Abstract
Hydrogen has antioxidant and anti-inflammatory effects on lung ischemia–reperfusion injury when it is inhaled by donor or/and recipient. This study examined the effects of lung inflation with 3% hydrogen during the cold ischemia phase on lung graft function in rats. The donor lung was inflated with 3% hydrogen, 40% oxygen, and 57% nitrogen at 5 mL/kg, and the gas was replaced every 20 min during the cold ischemia phase for 2 h. In the control group, the donor lung was inflated with 40% oxygen and 60% nitrogen at 5 mL/kg. The recipient was euthanized 2 h after orthotropic lung transplantation. The hydrogen concentration in the donor lung during the cold ischemia phase was 1.99–3%. The oxygenation indices in the arterial blood and pulmonary vein blood were improved in the hydrogen group. The inflammation response indices, including lung W/D ratio, the myeloperoxidase activity in the grafts, and the levels of IL-8 and TNF-α in serum, were significantly lower in the hydrogen group (5.2 ± 0.8, 0.76 ± 0.32 U/g, 340 ± 84 pg/mL, and 405 ± 115 pg/mL, respectively) than those in the control group (6.5 ± 0.7, 1.1 ± 0.5 U/g, 443 ± 94 pg/mL, and 657 ± 96 pg/mL, respectively (P < 0.05), and the oxidative stress indices, including the superoxide dismutase activity and the level of malonaldehyde in lung grafts were improved after hydrogen application. Furthermore, the lung injury score determined by histopathology, the cell apoptotic index, and the caspase-3 protein expression in lung grafts were decreased after hydrogen treatment, and the static pressure–volume curve of lung graft was improved by hydrogen inflation. In conclusion, lung inflation with 3% hydrogen during the cold ischemia phase alleviated lung graft injury and improved graft function.
Keywords: Hydrogen, donor, ischemia reperfusion injury, lung transplantation
Introduction
Primary graft dysfunction induced by ischemia–reperfusion injury (IRI) remains a major factor in the early mortality of lung transplantation.1,2 Preventing IRI in the lung graft has been the primary goal in recent years.
Lung preservation solution, such as low-potassium dextran (LPD) solution, is the most commonly used lung protective method during the cold ischemia phase.3,4 Different perfusion pressures and perfusion methods for the donor could also improve lung graft function,5 as could by using anti-platelet/endothelial cell adhesion molecule-1,6 relaxin,7 and steroids8 during IRI. Additionally, hypothermia (4–10℃) is an important method for donor lung perseveration during the ischemia phase,9,10 but the donor lung still could not be stored for a longer time.11 Thus, the preservation methods for donor lungs still need improvement.
The oxidative stress during IRI plays a pivotal role in lung injury.12 Inhibition or alleviation of oxidative stress during the cold storage phase was beneficial for IRI in lung grafts.9 Hydrogen, a novel antioxidant agent, has been shown to selectively reduce toxic reactive oxygen species (ROS) and to alleviate organ IRI in the kidneys, heart, brain, liver, small intestines, and lungs.13–17 Hydrogen also had protective effects against acute lung injury induced by IRI,18 ventilator use,19 hyperoxia,20 irradiation,21 or lipopolysaccharide (LPS)22 through antioxidant, anti-inflammatory, and anti-apoptotic activities. Thus, we hypothesized that hydrogen might have protective effects on lung graft injury if applied during the cold ischemia phase.
Materials and methods
All of the procedures in this study were approved by the Institutional Animal Care and Use Committee of Harbin Medical University.
Study design
Adult male Sprague-Dawley rats (240–300 g) were obtained from Vital River Laboratories (Beijing, China). The animals were housed in individual cages in a temperature-controlled room with a 12-h light/dark cycle and free access to food and water.
The animals were divided randomly into the sham group, the control group, and the hydrogen group. The left donor lung was inflated with 40% oxygen and 60% nitrogen or 3% hydrogen, 40% oxygen, and 57% nitrogen at 5 mL/kg (LiMing Gas Corporation, Harbin, China) in the control group and the hydrogen group during the cold ischemia phase. The trachea was sealed with a three-way stopcock, and the gas was replaced every 20 min. In the sham group, the rats were performed a thoracotomy, and were mechanical ventilated for 2.5 h.
Hydrogen concentration measurement
The metabolism of hydrogen in the donor lung was measured in the preliminary experiment (n = 10). The left donor lung was inflated with 3% hydrogen, 40% oxygen, and 57% nitrogen at 5 mL/kg using a 5 mL air-tight syringe through a three-way stopcock connected to the trachea, and was preserved at 4℃ for 20 min. Then, the gas was withdrawn from the lung alveoli with a 1 mL air-tight syringe, and the hydrogen concentration was measured using gas chromatography (GC-17A, Shimadzu Corporation, Kyoto, Japan).
Lung transplantation procedures
The donor rats were anesthetized using 60 mg/kg sodium pentobarbital intraperitoneally and were intubated through a tracheostomy. Then, the animals were ventilated with 40% oxygen and 60% nitrogen at a rate of 40–60 breaths/min, which was adjusted to maintain PaCO2 within 35–45 mmHg using a tidal volume of 10 mL/kg, and a positive end-expiratory pressure (PEEP) of 2 cm H2O for 10 min (Harvard Apparatus, South Natick, MA). Five minutes after heparin (1000 U/kg) administered via the tail vein, the donor was euthanised by exsanguinations. Then the rats were performed a thoracotomy, and the donor lungs were perfused with 20 mL of 4℃ LPD solution (prepared by Harbin Medical University according to a previous study23) with a pressure of 20 cm H2O. Subsequently, the donor lungs were inflated as the protocol and were stored in the LPD solution at 4℃ for 2 h. After a left thoracotomy through the fourth intercostal space, the orthotopic left lung transplantation was performed using a cuff technique.24,25 The anesthesia, operation, and ventilation settings for the rats from the recipients and the sham group were the same as those for the donor rats, and anesthesia was maintained using sodium pentobarbital with intermittent administration and pipecuronium bromide (0.4 mg·kg−1·h−1) intraperitoneally. Normal saline was infused at 10 mL·kg−1·h−1 through the tail veins in the donor and recipient rats. The recipient rats were sacrificed by exsanguination 2 h after lung transplantation. The mean arterial pressure (MAP) and body temperature were continuously recorded (Datex AS/3, Datex, Finland).
