Abstract
The aim of the present study was to investigate the overall clinical expression characteristics of the cluster of differentiation (CD)28 family receptors [CD28, inducible T-cell co-stimulator, programmed cell death protein 1 (PD-1), cytotoxic T-lymphocyte-associated protein 4 and B- and T-lymphocyte attenuator] on T cells in patients with chronic hepatitis B (CHB), analyze the correlations among these receptors and the clinical parameters, and to investigate the effects of PD-1 blockade on the receptor expression profiles, T-cell function and other biological effects. The expression characteristics of the CD28 family of receptors, the effects of PD-1 blockade on the receptor expression profiles and the levels of interferon (IFN)-γ were investigated in the T cells of patients with CHB. In addition, the transcription factor, T-box 21 (T-bet) and GATA binding protein 3 (GATA-3) mRNA expression levels were investigated in the peripheral blood mononuclear cells (PBMCs) of patients with CHB. The expression levels of the CD28 family receptors in the T cells of patients with CHB demonstrated distinct characteristics, for example levels of PD-1 and CTLA-4 on CD4 T cells and ICOS, PD-1, and BTLA on CD8 T cells were increased in cells from patients with CHB compared with those from the healthy individuals. A significant positive correlation was demonstrated among the serum HBV DNA titers and the levels of PD-1 on CD8+ T cells with the highest expression of PD-1 corresponding to viral levels >106 IU/ml. A significant positive correlation was observed between the serum HBV DNA titers and the expression levels of BTLA on CD8+ T cells with the highest expression of BTLA corresponding to viral levels >106 IU/ml. PD-1 blockade altered the expression profiles of CD28 family receptors in the T cells of patients with CHB, partly enhanced T cell function and increased the ratio of T-bet/GATA-3 mRNA in PBMCs. Thus, CD28 family receptors are potential clinical indicators for the rapid monitoring of changes in T cell function during CHB treatment. Furthermore, PD-1 blockade has a therapeutic potential that may be enhanced by modulating the expression of co-stimulatory and -inhibitory receptors of the CD28 family.
Keywords: cluster of differentiation 28 family of receptors, chronic hepatitis B, programmed cell death 1, programmed death-ligand 1
Introduction
Hepatitis B virus (HBV) is a persistent infection that has become a global threat to human health. Despite having an effective preventive vaccine, there are ~1 million deaths each year from complications resulting from chronic HBV infection. The present study suggests that functional depletion of T cell is an important feature of chronic HBV infection. The presence of increased quantities of antigen stimulation is thought to be an important reason for the T cell exhaustion in patients with chronic hepatitis B (CHB) (1,2). In the progression of chronic viral infection, abnormal expression of co-stimulatory and inhibitory receptors are often identified on the surfaces of exhaustive T cells, and vary in diversity and quantity depending on the course of the disease (3).
The cluster of differentiation (CD)28 family is the primary co-stimulatory molecule expressed on T cells. The CD28 family consists of the co-stimulatory receptors, CD28 and inducible T-cell co-stimulator (ICOS), and three co-inhibitory receptors, programmed cell death protein 1 (PD-1), cytotoxic T-lymphocyte-associated protein 4 (CTLA-4), and B- and T-lymphocyte attenuator (BTLA) (4). The members of the CD28 family possess a similar gene structure and they are important in the regulation of T cell function. Co-stimulatory receptors provide positive signals for T cells to promote proliferation. Co-inhibitory receptors produce inhibitory signals for T cells to limit, terminate and attenuate the T cell response (5). Upon ligand combination, these receptors regulate the function of various stages of T cell-mediated immune responses, including numerous positive or negative regulations of cellular and humoral immunity (6–8). T cells regulate immune responses by maintaining the balance between stimulation and inhibition. Understanding these pathways may provide novel insight into the diagnosis and treatment of viral diseases.
The association of the CD28 family of receptors with chronic human viral infections has been investigated in the past; however, previous research has been limited to a single study of co-stimulatory or co-inhibitory receptors in the human immunodeficiency virus (HIV) infection (9). Only a small number of studies have investigated HBV (10,11), whereas PD-1 has been researched to a greater extent and is well understood. The function of exhausted T cells in CHB has been demonstrated to be reversed or improved by PD-1 blockade in ex vivo studies (12-14). However, the underlying mechanism of this effect is not fully understood. Whether or not this blockade may tip the balance among the co-stimulatory or co-inhibitory receptors of the CD28 family or molecules requires further investigation, as the PD-1 blockade may trigger immune injury while effectively removing the virus.
The current study demonstrated the overall expression characteristics of the CD28 family of receptors and effects on T cells in persistent HBV infection, and correlated the expression with clinical parameters. In addition, in order to demonstrate the effect of PD-1 block on receptors of the CD28 family, cell function and differentiation, the expression levels of receptors in the CD28 family, interferon (IFN)γ and the ratio of T-box 21 (T-bet)/GATA binding protein 3 (GATA-3) mRNA in peripheral blood mononuclear cells (PBMCs) was tested. It was determined that blockage of PD-1 may lead to the increased expression of receptors of the CD28 family, cell function and differentiation may change accordingly. Thus, the therapeutic potential of PD-1 blockade demonstrates a correlation with the expression levels of the co-stimulatory and co-inhibitory receptors of the CD28 family in chronic HBV infection.
Materials and methods
Subjects
A total of 52 patients with CHB from the Department of Infectious Disease, Union Hospital (Wuhan, China) served as the experimental group and 26 healthy volunteers served as controls. Patients were diagnosed with CHB according to the guidelines for the prevention and treatment of the disease (15). Patients with other chronic liver diseases (i.e. caused by other viruses, autoimmunity, alcohol consumption, non-alcoholic fatty liver, medicines and toxins) were excluded. None of the patients had been treated with antiviral drugs or by immunological methods for a minimum of 1 year prior to recruitment. There were no statistically significant differences in age between the experimental and control groups. All patients were hepatitis B virus surface antigen (HBsAg) seropositive; 19 of which were hepatitis E antigen (HBeAg) seropositive. Patient alanine transaminase (ALT) levels ranged between 87 and 954 U/l (median, 205 U/l), and HBV DNA ranged between 5.67×102 and 4.98×108 IU/ml (median, 7.52×105 IU/ml). The polymerase chain reaction (PCR) method was used to screen for human leukocyte antigen (HLA)-A2+ and 22 HLA-A2+ patients were identified. The study was approved by the Medical Ethical Committee of Huazhong University of Science and Technology and written informed consent was obtained from all subjects.
