Abstract
Exploring drug targets based on disease-associated molecular mechanisms during development is crucial for the generation of novel prevention and treatment strategies for neurodevelopmental psychiatric conditions. We report that prefrontal cortex-specific postnatal knockdown of DISC1 via in utero electroporation combined with an inducible knockdown expression system drives deficits in synaptic GABAA function and dendritic development in pyramidal neurons, as well as abnormalities in sensorimotor gating, albeit without profound memory deficits. We show for the first time that DISC1 is specifically involved in regulating cell surface expression of α2 subunit-containing GABAA receptors in immature developing neurons, but not after full maturation. Notably, pharmacological intervention with α2/3 subtype-selective GABAA receptor positive allosteric modulators during the early postnatal period ameliorates dendritic deficits and behavioral abnormalities induced by knockdown of DISC1. These findings highlight a critical role of DISC1-mediated disruption of postnatal GABA signaling in aberrant prefrontal cortex maturation and function.
INTRODUCTION
γ-aminobutyric acid type A (GABAA) receptors are responsible for the majority of fast synaptic inhibition in the mature central nervous system.1 During pre and early postnatal periods, GABA exerts a depolarizing and excitatory action, regulating multiple processes of neuronal maturation, including dendritic development.2–4 Notably, dendritic abnormalities and deficits in GABA signaling, including alteration of GABAA receptors have been implicated in multiple neurodevelopmental psychiatric disorders, such as autism spectrum disorder, epilepsy, and schizophrenia.5–7 Accordingly, developmental GABAA receptor-mediated signaling is a unique molecular target to explore novel pharmacological treatment for these devastating conditions.
Many pharmacological agents targeting GABAA receptors are clinically available.5,8 Most prominently, benzodiazepines have diverse therapeutic actions by enhancing GABAA receptor channel functions.8 Nonetheless, due to their serious adverse effects including sedation, cognitive impairment, and potential for addiction/abuse, development of new positive modulators of GABAA receptors with lesser side effects is awaited.5,8 Many of these aversive effects are most likely due to activation of α1 subunit-containing GABAA receptor,8,9 leading researchers in academia and pharmaceutical companies to explore subtype-selective compounds without activity at α1 subunit-containing GABAA receptors.5,8 Although a number of subtype-selective GABAA receptor compounds have been tested in patients with neuropsychiatric conditions and animal models in adults,8,10–12 their action during earlier developmental phases has been poorly investigated.
In the present study, we explore the effect of postnatal intervention of subtype-selective positive allosteric modulators of α2/3-containing GABAA receptors on developmental deficits caused by knockdown of Disrupted-in-Schizophrenia-1 (DISC1), a genetic risk factor for major mental disorders.13 DISC1 is involved in multiple cellular processes in the developing cerebral cortex.14,15 A functional interaction of DISC1 and NKCC1, a cation-chloride cotransporter which plays a role in excitatory GABA function in hippocampal neurogenesis, has been demonstrated.16 In order to explore the specific role of DISC1 for developmental GABAA receptor-mediated signaling in the prefrontal cortex (PFC) during postnatal periods, we have developed a modified conditional knockdown method by using in utero electoroporation. This system allows us to suppress DISC1 expression, specifically in pyramidal neurons in the PFC during postnatal periods. This technique is an alternative method to genetically engineered animals, since region-specific genetic deletion is currently impractical.17–19 Our data demonstrates that postnatal knockdown of DISC1 impairs developmental GABAA receptor-mediated signaling, which can be reversed by subtype-selective positive allosteric small molecule modulators of GABAA receptors during the early postnatal period.
MATERIALS AND METHODS
In utero electroporation with a new electrode with a three-electrode configuration
In utero electroporation targeting the prefrontal cortex (PFC) region was performed by our published protocol with some modifications.20 Pregnant C57/BL6 mice (Charles River) were anesthetized at embryonic day 14.5 (E14.5) by intraperitoneal administration of a mixed solution of Ketamine HCl (100 mg/kg), Xylazine HCl (7.5 mg/kg), and Buprenorphine HCl (0.05 mg/kg). After the uterine horn was exposed by laparotomy, inducible shRNA to DISC1 (2 µg/µl) and CALNL-GFP (1 µg/µl) together with CAG-ERT2CreERT2 (1 µg/µl) (molar ratio, approximately 3:1:1) were injected into the bilateral ventricles with a glass micropipette made from a microcapillary tube (GD-1; Narishige). The embryo’s head in the uterus was held between the custom-made electrode, consisting of one positive and two negative pole disc-type electrodes (Nepagene). Electrode pulses (33V; 50 ms) were charged four times at intervals of 950 ms with an electroporator (CUY21EDIT; Nepagene). After electroporation, the uterine horn was replaced in the abdominal cavity to allow the embryos to continue to develop. Brains extracted from DISC1 knockdown and control animals were assessed to confirm GFP expression in proper PFC regions after behavioral characterization. All experiments were performed in accordance with the institutional guidelines for animal experiments.
Dendrite analysis
Dendrites of GFP-positive pyramidal neurons in layer II/III in medial PFC at postnatal day 7 (P7) were analyzed morphologically, according to modifications of previously published protocols.20,21 Z stacks of images were collected with a confocal microscope (LSM 510 Meta, Zeiss). Images of cells are taken using the 40 × oil-immersion objective lens as Z-series of images, averaged four times, taken at 0.8-µm intervals, 1024 × 1024 pixel resolution at a scan speed of 7 sec per section. Acquisition parameters were kept the same for all scans. Images were reconstructed by compressing the Z stacks, and then analyzed by using NeuronJ, plug-in software for ImageJ (http://www.imagescience.org/meijering/software/neuronj/) to trace primary, secondary, and tertiary branches of both apical and basal dendrites, and the number of dendrites per each category was calculated. In order to trace dendrites in a single GFP-positive neuron, the GFP-positive dendrites originating from other adjacent GFP-positive neurons were excluded based on observation of uncompressed z-scan images. In each condition, 10 neurons from independent brain slices were analyzed in a blinded manner. For quantification of dendritic complexity in pharmacological in vivo rescue experiments, a region of interest (ROI) was randomly selected (800 µm wide × 400 µm high) in layers I–III of medial PFC where GFP-positive cells were located. A z-series of 10 images were collected at 6.02-µm depth intervals with a confocal microscope (LSM710NLO-Meta, Zeiss). Acquisition parameters were kept the same for all images. Intensity of the GFP signal in the whole area and cell bodies in the ROI was measured from three images for each brain (total 10 brain sections for vehicle-treated mice and 10 brain sections from AZD7325-treated mice) by ImageJ software. The intensity ratio of GFP fluorescence of the processes (i.e., GFP signal in cell bodies subtracted from that in the whole area) to GFP fluorescence in the whole area was analyzed. Dendritic structure in cortical neurons at 7 DIV was analyzed by the previously published method with some modifications.22 The images of GFP-positive neurons were acquired with a florescence microscope (LSM510 Meta, Zeiss) in a blinded manner. We analyzed only neurons with typical morphological characteristics of pyramidal neurons (single axon and multiple dendrites) by excluding neurons which have atypical morphological characteristics, such as multiple axons or atypical neuronal processes. The total length of dendrites and dendrite branch number were determined by using NeuronJ, plug-in software for ImageJ. Dendritic complexity was analyzed by using sholl analysis. The number of dendrites that crossed a series of concentric circles at 40-µm intervals from the soma was counted.