Blood gas analysis
The blood was drawn through the left femoral artery, and the arterial blood gas was measured 5 min before transplantation (T0), 3 min after reperfusion, and every 30 min thereafter (T1–T5) (Rapid Lab 348, Bayer, Medfield, MA). At the end of the experiment, the pulmonary vein blood was collected from the left pulmonary veins of the recipients for blood gas analysis. In the sham group, the time point for blood gas analysis was consistent with the control group.
Measurement of inflammatory cytokines and oxidation–reduction indicators
The upper lobe of the lung graft, which was desiccated at 80℃ for 72 h, was used to measure the wet weight (W)/dry weight (D) ratio. The inferior lobe, which was stored at −80℃, was homogenized to measure the myeloperoxidase (MPO) activity using a special regent-box (Jiancheng Bio-Technology, Nanjing, China) and a spectrophotometer (UNICO UV-2100, Shanghai, China). One unit of MPO was defined as the quantity that degraded 1.0 mole of peroxide per minute at 37℃. The results were expressed as units per gram of lung tissue (U/g). The serum levels of interleukin (IL)-8 and tumor necrosis factor (TNF)-α were measured using an enzyme-linked immunosorbent assay (ELISA) kit (R&D Systems, Minneapolis, MN). The malonaldehyde (MDA) activity, which was expressed as nanomoles per milligram of protein (nmol/mg protein), and superoxide dismutase (SOD) activity, which was expressed as units per milligram of protein (U/mg protein), in the supernatants of the graft homogenates were determined using corresponding kits (Jiancheng Bio-Technology), respectively.
Histopathologic analysis
The lung tissues were fixed in 4% paraformaldehyde, embedded in paraffin, sectioned (4 µm thick), and stained with hematoxylin and eosin. A pathologist who was blinded to the study evaluated all of the sections. The evaluation was based on (1) neutrophil infiltration, (2) airway epithelial cell damage, (3) interstitial edema, (4) hyaline membrane formation, and (5) hemorrhage. Each section had fives scores that corresponded to the five criteria based on severity (normal = 0, minimal change = 1, mild change = 2, moderate change = 3, and severe change = 4). The lung injury score (LIS) for each criterion was recorded.26
Immunohistochemistry for the lung sections
Alveolar epithelial cells apoptosis was examined using terminal deoxynucleotidyl transferase dUTP nick end-labeling (TUNEL) assays (Zhongshan Golden Bridge Biotechnology, Beijing, China). The sections were stained with the brown 3,3’-diaminobenzidine (DAB) color-developing agent. Cell apoptosis was considered to be present when the cells had brown-yellow-stained nuclei. Positive cells were counted in five random high-power (×40) fields from every specimen to calculate the apoptotic index (AI), which was the number of positive cells per 100 cells in five different fields in the same section.27 Caspase-3 protein expression in the alveolar epithelial cell of the lung graft was measured by immunohistochemical staining (Cell Signaling Technology Inc, Boston, MA). Positive cells per section were counted in five random high-power (×40) fields from every specimen and were evaluated using an immunohistochemical score (IHS), which was calculated by multiplying the quantity score (an estimation of the percentage of immunoreactive cells) and the staining intensity score (an estimation of the staining intensity). The quantity score was illustrated as follows: no staining was scored as 0, 1–10% cell staining as a 1, 11–50% as a 2, 51–80% as a 3, and 81–100% as a 4. The staining intensity was rated on a scale of 0 to 3, with 0 = negative, 1 = weak, 2 = moderate, and 3 = strong. A pathologist examined all of the sections using a single-blind method.28
Measurement of the static pressure–volume curves of the lungs
A mid-thoracotomy was performed immediately after sacrifice, and the animals were connected to an apparatus to measure the static pressure–volume (P–V) curves of the lungs. The lung volumes were measured by raising the airway pressure to 30 cm H2O and then decreased to 0 cm H2O in a stepwise manner with 1 min of equilibration at each 5 cm H2O interval. The volume measurements were corrected for gas compression in the apparatus.29
Statistical analysis
The data were expressed as the mean values ± standard deviations or median (interquartile range) according to distribution. Differences in measured variables between groups were determined by one-way analysis of variance (ANOVA) followed by Student–Newman–Keuls (SNK) test or Kruskal–Wallis and Nemenyi tests. Repeated measurement data were analyzed by repeated-measures ANOVA. The data were statistically significant when P < 0.05.
Results
Transplantation-related data
The hydrogen concentration in the donor lung was 1.99 ± 0.26% at 20 min after inflation. Rats that had artificial lung damage, pleural hemorrhage, acute heart failure, or transplantation failure were excluded. Eight pairs of rats were included in each group ultimately. The MAP and heart rates were stable in each group, and MAP and heart rates after reperfusion in the control group and hydrogen group were lower than that in the sham group (P < 0.05) (Table 1).
Table 1.
The hydrodynamics parameters in rats from each group (mean ± SD, n = 8).a
| Group | T0 | T1 | T2 | T3 | T4 | T5 | |
|---|---|---|---|---|---|---|---|
| MAP (mm Hg) | Sham | 115 ± 9 | 113 ± 12 | 114 ± 7 | 113 ± 11 | 116 ± 8 | 118 ± 10 |
| Control | 122 ± 11 | 113 ± 9 | 100 ± 12* | 97 ± 13* | 96 ± 12* | 98 ± 8* | |
| Hydrogen | 123 ± 12 | 115 ± 10 | 103 ± 11* | 95 ± 14* | 97 ± 11* | 99 ± 11* | |
| HR (beats/min) | Sham | 363 ± 25 | 356 ± 24 | 346 ± 27 | 352 ± 21 | 348 ± 26 | 345 ± 28 |
| Control | 361 ± 26 | 374 ± 18 | 372 ± 20 | 365 ± 21 | 352 ± 24 | 351 ± 26 | |
| Hydrogen | 365 ± 24 | 371 ± 19 | 368 ± 23 | 363 ± 25 | 351 ± 28 | 354 ± 24 |
T0–T5 represented the following time points: baseline before transplantation, and 3 min, 30 min, 60 min, 90 min, and 120 min after reperfusion.
