Abstract
Background
Great biodiversity is a highlight of Brazilian flora. In contrast, the therapeutic potentialities of most species used in folk medicine remain unknown. Several of these species are commonly used to treat cancer. In this study, we investigated the cytotoxic activity of 18 plants from 16 families that are found in the northeast region of Brazil.
Methods
The following species were studied: Byrsonima sericea DC. (Malpighiaceae), Cupania impressinervia Acev. Rodr. var. (revoluta) Radlk (Sapindaceae), Duranta repens Linn. (Verbenaceae), Helicostylis tomentosa (Poepp. & Endl) Rusby (Moraceae), Himatanthus bracteatus (A.DC.) Woodson (Apocynaceae), Ipomoea purga (Wender.) Hayne (Convolvulaceae), Ixora coccinea Linn. (Rubiaceae), Mabea piriri Aubl. (Euphorbiaceae), Miconia minutiflora (Melastomataceae), Momordica charantia L. (Cucurbitaceae), Ocotea glomerata (Nees) Mez (Lauraceae), Ocotea longifolia Kunth (Oreodaphne opifera Mart. Nees) (Lauraceae), Pavonia fruticosa (Mill.) Fawc. & Rendle (Malvaceae), Psychotria capitata Ruiz & Pav. (Rubiaceae), Schefflera morototoni (Aubl.) Maguire, Steyerm. & Frodin (Araliaceae), Solanum paludosum Moric. (Solanaceae), Xylopia frutescens Aubl. (Annonaceae) and Zanthoxylum rhoifolium Lam. (Rutaceae). Their dried leaves, stems, flowers or fruits were submitted to different solvent extractions, resulting in 55 extracts. After incubating for 72 h, the cytotoxicity of each extract was tested against tumor cell lines using the alamar blue assay.
Results
The B. sericea, D. repens, H. bracteatus, I. purga, I. coccinea, M. piriri, O. longifolia and P. capitata extracts demonstrated the most potent cytotoxic activity. The chloroform soluble fractions of D. repens flowers and the hexane extract of I. coccinea flowers led to the isolation of quercetin and a mixture of α- and β-amyrin, respectively, and quercetin showed moderate cytotoxic activity.
Conclusion
The B. sericea, D. repens, H. bracteatus, I. purga, I. coccinea, M. piriri, O. longifolia and P. capitata plants were identified as having potent cytotoxic effects. Further investigations are required to determine the mechanisms of cytotoxicity exhibited and their in vivo activities. This work reinforces the need to understand the therapeutics potentialities of Brazilian medicinal plants.
Keywords: Cytotoxicity, Northeastern Brazil, Medicinal plants, Natural products
Background
In Brazil, the estimated number of described plant species (terrestrial and algae) ranges from 50,542 to 60,042 [1]; they are found in the following six different biomes: Amazônia (the Amazon rainforest in northern and central-western Brazil), Cerrado (the central Brazilian savanna), Mata Atlântica (the Atlantic rainforest ranging from sea level to the eastern highlands of Brazil), Caatinga (a xerophilous thorny forest found in northeastern Brazil), Pampa (the grasslands in southern Brazil) and Pantanal (periodically flooded grasslands by the Paraná and Paraguay rivers in central-western Brazil). Thus, Brazil is one of the most biodiverse nations in the world [1–3].
The cytotoxic potential of some Brazilian medicinal plants has been previously investigated [4–7]; however, due to the country’s great biodiversity, the therapeutic potentialities of most species remain unknown. In this work, the cytotoxic activities of 18 plants belonging to 16 families found in northeast Brazil were assessed against tumor cell lines. The following species were studied: Byrsonima sericea DC. (Malpighiaceae), Cupania impressinervia Acev. Rodr. var. (revoluta) Radlk (Sapindaceae), Duranta repens Linn. (Verbenaceae), Helicostylis tomentosa (Poepp. & Endl) Rusby (Moraceae), Himatanthus bracteatus (A.DC.) Woodson (Apocynaceae), Ipomoea purga (Wender.) Hayne (Convolvulaceae), Ixora coccinea Linn. (Rubiaceae), Mabea piriri Aubl. (Euphorbiaceae), Miconia minutiflora (Melastomataceae), Momordica charantia L. (Cucurbitaceae), Ocotea glomerata (Nees) Mez (Lauraceae), Ocotea longifolia Kunth (Oreodaphne opifera Mart. Nees) (Lauraceae), Pavonia fruticosa (Mill.) Fawc. & Rendle (Malvaceae), Psychotria capitata Ruiz & Pav. (Rubiaceae), Schefflera morototoni (Aubl.) Maguire, Steyerm. & Frodin (Araliaceae), Solanum paludosum Moric. (Solanaceae), Xylopia frutescens Aubl. (Annonaceae) and Zanthoxylum rhoifolium Lam. (Rutaceae). The plant species, family, vernacular name and traditional use in Brazil are shown in Table 1.
Table 1.
