Abstract
Background
Hypersensitivity reactions (HSR) during the perioperative period are unpredictable and can be life threatening. Prospective studies for evaluation of perioperative HSR are lacking and data on causative agents varies between different studies.
Objective
To prospectively determine the success of a comprehensive allergy evaluation plan for patients with HSR during anesthesia, including identification of causative agent and outcomes during subsequent anesthesia exposure.
Methods
All patients referred for perioperative HSR between November 2013 and March 2015, from a Boston teaching hospital, were evaluated using a standardized protocol with skin testing (ST) within 6 months of HSR. Comprehensive allergy evaluation included collection of patient information, including characteristics of HSR during anesthesia. We reviewed results of ST and/or test doses for all potential causative medications Event-related tryptase levels were reviewed when available.
Results
Over 17 months, 25 patients completed the comprehensive allergy evaluation. Fifty-two percent (13/25) were female with a median age of 52 (IQR 43–66) years. The most frequently observed HSR systems were cutaneous (68%), cardiovascular (64%), and pulmonary (24%). A culprit drug, defined as a positive ST, was identified in 36% (9/25) of patients. The most common agent identified was cefazolin (6/9). Following our comprehensive evaluation and management plan, seven (7/8, 88%) patients tolerated subsequent anesthesia.
Conclusions
Cefazolin was the most commonly identified cause of perioperative HSR in our study population. Skin testing patients within 6 months of a perioperative HSR may improve the odds of finding a positive result. Tolerance of subsequent anesthesia is generally achieved in patients undergoing our comprehensive evaluation.
Keywords: Drug, Allergy, Hypersensitivity, Reaction, Anesthesia, Perioperative, Anaphylaxis, Tryptase, Cefazolin
INTRODUCTION
Perioperative hypersensitivity reactions (HSRs) have been estimated to occur in 1:3,500 to 1:20,000 procedures, with a mortality rate of up to 9%.1 However, newer prospective data suggest this incidence may be as high as 1 in 385 operations.2 Several drugs are often administered simultaneously during general anesthesia, making identification of the causative agent(s) difficult. Identification of the culprit agent of a perioperative HSR is crucial to avoid re-exposure and prevent subsequent untoward outcomes.
Guidelines recommend comprehensive skin testing to chlorhexidine, all preoperative medications listed in the anesthetic record that were given prior to the HSR, and specific IgE testing to latex.3 The literature suggests that there is a geographic difference in the most frequently identified cause of perioperative HSRs. Studies from Europe have implicated neuromuscular blocking agents (NMBAs)4,5, while studies from the United States (US)6–8 more commonly implicate antibiotics as the cause of perioperative HSR.
A recent retrospective study described a 10-year period utilizing a specific evaluation and management plan to identify causative agents and provide recommendations for patients with HSR during anesthesia that require subsequent anesthesia.6 Limitations of this study were the absence of antibiotic skin testing in some patients due to lack of skin testing reagents and the prolonged and variable length of time from perioperative HSR to skin testing, which may have led to a higher rate of false-negative skin testing.9,10 Decreased availablity of beta-lactam testing may explain the low rate of a positive skin test upon evaluation (13 of 73 patients, or 18%) and antibiotics as the causal agent (3 of 13 patients, or 23%).
In the present study, we describe the application of a comprehensive management plan for perioperative HSRs prospectively over a 17-month period, including skin testing to all possible causative agents within 6 months of HSR, and assessing patients’ tolerability of subsequent anesthesia, when indicated.
METHODS
Study Design
We performed a prospective cohort study of all patients at Massachusetts General Hospital who were referred to Allergy/Immunology for perioperative HSR between November 2013 and March 2015. These patients were evaluated using a standardized protocol.6
Patients were identified based on referral information provided by a central patient scheduler. Patients that had a HSR during induction or maintenance of anesthesia, or during the immediate post-operative recovery period, were included.
Patients were excluded if they were under the age of 18 at time of evaluation, declined participation in the comprehensive evaluation, or did not complete the recommended comprehensive evaluation. The study was approved by the Partners Healthcare Institutional Review Board.
Comprehensive Allergy Evaluation
All patients referred to the Allergy/Immunology Unit for a perioperative HSR underwent evaluation involving a detailed review of the HSR obtained from the patient’s perioperative medical records. The review included anesthesia records, nursing records, operative and procedural reports, and consultation notes of the Allergy/Immunology Unit, when applicable. Patient information, including age, sex, atopic history, history of prior drug allergy, medications at the time of the surgery, history of previous surgery, serum specific immunoglobulin E (IgE) to latex, and serum tryptase levels (when available), were recorded. Documentation of perioperative organ system involvement also was recorded and included cutaneous, oropharyngeal, pulmonary, cardiovascular, gastrointestinal, or other system involvement. HSR severity was determined following an established grading scale (I–IV) with a grade of IV representing cardiac and/or respiratory arrest to classify the perioperative HSR (Table I).3
Table I.
