Abstract
Anaplastic lymphoma kinase (ALK) is a receptor tyrosine kinase (RTK) associated with alcohol dependence in humans and behavioral responses to ethanol in mice. To characterize the ability of ALK to control ethanol consumption, we treated mice with the ALK inhibitors TAE684 or alectinib before testing them for binge-like drinking using the drinking in the dark (DID) protocol. Mice treated with ALK inhibitors drank less ethanol than controls. In addition, TAE684 treatment abolished ethanol conditioned place preference (CPP), indicating that ALK regulates the rewarding properties of ethanol. Since the ventral tegmental area (VTA) is a key brain region involved in the rewarding effects of ethanol, we determined if Alk expression in the VTA is important for binge-like ethanol consumption. Mice expressing a short hairpin RNA (shRNA) targeting Alk in the VTA drank less ethanol compared to controls. ALK is expressed on dopamine (DA) neurons in the VTA, suggesting that ALK might regulate their firing properties. Extracellular recordings of putative DA neurons in VTA slices demonstrated that ALK inhibition did not affect the ability of ethanol to stimulate, or DA to inhibit, the firing of DA neurons. However, inhibiting ALK attenuated the time-dependent reversal of inhibition produced by moderate concentrations of DA, suggesting that ALK affects DA D2 autoreceptor (D2R) desensitization. Altered desensitization of the D2R changes the firing of DA neurons and is predicted to affect DA levels and alcohol drinking. These data support the possibility that ALK might be a novel target of pharmacotherapy for reducing excessive alcohol consumption.
Keywords: Alcohol addiction, dopamine receptor, ALK, binge-drinking, ventral tegmental area
INTRODUCTION
Binge drinking is a pattern of harmful alcohol use defined by the National Institute on Alcohol Abuse and Alcoholism as drinking an amount of alcohol in a short period of time (2 h) that is sufficient to produce blood alcohol concentrations ≥0.08%. Binge drinking accounts for over half of the approximately 80,000 deaths yearly that are attributed to alcohol use. Binge drinkers also put themselves at increased risk for the development of diseases affecting the cardiovascular system, liver and brain, among others (O'Keefe et al., 2007; Petit et al., 2014; Zakhari and Li, 2007). In the United States, the prevalence of binge drinking among adults is approximately 18% (Kanny et al., 2013) and binge drinking increases the risk of developing alcohol dependence (Bonomo et al., 2004). New pharmacotherapies aimed at reducing binge drinking would be useful in improving health outcomes and preventing alcoholism.
To find new molecular targets for the treatment of alcohol use disorders (AUDs), extensive efforts are underway to identify genes that regulate behavioral responses to ethanol. Variants in the human ALK gene are associated with alcohol dependence, ratings of subjective high after alcohol consumption, and sensitivity to the ataxic effects of alcohol (Lasek et al., 2011b; Wang et al., 2011). In addition, Alk knockout (AlkKO) mice exhibit altered behavioral responses to ethanol (Lasek et al., 2011b). The Alk gene encodes a receptor tyrosine kinase that is expressed throughout the developing and adult brain, with enriched expression in the olfactory bulbs, frontal cortex, striatum, hippocampus, thalamus, and midbrain nuclei (Bilsland et al., 2008; Iwahara et al., 1997; Vernersson et al., 2006). ALK appears to regulate neurogenesis and associative learning, since AlkKO mice show altered neurogenesis and subtle enhancements in spatial learning (Bilsland et al., 2008; Weiss et al., 2012). AlkKO mice also display less anxiety- and depression-like behaviors (Bilsland et al., 2008; Weiss et al., 2012). Finally, inhibition of ALK activity with TAE684 decreases cocaine sensitization and CPP (Lasek et al., 2011a). Together, these studies indicate that ALK is involved in learning and memory processes and behaviors related to psychiatric disorders such as addiction. Given the clinical and preclinical evidence suggesting that ALK regulates behaviors related to alcohol abuse, ALK is a potentially novel therapeutic target for the treatment of AUDs.