Treatment with GABAA positive allosteric modulator compounds
For in vitro assay, cortical neuron cultures expressing pSuper shRNA were treated daily with AZD7325 or AZD6280 (0.1 µM final concentration) for 7 days. To assess their effect on dendritic development in vivo, AZD7325 (10 µmol/kg body weight) with 40% (2-hydroxypropyl)-β-cyclodextrin or vehicle were injected intraperitoneally into postnatal DISC1 suppression mice for 1 week starting at postnatal day 4 (P4). Their pharmacological effects on dendritic structures and behavioral phenotypes were then assessed at P14 and 3 months old, respectively.
Details on the other experiments are provided in Supplementary Materials and Methods.
RESULTS
DISC1 is involved in cell surface expression of GABAA receptors in developing neurons, but not in mature neurons
GABAA receptors are heteropentameric ligand-gated ion channels, which are mainly composed of two α, two β, and one γ subunits in the brain.23 We first examined whether knockdown of DISC1 induced by lentivirus-based shRNA affects total and cell surface expression of α, β, and γ subunits of GABAA receptors in developing cultured cortical neurons. The knockdown effect of lentivirus-based shRNA to DISC1 on endogenous DISC1 was confirmed in developing cortical neurons (Supplementary Figure 1). It is noted that we have previously confirmed knockdown effect of the same lentivirus-based shRNA to DISC1 on endogenous DISC1 in mature neurons.24 Knockdown of DISC1 reduced cell surface expression of α2, β2, β3, and γ2 subunits, but not α1, α3, and α5 subunits, in developing cortical neurons at 7 days in vitro (7 DIV), whereas there were no changes in their total expression under these conditions (Figure 1a and Supplementary Figure 2). This was reversed by co-expressing RNAi-resistant wild-type DISC1 (Figure 1a). Decreased cell surface expression of these GABAA receptor subunits was also confirmed in the developing pyramidal neurons by immunocytochemical staining (Figure 1b). Neither total nor cell surface expression of the cation-chloride cotransporters, NKCC1 and KCC2, which are two major determinants for the excitatory and inhibitory neuronal response to GABA,2,25 were changed (Supplementary Figure 2). Notably, when DISC1 expression was suppressed in mature cortical neurons at 30 DIV, no difference in total or cell surface expression of these GABAA receptor subunits was observed between DISC1 knockdown and control neurons (Figure 1c). These results suggest that suppression of DISC1 expression specifically impairs cell surface expression of α2 subunit-containing GABAA receptors during early developmental periods.
Figure 1.
Suppression of DISC1 expression impairs cell surface expression of GABAA receptor in developing neurons, but not in mature neurons. (a) Reduction of cell surface expression of GABAA receptor by knockdown of DISC1 (DISC1 RNAi) in developing cortical neurons at 7 days in vitro (7 DIV). Cell surface proteins were labeled by biotinylation, precipitated by avidin-conjugated beads, and immunoblotted for GABAA receptor α2, β2/3, and γ2 subunits. Cell surface expression of GABAA receptor α2, β2/3, and γ2 subunits (red arrowheads) was reduced by knockdown of DISC1 (left panels), but there was no change in their total expression (right panels). Reduction of their cell surface expression was reversed by co-expression of RNAi-resistant DISC1 (DISC1R). (b) Reduction of cell surface expression of GABAA receptors by immunocytochemistry with antibodies against α2, β3, and γ2 subunits (red traced by dotted white lines) under nonpermeant conditions in developing cortical neurons with knockdown of DISC1 (green) at 7 DIV. Blue, nucleus. Scale bar, 20 µm. (c) Knockdown of DISC1 has no effect on total or cell surface expression of GABAA receptors in mature cortical neurons at 30 DIV. TfR, transferrin receptor. (d) Impairment of GABAA receptor-dependent increase in [Ca2+] by knockdown of DISC1 in developing cortical neurons. Calcium imaging was assessed using the developing primary cortical neurons at 7 DIV transfected with GCaMP3 (green) as well as shRNAs to DISC1 (DISC1 RNAi) or scrambled controls (Control RNAi), which carry scarlet protein (red). Scale bar, 20 µm. (e) Bath application of GABA (50 µM) elicited increase in intracellular Ca2+ concentration in neurons with control RNAi, whereas knockdown of DISC1 impaired Ca2+ influx. Increase of calcium influx induced by GABA was prevented by treatment with bicuculline (10 µM). n = 10 neurons per condition. Bars represent averages of each group in three independent experiments (a, c). *P < 0.05, **P < 0.01, ***P < 0.001 determined by one-way ANOVA with posthoc Bonferroni test (a, e) and Student’s t test (c)
To monitor whether DISC1 is involved in regulation of intracellular calcium concentration via GABA signaling, calcium imaging experiments were conducted in developing cortical neuron cultures expressing the genetically-encoded calcium indicator GCaMP3 together with shRNA to DISC1 or control scrambled shRNA, which carry a scarlet protein (Figure 1d). Consistent with previous studies, bath application of GABA evoked increases of intracellular calcium in neurons expressing control shRNA at 7 DIV,26 whereas knockdown of DISC1 diminished the effect of GABA administration on calcium influx (Figure 1e). We confirmed that GABA-induced increase of intracellular calcium was mediated by GABAA receptor, as treatment with bicuculline, a GABAA receptor antagonist, blocks the effect (Figure 1e).
Postnatal suppression of DISC1 expression by selective targeting of pyramidal neurons disrupts dendritic development in the PFC
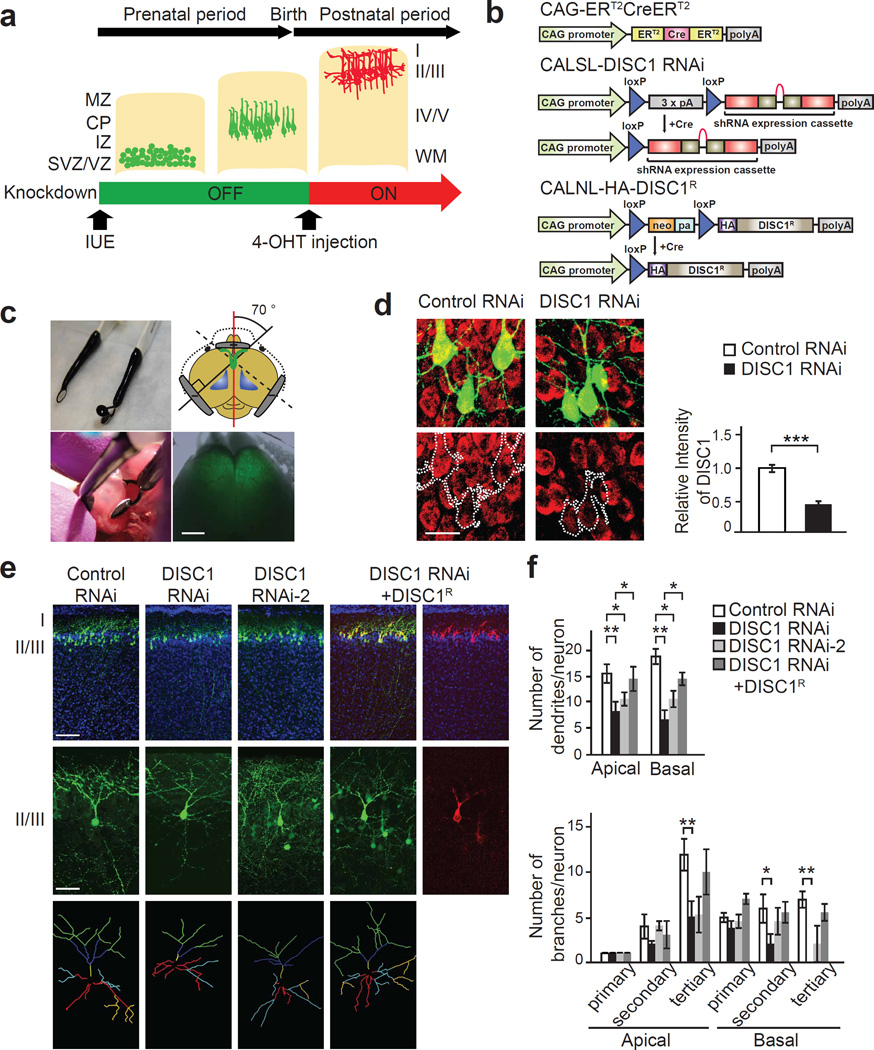
In order to investigate the effect of DISC1 in the PFC, specifically in the postnatal period, we utilized in utero electroporation with a previously developed Cre/loxP-mediated inducible gene expression system (Figure 2a and b)27. With this system, a stop codon flanked by two LoxP sites downstream from the CAG promoter is contained in microRNA30 (mir30)-based short hairpin RNA (shRNA) and overexpression plasmids. Cre recombinase fused to the mutated estrogen receptor is expressed under the control of the CAG promoter (CAG-ERT2CreERT2), in which Cre is only active in the presence of 4-hydroxytamoxifen (4-OHT). We designed specific conditional shRNAs to DISC1 with independent target sequences, containing the sequences of which knockdown effects on DISC1 expression were previously characterized by conventional shRNAs and lentivirus-mediated RNAi approaches.20,24,28–30 We also made an inducible plasmid expressing HA-tagged RNAi-resistant wild-type mouse DISC1, in which four silent mutations are contained in the target sequence of shRNA to DISC1 (CALNL-HA-DISC1R).28,30 We confirmed the inducible knockdown effect of DISC1 on exogenously overexpressed wild-type mouse DISC1 and overexpression of RNAi-resistant wild-type mouse DISC1 in HEK 293 cells treated with 4-OHT (Supplementary Figure 3), whose efficacies are comparable to lentivirus-based shRNA