Severity of Hypersensitivity Reactions during Anesthesia
| Grade | Symptoms |
|---|---|
| I | Cutaneous signs: urticaria, angioedema, generalized erythema |
| II | Measurable but not life-threatening symptoms: cutaneous signs, hypotension (defined as a decrease of more than 30% in blood pressure with tachycardia), respiratory symptoms (cough, difficulty with mechanical ventilation) |
| III | Life-threatening symptoms: cardiovascular collapse, tachycardia or bradycardia, arrhythmias, severe bronchospasm |
| IV | Cardiac and/or respiratory arrest |
Adapted from Ring J and Messmer K. Lancet 1977 Feb 26; 1(8009):466–9 and Mertes et al. J Investig Allergol Clin Immunol 2011; 21: 442–453
Formal collaboration was established between the Allergy/Immunology Unit and the Department of Anesthesia at our institution to capture cases of perioperative HSR. Initial steps included meetings with the Department of Anesthesia’s Quality Assurance Chair and lead nurse coordinator to discuss our cohort project and development of a referral checklist to ensure the best possible patient outcomes. We subsequently arranged a case based presentation at the Department of Anesthesia’s Mortality and Morbidity conference, to discuss commonly encountered drug allergy issues in the perioperative setting including associated hypersensitivity reactions. We encouraged sending serum total tryptase within 4 hours for any suspicion of a perioperative HSR and outpatient referral to Allergy. To ensure the vigilant capture of all possible or probable perioperative hypersensitivity reactions, we maintained weekly communication with the Anesthesia Department’s Quality Assurance team during the study period.
Skin testing was performed between four weeks and six months of the perioperative HSR to minimize false-negative results.11 Intradermal and/or skin prick testing was performed to all agents received prior to the HSR with published non-irritating skin testing concentrations. Inhalational agents, which have not been demonstrated to cause HSR and to which no standardized skin testing exists, were not skin tested. Skin testing during allergy evaluation was performed with non-irritating skin testing concentrations, and appropriately positive (histamine 6mg/mL) and negative (saline) controls (Table II). A positive skin prick reaction, suggesting an IgE-mediated cause, was considered if the diameter of the wheal was at least 3 mm larger than that induced by the negative control. If the skin prick test was negative, increasing concentrations of intradermal skin testing was performed, when available. A positive intradermal skin test reaction was considered if the diameter of the wheal had an increase of more than 3mm compared to the saline wheal, and had an associated erythematous flare.
Table II.
Skin Testing Reagents
| Medication | Concentration | SPT | ID Dilution |
|---|---|---|---|
| Atracurium30,31 | 10 mg/ml | Undiluted | 1:1000 |
| Benzylpenicilloyl polylysine32 | 6.0X10–5 M | Undiluted | Undiluted |
| Bupivacaine 0.25%31,33,34 | 0.25% | Undiluted | 1:100 |
| Cefazolin27,32 | 330mg/ml | Undiluted | 1:100 1:10 |
| Ceftriaxone27,32 | 100mg/ml | Undiluted | 1:100 1:10 |
| Cefuroxime27,32 | 100mg/ml | Undiluted | 1:100 1:10 |
| Ciprofloxacin35 | 2mg/ml | Undiluted | 1:1000 1:100 |
| Cisatracurium30,31 | 2 mg/ml | Undiluted | 1:1000 1:100 |
| Chlorhexidine Gluconate 0.5%31 | 1.2 mg/ml | Undiluted | 1:2500 |
| Chloroprocaine 1%31,33,34 | 1% | Undiluted | 1:100 |
| Etomidate31 | 2 mg/ml | Undiluted | 1:1000 1:100 1:10 |
| Fentanyl31 | 0.05 mg/ml | Undiluted | 1:10,000 1:1000 1:100 1:10 |
| Ketamine30,31 | 10mg/ml | Undiluted | 1:40 |
| Latex30 | Latex fragments soaked in saline | Undiluted Prick through dry glove Prick through wet glove |
N/A |
| Lidocaine 1%31,33,34 | 1% | Undiluted | 1:100 |
| Midazolam30,31 | 5 mg/ml | Undiluted | 1:20 1:10 |
| NPH insulin36,37 | 100 IU/ml | Undiluted | 1:1000 1:100 |
| Odansetron38,39 | 2 mg/ml | Undiluted | 1:100 |
| Pancuronium30,31 | 2 mg/ml | Undiluted | 1:1000 1:100 1:10 |
| Penicillin G32 | 1,000,0000 U/ml | 1:100 | 1:1000 1:100 |
| Propofol30,31,40 | 10 mg/ml | Undiluted | 1:1000 1:100 1:10 |
| Protamine sulfate41 | 10 mg/ml | 1:10 Undiluted |
1:100 1:10 Undiluted |
| Rocuronium30,31 | 10 mg/ml | Undiluted | 1:1000 1:100 |
| Succinylcholine30 | 20 mg/ml | Undiluted | 1:1000 |
| Tetracaine 1%33,34 | 1% | Undiluted | 1:100 |
| Vancomycin27 | 50 mg/ml | Undiluted | 1:10,000 |
| Vecuronium30,31 | 10 mg/ml | Undiluted | 1:1000 1:100 |
Abbreviations: SPT: Skin Prick Testing, ID: intradermal testing, IU: international units, ml: mililiters, mg: milligrams
Laboratory evaluation of patients including total baseline tryptase level (if perioperative tryptase value during HSR was elevated or absent) and specific IgE testing for latex if there was evidence of perioperative latex exposure. Latex specific IgE testing was performed using ImmunoCAP system (Upjohn-Pharmacia, Uppsala, Sweden) and, if negative, latex skin testing was performed (Table II).