Our goal was to determine if the potent ALK inhibitors, TAE684 and alectinib, alter binge-like drinking in mice. We used the DID model to examine the effect of ALK inhibition on binge drinking. In this model, C57BL/6 mice consume intoxicating amounts of alcohol, achieving blood ethanol concentrations in excess of 0.1% (Rhodes et al., 2005). We also tested TAE684 for the ability to regulate the rewarding effects of ethanol using the CPP test, since binge-drinking behavior is linked to heightened sensitivity to ethanol reward (Green and Grahame, 2008; King et al., 2011; King et al., 2014).
For ALK to alter alcohol-related behaviors, it must be involved in alcohol-sensitive pathways important for those behaviors. One such pathway is the mesolimbic DA system, which consists of DA neurons in the VTA that project to the nucleus accumbens (Acb). This system is clearly important for ethanol consumption and reward (Gonzales et al., 2004). ALK acting in the mesolimbic system to regulate alcohol drinking could alter physiological responses to ethanol or interfere with neuronal adaptations upon alcohol exposure. We therefore tested for the effect of ALK inhibition on physiological responses of VTA DA neurons. The present study provides a novel mechanistic link between ALK activity, responses of VTA neurons to DA, and binge drinking.
MATERIALS AND METHODS
Animals
Male C57BL/6J mice were used for the DID test, immunohistochemistry and electrophysiology. Male DBA/2J mice were used for ethanol CPP. All mice were 8 weeks old when purchased from the Jackson Laboratories (Bar Harbor, ME) and 9–12 weeks old during behavioral testing. Mice were group-housed in a temperature- and humidity-controlled environment under a 14 h light/dark cycle (lights on at 6 am and off at 8 pm), unless they underwent the DID test (described below). Animals had access to food and water ad libitum and were maintained and cared for in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals. All procedures performed on animals were approved by the University of Illinois at Chicago (UIC) Animal Care Committee.
DID test
The 4 day ethanol DID test was performed as described previously (Rhodes et al., 2005). Briefly, mice were individually housed in a 12 h reversed light/dark cycle room (lights off at 10 am and on at 10 pm) for 2 weeks prior to behavioral testing. The DID test was performed by replacing the water bottle 3 h into the dark cycle with a single sipper tube containing 20% ethanol in water. On the first 3 days (Monday, Tuesday, and Wednesday), mice were given access to the ethanol solution for 2 h. On the fourth day, mice were given access to the ethanol solution for 4 h and the volume consumed was measured at 2 and 4 h. Blood samples (20 µL) were collected immediately after the 4 h drinking session on day 4 to measure blood ethanol concentrations (BECs). Blood was collected in heparinized capillary tubes via tail vein puncture. BECs were determined using a nicotinamide adenine dinucleotide-alcohol dehydrogenase (NAD-ADH) enzymatic assay (Zapata et al., 2006). To test sucrose consumption, mice underwent a DID test identical to the ethanol consumption test, except the sipper tube contained 2% sucrose in water instead of ethanol.
Ethanol CPP
CPP training and testing were conducted in a modified mouse open field apparatus with 48-channel infrared beam detectors and Activity Monitor software (Med Associates, St. Albans, VT). Modifications of the open field apparatus to create a 2-chambered setup for CPP have been described previously (Hilderbrand and Lasek, 2014). On the 1st day of testing, mice were placed in the CPP apparatus and allowed access to both sides of the chamber for 30 min to measure baseline preference. Mice were then assigned to the non-preferred side for ethanol injections. Mice were conditioned over the next 8 weekdays with an intraperitoneal (i.p.) injection of 2 g/kg ethanol (20% v/v in 0.9% saline) on days 2, 4, 6 & 8 or saline (10 ml/kg) on days 3, 5, 7 & 9. Immediately after injection with ethanol or saline, mice were placed in the apparatus and confined to the appropriate side of the chamber for 5 min. On the 10th day, mice were not treated with any drug and were tested for preference by placing them in the apparatus with access to both sides of the chamber for 30 min. The preference score was calculated as time spent on the ethanol paired side post-conditioning minus pre-conditioning.