*P < 0.05 vs. sham group.
Hydrogen improved the oxygenation in recipients
The indices of blood gas analysis, including oxygenation index (partial pressure of arterial oxygen [PaO2]/fraction of inspired oxygen [FiO2]), base excess (BE) value, and pH value, were stable in the sham group. The PaO2/FiO2 in the control group was lower than the sham group (P < 0.05), and the PaO2/FiO2 in the hydrogen group was higher than the control group (P < 0.05). Additionally, the pH and BE values between the groups had similar tendency (Table 2). The pulmonary venous oxygen tension (PvO2)/FiO2 in the control group (372 ± 36 mm Hg) was lower than the sham group (490 ± 29 mm Hg), and the PvO2/FiO2 in the hydrogen group (425 ± 40 mm Hg) was higher than the control group (P < 0.05).
Table 2.
The indexes of arterial blood gas analysis in each group (mean ± SD, n = 8).a
| Group | T0 | T1 | T2 | T3 | T4 | T5 | |
|---|---|---|---|---|---|---|---|
| PaO2/FiO2 (mm Hg) | Sham | 453 ± 24 | 451 ± 18 | 447 ± 24 | 448 ± 24 | 450 ± 21 | 443 ± 24 |
| Control | 435 ± 28 | 342 ± 32* | 344 ± 29* | 345 ± 32* | 339 ± 29* | 325 ± 25* | |
| Hydrogen | 434 ± 30 | 349 ± 24* | 353 ± 31* | 358 ± 28* | 368 ± 27*# | 361 ± 27*# | |
| pH value | Sham | 7.38 ± 0.05 | 7.37 ± 0.07 | 7.38 ± 0.05 | 7.36 ± 0.04 | 7.37 ± 0.05 | 7.36 ± 0.07 |
| Control | 7.36 ± 0.04 | 7.32 ± 0.05 | 7.31 ± 0.07 | 7.26 ± 0.05* | 7.24 ± 0.05* | 7.21 ± 0.05* | |
| Hydrogen | 7.37 ± 0.04 | 7.32 ± 0.04 | 7.35 ± 0.07 | 7.36 ± 0.04# | 7.33 ± 0.06# | 7.31 ± 0.05# | |
| BE value | Sham | 0.04 ± 0.20 | 0.04 ± 0.28 | −0.21 ± 0.20 | −0.40 ± 0.50 | −0.59 ± 0.56 | −0.85 ± 0.64 |
| Control | 0.08 ± 0.02 | −1.79 ± 0.42* | −2.81 ± 0.55* | −3.55 ± 0.81* | −3.63 ± 0.59* | −3.96 ± 0.77* | |
| Hydrogen | 0.05 ± 0.04 | −1.70 ± 0.47* | −2.48 ± 0.47* | −2.69 ± 0.73*# | −3.01 ± 0.74* | −3.07 ± 0.70*# |
The PaO2/FiO2, pH values, and base excess (BE) values were decreased gradually after reperfusion in the control group and the hydrogen group. T0–T5 represents the following time points: baseline before transplantation, and 3 min, 30 min, 60 min, 90 min, and 120 min after reperfusion.
*P < 0.05 vs. sham group; #P < 0.05 vs. control group.
Hydrogen ameliorated the inflammatory response in recipients
The W/D ratio in the control group (6.5 ± 0.7) was higher than the sham group (4.8 ± 0.6) (P < 0.05), and the hydrogen group (5.2 ± 0.8) was lower than the control group, but the difference had no statistical significance. The MPO activity in the grafts had the similar tendency as W/D ratio. The levels of IL-8 in the serum in the control group (443 ± 94 pg/mL) and the hydrogen group (340 ± 84 pg/mL) were significantly higher than the sham group (34 ± 12 pg/mL) (P < 0.05), and the hydrogen group was lower than the control group (P < 0.05). The level of TNF-α in the serum had similar tendency as IL-8 (Table 3).
Table 3.
The indices of inflammatory response and oxidative stress in each group (mean ± SD, n = 8)
| Group | W/D ratio | MPO activity (U/g) | IL-8 (pg/mL) | TNF-α (pg/mL) | SOD (U/mg protein) | MDA (nmol/mg protein) |
|---|---|---|---|---|---|---|
| Sham | 4.8 ± 0.6 | 0.52 ± 0.13 | 34 ± 12 | 65 ± 16 | 71 ± 15 | 3.1 ± 0.8 |
| Control | 6.5 ± 0.7* | 1.12 ± 0.53* | 443 ± 94* | 657 ± 96* | 33 ± 9* | 7.7 ± 2.3* |
| Hydrogen | 5.2 ± 0.8# | 0.76 ± 0.32# | 340 ± 84*# | 405 ± 115*# | 51 ± 10*# | 4.2 ± 1.2# |
*P < 0.05 vs. sham group; #P < 0.05 vs. control group.
Hydrogen alleviated the oxidative stress in lung grafts
The SOD activity in the control group (33 ± 9 U/mg protein) was significantly lower than the sham group (71 ± 15 U/mg protein) (P < 0.05), and the hydrogen group (51 ± 10 U/mg protein) was significantly higher than the control group (P < 0.05). The MDA level in the control group (7.7 ± 2.3 nmol/mg protein) was significantly higher than the sham group (3.1 ± 0.8 nmol/mg protein) (P < 0.05), and the hydrogen group (4.2 ± 1.2 nmol/mg protein) was significantly lower than the control group (P < 0.05) (Table 3).