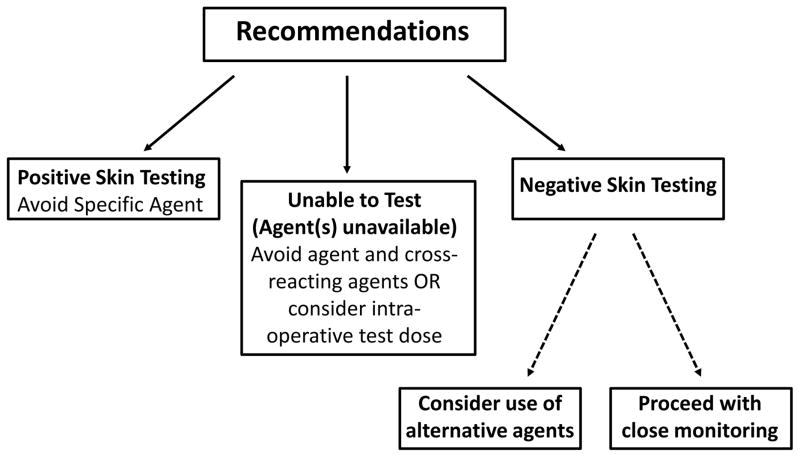
Following this comprehensive allergy evaluation, the allergy team made recommendations for subsequent use of anesthesia. A patient with a positive skin test, suggesting an IgE-mediated HSR, was advised to strictly avoid that specific agent. If a patient had negative skin testing, no specific medication avoidance was recommended. If skin testing was not available, or if skin testing was unable to be performed because of use of antihistamine agents or need for urgent surgery, patients were advised to avoid these medications and specific recommendations were made for safe alternatives (Figure 1). No specific recommendations for the use of pre-medications (i.e. anti-histamines or glucocorticoids) were made. Patient monitoring during subsequent anesthesia for signs and symptoms of a HSR was recommended.
Figure 1.
Recommendations for future anesthesia following evaluation. Recommendations were provided for subsequent procedures using the results of comprehensive allergy evaluation including skin testing to all available agents received immediately prior to the HSR and listed in the anesthetic record.
Follow-up Data Collection
Follow-up information on subsequent procedures was collected via standardized telephone interviewfor all patients that completed the comprehensive assessment. Patients who did not have follow-up information available in the electronic medical record were asked to sign medical record release to obtain their surgical and procedural records. All subsequent perioperative notes and anesthesia records were obtained and reviewed for details of exposure to medications evaluated during comprehensive allergy evaluation and development of HSRs.
RESULTS
Over the 17-month period, 30 patients were referred to the allergy practice with HSR during anesthesia; 25 were included in the analysis. Five patients were excluded due to the following: under the age of 18 at time of evaluation (n=1), declined participation in the comprehensive evaluation (n=2), or did not complete comprehensive allergyevaluation (n=2).
Patient Characteristics
Fifty two percent (n=13) of patients were female, with median age of 52 years (IQR 43–66) at the time of the initial HSR. The median time from HSR to initial allergy evaluation and skin testing was 32 days (IQR 3–89) and 61 days (IQR 49–146), respectively (Table III). At the time of the allergy evaluation 56% (n=14) of patients reported a prior history of drug allergy, 20% (n=5) reported a previous diagnosis of allergic rhinitis, 20% (n=5) reported a diagnosis of asthma, 12% (n=3) reported a history of food allergy, and no patients reported a previous diagnosis of eczema. No patients reported a previous history of allergy to any anesthetic agent.
Table III.
Patient Characteristics
| n = 25 | |
|---|---|
| Age at time of the HSR median (IQR) | 52 (43–66) |
| Sex: female No. (%) | 13 (52) |
| Time from HSR to allergy evaluation (days) median (IQR) | 32 (3–89) |
| Time from HSR to skin testing (days) median (IQR) | 61 (49–146) |
| Patient-reported history of drug allergy | 14 (56) |
| Patient-reported history of atopic disorders | |
| Asthma | 5 (20) |
| Allergic rhinitis | 5 (20) |
| Eczema | 0 (0) |
| Food allergy | 3 (12) |
| Any atopic disorder* | 6 (24) |
| Severity of perioperative HSR | |
| I | 7 (28) |
| II | 10 (40) |
| III | 6 (24) |
| IV | 2 (8) |
| Organ system involvement | |
| Cutaneous | 17 (68) |
| Oropharyngeal | 5 (20) |
| Pulmonary | 6 (24) |
| Cardiovascular | 16 (64) |
| Gastrointestinal | 3 (12) |
| Other | 3 (12) |
Defined as history of asthma, allergic rhinitis, eczema, or food allergy, Values are n (%) unless indicated otherwise
Abbreviations: HSR: hypersensitivity reaction, IQR: interquartile range,
Hypersensitivity Reactions During Anesthesia
Seven patients (28%) had a grade I HSR while 10 (40%) patients had a grade II HSR. Of the six patients with grade III HSR, all had hypotension with or without respiratory symptoms. Of the two patients with grade IV HSR, both had cardiac arrest and neither developed respiratory failure. During HSR, 17 patients (68%) experienced cutaneous signs and symptoms (urticaria, rash, flushing), five (20%) had oropharyngeal or mucosal symptoms (angioedema, throat tightness), six (24%) had pulmonary symptoms (bronchospasm, shortness of breath, wheezing), and 16 (64%) had cardiovascular signs (bradycardia, tachycardia, hypotension, arrhythmia). Serum total tryptase levels were obtained in 11 patients (44%) during their HSR; levels were elevated at greater than 11.4 ng/ml in 8 (73%) of these patients (Table IV). Ten tryptase values were obtained within 4 hours of the onset of the HSR and 8 were elevated (Table IV). One additional patient had a tryptase checked at eight hours, which was not elevated (6 ng/mL).
Table IV.
