Abstract
Gastrectomy (Gx) leads to osteopenia/osteoporosis in humans and animals. However, little is known about the influence of Gx on the cartilage in this regard. Recent studies have demonstrated a protective effect of 2-oxoglutaric acid (2-Ox) on bone and cartilage. Hence, the purpose of this study was to investigate whether 2-Ox can mitigate eventual Gx-induced cartilage impairment. Twenty female Sprague-Dawley rats were subjected to Gx and randomly divided into two groups: Gx + 2-Ox and Gx. Another 20 rats were sham-operated (ShO) and randomly divided into two groups: ShO + 2-Ox and ShO. The daily dose of 2-Ox administered to the rats in the drinking water was 0.43 g per 100 g rat. After eight weeks, rats were euthanized and femora and tibiae were collected. Histology and histomorphometry analyses of the articular cartilage and the growth plate were done. Gx resulted in a 32% (±44.5 femur, ±35.8 tibia) decrease in overall thickness of articular cartilage in both bones (femur: ShO 279.1 ± 48.5 vs. Gx 190.2 ± 38.4 µm, tibia: ShO 222.9 ± 50.3 µm vs. Gx 151.3 ± 52.6 µm) (in some zones up to 58 ± 28.0%), and in the growth plate up to 20% (±22.4) (femur: ShO 243.0 ± 34.0 vs. Gx 207.0 ± 33.7 µm, tibia: ShO 220.0 ± 24.6 µm vs. Gx 171.1 ± 16.1 µm). Gx altered the spatial distribution of thick and thin collagen fibers, and chondrocyte shape and size. 2-Ox administration prevented the reduction in both cartilages thickness (Gx + 2-Ox: articular cartilage 265.2 ± 53.8 µm, 235.6 ± 42.7 µm, growth plate 236.7 ± 39.2 µm, 191.3 ± 16.5 µm in femur and tibia, respectively), and abolished the spatial changes in collagen distribution and structure induced by Gx. Gx affects cartilage structure and thickness, however, 2-Ox administration mitigates these effects and showed protective and stimulatory properties. Our observations suggest that dietary 2-Ox can be used to offset some of the changes in hyaline cartilage, in particular articular cartilage, following bariatric surgeries.
Keywords: Histology, histomorphometry, gastrectomy, skeleton, development, 2-oxoglutaric acid
Introduction
Bariatric surgery is an efficient method of treatment for severely obese patients, patients with gastric cancer, trauma or complicated peptic ulcer disease. There is, however, a difference between the consequences of gastrectomy performed for an ulcer or a tumor, where large part of the stomach can be removed, and other types of bariatric surgeries, which are generally classified as restrictive procedures (laparoscopic adjustable gastric banding, sleeve gastrectomy) or restrictive with malabsorption procedures (Roux-en-Y gastric bypass, biliopancreatic diversion with a duodenal switch). Despite these differences, bariatric surgery patients are at risk of further systemic complications such as altered calcium homeostasis, altered vitamin D metabolism and absorption, increased bone turnover, and bone loss rate.1–4 Bone loss correlates with weight loss, however, the degree of bone loss after bariatric surgery is substantially higher.2,3
From the variety of bariatric surgery types, the gastrectomy (Gx) was chosen as a model for this study, since the effect of gastrectomy, on bone tissue is well defined and commonly results in the loss of trabecular and cortical bone which is associated with osteopenia in humans2–4 and experimental animals, regardless of gender.5–7 As a consequence of bone impairment, a high incidence of fractures of the hip and vertebrae is observed in human studies.8–10 Although the effects of Gx on the mineralization, morphology, and mechanical properties of bone have been well described, the mechanisms of Gx-evoked bone degeneration are still not fully understood.2–5,11–13 While possibilities such as the loss of particular stomach endocrine functions, general malnutrition or the lack of intrinsic nutritional elements promoting an imbalance in bone formation, and resorption leading to a loss in bone tissue are proposed, investigation into these proposed mechanisms is still in progress.2,6,11–15
With respect to the effects of Gx on bone, there is a dearth of information on the influence of this type of surgery on the cartilage in particular. One of the crucial and integral elements of long bones is the articular cartilage and growth plate. Since the articular cartilage allows for mobility within the skeleton and the growth plate enables its appropriate growth, the recognized influence of Gx on bone tissue may not be the only cause of skeletal malfunctions long term. Moreover, articular cartilage does not heal spontaneously under physiological circumstances due to natural avascularity and low cell turnover, which limits its capacity to self-regenerate.16,17
Despite recent advances in medicine, the treatment of cartilage lesions remains a complex problem, involving sophisticated techniques.18 Furthermore, cartilage lesions can lead to progression of osteoarthritis, the most common musculoskeletal disease which will become one of the leading causes of disability in developed ageing societies by the year 2020.16,19
Additionally, a growing body of evidence highlights a worldwide obesity epidemic in all age groups, specifically in children and young people.20 Although bariatric surgery is one of the effective and endorsed forms of treatment for patients who are overweight or obese, questions regarding the long-term safety of this procedure in severely obese children and adolescents have been raised.1 This was also the reason for us, in the present study, to choose of young/adolescent animals model, when the intensive growth and maturation of the skeleton occur.
It has been previously shown that 2-oxoglutaric acid (α-ketoglutaric acid) (2-Ox) administration reduces the changes evoked by Gx in bone tissue.7 Earlier studies showed that dietary 2-Ox counteracts the bone loss observed in ovariectomized rats21 and in post-menopausal women.22 Furthermore, recent studies displayed a protective effect of 2-Ox on long bones of fundectomized pigs.23 Most probably these observed effects were due to the role of 2-Ox derivatives such as proline, leucine, glutamine, glutamate, and other amino acids which are synthesized in the gastrointestinal tract from 2-Ox.24 Thus dietary 2-Ox may be an important factor for the improvement of protein anabolism, especially collagen anabolism. Pro-collagen proline in bone is converted into collagen hydroxyproline, the most abundant amino acid in bone collagen, in the presence of vitamin C and Fe2. Furthermore, 2-Ox, together with Fe2 have been proposed to be active participants in the conversion of proline to hydroxyproline. Moreover, 2-Ox acts as a cofactor for Fe2 absorption from the intestine.22,24,25
In earlier studies it was strongly hypostatized that impaired synthesis of collagen may contribute to the Gx-evoked osteopenia since the collagen matrix of the bone, trabecular structure of the bone in particular, was impaired by gastrectomy.7,22,25 Moreover, changes in bone metabolism markers related do osteoblast metabolism, and in turn to collagen synthesis, were observed in partial gastrectomy in pigs.23
Since impaired synthesis of collagen may contribute to the Gx-evoked osteopenia and hydroxyproline and proline are the main components of collagen, it is possible that 2-Ox may play an essential role in the development and preservation of cartilage and bone integrity.
Considering the above the purpose of this study was to determine if Gx leads to cartilage impairment in rats after two months and to investigate whether 2-Ox administration can mitigate the eventual Gx-induced cartilage impairment.