ALK inhibitor treatment in vivo
TAE684 and alectinib were purchased from Selleck Chemicals (Houston, TX). TAE684 was administered at a dose of 10 mg/kg in 90% PEG 300, 10% 1-methyl-2-pyrrolidinone vehicle. This dose is effective in inhibiting the growth of ALK-dependent tumors and attenuating behavioral responses to cocaine in mice (Galkin et al., 2007; Lasek et al., 2011a). Pharmacokinetic studies in mice have shown that maximal plasma levels of TAE684 are achieved 7 h after oral administration, the elimination half-life is approximately 12 h, bioavailability is 60–70%, and the brain to plasma ratio is 2:1 (Galkin et al., 2007; Zhang et al., 2012). Alectinib was administered at a dose of 60 mg/kg in 0.02N HCl, 10% DMSO, 10% Cremophor EL, 15% PEG400, and 15% 2-hydroxypropyl-β-cyclodextrin vehicle (Sakamoto et al., 2011). This dose regresses brain tumor xenografts in mice (Kodama et al., 2014). Pharmacokinetic studies in mice have shown that maximal plasma levels of alectinib are achieved approximately 7 h after oral administration, the half-life is approximately 9 h, and bioavailability is approximately 70% (Sakamoto et al., 2011). In rats, alectinib levels peak in the brain at about 8 h (Kodama et al., 2014). Mice were administered compounds or vehicle by oral gavage in a volume of approximately 0.1 mL. TAE684, alectinib, or vehicle was administered 5 h before each drinking session in the DID test (10–12 mice per group) or 5 h before each of the saline and ethanol conditioning sessions in the CPP test (13–17 mice per group). For the control CPP experiment in which we conditioned with TAE684 in the absence of ethanol, TAE684 was administered on days 2, 4, 6 & 8 and vehicle was administered on days 3, 5, 7 & 9, five h before injecting mice i.p. with saline (10 ml/kg) and confining them to one side of the apparatus for 5 min. The timing of ALK inhibitor administration in these experiments was chosen so that maximal plasma or brain levels would be achieved during behavioral testing (Galkin et al., 2007; Sakamoto et al., 2011).
Stereotaxic surgery and lentiviral injections
Replication-deficient lentiviruses expressing shScr or shAlk were produced as described previously (Lasek et al., 2011a). The vector backbone was pLL3.7, which contains shAlk or shScr under the control of the U6 promoter, and EGFP from a CMV promoter (Addgene #59299 and #59297, Cambridge, MA). For stereotaxic viral injections, mice were anesthetized with ketamine/xylazine, placed in a stereotaxic alignment system (David Kopf Instruments) and injected with 1.0 µl of virus solution bilaterally. VTA coordinates were: AP −3.2 mm, ML +/− 0.5 mm, DV −4.7 mm. Viral titers were approximately 3 × 107 pg p24 gag antigen/ml. Mice were allowed to recover for 3 weeks prior to commencing the DID experiment. Viral infection was confirmed by histology after the completion of the DID test. We used 12 mice per group and removed 1 mouse from each group in the analysis due to incorrect localization of viral infection. Acb injections were performed as previously described (Lasek et al., 2011a).
Immunofluorescent staining of brain sections
Mice were euthanized with pentobarbital followed by transcardial perfusion with cold phosphate-buffered saline (PBS) and 4% paraformaldehyde. Brains were processed as described previously (Kharazia et al., 2003). Primary antibodies for GFP and tyrosine hydroxylase (TH) detection were: rabbit anti-TH (Millipore, Temecula, CA) and mouse anti-GFP (Life Technologies, Carlsbad, CA). Primary antibodies for TH and ALK detection were: mouse anti-TH (Millipore) and rabbit anti-ALK, #3815 (Weiss et al., 2012). Images of lentiviral infection in the VTA were captured using an EVOS fl inverted fluorescence microscope with a 4× objective lens (Life Technologies) and images of ALK and TH staining in the VTA were captured using a Zeiss LSM 710 confocal microscope (Carl Zeiss, Thornwood, NY). For quantification of knockdown of ALK in the VTA after lentiviral infection, the intensity of ALK staining in the infected VTA was quantified from 2–3 sections per mouse and 9 mice per group (shScr, shAlk) three weeks after lentiviral infection. Intensity was measured using the National Institutes of Health ImageJ software.
