Abstract
Over the past decade, reported incidence of human metapneumovirus (hMPV) has increased owing to the use of molecular assays for diagnosis of respiratory viral infections in cancer patients. The seasonality of these infections, differences in sampling strategies across institutions, and small sample size of published studies make it difficult to appreciate the true incidence and impact of hMPV infections. In this systematic review, we summarized the published data on hMPV infections in hematopoietic cell transplant recipients and patients with hematologic malignancy, focusing on incidence, hMPV-associated lower respiratory tract infection (LRTI), mortality, prevention, and management with ribavirin and/or intravenous immunoglobulins. Although the incidence of hMPV infections and hMPV-associated LRTI in this patient population is similar to respiratory syncytial virus or parainfluenza virus and despite lack of directed antiviral therapy, the mortality rate remains low unless patients develop LRTI. In the absence of vaccine to prevent hMPV, infection control measures are recommended to reduce its burden in cancer patients.
Keywords: hMPV, stem cell transplantation, leukemia, lymphoma, cancer, mortality
1. Introduction
Use of sensitive polymerase chain reaction (PCR) assay has led to increased awareness and identification of human metapneumovirus (hMPV) as a common cause of respiratory viral infection in hematopoietic cell transplant (HCT) recipients and hematologic malignancy (HM) patients.
A recently discovered negative-sense RNA paramyxovirus, hMPV is genetically similar to respiratory syncytial virus (RSV) and it reportedly infects approximately 5%–9% of HCT recipients.[1, 2] Progression of upper respiratory tract infection (URTI) to the lower respiratory tract infection (LRTI) occurs in 21% to 40% of cases,[3] with reported fatality rates of up to 80% in HCT recipients if bronchoalveolar lavage is positive for hMPV.[4] Its presentation is clinically indistinguishable from that of other respiratory viruses, and its growth is unreliable on culture; thus, this viral infection is best diagnosed using PCR-based assays or direct antigen detection. The only drug that has been found to be active against this virus in vitro is ribavirin;[5, 6] however, there is a dearth of knowledge about this virus and its treatment.
Scattered case reports are available that describe the clinical disease spectrum, management, and overall outcomes of hMPV in cancer patients. Hence, we conducted a systematic review of all published data to summarize the incidence, risk factors, management, long-term outcomes, and associated mortality rates of hMPV infections with a focus on HM patients and HCT recipients. Advances in diagnostic methods, available or new investigational drugs, and vaccines are also discussed. Given the increase in the diagnosis of hMPV infections and the scarcity of available data, this review aims to help clinicians to better understand the implications of this infection in this specific patient population.
2. Materials and Methods
2.1 Search strategy and selection criteria
We conducted an electronic literature search using Medline via the Ovid, Embase, Web of Science, and Cochrane library databases in July 2015. The following Medical Subject Heading terms were used: human metapneumovirus, hematopoietic stem cell transplantation, bone marrow transplantation, leukemia, lymphoma, myeloma, hematologic malignancy and hematologic neoplasms. The references in all of the selected studies were also reviewed to identify additional articles that did not appear in the initial search. The full texts of the selected articles were reviewed by all the authors. Inclusion and exclusion criteria were defined a priori. The following inclusion criteria were used for selecting the articles:
HM patients and HCT recipients of any age and had been infected with hMPV,
Retrospective or prospective observational studies and randomized controlled trials
No time restriction for the study period.
Articles in English.
Exclusion criteria were as follows:
Review papers or meta-analyses,
Studies with duplicate data or incomplete information, and
Case reports and meeting abstracts were included due to limited literature on this topic. We also searched the Clinical Trials registry (U.S. National Institutes of Health, www.clinicaltrials.gov) to identify any registered clinical trials for hMPV infections.
a. Definitions
Infection episodes and subsequent outcomes were determined by the authors of the original articles using various definitions; however, below are the summarized versions of these definitions used for the current review.
hMPV case: patients with a positive nasal wash, nasopharyngeal swab, or bronchoalveolar lavage for hMPV by viral diagnostic test were included in this review.
hMPV-LRTI: was defined as the onset of respiratory symptoms with new or changing pulmonary infiltrates, as seen on chest x-ray or CT scan of chest and/or virus isolated from lower respiratory samples (e.g., endotracheal tube aspirate, sputum, or bronchoalveolar lavage fluid)
hMPV-mortality: Death was attributed to hMPV if a persistent or progressive infection with respiratory failure was identified at the time of death.
2.3 Data abstraction
Two authors (D.P.S. and P.K.S.) independently screened the abstracts using the predefined inclusion and exclusion criteria. Four authors (D.P.S., P.K.S., J.M.A., and F.E.C.) used standardized coding rules to abstract important variables from the final list of articles independently and discrepancies were resolved by discussion. Primary variables of interest for this study were incidence of hMPV infection, progression of hMPV-URTI to hMPV-LRTI and hMPV-associated mortality. Antiviral therapy included ribavirin (aerosolized, intravenous, or oral) alone or in combination with intravenous immunoglobulins (IVIGs). Effect of antiviral therapy was measured by comparing incidence rates of these outcomes in treated and untreated patients. Outcome data from selected full-text articles were validated by R.F.C. For studies reporting outcomes in HM patients and HCT recipients, the data abstraction was split into two parts to capture the characteristics and outcomes of each group, respectively.
2.4 Statistical analysis
Agreement between the two independent authors in the first and second phase of the full-text selection process was checked by calculating Cohen's Kappa. Outcomes (i.e., LRTI and death) were descriptively summarized as percentages. We compared treated and untreated patient outcomes using Chi-squared or Fisher exact tests, as appropriate. Odds ratios (ORs) were calculated with 95% confidence intervals (95% CIs). All statistical analyses were performed using STATA software version 13 (STATA Corp., College Station, TX, USA). A p value of 0.05 was considered significant.