Virologic determination
The presence or absence of HBsAg, HBeAg, antibody to hepatitis B surface antigen (anti-HBs), anti-HBe, anti-HBc, antibody to hepatitis C virus (anti-HCV), anti-hepatitis D virus (HDV) and anti-HIV (1/2) were determined using commercial enzyme immunoassay kits (Kehua Bio-Engineering Co., Ltd., Shanghai, China). HBV DNA quantification was performed at the laboratory of Hepatology and Infectious Disease, Union Hospital (Hubei, China) using a commercial PCR diagnostic kit (DaAn Gene Co., Ltd., Guangzhou, China). The cut-off value of HBV DNA was 5.0×102 IU/ml and the lower limit of HBV DNA detection was 1.0×102 IU/ml.
Synthetic peptides, antibodies and primers
A synthetic peptide with HLA-A2-restricted epitope HBcAg18-27 (FLPSDFFPSI) was synthesized by the Chinese Peptide Company (Hangzhou, China). Antibodies against CD8 allophycocyanin (APC; cat. no.17-0088; 1:20), CD8 fluorescein isothiocyanate (FITC; cat. no. 11-0088; 1:20), CD4 phycoerythrin (PE; cat. no. 12-0049; 1:20), CD4 FITC (cat. no. 11-0049; 1:20), CD3 peridinin chlorophyll protein (cat. no. 45-0037; 1:20), ICOS APC (cat. no. 17-9948; 1:20), CD28 PE (cat. no. 12-0289; 1:20), IFN-γ FITC (cat. no. 11-7319; 1:20), PD-L1 (cat. no. 16-5983; 1:200) and their corresponding isotype control antibodies (ICOS; cat. no. 17-4714; 1:20, CD28; cat. no. 12-4714; 1:20, IFN-γ; cat. no. 11-4714; 1:20, PD-L1; cat. no. 16-4714; 1:200) were purchased from eBioscience, Inc. (San Diego, CA, USA). Antibodies against PD-1 FITC (cat. no. 329904; 1:20), CTLA-4 APC (cat. no. 349908; 1:20), BTLA PE (cat. no. 344505; 1:20), CD28 (cat. no. 302902), their isotype control antibodies (PD-1; cat. no. 400107; 1:20, CTLA-4; cat. no. 400121; 1:20, BTLA; cat. no. 400211; 1:20) and 7-AAD Viability Staining Solution [7-amino-actinomycin D (7-AAD)] were purchased from Biolegend, Inc. (San Diego, CA, USA). PCR primers for HLA-A2 (forward, 5′-GTGGATAGAGCAGGAGGGT-3′ and reverse, 5′-CCAAGAGCGCAGGTCCTCT-3′) were purchased from Invitrogen; Thermo Fisher Scientific, Inc. Real-time quantitative primers specific for the transcription factors, T-bet (cat. no. QT00042217), GATA-3 (cat. no. QT00095501) and β-actin (cat. no. QT01680476) were purchased from Qiagen GmbH (Hilden, Germany).
Selection of HLA-A2+ individuals
Selection of HLA-A2+ individuals was completed by the method of ordinary PCR. A total of 5 ml venous blood was collected. DNA was extracted from fresh heparinized blood using a commercial blood DNA kit (Omega Bio-Tek, Inc., Norcross, GA, USA). HLA-A2+ individuals were selected by PCR as previously described (16). The cycling conditions for the PCR reaction were as follows: 94.0°C for 5 min, 30 cycles of 94.0°C for 45 sec, 60.0°C for 45 sec and 72.0°C for 45 sec. The products of PCR were determined using 2% agarose gel electrophoresis.
Isolation of PBMCs, in vitro culture and PD-1:PD-L1 blocking
PBMCs were isolated from fresh heparinized blood using Ficoll-Hypaque density gradient centrifugation at 800 × g for 20 min (HaoYang Biological Manufacture Co., Ltd., Tianjin, China) and were resuspended in RPMI-1640 medium supplemented with 10% fetal bovine serum and 1% penicillin-streptomycin (Gibco; Thermo Fisher Scientific, Inc.).
To assess the effects of blocking the PD-1:PD-L1 signaling pathway, PBMCs from HLA-A2+ individuals were cultured in a flat-bottomed 96-well plate (2–10×105 cells/well) in the presence of 5 µg/ml HBcAg18-27 peptide, 5 µg/ml anti-PD-L1 or control IgG for PD-1:PD-L1 for 10 days, supplemented with 25 IU/ml interleukin (IL)-2 (PeproTech, Inc., Rocky Hill, NJ, USA) and 0.5 µg/ml anti-CD28 at 0 and 5 days.
Flow cytometric analysis of CD28 family receptors and intracellular IFN-γ
PBMCs were surface-stained with anti-CD28, anti-ICOS, anti-PD-1, anti-CTLA-4, anti-BTLA, 7-AAD, anti-CD4, anti-CD8 and their corresponding isotype fluorescent antibodies. Subsequent to staining, cells were detected by flow cytometry (FACSCalibur; BD Biosciences, San Jose, CA, USA), and data were analyzed based on the levels of background of isotype-matched controls using the Flowjo 7.6.1 software (FlowJo, LLC., Ashland, OR, USA). In order to evaluate the effect of anti-PD-L1 on the expression of the CD28 family receptors on CD4+ and CD8+ T cells, cells cultured into 96-well plates in the presence of anti-PD-L1 or control antibody for 10 days, were collected, washed twice with PBS and analyzed as described above. For intracellular IFN-γ staining, following exposure to anti-PD-L1 for 10 days, cells were transferred from flat-bottomed 96-well plates to round-bottomed 96-well plates, washed once with RPMI-1640 with 10% FBS medium and incubated with 5 µg/ml HBcAg18-27 peptide, 0.5 µg/ml anti-CD28 and 1 µg/ml brefeldin A (eBioscience, Inc.) for 5 h at 37°C. The cells were washed twice with PBS and then stained with anti-CD4, anti-CD8, and 7-AAD and fixed. Cells were then fixed/permeabilized using Fixation/Permeabilization kit following manufacturer's protocol (BD Biosciences) for intracellular staining with anti-IFN-γ and analyzed as described above.
