Abstract
Osteoarthritis (OA) is an age-related degenerative joint disease. To date, its management is focused on symptoms (pain and inflammation). Studies suggest that fatty acids can reduce the expression of inflammatory and catalytic mediators, and improve in vivo joint function. Free fatty acid receptors (FFARs) such as G-protein coupled receptor 40 (GPR40) are proposed as attractive therapeutic targets to counteract inflammation and cartilage degradation observed in OA. This study aims to elucidate the involvement of GPR40 in OA. In this study, we used an in vitro model of OA, and surgically induced OA by ligament transection and partial meniscectomy in wild-type and GPR40 deficient mice. OA phenotype was investigated in vivo by histology and genes expression. We demonstrate that IL-1β-treated GPR40−/− chondrocytes secret more inflammatory mediators (nitric oxide, interleukin-6, prostaglandin E2) and active catabolic enzymes (metalloproteinase-2, -9 [MMP-2, MMP-9]), and show decreased anabolism (glycoaminoglycan) compared to GPR40+/+ cells. In accordance with these results, we show that GPR40−/− mice exhibit an aggravated OA-induced phenotype characterized by higher tidemark exposure, frequency of osteophyte formation and subchondral bone sclerosis. Altogether our results demonstrate that GPR40 deficiency leads to an extended OA phenotype, providing evidence that increasing GPR40 activity, by natural or synthetic ligands, could be a new strategy in the management of OA.
Keywords: Free fatty acid receptor (FFAR), G-protein coupled receptor 40, osteoarthritis, natural therapy
Introduction
Osteoarthritis (OA) is the most common degenerative joint disease. In 2003, the World Health Organization (WHO) reported that more than 50% of people over 65 years old have radiological evidence of OA.1 Although articular cartilage degradation is its main feature, the disease is now considered to damage the whole joint, involving synovitis, remodeling of subchondral bone and osteophyte formation.2 OA causes disability and leads to high morbidity in industrialized countries.3 The strongest risk factors for OA are injury, excessive mechanical stress, and aging.4 More recently, OA has been linked to obesity.5–7 Consequently OA is now considered as a major public health issue.
Dietary lipids derived from plants and animals encompass fatty acids (saturated, mono- and poly-unsaturated), their derivatives, and sterols. Beside their role in obesity development and subsequent impact on cartilage,8 dietary lipids have been reported to exert dual effects on inflammation with both pro- and anti-inflammatory effects depending on their structure and metabolism.9 Joint inflammation contributes to the induction of proteases that degrade cartilage during OA. In turn, the cartilage permeability increases and so does the dietary lipid availability for chondrocytes.8
In this regard, a growing body of evidence suggests a beneficial effect of dietary poly-unsaturated fatty acids (PUFAs) in rheumatoid diseases such as OA. Cho et al. showed that the consumption of n-3 PUFAs rich oil improved joint function and reduced pain in OA patients.10 Similarly, an animal model of spontaneous OA fed with a n-3 enriched diet exhibits a lower OA score associated to an increase of glycosaminoglycan (GAG) content and a decrease of MMP activity.11 In addition, Huang et al. showed that decreasing n-6/n-3 ratio by endogenous conversion of n-6 to n-3 fatty acids in mice delayed OA progression.12 In vitro experiments performed on chondrocyte cultures and cartilage explants stated that PUFAs (mainly n-3) reduced both inflammatory markers (interleukin-1β [IL-1β], prostaglandin E2 [PGE2], nitric oxide (NO)) and catabolism factors (MMP-13, A disintegrin and metalloproteinase with thrombospondin motifs (ADAMTS) -4 & -5) observed in OA joints.13,14 However, mechanisms of PUFA action are poorly understood, especially whether or not fatty acid receptors including liver X receptor (LXR), peroxisome-proliferator-activated receptor (PPAR), or G-protein coupled receptor (GPR) families are implicated in these effects.
Among potential receptors linking inflammation, lipids and health, the previously orphan GPR40 was shown to interact with medium to long-chain fatty acids and to regulate insulin secretion of pancreatic β-cells.15,16 Expression of GPR40 was also documented in leukocytes, macrophages, and bone marrow stromal cells17,18 that share common precursors with the bone and cartilage lineage. We recently demonstrated that GPR40 knock-out (GPR40−/−) mice exhibited osteoporotic features and that GPR40 stimulation preserved bone mass.19 Regarding the close relationships between bone and cartilage tissues behavior, the role of GPR40 in cartilage physiopathology was investigated in the present study. First, we performed in vitro experiments to investigate the consequence of GPR40 deficiency on chondrocyte metabolism. As IL-1β is a pro-inflammatory cytokine widely used to establish OA model in vitro,20 attention was given to GPR40 deficiency on IL-1β-treated articular chondrocytes. Then we evaluated the in vivo impact of GPR40 on articular cartilage integrity and on OA phenotype following an OA-inducing surgery in WT and GPR40+/+ mice. Taken together, our results provide evidence for a protective role of GPR40 in the cartilage and reveal new opportunities for OA management.
Materials and methods
Animals
GPR40−/− mice were obtained from AMGEN Inc. As described by Latour et al.,21 GPR40−/− mice were generated by replacing exon 2 of the Gpr40 gene with a LacZ gene, using homologous recombination in embryonic stem cells. The mice were backcrossed onto the C57BL/6 strain over nine generations. Wild-type B6 (GPR40+/+) littermates were used as controls. Mice were handled according to the European Guidelines for Care and Use of Laboratory Animals (EEC Directives 86/609/CEE of 24.11.1986). All experimental animal procedures conducted in the present study were approved by the Ethics Committee in Animal Experiment, CEMEA Auvergne (Agreement CE-13-12; Clermont-Ferrand, France).
Isolation and culture of mouse articular chondrocyte (MAC)
MACs were harvested from knee joints as previously described by Gosset et al.22 on 5–6 day-old newborn GPR40−/− mice and their respective wild-type littermates (C57BL6-J GPR40+/+ mice). Briefly, primary articular chondrocytes were isolated after digestion with collagenase D (Sigma, France) at 2.5 mg/mL for 1.5 h at 37℃ under shaking, followed by overnight incubation with collagenase D at 0.5 mg/mL. Chondrocytes were grown to confluence in Dulbecco’s Modified Eagle’s Medium (DMEM 4.5 g/L glucose) (Gibco, NY) supplemented with 10% fetal bovine serum (FBS, PAA Laboratories), 1% of an antibiotic/ anti-mycotic solution (PAA Laboratories, France) and 2 mmol/L of glutamine (Sigma, France). Cells were made quiescent for 24 h in DMEM medium with 0.2% FBS, and then they were stimulated with or without recombinant interleukin-1 beta (rIL-1β at 10 ng/mL (ImmunoTools, Germany) for 48 h. After IL-1β stimulation, culture media were collected, cells were lysed using lysis buffer (50 mmol/L Tris pH 7.8, 150 mmol/L NaCl, 0.5% sodium deoxycholate, 1% NP40), and each fraction was stored at −80℃ until analyses.