Perioperative HSR Associated Tryptase Values*
| Subject ID | Sex | Grade of Reaction | Total tryptase (ng/mL)† | Time to Tryptase (min) |
|---|---|---|---|---|
| 1 | M | III | 62 | 30 |
| 2 | M | III | 61 | 80 |
| 7 | M | II | 4 | 55 |
| 10 | M | III | 247 | 80 |
| 12 | F | III | 38 | 45 |
| 17 | F | II | 69 | 45 |
| 19 | M | II | 21 | 102 |
| 24 | F | IV | 182 | 70 |
| 26 | F | II | 25 | 160 |
| 27 | M | II | 3 | 170 |
| MEDIAN (IQR) | 50 (22–67) | 75 (48–97) |
all listed values were obtained within four hours of HSR onset,
tryptase reference range is <11.5 ng/mL.
Abbreviations: ng/mL: nanograms per mililiter, min: minutes, IQR: interquartile range
Comprehensive Allergy Evaluation and Management
Skin testing to agents administered perioperatively or latex specific IgE testing identified a causative agent in 9 (36%) of the 25 patients that completed the comprehensive evaluation. Of the patients with positive skin testing, all 9 (100%) were positive to antibiotics. Of these patients, six had positive skin testing to cefazolin; one patient had positive skin testing to penicillin (PCN) G; one patient had positive skin testing to ciprofloxacin; and one patient had positive skin testing to vancomycin (Table IV). In addition, two patients were found to have a persistently elevated tryptase levels. Both of these patients underwent evaluation for possible mastocytosis, with negative bone marrow biopsy results. There were no positive skin tests to any other agents administered perioperatively.
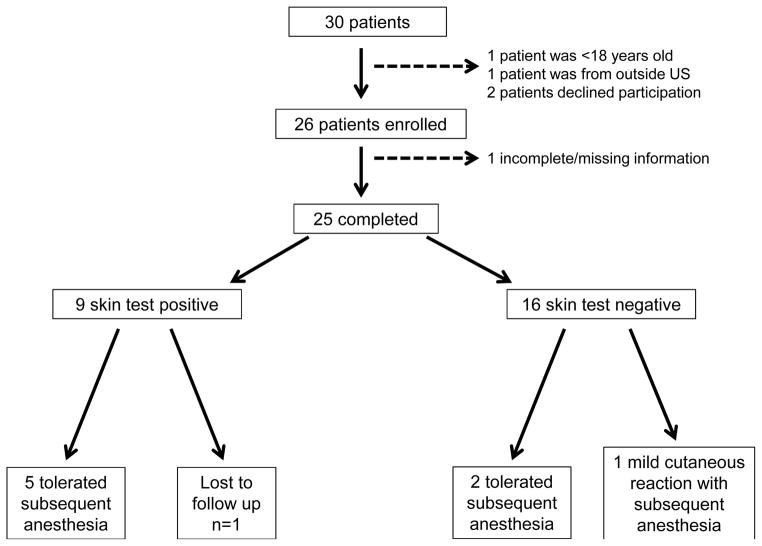
Following our management plan (Figure 2),6 8 patients underwent subsequent anesthesia with 7(88%) tolerating operative procedures regardless of the severity of the initial HSR (Figure 2). Only one of these patients had an elevated baseline tryptase value, but tolerated subsequent anesthesia without incident. Five ( 63%) of the patients had positive skin test results and alternative antibiotic agents were used during subsequent anesthesia without development of perioperative HSR. Premedication or the use of alternative agents-with the exception of antibiotics, was not recommended for patients undergoing subsequent anesthesia. In the remaining three patients with negative skin testing results, one (33%) developed a perioperative HSR after undergoing subsequent anesthesia. The perioperative HSR experienced by this patient was mild cutaneous reaction (erythematous patches only) that spontaneously resolved without intervention. Two (67%) of the patients underwent subsequent anesthesia using the same agents and did not develop a HSR. One (13%) patient underwent subsequent anesthesia without reaction; however, detailed anesthesia reports were not available.
Figure 2.
Outcomes after comprehensive evaluation. Of the 30 patients consecutively referred to our center, 4 were excluded (1 patient <18 years old, 1 patient lived outside US, 2 patients declined participation), 26 patients were enrolled and 25 patients completed our comprehensive evaluation. Of these 25 patients, 8 have undergone subsequent anesthesia and 1 was lost to follow up. All 5 patients with a positive skin test tolerated the subsequent anesthesia. One of three patients that had negative skin testing had a mild reaction with subsequent anesthesia.
DISCUSSION
Twenty-five patients completed a prospective comprehensive evaluation after perioperative HSR and a suspected IgE-mediated cause, with positive skin testing, was found in 36%, higher than 18% in a previously published study.6 We identified antibiotics as the most commonly identified cause of perioperative HSR by skin testing, with cefazolin having the most frequently observed positive skin testing. Tryptase levels, obtained during HSRs, aid in the evaluation of perioperative anaphylaxis. Nearly all patients who completed this evaluation and followed recommendations tolerated subsequent anesthesia, suggesting the value of allergy evaluation and management to both the patient and the anesthesiologist.