List of plants and their vernacular name and folk use in Brazil
| Plant species | Family | Vernacular name | Folk use in Brazil |
|---|---|---|---|
| Byrsonima sericea DC. | Malpighiaceae | Murici-da-mata | A decoction of the stem-bark is used to treat fevers, diarrhea, syphilis and kidney disease [26] |
| Cupania impressinervia Acev. Rodr. var. (revoluta) Radlk | Sapindaceae | Caboatã-de-rego | - |
| Duranta repens Linn. | Verbenaceae | Pingo-de-ouro | Ornamental [27] |
| Helicostylis tomentosa (Poepp. & Endl) Rusby | Moraceae | Amoreira-preta | Its fruit is used as a nutritional source [28] |
| Himatanthus bracteatus (A.DC.) Woodson | Apocynaceae | Janaguba | The latex is topically applied to treat external ulcers and tumors. It is dropped in a liter of water against inflammation and cancer [26] |
| Ipomoea purga (Wender.) Hayne | Convolvulaceae | Jalapa, batata-de-purga | It is used as an analgesic [29] |
| Ixora coccinea Linn. | Rubiaceae | Ixora | Ornamental [30] |
| Mabea piriri Aubl. | Euphorbiaceae | Canudo de cachimbo | - |
| Miconia minutiflora | Melastomataceae | Mundururu | Its fruit is used as a nutritional source [28] |
| Momordica charantia L. | Cucurbitaceae | Melão-de-sabiá, melão-de-são-caetano, galinha-de-melão | A decoction of a handful in a liter of water is used as a tea to treat diabetes, parasite infections, rheumatism and diarrhea [26] |
| Ocotea glomerata (Nees) Mez | Lauraceae | Caneleira, louro-branco, louro-abacate, louro-bravo | The leaves are used to treat hydropsy, digestive problems and high blood pressure [31, 32] |
| Ocotea longifolia Kunth (Oreodaphne opifera Mart. Nees) | Lauraceae | Canela de cheiro | Fruit oil is used to treat arthralgia and rheumatic diseases [33] |
| Pavonia fruticosa (Mill.) Fawc. & Rendle | Malvaceae | - | - |
| Psychotria capitata Ruiz & Pav. | Rubiaceae | - | - |
| Schefflera morototoni (Aubl.) Maguire, Steyerm. & Frodin | Araliaceae | Matatauba, Marupá | Its fruit is used as a nutritional source and to make local crafts [28] |
| Solanum paludosum Moric. | Solanaceae | Jurubeba-brava, jurubeba-roxa | An infusion of the roots is used to treat hepatic diseases. The fruit is used as a poison [26] |
| Xylopia frutescens Aubl. | Annonaceae | Embira, semente-de-embira | Its seeds and fruits are used as a digestive aid (a decoction of a teaspoon in a cup of water). It is used as a tea after meals [26] |
| Zanthoxylum rhoifolium Lam. | Rutaceae | Mamica de cadela, mamica de porca | The roots are used as a febrifuge, digestant and tonic. The stem bark is used to treat flatulence, colic, dyspepsia, ear aches, toothaches and snake bites [34] |
Methods
Plant material
Leaves, stems, fruits and/or flowers of B. sericea (voucher number 45628), C. impressinervia (voucher number 42953), D. repens (voucher number 25870), H. tomentosa (voucher number 20586), H. bracteatus (voucher number 50733), I. purga (voucher number 33255) and I. coccinea (voucher number 25861) were collected in February 2014 in the Municipality of Viçosa, Alagoas, Brazil. Leaves and/or stems of M. piriri (voucher number 45717), M. minutiflora (voucher number 49653), M. charantia (voucher number 34393), O. glomerata (voucher number 50577), O. longifolia (voucher number 50577), P. fruticosa (voucher number 45659) and P. capitata (voucher number 45620) were collected in March 2014 in the municipality of Rio Largo, Alagoas, Brazil. Leaves, stems and/or fruits of S. morototoni (voucher number 45659), S. paludosum (voucher number 37400), X. frutescens (voucher number 45677) and Z. rhoifolium (voucher number 49656) were collected in March 2014 in the Municipality of Murici, Alagoas, Brazil. All plants were collected in the Mata Atlântica bioma. The species were identified by Rosângela Pereira e Lyra Lemos, a plant taxonomist from the Instituto do Meio Ambiente de Alagoas IMA-AL. Voucher botanical specimens are stored at the Herbarium of the Instituto do Meio Ambiente de Alagoas IMA-AL. This work was performed according to the special authorization for access to genetic resources in Brazil # 010344/2014-4, issued by CNPq/MCTI.
Plant extractions
The dried leaves, stems, flowers or fruits were triturated and extracted at room temperature with ethanol or hexane. The solvent was removed to produce the correspondent crude extracts. The extraction steps for each sample were repeated three times with one-week intervals. Part of leaves, fruits or flowers ethanol extract was suspended in methanol:water (9:1) solution and extracted successively with ethyl acetate, chloroform, hexane or hexane:ethyl acetate (1:1) to produce the corresponding partitioned extracts.
Chemical fractionation
General procedures
Purification processes were performed through column chromatography (CC) using silica gel 60 (0.063-0.200 mm) as the stationary phase. Organic solvents or mixtures of increasing polarity were used as mobile phases. Silica gel 60 GF (Merck, Darmstadt, Germany) was used to perform analytical (0.25 mm) or preparative (0.75 mm) thin layer chromatographic (TLC) processes.
The 1H (400 MHz) and 13C (100 MHz) nuclear magnetic resonance (NMR) spectra were obtained on a Bruker Avance DRX-400 spectrometer (Bruker, Rheinstetten, Germany), operating at 300 K. The chemical shifts (δ) were expressed in units of ppm, using tetramethylsilane (TMS) as a reference (δH = δc = 0), and the coupling constants (J) were expressed in Hz. Chloroform-d was used as the solvent for the samples.
Isolation of compounds
The chloroform extract of D. repens flowers (7 g) was submitted to silica gel CC eluted with hexane and ethyl acetate (either pure or a mixture of increasing polarity), providing 50 fractions (5 mL each) that were grouped with hexane and ethyl acetate. After solvent evaporation, fractions 30–50 were purified with Sephadex-L water:methanol, yielding the compound quercetin (56, 11.0 mg). The compound structure was identified through a series of spectrometric data, such as NMR, as well as by comparison with data reported in the literature.
A hexane extract of I. coccinea flowers (26 g) was submitted to silica gel CC eluted with hexane, ethyl acetate and methanol (either pure or a mixture of increasing polarity), providing 100 fractions (5 mL each) that were grouped with hexane, ethyl acetate and methanol. After solvent evaporation from fractions 30 to 45 (eluted with hexane/ethyl acetate 8:2), a white solid (in the form of flakes) was obtained (57, 14.0 mg). This solid was identified as a mixture of α- and β-amyrin. The compounds structures were identified through a series of spectrometric data, such as NMR, as well as by comparison with data reported in the literature.