NO, PGE2, and IL-6 assays
NO production was estimated using a nitrate (NO3)/nitrite (NO2) detection kit (EnzoLife Siciences, Switzerland): NO concentration in the collected media was determined as the result of nitrate conversion to nitrite. Then nitrites were detected by a colorimetric reaction using the Griess reagent at 540 nm. NO species were determined from a standard curve of nitrate. Results are expressed in µmol/L of NO per mg of total cell proteins (determined by BCA assay [Sigma, France]).
PGE2 and IL-6 productions were measured in the media by a high sensitivity enzyme immuno-assay kit (EIA, EnzoLife Sciences) according to the manufacturer’s instructions. Concentrations were analyzed following serial dilutions in duplicate, read against standard curve and expressed in pg of PGE2 or IL-6 per mg of total cellular proteins determined by BCA assay (Sigma).
GAG assay
A dimethylmethylene blue (DMB) assay was used to detect GAG production in cell lysates. DMB solution was prepared at final concentration of 46 mmol/L in a pH 3 adjusted buffer: 40 mmol/L NaCl, 40 mmol/L glycine. Sample concentrations were determined by mixing 50 µL of cell extract with 200 µL of DMB reagent. Following 30 min incubation, the absorbance was read at 595 nm on an ELX808 IU spectrophotometer (BioTek Instruments, USA). The GAG content was determined using a standard curve of chondroitin sulfate (Sigma). Results are expressed as µg of GAG per mg of total proteins determined by BCA assay (Sigma).
Zymography
MMP-2, MMP-9, and MMP-13 expression and activity were determined by gelatinase zymography. Ten microliters of media were loaded onto a 7.5% SDS-polyacrylamide gel containing 1 mg/mL of gelatin. Following the electrophoresis the gel was incubated for 18 h in 0.1% Triton X-100, 0.2 mol/L NaCl, 5 mmol/L CaCl2 and 50 mmol/L Tris pH 7.4, and then stained with coomassie blue. Gel images were captured and the clear bands were analyzed using “ImageJ” image analysis software (www.imagej.nih.gov), and their optical density was expressed in arbitrary units. Graphs represent MMP activity expressed as the mean ± standard deviation of six independent measurements.
Surgical induction of OA
OA was surgically induced by a combination of ligament transection and partial meniscectomy mentioned as partial median meniscectomy (PMMx). The experiment was performed on 12-week-old male mice (n = 10 for both genotypes, GPR40+/+ and GPR40−/−) which were divided into two groups: sham-operated control mice (Sham) and induced OA mice (PMMx). Each mouse was given analgesics prior to surgery (0.4 mg/kg buprenorphine, Axience, France), and was anesthetized by intra-peritoneal injection of 100 mg/kg ketamine (Imalgene 1000®, Merial France) and 10 mg/kg xylazine (Rompun® 2%, Bayer France). Briefly, for PMMx, the joint capsule was incised in the medial side of the patella. The patella was dislocated to give greater exposure of the knee. The anterior attachment of the medial meniscus to the tibial plateau was transected and the anterior half of the meniscus was removed with careful precaution to avoid cartilage injuries.23,24 The operated field was irrigated with saline. In sham-operated control mice, the ligament was visualized but not transected. The skin was then closed with interrupted non-resorbable sutures (Vicryl®, Ethicon). Surgeries were performed on the both knees. Mice had normal mobility within 4 h after surgery.
Histology
Mice were euthanized eight weeks after surgery. Whole knee joints were either fixed in 4% paraformaldehyde for histology or carefully dissected to remove muscles and immediately frozen in liquid nitrogen, and kept at −80℃ for gene expression analyses. Fixed joints were decalcified in EDTA 0.5 mol/L pH 7.4 for one week at 4℃, and then embedded in paraffin. Five micrometer frontal sections were prepared from the entire knee using a Leica RM 2165 Microtome. Slides were stained with safranin-O and Fast green. Three sections of each knee were subjected to histomorphometry analyses.
Semi-quantitative scoring of osteoarthritic damages was performed by two-blinded observations using the Osteoarthritis Research Society International (OARSI)25 grade where 0 represents normal cartilage; 0.5: loss of Safranin-O with no structural lesions; 1: roughened articular surface and small fibrillations; 2: fibrillation below the superficial layer and some loss of lamina; 3: fibrillations extending to the calcified cartilage across less than 20% of the cartilage width; 4–5: fibrillations and erosions extending from 25% to 50%, and from 50% to 75% of the cartilage width; 6: cartilage erosion extending beyond 75% of the cartilage width. Histological scoring was performed on the four quadrants: medial and lateral femoral condyles and medial and lateral tibial plateaus. Results are expressed as the sum of all scores. Thickness of uncalcified and calcified articular cartilage (µm) was expressed as the mean of 10 repeated measurements performed in the middle zone of tibial cartilage. Cartilage area (uncalcified cartilage area/total cartilage area), tidemark exposure (expressed in % and corresponding to the ratio between the length of intra-articular-exposed tidemark and its total length: 100 − [Uncalcified cartilage length/total tidemark length]), subchondral bone density (bone area/tissue area ratio measured in a region of interest [ROI] located under articular cartilage area and extending to the growth plate), and osteophyte area (µm2) were evaluated manually using Image J software. Values represent mean ± standard error of the mean (SEM).
Serum bone turnover markers assays
The N-terminal fragment of collagen 1 (PINP), a bone formation marker, and the C-terminal fragment (CTX-1), a bone resorption marker, were detected in sera collected eight weeks after OA induction, by ELISA assay according to the manufacturer’s instructions (Immundiagnosticsystem, Germany). Results are expressed as ng of markers per mL of serum.
Relative gene expression
Frozen whole knees (from the tibia growth plate to the femoral condyles) were ground in liquid nitrogen to obtain a fine powder. Total RNA was extracted from knee powder using Trizol. RNA concentration was measured using a spectrophotometer NanoDrop ND-1000 (LabTech, Ringmer, UK). One microgram was used as template for single-strand cDNA synthesis with the DyNamoTM cDNA synthesis kit (FINNZYMES) in a 20 µL final volume containing 1x RT buffer, 0.5 µg of random primers and, 2 µL of M-MuLV RNase H+ reverse transcriptase. After an incubation at 42℃ for 50 min, reaction was stopped at 70℃ for 15 min. Five microliters of diluted cDNA (1:40 in water) were loaded in a 96-well PCR plate (Eppendorf, France). Amplification of the cDNA using specific primers (Table 1) and Express Sybr GreenER qPCR Super Mix Universal (Invitrogen, France) was performed using the Realplex2 Mastercycler (Eppendorf, France). After a 5 min 95℃ step for activation of DNA polymerase, cDNA was amplified by performing 40 two-step PCR cycles: a denaturation step (15 s at 95℃), followed by an annealing step (15 s at 58℃) and an extension step (15 s at 60℃). Q-PCR was performed in duplicate for PCR yield validation. Data were analyzed with the Realplex software (Eppendorf) and compared by the ΔΔCt method. Results were expressed relative to the housekeeping gene transcript amount and normalized to the GPR40+/+sham-operated condition.
