Abstract
Polo-like kinase 1 (Plk1) overexpression has been shown to occur in a wide range of tumors, prompting research and development of Plk1 inhibitors as a means of cancer treatment. This review discusses recent advances in the development of Plk1 inhibitors for cancer management. Plk1 inhibition has been shown to cause mitotic block and apoptosis of cells with higher mitotic index and therefore higher Plk1 expression. The potential of Plk1 inhibitors as cancer therapeutics has been widely investigated. However, a complete understanding of Plk1 biology/mechanism is yet to be fully achieved. Resistance to certain chemotherapeutic drugs has been linked to Plk1 overexpression, and Plk1-mediated mitotic events such as microtubule rearrangement have been found to reduce the efficacy of chemotherapeutic agents. The Plk1 inhibitor, volasertib, has shown considerable promise in clinical studies, having reached phase III trials. However, preclinical success with Plk1 inhibitors has not translated well into clinical success. In our view, combined therapies targeting other relevant pathways together with Plk1 may be vital to combat issues observed with monotherapy, especially resistance. In addition, research should also be directed towards understanding the mechanisms of Plk1 and designing additional next generations of specific, potent Plk1 inhibitors to target cancer.
Keywords: Plk1, Inhibitor, Cancer, Resistance, Clinical Trials
Introduction
Cancer is a major health problem across the globe and the second leading cause of deaths in the United States (1). The American Cancer Society estimates that in the USA alone, 1,685,210 new cases of cancer will be diagnosed, and 595,690 people will die of this disease, in the year 2016 (1). Cancer deaths are often caused by metastasis and recurrence due to acquired drug resistance. Traditional chemotherapy, including alkylating agents, antimetabolites and mitotic inhibitors [reviewed in (2)], are not adequate to manage most cancers, and often lead to drug-resistant and more aggressive cancers and metastasis [reviewed in (3)]. This has become increasingly problematic, substantially reducing the efficacy of available chemotherapy treatments. This has prompted research into new compounds that act on mechanistically-identified novel targets, such as the polo-like kinase-1 (Plk1), which can be used as monotherapy as well as in combination with existing chemotherapies, for a better management of cancer.
Polo, a serine/threonine kinase, was first described in Drosophila in 1988 (4) as a key regulator of the cell cycle. Building off of a sequence similarity to this kinase, as well as an S. Cerivisiae protein involved in the same part of the cell cycle, Cdc5, Golsteyn and colleagues were able to characterize a similar kinase in humans, polo-like kinase 1 (PLK1) (5). Since then, four other members have been added to this family of kinases [reviewed in (6)]. As shown in Figure 1, human Plks 1-4 contain an N terminal kinase domain and vary in the presence of one or more C terminal polo box domains. Plk5 differs, however, as it is the only polo-like kinase that lacks the majority of its kinase domain [reviewed in (7)]. Of the five known human Plks, Plk1 is the most extensively studied because of being the founding member and due to its key role in cell-cycle regulation [reviewed in (8)]. This review focuses on Plk1 and its mechanistic interactions and biological importance, but it is imperative to mention here that the other Plk family members have been implicated in important cellular signaling events on their own. This includes the involvement of Plk2 and Plk3 as tumor modulators owing to their role in p53 signaling, as well as the participation of Plk4 in centriole biogenesis during mitosis (9). Not much work has been done on Plk5 at this point, but it has been implicated in neuronal differentiation (10).
Figure 1. Plk family proteins.
The structures of the five Plk family member proteins are shown. The location of the kinase domains (KD) is shown in blue, whereas the polo box domains (PB) are represented in orange.
Plk1 in Cell-Cycle Regulation
Plk1 has been found to play a key role in cell cycle progression through mitosis via its effects on chromosome segregation, spindle assembly and cytokinesis [reviewed in (7)]. By means of phosphorylating CDC25, Plk1 activates the cyclin B/cdc2 complex, which triggers cell proliferation. A recent study has suggested that Plk1 and Monopolar spindle 1 (Mps1) act cooperatively at the beginning of mitosis to establish spindle assembly checkpoint (SAC) by recruiting the Mad1:C-Mad2 complex to the kinetochores (11). Additionally, the same study suggested that together Plk1 and Mps1 help maintain SAC in periods of mitotic arrest (11). Inhibition of Plk1 has been shown to delay acentriolar spindle formation during mitosis and impede chromosome alignment at the equator during metaphase, as well as promoting apoptosis (12, 13). In addition to having an essential role in mitosis, Plk1 has been shown to be an important regulator of the DNA damage checkpoint (14). This is because Plk1 mediates phosphorylation of the scaffold protein claspin and checkpoint kinase 2 (Chk2), which inhibits checkpoint kinase 1 (Chk1) and Chk2 activation respectively, and in turn prevents DNA repair during mitosis (15).
Based on a number of studies the mechanism of Plk1 is becoming increasingly clear during the cell-cycle progression. Plk1 has been shown to be activated in the cell by the kinase Aurora A and its co-factor Bora (16). Bora acts on Plk1 by altering its conformation, which in turn allows Aurora A to phosphorylate Plk1 on threonine residue 210, promoting mitotic entry (17). Once Plk1 has been activated, its expression accumulates during S phase, peaks during G2-M transition and declines rapidly upon mitotic exit (5). Interestingly, Plk1 has been shown to have the ability to induce degradation of Aurora A and Bora, suggesting the presence of a feedback loop (16). Less is known regarding the interaction of Plk1 with the other members of the Aurora kinase family. It was recently demonstrated that Aurora B-mediated phosphorylation of Plk1 at Threonine 210 activates its kinase activity at the kinetochore to promote precise chromosome segregation (18). Phosphorylation of mitotic centromere-associated kinesin (MCAK) by Plk1 was also determined to be necessary for efficient separation of the chromatids (18).
Given that Plk1 is so intimately involved in cell-cycle regulation pathways and DNA damage repair, it is not surprising that it has been implicated and found to be overexpressed in a variety of cancers including melanoma, colorectal cancer and non-small cell lung cancer [reviewed in (19)]. Similarly, in a bladder cancer study, Plk1 was found to be overexpressed and this overexpression was correlated with higher pathological grade (p=0.0024) and multiple tumors (p=0.0241) (20). Additionally, another study found that 3-year survival rates for esophageal carcinoma were significantly lower in patients with high-grade expression of Plk1 compared to low-grade Plk1 expression (p<0.05) (21). Due to these and other similar studies suggesting a correlation between Plk1 overexpression and poor prognosis, many researchers have started to explore anti-Plk therapies for cancer treatment. Interestingly, it has been found that Plk is involved in resistance to several anticancer drugs through various pathways. Below, we explore these resistance pathways and new compounds that have been developed to exploit Plk1's biological roles, as well as their potential therapeutic usefulness.
Plk1 in Therapy Resistance
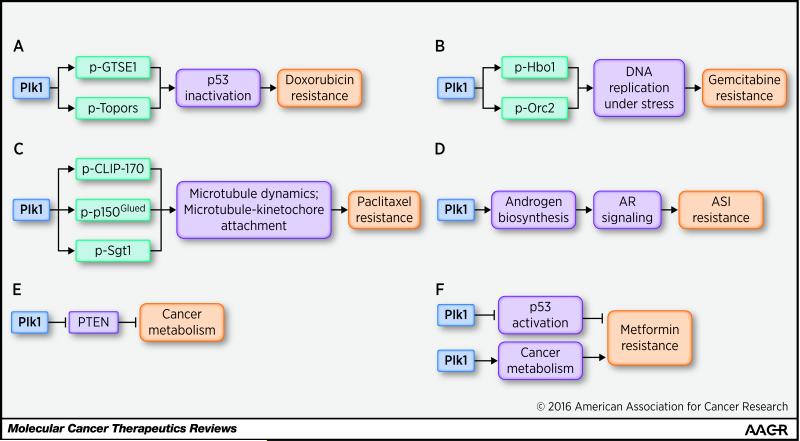
Likely because of its central role in cell-cycle progression and other cellular processes such as the DNA damage pathway, Plk1 has been found to be involved in the mechanisms of resistance to several chemotherapy drugs. As outlined in Figure 2, Plk1 has been found to be associated with drug resistance of a number of cancer chemotherapy drugs, including doxorubicin, paclitaxel, metformin, and gemcitabine. Some examples of these drugs and associated mechanisms are described below.
