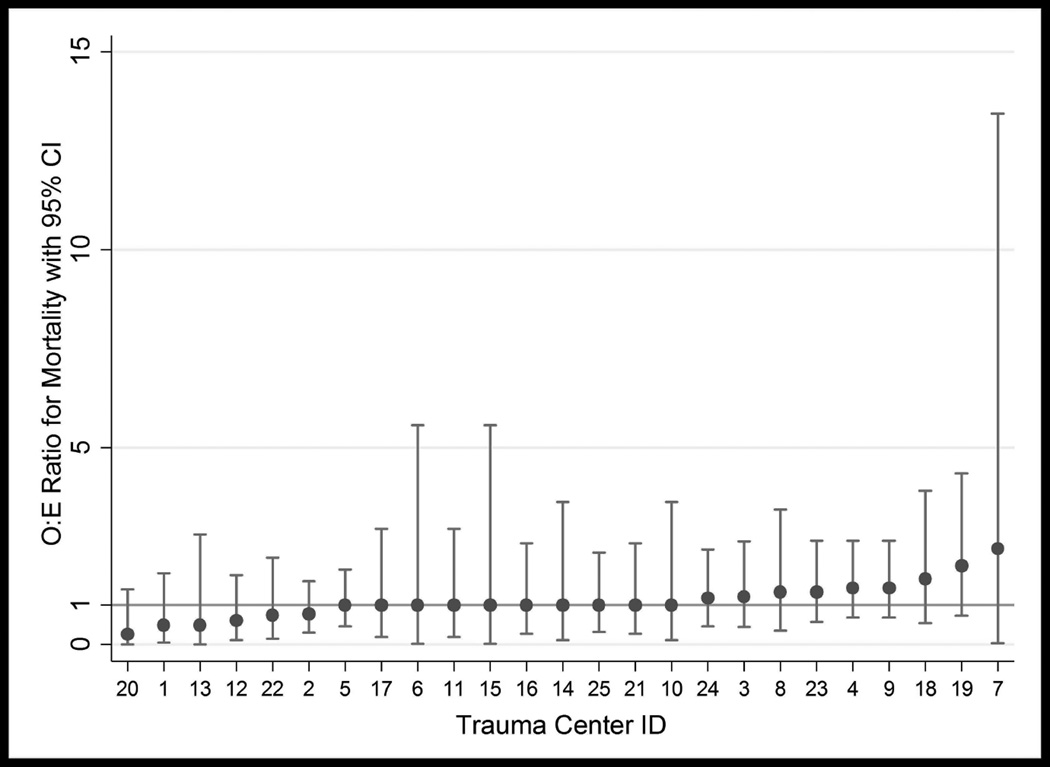
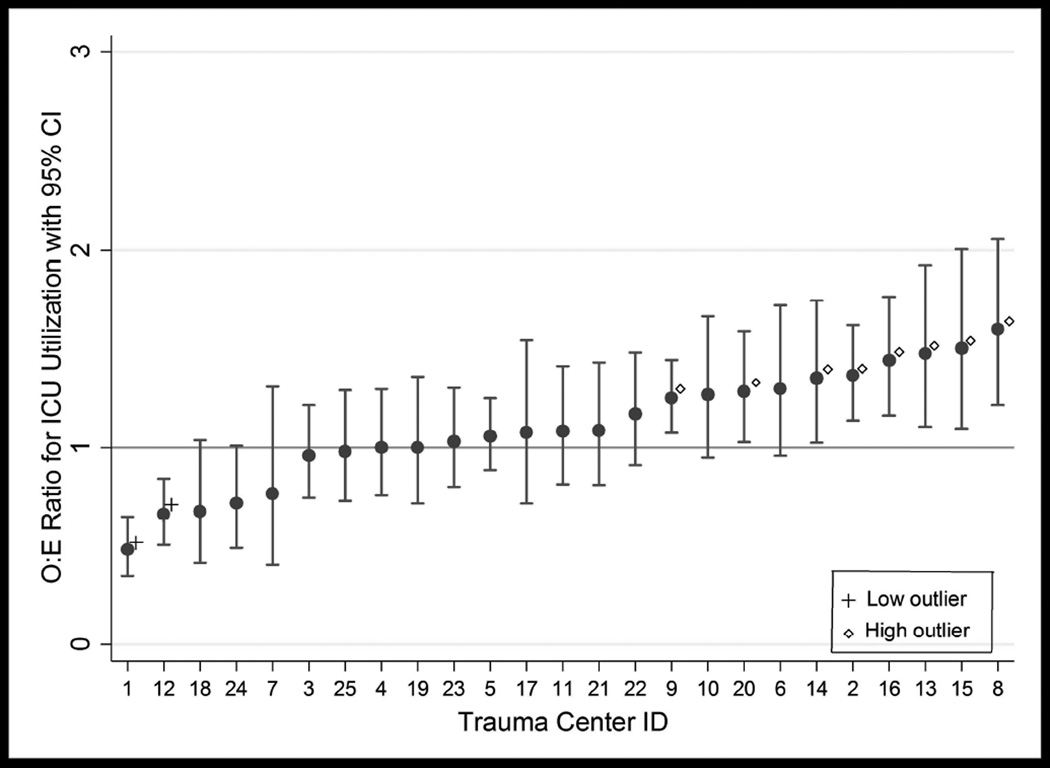
A total of 25 centers with at least 20 eligible patients were included in the center-level analysis, for a patient total of 2,521. Risk-adjusted ICU utilization rates derived from the multivariable logistic regression models ranged from 17.9% to 87.3% among centers, with a median rate of 68.3%. Risk-adjusted mortality rates ranged from 1.2% to 9.6% with a median mortality rate of 4.4%. No centers were classified as outliers for mortality (see Figure 3). For ICU utilization, there were 2 low outliers and 8 high outliers (See Figure 4). There was no correlation between O:E ratios for ICU utilization and mortality (rs = −0.2595, p=0.2103), or between ICU utilization rank and mortality rank (rs = −0.2192, p=0.2924).
Figure 3.
Pennsylvania Trauma Centers Observed-to-Expected Ratios for Mortality after Blunt Splenic Injury
Figure 4.
Pennsylvania Trauma Centers Observed-to-Expected Ratios for ICU Utilization after Blunt Splenic Injury
As shown in Table 4, the majority of patients (72.9%) did not receive any documented critical care procedure. This ranged from 63.6% in the lowest ICU utilization quartile to 77.7% in the highest quartile, and this difference was statistically significant with p<0.001. Likewise, the mean number of procedures performed per patient was significantly lower in the highest compared to the lowest quartile (0.4 vs. 0.7, p<0.001). For each procedure identified, centers in the highest quartile of risk-adjusted ICU utilization had lower rate of procedure utilization in their ICU patients than centers in the lowest quartile. This difference was statistically significant for mechanical and tracheostomy, but not for central line or arterial line placement.
Table 4.
Critical Care Procedure Utilization, by Quartile of Risk-Adjusted ICU Utilization
| ICU patients undergoing procedure; N (%) | ||||||
|---|---|---|---|---|---|---|
| Procedure | Total N=1,609 |
Quartile 1 (Lowest ICU utilization) N=297 |