3. Results
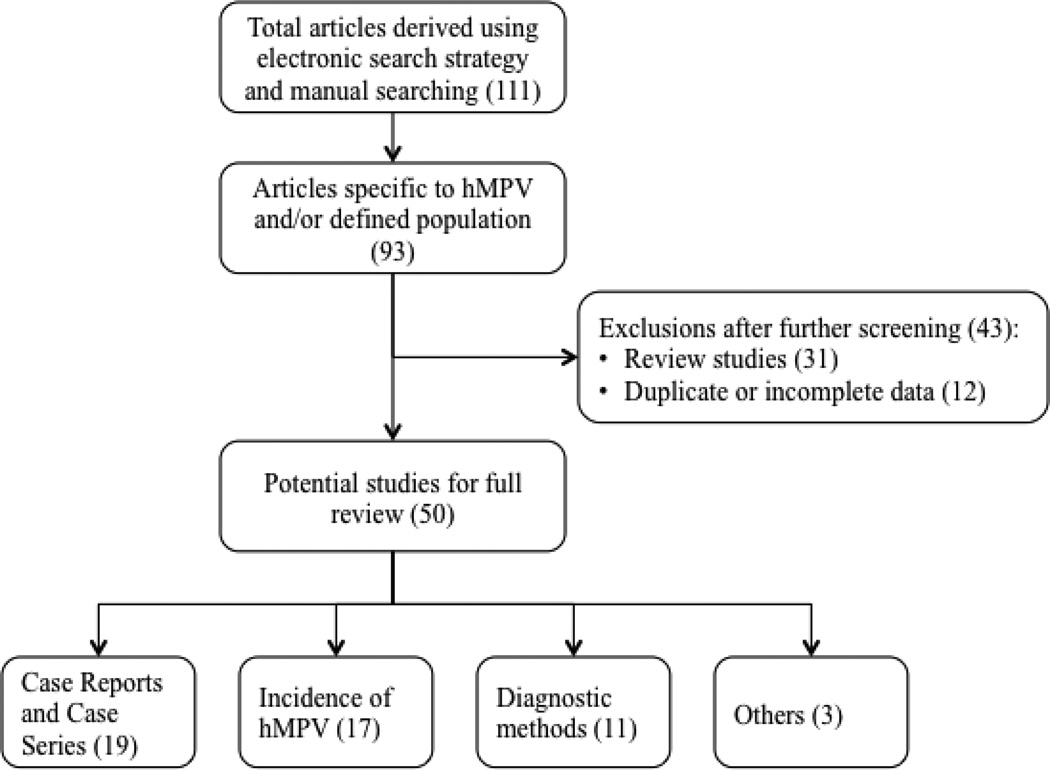
Of 111 abstracts retrieved for hMPV review (105 by electronic search and 6 by manual search), 93 were specific to hMPV infection in the defined population, hence screened further. Of these, 31 were review studies of respiratory viruses and 12 had overlapping data with an included study or had incomplete information; these were excluded from further review, leaving 50 full-text articles and abstracts (hMPV incidence [17], case reports and data on outcomes [19], diagnostic methods [11], and other studies [3]). Study screening flowchart is shown in Figure 1. The agreement between the two authors during the selection of abstracts and the selection of full-texts, as measured by Cohen's Kappa, was 0.942 [95% CI: 0.829 – 0.999] and 0.98 [0.94 – 0.984], respectively which is regarded as substantial to excellent.
Figure 1.
Flow diagram of study selection of hMPV infections in HM patients and HCT recipients
3.1 Clinical Presentation and Diagnosis
The clinical presentation of hMPV infection is similar to that of other respiratory infections, such as RSV and PIV, ranging from mild URTI to LRTI to progressive respiratory failure and death. Some of the common presenting symptoms of hMPV are fever, cough, nasal congestion, rhinorrhea, headache, sore throat, and dyspnea, along with other prodromal symptoms.[4, 7–16] Diagnosing hMPV infections is particularly dependent on the availability of RT-PCR because the viral culture yield is extremely low as a result of the delayed cytopathic effect of hMPV and masking by other concurrent viral infections.[12] Some earlier studies that used direct fluorescence antigen assay or culture reported an almost doubled yield with a better sensitive test such as RT-PCR.[2, 17]. A recent study found that RT-PCR detected high viral loads in bronchoalveolar lavage samples from 7 HCT recipients with hMPV-LRTI (median: 3.9 × 107 copies/ml) and viral RNA from serum samples of patients with pneumonia which may be indicative of severe disease.[18] Reported radiological findings of hMPV-LRTI include diffuse alveolar and interstitial infiltrates, bi-basilar tree-in-bud nodularity, ground glass opacities, scattered small nodules, basal shadowing, extensive air space consolidation, and pleural effusion.[4, 7, 9, 10, 13–15, 19]
3.2 Incidence of hMPV infections
The incidence of hMPV infections from 17 studies is shown in Table 1. Overall, 204 hMPV infections were identified of 4028 patients evaluated (studies examining HM patients, HCT recipients and combination of both); giving an incidence of 5% (reported range of 0% to 40%). The incidence of hMPV infections in HM patients (7% [18 of 272]) was not different from that observed for HCT recipients (7% [103 of 1504]). Furthermore, the hMPV infection rate in adult patients (7% [92 of 1346]) was slightly higher than that observed for children (4% [26 of 624). No specific risk factors for hMPV infection were reported in any of these studies.
Table 1.