Figure 2. Examples of how Plk1-associated kinase activity contributes to therapy resistance.
(A) Plk1 phosphorylation of its substrates GTSE-1 and Topors causes p53 inactivation which results in doxorubicin resistance (25, 26). (B) Plk1-associated kinase activity drives DNA replication under stress via modulating its substrates Hbo1 and Orc2, resulting in acquired gemcitabine resistance in pancreatic cancer cells (32, 34, 35). (C) Plk1 contributes to paclitaxel resistance by phosphorylating its substrates CLIP-170, p150Glued and Sgt1, thereby regulating microtubule dynamics and microtubule-kinetochore attachment (41-44). (D) Plk1-associated activation of AR signaling leads to ASI resistance in CRPC (47, 49). (E) Plk1 elevation causes inactivation of PTEN to modulate the metabolism in prostate cancer cells (56). (F) Inhibition of Plk1 enhances the efficacy of anti-neoplastic activity of metformin in prostate cancer via both p53 and metabolic pathways (52).
Role of Plk1 in doxorubicin resistance
Doxorubicin is a chemotherapy agent that appears to have several modes of action, including DNA intercalation, interference in DNA replication and repair, and free radical generation [reviewed in (22)]. This drug is used against many different kinds of cancer, but has issues with cytotoxicity and development of drug resistance. Interestingly, it has recently been shown that Plk1-mediated p53 inactivation contributes to doxorubicin resistance. In 2012, Forrest and colleagues showed that upon doxorubicin-induced DNA damage, activation of the classical ATM/Chk2/p53 pathway leads to a prolonged cell-cycle arrest to allow DNA repair to occur (23). A 2014 study, however, found that 26% of normal cells that undergo mitosis after doxorubicin treatment show evidence of centriole abnormalities, showing that the DNA repair mechanism is not enough to overcome all of the DNA damage done by these therapies (24). Upon completion of DNA repair, the p53 pathway has to be inactivated to allow cell-cycle re-entry. Therefore, premature p53 inactivation results in premature termination of the checkpoint, consequently contributing to doxorubicin resistance. Using U2OS cells, it was shown that this occurs through two Plk1 substrates that negatively regulate p53 (Figure 2A) (25, 26). The first substrate, GTSE1 (G2 and S phase-expressed 1), is expressed specifically during G2 and S phases of the cell-cycle, and is able to down regulate p53 by translocating into the nucleus, binding to and shuttling p53 out of the nucleus and inducing its degradation (27). Liu and colleagues found that Plk1 phosphorylation at S435 of GTSE-1 is required for doxorubicin-induced G2 checkpoint recovery and subsequent shuttling of p53 out of the nucleus to lead to its degradation during recovery from cell-cycle arrest (25). The second Plk1 substrate, Topors (topoisomerase I-binding protein), is an ubiquitin and SUMO E3 ligase towards p53. While Topors-mediated p53 ubiquitination leads to its degradation (28), Topors-associated p53 sumoylation is accompanied by an increase in the level of p53 protein (29). In another study, a series of experiments by Yang and colleagues showed that Plk1-mediated phosphorylation of Topors inhibits its sumoylation of p53, while p53 ubiquitination is enhanced, leading to p53 degradation (26, 30). Thus, it reasonable to expect that Plk1 inhibition in conjunction with doxorubicin could be useful in avoiding or limiting doxorubicin resistance, thereby enhancing its therapeutic index.
Role of Plk1 on gemcitabine response
Plk1-dependent DNA replication, under stress, has been found to reduce cellular response to gemcitabine. As an analogue of deoxycytidine, gemcitabine induces cell death via its inhibition of DNA replication (31). Therefore, understanding regulation of DNA replication will reveal potential mechanisms in the development of gemcitabine resistance. Song and colleagues showed that Plk1-associated kinase activity drives DNA replication under stress, resulting in acquired gemcitabine resistance in pancreatic cancer cells (Figure 2B) (32). Accordingly, two DNA replication factors, Orc2 and Hbo1 (histone acetyltransferase binding to Orc1), have been identified as Plk1 substrates. Plk1 phosphorylation of Orc2-S188 has been found to enhance its association with the origin of replication. Additionally, cells expressing the Orc2-S188A mutant fail to maintain the functional pre-replicative complex under various DNA replication stress conditions, such as UV and hydroxyurea (32, 33). Of significance, it was found that Plk1 phosphorylation of Orc2 promotes DNA replication in gemcitabine-treated Panc1 cells, as phosphorylation of Orc2 at a well-documented DNA replication origin, MCM4, was elevated upon gemcitabine treatment. The Orc2-S188A-expressing cells were shown to have reduced DNA replication compared with Orc2-expressing cells upon gemcitabine treatment, providing direct evidence that Plk1 phosphorylation of Orc2 affects cellular response to gemcitabine. Finally, Panc1 cells expressing Orc2-S188A were apparently much more sensitive to gemcitabine treatment than Orc2-expressing cells, indicated by elevated cleaved PARP, a marker for apoptosis (34). Hbo1, another Plk1 target, has been found to be a critical regulator of DNA replication as well. Experiments have shown that Plk1 phosphorylation of Hbo1 is required for chromatin loading of the minichromosome maintenance complex and replication licensing (35). Additionally, it has been shown that Plk1 phosphorylation of Hbo1 increases cFos expression and consequently elevates its target, MDR1, whose overexpression has a well-documented role in drug resistance (34). Based on this series of mechanistic studies, Song and colleagues conducted a study combining a Plk1 inhibitor and gemcitabine in a xenograft model. They showed that inhibition of Plk1 sensitizes Panc1-derived xenograft tumors to gemcitabine treatment (34), thereby providing evidence that was used to support a clinical trial that combines a Plk1 inhibitor with gemcitabine for pancreatic cancer treatment. Hopefully this approach can be used on other cancers in the future, as well.
Role of Plk1 in Taxol resistance
It has been demonstrated that Plk1 contributes to Taxol resistance via its ability to regulate microtubule dynamics and microtubule-kinetochore attachment. At the cellular level, taxanes, including docetaxel and Taxol, bind β-tubulin and inhibit microtubule depolymerization which, in actively dividing cells leads to mitotic arrest, followed by apoptosis (36). However, in contrast to cancer cells growing in vitro, cancer cells growing in vivo, particularly prostate cancer cells, often display very slow doubling times (37). This finding suggests that the clinical activity of taxanes is likely due to both mitotic effects and effects on microtubule stabilization in interphase. In support of this theory, it was recently proposed that the therapeutic effect of docetaxel in prostate cancer is largely due to its inhibitory role on androgen receptor (AR) signaling via its effect on microtubule dynamics in interphase (38, 39). It has been shown that Plk1 contributes to Taxol resistance via its ability to regulate microtubule dynamics and microtubule-kinetochore attachment (Figure 2C). Clip-170, p150Glued and Sgt1 have been identified as three Plk1 substrates in this regard. Clip-170, the founding member of the microtubule plus end family, has roles in various microtubule-dependent processes in both interphase and mitosis (40). Hou and colleagues showed that Plk1 phosphorylation of Clip-170 is essential for the formation of kinetochore-microtubule attachments in mitosis (41) and microtubule dynamics in interphase (42), thus contributing to Taxol resistance in prostate cancer. Li and colleagues demonstrated that Plk1 phosphorylation of p150Glued, a subunit of the dynein/dynactin motor complex and the major interacting protein of Clip-170, facilitates nuclear envelope breakdown during prophase via its regulation of microtubule dynamics (43). Finally, it was reported that Plk1 phosphorylates Sgt1, a co-chaperone for Hsp90, and that this phosphorylation event promotes kinetochore-microtubule attachment to ensure chromosome stability (44). With these mechanisms in mind, researchers can potentially use Plk1-targeted therapies to reduce or eliminate Taxol resistance, thereby allowing Taxol to be used more effectively in cancer management.