Preparation of brain slices for electrophysiology
Brain slices containing the VTA were prepared as described (Brodie et al., 1999). Coronal sections (400 µm thick) were cut on a vibratome in chilled cutting solution, then placed in artificial cerebrospinal fluid (aCSF, 35°C) and maintained for at least one hour before recording. Recordings were made up to 6 h after the slices were placed in the recording chamber. The composition of the aCSF in these experiments was (in mM): NaCl 126, KCl 2.5, NaH2PO4 1.24, CaCl2 2.4, MgSO4 1.3, NaHCO3 26, glucose 11. The composition of the cutting solution was (in mM): KCl 2.5, CaCl2 2.4, MgSO4 1.3, NaHCO3 26, glucose 11, and sucrose 220. Both solutions were saturated with 95% O2/5% CO2 (pH=7.4).
Extracellular recording
Extracellular recording electrodes were made from 1.5 mm diameter glass tubing with filament and were filled with 0.9% NaCl (tip resistance 2 – 5 MΩ). Firing rate was determined before and during drug application. Firing rate was calculated over 1 min intervals during the experiments; peak drug-induced changes in firing rate were expressed as the percentage change from the control firing rate. For the effects of ALK inhibitors on ethanol concentration-response curves, ethanol (40, 80, 120 mM) was tested before and after addition of the ALK inhibitor (100 nM TAE684 or alectinib in DMSO) to the extracellular medium. For the effects of ALK inhibitors on responses to acute DA, short application (5 min) of single concentrations of DA (1, 2, 5, or 10 µM) were added to slices in the presence of 0.1 % DMSO (control), TAE684, or alectinib. A washout period of at least 10 min was used between concentrations to permit the firing rate to return to baseline. To test reversal of DA inhibition (DIR, or desensitization), neurons of the VTA were incubated with DA for 40 min and firing was measured every 5 min over the 40 min period. DA concentrations were adjusted for each neuron so that inhibition at 5 min was between 50% and 95% (Nimitvilai and Brodie, 2010). This method controlled for differences in sensitivity between neurons, but also sometimes resulted in the mean concentrations of DA slightly differing between groups.
Statistical analysis
All data were expressed as the mean ± SEM and analyzed using Prism software (GraphPad, La Jolla, CA). DID data from the 2 h drinking sessions over 4 days was analyzed by 2 way repeated measures (RM) ANOVA for treatment and time. The 4 h DID, BEC and ethanol preference scores from CPP were analyzed using a Student’s t-test. Electrophysiology data were analyzed using a 2 way RM ANOVA with post-hoc Tukey’s multiple comparisons tests.