Quartile 2 N=385 |
Quartile 3 N=492 |
Quartile 4 (Highest ICU utilization) N=435 |
p-Value** |
| Mechanical ventilation* | 215 (13.4) | 59 (19.9) | 49 (12.7) | 71 (14.4) | 36 (8.3) | <0.001 |
| Central line placement* | 189 (11.8) | 38 (12.8) | 55 (14.3) | 51 (10.4) | 45 (10.4) | 0.305 |
| Arterial line placement or monitoring* |
173 (10.8) | 45 (15.2) | 31 (8.1) | 47 (9.6) | 50 (11.5) | 0.148 |
| Tracheostomy | 141 (8.8) | 41 (13.8) | 44 (11.4) | 32 (6.5) | 24 (5.5) | <0.001 |
| No critical care procedures identified |
1,172 (72.9) |
189 (63.6) | 280 (72.7) | 365 (74.2) | 338 (77.7) | <0.001 |
| Mean total critical care procedures per patient (sd) |
0.5 (1.0) | 0.7 (1.1) | 0.5 (1.02) | 0.5 (0.9) | 0.4 (0.9) | <0.001 |
limited to procedures occurring in the ICU
p-Values given for the difference Q1 vs. Q4
4. Discussion
In this cohort study of patients treated for splenic injury at 30 Pennsylvania trauma centers over a four-year period, we found substantial variation in ICU utilization amongst centers, but were unable to demonstrate either a relationship between ICU admission and mortality at the patient level, or between ICU utilization rates and mortality rates at the center level. Given that most patients with splenic injury are likely admitted to the ICU for hemodynamic monitoring, serial physical examinations, and serial CBCs, it is not surprising that the majority had no specific critical care procedure performed. However, we found an inverse relationship between ICU utilization and critical care procedure rates at the center level. This provides validation for our risk-adjustment ICU utilization model, and may indicate that high-utilization centers are admitting more patients to the ICU for observation alone. This group may include a subset of patients stable enough to do well with a lower level of care. Centers that admit a higher proportion of patients with splenic injuries to the ICU may be incurring high costs of care without improving patient outcomes.