Causative agents have varied between studies. Dong et al. in France, evaluated 1,253 patients with HSR during anesthesia, and were able to establish a diagnosis of IgE-mediated HSR in 786 cases (63%) while 467 cases (37%) were considered non-immune-mediated.4 The French investigators found that neuromuscular blocking agents (NMBAs) were the most frequent cause of anaphylaxis (47%), followed by latex (20%), antibiotics (18%), opioids (2%), colloids (2%), hypnotics (1%), and other substances including dyes (9%). Investigators from Spain reported a much higher incidence of antibiotics causing anaphylaxis (44%).12 Protamine, cisatracurium, amoxicillin-clavulanate, atracurium, and dipyrone (in order of most to least likely) were recently found in a prospective cohort from Spain as causes of an IgE mediated perioperative HSR.2 A case series from the United States of 38 patients with perioperative HSR found an IgE-mediated cause in 18 (47%), with 9 of these patients having positive skin testing to an antibiotic.8 Overall, cefazolin (7 patients) was the most common agent identified. In this same series, six of these patients had elevated tryptase levels, but negative skin testing, and a non–IgE-mediated reaction was suspected in the remaining cases. A more recent case series of 30 patients with perioperative HSR from the United States by Gonzalez-Estrada et al. found an IgE mediated cause in 17 (57%). Ten of these patients had positive skin testing to an antibiotic.7 The observed differences between these studies have several possible explanations including population level rates of sensitization and non-standard timing and methods of skin testing.
Similar to the recently published Spanish12 and US6,7 studies, we also found that antibiotics were the most frequently identified cause of perioperative HSR. Compared to our institution’s prior study,6 our higher rate of skin test positivity (36% vs. 18%) may be due to the availability of antibiotic skin testing and the reduced time interval to testing. It is unclear why our rates of skin test positivity are not as high as other recent studies, but may be explained by less severe perioperative HSR in our study compared to others (64% vs. 97% of patients with hypotension) or selection/referral bias present in the retrospective studies compared to our more inclusive cohort study. Cefazolin, a first generation cephalosporin, is the recommended first-line prophylactic agent for most procedures, including hip and knee surgeries, and in combination with metronidazole for colorectal surgery.13 Cefazolin does not share side chains with other cephalosporins or beta-lactam antibiotics. In patients with cefazolin allergy in the perioperative setting, other cephalosporins such as cefoxitin would be tolerated based on its different side chain.14 Data supporting cefazolin tolerance in PCN-allergic patients comes from only retrospective reviews subject that are subject to selection bias. In one review of 300 patients with a documented PCN allergy, only one had an adverse reaction to cefazolin.15 To date, we know of no specific reports of patients tolerance of PCNs given IgE mediated HSR to cefazolin. Previous perioperative HSR evaluation-related studies, including one from our institution,6 may have had a lower rate of antibiotic skin test positivity due to the lack of antibiotic skin testing reagents. Six of the nine patients identified with positive skin testing in our study were positive only to cefazolin. Given the findings of our study, we suggest anesthesiologists and proceduralists follow guidelines3 that recommend antibiotic prophylaxis be administered preoperatively to a monitored, awake patient before anesthesia induction.
Generally, we were able to evaluate patients with perioperative HSR within 1 month after HSR, and skin test patients within 2 months. This likely helped us to find a higher rate of positive skin tests, since previously published data report a decline over time in IgE antibodies in patients with drug allergy.6,9,10,16,17 In 2013 Lafuente et al, found 25 patients (57%) with positive skin testing when performed at 0–4 days (Stage 1) and again at 4–8 weeks (Stage 2) after perioperative HSR.17 Interestingly, a higher rate of skin test positivity was found in the Stage 1 (60%) compared to Stage 2 (40%) with three patients having positive skin testing only in Stage 1. This may be a consequence of false negative results when testing is delayed. In one study, the mean time to evaluation following HSR in skin test positive subjects was 12 months, compared to 36 months in those that were skin test negative.16 Avoidance of false negative skin testing can be achieved if performed at least four weeks after the HSR9,10 Therefore, a patient with HSR during anesthesia should be referred for evaluation, with HSR-specific skin testing completed within six weeks to six months of the reaction.
Our recommendation, after negative skin testing, was no specific medication avoidance. We certainly recognize the concern for false negative skin testing because our testing is not standardized with an established negative predictive value. Concern for false negative testing may be even higher when the timing of skin testing is not optimal. In our study, we identified only three patients with negative skin testing and all of these patients had mild to moderate HSR. If a patient experiences a severe HSR and has negative skin testing, there are no data to inform our best practices. It may be appropriate to suggest use of alternative agents in these cases. Further data are needed to optimize recommendations after negative skin testing.
Elevated or increased perioperative tryptase levels are seen in IgE and non-IgE mediated perioperative HSR. Similar to a recent study7 that found elevated tryptase levels in 100% (10/10) of cases of IgE-mediated anaphylaxis, our study showed 67% (6/9) of patients with an elevated tryptase having an IgE-mediated cause, compared to 0% (0/2) with a non-elevated tryptase level. An increase in serum tryptase compared to the baseline value is an indicator of mast cell activation even if the serum tryptase level is below the upper normal value of 11.4 ng/mL.18 While an elevated tryptase level is helpful in determining whether or not a perioperative HSR is IgE mediated it is certainly not diagnostic. Dong et al. reported in 2012 on 599 cases of perioperative HSR in which serum tryptase was measured and found a mean tryptase level of 13 ng/mL in 10.6% of cases labeled as non-IgE HSR.4 However, the information gained through obtaining a tryptase level during a potential perioperative HSR likely outweights cost involved in conducting the test. Given the importance of this test in helping the allergist evaluate patients with perioperative HSRs, we frequently engage the Department of Anesthesia with case-based educational events that demonstrate its utility. Performing perioperative tryptase testing in cases of suspected HSR has become a quality improvement goal shared by both departments.