Cells
Tumor cells lines B16-F10 (mouse melanoma), HepG2 (human hepatocellular carcinoma), K562 (human chronic myelocytic leukemia) and HL-60 (human promyelocytic leukemia) were donated by the Hospital A.C. Camargo, São Paulo, SP, Brazil. This panel of cell lines include different types of cell histology with different sensibility. Cells were maintained in Roswell Park Memorial Institute-1640 (RPMI-1640, Gibco-BRL, Gaithersburg, USA) medium supplemented with 10 % fetal bovine serum (Cultilab, Campinas, Brazil), 2 mM L-glutamine (Vetec Química Fina, Duque de Caxias, Brazil) and 50 μg/mL gentamycin (Novafarma, Anápolis, Brazil). Adherent cells were harvested by treatment with 0.25 % trypsin EDTA solution (Gibco-BRL, Gaithersburg, USA). All cell lines were cultured in cell culture flasks at 37 °C in 5 % CO2 and sub-cultured every 3–4 days to maintain exponential growth. All experiments were conducted with cells in the exponential growth phase. All cell lines were tested for mycoplasma with a Mycoplasma Stain Kit (Sigma-Aldrich, St Louis, MO, USA) and found to be free from contamination.
Heparinized blood (from healthy, 20- to 35-year-old, non-smoking donors who had not taken any drug for at least 15 days prior to the sampling) was collected, and peripheral blood mononuclear cells (PBMC) were isolated using a standard protocol, with Ficoll (Ficoll-Paque Plus, GE Healthcare Bio-Sciences AB, Sweden) density gradient centrifugation. PBMC were washed and resuspended at a concentration of 0.3 × 106 cells/mL in RPMI 1640 medium supplemented with 20 % fetal bovine serum, 2 mM L-glutamine, and 50 μg/mL gentamycin at 37 °C with 5 % CO2. In addition, concanavalin A (ConA, Sigma Chemical Co. St Louis, MO, USA) was used as a mitogen to trigger cell division in T-lymphocytes. ConA (10 μg/mL) was added at the beginning of the culture, and after 24 h, the cells were treated with the test drugs.
For all experiments, cell viability was examined using Trypan blue exclusion assays. Over 90 % of the cells were viable at the beginning of the culture.
Cytotoxicity assay
Cell viability was quantified using alamar blue assay, as previously described [8]. For all experiments, cells were seeded in 96-well plates (0.7 × 105 cells/mL for adherent cells or 0.3 × 106 cells/mL for suspended cells in 100 μL of medium). After 24 h (or immediately for the suspended cells), the samples, which were dissolved in dimethyl sulfoxide (DMSO, Sigma-Aldrich, St Louis, MO, USA) at a final concentration of 50 μg/mL, were added to each well and incubated for 72 h. Doxorubicin (purity >95 %, Laboratórios IMA S.A.I.C., Buenos Aires, Argentina) was used as the positive control. The negative control received the vehicle used to dilute the tested samples (0.5 % DMSO). Four (for cell lines) or 24 h (for PBMC) before the end of the incubation, 20 μL of stock solution (0.312 mg/mL) of the alamar blue (resazurin, Sigma-Aldrich, St Louis, MO, USA) were added to each well. The absorbance was measured using a SpectraMax 190 microplate reader (Molecular Devices, Sunnyvale, USA), and the drug effect was quantified as the percentage of control absorbance at 570 and 600 nm. The extracts that caused more than 75 % cell growth inhibition were tested again at concentrations varying from 0.39 to 50 μg/mL to determine the 50 % inhibitory concentration (IC50). Isolated compounds were tested at concentrations varying from 0.19 to 25 μg/mL.
Statistical analysis
Data are presented as the mean ± S.E.M. The IC50 values were obtained through nonlinear regression using the GraphPad program (Intuitive Software for Science, San Diego, USA).
Results and discussion
Fifty-five extracts were obtained from the plants studied, and their cytotoxicities were tested using the alamar blue assay after 72 h of incubation. The cytotoxicity of each extract was initially tested against two tumor cell lines (HepG2 and HL-60) at a final concentration of 50 μg/mL. The plant species and plant parts/solvent used for extract preparation are shown in Table 2. Concentration-response curves were generated and IC50 values were calculated against four tumor cell lines (B16-F10, HepG2, K562 and HL-60) and against one non-tumor cells (PBMC) for the extracts that caused more than 75 % cell growth inhibition (Table 3). In addition, the chloroform soluble fractions of D. repens flowers and the hexane extract I. coccinea flowers isolated quercetin and the mixture of α- and β-amyrin (Fig. 1), respectively, which were also tested for their cytotoxic activity (Table 3).
Table 2.