Materials and methods
Since the present study was a continuation of a former experiment, the description of all animal handling, feeding, and surgery procedures conducted during the study are the same as those featured in an earlier publication.7 Briefly, 40 (10 weeks old, 212.5 ± 2.5 g) female Sprague-Dawley rats were randomly and equally divided into four experimental groups. Twenty rats were sham operated (a midline abdominal incision, manipulation/handling of the stomach, without any change to the organ, and closure of the incision—ShO) and then further divided into a group receiving 2-Ox (ShO + 2-Ox) and one without 2-Ox (ShO). Another 20 rats were gastrectomized (glandular portion of the stomach, i.e. the acid-producing part and the pyloric antrum were resected and the remaining non-glandular part of the fore stomach was joined end-to-end with the duodenum—Gx) and then further divided into a group receiving 2-Ox (Gx + 2-Ox) and one without 2-Ox (Gx). Rats had free access to feed (standard rat feed pellets; Lactamin, Vadstena, Sweden) and water, controlled twice daily. The sodium salt of 2-Ox was administered at a dose of 14.6 g/L, in the drinking water. The average daily intake of 2-Ox was 0.43 g/100 g rat, which was calculated from water consumption and body weight. The rats were weighed before surgery and once every week after surgery.7 The study lasted for eight weeks to enclose the periods of adolescence and skeletal maturity, thus, to examine the active and not ossified growth plate where all morphological features of the growth plate, for growing animals, are maintained.25
To compensate for the loss of the intrinsic factor essential for absorption of vitamin B12 and for the poor absorption of iron due to the lack of gastric acid, the Gx rats received vitamin B12 (0.4 mg/kg Betolvex® 1 mg/mL, Dumex, Copenhagen, Denmark) and Fe3+ (20 mg/kg of ferric hydroxide polymaltose, Ferrum®, Vifor, St. Gallen, Switzerland) as previously described.11 Eight of the 40 rats died during surgery or shortly thereafter. The final distribution of rats in the groups was as follows: n = 7 in the ShO, n = 10 in the ShO + 2-Ox, n = 8 in the Gx, and n = 7 in the Gx + 2-Ox group. At the end of the study all rats were euthanized by exsanguination from the abdominal aorta under anesthesia, as previously described.7
The experiment was approved by the local Animal Welfare Committee, Lund, Sweden.
Tissue collection and analysis
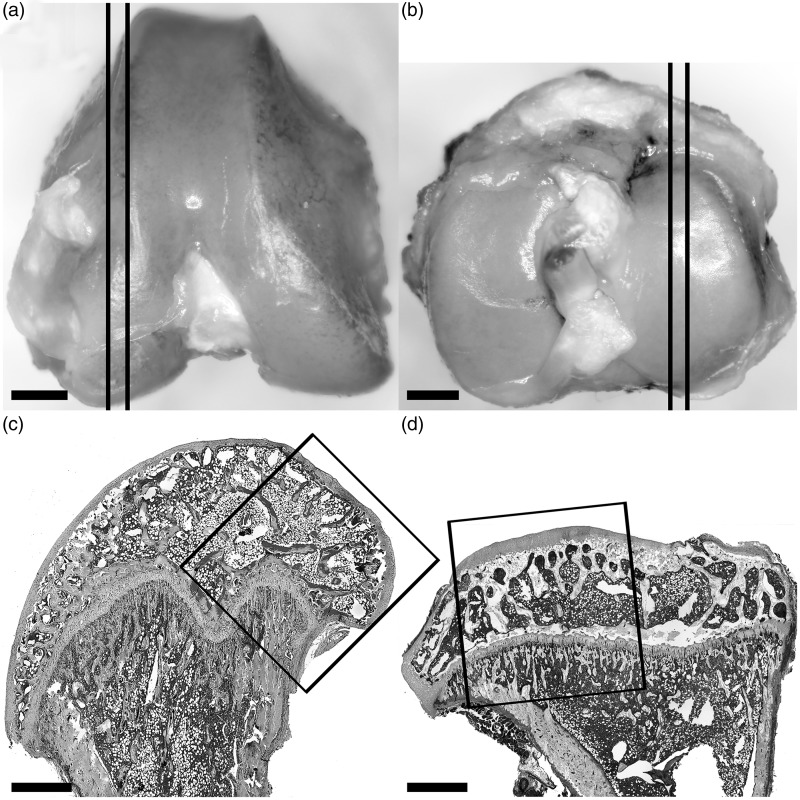
The left femora and tibiae were subjected to histology as previously described,7 except sections were cut at 4 µm thick and were stained using three methods: Goldner’s trichrome staining (GT) to assess the morphology of the growth plate and to distinguish it from the bone tissue as well as to cell counting purposes of both studied cartilages; picrosirus red staining (PSR) to assess the morphology of the articular cartilage and to evaluate the distribution of thick and thin collagen fibers of articular cartilage; and safranin O staining (SO) to assess the content and distribution of proteoglycans in articular cartilage.26,27 Moreover, an additional set of sections were not stained and were used for autofluorescence measurements purposes.7,25 Briefly, 56 sagittal sections, through the middle of the lateral condyle, for each femur and tibia (at 20 µm intervals after each series of 7 sections) were cut from each individual rat. Total thickness of section plane was 364 µm for each bone (total 112 sections per animal). Fourteen sections for each staining (with 164 µm interval between each 7 sections) were used. The site and size (approximately 3 mm in length of both analyzed cartilages) of the areas of interest, that were measured, were chosen on the basis of motoric properties of rat body, the knee joint in particular, and are shown in Figure 1. These selected sites are most laden by the weight of the body. Microscopic (two-dimensional) images of autofluorescence (magnification x200) and bright field (magnification x200 and x400) were collected using a confocal microscope (AXIOVERT 200 M, Carl Zeiss, Jena, Germany) equipped with a color digital camera (AxioCam HRc, Carl Zeiss, Jena, Germany), a halogen lamp, and a fluorescent lamp (excitation wavelength 450–490 nm). Moreover, sections stained with PSR were analyzed using an automated microscope (Olympus BX63, Tokyo, Japan) (magnification x200) equipped with filters to provide circularly polarized illumination (filters were aligned so that the background in the field of view was as dark as possible, that is, the filters were “crossed”) as previously described.27 Images were collected with a digital color camera (UC50 Olympus, Tokyo, Japan). To equally cover the regions of interest on both analyzed cartilages of both bones, 10 pictures (magnification x200) per slice and per staining were made from every animal. Pictures were taken on every third slice from articular cartilage and growth plate. For the purpose of cell measurements pictures of magnification x400 was used.
Figure 1.
The location selection and the size of areas of interest analyzed in individual bones. a: femoral distal epiphysis, b: tibial proximal epiphysis, c: femoral sagittal section, and d: tibial sagittal section. The site of sections are marked by black parallel lines and the distance between lines is approximately 360 µm. Square boxes indicate regions of interest in cartilage analysis. Scale bars: 1 mm
Structure of the growth plate and articular cartilage was examined by microscopic observation and the images collected were analyzed using graphical analysis software, ImageJ 1.48 g (National Institute of Health USA, http://rsb.info.nih.gov/ij/index.html). All measurements were completed by two histologists who were blinded to treatment groups.