In our study population, two patients had persistently elevated tryptase levels during HSR and after the event in follow up. Both of these patients underwent evaluation for possible systemic mastocytosis. Although uncommon, mast cell disorders were identified in 3 (4%) patients of our institutions’ prior perioperative HSR study.6 Further support of this statement comes from a previous study of mast cell disease in patients with drug HSRs, where 7 out of 86 (8%, 95%, confidence interval 3.8%–16.1%) patients with drug hypersensitivity had increased serum tryptase levels (>11.4 ng/ml), although only one patient was diagnosed with systemic mastocytosis after bone marrow biopsy.19 While incidence rates remain unknown, general anesthetics have been listed as risk factors for anaphylaxis in patients with mastocytosis,20–23 and guidelines have been published to minimize the risks of anesthesia in adult patients with systemic mastocytosis.24,25 It is therefore important to repeat a total tryptase measurement at least 24 hours after all signs and symptoms of a HSR have resolved for patients who develop perioperative HSR.
This study has several limitations, including its low number of subjects. Indeed, we only had nine patients have subsequent anesthesia, which is far too low to draw firm conclusions on the safety of subsequent anesthesia following our recommendations. However, this analysis provides a unique look at the clinical findings and recommendations made by allergists, and supports the utility an d safety of our approach. The creation of a system for referral of all intraoperative anaphylaxis cases helped reduce selection bias, but when compared to the other prospective perioperative HSR study2, we may have been referred the more severe reactions and may have missed more mild reactions. In select cases, we were limited by the delayed timing of skin testing or the patient’s preference not to pursue a comprehensive evaluation. This delay in skin testing could have led to false-negative results given the waning of skin test positivity over time.26 We are also limited by the lack of validated skin testing for most drugs, except for PCN, used in skin testing. For instance, a non-irritating concentration for ciprofloxacin remains a point of controversy in the literature and results should be interpreted with caution.27–29 It is possible that we identified antibiotics as the most common cause of IgE-mediated perioperative HSR because antibiotic testing is superior to testing for other agents. However, our protocols use the non-irritating concentrations informed by the best data currently available and consistent with the testing standards in the Drug Allergy Practice Parameters. Additionally, because anesthetic drugs require expert administration, patients in our study with negative skin tests did not undergo drug challenges to confirm the lack of drug hypersensitivity. Lastly, we report on only one Boston teaching hospital, so our experience here may not be generalizable to different patient populations, geographic areas or hospitals/clinics with different practices and resources.
In summary, we identified antibiotics—specifically, cefazolin—as the most likely cause of perioperative HSR in our patient cohort. Additionally, a serum tryptase test may be helpful in evaluating perioperative HSR. Furthermore, by prospectively enrolling patients in our institution’s comprehensive evaluation and management plan, patients were able to tolerate subsequent anesthesia.
Table V.
Causative Antibiotic Agents Identified by Skin Testing
| Subject ID | Sex | Grade of Reaction | Procedure Performed | Total Tryptase during HSR (ng/mL)* | Positive Skin Test |
|---|---|---|---|---|---|
| 2 | M | III | Muscle biopsy | 61 | Cefazolin |
| 6 | F | II | Lumpectomy | - | Cefazolin |
| 8 | F | I | Total knee replacement | - | Cefazolin |
| 10 | M | III | Renal transplantation | 243 | Cefazolin |
| 17 | F | II | Atrial fibrillation ablation | 69 | Vancomycin |
| 18 | M | III | Endoscopy with biopsy | - | PCN |
| 19 | M | II | Brachial plexus block | 21 | Cefazolin |
| 26 | F | II | Bilateral mastectomy | 25 | Ciprofloxacin |
| 29 | M | II | Rotator cuff repair | - | Cefazolin |
Tryptase reference range is <11.5 ng/mL.
Abbreviations: HSR: hypersensitivity reaction, ng/mL: nanograms per mililiter, M: male, F: female, PCN: penicillin
HIGHLIGHTS BOX.
1. What is already known about this topic?
Current knowledge of drug hypersensitivity reactions (HSRs) during anesthesia comes from retrospective studies. The most commonly implicated drugs causing perioperative HSR varies between countries.
2. What does this article add to our knowledge?
We describe a successful referral and treatment plan for patients with HSRs during anesthesia in a Boston teaching hospital.
3. How does this study impact current management guidelines?
The most commonly identified cause of perioperative HSRs was antibiotics. Allergists should evaluate patients with HSR during anesthesia to minimize risk with subsequent anesthesia.