Hydrogen improved lung graft histology
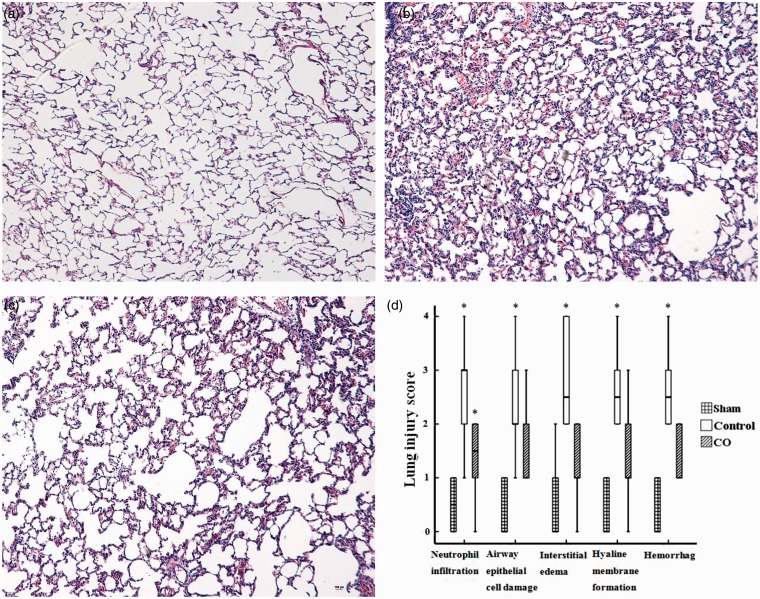
Microscopic findings in the lungs revealed normal lung parenchyma in the sham group (Figure 1a). Lung grafts from the control group (Figure 1b) showed extensive alterations, i.e. moderate to severe edema in the alveolar septum and spaces, hyaline membrane formation, and intra-alveolar hemorrhage occasionally. Much less severe changes were present in grafts from the hydrogen group (Figure 1c). Similarly, the LIS of neutrophil infiltration in the control group (3.0 [1.75–3.25]) was higher than the sham group (0.5 [0–1]) (P < 0.05), the hydrogen group (1.5 [0.75–2.0]) was lower than the control group, but the difference was not statistically significant. Simultaneously, the LIS of other criteria had similar tendency as the LIS of neutrophil infiltration (Figure 1d).
Figure 1.
Histological analysis of lung grafts (n = 6). Paraformaldehyde-fixed sections of lung grafts were stained with eosin and hematoxylin, and all pictures represent 10× original magnifications. (a) Sham group; (b) control group; (c) hydrogen group; (d) lung injury score for each criterion. The lung injury score for each criterion in the control group was higher than the sham group (P < 0.05), and the hydrogen group was lower than the control group, but the difference was not statistically significant. *P < 0.05 versus sham group; #P < 0.05 versus control group. (A color version of this figure is available in the online journal.)
Hydrogen decreased cell apoptosis in grafts
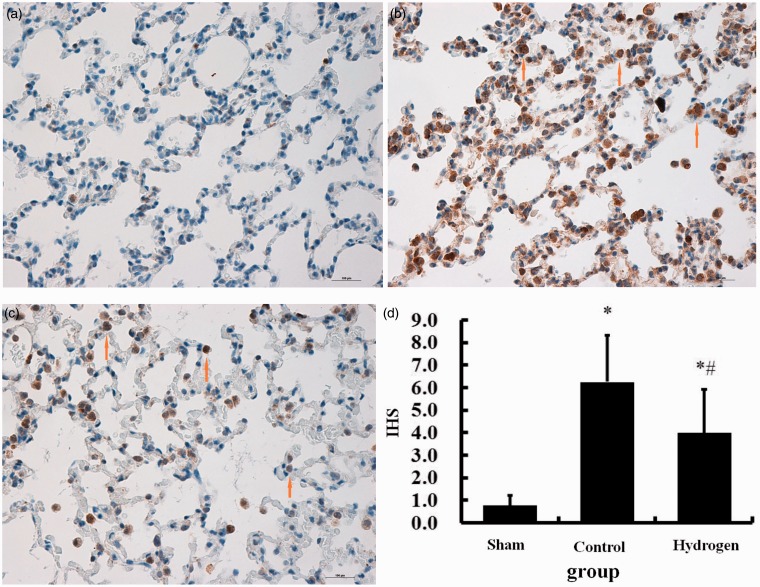
The number of apoptotic cells in the hydrogen group (Figure 2c) was lower than that in the control group (Figure 2b). The AI in the control group (52 ± 11) and hydrogen group (34 ± 9) was higher than the sham group (11 ± 3) (P < 0.05), and the hydrogen group was significantly lower than the control group (P < 0.05) (Figure 2). Similarly, the number of caspase-3 positive cells in the hydrogen group (Figure 3c) was lower than that in the control group (Figure 3b). The IHS of caspase-3 in the control group (6.2 ± 2.0) and hydrogen group (4.0 ± 1.8) was higher than the sham group (1.3 ± 0.4), and the IHS of caspase-3 in the hydrogen group was lower than the control group (Figure 3d).
Figure 2.
Alveolar epithelial cell apoptosis in lung grafts by TUNEL staining (n = 6). (a) Sham group; (b) control group; (c) hydrogen group. Apoptosis of the cell was represented by brown-yellow staining in the nuclei (arrows in b and c). All pictures represent 40× original magnifications. The number of apoptotic cells (AI) in the control group (52 ± 11) and hydrogen group (34 ± 9) was higher than the sham group (11 ± 3) (P < 0.05), and the hydrogen group was significantly lower than the control group (P < 0.05). *P < 0.05 versus sham group; #P < 0.05 versus control group. (A color version of this figure is available in the online journal.)
Figure 3.
Caspase-3 protein expression of alveolar epithelial cell in lung grafts (n = 6). (a) Sham group; (b) control group; (c) hydrogen group. The presence of brown granules in the cytoplasm indicated a positive cell (arrows in b and c). All pictures represent 40× original magnifications. The immunohistochemical score (IHS) in the control group (6.2 ± 2.0) and hydrogen group (4.0 ± 1.8) was higher than the sham group (1.3 ± 0.4) (P < 0.05), and the IHS of caspase-3 in the hydrogen group was lower than the control group (P < 0.05). *P < 0.05 versus sham group; #P < 0.05 versus control group. (A color version of this figure is available in the online journal.)