Measurements of growth plate width as the mean separation between boundaries of specified zones (the resting zone—between the upper border of the growth cartilage and the top of the first cell in the columns; proliferative zone—flat chondrocytes, where cells were in columns, as far down as the first cells showing a larger, more rounded shape; hypertrophy zone—where cells were larger and more rounded than proliferative cells and where the ratio of cell width to cell height becomes less than 2; calcified zone—between the mineralization front and the top of the first open lacuna, above where the matrix was mineralized and the cells began to show signs of apoptosis) were performed as previously described.25,28,29 Four zones of articular cartilage were defined and their width was measured (the superficial zone—articulating surface, with densely packed collagen fibrils and elongated chondrocytes, both parallel to the articular surface; the intermediate zone—with loosely packed, thicker collagen fibrils, aligned obliquely to the surface, and more rounded chondrocytes than in the superficial layer; the radial zone—with large diameter collagen fibrils, oriented perpendicular to the articular surface, the chondrocytes typically arranged in columnar fashion parallel to the collagen fibers and perpendicular to the joint line; the calcified zone—which rests directly on the subchondral bone) as previously described.29–31 The depth of the tidemark, which separates the superficial, intermediate, and radial zones from the calcified cartilage, was also measured.29,31
The quantity and chondrocyte dimensions, for growth plate and articular cartilage, were examined as follows: the number of chondrocytes/mm2 of hyaline cartilage, chondrocyte sectional area, mean and maximum diameter, and perimeter. Six thousand cells per animal were counted.
To assess the spatial distribution of thick and thin collagen fibers of articular cartilage the software’s color threshold function was applied to the images (taken in polarized light) of articular cartilage sections (stained in PSR) to isolate fibers of different color and hue. Such isolation facilitated assessment of the spatial distribution of various fiber populations. Subsequent software pixel counting function was used, and the amount of thin collagen fibers was calculated as a percent of cartilage sectional surface.27 Collagen content was calculated as a percentage of the area of each image (expressed in pixels).
The mean intensity of SO staining (measured in 8-bit grey value which showed an inverse correlation—the higher the result, the lower the amount of stain absorbed) was measured for proteoglycan content of the intercellular matrix of the cartilages examined. Moreover, visual assessment of proteoglycan distribution was done and Mankin’s histological and histochemical grading system for evaluation of articular cartilage was applied.32
Additionally, the mean intensity of intercellular matrix autofluorescence (measured in 8-bit grey value which showed a proportional correlation—the higher the result, the stronger the tissue autofluorescence) was measured for articular cartilage, since autofluorescence is related to the hydroxyproline content which in turn reflects the collagen content as shown by Hagiwara et al.33
Statistical analysis
All results are expressed as means ± SD or percentage of difference between means ± SD. Differences between means were tested by the use of a nested analysis of variance (ANOVA)34 and a post hoc Tukey’s unequal N honest significant difference (HSD) test as a correction for multiple comparisons, planed comparisons tests were used as well. A Nested ANOVA was used as the rats were paired as previously described.7 Normal distribution of the data was examined using the W Shapiro–Wilk test and equality of variance was tested by the Brown–Forsythe tests. When data were not normally distributed and/or there was an unequal variance of data, we made use of the Mann-Whitney U test with a Bonferroni correction for multiple comparisons, to analyze differences between means. P < 0.05 was considered statistically significant. All statistical analyses were carried out using STATISTICA 8.0 software (StatSoft, Inc. [2008]. STATISTICA [data analysis software system], version 8.0 www.statsoft.com).
Results
Articular cartilage and growth plate thickness
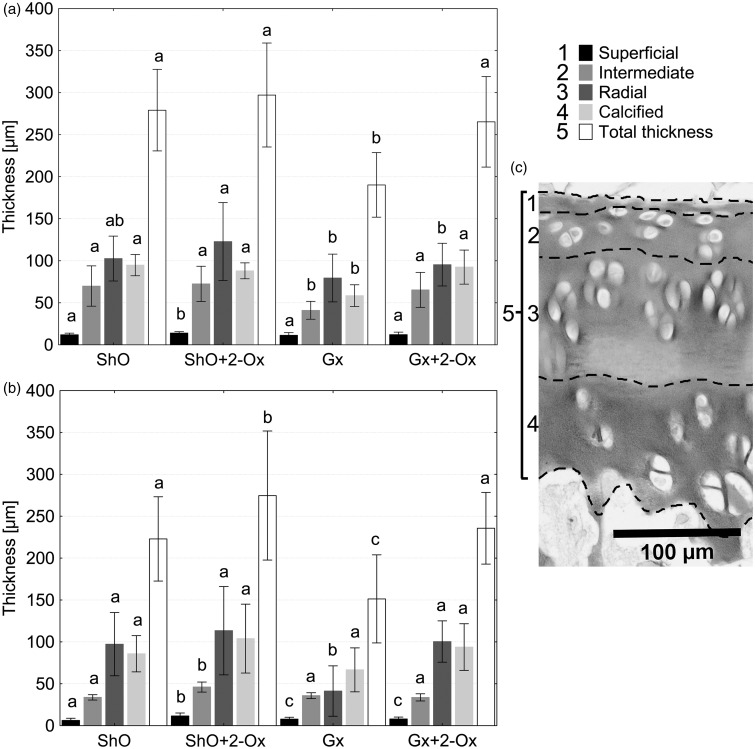
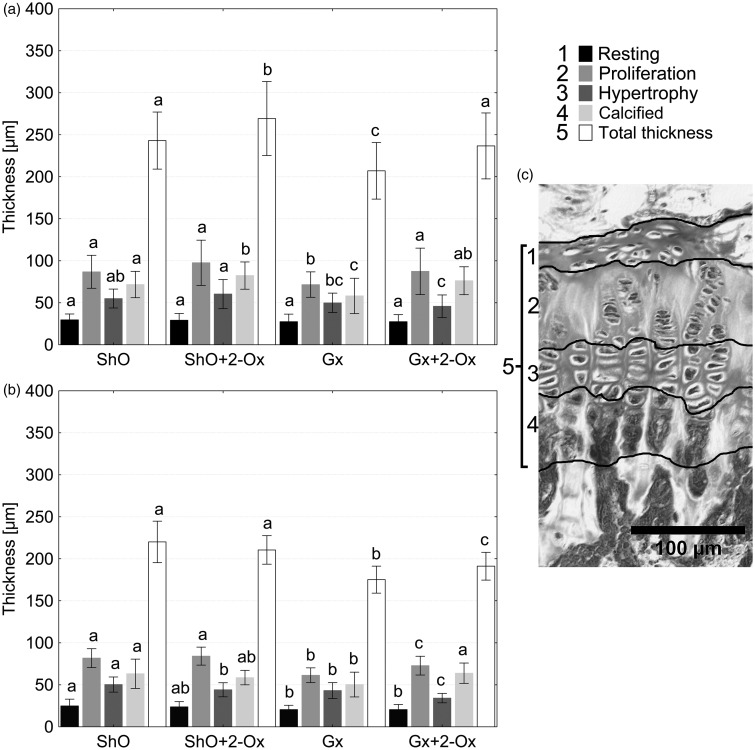
Articular cartilage and growth plate thickness are presented in Figures 2 and 3, respectively. Gastrectomy evoked a 32% decrease in total thickness of femoral (±44.5) and tibial (±35.8) articular cartilage (P < 0.001), and a 15% (±18.0) decrease in femoral and 20% (±22.4) in tibial growth plate (P < 0.001) in rats from the Gx group compared to ShO rats. Surprisingly, tibial superficial zone of articular cartilage was 24% (±0.7), significantly (P = 0.016) and tibial intermediate zone 6% (±1.0) not significantly (P = 0.274) thicker in Gx rats, whereas tibial resting zone of the growth plate was significantly (18 ± 2.2%, P = 0.049) thinner in this group compared to ShO rats. Gastrectomy did not exert any effect on superficial (articular cartilage) or resting (growth plate) zone in femur. The thickness of other three zones, both in the articular cartilage (except tibial intermediate) and in the growth plate, were decreased in rats from the Gx group compared to the control. Most pronounced changes were observed in femoral intermediate (42 ± 14.5%, P < 0.001) and calcified (38 ± 18.1%, P < 0.001) zones of articular cartilage and in proliferation (18 ± 7.6%, P = 0.006) and calcified (19 ± 6.8%, P = 0.026) zones of the growth plate. Regarding the tibia, significant decreases were observed in radial zone of articular cartilage (58 ± 28.0%, P < 0.001) and in proliferation (25 ± 10.2%, P < 0.001), hypertrophy (15 ± 3.5%, P = 0.016) and calcified (21 ± 6.5%, P = 0.006) zones of the growth plate. Although the thickness of femoral radial (22 ± 11.6% decrease) and tibial calcified (22 ± 9.6% decrease) zones of articular cartilage as well as femoral hypertrophy (9 ± 2.6% decrease) zone of the growth plate were lower in Gx rats than in the ShO group, these changes were not significant.