Expression of T-bet and GATA-3 genes in PBMCs
To assess the effect of anti-PD-L1 on T-bet and GATA-3 in PBMCs, total RNA from PBMCs treated with anti-PD-L1 or control antibody for 10 days was extracted using RNAiso Plus reagent (Takara Biotechnology Co., Ltd., Dalian, China) according to the manufacturer's instructions. T-bet, GATA-3 and β-actin mRNA were amplified by a Real Time One Step RT-PCR method with the One Step SYBR RT-PCR kit (Takara Biotechnology Co., Ltd.). The conditions used for qPCR were according to the manufacturer's protocol. PCR reactions were performed on a real-time fluorescent quantitative PCR analyzer (Bio-Rad Laboratories, Inc., Hercules, CA, USA). The conditions for reverse transcription reaction were 42.0°C for 5 min and 95.0°C for 10 sec. The cycling conditions for the PCR reaction were: 95.0°C for 5 sec, 57.0°C for 30 sec (+fluorescence detection), 39 cycles of 95.0°C for 5 sec and 57.0°C for 30 sec, melt curve 65.0–95.0°C (0.5°C increments for 5 sec, +fluorescence detection). The relative gene expression levels of T-bet and GATA-3 normalized to the corresponding β-actin were calculated automatically by the CFX Manager 2.1 software (Bio-Rad Laboratories, Inc.).
Statistical analysis
Data were analyzed using the GraphPad Prism 6.01 software (San Diego, USA). The Mann-Whitney U test was used to analyze the differences among the various groups. The Wilcoxon paired test was used to compare differences in expression inside and outside the cell membranes, prior and subsequent to blocking of PD-1:PD-L1. Correlations among various molecules, and the HBV DNA values and molecules were evaluated using the Spearman test. Data are presented as the mean ± standard error. A two-tailed test was used to analyze all data and P<0.05 was considered to indicate a statistically significant difference.
Results
T cells in CHB express different levels of CD28 family receptors compared with those of healthy individuals
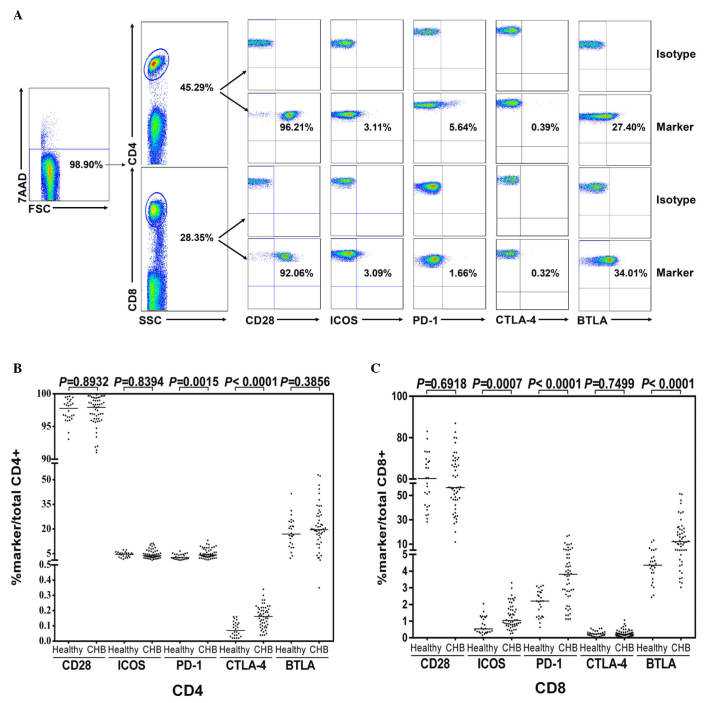
To compare the in vitro expression frequency of the CD28 family, 5 receptors on total peripheral CD4+ and CD8+ T cells from patients with CHB (n=52) and healthy controls (n=26) were investigated. Representative flow cytometric scatter plots of the CD28 family receptors on peripheral CD4+ and CD8+ T cells from a patient with CHB are demonstrated in Fig. 1A. Levels of PD-1 and CTLA-4 on CD4 T cells (Fig. 1B) and ICOS, PD-1, and BTLA on CD8 T cells (Fig. 1C) were increased in cells from patients with CHB compared with those from the healthy controls.
Figure 1.
Expression of CD28 family receptors on CD4+ and CD8+ T cells of patients with CHB. Peripheral blood mononuclear cells were isolated from healthy donors (n=26) or patients with CHB (n=52), and stained with anti-CD4, CD8, CD28, CTLA-4, PD-1, ICOS, BTLA or isotype control antibodies. Dead cells were excluded by 7-AAD staining. (A) Representative flow cytometric scatter plots of CD28 family receptors on peripheral CD4+ and CD8+ T cells from a patient with CHB. (B and C) Expression of CD28, CTLA-4, PD-1, ICOS and BTLA receptors. CD, cluster of differentiation; CHB, chronic hepatitis B; CTLA-4, cytotoxic T-lymphocyte-associated protein 4; PD-1, programmed cell death protein 1; ICOS, inducible T-cell co-stimulator; BTLA, B- and T-lymphocyte attenuator; 7-AAD, 7-aminoactinomycin D.