Measuring variation in ICU utilization and the impact of ICU admission is challenging, as population and severity of illness varies among centers.14 In this study, we identified a focused population of traumatically injured population—patients with blunt splenic injury—and used a rich source of clinical data to attempt to compare similar patients across institutions and in-hospital destinations. To our knowledge, no previous study has compared the outcomes of trauma patients admitted to the ICU to those of patients admitted elsewhere. However, Wunsch et al. studied ICU admission in Medicare patients undergoing major surgery, and found that hospitals with higher rates of ICU admission did not demonstrate any benefit in mortality.15 Likewise, with respect to medical conditions, Admon et al. found that patients admitted to the ICU for pulmonary embolism at high-utilization centers had relatively fewer critical care procedures performed, indicating variation in admission criteria with no impact on mortality.16 Safavi et al. found similar results in heart failure.2 Gershengorn et al. identified wide variation in ICU utilization for patients with diabetic ketoacidosis without any impact on mortality, and found that ICU admission for this condition was associated with an institution’s overall ICU admission rate, and that hospitals with a higher volume of DKA patients used the ICU less frequently in this population.14
Along with injury and illness severity, provider factors such as training and experience, and facility factors such as bed availability and staffing may inform the decision to admit a patient to the ICU. It is likely that centers differ as to which patients are considered to be safe for floor as opposed to ICU monitoring. Physician staffing and availability may also play a role, as an admitting surgeon at a busy center might be more inclined to admit an injured patient to the ICU, where another physician would be available. While the results of this study may not be adequate to change clinical practice in themselves, we believe that they should support clinical equipoise with respect to the role of ICU admission in blunt splenic injury. Further investigation is needed to understand the resources and processes in place at high- and low-ICU-utilization centers, and to optimize practice at all centers. Success rates greater than 67% have been reported for non-operative management even in patients with grade 3–5 injuries.5,17 Patients who are candidates for non-operative management are typically observed with serial physical examinations and hemoglobin levels, but evidence underlying the details of this process are limited.11 Individual centers have instituted clinical pathways or guidelines, using patient and injury characteristics to determine admission location and frequency of labs and imaging, with improvement in length of stay and no worsening of mortality.4,18 Previous investigations have found benefit to instituting practice guidelines for splenic injury,4,19,20 but have also shown that these guidelines are not widely known or used.21
Protocols for active observation of patients with splenic injuries could involve observing patients in the Emergency Department for a specified time period or according to specified physiologic criteria before determining an admission destination. Likewise, protocols for close observation on the ward or in the step down unit could make ICU admission unnecessary for some patients. We noted several independent predictors of ICU admission and mortality. Mechanism of injury, trauma alert level, ISS, and presence of additional abdominal injuries were significant predictors of both, while age, admitting physiology, blood transfusion in the ED, and history of cirrhosis or stroke predicted mortality. These factors could be incorporated into future guidelines to ensure that patients at highest risk of death are observed most closely. However, race and ethnicity, pre-hospital fluid resuscitation, and history of alcohol abuse predicted ICU admission but not mortality in this data set, and may be less valuable indicators of need for a higher level of care. Injury grade and additional injuries did not independently predict mortality, but are incorporated into ISS, which was an independent predictor.
We acknowledge several limitations to this work. While the registry we used included rich and detailed clinical data, there is no substitute for the judgment of the clinician at the bedside. Therefore, our risk adjustment may not have been complete, biasing our results toward the null. We did find that higher levels of trauma alert independently predicted ICU admission, perhaps reflecting the insight of the initial evaluating clinicians (either EMS or ED personnel) who set the alert level. Alert level was not an independent predictor of mortality, however. Injury severity score was substantially higher in ICU than in non-ICU patients. Future analyses using a larger set of patients could allow for additional analytic approaches, such as a matched analysis or instrumental variable analysis to compensate for this disparity more effectively. However, we believe that the multivariable logistic regression results we report do adequately adjust for differences in risk. Model fit was excellent for both groups (ACU= 0.9917 for non-ICU patients and 0.9368 for ICU patients in stratified analysis), providing reassurance that our model is adequate. We identified patients with splenic injuries, but many patients had other abdominal injuries, or injuries to other body regions, as well, and we could not clearly identify the motivation for ICU admission. While we controlled for the presence of additional injuries in our analysis, we cannot make recommendations for the management of a patient who would have been admitted to the ICU even without a splenic injury. Limiting this investigation to only those with isolated splenic injuries would have lowered our sample size considerably, and would also have failed to provide a true representation of the population of interest, as patients with splenic injuries indeed often have multiple additional injuries.