Incidence of hMPV infections, hMPV-LRTI, and hMPV mortality in HCT recipients and HM patients, n (%).
| Author, year | Study time period |
Location | Age | Number of patients evaluated (Symptomat ic/Asympto matic) |
Type of diagnostic test |
hMPV incidence (hMPV/patient s evaluated) |
hMPV-LRTI (LRTI/ number of hMPV infections) |
hMPV mortality (deaths/ number of hMPV infections) |
hMPV- deaths/hMPV- LRTI |
|---|---|---|---|---|---|---|---|---|---|
| HCT recipients | |||||||||
| Lee 2011[38]a | 2007–2009 | Seoul, Korea | Children | 211 (S) | PCR, culture | 0 | 0 | 0 | 0 |
| Fazekas-A, 2012[39]a | 2007–2009 | Vienna, Austria | Children | 31 (AS) | PCR | 8 (26) | NA | NA | NA |
| Srinivasan-A 2013[40]a | Oct 2010 – Sept 2011 |
Memphis, TN | Children | 42 (S) | PCR | 1 (2) | 1 (100) | NA | NA |
| Choi, 2013[41]a | 2007–2010 | Seoul, Korea | Children | 116 (S) | RT-PCR | 1 (1) | 0 | 0 | 0 |
| Martino, 2005[42]a | 1999–2003 | Barcelona, Spain | Adult | 177 (S) | IF, culture | 16 (9) | 5 (31) | 2 (13) | 2 (40) |
| Debiaggi, 2007[36]a | 2004–2006 | Italy | Adult | 53 (AS) | RT-PCR | 21 (40) | NA | NA | NA |
| Souza 2013[43]b | Mar 2008–Feb 2009 |
Sao Paulo, Brazil | Adult | 75 (S) | PCR | 11 (14.7) | NA | NA | NA |
| Peck, 2007[2]b | 2000–2004 | Seattle, WA | Any | 122 (AS) | Culture, DFA, PCR |
6 (5) | 0 | 0 | - |
| Debur, 2010[44]a | 2000–2002; 2006–2008 |
Curitiba, Brazil | Any | 189 (S) | RT-PCR | 18 (10) | 5 (28) | 0 | 0 |
| Wolfromm, 2011[45]a | 2007–2010 | Paris, France | Any | 127 (S) | PCR | 6 (5) | NA | 1 (17) | NA |
| Oliveira, 2008[46]a | 2001–2003 | Sao Paulo, Brazil | NA | 153 (S) | RT-PCR, IF | 11 (7) | 3 (27) | 1 (9) | 1 (33) |
| Mikulska, 2011[47]a | 2007–2010 | Genoa, Italy | NA | 208 (S) | PCR | 4 (2) | NA | NA | NA |
| HM patients | |||||||||
| Fazekas-B, 2012[39]a | 2007–2009 | Vienna, Austria | Children | 103 (AS) | PCR | 11 (11) | NA | NA | NA |
| Srinivasan-B, 2013[40] a |
Oct 2010 – Sept 2011 |
Memphis, TN | Children | 121 (S) | PCR | 5 (4) | 4 (80) | NA | NA |
| Chebotkevich, 2010[48] a |
2008–2009 | St. Petersburg, Russia | Any | 48 (S) | RT-PCR | 2 (4) | NA | NA | NA |
| HM and HCT patients | |||||||||
| Williams, 2005[1]a | 1999–2004 | Barcelona, Spain | Adult | 128 (S) | RT-PCR | 22 (17) | 9 (41) | 3 (14) | 3 (33) |
| Kwon, 2012 [49]c | Apr 2009– Aug 2011 |
Seoul, Korea | Adult | 176 (AS) | PCR | 1 (0.6) | NA | NA | NA |
| Park, 2013 [50]a | Jan 2009 – Feb 2012 |
Seoul, Korea | Adult | 737 (S) | PCR | 21 (2.8) | NA | NA | NA |
| Kamboj, 2008[17]b | 2005–2007 | NYC, USA | Any | 1211 (S) | DFA, RT-PCR, Shell vial assay |
39 (3) | NA | 0 | NA |
Single center investigator-initiated study;
samples collected during respiratory seasons;
only patients who underwent bronchoscopy
Data from Fazekas et al. (2012)[39] were split for hMPV incidence in the HM and HCT groups, respectively;
DFA = direct fluorescent antibody; hMPV-LRTI indicates human metapneumovirus-associated lower respiratory tract infections; HCT recipient = hematopoietic cell transplant recipients; HM = hematologic malignancy; IF = immunofluorescence; NA= not available; PCR = polymerase chain reaction; RT-PCR = reverse transcription polymerase chain reaction
3.3 hMPV-LRTI
The incidence of hMPV-LRTI in HM patients and HCT recipients with hMPV infections was reported by 9 studies and is displayed in Table 1. Twenty seven hMPV-LRTI cases of 80 hMPV infections were observed, giving an incidence of 34%. The reported range for hMPV-LRTI was 0% to 100%. Stratified hMPV-LRTI rates for HM patients were not available and a comparison with HCT recipients was not possible. Risk factors for hMPV-LRTI have not been studied in detail; hence it is difficult to identify high-risk patients. We identified two studies which examined relatively large-scale, single-institution, retrospective data on hMPV infections in cancer patients.[20, 21] The first study examined 118 HCT recipients with hMPV infections and found significant association between steroid use at ≥1mg/kg within 2 weeks prior to diagnosis (HR 5.10; p=0.004), low lymphocyte count (HR 3.43; p=0.011), and early onset of HMPV infection after HCT (before day 30 after HCT; HR 3.54; p=0.013) and progression to LRTI (adjusted hazards ratio = 4.1, P value = 0.04) based on bivariable Cox regression analysis.[20] Higher hMPV viral load in nasal wash at diagnosis was not identified as a significant predictor of LRTI progression in this study.[20] The second study examined 135 cancer patients including patients with HM (n=46), solid tumors (n=21), and HCT recipients (n=68) with hMPV infections. The overall incidence of LRTI was 44% and independent predictors of LRTI were nosocomial infection and hypoxia at presentation.[21]
3.4 hMPV-Mortality
As reported by the 9 studies that examined patients with hMPV infections (URTI and LRTI), 7 out of 119 patients died; giving a mortality rate of 6% (range: 0% to 17%). However, the mortality rate was significantly higher in patients who developed hMPV-LRTI (27%, 6 of 22) (OR: 5.9, 95% CI: 1.4, 23.3; P value = 0.0015). This finding remained same after including the five studies that only examined patients with hMPV-LRTI,[4, 22–25] with a mortality rate of 26% (14 deaths of 54 hMPV-LRTI cases). Similar findings were observed with overall low all-cause mortality (3%) and hMPV associated mortality (2%) at 30 days of follow-up after diagnosis of hMPV infections in 135 cancer patients; while no difference in 30-day all-cause mortality rate was identified between HM patients (4%) and HCT recipients (3%). [21] Statistical analysis to identify association of hMPV-mortality rates with different types of underlying HM was not possible due to insufficient data. To our knowledge, risk factors for hMPV mortality have not been reported in the current literature except in one study which reported that corticosteroids, oxygen requirement > 2L or mechanical ventilation and bone marrow as cell source were significant risk factors for respiratory virus-related mortality (not specific to hMPV-related mortality) in HCT recipients.[26]Characteristics, management, and outcomes of 47 hMPV infection cases published as case reports are displayed in supplemental table 1. Of these 47 patients with hMPV infections, 13 died and the mortality rate following hMPV-LRTI was 28%.