Hydrogen improved the compliance of lung graft
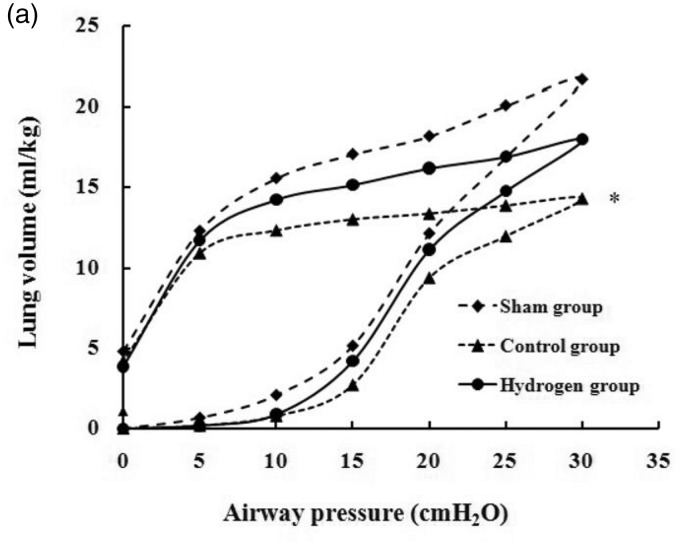
The values of static P–V curve in the control group were significantly lower than the sham group (P < 0.05), and the hydrogen group were higher than the control group, but the difference had no statistical significance (P = 0.071). At a pressure of 30 cm H2O, the volumes in the sham group, control group, and the hydrogen group were 21.7 ± 3.6 mL/kg, 14.2 ± 3.3 mL/kg, and 17.9 ± 3.2 mL/kg, respectively (Figure 4).
Figure 4.
The static P–V curve of the lungs (n = 8). Values were means. SD bars were omitted for clarity. *P < 0.05 versus sham group; #P < 0.05 versus control group
Discussion
Cold ischemia induces oxidative stress and inflammation, resulting in sodium pump inactivation, intracellular calcium overload, iron release, and cell death, ultimately leading to graft dysfunction.9 In this study, hydrogen inflation during the cold ischemia phase ameliorated the inflammatory response, oxidative stress, and alveolar epithelial cell apoptosis in the graft, improved lung graft histology, and lung graft function evaluated by blood gas analysis and static P–V curves.
Currently, the application of hydrogen has focused on inhalation, intraperitoneal injection, or drinking of hydrogen-rich saline.14,19,30 A saturated hydrogen LPD solution might be suitable for donor lung during the ischemia, but the process of the saturated hydrogen LPD solution preparation required special equipment. In addition, hydrogen-rich solution contains a very low dose of hydrogen (<0.6 mmol/L).13 The effective concentration of hydrogen was considered to be 2–4%.13,31 In this study, the hydrogen concentration was 1.99–3%, which was maintained at an effective level. Hydrogen has no risk of explosion at a concentration of less than 4% in the presence of oxygen, and hydrogen has no side effects in this concentration range in animals.13 Furthermore, hydrogen is able to penetrate cell membranes rapidly and reach target organs, and the excess hydrogen can be exhaled through respiration or dissipated by methanogenesis, sulfate reduction, and acetogenesis.32 Therefore, an overdose is impossible. These characteristics make hydrogen suitable for lung inflation. This study obtained an initial data about the hydrogen metabolism in the donor lung, and provided an innovative strategy for donor lung protection in the setting of the cold ischemic, that is, lung inflation with gases which had protective effect on lung injury when inhaled or used in other ways. Currently, hydrogen has already been used in some clinical treatment, such as metabolic syndrome, muscular diseases, and acute brain stem infarction,33–35 and non complication was found. Thus, the method used in this study might be applied in clinical practice directly.
Lung ischemia activates the release of proinflammatory cytokines, such as IL-8 and TNF-α, which activate neutrophils during perfusion.36 Neutrophils and ROS damage endothelial cells, leading to alterations in their permeability and to lung edema. MPO activity in the lungs is one of the most commonly used indices to measure the extent of neutrophil infiltration.37 In this study, hydrogen decreased the serum levels of IL-8 and TNF-α in the recipients and reduced the MPO levels in the lung grafts. These results were consistent with previous studies. Kawamura et al.18 found that hydrogen inhibited the up-regulation of TNF-α and IL-1β mRNAs in lung grafts. Sun et al.20 demonstrated that hydrogen decreased TNF-α in serum in a hyperoxic lung injury model. Other studies have also demonstrated that hydrogen mitigated the up-regulation of the inflammatory mediators (IL-6, TNF-α, and MPO) induced by IRI.30,38 Meanwhile, hydrogen ameliorated pulmonary edema and decreased the severity of lung injury in the grafts, as supported by W/D ratio and histologic analysis. Therefore, hydrogen might exert local protective effects against IRI in lung grafts through an anti-inflammatory mechanism.
Oxidative stress plays a pivotal role in IRI, which disrupts cellular metabolism and induces lipid peroxidation, protein denaturation, and DNA mutation, eventually leading to organ dysfunction.31 Hydrogen, as a selective anti-oxidant molecule, offers protective effects through free radical scavenging. In this study, the MDA level was reduced, and the SOD activity in the graft was activated by hydrogen. The results were similar to previous study, which found that the hydrogen-rich saline reduced the MDA level and increased SOD activity in lungs from a rabbit lung IRI model.38 In our previous study, the hydrogen inhalation in donors and recipients also alleviated the oxidative stress in lung grafts from a brain death donor.39 Therefore, we inferred that hydrogen had antioxidant effect when applied during the cold ischemia and reperfusion phase.