Correlations among the CD28 family receptors and clinical parameters in CHB
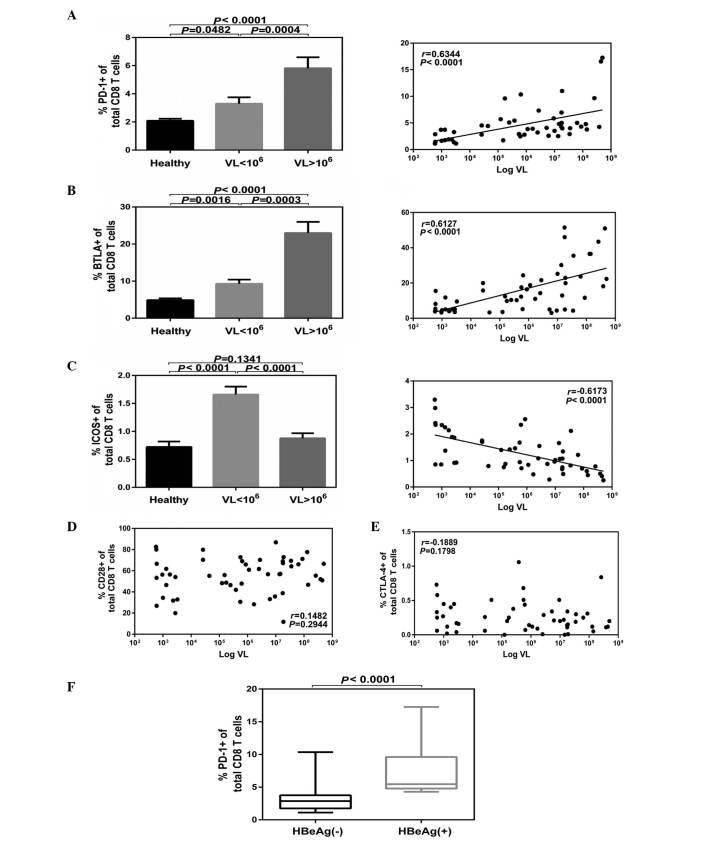
A significant positive correlation was demonstrated among the serum HBV DNA titers and the levels of PD-1 on CD8+ T cells with the highest expression of PD-1 corresponding to viremia levels >106 IU/ml (P<0.0001; Fig. 2A). A significant positive correlation was observed among the serum HBV DNA titers and the expression levels of BTLA on CD8+T cells with the highest expression of BTLA corresponding to viremia levels >106 IU/ml (P<0.0001; Fig. 2B). However, a significant inverse correlation was indicated among the serum HBV DNA titers and expression levels of ICOS on CD8+ T cells with the lowest expression of ICOS corresponding to viremia levels >106 IU/ml (P<0.001; Fig. 2C). No significant difference was demonstrated compared with the healthy controls (P>0.05; Fig. 2C). No correlation was observed among the serum HBV DNA titers and expression levels of CD28 (Fig. 2D) or CTLA-4 (Fig. 2E) on CD8+ T cells. In addition, no correlations were observed among the virological parameters and the abnormal expression of the CD28 family receptors on CD4+ T cells in patients with CHB (data not shown). Furthermore, expression of PD-1 on CD8+ T cells was significantly higher in the HBeAg+ group compared with the HBeAg− group (P<0.0001; Fig. 2F).
Figure 2.
Expression of CD28 family receptors associated with the HBV DNA levels. Patients were classified as low (n=27) and high (n=25) VL, and CD4 or CD8 cells expressing CD28 family receptors were analyzed. (A) PD-1 and (B) BTLA on CD8 T cells were positively correlated with VL, (C) ICOS were inversely correlated, while (D) CD28 and (E) CTLA-4 demonstrated no correlation with HBV DNA levels. (F) Cumulative PD-1 expression data on CD8+ T cells in HBeAg+ (n=19) and HBeAg− groups (n=33). Data are presented as the mean ± standard error. CD, cluster of differentiation; HBV, hepatitis B virus; PD-1, programmed cell death protein 1; BTLA, B- and T-lymphocyte attenuator; ICOS, inducible T-cell co-stimulator; CTLA-4, cytotoxic T-lymphocyte-associated protein 4; HBeAg, hepatitis E antigen; VL, viral levels.
Effects of blocking PD-1 on the expression profile of the CD28 family receptors and function of CD4 T cells in CHB
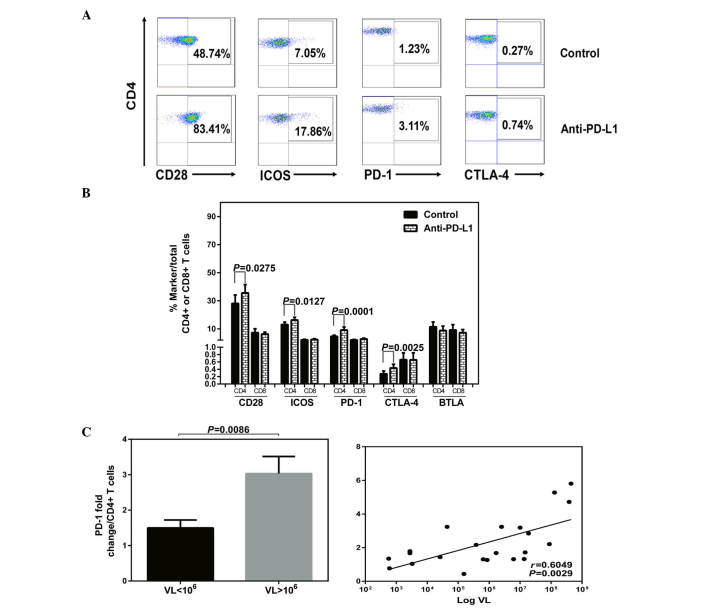
Besides assessing function, the effects of blocking PD-1 on the levels of co-stimulatory and co-inhibitory receptors were investigated. The expression of the CD28 family receptors and intracellular IFN-γ were measured simultaneously in T cells in HLA-A2+ patients with CHB following exposure to anti-PD-L1 or a control antibody. Increased expression of the receptors with PD-1 blockade or isotype antibody treatment are demonstrated by flow cytometric dot plots of peripheral CD4+ T cells from a patient with CHB (Fig. 3A). Following anti-PD-L1 exposure, the expression levels of CD28, ICOS, PD-1 and CTLA-4 were increased in the CD4+ T cells (Fig. 3B). No significant impact on the expression of the CD28 family receptors was observed on the CD8+ T cells following anti-PD-L1 exposure (Fig. 3B; P>0.05).
Figure 3.
Anti-PD-L1 treatment and expression of CD28 family receptors. Peripheral blood mononuclear cells derived from HLA-A2+ CHB patients (n=22) were isolated and cultured in the presence of anti-CD28, IL-2 and HBcAg18-27 peptide for 10 days. Cells were treated with anti-PD-L1 antibody or isotype antibody at a concentration of 5 µg/ml. (A) Representative flow cytometric dot plots of CD28 family receptors on CD4+ T cells with PD-1 blockade or isotype antibody treatment from a patient with CHB. (B) Expression of the receptors with PD-1 blockade or isotype antibody treatment. The fold-change of a single receptor was calculated by percentage following PD-1 blockade/before blockade. (C) Cumulative data showing fold-changes of PD-1 in CD4+ T cells with low and high VL, and correlation among HBV DNA titers and the fold-changes of PD-1 following anti-PD-L1 exposure. PD-L1; PD-ligand 1; CD, cluster of differentiation; CHB, chronic hepatits B; IL-2, interleukin 2; PD-1, programmed cell death protein 1; ICOS, inducible T-cell co-stimulator; CTLA-4, cytotoxic T-lymphocyte-associated protein 4; VL, viral levels.