Effect of Plk1 inhibition on androgen signaling blockade
In another important study, Plk1 inhibition was demonstrated to enhance the efficacy of androgen signaling blockade in CRPC. Because AR signaling continues to be essential in castration-resistant prostate cancer (CRPC), androgen signaling inhibitors (ASI), such as abiraterone and enzalutamide (MDV3100), are becoming the major approach to treat CRPC after docetaxel. However, the overall survival was only improved by five or two months in the phase III trials that compared abiraterone or enzalutamide with placebo in CRPC patients, respectively (45, 46). Recently, Zhang and colleagues found that Plk1 elevation leads to constitutively active AR signaling, eventually resulting in ASI resistance (Figure 2D) (47). Accordingly, it was shown that Plk1 is a positive regulator of AR signaling, likely due to its ability to enhance de novo androgen biosynthesis. It was further demonstrated that Plk1 increase led to activation of the SREBP (sterol regulatory element binding proteins) pathway, consequently resulting in increase of cholesterol, a precursor of androgen (47). More significantly, studies found that Plk1 inhibition sensitized enzalutamide-resistant MR49F cells to enzalutamide and its inhibition in combination with abiraterone inhibited patient-derived LuCaP35CR xenografts in a synergistic manner (47-49). This study supports the implementation of a clinical trial to combine a Plk1 inhibitor with ASIs to treat CRPC patients.
Effect of Plk1 inhibition on metformin's anti-proliferative response
Metformin, currently the first-line treatment for type 2 diabetes and has been shown to impact cellular metabolism, is currently being extensively investigated for its potential usefulness for cardiovascular diseases and cancer [(50, 51), and references therein]. Recently, Plk1 inhibition was shown to enhance the antineoplastic activity of metformin in prostate cancer (52). Cancer cells maintain tumor growth through increased glycolysis (Warburg effect) and glutaminolysis (glutamine addition) to satisfy the energy and carbon backbone needs, respectively (53). Garcia-Cao and colleagues have shown that PTEN inactivation leads to a tumor-promoting metabolic state by impacting both of these growth pathways (54). Interestingly, Plk1 elevation has been found to cause PTEN inactivation, increasing the cancer metabolism in prostate cancer cells (Figure 2E) (55). Subsequently, Plk1 phosphorylation of PTEN was shown to be responsible for its inactivation (56). Indeed, an in-depth understanding of potential upstream effectors of cancer metabolism may allow researchers to identify more appropriate drugs and combinations for cancer management. It is encouraging to note that metformin was found to inhibit the development of CRPC in a clinical trial with 3000 patients (57). Interestingly, inhibition of Plk1 enhances the antineoplastic activity of metformin in prostate cancer through both p53 and metabolic pathways (Figure 2F) (52). This gives us a hope that Plk1 inhibitors could enhance the anticancer activities of metformin or other metabolism and growth-regulating agents to the point that they could be clinically useful in cancer management.
Plk1 as a Drug Target
Due to the role of Plk1 in the cell-cycle and other pathways that are relevant to cancer progression, Plk1 is being considered as a ‘druggable target’ for development of drugs for the management of a variety of cancers. In this direction, considerable efforts are underway, from both drug companies and academic researchers. Some of these agents that have been explored are discussed below (and presented in Table 1), along with a review of their in vitro and in vivo successes and shortcomings.
Table 1. Plk inhibitor compounds.
Chemical structures of Plk1 inhibitors are shown along with their target and the phase/status of the trials discussed in the text. Further information regarding the trials and compound history may be found using the Clinical Trials Registry Number or reference provided.
| Compound | Structure | Target | Clinical Trial Phase | Clinical Trial Status | Ref. and NCT number |
|---|---|---|---|---|---|
| BI2536 |

|
ATP-binding domain | Phase II | Completed 2008 (primary) | (58, 59) NCT 00710710 |
| BI6727 (volasertib) |
|
ATP-binding domain | Phase III | Active, not recruiting | (63) NCT 01721876 |
| Tak960 |

|
ATP-binding domain | Phase I | Terminated 2013 | (73) NCT 01179399 |
| NMS-P937 |

|
ATP-binding domain | Phase I | Completed 2011 (primary) | (76, 77) NCT 01014429 |
| ON 01910.Na (rigosertib) |
|
Plk1 non-competitive and PI3K | Phase III | Completed 2015 | (80, 81) NCT 01360853 |
| GSK461364 |

|
ATP-binding domain | Phase I | Completed 2009 | (88, 90) NCT 00536835 |
| Poloxin |

|
Polo-box domain | None | N/A | (70, 71) |
| Poloxin-2 |

|
Polo-box domain | None | N/A | (72) |
BI 2536
One of the earliest Plk1 inhibitors, BI2536, was developed and described by the scientists at the Boehringer Ingelheim Company. In a study published in 2007, Steegmaier and colleagues showed that BI 2536 is a potent inhibitor of Plk1 that inhibits tumor growth in vitro as well as in vivo (58). BI 2536 was found to have an acceptable safety profile in some clinical trials (59). However, through extensive preclinical and early clinical testing, it is no longer used in monotherapy, possibly due to less than ideal response rates (60). Conversely, this first-generation Plk1 inhibitor has demonstrated efficacy when combined with other chemotherapeutic agents. BI2536 has been shown to sensitize neuroblastoma cells to vincristine antitumor activity. The cooperation of the two drugs resulted in inactivation of BCL-2 and cleavage of caspases 3 and 9 into their respective active fragments, promoting apoptosis (61). A synergistic effect was also observed between BI2536 and eribulin in neuroblastoma cells and in rhabdomyosarcoma where it decreased tumor growth in vivo (62).
Volasertib
Another Boehringer Ingelheim compound, volasertib (aka BI 6727), is an ATP-competitive inhibitor of Plk1 that was developed by tailoring the dihydropteridinone structure of BI 2536 (63). Volasertib has a very low IC50 of 0.87 umol/L and was shown to impede proliferation of acute myeloid leukemia (AML) cell lines (64). Additionally, a study in bladder cancer found that volasertib was a potent inducer of cell death and cell cycle arrest (65). Although BI6727 appeared promising as a monotherapy, in an effort to increase the efficacy of volasertib, it was also studied in combination with the MEK inhibitor Trematinib (JTP-74057) against cancer cells in vitro. The combination of drugs resulted in a dual G1 and G2-M arrest in an NRAS-mutant melanoma cell line (66). Additionally, cyclins D and E decreased whilst cyclin B and p21 increased and an overall reduction in melanoma growth was observed. Likewise, synergistic effects on clonogenic survival and cell proliferation were observed in prostate cancer lines when BI 6727 was combined with histone deacetylase (HDAC) inhibitors, which have previously been shown to downregulate Plk expression (67).