Apoptosis, another major feature of the IRI process, was activated by the caspase cascade and might have a deleterious impact on graft function.40 Fischer et al.41 demonstrated that apoptosis was a major contributor of graft injury. In this study, the AI and the caspase-3 protein expression in the alveolar epithelial cells of graft were inhibited by hydrogen. Another studies also found the similar effects. Shi et al.15 reported that hydrogen alleviated lung epithelial cell apoptosis. Xie et al.22 found that hydrogen decreases caspase-3 activity during LPS-induced lung injury. Therefore, the protective effects of hydrogen in the lung grafts might be due to its anti-apoptotic effects.
Currently, the molecule mechanism of hydrogen in protection for lung injury is still unclear. Hydrogen exerted protective effect in the ventilator-induced lung injury or LPS-induced acute lung injury through the up-regulation of hemeoxygenase-1 or down regulation of the nuclear factor kappa-B/B cell lymphoma 2 (NF-κB/Bcl-2) pathways.18,19,22 Kawamura et al.42 found that hydrogen reduced hyperoxic lung injury via the Nrf2 pathway in vivo. Additionally, Tanaka et al.43 suggested that the protective effect of hydrogen was associated with the up-regulation of pulmonary surfactant-related molecules, ATP synthases, and stress-response molecules in lung allograft. Although the signal transduction pathway has not been detected in this study, hydrogen exerted anti-inflammatory, anti-oxidative, and anti-apoptotic effects on graft when applied during the cold ischemia phase.
This study had some limitations. First, the time for lung inflation and the observed time after reperfusion was only 2 h, respectively. The long-term effects of hydrogen on lung IRI require further study. Second, we measured lung function and histopathologic changes to evaluate the effect of hydrogen, but not tissue and cell metabolism changes. Measuring the glucose levels in the perfusate of the donor lung before or the graft lung after transplantation might be more meaningful.44,45 As the contralateral lung was not clamped, the results of arterial blood gas analysis might not reflect the transplant lung function if the graft had none or low flow. Third, some indices between groups had no statistic difference, including the results of the blood gas analysis and the LIS. There may be two reasons, one was the sample size was small, and the other was the model used in this study was a mild lung graft injury model. Fourth, the controlled reperfusion method46 and steroids8 application after reperfusion might ameliorate the graft function. And the effects of hydrogen on lung graft function from different donors, such as brain death donor, should be further explored. And last, the specific mechanism by which hydrogen confers lung protection was not clarified. Further experiments, such as in vitro cell culture, are required.
In conclusion, donor lung inflation with 3% hydrogen during the cold ischemia phase had protective effects on lung graft injury through anti-inflammatory, anti-oxidative, and anti-apoptotic effects.
Acknowledgments
This study was supported by China Postdoctoral Science Foundation (Grant No. 20100471115 and 201104453), and National Natural Science Foundation of China (Grant No. 30901391).
Authors’ contributions
The first two authors contributed equally to this work. RL conceived and designed the research, acquired the data, and drafted the manuscript; XF conceived and designed the research, acquired the data, analyzed the data, performed statistical analysis, and drafted the manuscript; CM acquired the data, drafted the manuscript, and made critical revision of the manuscript for important intellectual content; JX conceived and designed the research, analyzed the data, and made critical revision of the manuscript for important intellectual content; JL analyzed the data and performed statistical analysis; WY analyzed the data and performed statistical analysis; LW conceived and designed the research, analyzed the data, performed statistical analysis, handled funding and supervision, and made critical revision of the manuscript for important intellectual content; HZ conceived and designed the research, analyzed the data, performed statistical analysis, handled funding and supervision, and made critical revision of the manuscript for important intellectual content.
References
- 1.Corris PA, Christie JD. Update in transplantation 2006. Am J Respir Crit Care Med 2007; 175: 432–5. [DOI] [PubMed] [Google Scholar]
- 2.Christie JD, Kotloff RM, Ahya VN, Tino G, Pochettino A, Gaughan C, DeMissie E, Kimmel SE. The effect of primary graft dysfunction on survival after lung transplantation. Am J Respir Crit Care Med 2005; 171: 1312–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Simoes EA, Pego-Fernandes PM, Cardoso PF, Pazetti R, Werebe E, de Oliveira Braga KA, Menezes A, Nepomuceno N, Soares PR, Correia AT, Jatene FB. Comparing the performance of rat lungs preserved for 6 or 12 hours after perfusion with low-potassium dextran or histidine-tryptophan-ketoglutarate. Transpl Proc 2011; 43: 1520–4. [DOI] [PubMed] [Google Scholar]