In addition, following anti-PD-L1 exposure, a significant positive correlation was detected among the HBV DNA titers and the fold changes of PD-1, with the highest fold-change of PD-1 in CD4+ T cells corresponding to viremia levels >106 IU/ml (VL<106 vs. VL>106, P=0.0086; Log VL vs. PD-1 fold change, P=0.0029; Fig. 3C).
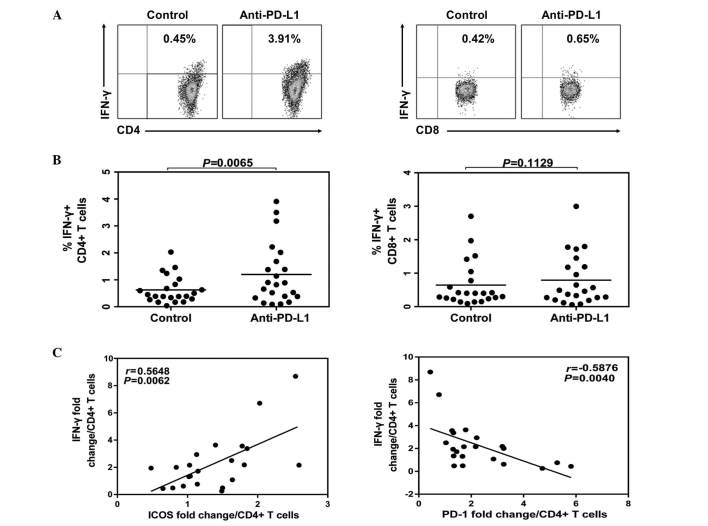
The representative flow cytometric dot plots of intracellular IFN-γ expression in CD4+ and CD8+ T cells with PD-1 blockade or isotype antibody treatment are demonstrated in Fig. 4. Following anti-PD-L1 treatment, the levels of IFN-γ were increased in CD4+ T cells (Fig. 4B) and IFN-γ fold changes in CD4+ T cells had a positive correlation with ICOS fold-change (P=0.0062; Fig. 4C). However, there was an inverse correlation with PD-1 fold-change (P=0.004; Fig. 4C). In the current study, no significant impact was demonstrated on the expression of IFN-γ in the CD8+ T cells following anti-PD-L1 treatment (Fig. 4B; P>0.05).
Figure 4.
IFN-γ expression of CD4 T cells following PD-1 blockade. Peripheral blood mononuclear cells derived from HLA-A2+ CHB patients were isolated and cultured with anti-CD28, IL-2 and HBcAg18-27 peptide for 10 days in present of anti-PD-L1 or isotype control. Cells were harvested, restimulated and stained by intracellular IFN-γ staining. (A) The representative flow cytometric dot plots of IFN-γ levels in CD4+ and CD8+ T cells. (B) Comparing percentage of IFN-γ producing in CD4 or CD8 T cells treated with anti-PD-L1 or isotype antibody. (C) The ability of IFN-γ production was positively correlated with ICOS while inversely correlated with PD-1 expression on CD4 T cells. IFN-, interferon-; CHB, chronic hepatitis B; IL-2, interleukin-2; PD-L1, PD-Ligand 1.
Effects of blocking PD-1 on the ratio of T-bet to GATA-3 in PBMCs in CHB
Blocking the PD-1:PD-L1 signaling pathway demonstrated a significant impact on the function of CD4+ T cells. The blocking step enhanced their expression levels in IFN-γ, but had no significant effect on the function of the CD8+ T cells. Changes in cell function tend to alter the expression levels of a variety of molecules in a cell; therefore, it is relatively limited to investigate the cell function by measuring certain protein levels. Due to this fact, the effect of blocking the PD-1:PD-L1 signaling pathway on total T-bet and GATA-3 mRNA levels from peripheral PBMCs was investigated.
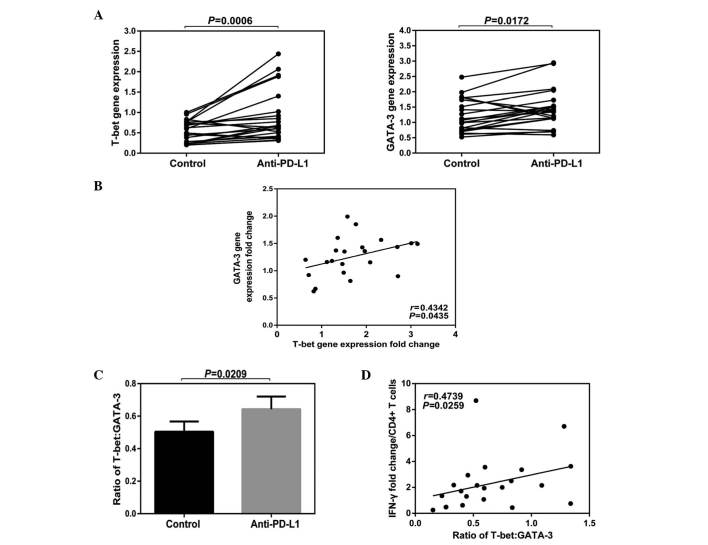
The results of the current study demonstrated that the total gene expression levels of T-bet and GATA-3 in PBMCs were increased following anti-PD-L1 exposure (T-bet Control vs. Anti-PD-L1; P=0.0006; GATA-3 Contol vs. Anti-PD-L1; P=0.0172; Fig. 5A). A positive correlation was demonstrated among the fold-changes of T-bet and GATA-3 (P=0.0435; Fig. 5B) and following anti-PD-L1 exposure, the rate of T-bet/GATA-3 was increased (P=0.0209; Fig. 5C). In addition, a positive correlation was identified between the IFN-γ fold-change and the rate of T-bet/GATA-3 (P=0.0259; Fig. 5D).
Figure 5.
Effects of PD-1 blockade on the production of T-bet and GATA-3 mRNA. Peripheral blood mononuclear cells derived from HLA-A2+ CHB patients were stimulated with HBcAg18-27 peptide for 10 days in the presence of anti-PD-L1 or isotype control. Totol mRNA was purified by TRIzol reagent, and T-bet and GATA-3 were qualified by real time PCR. (A) Expression of T-bet and GATA-3 mRNA in cells treated with anti-PD-L1 or isotype control. (B) Correlation among the gene expression fold changes of T-bet and GATA-3. (C) Ratio of T-bet: GATA-3 following anti-PD-L1 or isotype antibody treatment. (D) Correlation among the ratio of T-bet: GATA-3 and IFN-γ fold changes in CD4 T cells treated by anti-PD-L1 antibody. CHB, chronic hepatitis B; IFN-, interferon; PD-L1, PD-Ligand 1.