Growth inhibitory effects of plant extracts/fractions against tumor cell lines
| No. | Plant species | Parts of the planta (Extractionsb) | Cellc growth inhibition percentage (GI %)d | |
|---|---|---|---|---|
| HepG2 | HL60 | |||
| 01 | Byrsonima sericea DC. | F (e) | 25.03 ± 5.24 | 25.27 ± 8.79 |
| 02 | S (e) | 8.02 ± 6.85 | 27.32 ± 0.06 | |
| 03 | L (e) | 21.84 ± 2.64 | 64.71 ± 5.28 | |
| 04 | L (m/e) | 91.16 ± 0.64 | 102.7 ± 1.12 | |
| 05 | L (ea/e) | 92.14 ± 0.64 | 103.3 ± 7.26 | |
| 06 | L (c/e) | 39.96 ± 9.49 | 70.12 ± 11.24 | |
| 07 | L (h/e) | 91.62 ± 0.39 | 109.0 ± 2.52 | |
| 08 | Cupania impressinervia Acev. Rodr. var. (revoluta) Radlk | L (e) | 23.10 ± 5.59 | 55.19 ± 5.50 |
| 09 | Duranta repens Linn. | FL (m/e) | 14.09 ± 7.14 | 33.3 ± 0.92 |
| 10 | FL (ea/e) | 20.57 ± 7.67 | 28.36 ± 0.46 | |
| 11 | FL (c/e) | 79.94 ± 2.97 | 98.67 ± 6.35 | |
| 12 | Helicostylis tomentosa (Poepp. & Endl) Rusby | S (e) | 14.55 ± 3.30 | 53.67 ± 0,58 |
| 13 | L (e) | 7.96 ± 3.56 | 38.39 ± 7.33 | |
| 14 | Himatanthus bracteatus (A.DC.) Woodson | S (e) | 20.55 ± 1.61 | 29.38 ± 1.63 |
| 15 | L (e) | −9.90 ± 6.56 | −16.91 ± 4.61 | |
| 16 | L (h) | 14.78 ± 5.22 | 70.63 ± 2.62 | |
| 17 | L (m/e) | −0.77 ± 2.61 | 28.55 ± 3.25 | |
| 18 | L (ea/e) | 5.70 ± 2.61 | 56.16 ± 9.49 | |
| 19 | L (c/e) | 92.30 ± 1.50 | 93.63 ± 0.30 | |
| 20 | Ipomoea purga (Wender.) Hayne | S (e) | 98.39 ± 1.81 | 95.58 ± 1.33 |
| 21 | L (e) | 93.86 ± 2.70 | 106.4 ± 3.62 | |
| 22 | Ixora coccinea Linn. | F (e) | −2.97 ± 9.22 | 1.27 ± 3.86 |
| 23 | FL (h) | 21.47 ± 5.44 | −25.64 ± 9.33 | |
| 24 | L (m/e) | 9.25 ± 4.73 | 1.38 ± 1.35 | |
| 25 | L (ea/e) | 7.49 ± 5.16 | 32.25 ± 9.39 | |
| 26 | L (h/e) | 22.55 ± 3.23 | 58.78 ± 1.34 | |
| 27 | F (ea/e) | −5.53 ± 7.07 | 18.83 ± 5.74 | |
| 28 | F (c/e) | 94.93 ± 0.74 | 92.97 ± 2.39 | |
| 29 | Mabea piriri Aubl. | S (e) | 81.41 ± 8.27 | 92.35 ± 7.66 |
| 30 | L (e) | 12.48 ± 5.12 | 69.56 ± 0.17 | |
| 31 | Miconia minutiflora | S (e) | 25.64 ± 6.34 | 38.17 ± 1.16 |
| 32 | L (e) | 38.62 ± 2.72 | 66.54 ± 0.85 | |
| 33 | Momordica charantia L. | L (e) | −17.51 ± 2.97 | 9.30 ± 5.64 |
| 34 | Ocotea glomerata (Nees) Mez | S (e) | 49.49 ± 9.87 | 32.70 ± 9.32 |
| 35 | L (c/e) | 25.48 ± 7.87 | 52.0 ± 3.12 | |
| 36 | Ocotea longifolia Kunth (Oreodaphne opifera Mart. Nees) | S (e) | −7.43 ± 5.88 | 2.49 ± 0.33 |
| 37 | L (e) | 93.88 ± 2.12 | 94.70 ± 1.06 | |
| 38 | Pavonia fruticosa (Mill.) Fawc. & Rendle | S (e) | 44.81 ± 1.66 | 41.65 ± 6.51 |
| 39 | L (e) | 33.34 ± 4.48 | 55.82 ± 4.66 | |
| 40 | Psychotria capitata Ruiz & Pav. | S (e) | 15.82 ± 3.12 | 64.16 ± 0.32 |
| 41 | L (e) | 28.13 ± 8.45 | 22.00 ± 5.92 | |
| 42 | L (m/e) | 19.16 ± 8.90 | 51.72 ± 4.86 | |
| 43 | L (ea/e) | 95.14 ± 1.83 | 86.55 ± 4.20 | |
| 44 | L (c/e) | 96.92 ± 1.68 | 92.87 ± 0.06 | |
| 45 | L (h/e) | 12.28 ± 4.71 | −26.37 ± 1.89 | |
| 46 | L (h:ea/e) | 61.84 ± 9.74 | 48.62 ± 1.18 | |
| 47 | Schefflera morototoni (Aubl.) Maguire, Steyerm. & Frodin | S (e) | 42.73 ± 1.59 | −23.89 ± 3.53 |
| 48 | L (e) | 9.05 ± 7.07 | 63.74 ± 8.13 | |
| 49 | Solanum paludosum Moric. | S (e) | 17.69 ± 5.22 | 63.81 ± 2.08 |
| 50 | Xylopia frutescens Aubl. | S (e) | 19.36 ± 3.74 | 72.39 ± 2.90 |
| 51 | L (m/e) | 29.88 ± 2.90 | 18.19 ± 6.52 | |
| 52 | L (ea/e) | 19.46 ± 7.46 | 51.24 ± 5.86 | |
| 53 | L (c/e) | 71.00 ± 3.06 | 62.71 ± 4.30 | |
| 54 | L (h/e) | 23.49 ± 3.57 | 69.41 ± 0.88 | |
| 55 | Zanthoxylum rhoifolium Lam. | L (e) | 15.53 ± 5.03 | 14.00 ± 4.00 |
| Doxorubicine | 85.20 ± 0.78 | 86.34 ± 2.27 | ||
aParts of the plant: L leaves, S stem, F Fruits, FL flowers
bExtractions: h hexane, m methanol, e ethanol, m/e methanol/water partition from the ethanolic extract, ea/e ethyl acetate partition from the ethanolic extract, h/e hexane partition from the ethanolic extract, c/e chloroform partition from the ethanolic extract, h:ea/e, hexane:ethyl acetate (1:1) partition from the ethanolic extract
cCell lines: HepG2 (human hepatocellular carcinoma) and HL60 (human promyelocytic leukemia)
dGI % values are presented as the mean ± S.E.M. of two independents experiment performed in triplicate measured by the alamar blue assay after 72 h incubation. All extracts were tested at a concentration of 50 μg/mL. The negative control received the vehicle used to dilute the tested samples (0.5 % DMSO)
eDoxorubicin was used as the positive control
Table 3.