Table 1.
Forward and reverse primers used for quantitative PCRa
| Gene | GeneBank No. | Forward (5′–3′) | Reverse (5′–3′) | References |
|---|---|---|---|---|
| GPR40 | NM_1940S7.2 | GGCCCTATAATGCCTCCAAT | CCAGGACCTGTTCCCAAGTA | |
| GPR120 | NM_181748.2 | AGACTACCGACTCTTCCGCA | AAGAAAAGGGATGGCCAGAT | |
| Type II collagen (Col2Al) | NM_081163.3 | ATCTTGCCGCATCTGTGTGT | CTCCTTTCTGCCCCTTTGGC | |
| Aggrecan (ACAN) | NM_007424.2 | GGTCACTGTTACCGCCACTT | CCCCTTCGATAGTCCTGTCA | |
| COMP | NM_0166S5.2 | TGCGACGACGACATAGATGG | ACATCCCTCTGGTCTGGGTT | |
| MMP-2 | NM_008S10 | GCAGGAGACAAGTTCTGGAG | AGAAGTAGCTATGACCACCACCC | |
| MMP-9 | NM_013599 | ACTCACACGACATCTTCCAG | AGAAGGAGCCCTAGTTCAAG | 43 |
| MMP-13 | NM_008607.2 | GACCCACAGATGAGCACAGA | ATGTAAGGCCACCTCCACTG | |
| ADAMTS4 | NM_172845.2 | GGCAAGGACTATGACGC | TCAGCCCAAGGTGAGTG | 44 |
| ADAMTS5 | NM_011782.2 | CCTGCCCACCCAATGGTAAA | GTCCTCGGACACACACAGAG | |
| IL-1β | NM_OOS3613 | TTCAGGCAGGCAGTATCACTC | GAAGGTCCACGGGAAAGACAC | |
| TNF-α | NM_013693.2 | ATGGOCTCCCTCTCATCAGT | GCAGCCTTGTCCCTTGAAGA | |
| COX2 | NM_011198 | TGAGTACCGCAAACGCTTCT | ACGAGGTTTTTCCACCAGCA | |
| IL-6 | NM_031168 | TGAGAAAAGAGTTGTGCAATGG | TCTCTCTGAAGGACTCTGGCT | |
| iNOS | NM_010927 | TTTGTGCGAAGTGTCAGTGG | CAAACACCAAGCTCATGCGG | |
| Cyclophilin A | NM_00S907 | GGTGACTTTACACGCCATAATG | GGCTTCCACAATGTTCATGCC | 45 |
aPrimers were designed using Primer 3 software.46
Statistical analyses
As cartilage degradation is the main feature of OA, tidemark exposure was chosen as the main outcome to calculate the statistical power to demonstrate a significant difference between PMMx and control (sham-operated) mice. Based on previous data on tidemark exposure obtained with this animal model of OA, Gpower3 software determined that five animals per group are sufficient to observe a significant difference (at least 10%) with a power of 0.80.
Data were analyzed using the non-parametric Mann–Whitney test, except for the osteophyte frequencies that was compared using a χ2 test. Whatever the test, differences were considered to be statistically significant when P < 0.05. (*) Indicates significant differences to untreated or sham-operated condition, (§) to treated or PMMx GPR40+/+ condition, and (#) to untreated or sham-operated GPR40+/+ condition.
Results
GPR40−/− chondrocytes display an intense OA phenotype in response to IL-1β stimulation
We first examined the expression of GPR40 mRNA in mouse articular cartilage (Table 2). Gpr40 gene expression was detected in all tested tissues, similarly to the other free fatty acid receptor (FFAR) gene gpr120. Quantification of gene expression revealed that the detected amount of GPR40 mRNA in articular cartilage was 6-fold lower than in adipose tissue or 20-fold lower than in long bones such as femur.
Table 2.
GPR40 expression in mouse tissuesa
| mRNA relative expression |
||
|---|---|---|
| GPR40 | GPR120 | |
| Articular cartilage | 1.3 | 13.0 |
| Heart | 1.0 | 1.0 |
| Liver | 3.9 | 15.8 |
| Spleen | 37.0 | 12.0 |
| Lung | 5.8 | 12.9 |
| Gut | 7.3 | 9.6 |
| Brain | 3.5 | 4.5 |
| Adipose tissue | 7.8 | 186.1 |
| Muscle | 17.1 | 9.9 |
| Calvaria | 3.5 | 40.4 |
| Bone (femur) | 25.8 | 30.4 |
aExpression of free fatty receptors GPR40 and GPR120 in mouse tissues was investigated by RT-PCR. Relative expression was normalized to GAPDH (internal standard) and calculated using the ΔΔCt method.
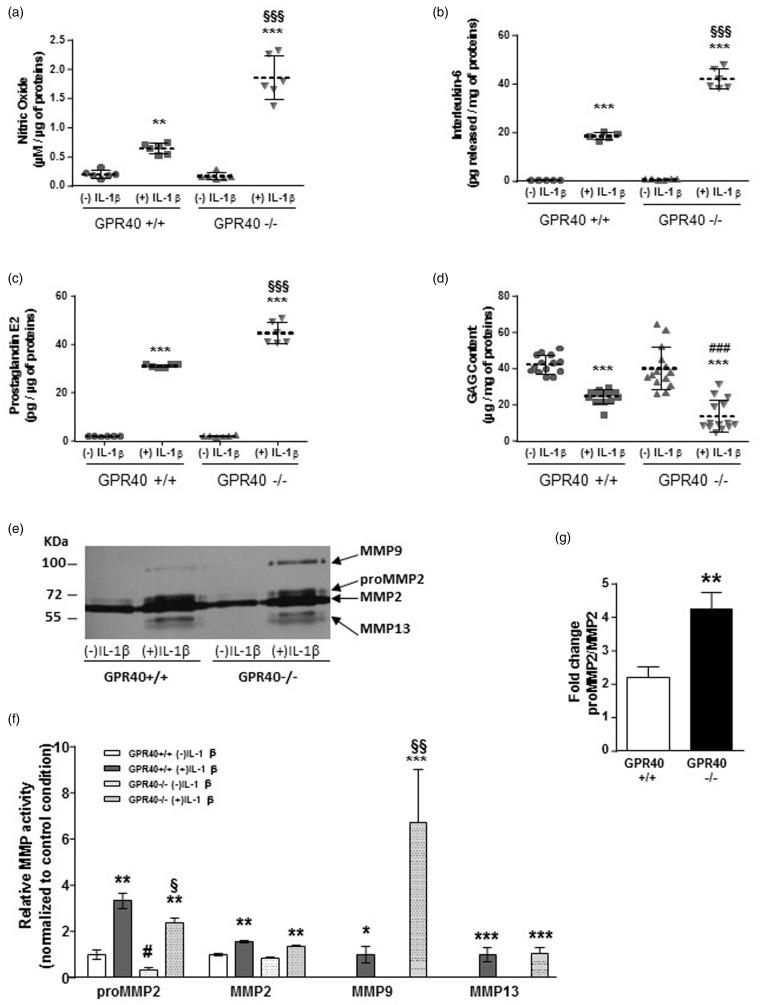
The impact of GPR40 deficiency on the metabolism of primary murine articular chondrocytes (MACs) was evaluated to address the role of GPR40 in articular cartilage physiopathology. We observed that GPR40 deficiency did not modify the inflammatory (PGE2, IL-6), anabolic (GAG), and catabolic activities (MMP-2, -9, -13) of MACs at basal level, i.e. without IL-1β stimulation (Figure 1). Next we stimulated chondrocytes with IL-1β to establish OA model in vitro. Incubation of GPR40+/+ MACs with 10 ng/mL rIL-1β for 72 h significantly increased the secretion of NO, IL-6, and PGE2 (Figure 1a–c). IL-1β-induced inflammatory response observed in GPR40−/− cells was significantly greater than those in GPR40+/+ cells (Figure 1a–c).