Discussion
The functional exhaustion of virus-specific T cells is an important feature of chronic HBV. Although the mechanism of T cell functional exhaustion in patients with CHB remains unclear, the co-stimulatory or co-inhibitory receptors on T cell surfaces vary in type and quantity. These receptors regulate and control T cell function, positively or negatively, in the progression of persistent human viral infections (3). The present study demonstrated that co-signaling molecules of the CD28 family are crucial in positive and negative regulation of T lymphocyte responses (17).
The results of the current study demonstrated that the overall expression status of the CD28 family receptors on peripheral T cells in patients with CHB had distinctive characteristics that were different from those of healthy control subjects. Furthermore, the results demonstrated that the CTLA-4 expression levels of CD4+ and CD8+ T cells, from patients with CHB and healthy controls, were weak, but were increased on global CD4+ T cells from patients with CHB compared with the healthy control subjects. CTLA-4 is expressed at low basal levels on naive T cells with increased expression following T cell activation (5). The present study hypothesized that the increased expression of CTLA-4 may be associated with increased frequencies of CD4+CD25+Foxp3+ regulatory T cells. CTLA-4 was demonstrated to be constitutively expressed on CD4+CD25+Foxp3+ regulatory T cells, which were increased in the peripheral blood of patients with CHB (18,19). The current study demonstrated that in chronic HBV infection, the expression of PD-1 on CD4+ and CD8+ T cells was increased. The level of PD-1 expression on CD8+ T cells in HBeAg-positive patients was significantly higher than that of HBeAg-negative patients, and PD-1 expression on the CD8+ T cells was positively correlated with HBV DNA titers. These data demonstrated that the expression of PD-1 was associated with the chronic HBV infection, and that HBeAg and HBV DNA promoted the expression of PD-1, resulting in injury to T cells. Highly expressed PD-1 is the primary indicator of exhausted T cells. In patients with chronic HBV infection, up-regulation of PD-1 on T cells has frequently been observed (20,21). The reason ICOS expression on CD8+ T cells in patients with CHB was increased compared with that in healthy controls remains unclear, and was negatively correlated with the HBV DNA levels. Previous studies have demonstrated that ICOS is a co-stimulatory molecule, and in HIV infection, ICOS expression on CD4+ and CD8+ T cells were up-regulated and associated with the disease process (22–24). However, previous studies have indicated that ICOS is similar to CD28 in structure and function, thus ICOS provided stimulatory signals to T cells affecting expansion, survival and differentiation (25,26). The results of the present study demonstrated that the BTLA expression on CD8+ T cells in patients with CHB was significantly higher compared with the healthy control subjects, and was positively correlated with the HBV DNA titers. No significant difference was demonstrated in the BTLA expression on CD4+ T cells between patients with CHB and healthy control subjects, which is consistent with the results of Nan et al (27). Previous studies demonstrated that, similar with PD-1 and CTLA-4, BTLA inhibits the activation of T cells, the initialization of CD4+ T cells, and the second reaction of CD4+ and CD8+ T cells (28,29). T cells express a diverse array of co-stimulatory and co-inhibitory receptors during chronic infection (3). It is likely that extrinsic regulation of T cells by a number of suppressive mechanisms may alter the balance of co-stimulatory versus co-inhibitory signals (30). Collectively, the balance of expression between the co-stimulatory and co-inhibitory receptors of the CD28 family may correlate with disease progression, suggesting that the co-expression profiles of these molecules are potential clinical indicators for rapid monitoring of changes in T cell function during CHB treatment.
Following anti-PD-L1 exposure, the expression profile of the CD28 family receptors in T-cells in patients with CHB was altered, and the receptor fold-changes were most prominent in the CD4+ T cells. The results of the present study demonstrated that blocking the PD-1:PD-L1 signaling pathway may enhance the peripheral CD4+ T cell function in patients with CHB. CD4+ T cells are involved in the antiviral response, which occurs primarily through the secretion of cytokines and supports CD8+ T cells. Similarly, virus-specific CD4+ T-cells have been indicated to lose efficacy in chronic viral infections (31). Therefore, the current study speculated that functional improvement of CD4+ T cells in CHB patients may promote functional recovery or reverse the exhausted CD8+ T cells. A previous study demonstrated that CD4+ T-cell activity is important for CD8+ T-cells to establish an effective response in the liver during viral infections (32). These data indicated that PD-1 blockade has a therapeutic potential, which may be enhanced by modulating the expression of co-stimulatory and co-inhibitory receptors of the CD28 family.
Although blocking the PD-1:PD-L1 signaling pathway did not effectively improve the function of the CD8+ T cells, it significantly enhanced the IFN-γ expression in the CD4+ T cells corresponding to the increasing rate of T-bet/GATA-3. T-bet and GATA-3 are transcription factors and T-bet promotes T helper (Th)0 cells to differentiate towards Th1 cells (33). GATA-3 makes Th0 cells differentiate into Th2 cells and the ratio of T-bet/GATA-3 determines the final direction of differentiation (33). T-bet is a member of the T-box transcription factor family, and promotes CD4+ T cells to secrete IFN-γ and induce the differentiation of CD4+ T cells (34). It may be speculated that the PD-1:PD-L1 signaling pathway may result in an increased ratio of T-bet/GATA-3 in CD4+ T cells, thus contributing to the secretion of CD4+ T cell IFN-γ and promoting the differentiation of CD4+ T cells from Th0 to Th1 cells. In addition, the current results demonstrated that the CTLA-4 expression on CD4+ T cells was significantly increased following blocking. Nasta et al (35) demonstrated that during the differentiation of helper T cells, CTLA-4 may inhibit the expression of GATA-3 mRNA, but not inhibit the T-bet mRNA expression. The present study hypothesized that the increased rate of T-bet/GATA-3 may have been due to the increase of the CTLA-4 expression on CD4+ T cells following blocking, which in turn promoted the Th0 cells to differentiate to Th1 cells, leading to improvement of T cell function. However, further research is required to confirm this.