Figure 2.
The effect of gastrectomy (Gx) and dietary 2-oxoglutaric acid (2-Ox) administration on the thickness of femoral (a) and tibial (b) articular cartilage of female rats. Appropriate zones of articular cartilage are indicated by different shades of gray scale and marked by segmented lines on cartilage section on the sample photograph (c). Different letters above the columns of the same color indicate differences between the groups with P < 0.05. ShO: sham operated rats. The 2-oxoglutaric acid sodium salt (0.43 g/100 g rat per day) was administered in the drinking water. Bars represent the mean, whiskers represent standard deviation
Figure 3.
The effect of gastrectomy (Gx) and dietary 2-oxoglutaric acid (2-Ox) administration on the thickness of femoral (a) and tibial (b) growth plate cartilage of female rats. Appropriate zones of the growth plate are indicated by different shades of gray scale and marked by segmented lines on cartilage section on the sample photograph (c). Different letters above the columns of the same color indicate differences between the groups with P < 0.05. ShO: sham operated rats. The 2-oxoglutaric acid sodium salt (0.43 g/100 g rat per day) was administered in the drinking water. Bars represent the mean, whiskers represent standard deviation
Dietary 2-Ox administration improved articular cartilage and growth plate thickness in both bones of gastrectomized rats (Figures 2 and 3). The total thickness of femoral and tibial articular cartilage as well as femoral growth plate in the Gx + 2-Ox group was significantly higher (P < 0.001, P < 0.001 and P = 0.001, respectively) than in Gx rats, reaching the level of control rats from the ShO group. The total thickness of tibial growth plate in Gx + 2-Ox rats was also higher (10 ± 7.9%, P = 0.012) than in Gx rats, however not as high as in the control group. Considering zones of articular cartilages, the treatment with 2-Ox increased the thickness of intermediate (59 ± 12.2%, P = 0.001) and calcified (58 ± 17.0%, P < 0.001) zones of femoral articular cartilage, and radial (143 ± 29.6%, P < 0.001) zone of tibial articular cartilage in the Gx + 2-Ox group when compared to Gx rats. Accordingly, proliferation and calcified zones of the growth plate in femur (22 ± 7.9%, P = 0.018 and 31 ± 9.0%, P < 0.001, respectively) and tibia (19 ± 5.7%, P < 0.001 and 27 ± 6.8%, P < 0.001, respectively) were thicker in Gx + 2-Ox rats compared with the Gx group. Although, the thickness of hypertrophy zone in tibial growth plate was thinner (21 ± 4.5%, P < 0.001) in Gx + 2-Ox rats than in the Gx group.
The 2-Ox administered to sham operated rats moderately stimulated hyaline cartilage comparing with the ShO group. The total thickness of articular cartilage was elevated both in femur (6 ± 9.0%) and tibia (23 ± 25.9%), however this change was significant only in tibia (P = 0.004). Conversely, the femoral growth plate was thicker in the ShO + 2-Ox group (11 ± 13.1%, P = 0.027) comparing with ShO rats, whereas tibial growth plate was insignificantly thinner (4 ± 4.8%) in sham operated rats receiving 2-Ox than in the ShO group. Interestingly the hypertrophy zone of tibial growth plate in the ShO + 2-Ox group was (12 ± 3.1%, P = 0.036) thinner compared with the ShO group.
Gastrectomy significantly decreased the depth of the tidemark of femoral articular cartilage in the Gx group (131.7 ± 34.0 µm) comparing with ShO rats (184.4 ± 40.7 µm) (P = 0.003). Tibial articular cartilage was similarly changed (Gx rats 84.7 ± 32.7 µm; ShO rats 137.1 ± 41.1 µm) (P < 0.001). Treatment with 2-Ox abolished those changes. Rats from the Gx + 2-Ox group had thicker tissue above the tidemark in femur (172.8 ± 44.6 µm) and tibia (141.9 ± 25.4 µm) than rats from Gx group (P = 0.028 and P < 0.001, respectively). Moreover, sham operated rats receiving 2-Ox had thicker cartilage above the tidemark in tibial articular cartilage (170.8 ± 58.2 µm) than animals in the ShO group (137.1 ± 41.1 µm) (P = 0.011).
Quantitative and qualitative evaluation of growth plate and articular cartilage chondrocytes
The effect of Gx and dietary 2-Ox administration on the number and morphological properties of the growth plate and articular cartilage chondrocytes of the femur and tibia are presented in Tables 1 and 2. Gx evoked a 6–8% reduction in chondrocyte number in both bones and cartilage types examined, compared to the ShO group (P = 0.093 for femoral and P = 0.079 for tibial growth plate, P = 0.23 for femoral and P = 0.96 for tibial articular cartilage). On the other hand, 2-Ox administration increased the number of chondrocytes in femoral and tibial growth plate and in femoral articular cartilage of Gx rats by 20%, 17%, and 20%, respectively, compared to ShO rats. This increase was statistically insignificant due to a high SD. Comparison of the same particular sites between Gx + 2-Ox and Gx groups showed a substantial enhancement of chondrocyte number evoked by dietary 2-Ox administration, by 27% for both the femoral and tibial growth plate, and 28% for femoral articular cartilage (P = 0.018, P = 0.015 and P = 0.049, respectively). Interestingly, 2-Ox administered to sham operated rats decreased chondrocyte number in femoral articular cartilage by 27% (P = 0.045), and by 13% in the tibial growth plate (P = 0.57) compared to the ShO group.
Table 1.
The effect of gastrectomy (Gx) and dietary 2-oxoglutaric acid (2-Ox) administration on the quantity and dimensions of growth plate cartilage chondrocytes of female rats
| ShO |
ShO + 2-Ox |
Gx |
Gx + 2-Ox |
|||||
|---|---|---|---|---|---|---|---|---|
| Parameter | Mean | SD | Mean | SD | Mean | SD | Mean | SD |
| Femur | ||||||||
| Cell number/mm2 of cartilage | 1853ab | 218 | 1835ab | 332 | 1750a | 364 | 2220b | 489 |
| Cell sectional area (µm2) | 33.9a | 15.6 | 30.2b | 12.0 | 33.3a | 15.0 | 35.8c | 17.4 |
| Mean diameter (µm) | 7.9a | 1.8 | 7.4b | 1.6 | 7.7c | 1.9 | 8.1a | 2.3 |
| Maximum diameter (µm) | 9.1a | 2.1 | 8.4b | 1.9 | 8.8c | 2.3 | 9.2a | 2.6 |
| Perimeter (µm) | 23.7a | 6.0 | 22.2b | 6.1 | 23.2a | 6.6 | 24.5c | 8.2 |
| Tibia | ||||||||
| Cell number/mm2 of cartilage | 2115ab | 340 | 1844a | 410 | 1946a | 328 | 2473b | 470 |
| Cell sectional area (µm2) | 31.7a | 13.2 | 31.4a | 13.6 | 31.1a | 14.6 | 33.5a | 31.2 |
| Mean diameter (µm) | 8.5ac | 2.0 | 8.2ab | 2.0 | 8.2b | 1.9 | 8.7c | 2.5 |
| Maximum diameter (µm) | 9.9a | 2.3 | 9.5b | 2.3 | 9.5b | 2.2 | 10.1a | 2.8 |
| Perimeter (µm) | 24.8ab | 6.7 | 24.2a | 7.6 | 24.3a | 7.7 | 25.7b | 10.1 |
ShO: sham operated rats; the 2-oxoglutaric acid sodium salt (0.43 g/100 g rat per day) was administered in the drinking water. Data are expressed as mean ± SD. Superscript letters indicate significant differences between groups at P < 0.05.