- 4.Sugimoto R, Tanaka Y, Noda K, Kawamura T, Toyoda Y, Billiar TR, McCurry KR, Nakao A. Preservation solution supplemented with biliverdin prevents lung cold ischaemia/reperfusion injury. Eur J Cardiothorac Surg 2012; 42: 1035–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Van Raemdonck D. Thoracic organs: current preservation technology and future prospects; part 1: lung. Curr Opin Organ Transpl 2010; 15: 150–5. [DOI] [PubMed] [Google Scholar]
- 6.Preissler G, Loehe F, Huff IV, Ebersberger U, Shuvaev VV, Bittmann I, Hermanns I, Kirkpatrick JC, Fischer K, Eichhorn ME, Winter H, Jauch KW, Albelda SM, Muzykantov VR, Wiewrodt R. Targeted endothelial delivery of nanosized catalase immunoconjugates protects lung grafts donated after cardiac death. Transplantation 2011; 92: 380–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Alexiou K, Matschke K, Westphal A, Stangl K, Dschietzig T. Relaxin is a candidate drug for lung preservation: relaxin-induced protection of rat lungs from ischemia-reperfusion injury. J Heart Lung Transpl 2010; 29: 454–60. [DOI] [PubMed] [Google Scholar]
- 8.Paulus P, Holfeld J, Urbschat A, Mutlak H, Ockelmann PA, Tacke S, Zacharowski K, Reissig C, Stay D, Scheller B. Prednisolone as preservation additive prevents from ischemia reperfusion injury in a rat model of orthotopic lung transplantation. PLoS One 2013; 8: e73298–e73298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.de Perrot M, Liu M, Waddell TK, Keshavjee S. Ischemia-reperfusion-induced lung injury. Am J Respir Crit Care Med 2003; 167: 490–511. [DOI] [PubMed] [Google Scholar]
- 10.Rega FR, Jannis NC, Verleden GM, Flameng WJ, Lerut TE, Van Raemdonck DE. Should we ventilate or cool the pulmonary graft inside the non-heart-beating donor? J Heart Lung Transpl 2003; 22: 1226–33. [DOI] [PubMed] [Google Scholar]
- 11.Munshi L, Keshavjee S, Cypel M. Donor management and lung preservation for lung transplantation. Lancet Respir Med 2013; 1: 318–28. [DOI] [PubMed] [Google Scholar]
- 12.Heffner JE, Repine JE. Pulmonary strategies of antioxidant defense. Am Rev Respir Dis 1989; 140: 531–54. [DOI] [PubMed] [Google Scholar]
- 13.Ohsawa I, Ishikawa M, Takahashi K, Watanabe M, Nishimaki K, Yamagata K, Katsura K, Katayama Y, Asoh S, Ohta S. Hydrogen acts as a therapeutic antioxidant by selectively reducing cytotoxic oxygen radicals. Nat Med 2007; 13: 688–94. [DOI] [PubMed] [Google Scholar]
- 14.Ji X, Tian Y, Xie K, Liu W, Qu Y, Fei Z. Protective effects of hydrogen-rich saline in a rat model of traumatic brain injury via reducing oxidative stress. J Surg Res 2012; 178: e9–16. [DOI] [PubMed] [Google Scholar]
- 15.Shi J, Yao F, Zhong C, Pan X, Yang Y, Lin Q. Hydrogen saline is protective for acute lung ischaemia/reperfusion injuries in rats. Heart Lung Circ 2012; 21: 556–63. [DOI] [PubMed] [Google Scholar]
- 16.Wang F, Yu G, Liu SY, Li JB, Wang JF, Bo LL, Qian LR, Sun XJ, Deng XM. Hydrogen-rich saline protects against renal ischemia/reperfusion injury in rats. J Surg Res 2011; 167: e339–44. [DOI] [PubMed] [Google Scholar]
- 17.Hayashida K, Sano M, Ohsawa I, Shinmura K, Tamaki K, Kimura K, Endo J, Katayama T, Kawamura A, Kohsaka S, Makino S, Ohta S, Ogawa S, Fukuda K. Inhalation of hydrogen gas reduces infarct size in the rat model of myocardial ischemia-reperfusion injury. Biochem Biophys Res Commun 2008; 373: 30–5. [DOI] [PubMed] [Google Scholar]
- 18.Kawamura T, Huang CS, Peng X, Masutani K, Shigemura N, Billiar TR, Okumura M, Toyoda Y, Nakao A. The effect of donor treatment with hydrogen on lung allograft function in rats. Surgery 2011; 150: 240–9. [DOI] [PubMed] [Google Scholar]
- 19.Huang CS, Kawamura T, Peng X, Tochigi N, Shigemura N, Billiar TR, Nakao A, Toyoda Y. Hydrogen inhalation reduced epithelial apoptosis in ventilator-induced lung injury via a mechanism involving nuclear factor-kappa B activation. Biochem Biophys Res Commun 2011; 408: 253–8. [DOI] [PubMed] [Google Scholar]
- 20.Sun Q, Cai J, Liu S, Liu Y, Xu W, Tao H, Sun X. Hydrogen-rich saline provides protection against hyperoxic lung injury. J Surg Res 2011; 165: e43–9. [DOI] [PubMed] [Google Scholar]
- 21.Terasaki Y, Ohsawa I, Terasaki M, Takahashi M, Kunugi S, Dedong K, Urushiyama H, Amenomori S, Kaneko-Togashi M, Kuwahara N, Ishikawa A, Kamimura N, Ohta S, Fukuda Y. Hydrogen therapy attenuates irradiation-induced lung damage by reducing oxidative stress. Am J Physiol Lung Cell Mol Physiol 2011; 301: L415–26. [DOI] [PubMed] [Google Scholar]
- 22.Xie K, Yu Y, Huang Y, Zheng L, Li J, Chen H, Han H, Hou L, Gong G, Wang G. Molecular hydrogen ameliorates lipopolysaccharide-induced acute lung injury in mice through reducing inflammation and apoptosis. Shock 2012; 37: 548–55. [DOI] [PubMed] [Google Scholar]
- 23.Pizanis N, Petrov A, Heckmann J, Wiswedel I, Wohlschlager J, de Groot H, Jakob H, Rauen U, Kamler M. A new preservation solution for lung transplantation: evaluation in a porcine transplantation model. J Heart Lung Transpl 2012; 31: 310–7. [DOI] [PubMed] [Google Scholar]
- 24.Mizuta T, Kawaguchi A, Nakahara K, Kawashima Y. Simplified rat lung transplantation using a cuff technique. J Thorac Cardiovasc Surg 1989; 97: 578–81. [PubMed] [Google Scholar]
- 25.Sugimoto R, Nakao A, Nagahiro I, Kohmoto J, Sugimoto S, Okazaki M, Yamane M, Inokawa H, Oto T, Tahara K, Zhan J, Sano Y, McCurry KR. Experimental orthotopic lung transplantation model in rats with cold storage. Surg Today 2009; 39: 641–5. [DOI] [PubMed] [Google Scholar]