IC50 values of plant extracts/fractions and isolated compounds against tumor cell lines
| No. | Plant species | Parts of planta (Extractionsb) | Cellsc (IC50 in μg/mL)d | ||||
|---|---|---|---|---|---|---|---|
| B16-F10 | HepG2 | HL60 | K562 | PBMC | |||
| 04 | Byrsonima sericea DC. | L (m/e) | 13.46 | 27.95 | 32.56 | >50 | 33.81 |
| 10.47–17.30 | 20.79–37.60 | 25.44–41.66 | 26.45–43.23 | ||||
| 05 | L (ea/e) | 17.44 | 41.53 | 28.21 | >50 | >50 | |
| 13.95–21.81 | 35.31–48.85 | 21.18–37.57 | |||||
| 07 | L (h/e) | 16.89 | 26.19 | 16.14 | 28.32 | 17.58 | |
| 14.63–19.49 | 23.14–29.64 | 13.85–18.82 | 16.96–47.29 | 10.01–30.86 | |||
| 11 | Duranta repens Linn. | L (c/e) | 29.00 | 33.78 | 22.41 | 21.12 | >50 |
| 24.51–34.30 | 29.44–38.75 | 14.10–35.62 | 17.77–25.11 | ||||
| 56 | Quercetin | - | 12.12 | 12.15 | 8.81 | 14.93 | 14.39 |
| 9.92–14.81 | 9.82–15.05 | 7.28–10.65 | 13.90–16.04 | 11.18–18.52 | |||
| 19 | Himatanthus bracteatus (A.DC.) Woodson | L (c/e) | 22.43 | 24.75 | 23.06 | 29.85 | 12.53 |
| 20.80–24.18 | 21.70–28.22 | 20.62–25.79 | 26.87–33.15 | 8.28–18.96 | |||
| 20 | Ipomoea purga (Wender.) Hayne | S (e) | 7.39 | 8.52 | 9.39 | 8.53 | 6.99 |
| 6.03–9.07 | 7.51–9.65 | 7.81–11.30 | 7.61–9.56 | 4.43–11.02 | |||
| 21 | L (e) | 16.30 | 29.63 | 32.64 | 28.95 | 30.71 | |
| 11.80–22.51 | 25.59–34.31 | 28.15–37.85 | 24.39–34.36 | 24.04–39.24 | |||
| 28 | Ixora coccinea Linn. | F (c/e) | 40.41 | 38.25 | 36.92 | >50 | >50 |
| 36.60–44.63 | 30.46–48.04 | 33.52–40.67 | |||||
| 57 | Mixture of α- and β-amyrin | - | 23.21 | 24.09 | >25 | >25 | Nd |
| 18.51–29.11 | 18.71–31.01 | ||||||
| 29 | Mabea piriri Aubl. | S (e) | 17.76 | 32.98 | 22.72 | 21.89 | >50 |
| 13.42–23.51 | 27.61–39.40 | 13.26–38.91 | 15.52–30.87 | ||||
| 37 | Ocotea longifolia Kunth (Oreodaphne opifera Mart. Nees) | L (e) | 37.07 | 37.34 | 31.00 | 43.34 | 21.06 |
| 32.10–42.81 | 31.34–44.49 | 21.04–45.69 | 39.47–47.58 | 17.48–25.37 | |||
| 43 | Psychotria capitata Ruiz & Pav. | L (ea/e) | 15.58 | 18.32 | 18.91 | 31.38 | 12.73 |
| 11.29–21.51 | 14.91–22.50 | 13.93–25.67 | 22.78–43.22 | 7.43–21.81 | |||
| 44 | L (c/e) | 27.06 | 37.06 | 36.06 | 16.45 | 29.14 | |
| 19.26–38.02 | 31.54–43.53 | 28.65–45.40 | 13.16–20.56 | 22.46–37.82 | |||
| Doxorubicine | 0.08 | 0.03 | 0.02 | 0.06 | 0.14 | ||
| 0.06–0.11 | 0.02–0.03 | 0.02–0.02 | 0.04–0.09 | 0.02–1.08 | |||
Nd Not determined
aParts of the plant: L leaves, S stem, and F Fruits
bExtractions: h hexane, m methanol, e ethanol, m/e methanol/H2O partition from the ethanolic extract, ea/e ethyl acetate partition from the ethanolic extract, h/e hexane partition from the ethanolic extract, c/e chloroform partition from the ethanolic extract, h:ea/e hexane:ethyl acetate (1:1) partition from the ethanolic extract
cTumor cells: B16-F10 (mouse melanoma), HepG2 (human hepatocellular carcinoma), HL-60 (human promyelocytic leukemia) and K562 (human chronic myelocytic leukemia). Non-tumor cells: PBMC (human peripheral blood mononuclear cells activated with concanavalin A – human lymphoblast)
dData are presented as IC50 values in μg/mL and their 95 % confidence interval obtained by nonlinear regression from three independent experiments performed in duplicate, measured using alamar blue assay after 72 h incubation. The negative control received the vehicle used to dilute the tested samples (0.5 % DMSO)
eDoxorubicin was used as the positive control
Fig 1.
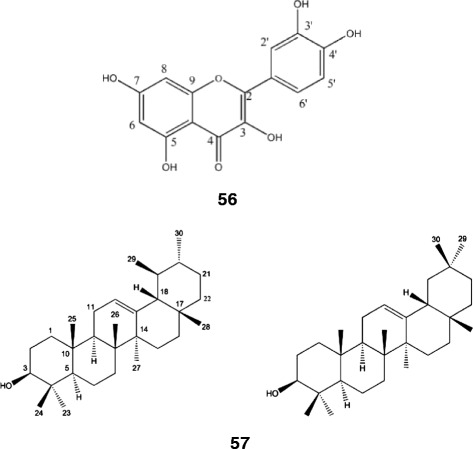
The chemical structures of quercetin (56) isolated from the chloroform soluble fractions of Duranta repens flowers and α- and β-amyrin (57) isolated from the hexane extract of Ixora coccinea flowers
In the preclinical cytotoxic drug screening program used in this work, which is based in the United States National Cancer Institute program, only those extracts that present IC50 values below 30 μg/mL and pure compounds with IC50 values below 4 μg/mL in tumor cell line assays are considered to be promising for anticancer drug development [9–11]. Therefore, the B. sericea, D. repens, H. bracteatus, I. purga, I. coccinea, M. piriri, O. longifolia and P. capitata extracts demonstrated promising cytotoxic activity. Doxorubicin, a clinical useful chemotherapy drug, was used as the positive control and presented IC50 values ranged from 0.02 to 0.08 μg/mL for HL-60 and B16-F10, respectively.