Table 2.
The effect of gastrectomy (Gx) and dietary 2-oxoglutaric acid (2-Ox) administration on the quantity and dimensions of articular cartilage chondrocytes of female rats
| ShO |
ShO + 2-Ox |
Gx |
Gx + 2-Ox |
|||||
|---|---|---|---|---|---|---|---|---|
| Parameter | Mean | SD | Mean | SD | Mean | SD | Mean | SD |
| Femur | ||||||||
| Cell number/mm2 of cartilage | 1614a | 303 | 1180b | 275 | 1510ab | 340 | 1936c | 398 |
| Cell sectional area (µm2) | 69.2a | 18.4 | 74.5a | 21.6 | 68.5a | 23.6 | 70.7a | 21.0 |
| Mean diameter (µm) | 9.9a | 1.3 | 10.2a | 1.4 | 9.8a | 1.6 | 10.0a | 1.4 |
| Maximum diameter (µm) | 11.6a | 1.7 | 11.9a | 1.6 | 11.3a | 1.9 | 11.4a | 1.7 |
| Perimeter (µm) | 32.1a | 4.6 | 33.2a | 4.9 | 32.1a | 5.4 | 32.6a | 4.6 |
| Tibia | ||||||||
| Cell number/mm2 of cartilage | 1365a | 391 | 1280a | 238 | 1287a | 134 | 1293a | 134 |
| Cell sectional area (µm2) | 88.6ac | 28.1 | 78.6ab | 22.4 | 71.9b | 29.5 | 94.8c | 28.8 |
| Mean diameter (µm) | 11.1a | 1.7 | 10.5b | 1.5 | 10.0b | 1.9 | 11.6c | 1.7 |
| Maximum diameter (µm) | 12.9a | 2.1 | 12.1b | 1.7 | 11.7b | 2.3 | 13.4a | 2.0 |
| Perimeter (µm) | 36.2ac | 6.1 | 34.6a | 5.1 | 32.8b | 6.5 | 37.8c | 5.6 |
ShO: sham operated rats; the 2-oxoglutaric acid sodium salt (0.43 g/100 g rat per day) was administered in the drinking water. Data are expressed as mean ± SD. Superscript letters indicate significant differences between groups at P < 0.05.
Gx had no effect on the chondrocyte sectional area, except for the tibial articular cartilage where a 19% decrease was observed compared to the ShO group (P < 0.001). This effect was reversed by the 2-Ox administration in the Gx + 2-Ox group. Gx rats receiving 2-Ox had an increased cell sectional area in the femoral growth plate by over 15% compared to other groups (P < 0.001 for all comparisons). Nonetheless, 2-Ox administration decreased cell sectional area in the femoral growth plate of sham operated rats by over 10% (P < 0.001 for all comparisons).
The mean and maximum diameter of chondrocytes were decreased by Gx in femoral and tibial growth plates and tibial articular cartilage compared to the ShO group (P < 0.001 for all comparisons). There were no significant differences in femoral articular cartilage in this regard. 2-Ox generally abolished the influence of Gx on these two parameters in the Gx + 2-Ox group compared to the Gx group. However, sham operated rats receiving 2-Ox had significantly shorter chondrocyte maximal diameter in femoral and tibial growth plates and tibial articular cartilage than rats from the ShO group (P < 0.001 for all comparisons). Also, the mean diameter of chondrocytes in the femoral growth plate was lowered by 2-Ox (P < 0.001) compared to the ShO group.
Gx caused a decrease in chondrocyte perimeter in tibial articular cartilage (P < 0.001), showing no significant effect in the other sites examined, compared to the ShO group. The 2-Ox abolished the influence of Gx in the Gx + 2-Ox group, in this regard.
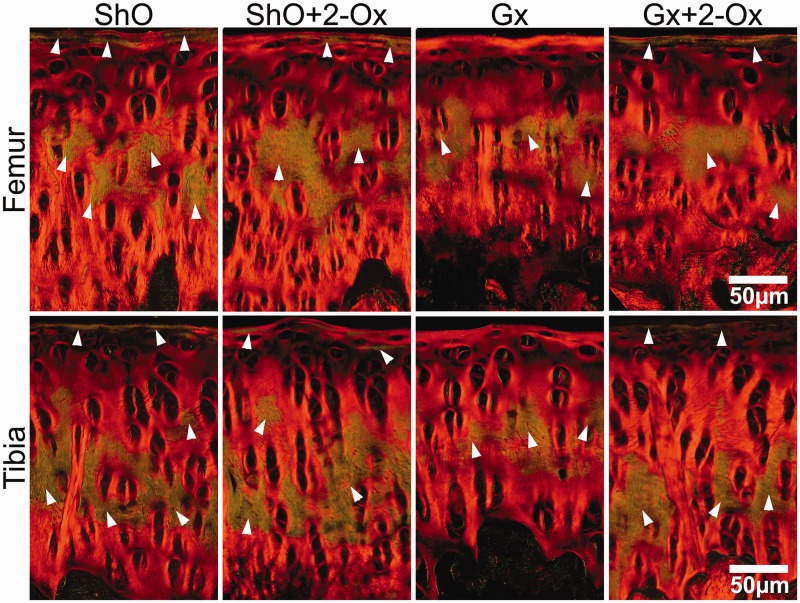
The distribution of thick and thin collagen fibers of articular cartilage and matrix autofluorescence
The PSR staining method and polarized light allowed us to distinguish between the larger collagen fibers (orange to red color), and the thinner ones, including reticular fibers (green color). The effect of Gx and dietary 2-Ox on the distribution of thick and thin collagen fibers of femoral and tibial articular cartilage is presented in Figure 4. The distribution of thin and thick collagen fibers was heterogeneous in both bones examined. Majority of thin fibers were present in the superficial and radial zone of femoral and tibial articular cartilage. Gastrectomy exerted its effects mostly within the superficial zone of cartilage in both bones, where thin fibers were almost absent compared to the ShO group. However, reduction of thin collagen fibers, observed in radial zone of gastrectomized rats (11% femur, 9% tibia), was much less pronounced compared to the ShO group (P = 0.048 and P = 0.043, respectively). Administration of 2-Ox reversed the effect of Gx in the superficial zone of articular cartilage in both bones examined. The amount of thin collagen fibers, in the radial zone, increased significantly in the Gx + 2-Ox group only in tibia (17%, P = 0.011), whereas in femur the improvement was less pronounced (7%, P = 0.059), compared with Gx rats. Moreover, there was 14% less thin collagen fibers in the superficial zone of rats from the ShO + 2-Ox group than in the control (P = 0.037), and no significant differences in radial zone between these groups, with respect to sectional surface of these particular zones.