- 26.Pirat A, Zeyneloglu P, Aldemir D, Yucel M, Ozen O, Candan S, Arslan G. Pretreatment with simvastatin reduces lung injury related to intestinal ischemia-reperfusion in rats. Anesth Analg 2006; 102: 225–32. [DOI] [PubMed] [Google Scholar]
- 27.Lu MP, Du LZ, Gu WZ, Chen XX. Nitric oxide inhalation inhibits inducible nitric oxide synthase but not nitrotyrosine formation and cell apoptosis in rat lungs with meconium-induced injury. Acta Pharmacol Sin 2005; 26: 1123–9. [DOI] [PubMed] [Google Scholar]
- 28.Soslow RA, Dannenberg AJ, Rush D, Woerner BM, Khan KN, Masferrer J, Koki AT. COX-2 is expressed in human pulmonary, colonic, and mammary tumors. Cancer 2000; 89: 2637–45. [DOI] [PubMed] [Google Scholar]
- 29.Albaiceta GM, Taboada F, Parra D, Luyando LH, Calvo J, Menendez R, Otero J. Tomographic study of the inflection points of the pressure-volume curve in acute lung injury. Am J Respir Crit Care Med 2004; 170: 1066–72. [DOI] [PubMed] [Google Scholar]
- 30.Kawamura T, Huang CS, Tochigi N, Lee S, Shigemura N, Billiar TR, Okumura M, Nakao A, Toyoda Y. Inhaled hydrogen gas therapy for prevention of lung transplant-induced ischemia/reperfusion injury in rats. Transplantation 2010; 90: 1344–51. [DOI] [PubMed] [Google Scholar]
- 31.Weyker PD, Webb CA, Kiamanesh D, Flynn BC. Lung ischemia reperfusion injury: a bench-to-bedside review. Semin Cardiothorac Vasc Anesth 2013; 17: 28–43. [DOI] [PubMed] [Google Scholar]
- 32.Hertzler SR, Savaiano DA, Levitt MD. Fecal hydrogen production and consumption measurements. Response to daily lactose ingestion by lactose maldigesters. Dig Dis Sci 1997; 42: 348–53. [DOI] [PubMed] [Google Scholar]
- 33.Nakao A, Toyoda Y, Sharma P, Evans M, Guthrie N. Effectiveness of hydrogen rich water on antioxidant status of subjects with potential metabolic syndrome-an open label pilot study. J Clin Biochem Nutr 2010; 46: 140–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Ito M, Ibi T, Sahashi K, Ichihara M, Ohno K. Open-label trial and randomized, double-blind, placebo-controlled, crossover trial of hydrogen-enriched water for mitochondrial and inflammatory myopathies. Med Gas Res 2011; 1: 24–24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Ono H, Nishijima Y, Adachi N, Tachibana S, Chitoku S, Mukaihara S, Sakamoto M, Kudo Y, Nakazawa J, Kaneko K, Nawashiro H. Improved brain MRI indices in the acute brain stem infarct sites treated with hydroxyl radical scavengers, edaravone and hydrogen, as compared to edaravone alone. A non-controlled study. Med Gas Res 2011; 1: 12–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Suarez Lopez VJ, Minambres E, Robles Arista JC, Ballesteros MA. Primary graft dysfunction after lung transplantation. Med Intensiva 2012; 36: 506–12. [DOI] [PubMed] [Google Scholar]
- 37.Goldblum SE, Wu KM, Jay M. Lung myeloperoxidase as a measure of pulmonary leukostasis in rabbits. J Appl Physiol 1985; 59: 1978–85. [DOI] [PubMed] [Google Scholar]
- 38.Li H, Zhou R, Liu J, Li Q, Zhang J, Mu J, Sun X. Hydrogen-rich saline attenuates lung ischemia-reperfusion injury in rabbits. J Surg Res 2012; 174: e11–6. [DOI] [PubMed] [Google Scholar]
- 39.Zhou H, Fu Z, Wei Y, Liu J, Cui X, Yang W, Ding W, Pan P, Li W. Hydrogen inhalation decreases lung graft injury in brain-dead donor rats. J Heart Lung Transpl 2013; 32: 251–8. [DOI] [PubMed] [Google Scholar]
- 40.Elmore S. Apoptosis: a review of programmed cell death. Toxicol Pathol 2007; 35: 495–516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Fischer S, Maclean AA, Liu M, Cardella JA, Slutsky AS, Suga M, Moreira JF, Keshavjee S. Dynamic changes in apoptotic and necrotic cell death correlate with severity of ischemia-reperfusion injury in lung transplantation. Am J Respir Crit Care Med 2000; 162: 1932–9. [DOI] [PubMed] [Google Scholar]
- 42.Kawamura T, Wakabayashi N, Shigemura N, Huang CS, Masutani K, Tanaka Y, Noda K, Peng X, Takahashi T, Billiar TR, Okumura M, Toyoda Y, Kensler TW, Nakao A. Hydrogen gas reduces hyperoxic lung injury via the Nrf2 pathway in vivo. Am J Physiol Lung Cell Mol Physiol 2013; 304: L646–56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Tanaka Y, Shigemura N, Kawamura T, Noda K, Isse K, Stolz DB, Billiar TR, Toyoda Y, Bermudez CA, Lyons-Weiler J, Nakao A. Profiling molecular changes induced by hydrogen treatment of lung allografts prior to procurement. Biochem Biophys Res Commun 2012; 425: 873–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Valenza F, Rosso L, Pizzocri M, Salice V, Umbrello M, Conte G, Stanzi A, Colombo J, Gatti S, Santambrogio L, Iapichino G, Gattinoni L. The consumption of glucose during ex vivo lung perfusion correlates with lung edema. Transpl Proc 2011; 43: 993–6. [DOI] [PubMed] [Google Scholar]
- 45.Koike T, Yeung JC, Cypel M, Rubacha M, Matsuda Y, Sato M, Waddell TK, Liu M, Keshavjee S. Kinetics of lactate metabolism during acellular normothermic ex vivo lung perfusion. J Heart Lung Transpl 2011; 30: 1312–9. [DOI] [PubMed] [Google Scholar]
- 46.Santana Rodríguez N, Llontop Santisteban P, López García A, Clavo B, Ponce González MÁ, Rodríguez Bermejo JC, García Castellano JM, García Herrera R, Zerecero K, Ruíz Caballero JA, Brito Godoy Y, Varela De Ugarte A. Technical modifications of the orthotopic lung transplantation model in rats with brain-dead donors. Arch Bronconeumol 2011; 47: 488–94. [DOI] [PubMed] [Google Scholar]