RESULTS
ALK inhibition attenuates binge-like ethanol consumption
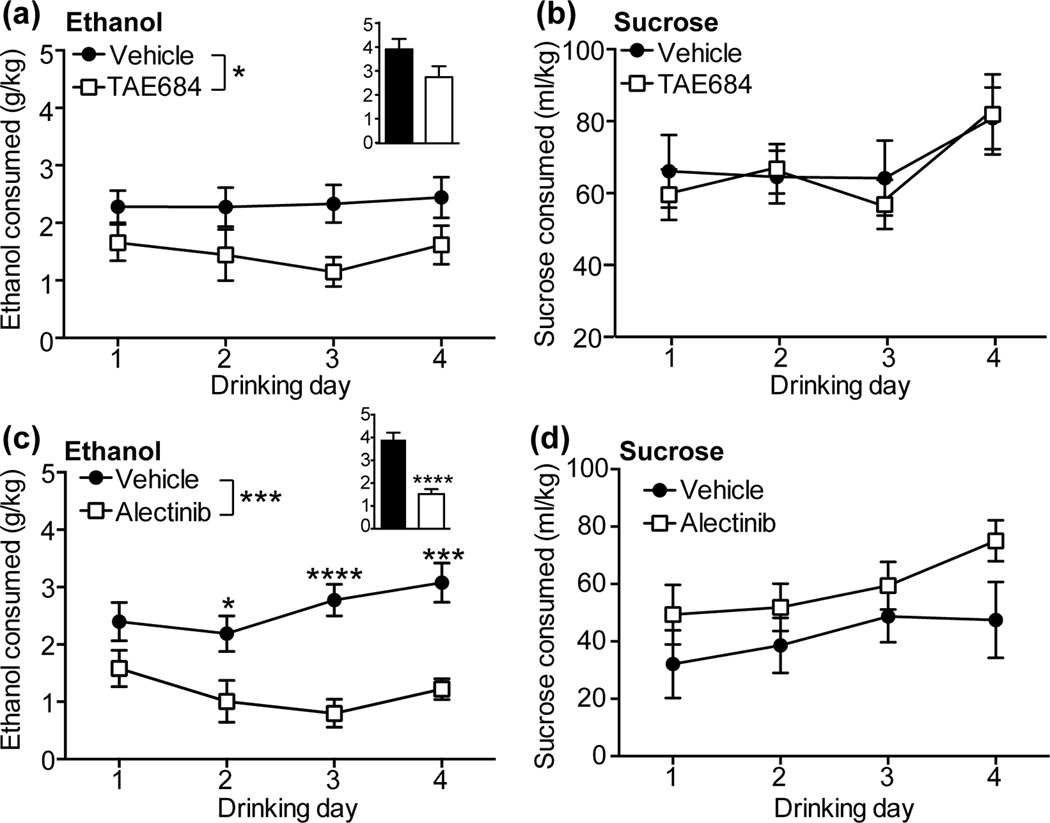
To examine how ALK inhibition might affect binge-like ethanol consumption, we treated mice with the ALK inhibitor TAE684 and found that they consumed significantly less ethanol during the 2 h drinking sessions over the 4 day DID experiment when compared to mice treated with vehicle (Fig. 1a; 2 way RM ANOVA, treatment [F(1,22) = 5.33, P = 0.031]). Mice treated with TAE684 consumed 37% less ethanol overall when compared to mice treated with vehicle. We also examined ethanol intake after 4 h of drinking on day 4 and observed a trend towards decreased ethanol consumption (a 31% reduction) [t(22) = 1.87, P = 0.076]. Finally, we measured BECs after the 4 h drinking session. Consistent with decreased ethanol consumption in mice treated with TAE684, BECs were lower in mice treated with TAE684 (vehicle, 62.9 ± 29.7 mg%; TAE684, 2.4 ± 2.4 mg%, [t(10) = 2.02, P = 0.07]). Treatment of mice with TAE684 did not affect sucrose drinking (Fig. 1b). We performed additional experiments to determine if TAE684 alters the acute sedative response to ethanol or ethanol clearance from the blood. TAE684 did not affect recovery time from the sedative effect of ethanol in the ethanol-induced loss of the righting reflex test, nor did TAE684 alter ethanol clearance from the blood (Fig. S1). Together, these data suggest that inhibition of ALK affects binge-like ethanol drinking without altering ethanol metabolism or the consumption of natural rewards such as sucrose.
Figure 1.
ALK inhibition attenuates binge-like ethanol consumption. (a) Ethanol consumed (g/kg) during 2 h of drinking on days 1–4 in mice treated with vehicle (n=12) or 10 mg/kg TAE684 (n=12). TAE684 treatment led to an average 37% decrease in ethanol consumption compared to vehicle treatment over 4 days. Inset bar graph shows the amount of ethanol consumed during 4 h on day 4. (b) Volume of 2% sucrose solution (ml/kg) consumed during 2 h of drinking on days 1–3 and 4 h on day 4 in mice treated with vehicle (n=10) or TAE684 (n=10). (c) Ethanol consumed during 2 h of drinking on days 1–4 in mice treated with vehicle (n=10) or 60 mg/kg alectinib (n=10). Alectinib treatment led to an average 61% decrease in ethanol consumption over 4 days. Inset bar graph shows the amount of ethanol consumed during 4 h on day 4. (d) Volume of 2% sucrose consumed during 2 h of drinking on days 1–3 and 4 h on day 4 in mice treated with vehicle (n=5) or alectinib (n=5). *p < 0.05, ***p < 0.001, ****p < 0.0001 by 2 way ANOVA or t-test.