ABBREVIATIONS
- HSR(s)
Hypersensitivity reaction(s)
- NMBAs
Neuromuscular blocking agents
- IgE
Immunoglobulin E
- PCN
penicillin
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Galvão VR, Giavina-Bianchi P, Castells M. Perioperative anaphylaxis. Current allergy and asthma reports. 2014;14:452. doi: 10.1007/s11882-014-0452-6. [DOI] [PubMed] [Google Scholar]
- 2.Berroa F, Lafuente A, Javaloyes G, Cabrera-Freitag P, de la Borbolla JM, Moncada R, Goikoetxea MJ, Sanz ML, Ferrer MGG. The Incidence of Perioperative Hypersensitivity Reactions: A Single-Center, Prospective, Cohort Study. Anesth Analg. 2015 doi: 10.1213/ANE.0000000000000776. [DOI] [PubMed] [Google Scholar]
- 3.Mertes PM, et al. Reducing the risk of anaphylaxis during anesthesia: 2011 updated guidelines for clinical practice. J Investig Allergol Clin Immunol. 2011;21:442–453. [PubMed] [Google Scholar]
- 4.Dong SW, Mertes PM, Petitpain N, Hasdenteufel F, Malinovsky JM. Hypersensitivity reactions during anesthesia. Results from the ninth French survey (2005–2007) Minerva Anestesiol. 2012;78:868–878. [PubMed] [Google Scholar]
- 5.Antunes J, Kochuyt A-M, Ceuppens JL. Perioperative allergic reactions: experience in a Flemish referral centre. Allergol Immunopathol (Madr) 42:348–54. doi: 10.1016/j.aller.2013.08.001. [DOI] [PubMed] [Google Scholar]
- 6.Guyer AC, et al. Comprehensive Allergy Evaluation Is Useful in the Subsequent Care of Patients with Drug Hypersensitivity Reactions During Anesthesia. J Allergy Clin Immunol Pract. 2015;3:94–100. doi: 10.1016/j.jaip.2014.11.011. [DOI] [PubMed] [Google Scholar]
- 7.Gonzalez-Estrada A, Pien LC, Zell K, Wang X-F, Lang DM. Antibiotics Are an Important Identifiable Cause of Perioperative Anaphylaxis in the United States. J Allergy Clin Immunol Pract. 2015;3:101–105.e1. doi: 10.1016/j.jaip.2014.11.005. [DOI] [PubMed] [Google Scholar]
- 8.Gurrieri C, et al. Allergic reactions during anesthesia at a large united states referral center. Anesth Analg. 2011;113:1202–1212. doi: 10.1213/ANE.0b013e31822d45ac. [DOI] [PubMed] [Google Scholar]
- 9.Hesterberg PE, et al. Risk stratification for desensitization of patients with carboplatin hypersensitivity: clinical presentation and management. J Allergy Clin Immunol. 2009;123:1262–7.e1. doi: 10.1016/j.jaci.2009.02.042. [DOI] [PubMed] [Google Scholar]
- 10.Patil SU, et al. A protocol for risk stratification of patients with carboplatin-induced hypersensitivity reactions. J Allergy Clin Immunol. 2012;129:443–447. doi: 10.1016/j.jaci.2011.10.010. [DOI] [PubMed] [Google Scholar]
- 11.Goldberg A, Confino-Cohen R. Timing of venom skin tests and IgE determinations after insect sting anaphylaxis. J Allergy Clin Immunol. 1997;100:182–184. doi: 10.1016/s0091-6749(97)70222-7. [DOI] [PubMed] [Google Scholar]
- 12.Lobera TI, et al. Study of hypersensitivity reactions and anaphylaxis during anesthesia in Spain. J Investig Allergol Clin Immunol. 2008;18:350–356. [PubMed] [Google Scholar]
- 13.Bratzler DW, et al. Clinical practice guidelines for antimicrobial prophylaxis in surgery. Am J Heal Pharm. 2013;70:195–283. doi: 10.2146/ajhp120568. [DOI] [PubMed] [Google Scholar]
- 14.Pichichero ME, Zagursky R. Penicillin and Cephalosporin allergy. Annals of Allergy, Asthma and Immunology. 2014;112:404–412. doi: 10.1016/j.anai.2014.02.005. [DOI] [PubMed] [Google Scholar]
- 15.Goodman EJ, et al. Cephalosporins can be given to penicillin-allergic patients who do not exhibit an anaphylactic response. J Clin Anesth. 2001;13:561–564. doi: 10.1016/s0952-8180(01)00329-4. [DOI] [PubMed] [Google Scholar]
- 16.Krøigaard M, Garvey LH, Menné T, Husum B. Allergic reactions in anaesthesia: Are suspected causes confirmed on subsequent testing? Br J Anaesth. 2005;95:468–471. doi: 10.1093/bja/aei198. [DOI] [PubMed] [Google Scholar]
- 17.Lafuente A, et al. Early skin testing is effective for diagnosis of hypersensitivity reactions occurring during anesthesia. Allergy Eur J Allergy Clin Immunol. 2013;68:820–822. doi: 10.1111/all.12154. [DOI] [PubMed] [Google Scholar]
- 18.Borer-Reinhold M, et al. An increase in serum tryptase even below 11.4 ng/mL may indicate a mast cell-mediated hypersensitivity reaction: a prospective study in Hymenoptera venom allergic patients. Clin Exp Allergy. 2011;41:1777–83. doi: 10.1111/j.1365-2222.2011.03848.x. [DOI] [PubMed] [Google Scholar]
- 19.Bonadonna P, et al. How much specific is the association between hymenoptera venom allergy and mastocytosis? Allergy Eur J Allergy Clin Immunol. 2009;64:1379–1382. doi: 10.1111/j.1398-9995.2009.02108.x. [DOI] [PubMed] [Google Scholar]
- 20.Brockow K, Bonadonna P. Drug allergy in mast cell disease. Current Opinion in Allergy and Clinical Immunology. 2012;12:354–360. doi: 10.1097/ACI.0b013e328355b7cb. [DOI] [PubMed] [Google Scholar]