3.5 Antiviral therapy
In few case reports, ribavirin-based therapy has been found (anecdotally) to be effective at preventing death from hMPV in immunocompromised patients.[10, 13–15, 19] However, most of these reports had no control group. Available data from case reports on the use of antiviral therapy and outcomes in 47 patients with hMPV infections is shown in supplemental table 1. Four out of 13 patients who died had received antiviral therapy (2 AR, 1 IVIG, and 1 AR + IVIG). Of the 34 patients who survived, 19 patients had not received any antiviral therapy (supplemental table 1). Bivariable regression analysis using data from Table 2 showed that the ribavirin therapy did not have a significant protective effect against hMPV-mortality (0.58 [0.18 – 1.85]; P = 0.337). A recent study compared the mortality rate in 23 HCT recipients with hMPV-LRTI who were treated or not with aerosolized ribavirin and intravenous polyclonal immunoglobulins and the results were discouraging, with similar mortality rates as in the untreated group but higher hypoxemia rates.[26] In addition, a recent case series that included 31 children with cancer (14 with leukemia) reported that all children recovered from their illness with no antiviral-directed therapy; only one patient had prolonged respiratory symptoms.[27] In the cohort of 135 cancer patients with hMPV infections (21), 3 patients with hMPV-LRTI (2 HCT and 1 HM) and 1 patient with hMPV-URI (1 HCT) were treated with AR; one patient with HM and LRTI died at day 22.
4. Discussion
In this systematic review, we attempted to summarize all published data on hMPV infections in HM patients and HCT recipients to generate meaningful conclusions about their incidence, LRTI and mortality rates, management including antiviral therapy, and prevention measures, including vaccines
We identified high rates of hMPV infections in HM patients and HCT recipients, similar to that observed for other respiratory viruses such as RSV and PIV. The incidence varied widely, which could be attributed to factors such as patient sampling methods (symptomatic versus asymptomatic), patient age, transplant and underlying malignancy type, season and study period, publication bias, and diagnostic method used (i.e., RT-PCR vs. direct fluorescence antigen assay vs. culture). Rates of hMPV infections were similar for HCT recipients as well as HM patients. Furthermore, high morbidity rates were reported with approximately one-third of hMPV infections developing LRTI (34%). Despite lack of a directed antiviral therapy, the virus-associated mortality rates remained low for hMPV infections (6%). However, patients who progressed to LRTI stage experienced higher mortality rates (27%). Thus, it is important to prevent LRTI in patients with hMPV infections. No hMPV-specific risk factors for increased mortality have been identified. A recent small study comparing mortality rates in 23 RSV-LRTI versus 23 hMPV-LRTI in HCT recipients identified that steroid therapy, oxygen requirement >2 L or mechanical ventilation, and bone marrow as cell source were significant risk factors for overall and virus-related mortality; however, these risk factors were not specific to RSV or hMPV.[26]
Ribavirin’s role in treating hMPV infections in HCT recipients and HM patients is still not known. Few studies report successful treatment of hMPV infections with ribavirin, with and without intravenous polyclonal immunoglobulins; [10, 13–15, 19] however; the data are limited to small case series or case reports. Ribavirin has been hypothesized to have a dual mode of action against hMPV: it has a direct antiviral effect on hMPV, and it may awaken the hypo-responsive immune system after prolonged viremia.[28] We did not observe a significant protective effect of ribavirin in preventing hMPV mortality; however, the sample size was small and adjustment for other confounding factors was not possible (supplemental table 1). Thus, efficacy of ribavirin in preventing LRTI and mortality from these infections needs to be further investigated in the absence of investigational agents for this infection using a large retrospective cohort, multi-institutional preferably, or a randomized clinical trial that could be very difficult to conduct in a timely fashion. Standard IVIG administration can inhibit hMPV replication in vitro [5] but its potential benefit in HCT recipients could not be determined from current published studies. We concur with the guidelines from the Fourth European Conference on Infections in Leukemia (ECIL-4) that treatment recommendations are currently not possible for hMPV infections in cancer patients.[29] Advances have been reported in the generation of polyfunctional hMPV specific Tcells producing multiple cytokines (IFN, TNF, GM-CSF) and effector molecules (Granzyme B) upon stimulation with cognate antigen.[30] These T-cells were specific for F, N, M2-1 and P antigens in HCT recipient who cleared hMPV infection; thus a potential adoptive T-Cell therapy to prevent and/or treat hMPV infections in immunocompromised patients is promising.[30]
Our search of the clinical trials registry and the published articles did not identify reports of any new investigational drug being developed for the treatment of hMPV infections. However, few monoclonal antibodies (moAb) are being developed for prevention and treatment of hMPV infection. MoAb 338 that targets hMPV fusion protein has showed potential usefulness for hMPV prevention and promising results in vitro and in vivo where it reduced pulmonary viral titers, limited bronchial hyperactivity and prevented severe acute manifestations.[31, 32] Furthermore, Human Fab DS7 (human moAb fragment) showed prophylactic and therapeutic benefits for severe hMPV infections in vitro and in mouse models.[33, 34] In addition, a phase 1, double-blind, placebo-controlled, study is ongoing to determine the safety and immunogenicity of a recombinant, live, attenuated rHMPV-Pa vaccine delivered intranasally in healthy adults (aged 18 to 49 yrs) and hMPV-seropositive and sero-negative children (NCT01255410). Although live, attenuated virus vaccine may be promising in young children and in healthy adults, its use in immunocompromised cancer patients may not be recommended. Finally, a specific class of viral subunit vaccine, virus-like particles (VLPs) that mimic the overall viral structure and antigenic conformation of F and G proteins induced significant cross-protective immunity in mice. These VLPs offer a distinct advantage of safe, specific, and sustained immune response against hMPV infections and may be a viable candidate for further trials in cancer patients.[35]
In the absence of an effective drug or vaccine, infection control measures play a key role in decreasing the incidence of hMPV infections, preventing nosocomial outbreaks, and subsequent morbidity and mortality in immunocompromised patients. However, hMPV infections in HCT recipients may be subclinical,[36] making it difficult to halt transmission of this virus. In addition, prolonged viral shedding of up to 90 days in immunocompromised hosts has been reported.[37] Thus, including hMPV testing in the routine surveillance of symptomatic HCT recipients and HM patients would be beneficial for early identification, allowing patients to be placed on contact isolation when needed.