Figure 1.
GPR40 deficiency induces a more severe OA phenotype in mouse articular chondrocytes in response to an inflammatory stimulus. Secretion of inflammatory mediators such as nitric oxide (a), interleukin-6 (b), and prostaglandin E2 (c) was measured in the culture medium of GPR40+/+ and GPR40−/− chondrocytes, treated with or without 10 ng/mL IL-1β for 72 h (n = 6). Extracellular GAG content (d) was measured in chondrocyte cultures (n = 14). IL-1β-induced matrix metalloproteinase activities were investigated in culture medium by zymography (n = 6). The picture shows a representative gelatin gel stained by coomassie blue (e). Relative MMP activity was quantified by densitometry (f). The fold-change of proMMP2/MMP2 ratio induced by IL-1β treatment was also calculated (g). Results are represented with means ± SEM. (*), (#), and (§) indicate significant differences compared to IL-1β untreated group, IL-1β treated GPR40+/+ group and to IL-1β untreated GPR40+/+ group, respectively
Here, we measured GAG matrix content and demonstrated a significant decrease of GAG content after IL-1β exposure compared to untreated cells both in GPR40+/+ and GPR40−/− cultures (Figure 1d). This decrease of GAG content was significantly higher in IL-1β-induced GPR40−/− cells (−67 ± 5%) than in IL-1β-induced GPR40+/+ cells (−41 ± 2%). Then, we analyzed the role of GPR40 deficiency on catalytic enzyme activity by a densitometry measure on a gelatin zymography (Figure 1e–g). Our results reveal that MMP-2 (pro- and active), MMP-9, and MMP-13 were induced by IL-1β (Figure 1e,f). In GPR40−/− cells, stimulation with IL-1β significantly enhanced MMP-9 catalytic activity up to six-fold as compared to the level in GPR40+/+ cells (Figure 1f,g). No difference was found for IL-1β-induced MMP-13 activity between GPR40+/+ and GPR40−/− chondrocytes.
Altogether, these data, obtained on an in vitro-induced OA model, demonstrated that IL-1β-induced inflammation, extracellular matrix loss, and catabolic activity were enhanced in GPR40-deficient chondrocytes.
GPR40−/− mice do not spontaneously exhibit OA phenotype
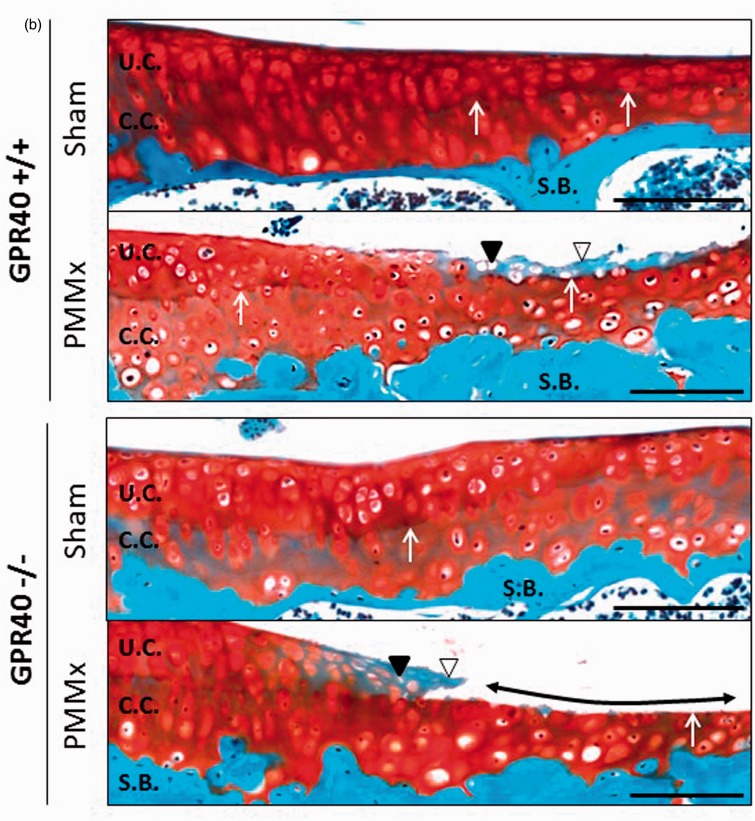
To further investigate the potential role of GPR40 in cartilage physiopathology, we next characterized the knee cartilage integrity on five-month-old GPR40+/+ and GPR40−/− mice. Safranin O/fast green staining demonstrated that the knee articular cartilage was intact both in GPR40+/+ and GPR40−/− strains, without any sign of cartilage damage (Figure 2a). Moreover, the thickness of cartilage (uncalcified and calcified) measured on the tibial plateau was similar in both genotypes (supplementary Figure 1).
Figure 2.