- 21.Casale TB, Bowman S, Kaliner M. Induction of human cutaneous mast cell degranulation by opiates and endogenous opioid peptides: evidence for opiate and nonopiate receptor participation. J Allergy Clin Immunol. 1984;73:775–781. doi: 10.1016/0091-6749(84)90447-0. [DOI] [PubMed] [Google Scholar]
- 22.González De Olano D, et al. Prevalence of allergy and anaphylactic symptoms in 210 adult and pediatric patients with mastocytosis in Spain: A study of the Spanish network on mastocytosis (REMA) Clin Exp Allergy. 2007;37:1547–1555. doi: 10.1111/j.1365-2222.2007.02804.x. [DOI] [PubMed] [Google Scholar]
- 23.Valent P, et al. Definitions, criteria and global classification of mast cell disorders with special reference to mast cell activation syndromes: a consensus proposal. Int Arch Allergy Immunol. 2012;157:215–25. doi: 10.1159/000328760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Brockow K, Jofer C, Behrendt H, Ring J. Anaphylaxis in patients with mastocytosis: A study on history, clinical features and risk factors in 120 patients. Allergy Eur J Allergy Clin Immunol. 2008;63:226–232. doi: 10.1111/j.1398-9995.2007.01569.x. [DOI] [PubMed] [Google Scholar]
- 25.Chaar CIO, Bell RL, Duffy TP, Duffy AJ. Guidelines for safe surgery in patients with systemic mastocytosis. The American surgeon. 2009;75:74–80. [PubMed] [Google Scholar]
- 26.Blanca M, et al. Natural evolution of skin test sensitivity in patients allergic to beta-lactam antibiotics. J Allergy Clin Immunol. 1999;103:918–924. doi: 10.1016/s0091-6749(99)70439-2. [DOI] [PubMed] [Google Scholar]
- 27.Empedrad R, Darter AL, Earl HS, Gruchalla RS. Nonirritating intradermal skin test concentrations for commonly prescribed antibiotics. J Allergy Clin Immunol. 2003;112:629–30. doi: 10.1016/s0091-6749(03)01783-4. [DOI] [PubMed] [Google Scholar]
- 28.Seitz CS, Bröcker EB, Trautmann A. Diagnostic testing in suspected fluoroquinolone hypersensitivity. Clin Exp Allergy. 2009;39:1738–45. doi: 10.1111/j.1365-2222.2009.03338.x. [DOI] [PubMed] [Google Scholar]
- 29.Blanca-López N, et al. Hypersensitivity reactions to fluoroquinolones: analysis of the factors involved. Clin Exp Allergy. 2013;43:560–7. doi: 10.1111/cea.12099. [DOI] [PubMed] [Google Scholar]
- 30.Thong BYH, Yeow-Chan Anaphylaxis during surgical and interventional procedures. Ann Allergy Asthma Immunol. 2004;92:619–628. doi: 10.1016/S1081-1206(10)61427-5. [DOI] [PubMed] [Google Scholar]
- 31.Mertes PM, et al. Perioperative Anaphylaxis. Immunol Allergy Clin North Am. 2009;29:429–451. doi: 10.1016/j.iac.2009.04.004. [DOI] [PubMed] [Google Scholar]
- 32.Drug allergy: an updated practice parameter. Ann Allergy Asthma Immunol. 2010;105:259–273. doi: 10.1016/j.anai.2010.08.002. [DOI] [PubMed] [Google Scholar]
- 33.McClimon B, Rank M, Li J. The predictive value of skin testing in the diagnosis of local anesthetic allergy. Allergy Asthma Proc. 2011;32:95–98. doi: 10.2500/aap.2011.32.3417. [DOI] [PubMed] [Google Scholar]
- 34.Berkun Y, Ben-Zvi A, Levy Y, Galili D, Shalit M. Evaluation of adverse reactions to local anesthetics: experience with 236 patients. Ann Allergy Asthma Immunol. 2003;91:342–345. doi: 10.1016/S1081-1206(10)61680-8. [DOI] [PubMed] [Google Scholar]
- 35.Brož P, et al. Nonirritant intradermal skin test concentrations of ciprofloxacin, clarithromycin, and rifampicin. Allergy. 2012;67:647–52. doi: 10.1111/j.1398-9995.2012.02807.x. [DOI] [PubMed] [Google Scholar]
- 36.Adachi A, Fukunaga A, Horikawa T. A case of human insulin allergy induced by short-acting and intermediate-acting insulin but not by long-acting insulin. Int J Dermatol. 2004;43:597–9. doi: 10.1111/j.1365-4632.2004.02141.x. [DOI] [PubMed] [Google Scholar]
- 37.Castéra V, Dutour-Meyer A, Koeppel M, Petitjean C, Darmon P. Systemic allergy to human insulin and its rapid and long acting analogs: successful treatment by continuous subcutaneous insulin lispro infusion. Diabetes Metab. 2005;31:391–400. doi: 10.1016/s1262-3636(07)70210-0. [DOI] [PubMed] [Google Scholar]
- 38.Bousquet PJ, Co-Minh HB, Demoly P. Isolated urticaria to ondansetron and successful treatment with granisetron. Allergy Eur J Allergy Clin Immunol. 2005;60:543–544. doi: 10.1111/j.1398-9995.2005.00754.x. [DOI] [PubMed] [Google Scholar]
- 39.Demir HA, et al. Anaphylactic reaction owing to ondansetron administration in a child with neuroblastoma and safe use of granisetron: a case report. Journal of pediatric hematology/oncology : official journal of the American Society of Pediatric Hematology/Oncology. 2010;32:e341–e342. doi: 10.1097/MPH.0b013e3181ea214a. [DOI] [PubMed] [Google Scholar]
- 40.Laxenaire MC, Mata-Bermejo E, Moneret-Vautrin DA, Gueant JL. Life-threatening anaphylactoid reactions to propofol (Diprivan) Anesthesiology. 1992;77:275–280. doi: 10.1097/00000542-199208000-00009. [DOI] [PubMed] [Google Scholar]
- 41.Chong YY, Caballero MR, Lukawska J, Dugué P. Anaphylaxis during general anaesthesia: one-year survey from a British allergy clinic. Singapore Med J. 2008;49:483–7. [PubMed] [Google Scholar]