to DISC1 (Supplementary Figure 1). To manipulate targeted gene expression in the bilateral PFC, we have developed a modified in utero electroporation method with a new electrode, which overcomes inefficiencies of our previous method which required a double electroporation step.20 The new electrode has a three-pole configuration, composed of one positive pole and two negative poles, which was confirmed to target the bilateral PFC more precisely (Figure 2c). In the developing cerebral cortex, pyramidal neurons originate from the ventricular zone and migrate radially into the cerebral cortex.18,19 Given that interneurons migrate tangentially from a ganglionic eminence31 and glial cells are produced at late embryonic and early postnatal days,32 the plasmid constructs introduced by in utero electroporation in the mid-embryonic periods are exclusively integrated into a lineage of pyramidal neurons as previously shown.20 The efficacy of the modified technique with the new electrode was tested by a single electroporation of the inducible overexpression plasmid of green fluorescent protein (CALNL-GFP) and CAG-ERT2CreERT2 into PFC at embryonic day 14.5 (E14.5), followed by intraperitoneal injection of 4-OHT at postnatal day 2 (P2). We confirmed that GFP was extensively expressed in the bilateral PFC areas, simultaneously, at P7 (Figure 2c).
Figure 2.
Postnatal suppression of DISC1 expression by selective targeting of pyramidal neurons in the prefrontal cortex (PFC) impairs dendritic development. (a) Schematic representation showing gene targeting by using the Cre/loxP system via in utero electroporation. Cells expressing inducible plasmids (green) that have migrated to the proper cortical layer; red cells represent post-migratory neurons, in which inducible plasmids were activated by intraperitoneal injection of 4-hydroxytamoxifen (4-OHT) during the postnatal period. MZ, marginal zone; CP, cortical plate; IZ, intermediate zone; SVZ/VZ, subventricular zone/ventricular zone. IUE, in utero electroporation. (b) 4-OHT activates Cre recombinase (Cre) comprising two mutated estrogen receptor (ER) binding regions of CAG-ERT2CreERT2 vector. Mir30-based shRNAs to DISC1 and HA-tagged RNAi-resistant DISC1 are expressed under control of the CAG promoter in the presence of 4-OHT-activated Cre (CALSL-DISC1 RNAi and CALNL-HA-DISC1R). (c) Three-electrode configuration for in utero electroporation to target the bilateral PFC. By using the custom-made electrode (upper panels), CALNL-GFP was simultaneously electroporated into the right and left side of PFC at embryonic day 14.5 (E14.5) and GFP expression was confirmed in the bilateral PFC at postnatal day 7 (P7) (lower panels). Scale bar, 1 mm. (d) Inducible DISC1 RNAi or Control RNAi were co-introduced with CAG-ERT2CreERT2 and CALNL-GFP in the PFC at E14.5 followed by intraperitoneal injection of 4-OHT at P2. Efficient suppression of DISC1 immunoreactivity (red traced by dotted white lines) in GFP-positive pyramidal neurons (green) is shown in the medial PFC at P7. n = 30 neurons from three mice per condition. Scale bar, 10 µm. (e, f) Mice with postnatal knockdown of DISC1 in pyramidal neurons of the medial PFC show impaired dendritic development, but not radial neuronal migration. Total number of dendrites and dendrite branches in each category (primary, secondary, and tertiary of apical and basal dendrites) was analyzed at P7. DISC1 RNAi has a stronger effect than RNAi-2. Inducible co-expression with RNAi-resistant DISC1 (red) normalized dendritic abnormalities induced by knockdown of DISC1. In each condition, 10 neurons from independent brain slices were analyzed in a blinded manner. Traced dendrites were categorized as primary apical (yellow) and basal (red), secondary apical (blue) and basal (cyan) dendrites, as well as tertiary apical (green) and basal (yellow) dendrites, respectively. Only neurons with typical morphological characteristics of pyramidal neurons (single thick axon arising from the basal side of the soma with multiple basal dendrites and apical dendrites extending vertically to layer I) were analyzed. Green, GFP; blue, nucleus. Scale bar, 100 µm (upper panels), 20 µm (middle panels). *P < 0.05, **P < 0.01, and ***P < 0.001 determined by Student’s t test (d) and one-way ANOVA with posthoc Bonferroni test (f).
We adapted this device to test the in vivo knockdown effect of DISC1 during the postnatal period by use of the Cre/loxP-mediated expression system. Inducible shRNA to DISC1 (DISC1 RNAi) or scrambled control shRNA (Control RNAi), together with CALNL-GFP and CAG-ERT2CreERT2, were electroporated into the developing PFC at E14.5, and pups were given 4-OHT at P2, when early postnatal period dendrite growth occurs. We have previously reported that in utero electroporation can be used to specifically target approximately 20 % of pyramidal neurons in layers II/III of the PFC.20 Consistently, the present study found that GFP-positive cells are confined to CaMKII-positive pyramidal neurons, approximately 20% of which are GFP-positive in layers II/III of the medial PFC (Supplementary Figure 4). Knockdown of endogenous DISC1 in pyramidal neurons in the medial PFC was confirmed at P7 via immunohistochemistry, utilizing a previously characterized DISC1 antibody (Figure 2d).33
Dendritic structure is regulated by multiple mechanisms, including GABAA receptor-mediated signaling in early postnatal periods,2,3 and is a critical component for neuronal information processing through synaptic integration.34 To evaluate the importance of DISC1 in dendritic growth specifically in post-migratory neurons, we monitored the effects of postnatal suppression of DISC1 expression on dendritic development in the medial PFC by use of the aforementioned Cre/loxP-mediated expression system via in utero electroporation. When observed at P7 in the medial PFC, a period of robust dendritic growth, numbers of apical and basal dendrites, as well as secondary branches of basal dendrites and tertiary branches of both apical and basal dendrites were reduced by the postnatal knockdown of DISC1, without deficits in radial neuronal migration (Figure 2e, f, and Supplementary Figure 5). The reduction in number of dendrites and branches was seen in a knockdown effect-dependent manner and reversed by inducible co-expression of RNAi-resistant wild-type DISC1 (Figure 2e and f), suggesting that the observed phenotype is specifically due to knockdown of DISC1. We previously reported that conventional knockdown of DISC1 in the medial PFC starting at prenatal periods results in reduction of immunoreactivity of paravalbumin (PV), a marker of the fast spiking GABAergic interneurons.20 When DISC1 expression was suppressed at the postnatal periods by inducible knockdown approaches, we observed no significant change in the number of PV-positive neurons in the medial PFC at young adulthood (Supplementary Figure 6), indicating that knockdown of DISC1 in the postnatal periods is not sufficient for impairment of PV-positive neuron maturation.