GPR40 deficiency results in an enhanced joint injury in surgical-induced OA knees. (a) Representative histological cross-sections of GPR40+/+ and GPR40−/− joint knees, eight weeks after surgical OA induction (PMMx) or not (sham-operated), stained with Safranin O/fast green. Black arrow heads show area of cartilage degradation and tidemark exposure; asterisks localize subchondral bone (S.B.) sclerosis; dotted lines delimit osteophytes at the merge of tibia. Bars represent 1 mm (magnification x5). (b) Representative focused images from articular cartilage of the medial tibial plateau. White arrow heads indicate loss of safranin O staining; black arrow heads show empty chondroplast due to chondrocyte apoptosis, white arrows localize the tidemark (between uncalcified [UC] and calcified cartilage [CC]); double head arrows demonstrate tidemark exposures. Bars represent 500 µm (magnification x10). (c) OARSI grade. Bars represent means of the summed scores for all quadrants of each group ± SEM (n = 5). (d and e) Quantification of the uncalcified/total cartilage area ratio and of the Tidemark exposure, respectively, both reflecting cartilage breakdown on the medial tibia plateau. Results are presented as means ± SEM (n = 5). (f) Bone volume fraction (bone area/tissue area) measured in the subchondral bone (medial tibia). (g) Representative images focused on the median tibial edge. Bars represent 500 µm (magnification x10). Dotted lines circle osteophyte area. (h) Frequency of osteophyte formation in PMMx groups and mean area of osteophyte per group (µm2) ± SEM. (*), (#), and (§) indicate significant differences compared to IL-1β untreated group, IL-1β treated GPR40+/+ group and to IL-1β untreated GPR40+/+ group, respectively. (A color version of this figure is available in the online journal.)
Consistently with the phenotype observed on chondrocytes without OA stimulus, these results show that GPR40 deficiency alone is not sufficient to induce a histological modification of cartilage on five-month-old mice.
GPR40−/− mice develop aggravated OA-induced phenotype
The effect of whole-body GPR40 deficiency on OA was investigated in a mouse model of OA induced by knee joint instability (PMMx). In this study, lateral and medial sides of the joint were analyzed. After eight weeks of OA induction, as expected cartilage degradation was mostly observed on the medial tibial plateau of PMMx mice (Figure 2a). As a consequence of median destabilization, some OA lesions were detected on the lateral side of the joint. OA lesions, as illustrated on median side of the tibial plateau (Figure 2b), were characterized at a tissue level by a clustering of chondrocytes in the superficial area of cartilage and a loss of Safranin O staining in the cartilage breakdown area associated with a chondrocyte death. These features were observed on PMMx-GPR40−/− and PMMx-GPR40+/+ knees. OA severity determined by OARSI score (Figure 2c) was found significantly higher in PMMx knees compared to sham joints. Interestingly, we observed that cartilage lesions were more spread in GPR40−/− mice than in GPR40+/+ littermates resulting in a slight increase of the semi-quantitative histological grade (+40%, P = 0.08). These lesions were characterized by a loss of the uncalcified cartilage. Quantification of the uncalcified/total cartilage surface ratio showed that uncalcified cartilage breakdown in the medial tibial plateau represented a 50% decrease of cartilage surface in GPR40−/− mice (Figure 2d). As a consequence of cartilage loss, exposure of the boundary between the uncalcified and calcified cartilage layers (tidemark) was found significantly increased in GPR40−/− as compared to GPR40+/+ OA knees (46 ± 7% and 26 ± 2%, respectively, P = 0.01) (Figure 2e).
We also observed a modification of the subchondral bone microarchitecture. Indeed in PMMx group, histological observations reveal a densification of the trabecular bone network of the tibial plateaus also called bone sclerosis (Figure 2a). To quantify the bone sclerosis, bone volume fraction was measured in a ROI located under the articular cartilage area, and extending to the growth plate (Figure 2f). In sham-operated mice, bone area/tissue area ratio was significantly lower in GPR40−/− mice than GPR40+/+ mice (−20%). In GPR40−/− tibial plateau, the extended cartilage degradation was associated with an increased bone sclerosis phenotype characterized by a 30% higher bone volume compared to the respective sham control. However, no difference was observed for bone volume fraction in PMMx group between GPR40+/+ and GPR40−/− mice.
These results show that surgical destabilization of the joint induced a rapid and intense OA phenotype leading to the development of advanced stage pathology such as osteophyte formation. Osteophyte formation was only observed on the medial tibial plateaus of the knee joint with a higher frequency in GPR40−/− mice than in GPR40+/+ mice (Figure 2g,h). When osteophytes were present, total osteophyte area was measured. Quantification showed that GPR40−/− osteophyte size was 30% lower than in GPR40+/+ OA joints.
We performed additional experiments to determine the bone remodeling status in each mouse group. Thus, the bone formation marker, N-terminal fragment of collagen 1 (PINP) and the bone resorption marker, C-terminal fragment (CTX-1) were detected in mouse sera after eight weeks of OA induction (Figure 3). We first observed a significant decrease of PINP when OA was surgically induced in GPR40+/+ (−22%). In contrast, the serum levels of PINP and CTX1 were not modified in response to OA induction in GPR40−/− mice supporting that GPR40 play a role in the control of the bone mass. In addition, a significant increase of the bone resorption marker in sham-operated GPR40−/− sera compared to control sera was detected (+16%). These results sustain that bone remodeling may be involved in OA and hypothesize that GPR40 may protect from altered bone resorption in OA.
Figure 3.
GPR40 deficiency sustains bone status in surgical-induced OA mice. The bone formation marker, N-terminal fragment of collagen 1 (PINP), and the bone resorption marker, C-terminal fragment (CTX-1), were detected in mouse sera. Results are presented as mean ± SEM; *, #: P < 0.05, non-parametric Mann–Whitney test (n = 5 per group). (*) and (#) and indicate significant differences compared to sham-operated GPR40+/+ group and PMMx-GPR40+/+ group, respectively
Taken together, these results provide evidence that the absence of GPR40 expression in a surgical-induced OA model results in an aggravated OA phenotype in tibiofemoral joints characterized by an extended cartilage breakdown associated to an elevated osteophyte formation, a greater bone sclerosis and a higher bone resorption.
GPR40−/− joints exhibit increased expression of inflammatory and catabolic markers
To further assess the aggravated cartilage breakdown in GPR40 knock-out mice compared to wild-type mice, we analyzed the expression of a wide range of key markers implicated in OA after eight weeks of induction (Figure 4). Using quantitative PCR, we first determined the expression of inflammatory factors (Figure 4a). PMMx GPR40−/− joints showed a slightly increased expression of IL-6 (1.54 fold-change), inducible iNOS (1.44 fold-change) and cyclooxygenase COX-2 (1.58 fold-change), and a significant elevation of TNF-α expression (1.78 fold-change, P = 0.02) compared to PMMx GPR40+/+ joints (Figure 4a). Overall the inflammatory level measured in GPR40−/− knees was higher than in GPR40+/+. In addition to inflammation, we observed an elevated expression of catabolic enzymes such as MMP-2 (1.44 fold-change, P = 0.04), ADAMST4 (1.72 fold-change) and ADAMST5 (1.65 fold-change, P = 0.01) in GPR40 deficient joints (Figure 4b). Cartilage anabolism was also investigated and we showed an increase of COMP and collagen X expressions in PMMx groups compared to sham-operated groups (Figure 4c). However, we noted that the fold-changes measured between PMMx and Sham groups were lower in GPR40−/− knees than in GPR40+/+ knees (1.28 versus 1.79, P = 0.04 for COMP and 1.28 versus 1.8, P = 0.16 for ColX, respectively). In addition, the expression of collagen type 2 was significantly lower in GPR40−/− knees than in GPR40+/+ knees (COL2A1, P = 0.02), suggesting that cartilage anabolism was more decreased in surgical-induced OA knees from GPR40−/− mice than GPR40+/+ mice.