We tabulated critical care procedures to estimate the need for ICU care in this population, but this representation may not be accurate in a population whose primary need is for close monitoring. It is also possible that close observation obviated the need for these procedures in some marginal patients admitted to the ICU at high utilization centers.
We included patients who underwent initial operative management of their injuries, and designated them as ICU vs. non-ICU patients based on their post-operative location. We chose to include these patients out of concern that different selection criteria for operation at the different centers would bias our population if we excluded all operative patients. On the other hand, it is possible that including these patients biased our results if a patient who would have gone to the ICU at one center went to the OR and subsequently to the regular hospital floor at another center. In surviving patients not undergoing operation, diagnoses were based on imaging, and may be less accurate than operative or autopsy diagnoses. Likewise, although the trauma registry is maintained by dedicated, trained registrars at each center, we cannot rule out differences in coding practice that might be related to ICU utilization.
5. Conclusions
As non-operative management of splenic injuries has become more common, variation in practice within this sphere is increasingly evident and relevant. In this cohort study, risk-adjusted ICU utilization rates for splenic trauma varied widely among trauma centers, with no clear relationship to mortality. Standardizing ICU admission criteria could improve resource utilization without increasing mortality, and prospective study is needed to delineate which patients truly require intensive monitoring in an ICU setting.
Acknowledgments
No specific funding support was obtained for this research, but Dr. Kaufman was supported by a training grant from the National Heart, Lung and Blood Institute, T32 HL-98054-6.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Author Contributions: E.J. Kaufman and D.N. Holena are responsible for the study concept, design, and analysis. E.J. Kaufman drafted the manuscript. All authors contributed to critical review of the manuscript.
Conflicts of Interest: The authors report no proprietary or commercial interest in any product mentioned or concept discussed in this manuscript.
Contributor Information
Douglas J. Wiebe, Email: dwiebe@exchange.upenn.edu.
Niels D. Martin, Email: niels.martin@uphs.upenn.edu.
Jose L. Pascual, Email: jose.pascual@uphs.upenn.edu.
Patrick M. Reilly, Email: reillyp@uphs.upenn.edu.
Daniel N. Holena, Email: daniel.holena@uphs.upenn.edu.
References
- 1.Valley TS, Sjoding MW, Ryan AM, Iwashyna TJ, Cooke CR. Association of intensive care unit admission with mortality among older patients with pneumonia. JAMA. 2015;314(12):1272–1279. doi: 10.1001/jama.2015.11068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Safavi KC, Dharmarajan K, Kim N, et al. Variation exists in rates of admission to intensive care units for heart failure patients across hospitals in the United States. Circulation. 2013;127(8):923–929. doi: 10.1161/CIRCULATIONAHA.112.001088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Harbrecht BG, Zenati MS, Ochoa JB, Puyana JC, Alarcon LH, Peitzman AB. Evaluation of a 15-year experience with splenic injuries in a state trauma system. Surgery. 2006;141(2):229–238. doi: 10.1016/j.surg.2006.06.032. [DOI] [PubMed] [Google Scholar]
- 4.Izu BS, Ryan M, Markert RJ, Ekeh AP, McCarthy MC. Impact of splenic injury guidelines on hospital stay and charges in patients with isolated splenic injury. Surgery. 2009;146(4):787. doi: 10.1016/j.surg.2009.06.021. [DOI] [PubMed] [Google Scholar]
- 5.Shapiro MJ, Krausz C, Durham RM, Mazuski JE. Overuse of splenic scoring and computed tomographic scans. J Trauma. 1999;47(4):651–658. doi: 10.1097/00005373-199910000-00008. [DOI] [PubMed] [Google Scholar]