To extend these findings to a new clinically available ALK inhibitor, we treated mice with alectinib and found that they consumed significantly less ethanol over the 4 days of DID testing compared to vehicle-treated mice (Fig. 1c; 2 way RM ANOVA, treatment [F(1,18) = 19.3, P = 0.0004], time [F(3,54) = 2.44, P = 0.074], treatment×time interaction [F(3,54) = 3.21, P = 0.03]). Overall, mice treated with alectinib consumed 56% less ethanol over the four 2 h drinking sessions when compared to vehicle-treated mice. Post-hoc Holm-Sidak’s multiple comparisons test demonstrated a significant difference between vehicle- and alectinib-treated mice on days 2, 3 and 4. Ethanol consumption was also significantly reduced by 61% in alectinib-treated mice after 4 h of drinking on day 4 [t(18) = 6.25, P < 0.0001]. Similar to what we observed in mice treated with TAE684, alectinib did not affect consumption of 2% sucrose (Fig. 1d). We also examined two-bottle choice drinking in mice treated with alectinib to determine if ALK inhibition alters ethanol preference. Mice were given the choice between sipper tubes filled with water or 20% ethanol in a 4 day DID experiment. We found that mice treated with alectinib drank significantly less ethanol in this procedure, similar to what we observed in the one bottle DID experiment (Fig. S2). The overall reduction in ethanol consumption over 4 days was 78%. Importantly, mice treated with alectinib demonstrated significantly reduced preference for ethanol (Fig. S2, a 75% reduction overall), indicating that alectinib did not affect general fluid consumption, but specifically reduced ethanol drinking. Collectively, these data indicate that ALK inhibitors reduce ethanol consumption without affecting sucrose consumption, ethanol-induced sedation, or ethanol clearance.
ALK inhibition attenuates ethanol reward
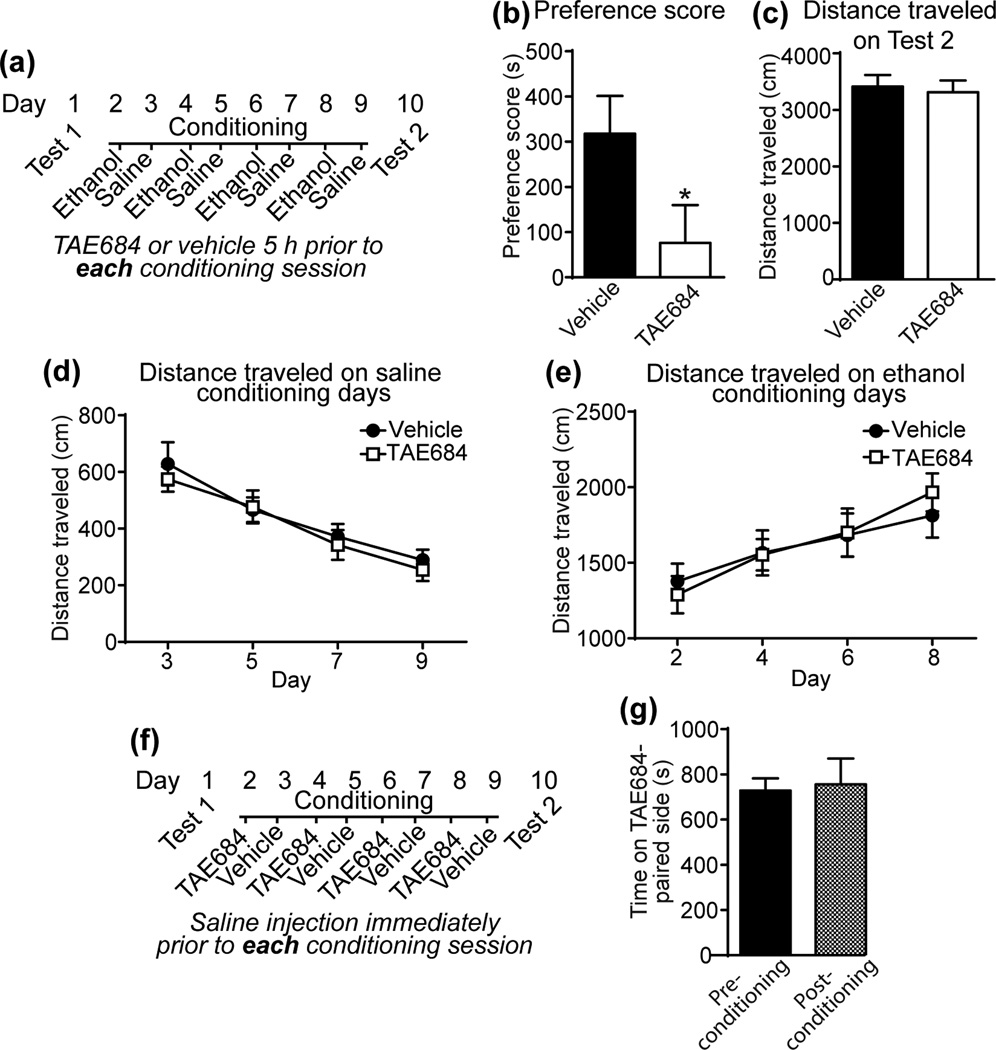
The reduced ethanol consumption in mice treated with ALK inhibitors suggests that ALK might be involved in the rewarding properties of ethanol. To test this, we performed ethanol CPP in DBA/2J mice treated with TAE684 or vehicle. DBA/2J mice were chosen because this inbred mouse strain develops robust CPP for ethanol (Cunningham et al., 1992). DBA/2J mice were treated with TAE684 or vehicle daily prior to ethanol and saline conditioning sessions (Fig. 2a). TAE684 treatment decreased the ethanol place preference score by 76% (Fig. 2b, [t(27) = 2.02, p = 0.053]). Prior TAE684 treatment had no effect on the distance traveled on test day (Fig. 2c). We also analyzed the locomotor activity data from the saline and ethanol conditioning days. TAE684 treatment did not affect the distance traveled in mice injected with saline or ethanol (Fig. 2d, e). Interestingly, we found that DBA/2J mice sensitized to the locomotor-stimulating effect of ethanol during the conditioning procedure, as reported previously (Phillips et al., 1994), but mice treated with TAE684 sensitized to the same extent as vehicle-treated mice (Fig. 2e; 2 way RM ANOVA, time [F(3, 72) = 24.78, P < 0.0001]). To test if TAE684 is rewarding or aversive on its own (i.e., in the absence of ethanol), we performed a control experiment in which mice were place-conditioned with TAE684 (Fig. 2f). Conditioning with TAE684 did not alter the time spent on the TAE684-paired side of the apparatus, indicating that ALK inhibition in the absence of ethanol does not induce preference or aversion (Fig. 2g). These data suggest that ALK inhibition attenuates the rewarding properties of ethanol without affecting ethanol-induced locomotor sensitization.
Figure 2.
ALK inhibition attenuates ethanol reward. (a) Schematic showing treatment conditions for the ethanol CPP test. (b) Preference score after ethanol conditioning in mice treated with vehicle (n=16) or 10 mg/kg TAE684 (n=13), showing that TAE684 significantly decreased ethanol CPP by 76%, *p = 0.05 by t-test. (c) Distance traveled (cm) during the 30 min preference test. (d) Distance traveled in 5 min immediately after a saline injection on conditioning days 3, 5, 7 & 9. (e) Distance traveled in 5 min after a 2 g/kg ethanol injection on conditioning days 2, 4, 6 & 8. Note that the mice sensitized to the ethanol injections, but there was no effect of TAE684 treatment on ethanol sensitization. (f) Schematic showing the treatment conditions for the TAE684 CPP test (in the absence of ethanol). (g) Time (s) spent on the TAE684-paired side before conditioning (pre-conditioning) and after conditioning (post-conditioning) in mice conditioned with 10 mg/kg TAE684 (n=11) instead of ethanol. Time spent on the TAE684-paired side did not change after conditioning.