One of the limitations of this systematic review is the exclusion of non-English language publications, especially from developing nations. In addition, our reliance on secondary data may be subject to interpretation errors; we tried to minimize this by having two different investigators (D.P.S. and P.K.S.) validate the data; and outcomes were reconfirmed by R.F.C. Most of the studies were retrospective and non-randomized in nature; hence, the results of this systematic review should be interpreted with caution. We attempted to decrease the publication bias by including almost all the published studies with minimal exclusion criteria. Another limitation of these studies was the lack of standardized definition for hMPV-LRTI. Since this review included a heterogeneous study population, we could not conduct meta-regression analyses to identify the independent effects of various host risk factors on the progression to LRTI. Finally, due to inconsistent reporting, the role of co-infections and antibiotics usage in preventing progression to hMPV-LRTI could not be systematically evaluated.
In summary, to our knowledge, this is the first comprehensive review on hMPV infections in HM patients and HCT recipients attempting to determine the actual incidence of these infections and subsequent LRTI and mortality in this patient population. Based on this review, we identified that there is high morbidity with low mortality associated with hMPV infections, unless the infection progresses to LRTI stage. Further studies focusing on hMPV infections in cancer patients are needed to determine the risk factors associated with LRTI and mortality and the role of existing antiviral drugs in preventing severe disease outcomes.
Supplementary Material
Highlights.
First systematic review on hMPV infections in HM patients and HCT recipients
hMPV incidence, risk factors, morbidity, and mortality reviewed from all published studies
Data on antiviral therapy, ongoing clinical trials and vaccine trials are also examined
Acknowledgments
We are grateful to our librarian, Ms. Yimin Geng, The University of Texas MD Anderson Research Medical Library for her assistance with electronic search for this review. We also thank Ms. Ann Sutton, Department of Scientific Publications, The University of Texas MD Anderson Cancer Center, for her editorial support.
FUNDING SUPPORT
This research was supported in part by the National Institutes of Health through MD Anderson’s Cancer Center Support Grant CA016672.
Abbreviations
- ALL
acute lymphoblastic leukemia
- Allo-HCT
allogeneic HCT
- AML
acute myeloid leukemia
- auto-HCT
autologous HCT
- CI
confidence interval
- CLL
chronic lymphocytic leukemia
- CML
chronic myelogenous leukemia
- DFA
Direct fluorescent antibody
- HCT
hematopoietic cell transplant
- HM
hematologic malignancy
- hMPV
human metapneumovirus
- IF
immunofluorescence
- LRTI
lower respiratory tract infection
- MDS
myelodysplastic syndrome
- MM
multiple myeloma
- MMF
mycophenolate mofetil
- MMRD
mismatched related donor
- MoAb
monoclonal antibody
- MRD
matched related donor
- MUD
matched unrelated donor
- OR
Odds ratio
- PCR
polymerase chain reaction
- RSV
respiratory syncytial virus
- RT-PCR
reverse transcriptase polymerase chain reaction
- SCID
severe combined immunodeficiency
- URTI
upper respiratory tract infection
- VLP
virus-like particle
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
AUTHOR CONTRIBUTIONS: D.P.S. and R.F.C. designed the study, D.P.S., P.K.S. screened the abstracts; D.P.S., P.K.S., F.E.C., and J.M.A. extracted data from full text articles, D.P.S and R.F.C. wrote the manuscript and all authors reviewed the full text articles and provided critical feedback and final approval for the manuscript.
DECLARATION OF CONFLICTS OF INTEREST
The authors have no conflicts of interest to declare.