Figure 4.
GPR40 deficiency alters expression of inflammatory, catabolic, and anabolic cartilage markers. Expression of a: inflammatory genes (IL-1β, IL-6, TNF-α, COX-2, iNOS), b: catalytic enzymes (MMP-2, -9, -13, ADAMST-4 and -5) and c: markers of cartilage anabolism (ACAN, COMP, Col2A1, ColX and TGFβ) in GPR40+/+ and GPR40−/− joint knees were normalized to that of the respective cyclophilin A (internal standard) and then to results obtained in GPR40+/+ sham group (value = 1). Results are presented as means ± SEM (n = 5). Values indicate fold-changes between compared groups. Statistics (P value) performed on fold-changes, using a posteriori Mann–Whitney test is indicated on the graphs
All these results indicate that the GPR40 deficiency leads to a higher expression of inflammatory markers and catabolic enzymes, associated to a reduced cartilage formation in advanced stage of OA, compared to a control situation. These observations are in agreement with the extend cartilage degradation observed on histological sections of OA-induced GPR40−/− knees.
Discussion
In the present study, we report for the first time that the GPR40 receptor is involved in OA severity. In vitro and in vivo experiments demonstrated that GPR40 deficiency alone is not sufficient to induce a histological modification of cartilage or a change of the basal metabolism of chondrocytes. However in vitro- and in vivo-induced OA features are much more severe in GPR40 deficient models providing evidence that GPR40 activation may protect joints for OA.
Indeed, results obtained from articular chondrocyte cultures support a protective role of GPR40 on chondrocytes. Activation of chondral IL-1β receptors induces myeloid differentiation factor 88 (MyD88)-dependent signaling and subsequent nuclear factor-kappa-B (NF-κB) transcription activity that drives pro-OA mediators expression including MMPs, ADAMTS, and pro-inflammatory cytokines.26,27 Recently, activation of FFAR1 and FFAR3 by endogeneous or synthetic ligands has been shown to inhibit NF-κB activation in both osteoclasts and macrophages.19,28 In the light of these data, inhibition of NF-κB signaling pathways may contribute to explain the protective role of GPR40 in OA.
To explore the role of GPR40 in cartilage and OA, we used a whole-body GPR40 deficient mouse strain. Since OA spontaneously occurs with aging in C57BL/6 inbred and genetically modified mouse strain,29 we first assessed the impact of GPR40 deficiency on the integrity of joint knee. No OA features were observed in five-month-old GPR40−/− mice without any OA induction, consistent with the absence of in vitro chondral disorders. These data contrast the osteoporotic features observed in GPR40−/− mice at the same age, and would suggest that bone changes observed in GPR40 knock-out mice are not sufficient to drive OA. Nevertheless, the histological analyses were performed at the age of five months, while OA occurs during aging.30 In the C57BL/6 mouse strain, OA arises after over one year old;31,32 thus an aggravated age-related spontaneous OA phenotype in GPR40−/− mice remains to be deciphered.
Altered joint loading due to over-weight or instability of the joint represents a significant risk factor for the onset and the progression of OA in humans.33–35 Accordingly, we used a model of surgical joint destabilization that mimics the OA histopathology and molecular pathology observed in human and thus strongly supports the relevance of our data. We demonstrated that GPR40 deficiency worsens the aforementioned joint tissue injuries observed in OA and that it is not linked to an increased joint loading due to over-weight (supplementary Figure 2).
In addition osteophytes, another adverse development in OA progression, were more frequent in OA-GPR40−/− than in OA-GPR40+/+ joints, but their area tended to be lower. Excessive mechanical loading, a well-known factor that controls endochondral ossification,36 may participate in this uncontrolled ectopic bone formation. However, this increased endochondral ossification in GPR40−/− mice contrasts with the altered bone mass observed in long bone. It is worth noting that stimulation of GPR40 in osteoblastic cells results in an inhibition of their mineralization capabilities (unpublished data). Consequently, one may speculate that these seemingly conflicting observations may in fact result from higher remodeling in GPR40−/− mice. Serum detection of bone remodeling markers strengthens that bone resorption is increased in GPR40−/− mice compared to GPR40+/+ mice. A growing body of evidence suggests that osteoclasts may be involved in OA.37 Overall these results raise the hypothesis that bone expressed GPR40 is a promising target to counteract OA.
From a nutritional point of view, omega-3 (n-3) PUFAs have been evidenced to modulate inflammation and cartilage degradation,13,38,39 that contribute to prevent, slow down or rescue OA phenotype, and to improve mobility and activity.11 With regards to our data pointing to a protective role of GPR40 in an OA context, and to the fact that fatty acids are well-known natural ligands of GPR40, one can speculate that these results originate from a GPR40 involvement.
No polymorphism or differential expression of GPR40 has been reported in human OA cartilage compared to healthy cartilage. However, data from expression arrays (GEO database, Pubmed) indicate that GPR40 expression increased during differentiation by chondrocytes isolated from healthy human articular cartilage.40 In addition, expression arrays from Dehne et al. reveal an over expression of GPR40 in human OA chondrocytes compared to healthy chondrocytes.41 These results are in contrast to the constant GPR40 expression observed in the OA-induced mouse (unpublished data) and rat.42 Further investigations are needed on cartilage tissue to support that GPR40 is over expressed in human OA and that it could be an attractive target to treat OA.
In conclusion, our results clearly demonstrate that GPR40 deficiency heightens inflammation, cartilage catabolism, and subchondral bone remodeling resulting in an aggravated OA phenotype (both in vitro and in vivo). In addition, our results sustain that an increased bone remodeling observed in GPR40−/− likely contributes to worsen OA. All together these data give new insight into the ability of GPR40 to prevent or protect from OA and open new avenues to design innovative strategies for OA management especially by nutritional approaches.
ACKNOWLEDGMENTS
We thank Vincent Poitout (Centre de Recherche du Diabète de Montréal; Centre de Recherche du Centre Hospitalier de l’Université de Montréal (CRCHUM)) and AMGEN Inc (USA) for giving us the opportunity to work on GPR40−/− mice; Julien Hermet (INRA, UMR1019 - Unité Expérimentale de Nutrition, Clermont-Ferrand, France) for technical assistance in the GPR40−/− mouse facility. We are grateful to Chantal Bourget (INSERM 1026, Bordeaux, France) for her help and advice on zymography, and Eric Hay (INSERM 606, Paris, France) for his expertise on OARSI grading. We express gratitude towards Brigitte Gaillard (INRA, Unité de Microbiologie, Clermont-Ferrand, France) for the access to the microscopy platform. This work was supported by the French National Institute for Agricultural Research.