Postnatal suppression of DISC1 expression in the PFC impairs developmental synaptic GABA function
In order to examine the importance of postnatal expression of DISC1 for synaptic expression of α2 subunit-containing GABAA receptors under in vivo conditions, we analyzed cellular distribution of GABAA receptors and gephyrin, a GABA synaptic scaffold protein in the medial PFC of mice with postnatal suppression of DISC1 expression. Although inducible knockdown of DISC1 does not affect the number of gephyrin puncta and colocalization of gephyrin and vesicular GABA transporter (vGAT), a presynaptic marker for GABAergic synapses, a reduction in colocalization of gephyrin and α2 subunit of GABAA receptors was observed in the dendrites of developing pyramidal neurons (Figure 3a and b). These results indicate that knockdown of DISC1 reduces expression of α2 subunit in the GABAergic synapses in a cell-autonomous manner.
Figure 3.
Postnatal suppression of DISC1 expression in the prefrontal cortex (PFC) impairs developmental synaptic GABA function. (a) Immunohistochemical analysis with antibodies against GABAA receptor α2 subunits (red) and gephyrin, a GABA synaptic scaffold protein (green), in developing pyramidal neurons (blue) of the medial PFC in mice with postnatal knockdown of DISC1 at P14. Reduction of colocalization of GABAA receptor α2 subunits and gephyrin are observed in postnatal DISC1 knockdown mice. Scale bar, 10 µm. n = 5 neurons per condition. (b) Immunohistochemical analysis with antibodies against vesicular GABA transporter (vGAT, a presynaptic marker, indicated in red) and gephyrin (a GABA synaptic scaffold protein, indicated in green), in the developing pyramidal neurons (blue) of the medial PFC in mice with postnatal knockdown of DISC1 and control mice at P14. No changes in the number of gephyrin puncta and colocalization of vGAT and gephyrin are observed in postnatal DISC1 knockdown mice compared to control mice. Scale bar, 10 µm. n=5 neurons per condition. (c, d) Acute brain slice electrophysiology recordings from P14 mice show that postnatal suppression of DISC1 depresses GABAergic transmission. Representative traces depicting GABA-mediated miniature postsynaptic currents (GABA-mediated mPSCs) from each condition are shown. Summary graphs showing the amplitude of GABA-mediated mPSCs are decreased by postnatal DISC1 suppression, while DISC1 suppression does not affect on mPSCs frequency. All recordings were performed in the presence of D,L AP5 (100 µM), NBQX (10 µM), and TTX (1 µM). n = 10–15 neurons per condition. *P < 0.05 determined by Student’s t test (a, d).
Postnatal suppression of DISC1 expression may affect synaptic GABAA receptor function. To test this idea, synaptic GABAA receptor function was measured by whole-cell electrophysiology in postnatal PFC brain slices of inducible DISC1 knockdown mice and control mice with inducible scrambled control shRNA. Consistent with immunohistochemical data (Figure 3a and b), we found significant reductions in the amplitude of GABA-mediated miniature postsynaptic currents (GABA-mediated mPSCs) in neurons with postnatal suppression of DISC1 expression under pharmacological blockade of NMDA receptors (100 µM DL-AP5), AMPA receptors (10 µM NBQX), and sodium channels (1 µM TTX), whereas no difference was observed in the frequency of GABA-mediated mPSCs between both groups of mice (Figure 3c and d).
Postnatal suppression of DISC1 expression in the PFC impairs sensorimotor gating, albeit without profound memory deficits, in young adulthood
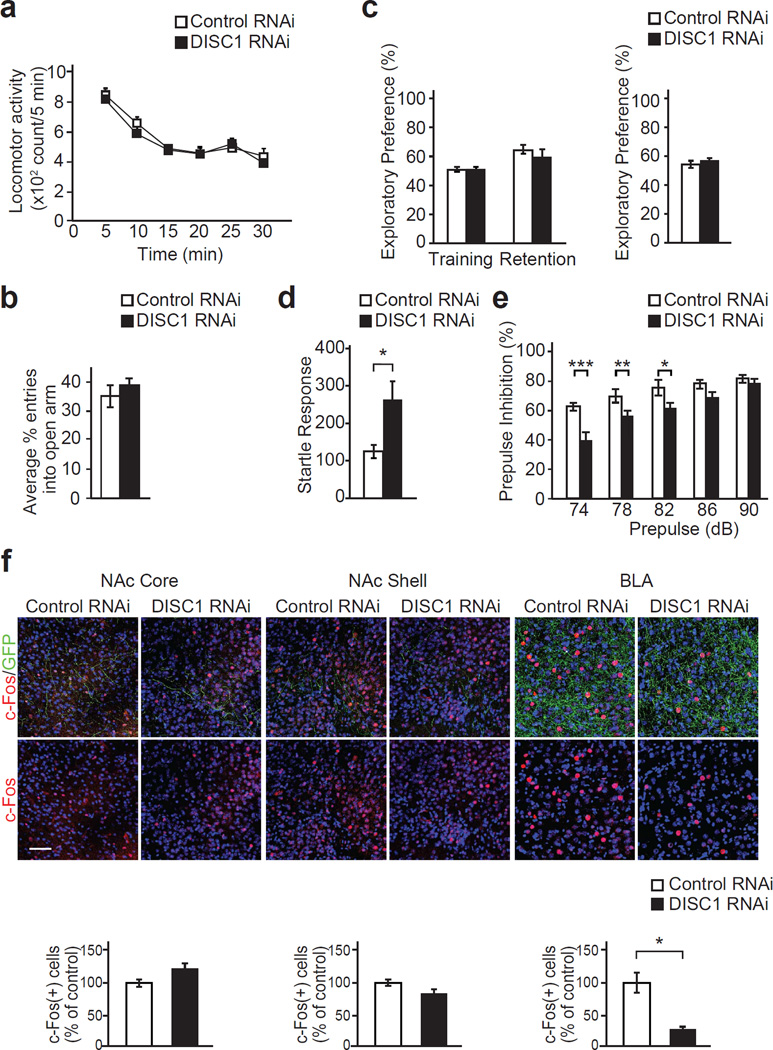
We next examined whether postnatal knockdown of DISC1 in the PFC results in long lasting behavioral alterations. Given that changes in body weight may reflect nonspecific developmental abnormalities and resultant behaviors, we examined differences in body weight between inducible DISC1 knockdown mice and control mice with inducible scrambled control shRNA at 3 months of age, but observed no difference (Supplementary Figure 7). Animals were then evaluated in the open field test and elevated plus maze test to measure locomotor activity and anxiety-related behavior, which might confound assessment of cognitive behaviors. There was no difference in either test between both groups of mice (Figure 4a and b). Our previous study established that knockdown of DISC1 in the PFC starting from the embryonic period resulted in deficits in cognitive function and sensory information processing, assessed by the novel object recognition test (NORT) and prepulse inhibition test (PPI).20 To examine whether postnatal suppression of DISC1 expression is sufficient to elicit these behavioral abnormalities, NORT and PPI were performed in 3-month-old animals with inducible knockdown of DISC1. In the NORT, no difference was observed between inducible DISC1 knockdown mice and controls for total exploration time and exploratory preference of the novel object and its location, suggesting that postnatal suppression of DISC1 expression in the PFC has no major impact on object and spatial recognition memory (Figure 4c and Supplementary Figure 8). Of note, we found a significant increase in the baseline of acoustic startle response as well as a reduction in PPI with prepulses of 74 dB, 78 dB, and 82 dB in the inducible DISC1 knockdown mice (Figure 4d and e), indicating that postnatal expression of DISC1 in the PFC is required for normal sensorimotor gating. To examine the neuronal circuit mechanisms underlying these behavioral alterations induced by knockdown of DISC1, expression of c-Fos, an immediate early gene, was evaluated in several brain regions where we demonstrated existence of direct projections from GFP-positive pyramidal neurons in the PFC after the mice were given the pulse in PPI experiments (Supplementary Figure 9a). These brain areas include the nucleus accumbens core and shell as well as the basolateral amygdala, which are implicated in both the acoustic startle response and PPI.35–37 We found that induction of c-Fos in the basolateral amygdala, but not in the nucleus accumbens core and shell, was impaired by postnatal knockdown of DISC1 (Figure 4f). Importantly double-labeling experiments revealed that observed induction of c-Fos elevation in the basolateral amygdala is occurring predominantly in GAD67-positive GABAergic neurons (Supplementary Figure 9b).