In conclusion, the results of the current study demonstrate that the expression profiles of co-stimulatory and co-inhibitory receptors of the CD28 family serve an important role in chronic HBV infection. In particular, the expression profiles of the CD28 family receptors, and the ratio of T-bet/GATA-3 gene expression were changed when T cell function was enhanced following PD-1 blockade. These results suggested that the balance between these molecules may have an important correlation with the improvement of T cell function induced by PD-1 blockade. However, considering the complexity of correlations among various receptors on CD4+ or CD8+ T cells, additional studies are required to assess whether PD-1 blockade may lead to the possibly life-threatening damage, which may result from biological effects when T cell function is improved by PD-1 blockade. This may aid to better understand the pathogenic mechanism of HBV infection, and provide novel means to assess curative effects, evaluate prognosis, as well as to investigate novel immunological therapeutic strategies in future.
Acknowledgments
The present study would like to thank Professor Haiming Wei (University of Science and Technology of China, Hefei, China) for assisting with the writing of this manuscript. The study was supported by grants from the National Major Science and Technology Project for Infectious Diseases of China (grant nos. 2008ZX10002-011, 2012ZX10004503 and 2013ZX10002001), the International Science & Technology Cooperation Program of China (grant no. 2011DFA31030), the Natural Science Foundation of China (grant nos. 81271884 and 81461130019) and the Deutsche Forschungsgemeinschaft (Transregio TRR60).
References
- 1.Yi JS, Cox MA, Zajac AJ. T-cell exhaustion: Characteristics, causes and conversion. Immunology. 2010;129:474–481. doi: 10.1111/j.1365-2567.2010.03255.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Wherry EJ. T cell exhaustion. Nat Immunol. 2011;12:492–499. doi: 10.1038/ni.2035. [DOI] [PubMed] [Google Scholar]
- 3.Crawford A, Wherry EJ. The diversity of costimulatory and inhibitory receptor pathways and the regulation of antiviral T cell responses. Curr Opin Immunol. 2009;21:179–186. doi: 10.1016/j.coi.2009.01.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Chen L, Flies DB. Molecular mechanisms of T cell co-stimulation and co-inhibition. Nat Rev Immunol. 2013;13:227–242. doi: 10.1038/nri3405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Carreno BM, Carter LL, Collins M. Therapeutic opportunities in the B7/CD28 family of ligands and receptors. Curr Opin Pharmacol. 2005;5:424–430. doi: 10.1016/j.coph.2005.02.003. [DOI] [PubMed] [Google Scholar]
- 6.Anand S, Chen L. Control of autoimmune diseases by the B7-CD28 family molecules. Curr Pharm Des. 2004;10:121–128. doi: 10.2174/1381612043453450. [DOI] [PubMed] [Google Scholar]
- 7.Ma L, Cai YJ, Yu L, Feng JY, Wang J, Li C, Niu JQ, Jiang YF. Treatment with telbivudine positively regulates antiviral immune profiles in Chinese patients with chronic hepatitis B. Antimicrob Agents Chemother. 2013;57:1304–1311. doi: 10.1128/AAC.02181-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Zelinskyy G, Myers L, Dietze KK, Gibbert K, Roggendorf M, Liu J, Lu M, Kraft AR, Teichgräber V, Hasenkrug KJ, Dittmer U. Virus-specific CD8+ T cells upregulate programmed death-1 expression during acute friend retrovirus infection but are highly cytotoxic and control virus replication. J Immunol. 2011;187:3730–3737. doi: 10.4049/jimmunol.1101612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Tassiopoulos K, Landay A, Collier AC, Connick E, Deeks SG, Hunt P, Lewis DE, Wilson C, Bosch R. CD28-negative CD4+ and CD8+ T cells in antiretroviral therapy-naive HIV-infected adults enrolled in adult clinical trials group studies. J Infect Dis. 2012;205:1730–1738. doi: 10.1093/infdis/jis260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Schurich A, Khanna P, Lopes AR, Han KJ, Peppa D, Micco L, Nebbia G, Kennedy PT, Geretti AM, Dusheiko G, Maini MK. Role of the coinhibitory receptor cytotoxic T lymphocyte antigen-4 on apoptosis-Prone CD8 T cells in persistent hepatitis B virus infection. Hepatology. 2011;53:1494–1503. doi: 10.1002/hep.24249. [DOI] [PubMed] [Google Scholar]
- 11.Lopes AR, Kellam P, Das A, Dunn C, Kwan A, Turner J, Peppa D, Gilson RJ, Gehring A, Bertoletti A, Maini MK. Bim-mediated deletion of antigen-specific CD8 T cells in patients unable to control HBV infection. J Clin Invest. 2008;118:1835–1845. doi: 10.1172/JCI33402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Fisicaro P, Valdatta C, Massari M, Loggi E, Biasini E, Sacchelli L, Cavallo MC, Silini EM, Andreone P, Missale G, Ferrari C. Antiviral intrahepatic T-cell responses can be restored by blocking programmed death-1 pathway in chronic hepatitis B. Gastroenterology. 2010;138:682–693. 693.e1–e4. doi: 10.1053/j.gastro.2009.09.052. [DOI] [PubMed] [Google Scholar]
- 13.Schurich A, Pallett LJ, Lubowiecki M, Singh HD, Gill US, Kennedy PT, Nastouli E, Tanwar S, Rosenberg W, Maini MK. The third signal cytokine IL-12 rescues the anti-viral function of exhausted HBV-specific CD8 T cells. PLoS Pathog. 2013;9:e1003208. doi: 10.1371/journal.ppat.1003208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Bengsch B, Martin B, Thimme R. Restoration of HBV-specific CD8+ T-cell function by PD-1 blockade in inactive carrier patients is linked to T-cell differentiation. J Hepatol. 2014;61:1212–1219. doi: 10.1016/j.jhep.2014.07.005. [DOI] [PubMed] [Google Scholar]