Figure 4.
The effect of gastrectomy (Gx) and dietary 2-oxoglutaric acid (2-Ox) administration on the distribution of thick and thin collagen fibers of femoral and tibial articular cartilage of female rats. Representative samples of the microscopic images collected in polarized light. Magnification for each image in the row indicated by white scale bar. The larger collagen fibers are orange to red, and the thinner ones, including reticular fibers, are green. The surface of the cartilage is directed toward the top of every sample picture. White arrowheads indicate green, thin collagen fibers. ShO: sham operated rats. The 2-oxoglutaric acid sodium salt (0.43 g/100 g rat per day) was administered in the drinking water. (A color version of this figure is available in the online journal.)
Collagen/hydroxyproline content, measured as intercellular matrix autofluorescence, of femoral articular cartilage increased in ShO + 2-Ox (136.5 ± 20.7) and Gx + 2-Ox (142.9 ± 12.0) groups compared to the ShO (89.0 ± 13.6) and Gx groups (94.7 ± 22.8) (P < 0.001 and P < 0.001). However, in tibial articular cartilage, this parameter was increased only in the Gx + 2-Ox group (142.9 ± 17.1) compared to the ShO (112.5 ± 27.4), ShO + 2-Ox (111.6 ± 33.0), and Gx groups (110.7 ± 14.1) (P = 0.039, P = 0.031, P = 0.026, respectively).
The content and distribution of proteoglycans in growth plate and articular cartilage
There were no visible degenerative changes or damage to the articular cartilage and no differences in Mankin’s score between all groups. There was no gradient in SO staining. However, rats from the Gx + 2-Ox group had a significantly brighter SO staining (mean intensity 123.4 ± 8.0 of 8-bit gray scale) in femoral articular cartilage than the other groups (111.2 ± 4.6 for ShO, 110.8 ± 7.2 for ShO + 2-Ox, 110.0 ± 9.1 for Gx) (P = 0.001), and there were no significant differences among these groups as well as in tibial articular cartilage (113.7 ± 19.1 for ShO, 109.5 ± 11.1 for ShO + 2-Ox, 105.7 ± 13.8 for Gx, 109.6 ± 10.2 for Gx + 2-Ox). There were no differences in proteoglycan content and distribution in growth plate cartilage.
Discussion
The role of the stomach in bone maintenance and remodeling has been proven in many earlier studies on humans and laboratory animals, where the development of osteopenia/osteoporosis is evoked by partial or total gastrectomy.2,7,9,10,12,13,23,35,36 However, little is known about the effects of Gx on the cartilage in this regard.
Hyaline cartilage is a heterogeneous tissue which is involved in the creation of most bones during early development, enabling the longitudinal growth of bones and forming the bearing surfaces of all synovial joints, thereby largely contributing to the extraordinary functional capacities of bone. Articular cartilage has great durability and, unfortunately, a limited ability to maintain and repair itself.37 Hence, based on the data available on Gx and the related risk of bone loss and fractures, as previously mentioned, the present study examined the effect of this particular type of surgery on the morphological properties of the growth plate and articular cartilage in a rat gastrectomy model. Since previous studies have shown a protective effect of 2-Ox administration on bone loss after gastrectomy, fundectomy, antrectomy, ovariectomy, and in post-menopausal women,7,21–23,35,38 it was assumed that some of the gastrectomy-induced changes within cartilage could also be mitigated or even abolished by 2-Ox administration. Bearing in mind results from recent studies involving growth regulation, prenatal programming, and glucocorticoids or proton pump inhibitor therapies, all related to the growth, maintenance, and metabolism of bone and cartilage,25,39–41 we felt it was necessary and important to elucidate the protective and stimulatory potential of 2-Ox on cartilage in this regard.
Results obtained have shown that Gx affects mainly cartilage thickness, cells morphology, and collagens distribution and synthesis. However, no Gx-induced effects on bone length have previously been observed.36,42,43 Direct measurement of cartilage thickness showed a decrease in the total as well as in particular zones thickness, in both examined cartilages, evoked by Gx, which was abolished by 2-Ox administration. Thus, since the height of the hypertrophy zone reflects the rate of bone growth,28 it may be possible that Gx affects not only bone resorption, as concluded by Mühlbauer et al.,42 but also bone formation, since both the proliferative and hypertrophy zones were thinner in the femoral and tibial growth plate of Gx rats in the present study. Nevertheless, independently of performed surgery, 2-Ox probably shortened the time of chondrocyte passage through the hypertrophy zone and, at the same time, stimulated proliferation. Such hypothesis could be the possible explanation for observed changes in chondrocyte number and their dimensions, as well as differences in the thickness of hypertrophy and proliferative zones, especially in stress circumstances and surplus of energy source. Since this observation differs from recent study on non-operated, adolescent rats,25 where there was no effect of 2-Ox on proliferation and hypertrophy zones, simultaneously it strengthens assumptions on specific regulatory mechanism involving 2-Ox in organism under stress.25,39,41
Chondrocytes within various zones differ in shape due to constantly altered metabolism.37 Moreover, chondrocyte mean and specifically maximal diameter (which is parallel to the growth plate surface37) were lowered by Gx, while cell sectional area was unaffected, which means that these cells were less compressed.44 However, the Gx rats were only 8% lighter than in the ShO group at the end of the experiment.7
One can speculate that in both cases, Gx and 2-Ox supplementation, chondrocyte response (changes in shape and size) was the compensation to the malnutrition or enhanced amount of glutamine serving as an energy source, respectively. Furthermore, chondrocytes in the Gx + 2-Ox group became bigger and more numerous in order to enhance collagen synthesis, when glutamine is delivered. Moreover, probably 2-Ox strengthened chondrocyte metabolism while reducing their quantity in ShO + 2-Ox group.
Reduction in body mass is an advantage of bariatric surgery in overweight patients and is associated with the abolishment of some comorbid conditions like the pain related to degenerative joint diseases.45,46 However, there are some deleterious changes, involving collagens distribution and cartilage thickness, which occur in articular cartilage following Gx, as revealed in the present animal study. On the other hand, rats from the Gx + 2-Ox group had a much higher articular cartilage thickness than those from the Gx group, despite the lack of significant differences in body mass.7 Hence, it can be presumed that the reduced cartilage thickness after Gx was not necessarily related to a lower bone load (considering the association of the cartilage thickness to the functional demand25,47,48).
Furthermore, the collagen fiber meshwork, which gives cartilage its form and tensile strength37 was also affected by Gx (especially reduced amount of thin collagen fibers), whereas dietary 2-Ox administration restored the distribution of thin and thick collagen fibers. However, one may speculate that the cohesiveness between the collagen fibers might not have been fully restored. Still, the collagen network was improved by 2-Ox administration which was similar to previous findings.7,25,39,41
Additionally, the mean intensity of intercellular matrix autofluorescence, in articular cartilage, was significantly elevated in rats treated with dietary 2-Ox, which reflects possible higher amount of hydroxyproline, which in turn reflects the collagen content. Although, levels of circulating amino acids were not measured in our study, the positive correlation between hydroxyproline and autofluorescence was previously shown by Hagiwara et al.33 Moreover, both autofluorescence and the amount of thin collagen fibers in articular cartilage were elevated in rats receiving 2-Ox, which means elevated collagen synthesis. The positive response to 2-Ox may be through the increased synthesis of proline and hydroxyproline, two main amino acids of collagen. Furthermore, in long-term studies on fundectomized pigs, dietary 2-Ox supplementation increased circulating plasma levels of the growth hormone, and insulin-like growth factor-1.23 Moreover, enteral administration of 2-Ox increases arterial plasma proline level whereas its parenteral treatment decreases plasma concentration of glutamine as shown by Kristensen et al.24 in pigs. Bearing in mind the function of 2-Ox as an energy donor and the stimulator of protein synthesis, particularly proline (which plays a key role in collagen formation), arginine, and asparagine,22,24 we conclude that this anabolic role of 2-Ox may be the main mechanism underlying the beneficial effects on cartilage (especially collagen) of gastrectomized rats.