- 6.Zarzaur BL, Kozar R, Myers JG, et al. The splenic injury outcomes trial: An American Association for the Surgery of Trauma multi-institutional study. Journal of Trauma and Acute Care Surgery. 2015;79(3):335. doi: 10.1097/TA.0000000000000782. [DOI] [PubMed] [Google Scholar]
- 7.Pachter HL, Guth AA, Hofstetter SR, Spencer FC. Changing patterns in the management of splenic trauma. Annals of Surgery. 1998;227(5):708. doi: 10.1097/00000658-199805000-00011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Longo WE, Baker CC, McMillen MA, Modlin IM, Degutis LC, Zucker KA. Nonoperative management of adult blunt splenic trauma. Criteria for successful outcome. Annals of Surgery. 1989;210(5):626–629. doi: 10.1097/00000658-198911000-00010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Zarzaur BL, Kozar RA, Fabian TC, Coimbra R. A survey of American Association for the Surgery of Trauma member practices in the management of blunt splenic injury. J Trauma. 2011;70(5):1026–1031. doi: 10.1097/TA.0b013e318217080c. [DOI] [PubMed] [Google Scholar]
- 10.Wisner DH, Kuppermann N, Cooper A, et al. Management of children with solid organ injuries after blunt torso trauma. Journal of Trauma and Acute Care Surgery. 2015;79(2):206. doi: 10.1097/TA.0000000000000731. [DOI] [PubMed] [Google Scholar]
- 11.Stassen NA, Bhullar I, Cheng JD, et al. Selective nonoperative management of blunt splenic injury. Journal of Trauma and Acute Care Surgery. 2012;73:S294. doi: 10.1097/TA.0b013e3182702afc. [DOI] [PubMed] [Google Scholar]
- 12.Pennsylvania Trauma Systems Foundation. 2014 PTOS Patient Inclusion Criteria. [Accessed 1/19/2016]; http://www.ptsf.org/ [Google Scholar]
- 13.Moore EE, Cogbill TH, Malangoni MA, Jurkovich GJ, Champion HR. Scaling system for organ specific injuries. [Accessed 1/18/2016];American Association for the Surgery of Trauma. www.aast.org/asset.axd?id=56ef079d-229c-45f2-9b18-c3825e450e65&t=633867256925730000. [Google Scholar]
- 14.Gershengorn HB, Iwashyna TJ, Cooke CR, Scales DC, Kahn JM, Wunsch H. Variation in use of intensive care for adults with diabetic ketoacidosis. Crit Care Med. 2012;40(7):2009. doi: 10.1097/CCM.0b013e31824e9eae. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Wunsch H, Gershengorn HB, Cooke CR, et al. Use of intensive care services for Medicare beneficiaries undergoing major surgical procedures. Anesthesiology. 2016 doi: 10.1097/ALN.0000000000001024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Admon AJ, Seymour CW, Gershengorn HB, Wunsch H, Cooke CR. Hospital-level variation in ICU admission and critical care procedures for patients hospitalized for pulmonary embolism. Chest. 2014;146(6):1452–1461. doi: 10.1378/chest.14-0059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Goan YG, Huang MS, Lin JM. Nonoperative management for extensive hepatic and splenic injuries with significant hemoperitoneum in adults. J Trauma Injury Infect Crit Care. 1998;45(2):360. doi: 10.1097/00005373-199808000-00026. [DOI] [PubMed] [Google Scholar]
- 18.Dervan LA, King MA, Cuschieri J, Rivara FP, Weiss NS. Pediatric solid organ injury operative interventions and outcomes at Harborview Medical Center, before and after introduction of a solid organ injury pathway for pediatrics. Journal of Trauma and Acute Care Surgery. 2015;79(2):215. doi: 10.1097/TA.0000000000000726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Gutierrez IM, Zurakowski D, Chen Q, Mooney DP. Clinical practice guidelines (CPGs) reduce costs in the management of isolated splenic injuries at pediatric trauma centers. Langenbecks Arch Surg. 2012;398(2):313–315. doi: 10.1007/s00423-012-1003-z. [DOI] [PubMed] [Google Scholar]
- 20.Notrica DM, Eubanks JW, Tuggle DW, et al. Nonoperative management of blunt liver and spleen injury in children. Journal of Trauma and Acute Care Surgery. 2015;79(4):683. doi: 10.1097/TA.0000000000000808. [DOI] [PubMed] [Google Scholar]
- 21.Bowman SA, Bulger E, Sharar SR, Maham SA, Smith SD. Variability in pediatric splenic injury care: results of a national survey of general surgeons. Archives Of Surgery. 2010;145(11):1048–1053. doi: 10.1001/archsurg.2010.228. [DOI] [PubMed] [Google Scholar]