Author contributions
The present work was carried out in collaboration between all authors. LM and YW defined the research theme. CP provided GPR40 knock-out mice. LM designed methods and experiments, carried out the experiments, analyzed the data and wrote the paper. SM co-performed the experiments. YW and VC revised the article. All authors have approved the manuscript.
References
- 1.Felson DT, Zhang Y. An update on the epidemiology of knee and hip osteoarthritis with a view to prevention. Arthritis Rheumat 1998; 41: 1343–55. [DOI] [PubMed] [Google Scholar]
- 2.Loeser RF, Goldring SR, Scanzello CR, Goldring MB. Osteoarthritis: a disease of the joint as an organ. Arthritis Rheumat 2012; 64: 1697–707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Walker-Bone K, Javaid K, Arden N, Cooper C. Regular review: medical management of osteoarthritis. BMJ 2000; 321: 936–40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Loeser RF. Age-related changes in the musculoskeletal system and the development of osteoarthritis. Clin Geriatr Med 2010; 26: 371–86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Berenbaum F, Eymard F, Houard X. Osteoarthritis, inflammation and obesity. Curr Opin Rheumatol 2013; 25: 114–8. [DOI] [PubMed] [Google Scholar]
- 6.Jiang L, Tian W, Wang Y, Rong J, Bao C, Liu Y, Zhao Y, Wang C. Body mass index and susceptibility to knee osteoarthritis: a systematic review and meta-analysis. Joint Bone Spine 2012; 79: 291–7. [DOI] [PubMed] [Google Scholar]
- 7.Nuesch E, Dieppe P, Reichenbach S, Williams S, Iff S, Juni P. All cause and disease specific mortality in patients with knee or hip osteoarthritis: population based cohort study. BMJ 2011; 342: d1165–d1165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Villalvilla A, Gomez R, Largo R, Herrero-Beaumont G. Lipid transport and metabolism in healthy and osteoarthritic cartilage. Int J Mol Sci 2013; 14: 20793–808. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Calder PC. Polyunsaturated fatty acids, inflammation, and immunity. Lipids 2001; 36: 1007–24. [DOI] [PubMed] [Google Scholar]
- 10.Cho SH, Jung YB, Seong SC, Park HB, Byun KY, Lee DC, Song EK, Son JH. Clinical efficacy and safety of lyprinol, a patented extract from New Zealand green-lipped mussel (Perna Canaliculus) in patients with osteoarthritis of the hip and knee: a multicenter 2-month clinical trial. Eur Ann Allergy Clin Immunol 2003; 35: 212–6. [PubMed] [Google Scholar]
- 11.Knott L, Avery NC, Hollander AP, Tarlton JF. Regulation of osteoarthritis by omega-3 (n-3) polyunsaturated fatty acids in a naturally occurring model of disease. Osteoarthritis Cartilage 2011; 19: 1150–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Huang MJ, Wang L, Jin DD, Zhang ZM, Chen TY, Jia CH, Wang Y, Zhen XC, Huang B, Yan B, Chen YH, Li SF, Yang JC, Dai YF, Bai XC. Enhancement of the synthesis of n-3 PUFAs in fat-1 transgenic mice inhibits mTORC1 signalling and delays surgically induced osteoarthritis in comparison with wild-type mice. Ann Rheum Dis 2013; 73: 1719–27. [DOI] [PubMed] [Google Scholar]
- 13.Curtis CL, Hughes CE, Flannery CR, Little CB, Harwood JL, Caterson B. n-3 fatty acids specifically modulate catabolic factors involved in articular cartilage degradation. J Biol Chem 2000; 275: 721–4. [DOI] [PubMed] [Google Scholar]
- 14.Zainal Z, Longman AJ, Hurst S, Duggan K, Caterson B, Hughes CE, Harwood JL. Relative efficacies of omega-3 polyunsaturated fatty acids in reducing expression of key proteins in a model system for studying osteoarthritis. Osteoarthritis Cartilage 2009; 17: 896–905. [DOI] [PubMed] [Google Scholar]
- 15.Briscoe CP, Tadayyon M, Andrews JL, Benson WG, Chambers JK, Eilert MM, Ellis C, Elshourbagy NA, Goetz AS, Minnick DT, Murdock PR, Sauls HR, Jr, Shabon U, Spinage LD, Strum JC, Szekeres PG, Tan KB, Way JM, Ignar DM, Wilson S, Muir AI. The orphan G protein-coupled receptor GPR40 is activated by medium and long chain fatty acids. J Biol Chem 2003; 278: 11303–11. [DOI] [PubMed] [Google Scholar]
- 16.Itoh Y, Kawamata Y, Harada M, Kobayashi M, Fujii R, Fukusumi S, Ogi K, Hosoya M, Tanaka Y, Uejima H, Tanaka H, Maruyama M, Satoh R, Okubo S, Kizawa H, Komatsu H, Matsumura F, Noguchi Y, Shinohara T, Hinuma S, Fujisawa Y, Fujino M. Free fatty acids regulate insulin secretion from pancreatic beta cells through GPR40. Nature 2003; 422: 173–6. [DOI] [PubMed] [Google Scholar]
- 17.Kaplamadzhiev DB, Hisha H, Adachi Y, Ikehara S, Tonchev AB, Boneva NB, Pyko IV, Kikuchi M, Nakaya M, Wakayama T, Iseki S, Yamashima T. Bone marrow-derived stromal cells can express neuronal markers by DHA/GPR40 signaling. Biosci Trends 2010; 4: 119–29. [PubMed] [Google Scholar]
- 18.Kebede MA, Alquier T, Latour MG, Poitout V. Lipid receptors and islet function: therapeutic implications? Diabetes Obes Metab 2009; 11: 10–20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Wauquier F, Philippe C, Leotoing L, Mercier S, Davicco MJ, Lebecque P, Guicheux J, Pilet P, Miot-Noirault E, Poitout V, Alquier T, Coxam V, Wittrant Y. The free fatty acid receptor G protein-coupled receptor 40 (GPR40) protects from bone loss through inhibition of osteoclast differentiation. J Biol Chem 2013; 288: 6542–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Fernandes JC, Martel-Pelletier J, Pelletier JP. The role of cytokines in osteoarthritis pathophysiology. Biorheology 2002; 39: 237–46. [PubMed] [Google Scholar]
- 21.Latour MG, Alquier T, Oseid E, Tremblay C, Jetton TL, Luo J, Lin DC, Poitout V. GPR40 is necessary but not sufficient for fatty acid stimulation of insulin secretion in vivo. Diabetes 2007; 56: 1087–94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Gosset M, Berenbaum F, Thirion S, Jacques C. Primary culture and phenotyping of murine chondrocytes. Nature Protoc 2008; 3: 1253–60. [DOI] [PubMed] [Google Scholar]