The present findings also confirmed the recent assumptions about specific mechanism of metabolic regulation involving 2-Ox, which presumably is activated in a state of health problems.25,39,41
Since gastric surgeries are frequently used to induce weight loss in obese patients, particularly young, growing obese patients the present findings may have clinical implications. The present study served as a great opportunity to enhance the knowledge of the side effects of Gx on hyaline cartilage (articular and growth plate), simultaneously, to determine possible protective influences of dietary 2-Ox administration. The obtained results indicate that Gx greatly affects hyaline cartilage and dietary 2-Ox administration is capable of counteracting the Gx-evoked impairment of this crucial tissue.
Acknowledgement
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Author contributions
PD involved in conception and design, collection and assembly of data, analysis and interpretation of the data, statistical expertise, drafting of the article, final approval of the article, provision of study materials, and administrative, technical, or logistic support; ET involved in analysis and interpretation of the data, critical revision of the article for important intellectual content, and final approval of the article; PK involved in analysis of the data, final approval of the article, administrative, technical, or logistic support; SGP involved in conception and design, coding groups, interpretation of the data, critical revision of the article for important intellectual content, final approval of the article, and provision of study materials.
Declaration of conflicting interests
The author(s) declare(s) that there is no conflict of interest.
REFERENCES
- 1.Alqahtani AR, Antonisamy B, Alamri H, Elahmedi M, Zimmerman VA. Laparoscopic sleeve gastrectomy in 108 obese children and adolescents aged 5 to 21 years. Ann Surg 2012; 256: 266–73. [DOI] [PubMed] [Google Scholar]
- 2.Sanghera TS, Kang SN, Hamdan K. Metabolic bone disease and bariatric surgery. Rheumatol Curr Res 2012; S5: 001–001. [Google Scholar]
- 3.Williams SE. Metabolic bone disease in the bariatric surgery patient. J Obes. Epub ahead of print 2011. DOI:10.1155/2011/634614. [DOI] [PMC free article] [PubMed]
- 4.Baek KH, Jeon HM, Lee SS, Lim DJ, Oh KW, Lee WY, Rhee EJ, Han JH, Cha BY, Lee KW, Son HY, Kang SK, Kang MI. Short-term changes in bone and mineral metabolism following gastrectomy in gastric cancer patients. Bone 2008; 42: 61–7. [DOI] [PubMed] [Google Scholar]
- 5.Rümenapf G, Schwille PO, Erben RG, Schreiber M, Fries W, Schmiedl A, Hohenberger W. Osteopenia following total gastrectomy in the rat – state of mineral metabolism and bone histomorphometry. Eur Surg Res 1997; 29: 209–21. [DOI] [PubMed] [Google Scholar]
- 6.Lehto-Axtelius D, Surve V, Johnell O, Håkanson R. Effect of calcium deficiency and calcium supplementation on gastrectomy-induced osteopenia in the young male rat. Scand J Gastroenterol 2002; 37: 299–306. [DOI] [PubMed] [Google Scholar]
- 7.Dobrowolski P, Piersiak T, Surve VV, Kruszewska D, Gawron A, Pacuska P, Håkanson R, Pierzynowski SG. Dietary α-ketoglutarate reduces gastrectomy-evoked loss of calvaria and trabecular bone in female rats. Scand J Gastroenterol 2008; 43: 551–8. [DOI] [PubMed] [Google Scholar]
- 8.Zittel T, Zeeb B, Maier G, Kaiser GW, Zwirner M, Liebich H, Starlinger M, Becker HD. High prevalence of bone disorders after gastrectomy. Am J Surg 1997; 174: 431–8. [DOI] [PubMed] [Google Scholar]
- 9.Lim JS, Kim SB, Bang HY, Cheon GJ, Lee JI. High prevalence of osteoporosis in patients with gastric adenocarcinoma following gastrectomy. World J Gastroenterol 2007; 13: 6492–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lim JS, Lee JI. Prevalence, pathophysiology, screening and management of osteoporosis in gastric cancer patients. J Gastric Cancer 2011; 11: 7–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Persson P, Gagnemo-Persson R, Chen D, Axelson J, Nylander AG, Johnell O, Håkanson R. Gastrectomy causes bone loss in the rat: Is lack of gastric acid responsible? Scand J Gastroenterol 1993; 28: 301–6. [DOI] [PubMed] [Google Scholar]
- 12.Lehto-Axtelius D, Chen D, Surve V, Håkanson R. Post-gastrectomy osteopenia in the rat. Bone structure is preserved by retaining 10%–30% of the oxyntic gland area. Scand J Gastroenterol 2002; 37: 437–43. [DOI] [PubMed] [Google Scholar]
- 13.Viégas M, Vasconcelos RS, Neves AP, Diniz ET, Bandeira F. Bariatric surgery and bone metabolism a systematic review. Arq Bras Endocrinol Metab 2010; 54: 158–63. [DOI] [PubMed] [Google Scholar]
- 14.Rümenapf G, Schwille PO, Erben RG, Schreiber M, Bergé B, Fries W, Schmiedl A, Koroma S, Hohenberger W. Gastric fundectomy in the rat: effects on mineral and bone metabolism, with emphasis on the gastrin-calcitonin-parathyroid hormone-vitamin D axis. Calcif Tissue Int 1998; 63: 433–41. [DOI] [PubMed] [Google Scholar]
- 15.Zittel T, Maier G, Starlinger M, Becker HD. Calcium and bone metabolism after gastrectomy. Chirurg 1997; 68: 784–8. [DOI] [PubMed] [Google Scholar]
- 16.Inui A, Iwakura T, Reddi AH. Human stem cells and articular cartilage regeneration. Cells 2012; 1: 994–1009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Heng BC, Cao T, Lee EH. Directing stem cell differentiation into the chondrogenic lineage in vitro. Steam Cells 2004; 22: 1152–67. [DOI] [PubMed] [Google Scholar]
- 18.Matsiko A, Levingstone TJ, O’Brien FJ. Advanced strategies for articular cartilage defect repair. Materials 2013; 6: 637–68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Wei Y, Zeng W, Wan R, Wang J, Zhou Q, Qiu S, Singh SR. Chondrogenic differentiation of induced pluripotent stem cells from osteoarthritic chondrocytes in alginate matrix. Eur Cell Mater 2012; 23: 1–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lobstein T, Baur L, Uauy R. Obesity in children and young people: a crisis in public health. Obes Rev 2004; 5: 4–85. [DOI] [PubMed] [Google Scholar]
- 21.Radzki RP, Bienko M, Pierzynowski SG. Anti-osteopenic effect of alpha-ketoglutarate sodium salt on ovariectomized rats. J Bone Miner Metab 2012; 30: 651–9. [DOI] [PubMed] [Google Scholar]
- 22.Harrison AP, Pierzynowski SG. Biological effects of 2-oxoglutarate with particular emphasis on the regulation of protein, mineral and lipid absorption/metabolism, muscle performance, kidney function, bone formation and cancerogenesis, all viewed from a healthy ageing perspective state of the art – review article. J Physiol Pharmacol 2008; 59: 91–106. [PubMed] [Google Scholar]
- 23.Śliwa E. 2-Oxoglutaric acid administration diminishes fundectomy-induced osteopenia in pigs. J Anim Physiol Anim Nutr (Berl) 2010; 94: 86–95. [DOI] [PubMed] [Google Scholar]
- 24.Kristensen NB, Jungvid H, Fernandes JA, Pierzynowski SG. Absorption and metabolism of α-ketoglutarate in growing pigs. J Anim Physiol Anim Nutr 2002; 86: 239–45. [DOI] [PubMed] [Google Scholar]
- 25.Dobrowolski P, Tomaszewska E, Bienko M, Radzki RP, Pierzynowski SG. The effect of dietary administration of 2-oxoglutaric acid on the cartilage and bone of growing rats. Br J Nutr 2013; 110: 651–8. [DOI] [PubMed] [Google Scholar]
- 26.Suvara SK, Layton C, Bancroft JD. Bancroft's Theory and practice of histological techniques. 7th ed. Churchill Livingstone Elsevier, London, 2012.