The pharmacology activity of B. sericea has been previously reported; the ethanolic extract from its leaves has gastroprotective properties that are mediated by nitric oxide and the K+ ATP channel. Phytochemical studies have revealed the presence of flavonoids (i.e., rutin, isoquercitrin, kaempferol 3-O-rutinoside and quercetin). However, its cytotoxic activity has not been previously reported [12]. Previous studies of D. repens have reported the presence of triterpenes, flavonoids, steroids, C-alkylated flavonoids and acetosides, as well as some alkaloids. Interestingly, durantanin IV and V and E/Z acteoside isolated from the leaves of D. repens have demonstrated significant cytotoxic activity against a HepG2 cell line [13]. Jalapinoside, a macrocyclic bisdesmoside resin glycoside, was recently isolated from the roots of I. purga, and its cytotoxic activity was evaluated. Jalapinoside was not cytotoxic (IC50 > 10 μg/mL), but reversal of multidrug resistance was observed using vinblastine-resistant human breast carcinoma cells [14]. In this study, we isolated the flavonol quercetin from the flowers of D. repens, which demonstrated a cytotoxic effect (with IC50 values ranging from 8.81 to 14.93 μg/mL on tumor cell lines HL-60 and K562, respectively). Quercetin has been previously reported as a cytotoxic agent because of its ability to induce apoptosis [15, 16].
The antitumor activity of flowers of I. coccinea was previously studied in murine models. Inhibited tumor growth and increased life spans were observed in Dalton’s lymphoma and Ehrlich ascites carcinoma-bearing mice [17]. Moreover, ixorapeptide I, a peptide derived from I. coccinea, displayed cytotoxicity against the Hep3B liver tumor cell line, with an IC50 value of 3.36 μg/mL [18]. Interestedly, although the use of I. coccinea is currently only ornamental in Brazil, its flowers are an ingredient in an Ayurvedic cancer formulation in India [19, 20]. The flowers of I. coccinea are used to treat cancer, leucorrhoea, dysentery, dysmenorrhea, hemoptysis and hypertension; its leaves are used to pacify vitiated pitta, skin diseases, colic, flatulence, diarrhea, indigestion, ulcers and wounds, and they are also used as an antiseptic [20, 21]. The roots of I. coccinea are used as an astringent and antiseptic against scabies and other skin diseases [20]. In this study, the fractionation of the extract of I. coccinea flowers resulted in the mixture of α- and β-amyrin, which demonstrated weak cytotoxic activity toward tumor cell lines. In fact, no potent cytotoxic activity has been reported for these compounds.
Prieto et al. [22] have reported that essential oil from the leaves of O. longifolia, of which α-terpinolene and α-phellandrene are the primary ingredients, demonstrates antifungal activity against Sitophilus zeamais. However, the cytotoxic activity of the O. longifolia plant has not been previously reported. No previous biological effects have been reported for the medicinal plants H. bracteatus, M. piriri and P. capitata.
The extracts from C. impressinervia, H. tomentosa, M. minutiflora, M. charantia, O. glomerata, P. fruticosa, S. morototoni, S. paludosum, X. frutescens and Z. rhoifolium showed no pronounced cytotoxic effects. Among these plants, essential oil from the leaves of X. frutescens has been previously assessed for its cytotoxic and antitumor effects; the oil demonstrated potent in vitro cytotoxic activity against tumor cell lines from different histotypes and in vivo antitumor activity in sarcoma 180 murine model [23]. Although essential oil from Z. rhoifolium leaves and M. charantia fruit extract have been previously assessed for cytotoxicity, they demonstrated no cytotoxic effects [24, 25]. The cytotoxic potential of C. impressinervia, H. tomentosa, M. minutiflora, O. glomerata, P. fruticose, S. morototoni and S. paludosum plants has never been investigated.
Conclusion
In conclusion, we screened the cytotoxic potential of 55 extracts from 18 plants found in northeast Brazil, and extracts from B. sericea, D. repens, H. bracteatus, I. purga, I. coccinea, M. piriri, O. longifolia and P. capitata demonstrated potent cytotoxic effects. The fractionation of D. repens and I. coccinea extracts led to the isolation of quercetin and the mixture of α- and β-amyrin, respectively, and quercetin showed moderate cytotoxic activity. Fractionation of the other plants should be performed to identify their active constituents, and further investigations are required to determine their mechanisms of cytotoxicity and in vivo activities. This work reinforces the need to understand the therapeutic potentialities of Brazilian medicinal plants.
Abbreviations
CC, column chromatography; ConA, concanavalin A; DMSO, dimethyl sulfoxide; IC50, 50 % inhibitory concentration; NMR, nuclear magnetic resonance; PBMC, peripheral blood mononuclear cells; RPMI-1640, roswell park memorial institute-1640; TLC, thin layer chromatography; TMS, tetramethylsilane.
Acknowledgments
The authors are grateful to Edileuza Soares Passos for providing technical assistance and to Rosângela Pereira e Lyra Lemos of the IMA-AL for providing the botanical identifications.
Funding
This work was financially supported by the following Brazilian agencies: CAPES (Coordenação de Aperfeiçoamento de Pessoal de Nível Superior), CNPq (Conselho Nacional de Desenvolvimento Cientifico e Tecnológico) and FAPESB (Fundação de Amparo à Pesquisa e à Inovação Tecnológica do Estado da Bahia).
Availability of data and materials
We have presented our primary data in the form of figure and tables. The datasets supporting the conclusions of this article are included within the article.
Authors’ contributions
Conceived and designed the experiments: TBCS, MBPS and DPB. Performed the experiments: CODC, AFCG, LMB, ACBCR, MCSM, AAD and TRS. Analyzed the data: TBCS and DPB. Contributed the reagents/materials/analysis tools: TBCS, MBPS and DPB. Wrote the paper: TBCS, MBPS and DPB. All authors read and approved the final manuscript for submission.