Figure 4.
Postnatal suppression of DISC1 expression in the prefrontal cortex (PFC) causes deficits in sensorimotor gating in young adulthood. (a) Postnatal knockdown of DISC1 in the PFC has no effect on locomotion as indicated by the open field test. (b) Postnatal suppression of DISC1 expression in the PFC has no effect on anxiety as indicated by the elevated plus maze test. (c) In the novel object recognition test, there were no differences in exploratory preference during training and retention sessions (left graph), and no abnormality in spatial recognition memory in postnatal DISC1 knockdown mice (right graph). (d) Excess acoustic startle response was observed in inducible DISC1 knockdown mice. n = 12 per condition. (e) Impairment of prepulse inhibition in the postnatal DISC1 suppression mice, when the 120 dB pulse was preceded by prepulses of 74, 78, and 82 dB. There were significant effects of genotype (F(1,110) = 53.92, P < 0.0001), prepulse intensity (F(4,110) = 29.93, P < 0.0001), and genotype-prepulse interaction (F(4,110) = 3.15, P = 0.0172). n = 12 per condition. (f) Projections from the PFC to nucleus accumbens core (NAc core), nucleus accumbens shell (NAc shell), and basolateral amygdala (BLA) were visualized by immunohistochemistry with antibodies against GFP (green). Postnatal DISC1 knockdown induced reductions in elevation of c-Fos expression (red) in the BLA, but not in the NAc core and NAc shell, following PPI. Blue, nucleus. Scale bar, 100 µm. n = 10 per condition. *P < 0.05, **P < 0.01, and ***P < 0.001 determined by Student’s t test (d, f) and two-way repeated measures ANOVA followed by the Bonferroni test (e).
Subtype-selective positive allosteric modulators of α2/3-containing GABAA receptors reverse dendritic abnormalities induced by suppression of DISC1 in developing cortical neurons
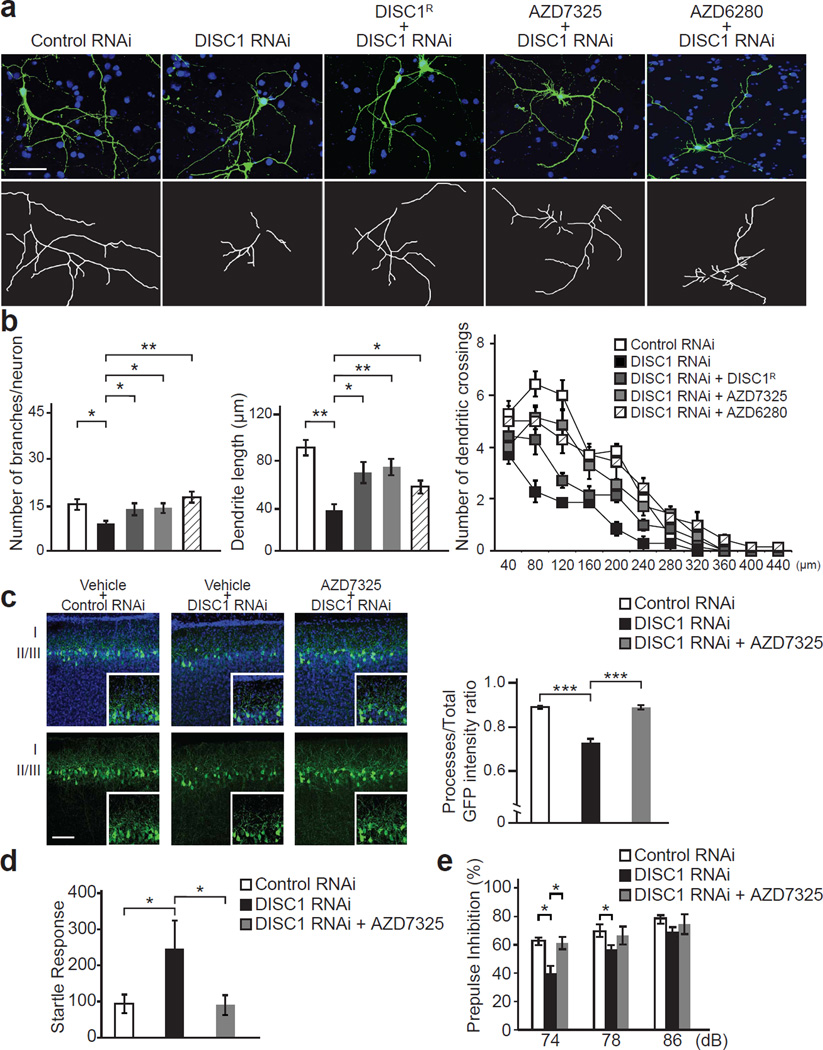
Results thus far indicate that postnatal knockdown of DISC1 causes a reduction in cell surface expression of α2 subunit-containing GABAA receptors, impaired postsynaptic GABAA receptor signaling, dendritic abnormalities in developing pyramidal neurons, and behavioral alterations including excessive startle response and deficits in PPI. We therefore investigated whether pharmacological activation of GABAA receptors might overcome these abnormalities due to suppression of DISC1 expression. A variety of subtype-selective compounds of GABAA receptors have recently been synthesized.8 We tested the potential of two novel subtype-selective positive allosteric modulators of α2/3-containing GABAA receptors (AZD7325 and AZD6280) (Supplementary Figure 10) to overcome dendritic alterations induced by knockdown of DISC1 in developing cortical neuron cultures. These compounds were previously shown to have modest efficacy on α2/3-containing GABAA receptors and low efficacy on α1 or α5 containing subtypes.38 These observations were confirmed by electrophysiological characterization in Xenopus oocytes expressing α1, α2, α3, or α5-containing GABAA receptors (Supplementary Figure 11). Application of either AZD7325 or AZD6280 rescued dendritic abnormalities induced by knockdown of DISC1, including both numbers of branches as well as total dendritic length and complexity; this effect is comparable to that of overexpression of RNAi-resistant wild-type DISC1 (Figure 5a, b and Supplementary Figure 12). We also found that treatment with AZD7325 does not affect impaired cell surface expression of α2 subunit induced by knockdown of DISC1 (Supplementary Figure 13). We then examined whether transient administration of AZD7325 during the early postnatal period ameliorates observed neuronal and behavioral deficits in inducible DISC1 suppression mice. AZD7325 (10 µmol/kg body weight) and vehicle were administered intraperitoneally daily for 1 week starting from P4. Dendritic quantity in the mPFC was measured at P14 by comparing fluorescence intensity. We observed that transient pharmacological early intervention with AZD7325 reversed dendritic deficits caused by postnatal DISC1 suppression (Figure 5c). The effect of treatment with AZD7325 on abnormalities in acoustic startle response and PPI was then assessed in 3-month-old animals. In addition to normalization of the elevated startle response, a reduction in PPI with prepulses of 74 dB was rescued by early postnatal treatment with AZD7325 (Figure 5d and e).