REFERENCES
- 1.Williams JV, Martino R, Rabella N, Otegui M, Parody R, Heck JM, Crowe JE., Jr A prospective study comparing human metapneumovirus with other respiratory viruses in adults with hematologic malignancies and respiratory tract infections. Journal of Infectious Diseases. 2005;192:1061–1065. doi: 10.1086/432732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Peck AJ, Englund JA, Kuypers J, Guthrie KA, Corey L, Morrow R, Hackman RC, Cent A, Boeckh M. Respiratory virus infection among hematopoietic cell transplant recipients: evidence for asymptomatic parainfluenza virus infection. Blood. 2007;110:1681–1688. doi: 10.1182/blood-2006-12-060343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Renaud C, Campbell AP. Changing epidemiology of respiratory viral infections in hematopoietic cell transplant recipients and solid organ transplant recipients. Current Opinion in Infectious Diseases. 2011;24:333–343. doi: 10.1097/QCO.0b013e3283480440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Englund JA, Boeckh M, Kuypers J, Nichols WG, Hackman RC, Morrow RA, Fredricks DN, Corey L. Brief communication: fatal human metapneumovirus infection in stem-cell transplant recipients. Annals of Internal Medicine. 2006;144:344–349. doi: 10.7326/0003-4819-144-5-200603070-00010. [DOI] [PubMed] [Google Scholar]
- 5.Wyde PR, Chetty SN, Jewell AM, Boivin G, Piedra PA. Comparison of the inhibition of human metapneumovirus and respiratory syncytial virus by ribavirin and immune serum globulin in vitro. Antiviral Res. 2003;60:51–59. doi: 10.1016/s0166-3542(03)00153-0. [DOI] [PubMed] [Google Scholar]
- 6.Hamelin ME, Prince GA, Boivin G. Effect of ribavirin and glucocorticoid treatment in a mouse model of human metapneumovirus infection. Antimicrob Agents Chemother. 2006;50:774–777. doi: 10.1128/AAC.50.2.774-777.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Pelletier G, Déry P, Abed Y, Boivin G. Respiratory tract reinfections by the new human Metapneumovirus in an immunocompromised child. Emerg Infect Dis. 2002;8:976–978. doi: 10.3201/eid0809.020238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Cane PA, van den Hoogen BG, Chakrabarti S, Fegan CD, Osterhaus ADME. Human metapneumovirus in a haematopoietic stem cell transplant recipient with fatal lower respiratory tract disease. Bone Marrow Transplantation. 2003;31:309–310. doi: 10.1038/sj.bmt.1703849. [DOI] [PubMed] [Google Scholar]
- 9.Huck B, Egger M, Bertz H, Peyerl-Hoffman G, Kern WV, Neumann-Haefelin D, Falcone V. Human metapneumovirus infection in a hematopoietic stem cell transplant recipient with relapsed multiple myeloma and rapidly progressing lung cancer. Journal of Clinical Microbiology. 2006;44:2300–2303. doi: 10.1128/JCM.00152-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kamble RT, Bollard C, Demmler G, LaSala PR, Carrum G. Human metapneumovirus infection in a hematopoietic transplant recipient. Bone Marrow Transplantation. 2007;40:699–700. doi: 10.1038/sj.bmt.1705781. [DOI] [PubMed] [Google Scholar]
- 11.Muller A, Kupfer B, Vehreschild J, Cornely O, Kaiser R, Seifert H, Viazov S, Tillmann RL, Franzen C, Simon A, Schildgen O. Fatal pneumonia associated with human metapneumovirus (HMPV) in a patient with myeloid leukemia and adenocarcinoma in the lung. European Journal of Medical Research. 2007;12:183–184. [PubMed] [Google Scholar]
- 12.Abed Y, Boivin G. Human metapneumovirus infection in immunocompromised child. Emerging Infectious Diseases. 2008;14 doi: 10.3201/eid1405.071459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bonney D, Razali H, Turner A, Will A. Successful treatment of human metapneumovirus pneumonia using combination therapy with intravenous ribavirin and immune globulin: Correspondence. British Journal of Haematology. 2009;145:667–669. doi: 10.1111/j.1365-2141.2009.07654.x. [DOI] [PubMed] [Google Scholar]
- 14.Shahda S, Carlos WG, Kiel PJ, Khan BA, Hage CA. The human metapneumovirus: a case series and review of the literature. Transplant Infectious Disease. 2011;13:324–328. doi: 10.1111/j.1399-3062.2010.00575.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Shachor-Meyouhas Y, Ben-Barak A, Kassis I. Treatment with oral ribavirin and IVIG of severe human metapneumovirus pneumonia (HMPV) in immune compromised child. Pediatric Blood and Cancer. 2011;57:350–351. doi: 10.1002/pbc.23019. [DOI] [PubMed] [Google Scholar]
- 16.van Dijk NM, Linssen CFM, Bussink M, Span LFR, van Mook WNKA. Critical illness after horizontal nosocomial transmission of human metapneumovirus in the haematology ward. Netherlands Journal of Critical Care. 2012;16:22–25. [Google Scholar]
- 17.Kamboj M, Gerbin M, Huang C-K, Brennan C, Stiles J, Balashov S, Park S, Kiehn TE, Perlin DS, Pamer EG, Sepkowitz KA. Clinical characterization of human metapneumovirus infection among patients with cancer. Journal of Infection. 2008;57:464–471. doi: 10.1016/j.jinf.2008.10.003. [DOI] [PubMed] [Google Scholar]
- 18.Campbell AP, Chien JW, Kuypers J, Englund JA, Wald A, Guthrie KA, Corey L, Boeckh M. Respiratory Virus Pneumonia after Hematopoietic Cell Transplantation (HCT): Associations between Viral Load in Bronchoalveolar Lavage Samples, Viral RNA Detection in Serum Samples, and Clinical Outcomes of HCT. Journal of Infectious Diseases. 2010;201:1404–1413. doi: 10.1086/651662. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hofmeyr A, Dunlop L, Ling S, Fiakos E, Maley M. Ribavirin treatment for human metapneumovirus and methicillin-resistant Staphylococcus aureus co-infection in adult haematological malignancy. Clinical Microbiology and Infection. 2011;17:S812. [Google Scholar]