Competing interests
The authors declare that they have no competing interests.
Consent for publication
All study participants provided informed consent for the data publication.
Ethics approval and consent to participate
For the PBMC collection, the Research Ethics Committee of the Oswaldo Cruz Foundation (Salvador, Bahia, Brazil) approved the experimental protocol (number 031019/2013). All participants provided written informed consent to participate in the study.
References
- 1.Lewinsohn TL, Prado PI. Quantas espécies há no Brasil? Megadiversidade. 2005;1(1):36–42. [Google Scholar]
- 2.Melo JG, Santos AG, Amorim ELC, Nascimento SC, Albuquerque UP. Medicinal plants used as antitumor agents in Brazil: an ethnobotanical approach. Evid Based Complement Alternat Med. 2011;2011:365359. doi: 10.1155/2011/365359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Forzza RC, Baumgratz JFA, Bicudo CEM, Canhos DAL, Carvalho AA, Jr, Coelho MAN, Costa AF, Costa DP, Hopkins MG, Leitman PM, Lohmann LG, Lughadha EN, Maia LC, Martinelli G, Menezes M, Morim MP, Peixoto AL, Pirani JR, Prado J, Queiroz LP, Souza S, Souza VC, Stehmann JR, Sylvestre LS, Walter BMT, Zappi DC. New brazilian floristic list highlights conservation challenges. Bioscience. 2012;62(1):39–45. doi: 10.1525/bio.2012.62.1.8. [DOI] [Google Scholar]
- 4.Mesquita ML, de Paula JE, Pessoa C, de Moraes MO, Costa-Lotufo LV, Grougnet R, Michel S, Tillequin F, Espindola LS. Cytotoxic activity of Brazilian Cerrado plants used in traditional medicine against cancer cell lines. J Ethnopharmacol. 2009;123(3):439–45. doi: 10.1016/j.jep.2009.03.018. [DOI] [PubMed] [Google Scholar]
- 5.Ozi JM, Suffredini IB, Paciencia M, Frana SA, Dib LL. In vitro cytotoxic effects of Brazilian plant extracts on squamous cell carcinoma of the oral cavity. Braz Oral Res. 2011;25(6):519–25. doi: 10.1590/S1806-83242011000600008. [DOI] [PubMed] [Google Scholar]
- 6.Ferreira PM, Farias DF, Viana MP, Souza TM, Vasconcelos IM, Soares BM, Pessoa C, Costa-Lotufo LV, Moraes MO, Carvalho AF. Study of the antiproliferative potential of seed extracts from Northeastern Brazilian plants. An Acad Bras Cienc. 2011;83(3):1045–58. doi: 10.1590/S0001-37652011005000017. [DOI] [PubMed] [Google Scholar]
- 7.Ribeiro SS, de Jesus AM, dos Anjos CS, da Silva TB, Santos AD, de Jesus JR, Andrade MS, Sampaio TS, Gomes WF, Alves PB, Carvalho AA, Pessoa C, de Moraes MO, Pinheiro ML, Prata AP, Blank AF, Silva-Mann R, Moraes VR, Costa EV, Nogueira PC, Bezerra DP. Evaluation of the cytotoxic activity of some Brazilian medicinal plants. Planta Med. 2012;78(14):1601–6. doi: 10.1055/s-0032-1315043. [DOI] [PubMed] [Google Scholar]
- 8.Ahmed SA, Gogal RM, Walsh JE. A new rapid and simple non-radioactive assay to monitor and determine the proliferation of lymphocytes an alternative to [3H] thymidine incorporation assay. J Immunol Methods. 1994;170(2):211–24. doi: 10.1016/0022-1759(94)90396-4. [DOI] [PubMed] [Google Scholar]
- 9.Suffness M, Pezzuto JM. Assays related to cancer drug discovery. In: Hostettmann K, editor. Methods in plant biochemistry: assays for bioactivity. London: Academic Press; 1990. pp. 71–133. [Google Scholar]
- 10.Quintans JSS, Soares BM, Ferraz RPC, Oliveira ACA, Silva TB, Menezes LRA, Sampaio MFC, Prata APN, Moraes MO, Pessoa C, Antoniolli AR, Costa EV, Bezerra DP. Chemical constituents and anticancer effects of the essential oil from leaves of Xylopia laevigata. Planta Med. 2013;79(2):123–30. doi: 10.1055/s-0032-1328091. [DOI] [PubMed] [Google Scholar]
- 11.Rodrigues ACBC, Bomfim LM, Neves SP, Menezes LRA, Dias RB, Soares MBP, Prata APN, Rocha CAG, Costa EV, Bezerra DP. Antitumor properties of the essential oil from the leaves of Duguetia gardneriana. Planta Med. 2015;81(10):798–803. doi: 10.1055/s-0035-1546130. [DOI] [PubMed] [Google Scholar]
- 12.Rodrigues PA, Morais SM, Souza CM, Magalhaes DV, Vieira IGP, Andrade GM, Rao VS, Santos FA. Gastroprotective effect of Byrsonima sericea DC leaf extract against ethanol-induced gastric injury and its possible mechanisms of action. An Acad Bras Cienc. 2012;84(1):113–22. doi: 10.1590/S0001-37652012005000011. [DOI] [PubMed] [Google Scholar]
- 13.Ahmed WS, Mohamed MA, El-Dib RA, Hamed MM. New triterpene saponins from Duranta repens Linn. and their cytotoxic activity. Molecules. 2009;14(5):1952–65. doi: 10.3390/molecules14051952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Bautista E, Fragoso-Serrano M, Pereda-Miranda R. Jalapinoside, a macrocyclic bisdesmoside from the resin glycosides of Ipomea purga, as a modulator of multidrug resistance in human cancer cells. J Nat Prod. 2015;78(1):168–72. doi: 10.1021/np500762w. [DOI] [PubMed] [Google Scholar]