Figure 5.
Subtype-selective positive allosteric modulators of α2/3-containing GABAA receptors reverse dendritic abnormalities and altered behaviors induced by knockdown of DISC1. (a) Representative images showing effect of co-expressing RNAi-resistant DISC1 (DISC1R), and 7-day treatment with positive allosteric modulators of α2/3-containing GABAA receptors, AZD7325 (0.1 µM) and AZD6280 (0.1 µM), in primary cortical neuron expressing DISC1 RNAi carrying GFP protein (green). Traced dendrites were depicted as white lines (lower panels). Blue, nucleus. Scale bar, 100 µm. (b) Quantification of dendritic morphometry demonstrates that reduction of dendrite growth and branches as well as altered dendrite complexity induced by knockdown of DISC1 was ameliorated by co-expression with RNAi-resistant DISC1, in which the effect is comparable to treatment with AZD7325 and AZD6280. We analyzed only neurons with typical morphological characteristics of pyramidal neurons (single axon and multiple dendrites). n = 7 per condition. (c) Representative image showing dendritic structure of GFP-labeled pyramidal neurons in medial PFC from postnatal DISC1 suppression mice after intraperitoneal injection of AZD7325 (10 µmol/kg body weight) or vehicle for 1 week in the early postnatal period. Ratio of GFP fluorescence intensity in dendrites to total GFP fluorescence intensity was increased in AZD7325-treated inducible DISC1 knockdown mice, compared to those of vehicle-treated ones. Blue, nucleus. Scale bar, 100 µm. Ten brain slice images per condition were analyzed in a blinded manner. (d) Startle response to 120dB acoustic pulse was increased in the postnatal DISC1 knockdown mice compared to the control mice, and this excessive response was normalized by early postnatal treatment with AZD7325. (e) Impairment of prepulse inhibition in the postnatal DISC1 knockdown mice was rescued by AZD7325 treatment, significantly ameliorated at 74dB prepulse, and partially ameliorated at 78dB. *P < 0.05, **P < 0.01, and ***P < 0.001 determined by one-way ANOVA with posthoc Bonferroni test (b, c, d, e).
DISCUSSION
Our data indicate that DISC1 is involved in regulating cell surface expression of α2 subunit-containing GABAA receptors and postsynaptic GABAA receptor signaling during early postnatal periods. Given that knockdown of DISC1 has no effect on total protein expression levels of GABAA receptors, reduction of cell surface expression of GABAA receptors is unlikely to result from slow maturation of these neurons caused by knockdown of DISC1. Although molecular mechanisms determining how DISC1 is involved in cell surface expression of α2 subunit-containing GABAA receptors in developing neurons are still unclear, it is worth noting that DISC1 has been reported to bind to the kinesin family member 5 that regulates GABAA receptor intracellular transport.39,40 Thus DISC1 may participate in the targeting of these receptors to the cell surface or specific neuronal compartments. Multiple DISC1-associated signaling pathways are involved in dendritic growth.14,15,41 However, our data demonstrate that pharmacological potentiation of GABAA receptor-mediated signaling rescued dendritic abnormalities caused by knockdown of DISC1, suggesting that DISC1 may be involved in developmental control of dendrite growth through modulation of GABA signaling. Of note, the effect of DISC1 on cell surface expression of GABAA receptors is specific to the developmental phase and treatment with subtype-selective positive allosteric modulators of α2/3-containing GABAA receptors during the early postnatal period alleviated dendritic and behavioral abnormalities, though it remains an open question whether the observed abnormalities induced by knockdown of DISC1 could also be rescued by other subtype-selective modulators.
Based on our results, we assume that reduced GABAergic inhibition in the basolateral amygdala may contribute to dysfunction in medial PFC-basolateral amygdala circuits that underlies the observed behavioral changes. Changes in the activity of circuits connecting these brain regions may represent a potential mechanism that contributes to the enhanced acoustic startle response and impaired sensorimotor gating caused by postnatal knockdown of DISC1. This hypothesis is supported by previous findings that pharmacological blockade of GABAA receptors in the basolateral amygdala leads to an enhanced startle response and deficits in PPI.42 We have previously demonstrated that knockdown of DISC1 beginning at prenatal periods leads to abnormalities in dopaminergic circuit maturation.20 Given that mesocortical and mesolimbic dopaminergic projections are involved in regulating sensorimotor gating,35 it would be interesting to elucidate how postnatal knockdown of DISC1 in the PFC affects dopaminergic circuits, whose disturbance may also underlie excess acoustic startle response and PPI deficits observed in our mouse models.
Pharmacological targeting of excitatory GABA signaling during early development has recently started to be tested. Tyzio et al. have reported that maternal treatment with bumetanide, a NKCC1 blocker, ameliorates autism-associated electrophysiological and behavioral characteristics in animal models with in utero exposure to valprorate and mouse model carrying the fragile X mutation.43 In contrast, perinatal treatment with bumetanide has detrimental effects on dendritic maturation and behaviors.44 The discrepancy between these studies cautions us that dose and timing of pharmacological modulators to alter developmental GABA signaling is critical to obtain beneficial effects on disease conditions.
To the best of our knowledge, this is the first study demonstrating the utility of a conditional knockdown system with in utero electroporation for gene targeting in a spatial and temporal manner. This approach allowed us to segregate a role for DISC1 in dendritic development, independent from the secondary effects of DISC1 on dendrites caused by disturbed cell proliferation and neuronal migration. DISC1 is involved in diverse cellular processes in the developing cerebral cortex.14,15 These include cell proliferation, neuronal migration, and dendrite growth, which have been demonstrated in various rodent models.20,28–30,45–52 Nonetheless, how DISC1-mediated pathways in specific developmental periods are involved in these phenotypes is unclear. Here we report that suppression of DISC1 expression in the PFC in the postnatal period impaired developmental synaptic GABAA receptor function and was sufficient to evoke deficits in dendritic growth and sensorimotor gating in young adulthood. Similarly, indirect disruption of early GABA excitatory action caused by perinatal treatment with bumetanide, results in deficits in dendritic formation in cortical neurons and sensorimotor gating.44
Although it is laboriously difficult to investigate effective therapeutic strategies to restore function due to a genetic predisposition during brain development in humans, our study provides proof-of-principle that pharmacological intervention during specific postnatal periods may be critical. Thus, mechanism-based pharmacological application during development is warranted to be tested in animal models.
Supplementary Material
ACKNOWLEDGEMENTS
We thank Drs. Pamela Talalay and Keri Martinowich, as well as Ms. Nada Rendradjaja for critical reading of the manuscript. We thank Drs. Kozo Kaibuchi and Keisuke Kuroda for providing us with DISC1 antibodies and Dr. Miho Terunuma for advice on biotinylation assays. This work was supported by R01 MH-091230 (A.K.), P50 MH-094268 (A.K.), R21AT008547 (A.K.), K01 MH-086050 (B.J.M.), Brain & Behavior Research Foundation (A.K.), and Brain Science Foundation (A.K.).