Like many chemotherapy drugs, it is possible for volasertib to become less effective over time as the cancer cells develop resistance. Overexpression of the ATP-binding cassette (ABC) drug transporter ABCB1 has been shown to reduce the effect of chemotherapeutic agents on cancer cells by utilizing ATP hydrolysis to transport the drugs out of cancer cells (68). Recently, Wu and colleagues found that the resistance is conferred via this mode of action with volasertib (69). Therefore, using a modulator of ABCB1 as a cotreatment in future trials may be an effective method to prevent this route of resistance.
Poloxin
Reindl and colleagues, in 2008, developed a small molecule called Poloxin that was shown to interfere with the function of the polo-box domain (PBD) of Plk1 (70). Yuan and colleagues demonstrated that Poloxin induced centrosome fragmentation and abnormal spindle and chromosome misalignment by modulating Kizuna, a key substrate of Plk1 at centrosomes (71). Poloxin also caused mitotic arrest and apoptosis of a number of cancer cell lines (71). Further, Poloxin significantly suppressed tumor growth in xenograft mouse models (71). These data suggested that targeting the PBD by agents such as Poloxin may be a useful strategy for cancer management.
In an effort to further improve the efficacy of Poloxin, recently, Scharow and colleagues reported identification of the optimized analog Poloxin-2, which was shown to have significantly improved potency and selectivity over Poloxin (72). Poloxin-2 arrests mitosis in human tumor cell lines and induces apoptosis at lower micromolar concentrations (IC50=1.36 umol/L), as compared with the IC50 of 4.8 umol/L of its predecessor (70). However, further in-depth studies are needed to determine its efficacy, both in mono- and multiple therapy regimens.
Other Plk1 inhibitors
Given the potential ‘druggability’ of Plk1 for cancer therapies, several new drugs targeting Plk1 have been reported in the last few years. These drugs are starting to be explored, so not much information is available on them. One such agent, Tak 960, was discovered through optimization of a novel series of pyrimidodiazepinone Plk1 inhibitors (73). Tak960 has an IC50 of 2 nmol/L, and a study in HT-29 colorectal cells found that they were arrested in G2-M phase when treated with Tak960 (74). The same study found that aberrant spindle accumulation was apparent with higher doses of Tak960. Another study demonstrated that Tak960 causes an induction of apoptosis, possibly due to a down regulation of Mcl-1 in sarcoma cells (75). Another Plk1 inhibitor, NMS-P937, has shown notable success in the reduction of osteosarcoma tumor growth (76). Interestingly, this inhibitor is a pyrazoloquinazoline that can be administered orally rather than via intravenous administration like other Plk1 inhibitors (77). With an IC50 of 36 nmol/L, NMS-P937 has also shown high selectivity against Plk1 in acute myeloid leukemia-NS8 cells (78). In vivo, the inhibitor significantly prolonged median survival time (MST) compared with standard therapies for AML (51 vs. 41 days, respectively), the inhibitor also led to a significantly longer MST in comparison with cytarabine (62 vs. 36 days). NMS-P937 has started phase 1 trials for patients with advanced metastatic solid tumors, but no results have been published as of yet.
Plk1 Inhibitors in Clinical Trials
In addition to the studies mentioned above, several drugs that target Plk1 are being tested in clinical trials following their preclinical success. Below, we discuss some of the completed Plk1-targeting clinical trials (also outlined in Table 1) and their results in various human cancers.
Rigosertib (ON 01910.Na)
The benzyl sulfone analogue rigosertib acts as a Ras mimetic and as a non-ATP-competitive small molecule inhibitor of Plk1 and PI3K (79, 80). Although not Plk1 specific, rigosertib is an interesting compound, owing to its dual targetability affecting both Plk1 and PI3K, which are important pathways in cancer progression. In a phase 1 study, oral rigosertib was administered to patients with advanced solid malignancies (81). A response rate of 4.6% (2 of 46 patients) was observed, with 1 patient achieving complete response and the other reaching partial response status; both patients had head and neck squamous cell carcinoma. Additionally, 8 patients maintained stable disease for ≥ 12 weeks, thus the overall response rate (complete response (CR) + partial response (PR) + stable disease) of the study was 21.7% (10/46 patients). Urinary toxicities were the most common side effect and the recommended dose of oral rigosertib was determined to be 560 mg twice daily (81).
As mentioned above, Plk1 inhibitors have been used by some researchers successfully to sidestep acquired gemcitabine resistance in certain cancer lines. This idea was brought forward into a phase III trial of metastatic pancreatic adenocarcinoma, when gemcitabine monotherapy was compared with a combination of gemcitabine and rigosertib. The combination produced a greater partial response in patients (19%) than with gemcitabine alone (13%). However, the median overall survival was 3 months shorter in the combination group than in gemcitabine alone (6.1 months vs. 6.3 months). The combination was thus deemed unsuccessful at improving survival rates (82).
Volasertib
A number of clinical trials involving volasertib have been undertaken in recent years. In a phase 1 study, two dosing schedules of volasertib were investigated in patients with advanced solid cancers (83). Patients on schedule A had IV volasertib on day 1 every 3 weeks, whilst patients on schedule B had IV volasertib on days 1 and 8 every 3 weeks. The maximum tolerated dose was 300 mg and 150 mg, respectively, and the main adverse effects were hematological, including neutropenia, leukopenia and thrombocytopenia. Two patients achieved partial response, one from each schedule group. Overall, 26 patients (44.1%) showed stable disease, 14 patients in schedule A (43.1%) and 12 in schedule B (44.4%) (83). However, in a separate phase 2 trial, patients with metastatic urothelial carcinoma were infused with 300 mg volasertib daily for 21 days. 46% of patients escalated to 350 mg which suggests a manageable safety profile. However, antitumor activity was limited as only 14% of the 50 patients achieved a partial response (84). It is likely that volasertib will be best used with only certain cancer types, as it appears to have better response rates in some than others. To overcome this, combination therapy trials are being actively pursued, and the results of a few clinical trials have been released on certain combinations.
Recently, a phase I, dose-escalation study of volasertib in combination with Nintedanib, a potent angiokinase inhibitor of PDGF, VEGF, and bFGF receptors was reported in advanced solid tumors (85). In patients with solid tumors, one complete response and one partial response were observed at a dosage of 300 mg volasertib plus 200 mg nintedanib. The combination was considered well tolerated with manageable adverse effects (85). In another combination phase II trial, volasertib combined with low dose of the chemotherapeutic agent cytarabine was shown to have an improved response rate and longer survival rates compared to cytarabine monotherapy (86). Based on these positive results volasertib is now in phase III trials to determine its efficacy and safety on a larger sample of patients. This newest trial involves 660 patients with untreated acute myeloid leukemia; half the participants will receive volasertib in combination with low dose cytarabine while the other group will be treated with cytarabine alone. This study is expected to be completed in 2016 (87).
GSK461364
Another ATP-competitive Plk1 inhibitor that is being investigated in clinical trials is the compound GSK461364. This thiophene amide developed by GlaxoSmithKline has been shown to promote G2-M arrest in tumor tissues (88, 89). In a phase I trial, patients with solid malignancies were treated with escalating doses of GSK461364 (90). Group 1 were dosed on days 1, 8, and 15 of 28-day cycles and group 2 on days 1, 2, 8, 9, 15, and 16 of 28-day cycles. Dose limiting toxicities occurred at 300 mg and 225 mg, respectively; with the most common toxicity identified as venous thrombotic emboli (90). The recommended dose for phase II was found to be 225 mg in a group 1 schedule, in combination with an anticoagulant to counteract adverse effects of the inhibitor treatment (90). GSK461364 has also been found to inhibit ABCB1-mediated calcein-AM outflow (91). However, the antitumor activity of GSK461364 is shown to be reduced when ABCB1 is overexpressed. Like the results found in vitro, re-sensitization to GSK461364 occurs when ABCB1 is inhibited (91).