- 23.Glasson SS, Blanchet TJ, Morris EA. The surgical destabilization of the medial meniscus (DMM) model of osteoarthritis in the 129/SvEv mouse. Osteoarthritis Cartilage 2007; 15: 1061–9. [DOI] [PubMed] [Google Scholar]
- 24.Kamekura S, Hoshi K, Shimoaka T, Chung U, Chikuda H, Yamada T, Uchida M, Ogata N, Seichi A, Nakamura K, Kawaguchi H. Osteoarthritis development in novel experimental mouse models induced by knee joint instability. Osteoarthritis Cartilage 2005; 13: 632–41. [DOI] [PubMed] [Google Scholar]
- 25.Glasson SS, Chambers MG, Van Den Berg WB, Little CB. The OARSI histopathology initiative - recommendations for histological assessments of osteoarthritis in the mouse. Osteoarthritis Cartilage 2010; 18: S17–23. [DOI] [PubMed] [Google Scholar]
- 26.Ahmad R, Sylvester J, Zafarullah M. MyD88, IRAK1 and TRAF6 knockdown in human chondrocytes inhibits interleukin-1-induced matrix metalloproteinase-13 gene expression and promoter activity by impairing MAP kinase activation. Cell Signall 2007; 19: 2549–57. [DOI] [PubMed] [Google Scholar]
- 27.Su SL, Tsai CD, Lee CH, Salter DM, Lee HS. Expression and regulation of Toll-like receptor 2 by IL-1beta and fibronectin fragments in human articular chondrocytes. Osteoarthritis Cartilage 2005; 13: 879–86. [DOI] [PubMed] [Google Scholar]
- 28.Oh DY, Talukdar S, Bae EJ, Imamura T, Morinaga H, Fan W, Li P, Lu WJ, Watkins SM, Olefsky JM. GPR120 is an omega-3 fatty acid receptor mediating potent anti-inflammatory and insulin-sensitizing effects. Cell 2010; 142: 687–98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Helminen HJ, Saamanen AM, Salminen H, Hyttinen MM. Transgenic mouse models for studying the role of cartilage macromolecules in osteoarthritis. Rheumatology (Oxford, England) 2002; 41: 848–56. [DOI] [PubMed] [Google Scholar]
- 30.Stoop R, van der Kraan PM, Buma P, Hollander AP, Billinghurst RC, Poole AR, van den Berg WB. Type II collagen degradation in spontaneous osteoarthritis in C57Bl/6 and BALB/c mice. Arthritis Rheumat 1999; 42: 2381–9. [DOI] [PubMed] [Google Scholar]
- 31.Loeser RF, Olex AL, McNulty MA, Carlson CS, Callahan MF, Ferguson CM, Chou J, Leng X, Fetrow JS. Microarray analysis reveals age-related differences in gene expression during the development of osteoarthritis in mice. Arthritis Rheumat 2012; 64: 705–17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.McNulty MA, Loeser RF, Davey C, Callahan MF, Ferguson CM, Carlson CS. Histopathology of naturally occurring and surgically induced osteoarthritis in mice. Osteoarthritis Cartilage 2012; 20: 949–56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Cooper C, Snow S, McAlindon TE, Kellingray S, Stuart B, Coggon D, Dieppe PA. Risk factors for the incidence and progression of radiographic knee osteoarthritis. Arthritis Rheumat 2000; 43: 995–1000. [DOI] [PubMed] [Google Scholar]
- 34.Guilak F. Biomechanical factors in osteoarthritis. Best Pract Res 2011; 25: 815–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Egloff C, Hugle T, Valderrabano V. Biomechanics and pathomechanisms of osteoarthritis. Swiss Med Wkly 2012; 142: w13583–w13583. [DOI] [PubMed] [Google Scholar]
- 36.Claes LE, Heigele CA. Magnitudes of local stress and strain along bony surfaces predict the course and type of fracture healing. J Biomech 1999; 32: 255–66. [DOI] [PubMed] [Google Scholar]
- 37.Martinez-Calatrava MJ, Prieto-Potin I, Roman-Blas JA, Tardio L, Largo R, Herrero-Beaumont G. RANKL synthesized by articular chondrocytes contributes to juxta-articular bone loss in chronic arthritis. Arthritis Res Ther 2012; 14: R149–R149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Curtis CL, Rees SG, Cramp J, Flannery CR, Hughes CE, Little CB, Williams R, Wilson C, Dent CM, Harwood JL, Caterson B. Effects of n-3 fatty acids on cartilage metabolism. Proc Nutr Soc 2002; 61: 381–9. [DOI] [PubMed] [Google Scholar]
- 39.Curtis CL, Rees SG, Little CB, Flannery CR, Hughes CE, Wilson C, Dent CM, Otterness IG, Harwood JL, Caterson B. Pathologic indicators of degradation and inflammation in human osteoarthritic cartilage are abrogated by exposure to n-3 fatty acids. Arthritis Rheumat 2002; 46: 1544–53. [DOI] [PubMed] [Google Scholar]
- 40.Bernstein P, Sticht C, Jacobi A, Liebers C, Manthey S, Stiehler M. Expression pattern differences between osteoarthritic chondrocytes and mesenchymal stem cells during chondrogenic differentiation. Osteoarthritis Cartilage 2010; 18: 1596–607. [DOI] [PubMed] [Google Scholar]
- 41.Dehne T, Karlsson C, Ringe J, Sittinger M, Lindahl A. Chondrogenic differentiation potential of osteoarthritic chondrocytes and their possible use in matrix-associated autologous chondrocyte transplantation. Arthritis Res Ther 2009; 11: R133–R133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Appleton CT, Pitelka V, Henry J, Beier F. Global analyses of gene expression in early experimental osteoarthritis. Arthritis Rheumat 2007; 56: 1854–68. [DOI] [PubMed] [Google Scholar]
- 43.Flajollet S, Tian TV, Huot L, Tomavo N, Flourens A, Holder-Espinasse M, et al. Increased adipogenesis in cultured embryonic chondrocytes and in adult bone marrow of dominant negative Erg transgenic mice. PloS one 2012;7:e48656. [DOI] [PMC free article] [PubMed]
- 44.Gabay O, Sanchez C, Salvat C, Chevy F, Breton M, Nourissat G, et al. Stigmasterol: a phytosterol with potential anti-osteoarthritic properties. Osteoarthritis and cartilage/OARS, Osteoarthritis Research Society 2010;18:106–16. [DOI] [PubMed]
- 45.Boudiffa M, Wade-Gueye NM, Guignandon A, Vanden-Bossche A, Sabido O, Aubin JE, et al. Bone sialoprotein deficiency impairs osteoclastogenesis and mineral resorption in vitro. J Bone Miner Res 2010;25:2669–79. [DOI] [PubMed]
- 46.Rozen S, Skaletsky H, J. Primer3 on the WWW for general users and for biologist programmers. In: Krawetz S, Misener S, editors. Bioinformatics Methods and Protocols: Methods in Molecular Biology. Totowa, NJ: Hummana Press, 2000, pp.365–86. [DOI] [PubMed]