Footnotes
CONFLICT OF INTEREST
A.C. and N.J.B. are full-time employees and shareholders in AstraZeneca. S.J.M serves as a consultant for SAGE therapeutics and AstraZeneca, relationships that are regulated by Tufts University.
REFERENCES
- 1.Fritschy JM, Panzanelli P. GABAA receptors and plasticity of inhibitory neurotransmission in the central nervous system. Eur J Neurosci. 2014;39:1845–1865. doi: 10.1111/ejn.12534. [DOI] [PubMed] [Google Scholar]
- 2.Ben-Ari Y. Excitatory actions of gaba during development: the nature of the nurture. Nat Rev Neurosci. 2002;3:728–739. doi: 10.1038/nrn920. [DOI] [PubMed] [Google Scholar]
- 3.Cancedda L, Fiumelli H, Chen K, Poo MM. Excitatory GABA action is essential for morphological maturation of cortical neurons in vivo. J Neurosci. 2007;27:5224–5235. doi: 10.1523/JNEUROSCI.5169-06.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Le Magueresse C, Monyer H. GABAergic interneurons shape the functional maturation of the cortex. Neuron. 2013;77:388–405. doi: 10.1016/j.neuron.2013.01.011. [DOI] [PubMed] [Google Scholar]
- 5.Charych EI, Liu F, Moss SJ, Brandon NJ. GABA(A) receptors and their associated proteins: implications in the etiology and treatment of schizophrenia and related disorders. Neuropharmacology. 2009;57:481–495. doi: 10.1016/j.neuropharm.2009.07.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Cellot G, Cherubini E. GABAergic signaling as therapeutic target for autism spectrum disorders. Front Pediatr. 2014;2:70. doi: 10.3389/fped.2014.00070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hoftman GD, Lewis DA. Postnatal developmental trajectories of neural circuits in the primate prefrontal cortex: identifying sensitive periods for vulnerability to schizophrenia. Schizophr Bull. 2011;37:493–503. doi: 10.1093/schbul/sbr029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Rudolph U, Knoflach F. Beyond classical benzodiazepines: novel therapeutic potential of GABAA receptor subtypes. Nat Rev Drug Discov. 2011;10:685–697. doi: 10.1038/nrd3502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Crestani F, Martin JR, Mohler H, Rudolph U. Mechanism of action of the hypnotic zolpidem in vivo. Br J Pharmacol. 2000;131:1251–1254. doi: 10.1038/sj.bjp.0703717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Han S, Tai C, Jones CJ, Scheuer T, Catterall WA. Enhancement of inhibitory neurotransmission by GABAA receptors having alpha2,3-subunits ameliorates behavioral deficits in a mouse model of autism. Neuron. 2014;81:1282–1289. doi: 10.1016/j.neuron.2014.01.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lewis DA, Cho RY, Carter CS, Eklund K, Forster S, Kelly MA, et al. Subunit-selective modulation of GABA type A receptor neurotransmission and cognition in schizophrenia. Am J Psychiatry. 2008;165:1585–1593. doi: 10.1176/appi.ajp.2008.08030395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Buchanan RW, Keefe RS, Lieberman JA, Barch DM, Csernansky JG, Goff DC, et al. A randomized clinical trial of MK-0777 for the treatment of cognitive impairments in people with schizophrenia. Biol Psychiatry. 2011;69:442–449. doi: 10.1016/j.biopsych.2010.09.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Millar JK, Wilson-Annan JC, Anderson S, Christie S, Taylor MS, Semple CA, et al. Disruption of two novel genes by a translocation co-segregating with schizophrenia. Hum Mol Genet. 2000;9:1415–1423. doi: 10.1093/hmg/9.9.1415. [DOI] [PubMed] [Google Scholar]
- 14.Brandon NJ, Sawa A. Linking neurodevelopmental and synaptic theories of mental illness through DISC1. Nat Rev Neurosci. 2011;12:707–722. doi: 10.1038/nrn3120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kamiya A, Sedlak TW, Pletnikov MV. DISC1 Pathway in Brain Development: Exploring Therapeutic Targets for Major Psychiatric Disorders. Front Psychiatry. 2012;3:25. doi: 10.3389/fpsyt.2012.00025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kim JY, Liu CY, Zhang F, Duan X, Wen Z, Song J, et al. Interplay between DISC1 and GABA signaling regulates neurogenesis in mice and risk for schizophrenia. Cell. 2012;148:1051–1064. doi: 10.1016/j.cell.2011.12.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Manent JB, Wang Y, Chang Y, Paramasivam M, LoTurco JJ. Dcx reexpression reduces subcortical band heterotopia and seizure threshold in an animal model of neuronal migration disorder. Nat Med. 2009;15:84–90. doi: 10.1038/nm.1897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.LoTurco J, Manent JB, Sidiqi F. New and improved tools for in utero electroporation studies of developing cerebral cortex. Cereb Cortex. 2009;19(Suppl 1):i120–i125. doi: 10.1093/cercor/bhp033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Taniguchi Y, Young-Pearse T, Sawa A, Kamiya A. In utero electroporation as a tool for genetic manipulation in vivo to study psychiatric disorders: from genes to circuits and behaviors. Neuroscientist. 2012;18:169–179. doi: 10.1177/1073858411399925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Niwa M, Kamiya A, Murai R, Kubo K, Gruber AJ, Tomita K, et al. Knockdown of DISC1 by in utero gene transfer disturbs postnatal dopaminergic maturation in the frontal cortex and leads to adult behavioral deficits. Neuron. 2010;65:480–489. doi: 10.1016/j.neuron.2010.01.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Lai WS, Xu B, Westphal KG, Paterlini M, Olivier B, Pavlidis P, et al. Akt1 deficiency affects neuronal morphology and predisposes to abnormalities in prefrontal cortex functioning. Proc Natl Acad Sci U S A. 2006;103:16906–16911. doi: 10.1073/pnas.0604994103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Takemoto-Kimura S, Ageta-Ishihara N, Nonaka M, Adachi-Morishima A, Mano T, Okamura M, et al. Regulation of dendritogenesis via a lipid-raft-associated Ca2+/calmodulin-dependent protein kinase CLICK-III/CaMKIgamma. Neuron. 2007;54:755–770. doi: 10.1016/j.neuron.2007.05.021. [DOI] [PubMed] [Google Scholar]
- 23.Moss SJ, Smart TG. Constructing inhibitory synapses. Nat Rev Neurosci. 2001;2:240–250. doi: 10.1038/35067500. [DOI] [PubMed] [Google Scholar]