- 15.Siegelin MD, Reuss DE, Habel A, Rami A, von Deimling A. Quercetin promotes degradation of survivin and thereby enhances death-receptor-mediated apoptosis in glioma cells. Neuro Oncol. 2009;11(2):122–31. doi: 10.1215/15228517-2008-085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Jakubowicz-Gil J, Langner E, Bądziul D, Wertel I, Rzeski W. Apoptosis induction in human glioblastoma multiforme T98G cells upon temozolomide and quercetin treatment. Tumour Biol. 2013;34(4):2367–78. doi: 10.1007/s13277-013-0785-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Latha PG, Panikkar KR. Cytotoxic and antitumor principles from Ixora coccinea flowers. Cancer Lett. 1998;130(1-2):197–202. doi: 10.1016/S0304-3835(98)00140-2. [DOI] [PubMed] [Google Scholar]
- 18.Lee CL, Liao YC, Hwang TL, Wu CC, Chang FR, Wu YC. Ixorapeptide I and ixorapeptide II, bioactive peptides isolated from Ixora coccinea. Bioorg Med Chem Lett. 2010;20(24):7354–7. doi: 10.1016/j.bmcl.2010.10.058. [DOI] [PubMed] [Google Scholar]
- 19.Panikkar KR, Bhanumathy P, Raghunath PN. Antitumour activity of an ayurvedic oil preparation. Ancient Sci Life. 1986;6(2):107–8. [PMC free article] [PubMed] [Google Scholar]
- 20.Kharat AR, Nambiar VV, Tarkasband YS, Pujari RR. A review on phytochemical and pharmacological activity of genus Ixora. IJRPC. 2013;3(3):628–35. [Google Scholar]
- 21.John D. One hundred useful raw drugs of Kani tribes of trivandrum forest division, Kerala, India. Int J Crude Drug Res. 1984;22(1):17–39. doi: 10.3109/13880208409070646. [DOI] [Google Scholar]
- 22.Prieto JA, Pabon LC, Patino OJ, Delgado WA, Cuca LE. Chemical constituents and insecticidal and antifungal activities of the essential oils of leaves of two Colombian species of the genus Ocotea (Lauraceae) Rev Colomb Quim. 2010;39(2):199–209. [Google Scholar]
- 23.Ferraz RP, Cardoso GM, da Silva TB, Fontes JE, Prata AP, Carvalho AA, Moraes MO, Pessoa C, Costa EV, Bezerra DP. Antitumour properties of the leaf essential oil of Xylopia frutescens Aubl. (Annonaceae) Food Chem. 2013;141(1):196–200. doi: 10.1016/j.foodchem.2013.02.114. [DOI] [PubMed] [Google Scholar]
- 24.Masuda T, Oyama Y, Yamamoto N, Umebayashi C, Nakao H, Toi Y, Takeda Y, Nakamoto K, Kuninaga H, Nishizato Y, Nonaka A. Cytotoxic screening of medicinal and edible plants in Okinawa, Japan, and identification of the main toxicconstituent of Rhodea japonica (Omoto) Biosci Biotechnol Biochem. 2003;67(6):1401–4. doi: 10.1271/bbb.67.1401. [DOI] [PubMed] [Google Scholar]
- 25.Boehme AK, Noletto JA, Haber WA, Setzer WN. Bioactivity and chemical composition of the leaf essential oils of Zanthoxylum rhoifolium and Zanthoxylum setulosum from Monteverde, Costa Rica. Nat Prod Res. 2008;22(1):31–6. doi: 10.1080/14786410601130224. [DOI] [PubMed] [Google Scholar]
- 26.Agra MF, Freitas PF, Barbosa-Filho JM. Synopsis of the plants known as medicinal and poisonous in Northeast of Brazil. Braz J Pharmacog. 2007;17(1):114–40. [Google Scholar]
- 27.Amaral GC, Brito LPS, Avelino RC, Silva Júnior JV, Beckmann-Cavalcante MZ, Cavalcante IHL. Seedlings production of Duranta repens l. using cuttings process. Rev Ciênc Agrár. 2012;35(1):134–42. [Google Scholar]
- 28.Costa LCB, Rocha EA, Silva LAM, Jardim JG, Silva DC, Gaião LO, Moreira RCT. Levantamento preliminar das espécies vegetais com potencial econômico no parque municipal da Boa Esperança, Ilhéus, Bahia, Brasil. Acta Farm Bonaerense. 2006;25(2):184–91. [Google Scholar]
- 29.Madaleno IM. Popular medicinal plants from São Luis, Brazil. Bol Mus Para Emílio Goeldi Cienc Hum. 2011;6(2):273–86. doi: 10.1590/S1981-81222011000200002. [DOI] [Google Scholar]
- 30.Siviero A, Delunardo TA, Haverroth M, Oliveira LC, Roman ALC, Mendonça AMS. Ornamental plants in urban homegardens of Rio Branco, Brazil. Bol Mus Para Emílio Goeldi Cienc Hum. 2014;9(3):797–813. doi: 10.1590/1981-81222014000300015. [DOI] [Google Scholar]
- 31.Martins JEC. Plantas medicinais de uso na amazônia. 2. São Paulo: Graficentro-CEJUP; 1989. [Google Scholar]
- 32.De Paula JE, Alves JLH. Madeiras naturais: anatomia, dendrologia, dendrometria, produção e uso. Brasília: Fundação Mokiti Okada-MOA; 1997. [Google Scholar]
- 33.Breitbach UB, Niehues M, Lopes NP, Faria JEQ, Brandão MGL. Amazonian Brazilian medicinal plants described by C.F.P. von Martius in the 19th century. J Ethnopharmacol. 2013;147(1):180–9. doi: 10.1016/j.jep.2013.02.030. [DOI] [PubMed] [Google Scholar]
- 34.Cruz GL. Dicionário de plantas úteis do Brasil. In: Bertrand Brasil. 5th ed. Rio de janeiro; 1995. p. 436
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
We have presented our primary data in the form of figure and tables. The datasets supporting the conclusions of this article are included within the article.