Conclusions
As described above, Plk1 inhibition is being viewed as a potential strategy to develop novel therapeutics for cancer management. A number of Plk1 inhibitors, some of which have been described here, are in various stages of development and clinical trials. In a number of cases, the results of preclinical studies have not been found to be translatable to the clinical trials. For example, BI 2536 showed promise in preclinical work, but was not as effective in monotherapy regimens in clinical trials. Nevertheless, a variety of different cancers have responded in some manner to the Plk1 inhibitor drugs in clinical trials, either by exhibiting a partial or complete response or by stabilizing the disease. Volasertib is arguably one of the most effective Plk1 inhibitors in vitro and in vivo to date and has shown potential in phase I and II trials, and is being tested in a Phase III trial. This inhibitor has also been tried in combination with other inhibitors with encouraging success.
PBD inhibitors are unique to polo-like kinases and are therefore much more specific than inhibitors that target the ATP-binding domain, which can be found in over 500 protein kinases and is vital for kinase activity (92). However, the majority of inhibitors currently in trials are directed towards the less specific ATP-binding domain. The PBD inhibitor Poloxin is showing promise in early preclinical trials but more research is required to efficiently target the PBD. Using this drug or other PBD inhibitors in combination with another chemotherapy or pathway-specific inhibitor drug would likely give a more targeted response than with other Plk1 inhibitors. Combination treatments have become more frequently used in trials in recent years in order to combat limitations of monotherapy. In a number of studies, the observed response of combination has been better and/or synergistic compared with monotherapy. The main side effects associated with Plk1 inhibitors have not been significantly adverse and can likely be managed by other drugs such as anticoagulants, though it is important to consider the additional side effects that may occur with two drugs over one.
Like a number of other targeted therapies, resistance appears to be an omnipresent problem with small-molecule Plk1 inhibitors, possibly due to Plk1s– many roles/interaction in the cell cycle. This seems to be only addressable by combination therapies. In this direction, it may also be useful to attempt two agents that have an ability to target Plk1 as well as other pathways important for specific cancers. In addition, the next generation of Plk1 inhibitors with improved pharmacokinetic profile may also be suitable.
Acknowledgments
Financial Information: This work was partially supported by funding from the NIH (R01AR059130 and R01CA176748; to N. Ahmad) and the Department of Veterans Affairs (VA Merit Review Award 1I01BX001008; to N. Ahmad).
Footnotes
Conflicts of Interest: The authors have no conflicts of interest to disclose.
References
- 1.Siegel RL, Miller KD, Jemal A. Cancer statistics, 2016. CA Cancer J Clin. 2016;66:7–30. doi: 10.3322/caac.21332. [DOI] [PubMed] [Google Scholar]
- 2.Gerber DE. Targeted therapies: a new generation of cancer treatments. Am Fam Physician. 2008;77:311–9. [PubMed] [Google Scholar]
- 3.Nastiuk KL, Krolewski JJ. Opportunities and challenges in combination gene cancer therapy. Adv Drug Deliv Rev. 2016;98:35–40. doi: 10.1016/j.addr.2015.12.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Sunkel CE, Glover DM. polo, a mitotic mutant of Drosophila displaying abnormal spindle poles. J Cell Sci. 1988;89:25–38. doi: 10.1242/jcs.89.1.25. [DOI] [PubMed] [Google Scholar]
- 5.Golsteyn RM, Schultz SJ, Bartek J, Ziemiecki A, Ried T, Nigg EA. Cell cycle analysis and chromosomal localization of human Plk1, a putative homologue of the mitotic kinases Drosophila polo and Saccharomyces cerevisiae Cdc5. J Cell Sci. 1994;107:1509–17. doi: 10.1242/jcs.107.6.1509. [DOI] [PubMed] [Google Scholar]
- 6.Liu X. Targeting Polo-Like Kinases: A Promising Therapeutic Approach for Cancer Treatment. Transl Oncol. 2015;8:185–95. doi: 10.1016/j.tranon.2015.03.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.de Cárcer G, Manning G, Malumbres M. From Plk1 to Plk5: Functional evolution of polo-like kinases. Cell Cycle. 2011;10:2255–62. doi: 10.4161/cc.10.14.16494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Lens SMA, Voest EE, Medema RH. Shared and separate functions of polo-like kinases and aurora kinases in cancer. Nat Rev Cancer. 2010;10:825–41. doi: 10.1038/nrc2964. [DOI] [PubMed] [Google Scholar]
- 9.Luo YB, Kim NH. PLK4 Is Essential for Meiotic Resumption in Mouse Oocytes. Biol Reprod. 2015;92:101, 1–8. doi: 10.1095/biolreprod.114.124065. [DOI] [PubMed] [Google Scholar]
- 10.de Cárcer G, Escobar B, Higuero AM, Garcia L, Anson A, Perez G, et al. Plk5, a polo box domain-only protein with specific roles in neuron differentiation and glioblastoma suppression. Mol Cell Biol. 2011;31:1225–39. doi: 10.1128/MCB.00607-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.von Schubert C, Cubizolles F, Bracher JM, Sliedrecht T, Kops GJ, Nigg EA. Plk1 and Mps1 Cooperatively Regulate the Spindle Assembly Checkpoint in Human Cells. Cell Rep. 2015;12:66–78. doi: 10.1016/j.celrep.2015.06.007. [DOI] [PubMed] [Google Scholar]
- 12.Baran V, Brzakova A, Rehak P, Kovarikova V, Solc P. PLK1 regulates spindle formation kinetics and APC/C activation in mouse zygote. Zygote. 2015:1–8. doi: 10.1017/S0967199415000246. [DOI] [PubMed] [Google Scholar]
- 13.Liu X, Erikson RL. Polo-like kinase (Plk)1 depletion induces apoptosis in cancer cells. Proc Natl Acad Sci U S A. 2003;100:5789–94. doi: 10.1073/pnas.1031523100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Takaki T, Trenz K, Costanzo V, Petronczki M. Polo-like kinase 1 reaches beyond mitosis—cytokinesis, DNA damage response, and development. Curr Opin Cell Biol. 2008;20:650–60. doi: 10.1016/j.ceb.2008.10.005. [DOI] [PubMed] [Google Scholar]
- 15.Shaltiel IA, Krenning L, Bruinsma W, Medema RH. The same, only different – DNA damage checkpoints and their reversal throughout the cell cycle. J Cell Sci. 2015;128:607–20. doi: 10.1242/jcs.163766. [DOI] [PubMed] [Google Scholar]