- 15.Chinese Society of Hepatology and Chinese Society of Infectious Diseases Chinese Medical Association The guideline of prevention and treatment for chronic hepatitis B (2010 version) Zhonghua Gan Zang Bing Za Zhi. 2011;19:13–24. doi: 10.3760/cma.j.issn.1007-3418.2011.01.007. In Chinese. [DOI] [PubMed] [Google Scholar]
- 16.Bunce M. PCR-sequence-specific primer typing of HLA class I and class II alleles. Methods Mol Biol. 2003;210:143–171. doi: 10.1385/1-59259-291-0:143. [DOI] [PubMed] [Google Scholar]
- 17.Mikami N, Sakaguchi S. CD28 signals the differential control of regulatory T cells and effector T cells. Eur J Immunol. 2014;44:955–957. doi: 10.1002/eji.201444513. [DOI] [PubMed] [Google Scholar]
- 18.Deng M, Li MH, Liu SA, Liu F, Sang Y, Song SJ, Zang SF, Guan XP, Yao X, Wu XP, et al. Studies about the level of CD4+ CD25+ regulatory T cells and relation between expression of Foxp3 and CD127 in peripheral blood of chronic HBV infection. Zhonghua Shi Yan He Lin Chuang Bing Du Xue Za Zhi. 2010;24:21–23. In Chinese. [PubMed] [Google Scholar]
- 19.Sojka DK, Hughson A, Fowell DJ. CTLA-4 is required by CD4+CD25+ Treg to control CD4+ T-cell lymphopenia-induced proliferation. Eur J Immunol. 2009;39:1544–1551. doi: 10.1002/eji.200838603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Li M, Sun XH, Zhu XJ, Jin SG, Zeng ZJ, Zhou ZH, Yu Z, Gao YQ. HBcAg induces PD-1 upregulation on CD4+T cells through activation of JNK, ERK and PI3K/AKT pathways in chronic hepatitis-B-infected patients. Lab Invest. 2012;92:295–304. doi: 10.1038/labinvest.2011.157. [DOI] [PubMed] [Google Scholar]
- 21.Liu XY, Shi F, Zhao H, Wang HF. Research of PD-1 expression in CD8+ T cell of peripheral blood with HBV-associated acute-on-chronic liver failure. Zhonghua Shi Yan He Lin Chuang Bing Du Xue Za Zhi. 2010;24:125–127. In Chinese. [PubMed] [Google Scholar]
- 22.Prendergast A, Klenerman P, Goulder P. Expression of inducible costimulator (ICOS) on T cells is associated with HIV disease progression. Aids Research and Human Retroviruses. 2008;24:130. [Google Scholar]
- 23.Jurado JO, Pasquinelli V, Alvarez IB, Martínez GJ, Laufer N, Sued O, Cahn P, Musella RM, Abbate E, Salomón H, Quiroga M. ICOS, SLAM and PD-1 expression and regulation on T lymphocytes reflect the immune dysregulation in patients with HIV-related illness with pulmonary tuberculosis. J Int AIDS Soc. 2012;15:17428. doi: 10.7448/IAS.15.2.17428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Rudd CE, Taylor A, Schneider H. CD28 and CTLA 4 coreceptor expression and signal transduction. Immuno Rev. 2009;229:12–26. doi: 10.1111/j.1600-065X.2009.00770.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.van Berkel ME, Oosterwegel MA. CD28 and ICOS: Similar or separate costimulators of T cells? Immunol Lett. 2006;105:115–122. doi: 10.1016/j.imlet.2006.02.007. [DOI] [PubMed] [Google Scholar]
- 26.Simpson TR, Quezada SA, Allison JP. Regulation of CD4 T cell activation and effector function by inducible costimulator (ICOS) Curr Opin Immunol. 2010;22:326–332. doi: 10.1016/j.coi.2010.01.001. [DOI] [PubMed] [Google Scholar]
- 27.Nan XP, Zhang Y, Yu HT, Li Y, Sun RL, Wang JP, Bai XF. Circulating CD4+CD25 high regulatory T cells and expression of PD-1 and BTLA on CD4+ T cells in patients with chronic hepatitis B virus infection. Viral Immunol. 2010;23:63–70. doi: 10.1089/vim.2009.0061. [DOI] [PubMed] [Google Scholar]
- 28.Watanabe N, Gavrieli M, Sedy JR, Yang J, Fallarino F, Loftin SK, Hurchla MA, Zimmerman N, Sim J, Zang X, et al. BTLA is a lymphocyte inhibitory receptor with similarities to CTLA-4 and PD-1. Nat Immunol. 2003;4:670–679. doi: 10.1038/ni944. [DOI] [PubMed] [Google Scholar]
- 29.Otsuki N, Kamimura Y, Hashiguchi M, Azuma M. Expression and function of the B and T lymphocyte attenuator (BTLA/CD272) on human T cells. Biochem Biophys Res Commun. 2006;344:1121–1127. doi: 10.1016/j.bbrc.2006.03.242. [DOI] [PubMed] [Google Scholar]
- 30.Maini MK, Schurich A. The molecular basis of the failed immune response in chronic HBV: Therapeutic implications. J Hepatol. 2010;52:616–619. doi: 10.1016/j.jhep.2009.12.017. [DOI] [PubMed] [Google Scholar]
- 31.Brooks DG, Teyton L, Oldstone MB, McGavern DB. Intrinsic functional dysregulation of CD4 T cells occurs rapidly following persistent viral infection. J Virol. 2005;79:10514–10527. doi: 10.1128/JVI.79.16.10514-10527.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Trautmann T, Kozik JH, Carambia A, Richter K, Lischke T, Schwinge D, Mittrücker HW, Lohse AW, Oxenius A, Wiegard C, Herkel J. CD4+ T-cell help is required for effective CD8+ T cell-mediated resolution of acute viral hepatitis in mice. PLoS One. 2014;9:e86348. doi: 10.1371/journal.pone.0086348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Chakir H, Wang H, Lefebvre DE, Webb J, Scott FW. T-bet/GATA-3 ratio as a measure of the Th1/Th2 cytokine profile in mixed cell populations: Predominant role of GATA-3. J Immunol Methods. 2003;278:157–169. doi: 10.1016/S0022-1759(03)00200-X. [DOI] [PubMed] [Google Scholar]
- 34.Szabo SJ, Sullivan BM, Stemmann C, Satoskar AR, Sleckman BP, Glimcher LH. Distinct effects of T-bet in TH1 lineage commitment and IFN-gamma production in CD4 and CD8 T cells. Science. 2002;295:338–342. doi: 10.1126/science.1065543. [DOI] [PubMed] [Google Scholar]
- 35.Nasta F, Ubaldi V, Pace L, Doria G, Pioli C. Cytotoxic T-lymphocyte antigen-4 inhibits GATA-3 but not T-bet mRNA expression during T helper cell differentiation. Immunology. 2006;117:358–367. doi: 10.1111/j.1365-2567.2005.02309.x. [DOI] [PMC free article] [PubMed] [Google Scholar]