- 27.Rich L, Whittaker P. Collagen and picrosirius red staining: a polarized light assessment of fibrillar hue and spatial distribution. Braz J Morphol Sci 2005; 22: 97–104. [Google Scholar]
- 28.Roach HI, Mehta G, Oreffo ROC, Clarke NM, Cooper C. Temporal analysis of rat growth plates cessation of growth with age despite presence of a physis. J Histochem Cytochem 2003; 51: 373–83. [DOI] [PubMed] [Google Scholar]
- 29.Tomaszewska E, Dobrowolski P, Puzio I. Morphological changes of the cartilage and bone in newborn piglets evoked by experimentally induced glucocorticoid excess during pregnancy. J Anim Physiol Anim Nutr 2013; 98: 785–96. [DOI] [PubMed] [Google Scholar]
- 30.Hunziker EB, Quinn TM, Hauselmann HJ. Quantitative structural organization of normal adult human articular cartilage. Osteoarthritis Cartilage 2002; 10: 564–72. [DOI] [PubMed] [Google Scholar]
- 31.Claassen H, Schicht M, Paulsen F. Impact of sex hormones, insulin, growth factors and peptides on cartilage health and disease. Prog Histochem Cytochem 2011; 45: 239–93. [DOI] [PubMed] [Google Scholar]
- 32.Bobinac D, Spanjol J, Zoricic S, Maric I. Changes in articular cartilage and subchondral bone histomorphometry in osteoarthritic knee joints in humans. Bone 2003; 32: 284–90. [DOI] [PubMed] [Google Scholar]
- 33.Hagiwara Y, Hattori K, Aoki T, Ohgushi H, Ito H. Autofluorescence assessment of extracellular matrices of a cartilage-like tissue construct using a fluorescent image analyser. J Tissue Eng Regen Med 2011; 5: 163–8. [DOI] [PubMed] [Google Scholar]
- 34.Sokal RR, Rohlf FJ. Biometry, 3rd ed New York, NY: W.H. Freeman, 1995, pp. 272–308. [Google Scholar]
- 35.Lehto-Axtelius D, Stenström M, Johnell O. Osteopenia after gastrectomy, fundectomy or antrectomy: an experimental study in the rat. Regul Pept 1998; 78: 41–50. [DOI] [PubMed] [Google Scholar]
- 36.Surve V, Andersson N, Alatalo S, Lehto-Axtelius D, Halleen J, Väänänen K, Håkanson R. Does combined gastrectomy and ovariectomy induce greater osteopenia in young female rats than gastrectomy alone? Calcif Tissue Int 2001; 69: 274–80. [DOI] [PubMed] [Google Scholar]
- 37.Buckwalter JA, Mankin HJ, Grodzinsky AJ. Articular cartilage and osteoarthritis. Instr Course Lect 2005; 54: 465–80. [PubMed] [Google Scholar]
- 38.Filip RS, Pierzynowski SG, Lindegard B, Wernerman J, Haratym-Maj A, Podgurniak M. Alpha-ketoglutarate decreases serum levels of C-terminal cross-linking telopeptide of type I collagen (CTX) in postmenopausal women with osteopenia: six-month study. Int J Vitam Nutr Res 2007; 77: 89–97. [DOI] [PubMed] [Google Scholar]
- 39.Dobrowolski P, Tomaszewska E, Radzki RP, Bienko M, Wydrych J, Zdybel A, Pierzynowski SG. Can 2-oxoglutarate prevent changes in bone evoked by omeprazole? Nutrition 2013; 29: 556–61. [DOI] [PubMed] [Google Scholar]
- 40.Śliwa E, Adaszek Ł, Tatara M, Dobrowolski P. Short- and long-term consequences on biochemical markers after fundectomy in pigs supplemented with 3-hydroxy-3-methylbutyrate and alpha-ketoglutarate. Berl Munch Tierarztl Wochenschr 2010; 123: 397–405. [PubMed] [Google Scholar]
- 41.Tomaszewska E, Dobrowolski P, Wydrych J. Postnatal administration of 2-oxoglutaric acid improves articular and growth plate cartilages and bone tissue morphology in pigs prenatally treated with dexamethasone. J Physiol Pharmacol 2012; 63: 547–54. [PubMed] [Google Scholar]
- 42.Mühlbauer RC, Schenk RK, Chen D, Lehto-Axtelius D, Håkanson R. Morphometric analysis of gastrectomy-evoked osteopenia. Calcif Tissue Int 1998; 62: 323–6. [DOI] [PubMed] [Google Scholar]
- 43.Andersson N, Surve VV, Lehto-Axtelius D, Ohlsson C, Håkanson R, Andersson K, Ryberg B. Drug-induced prevention of gastrectomy- and ovariectomy-induced osteopenia in the young female rat. J Endocrinol 2002; 175: 695–703. [DOI] [PubMed] [Google Scholar]
- 44.Guilak F, Jones WR, Ting-Beall HP, Lee GM. The deformation behavior and mechanical properties of chondrocytes in articular cartilage. Osteoarthritis Cartilage 1999; 7: 59–70. [DOI] [PubMed] [Google Scholar]
- 45.Sugerman HJ, Sugerman EL, DeMaria EJ, Kellum JM, Kennedy C, Mowery Y, Wolfe LG. Bariatric surgery for severely obese adolescents. J Gastrointest Surg 2003; 7: 102–7. [DOI] [PubMed] [Google Scholar]
- 46.Vincent HK, Heywood K, Connelly J, Hurley RW. Obesity and weight loss in the treatment and prevention of osteoarthritis. PMR 2012; 4: 59–67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Eckstein F, Muller-Gerbl M, Putz R. Distribution of subchondral bone density and cartilage thickness in the human patella. J Anat 1992; 180: 425–33. [PMC free article] [PubMed] [Google Scholar]
- 48.Koszyca B, Fazzalari NL, Vernon-Roberts B. Quantitative analysis of the bone cartilage interface within the knee. Knee 1996; 3: 23–31. [Google Scholar]