- 16.Chan EH, Santamaria A, Sillje HH, Nigg EA. Plk1 regulates mitotic Aurora A function through betaTrCP-dependent degradation of hBora. Chromosoma. 2008;117:457–69. doi: 10.1007/s00412-008-0165-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Seki A, Coppinger JA, Jang CY, Yates JR, Fang G. Bora and Aurora A Cooperatively Activate Plk1 and Control the Entry into Mitosis. Science. 2008;320:1655–8. doi: 10.1126/science.1157425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Shao H, Huang Y, Zhang L, Yuan K, Chu Y, Dou Z, et al. Spatiotemporal dynamics of Aurora B-PLK1-MCAK signaling axis orchestrates kinetochore bi-orientation and faithful chromosome segregation. Sci Rep. 2015;5:12204. doi: 10.1038/srep12204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Takai N, Hamanaka R, Yoshimatsu J, Miyakawa I. Polo-like kinases (Plks) and cancer. Oncogene. 2005;24:287–91. doi: 10.1038/sj.onc.1208272. [DOI] [PubMed] [Google Scholar]
- 20.Yamamoto Y, Matsuyama H, Kawauchi S, Matsumoto H, Nagao K, Ohmi C, et al. Overexpression of Polo-Like Kinase 1 (PLK1) and Chromosomal Instability in Bladder Cancer. Oncology. 2006;70:231–7. doi: 10.1159/000094416. [DOI] [PubMed] [Google Scholar]
- 21.Tokumitsu Y, Mori M, Tanaka S, Akazawa K, Nakano S, Niho Y. Prognostic significance of polo-like kinase expression in esophageal carcinoma. Int J Oncol. 1999;15:687–92. doi: 10.3892/ijo.15.4.687. [DOI] [PubMed] [Google Scholar]
- 22.Rivankar S. An overview of doxorubicin formulations in cancer therapy. J Cancer Res Ther. 2014;10:853–8. doi: 10.4103/0973-1482.139267. [DOI] [PubMed] [Google Scholar]
- 23.Forrest RA, Swift LP, Rephaeli A, Nudelman A, Kimura K, Phillips DR, et al. Activation of DNA damage response pathways as a consequence of anthracycline-DNA adduct formation. Biochem Pharmacol. 2012;83:1602–12. doi: 10.1016/j.bcp.2012.02.026. [DOI] [PubMed] [Google Scholar]
- 24.Olsen TW, Palejwala NV, Lee LB, Bergstrom CS, Yeh S. Chorioretinal folds: associated disorders and a related maculopathy. Am J Ophthalmol. 2014;157:1038–47. doi: 10.1016/j.ajo.2014.02.021. [DOI] [PubMed] [Google Scholar]
- 25.Liu XS, Li H, Song B, Liu X. Polo-like kinase 1 phosphorylation of G2 and S-phase-expressed 1 protein is essential for p53 inactivation during G2 checkpoint recovery. EMBO Rep. 2010;11:626–32. doi: 10.1038/embor.2010.90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Yang X, Li H, Zhou Z, Wang WH, Deng A, Andrisani O, et al. Plk1-mediated phosphorylation of Topors regulates p53 stability. J Biol Chem. 2009;284:18588–92. doi: 10.1074/jbc.C109.001560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Monte M, Benetti R, Collavin L, Marchionni L, Del Sal G, Schneider C. hGTSE-1 expression stimulates cytoplasmic localization of p53. J Biol Chem. 2004;279:11744–52. doi: 10.1074/jbc.M311123200. [DOI] [PubMed] [Google Scholar]
- 28.Rajendra R, Malegaonkar D, Pungaliya P, Marshall H, Rasheed Z, Brownell J, et al. Topors functions as an E3 ubiquitin ligase with specific E2 enzymes and ubiquitinates p53. J Biol Chem. 2004;279:36440–4. doi: 10.1074/jbc.C400300200. [DOI] [PubMed] [Google Scholar]
- 29.Weger S, Hammer E, Heilbronn R. Topors acts as a SUMO-1 E3 ligase for p53 in vitro and in vivo. FEBS Lett. 2005;579:5007–12. doi: 10.1016/j.febslet.2005.07.088. [DOI] [PubMed] [Google Scholar]
- 30.Yang X, Li H, Deng A, Liu X. Plk1 phosphorylation of Topors is involved in its degradation. Mol Biol Rep. 2010;37:3023–8. doi: 10.1007/s11033-009-9871-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ewald B, Sampath D, Plunkett W. ATM and the Mre11-Rad50-Nbs1 complex respond to nucleoside analogue-induced stalled replication forks and contribute to drug resistance. Cancer Res. 2008;68:7947–55. doi: 10.1158/0008-5472.CAN-08-0971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Song B, Liu XS, Davis K, Liu X. Plk1 phosphorylation of Orc2 promotes DNA replication under conditions of stress. Mol Cell Biol. 2011;31:4844–56. doi: 10.1128/MCB.06110-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Song B, Davis K, Liu XS, Lee HG, Smith M, Liu X. Inhibition of Polo-like kinase 1 reduces beta-amyloid-induced neuronal cell death in Alzheimer's disease. Aging (Albany NY) 2011;3:846–51. doi: 10.18632/aging.100382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Song B, Liu XS, Rice SJ, Kuang S, Elzey BD, Konieczny SF, et al. Plk1 phosphorylation of orc2 and hbo1 contributes to gemcitabine resistance in pancreatic cancer. Mol Cancer Ther. 2013;12:58–68. doi: 10.1158/1535-7163.MCT-12-0632. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Wu ZQ, Liu X. Role for Plk1 phosphorylation of Hbo1 in regulation of replication licensing. Proc Natl Acad Sci U S A. 2008;105:1919–24. doi: 10.1073/pnas.0712063105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Jordan MA, Wilson L. Microtubules as a target for anticancer drugs. Nat Rev Cancer. 2004;4:253–65. doi: 10.1038/nrc1317. [DOI] [PubMed] [Google Scholar]
- 37.Berges RR, Vukanovic J, Epstein JI, CarMichel M, Cisek L, Johnson DE, et al. Implication of cell kinetic changes during the progression of human prostatic cancer. Clin Cancer Res. 1995;1:473–80. [PMC free article] [PubMed] [Google Scholar]
- 38.Thadani-Mulero M, Nanus DM, Giannakakou P. Androgen receptor on the move: boarding the microtubule expressway to the nucleus. Cancer Res. 2012;72:4611–5. doi: 10.1158/0008-5472.CAN-12-0783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Darshan MS, Loftus MS, Thadani-Mulero M, Levy BP, Escuin D, Zhou XK, et al. Taxane-induced blockade to nuclear accumulation of the androgen receptor predicts clinical responses in metastatic prostate cancer. Cancer Res. 2011;71:6019–29. doi: 10.1158/0008-5472.CAN-11-1417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Perez F, Diamantopoulos GS, Stalder R, Kreis TE. CLIP-170 highlights growing microtubule ends in vivo. Cell. 1999;96:517–27. doi: 10.1016/s0092-8674(00)80656-x. [DOI] [PubMed] [Google Scholar]
- 41.Li H, Liu XS, Yang X, Wang Y, Turner JR, Liu X. Phosphorylation of CLIP-170 by Plk1 and CK2 promotes timely formation of kinetochore-microtubule attachments. EMBO J. 2010;29:2953–65. doi: 10.1038/emboj.2010.174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Hou X, Li Z, Huang W, Li J, Staiger C, Kuang S, et al. Plk1-dependent microtubule dynamics promotes androgen receptor signaling in prostate cancer. Prostate. 2013;73:1352–63. doi: 10.1002/pros.22683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Li H, Liu XS, Yang X, Song B, Wang Y, Liu X. Polo-like kinase 1 phosphorylation of p150Glued facilitates nuclear envelope breakdown during prophase. Proc Natl Acad Sci U S A. 2010;107:14633–8. doi: 10.1073/pnas.1006615107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Liu XS, Song B, Tang J, Liu W, Kuang S, Liu X. Plk1 phosphorylates Sgt1 at the kinetochores to promote timely kinetochore-microtubule attachment. Mol Cell Biol. 2012;32:4053–67. doi: 10.1128/MCB.00516-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Beer TM, Armstrong AJ, Rathkopf DE, Loriot Y, Sternberg CN, Higano CS, et al. Enzalutamide in metastatic prostate cancer before chemotherapy. N Engl J Med. 2014;371:424–33. doi: 10.1056/NEJMoa1405095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Ryan CJ, Molina A, Griffin T. Abiraterone in metastatic prostate cancer. N Engl J Med. 2013;368:1458–9. doi: 10.1056/NEJMc1301594. [DOI] [PubMed] [Google Scholar]