- 20.Seo S, Gooley TA, Kuypers JM, Stednick Z, Jerome KR, Englund JA, Boeckh M. Human Metapneumovirus Infections following Hematopoietic Cell Transplantation: Factors Associated with Disease Progression. Clin Infect Dis. 2016 doi: 10.1093/cid/ciw284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.El Chaer F, Shah DP, Kmeid J, Ariza-Heredia E, Hosing C, Chemaly RF. ID Week. San Diego, California: 2015. Human Metapenumovirus (hMPV) infections in 135 cancer patients. [Google Scholar]
- 22.Muller A, Klinkenberg D, Vehreschild J, Cornely O, Tillmann RL, Franzen C, Simon A, Schildgen O. Low prevalence of human metapneumovirus and human bocavirus in adult immunocompromised high risk patients suspected to suffer from Pneumocystis pneumonia. Journal of Infection. 2009;58:227–231. doi: 10.1016/j.jinf.2009.01.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Godet C, Le Goff J, Beby-Defaux A, Robin M, Raffoux E, Arnulf B, Roblot F, Frat JP, Maillard N, Tazi A, Bergeron A. Human metapneumovirus pneumonia in patients with hematological malignancies. J Clin Virol. 2014;61:593–596. doi: 10.1016/j.jcv.2014.08.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Span NM, van Dijk NM, Linssen CFM, van Mook WNKA WNKA. Human metapneumovirus in immunocompromised patients with hAematological malignancies: awareness with caution and respect! Bone Marrow Transplantation. 2009;43:S59. [Google Scholar]
- 25.Egli A, Bucher C, Dumoulin A, Stern M, Buser A, Bubendorf L, Gregor M, Servida P, Sommer G, Bremerich J, Gratwohl A, Khanna N, Widmer AF, Battegay M, Tamm M, Hirsch HH, Halter JP. Human metapneumovirus infection after allogeneic hematopoietic stem cell transplantation. Infection. 2012;40:677–684. doi: 10.1007/s15010-012-0279-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Renaud C, Xie H, Seo S, Kuypers J, Cent A, Corey L, Leisenring W, Boeckh M, Englund JA. Mortality rates of human metapneumovirus and respiratory syncytial virus lower respiratory tract infections in hematopoietic cell transplantation recipients. Biol Blood Marrow Transplant. 2013;19:1220–1226. doi: 10.1016/j.bbmt.2013.05.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Ali M, Baker J, Richardson SE, Allen U, Abla O. Human metapneumovirus infection in children with cancer. Blood; Conference: 52nd Annual Meeting of the American Society of Hematology, ASH.2010. [Google Scholar]
- 28.Safdar A. Immune modulatory activity of ribavirin for serious human metapneumovirus disease: early i.v. therapy may improve outcomes in immunosuppressed SCT recipients. Bone Marrow Transplantation. 2008;41:707–708. doi: 10.1038/bmt.2008.80. [DOI] [PubMed] [Google Scholar]
- 29.Hirsch HH, Martino R, Ward KN, Boeckh M, Einsele H, Ljungman P. Fourth European Conference on Infections in Leukaemia (ECIL-4): guidelines for diagnosis and treatment of human respiratory syncytial virus, parainfluenza virus, metapneumovirus, rhinovirus, and coronavirus. Clin Infect Dis. 2013;56:258–266. doi: 10.1093/cid/cis844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Tzannou I, Nicholas S, Misra A, Katari U, Orange J, Vera J, Heslop H, Rooney C, Leen A. Biology of Blood and Marrow Transplantation. San Diego, CA: 2015. Adoptive T-Cell Therapy to Prevent and Treat Human Metapneumovirus (hMPV) Infections Post Hematopoietic Stem Cell Transplant (HSCT) BMT Tandem Meeting; p. S170. [Google Scholar]
- 31.Hamelin ME, Couture C, Sackett M, Kiener P, Suzich J, Ulbrandt N, Boivin G. The prophylactic administration of a monoclonal antibody against human metapneumovirus attenuates viral disease and airways hyperresponsiveness in mice. Antivir Ther. 2008;13:39–46. [PubMed] [Google Scholar]
- 32.Ulbrandt ND, Ji H, Patel NK, Riggs JM, Brewah YA, Ready S, Donacki NE, Folliot K, Barnes AS, Senthil K, Wilson S, Chen M, Clarke L, MacPhail M, Li J, Woods RM, Coelingh K, Reed JL, McCarthy MP, Pfarr DS, Osterhaus AD, Fouchier RA, Kiener PA, Suzich JA. Isolation and characterization of monoclonal antibodies which neutralize human metapneumovirus in vitro and in vivo. J Virol. 2006;80:7799–7806. doi: 10.1128/JVI.00318-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Williams JV, Chen Z, Cseke G, Wright DW, Keefer CJ, Tollefson SJ, Hessell A, Podsiad A, Shepherd BE, Sanna PP, Burton DR, Crowe JE, Williamson RA. A recombinant human monoclonal antibody to human metapneumovirus fusion protein that neutralizes virus in vitro and is effective therapeutically in vivo. J Virol. 2007;81:8315–8324. doi: 10.1128/JVI.00106-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Hamelin ME, Gagnon C, Prince GA, Kiener P, Suzich J, Ulbrandt N, Boivin G. Prophylactic and therapeutic benefits of a monoclonal antibody against the fusion protein of human metapneumovirus in a mouse model. Antiviral Res. 2010;88:31–37. doi: 10.1016/j.antiviral.2010.07.001. [DOI] [PubMed] [Google Scholar]
- 35.Lévy C, Aerts L, Hamelin M, Granier C, Szécsi J, Lavillette D, Boivin G, Cosset FL. Virus-like particle vaccine induces cross-protection against human metapneumovirus infections in mice. Vaccine. 2013;31:2778–2785. doi: 10.1016/j.vaccine.2013.03.051. [DOI] [PubMed] [Google Scholar]
- 36.Debiaggi M, Canducci F, Terulla C, Sampaolo M, Marinozzi MC, Alessandrino PE, Colombo AA, Caldera D, Bragotti LZ, Migliavacca R, Bianchi E, Romero E, Clementi M. Long-term study on symptomless human metapneumovirus infection in hematopoietic stem cell transplant recipients. New Microbiologica. 2007;30:255–258. [PubMed] [Google Scholar]