- 24.Hayashi-Takagi A, Takaki M, Graziane N, Seshadri S, Murdoch H, Dunlop AJ, et al. Disrupted-in-Schizophrenia 1 (DISC1) regulates spines of the glutamate synapse via Rac1. Nat Neurosci. 2010;13:327–332. doi: 10.1038/nn.2487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Cellot G, Cherubini E. Functional role of ambient GABA in refining neuronal circuits early in postnatal development. Front Neural Circuits. 2013;7:136. doi: 10.3389/fncir.2013.00136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Porcher C, Hatchett C, Longbottom RE, McAinch K, Sihra TS, Moss SJ, et al. Positive feedback regulation between gamma-aminobutyric acid type A (GABA(A)) receptor signaling and brain-derived neurotrophic factor (BDNF) release in developing neurons. J Biol Chem. 2011;286:21667–21677. doi: 10.1074/jbc.M110.201582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Matsuda T, Cepko CL. Controlled expression of transgenes introduced by in vivo electroporation. Proc Natl Acad Sci U S A. 2007;104:1027–1032. doi: 10.1073/pnas.0610155104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Ishizuka K, Kamiya A, Oh CE, Kanki H, Seshadri S, Robinson FJ, et al. A DISC1-dependent switch from neuronal proliferation to migration in the developing cortex. Nature. 2011 doi: 10.1038/nature09859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kamiya A, Kubo K, Tomoda T, Takaki M, Youn R, Ozeki Y, et al. A schizophrenia-associated mutation of DISC1 perturbs cerebral cortex development. Nat Cell Biol. 2005;7:1167–1178. doi: 10.1038/ncb1328. [DOI] [PubMed] [Google Scholar]
- 30.Kubo K, Tomita K, Uto A, Kuroda K, Seshadri S, Cohen J, et al. Migration defects by DISC1 knockdown in C57BL/6, 129X1/SvJ, and ICR strains via in utero gene transfer and virus-mediated RNAi. Biochem Biophys Res Commun. 2010;400:631–637. doi: 10.1016/j.bbrc.2010.08.117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Marin O, Rubenstein JL. Cell migration in the forebrain. Annu Rev Neurosci. 2003;26:441–483. doi: 10.1146/annurev.neuro.26.041002.131058. [DOI] [PubMed] [Google Scholar]
- 32.Sauvageot CM, Stiles CD. Molecular mechanisms controlling cortical gliogenesis. Curr Opin Neurobiol. 2002;12:244–249. doi: 10.1016/s0959-4388(02)00322-7. [DOI] [PubMed] [Google Scholar]
- 33.Seshadri S, Kamiya A, Yokota Y, Prikulis I, Kano S, Hayashi-Takagi A, et al. Disrupted-in-Schizophrenia-1 expression is regulated by beta-site amyloid precursor protein cleaving enzyme-1-neuregulin cascade. Proc Natl Acad Sci U S A. 2010;107:5622–5627. doi: 10.1073/pnas.0909284107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Konur S, Ghosh A. Calcium signaling and the control of dendritic development. Neuron. 2005;46:401–405. doi: 10.1016/j.neuron.2005.04.022. [DOI] [PubMed] [Google Scholar]
- 35.Swerdlow NR, Geyer MA, Braff DL. Neural circuit regulation of prepulse inhibition of startle in the rat: current knowledge and future challenges. Psychopharmacology (Berl) 2001;156:194–215. doi: 10.1007/s002130100799. [DOI] [PubMed] [Google Scholar]
- 36.Lee Y, Davis M. Role of the hippocampus, the bed nucleus of the stria terminalis, and the amygdala in the excitatory effect of corticotropin-releasing hormone on the acoustic startle reflex. J Neurosci. 1997;17:6434–6446. doi: 10.1523/JNEUROSCI.17-16-06434.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Koch M. The neurobiology of startle. Prog Neurobiol. 1999;59:107–128. doi: 10.1016/s0301-0082(98)00098-7. [DOI] [PubMed] [Google Scholar]
- 38.Alhambra C, Becker C, Blake T, Chang AH, Damewood JR, Jr, Daniels T, et al. Development and SAR of functionally selective allosteric modulators of GABAA receptors. Bioorg Med Chem. 2011;19:2927–2938. doi: 10.1016/j.bmc.2011.03.035. [DOI] [PubMed] [Google Scholar]
- 39.Twelvetrees AE, Yuen EY, Arancibia-Carcamo IL, MacAskill AF, Rostaing P, Lumb MJ, et al. Delivery of GABAARs to synapses is mediated by HAP1-KIF5 and disrupted by mutant huntingtin. Neuron. 2010;65:53–65. doi: 10.1016/j.neuron.2009.12.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Taya S, Shinoda T, Tsuboi D, Asaki J, Nagai K, Hikita T, et al. DISC1 regulates the transport of the NUDEL/LIS1/14-3-3epsilon complex through kinesin-1. J Neurosci. 2007;27:15–26. doi: 10.1523/JNEUROSCI.3826-06.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Porteous DJ, Millar JK, Brandon NJ, Sawa A. DISC1 at 10: connecting psychiatric genetics and neuroscience. Trends Mol Med. 2011;17:699–706. doi: 10.1016/j.molmed.2011.09.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Fendt M, Schwienbacher I, Koch M. Amygdaloid N-methyl-D-aspartate and gamma-aminobutyric acid(A) receptors regulate sensorimotor gating in a dopamine-dependent way in rats. Neuroscience. 2000;98:55–60. doi: 10.1016/s0306-4522(00)00086-5. [DOI] [PubMed] [Google Scholar]
- 43.Tyzio R, Nardou R, Ferrari DC, Tsintsadze T, Shahrokhi A, Eftekhari S, et al. Oxytocin-mediated GABA inhibition during delivery attenuates autism pathogenesis in rodent offspring. Science. 2014;343:675–679. doi: 10.1126/science.1247190. [DOI] [PubMed] [Google Scholar]
- 44.Wang DD, Kriegstein AR. Blocking early GABA depolarization with bumetanide results in permanent alterations in cortical circuits and sensorimotor gating deficits. Cereb Cortex. 2011;21:574–587. doi: 10.1093/cercor/bhq124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Young-Pearse TL, Suth S, Luth ES, Sawa A, Selkoe DJ. Biocemical and Functional Interaction of Disrupted-in-Schizophrenia 1 and Amyloid Precursor Protein Regulates Neuronal Migration during Mammalian Cortical Development. J Neurosci. 2010;30:10431–10440. doi: 10.1523/JNEUROSCI.1445-10.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Kvajo M, McKellar H, Arguello PA, Drew LJ, Moore H, MacDermott AB, et al. A mutation in mouse Disc1 that models a schizophrenia risk allele leads to specific alterations in neuronal architecture and cognition. Proc Natl Acad Sci U S A. 2008;105:7076–7081. doi: 10.1073/pnas.0802615105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Pletnikov MV, Ayhan Y, Nikolskaia O, Xu Y, Ovanesov MV, Huang H, et al. Inducible expression of mutant human DISC1 in mice is associated with brain and behavioral abnormalities reminiscent of schizophrenia. Mol Psychiatry. 2008;13:173–186. 115. doi: 10.1038/sj.mp.4002079. [DOI] [PubMed] [Google Scholar]
- 48.Lee FH, Fadel MP, Preston-Maher K, Cordes SP, Clapcote SJ, Price DJ, et al. Disc1 point mutations in mice affect development of the cerebral cortex. J Neurosci. 2011;31:3197–3206. doi: 10.1523/JNEUROSCI.4219-10.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Duan X, Chang JH, Ge S, Faulkner RL, Kim JY, Kitabatake Y, et al. Disrupted-In-Schizophrenia 1 regulates integration of newly generated neurons in the adult brain. Cell. 2007;130:1146–1158. doi: 10.1016/j.cell.2007.07.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Shen S, Lang B, Nakamoto C, Zhang F, Pu J, Kuan SL, et al. Schizophrenia-related neural and behavioral phenotypes in transgenic mice expressing truncated Disc1. J Neurosci. 2008;28:10893–10904. doi: 10.1523/JNEUROSCI.3299-08.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Maher BJ, LoTurco JJ. Disrupted-in-schizophrenia (DISC1) functions presynaptically at glutamatergic synapses. PLoS One. 2012;7:e34053. doi: 10.1371/journal.pone.0034053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Mao Y, Ge X, Frank CL, Madison JM, Koehler AN, Doud MK, et al. Disrupted in schizophrenia 1 regulates neuronal progenitor proliferation via modulation of GSK3beta/beta-catenin signaling. Cell. 2009;136:1017–1031. doi: 10.1016/j.cell.2008.12.044. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.