- 47.Zhang Z, Hou X, Shao C, Li J, Cheng JX, Kuang S, et al. Plk1 inhibition enhances the efficacy of androgen signaling blockade in castration-resistant prostate cancer. Cancer Res. 2014;74:6635–47. doi: 10.1158/0008-5472.CAN-14-1916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Thoma C. Prostate cancer: PLK-1 inhibition improves abiraterone efficacy. Nat Rev Urol. 2014;11:603. doi: 10.1038/nrurol.2014.287. [DOI] [PubMed] [Google Scholar]
- 49.Zhang Z, Chen L, Wang H, Ahmad N, Liu X. Inhibition of Plk1 represses androgen signaling pathway in castration-resistant prostate cancer. Cell Cycle. 2015;14:2142–8. doi: 10.1080/15384101.2015.1041689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Foretz M, Guigas B, Bertrand L, Pollak M, Viollet B. Metformin: from mechanisms of action to therapies. Cell Metab. 2014;20:953–66. doi: 10.1016/j.cmet.2014.09.018. [DOI] [PubMed] [Google Scholar]
- 51.Chan AT. Metformin for cancer prevention: a reason for optimism. Lancet Oncol. 2016 doi: 10.1016/S1470-2045(16)00006-1. Epub ahead of print. [DOI] [PubMed] [Google Scholar]
- 52.Shao C, Ahmad N, Hodges K, Kuang S, Ratliff T, Liu X. Inhibition of Polo-like Kinase 1 (Plk1) Enhances the Anti-neoplastic Activity of Metformin in Prostate Cancer. J Biol Chem. 2015;290:2024–33. doi: 10.1074/jbc.M114.596817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Locasale JW, Cantley LC. Metabolic flux and the regulation of mammalian cell growth. Cell Metab. 2011;14:443–51. doi: 10.1016/j.cmet.2011.07.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Garcia-Cao I, Song MS, Hobbs RM, Laurent G, Giorgi C, de Boer VC, et al. Systemic elevation of PTEN induces a tumor-suppressive metabolic state. Cell. 2012;149:49–62. doi: 10.1016/j.cell.2012.02.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Liu XS, Song B, Elzey BD, Ratliff TL, Konieczny SF, Cheng L, et al. Polo-like kinase 1 facilitates loss of Pten tumor suppressor-induced prostate cancer formation. J Biol Chem. 2011;286:35795–800. doi: 10.1074/jbc.C111.269050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Li Z, Li J, Bi P, Lu Y, Burcham G, Elzey BD, et al. Plk1 phosphorylation of PTEN causes a tumor-promoting metabolic state. Mol Cell Biol. 2014;34:3642–61. doi: 10.1128/MCB.00814-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Spratt DE, Zhang C, Zumsteg ZS, Pei X, Zhang Z, Zelefsky MJ. Metformin and prostate cancer: reduced development of castration-resistant disease and prostate cancer mortality. Eur Urol. 2013;63:709–16. doi: 10.1016/j.eururo.2012.12.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Steegmaier M, Hoffmann M, Baum A, Lenart P, Petronczki M, Krssak M, et al. BI 2536, a potent and selective inhibitor of polo-like kinase 1, inhibits tumor growth in vivo. Curr Biol. 2007;17:316–22. doi: 10.1016/j.cub.2006.12.037. [DOI] [PubMed] [Google Scholar]
- 59.Frost A, Mross K, Steinbild S, Hedbom S, Unger C, Kaiser R, et al. Phase i study of the Plk1 inhibitor BI 2536 administered intravenously on three consecutive days in advanced solid tumours. Curr Oncol. 2012;19:e28–35. doi: 10.3747/co.19.866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Mross K, Dittrich C, Aulitzky WE, Strumberg D, Schutte J, Schmid RM, et al. A randomised phase II trial of the Polo-like kinase inhibitor BI 2536 in chemo-naïve patients with unresectable exocrine adenocarcinoma of the pancreas – a study within the Central European Society Anticancer Drug Research (CESAR) collaborative network. Br J Cancer. 2012;107:280–6. doi: 10.1038/bjc.2012.257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Czaplinski S, Hugle M, Stiehl V, Fulda S. Polo-like kinase 1 inhibition sensitizes neuroblastoma cells for vinca alkaloid-induced apoptosis. Oncotarget. 2016;7:8700–11. doi: 10.18632/oncotarget.3901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Stehle A, Hugle M, Fulda S. Eribulin synergizes with Polo-like kinase 1 inhibitors to induce apoptosis in rhabdomyosarcoma. Cancer Lett. 2015;365:37–46. doi: 10.1016/j.canlet.2015.04.011. [DOI] [PubMed] [Google Scholar]
- 63.Rudolph D, Steegmaier M, Hoffmann M, Grauert M, Baum A, Quant J, et al. BI 6727, A Polo-like Kinase Inhibitor with Improved Pharmacokinetic Profile and Broad Antitumor Activity. Clin Cancer Res. 2009;15:3094–102. doi: 10.1158/1078-0432.CCR-08-2445. [DOI] [PubMed] [Google Scholar]
- 64.Rudolph D, Impagnatiello MA, Blaukopf C, Sommer C, Gerlich DW, Roth M, et al. Efficacy and Mechanism of Action of Volasertib, a Potent and Selective Inhibitor of Polo-Like Kinases, in Preclinical Models of Acute Myeloid Leukemia. J Pharmacol Exp Ther. 2015;352:579–89. doi: 10.1124/jpet.114.221150. [DOI] [PubMed] [Google Scholar]
- 65.Brassesco MS, Pezuk JA, Morales AG, de Oliveira JC, Roberto GM, da Silva GN, et al. In vitro targeting of Polo-like kinase 1 in bladder carcinoma: comparative effects of four potent inhibitors. Cancer Biol Ther. 2013;14:648–57. doi: 10.4161/cbt.25087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Posch C, Cholewa BD, Vujic I, Sanlorenzo M, Ma J, Kim ST, et al. Combined Inhibition of MEK and Plk1 Has Synergistic Antitumor Activity in NRAS Mutant Melanoma. J Invest Dermatol. 2015;135:2475–83. doi: 10.1038/jid.2015.198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Wissing MD, Mendonca J, Kortenhorst MSQ, Kaelber NS, Gonzalez M, Kim E, et al. Targeting prostate cancer cell lines with polo-like kinase 1 inhibitors as a single agent and in combination with histone deacetylase inhibitors. FASEB J. 2013;27:4279–93. doi: 10.1096/fj.12-222893. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Burger H, van Tol H, Brok M, Wiemer EA, de Bruijn EA, Guetens G, et al. Chronic imatinib mesylate exposure leads to reduced intracellular drug accumulation by induction of the ABCG2 (BCRP) and ABCB1 (MDR1) drug transport pumps. Cancer Biol Ther. 2005;4:747–52. doi: 10.4161/cbt.4.7.1826. [DOI] [PubMed] [Google Scholar]
- 69.Wu CP, Hsieh CH, Hsiao SH, Luo SY, Su CY, Li YQ, et al. Human ATP-Binding Cassette Transporter ABCB1 Confers Resistance to Volasertib (BI 6727), a Selective Inhibitor of Polo-like Kinase 1. Mol Pharm. 2015;12:3885–95. doi: 10.1021/acs.molpharmaceut.5b00312. [DOI] [PubMed] [Google Scholar]