- 37.Debiaggi M, Canducci F, Sampaolo M, Marinozzi MC, Parea M, Terulla C, Colombo AA, Alessandrino EP, Bragotti LZ, Arghittu M, Goglio A, Migliavacca R, Romero E, Clementi M. Persistent symptomless human metapneumovirus infection in hematopoietic stem cell transplant recipients. Journal of Infectious Diseases. 2006;194:474–478. doi: 10.1086/505881. [DOI] [PubMed] [Google Scholar]
- 38.Lee JH, Jang JH, Yoo KH, Sung KW, Koo HH, Lee NY, Ki CS. Incidence, clinical features, and outcome of respiratory virus infections in paediatric patients after haematopoietic stem cell transplantation. Bone Marrow Transplantation. 2011;46:S228. [Google Scholar]
- 39.Fazekas T, Eickhoff P, Rauch M, Verdianz M, Attarbaschi A, Dworzak M, Peters C, Hammer K, Vecsei A, Potschger U, Lion T. Prevalence and clinical course of viral upper respiratory tract infections in immunocompromised pediatric patients with malignancies or after hematopoietic stem cell transplantation. Journal of Pediatric Hematology/Oncology. 2012;34:442–449. doi: 10.1097/MPH.0b013e3182580bc8. [DOI] [PubMed] [Google Scholar]
- 40.Srinivasan A, Gu ZM, Smith T, Morgenstern M, Sunkara A, Kang GL, Srivastava DK, Gaur AH, Leung W, Hayden RT. Prospective Detection of Respiratory Pathogens in Symptomatic Children With Cancer. Pediatric Infectious Disease Journal. 2013;32:E99–E104. doi: 10.1097/INF.0b013e31827bd619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Choi JH, Choi EH, Kang HJ, Park KD, Park SS, Shin HY, Lee HJ, Ahn HS. Respiratory viral infections after hematopoietic stem cell transplantation in children. Journal of Korean Medical Science. 2013;28:36–41. doi: 10.3346/jkms.2013.28.1.36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Martino R, Porras RP, Rabella N, Williams JV, Ramila E, Margall N, Labeaga R, Crowe JE, Coll P, Sierra J. Prospective study of the incidence, clinical features, and outcome of symptomatic upper and lower respiratory tract infections by respiratory viruses in adult recipients of hematopoietic stem cell transplants for hematologic malignancies. Biology of Blood and Marrow Transplantation. 2005;11:781–796. doi: 10.1016/j.bbmt.2005.07.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Souza JS, Watanabe A, Carraro E, Granato C, Bellei N. Severe metapneumovirus infections among immunocompetent and immunocompromised patients admitted to hospital with respiratory infection. J Med Virol. 2013;85:530–536. doi: 10.1002/jmv.23477. [DOI] [PubMed] [Google Scholar]
- 44.Debur MC, Vidal LR, Stroparo E, Nogueira MB, Almeida SM, Takahashi GA, Rotta I, Pereira LA, Silveira CS, Bonfim CM, Raboni SM. Human metapneumovirus infection in hematopoietic stem cell transplant recipients. Transplant Infectious Disease. 2010;12:173–179. doi: 10.1111/j.1399-3062.2009.00465.x. [DOI] [PubMed] [Google Scholar]
- 45.Wolfromm A, Porcher R, Legoff J, De Latour RP, Xhaard A, Ribaud P, Scieux C, Bergeron A, Socie G, Robin M. Viral respiratory infections after allogeneic hemaotpoietic stem cell transplantation: Outcome and prognosis. Blood; Conference: 53rd Annual Meeting of the American Society of Hematology, ASH.2011. [Google Scholar]
- 46.Oliveira RR, Machado AF, Tateno AF, Boas LSV, Pannuti CS, Machado CM. Frequency of human metapneumovirus infection in hematopoietic SCT recipients during 3 consecutive years. Bone Marrow Transplantation. 2008;42:265–269. doi: 10.1038/bmt.2008.153. [DOI] [PubMed] [Google Scholar]
- 47.Mikulska M, Del Bono V, Dini S, Raiola A, Dominietto A, Bregante S, Van Lint M, Orsi A, Ansaldi F, Bacigalupo A, Viscoli C. Respiratory viral infections after allogeneic haematopoietic stem cell transplantation. Clinical Microbiology and Infection. 2011;17:S615. [Google Scholar]
- 48.Chebotkevich V. The challenge of respiratory viral infections in onco-haematological patients. Clinical Microbiology and Infection. 2010;16:S705. [Google Scholar]
- 49.Kwon JC, Cho SY, Kim SH, Park SH, Choi SM, Lee DG, Choi JH, Yoo JH. Microbiology from bronchoscopy in haematologic patients: Comparison of stem cell recipients with nonrecipients. Clinical Microbiology and Infection. 2012;18:616. [Google Scholar]
- 50.Park SY, Baek S, Lee SO, Choi SH, Kim YS, Woo JH, Sung H, Kim MN, Kim DY, Lee JH, Lee KH, Kim SH. Efficacy of oral ribavirin in hematologic disease patients with paramyxovirus infection: Analytic strategy using propensity scores. Antimicrobial Agents and Chemotherapy. 2013;57:983–989. doi: 10.1128/AAC.01961-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Ghattas C, Mossad SB. Fatal human metapneumovirus and influenza B virus coinfection in an allogeneic hematopoietic stem cell transplant recipient. Transpl Infect Dis. 2012;14:E41–E43. doi: 10.1111/j.1399-3062.2012.00766.x. [DOI] [PubMed] [Google Scholar]
- 52.Dokos C, Masjosthusmann K, Rellensmann G, Werner C, Schuler-Lüttmann S, Müller KM, Schiborr M, Ehlert K, Groll AH. Fatal human metapneumovirus infection following allogeneic hematopoietic stem cell transplantation. Transpl Infect Dis. 2013;15:E97–E101. doi: 10.1111/tid.12074. [DOI] [PubMed] [Google Scholar]
- 53.Kitanovski L, Kopriva S, Pokorn M, Dolničar MB, Rajić V, Stefanović M, Jazbec J. Treatment of severe human metapneumovirus (hMPV) pneumonia in an immunocompromised child with oral ribavirin and IVIG. J Pediatr Hematol Oncol. 2013;35:e311–e313. doi: 10.1097/MPH.0b013e3182915d2d. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.