- 70.Reindl W, Yuan J, Kramer A, Strebhardt K, Berg T. Inhibition of polo-like kinase 1 by blocking polo-box domain-dependent protein-protein interactions. Chem Biol. 2008;15:459–66. doi: 10.1016/j.chembiol.2008.03.013. [DOI] [PubMed] [Google Scholar]
- 71.Yuan J, Sanhaji M, Krämer A, Reindl W, Hofmann M, Kreis N-N, et al. Polo-Box Domain Inhibitor Poloxin Activates the Spindle Assembly Checkpoint and Inhibits Tumor Growth in Vivo. Am J Pathol. 2011;179:2091–9. doi: 10.1016/j.ajpath.2011.06.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Scharow A, Raab M, Saxena K, Sreeramulu S, Kudlinzki D, Gande S, et al. Optimized Plk1 PBD Inhibitors Based on Poloxin Induce Mitotic Arrest and Apoptosis in Tumor Cells. ACS Chem Biol. 2015;10:2570–9. doi: 10.1021/acschembio.5b00565. [DOI] [PubMed] [Google Scholar]
- 73.Nie Z, Feher V, Natala S, McBride C, Kiryanov A, Jones B, et al. Discovery of TAK-960: An orally available small molecule inhibitor of polo-like kinase 1 (PLK1). Bioorg Med Chem Lett. 2013;23:3662–6. doi: 10.1016/j.bmcl.2013.02.083. [DOI] [PubMed] [Google Scholar]
- 74.Hikichi Y, Honda K, Hikami K, Miyashita H, Kaieda I, Murai S, et al. TAK-960, a Novel, Orally Available, Selective Inhibitor of Polo-Like Kinase 1, Shows Broad-spectrum Preclinical Antitumor Activity in Multiple Dosing Regimens. Mol Cancer Ther. 2012;11:700–9. doi: 10.1158/1535-7163.MCT-11-0762. [DOI] [PubMed] [Google Scholar]
- 75.Nair JS, Schwartz GK. Inhibition of polo like kinase 1 in sarcomas induces apoptosis that is dependent on Mcl-1 suppression. Cell Cycle. 2015;14:3101–11. doi: 10.1080/15384101.2015.1078033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Sero V, Tavanti E, Vella S, Hattinger C, Fanelli M, Michelacci F, et al. Targeting polo-like kinase 1 by NMS-P937 in osteosarcoma cell lines inhibits tumor cell growth and partially overcomes drug resistance. Invest New Drugs. 2014;32:1167–80. doi: 10.1007/s10637-014-0158-6. [DOI] [PubMed] [Google Scholar]
- 77.Beria I, Ballinari D, Bertrand JA, Borghi D, Bossi RT, Brasca MG, et al. Identification of 4,5-dihydro-1H-pyrazolo[4,3-h]quinazoline derivatives as a new class of orally and selective Polo-like kinase 1 inhibitors. J Med Chem. 2010;53:3532–51. doi: 10.1021/jm901713n. [DOI] [PubMed] [Google Scholar]
- 78.Casolaro A, Golay J, Albanese C, Ceruti R, Patton V, Cribioli S, et al. The Polo-Like Kinase 1 (PLK1) Inhibitor NMS-P937 Is Effective in a New Model of Disseminated Primary CD56(+) Acute Monoblastic Leukaemia. PLoS One. 2013;8:e58424. doi: 10.1371/journal.pone.0058424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Komrokji RS, Raza A, Lancet JE, Ren C, Taft D, Maniar M, et al. Phase I clinical trial of oral rigosertib in patients with myelodysplastic syndromes. Br J Haematol. 2013;162:517–24. doi: 10.1111/bjh.12436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Gumireddy K, Reddy MV, Cosenza SC, Boominathan R, Baker SJ, Papathi N, et al. ON01910, a non-ATP-competitive small molecule inhibitor of Plk1, is a potent anticancer agent. Cancer Cell. 2005;7:275–86. doi: 10.1016/j.ccr.2005.02.009. [DOI] [PubMed] [Google Scholar]
- 81.Bowles DW, Diamond JR, Lam ET, Weekes CD, Astling DP, Anderson RT, et al. Phase I Study of Oral Rigosertib (ON 01910.Na), a Dual Inhibitor of the PI3K and Plk1 Pathways, in Adult Patients with Advanced Solid Malignancies. Clin Cancer Res. 2014;20:1656–65. doi: 10.1158/1078-0432.CCR-13-2506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.O'Neil BH, Scott AJ, Ma WW, Cohen SJ, Aisner DL, Menter AR, et al. A phase II/III randomized study to compare the efficacy and safety of rigosertib plus gemcitabine versus gemcitabine alone in patients with previously untreated metastatic pancreatic cancer. Ann Oncol. 2015;26:1923–9. doi: 10.1093/annonc/mdv264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Lin CC, Su WC, Yen CJ, Hsu CH, Su WP, Yeh KH, et al. A phase I study of two dosing schedules of volasertib (BI 6727), an intravenous polo-like kinase inhibitor, in patients with advanced solid malignancies. Br J Cancer. 2014;110:2434–40. doi: 10.1038/bjc.2014.195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Stadler WM, Vaughn DJ, Sonpavde G, Vogelzang NJ, Tagawa ST, Petrylak DP, et al. An open-label, single-arm, phase 2 trial of the polo-like kinase inhibitor volasertib (BI 6727) in patients with locally advanced or metastatic urothelial cancer. Cancer. 2014;120:976–82. doi: 10.1002/cncr.28519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.de Braud F, Cascinu S, Spitaleri G, Pilz K, Clementi L, Liu D, et al. A phase I, dose-escalation study of volasertib combined with nintedanib in advanced solid tumors. Ann Oncol. 2015;26:2341–6. doi: 10.1093/annonc/mdv354. [DOI] [PubMed] [Google Scholar]
- 86.Dohner H, Lubbert M, Fiedler W, Fouillard L, Haaland A, Brandwein JM, et al. Randomized, phase 2 trial of low-dose cytarabine with or without volasertib in AML patients not suitable for induction therapy. Blood. 2014;124:1426–33. doi: 10.1182/blood-2014-03-560557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Gjertsen BT, Schöffski P. Discovery and development of the Polo-like kinase inhibitor volasertib in cancer therapy. Leukemia. 2015;29:11–9. doi: 10.1038/leu.2014.222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Gilmartin AG, Bleam MR, Richter MC, Erskine SG, Kruger RG, Madden L, et al. Distinct concentration-dependent effects of the polo-like kinase 1-specific inhibitor GSK461364A, including differential effect on apoptosis. Cancer Res. 2009;69:6969–77. doi: 10.1158/0008-5472.CAN-09-0945. [DOI] [PubMed] [Google Scholar]
- 89.Schöffski P. Polo-Like Kinase (PLK) Inhibitors in Preclinical and Early Clinical Development in Oncology. Oncologist. 2009;14:559–70. doi: 10.1634/theoncologist.2009-0010. [DOI] [PubMed] [Google Scholar]
- 90.Olmos D, Barker D, Sharma R, Brunetto AT, Yap TA, Taegtmeyer AB, et al. Phase I Study of GSK461364, a Specific and Competitive Polo-like Kinase 1 Inhibitor, in Patients with Advanced Solid Malignancies. Clin Cancer Res. 2011;17:3420–30. doi: 10.1158/1078-0432.CCR-10-2946. [DOI] [PubMed] [Google Scholar]
- 91.Wu CP, Hsiao SH, Sim HM, Luo SY, Tuo WC, Cheng HW, et al. Human ABCB1 (P-glycoprotein) and ABCG2 Mediate Resistance to BI 2536, a Potent and Selective Inhibitor of Polo-like Kinase 1. Biochem Pharmacol. 2013;86:904–13. doi: 10.1016/j.bcp.2013.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Shin S-B, Woo S-U, Yim H. Differential Cellular Effects of Plk1 Inhibitors Targeting the ATP-binding Domain or Polo-box Domain. J Cell Physiol. 2015;230:3057–67. doi: 10.1002/jcp.25042. [DOI] [PubMed] [Google Scholar]