ALK is expressed in DA neurons and in non-DA cells in the VTA
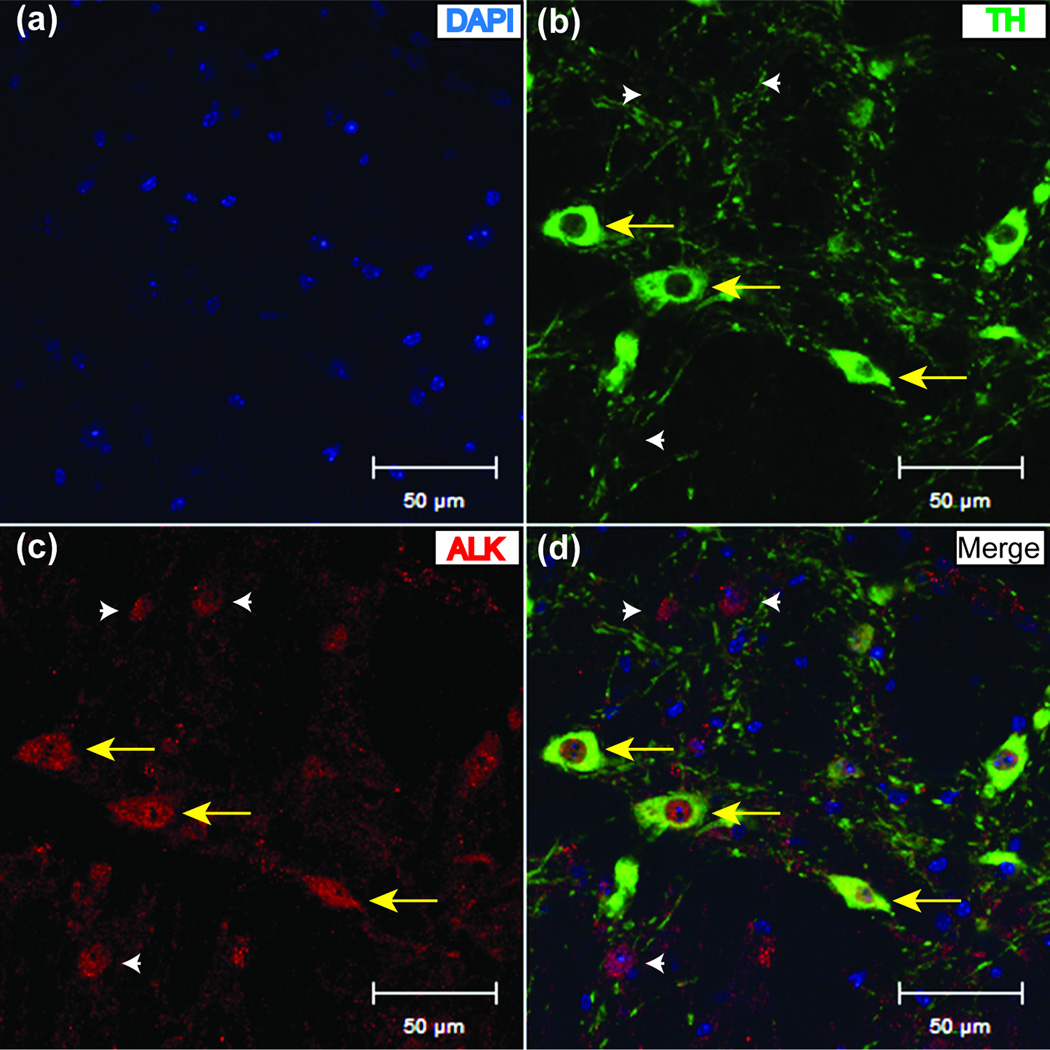
The VTA mediates the rewarding properties of ethanol, and several groups have demonstrated that the VTA plays a role in binge-like ethanol consumption in mice (Gonzales et al., 2004). As a result, the VTA is one region where ALK might act to control ethanol consumption. We analyzed ALK expression in the VTA using immunohistochemistry. Antibodies to ALK and TH (an enzyme involved in the biosynthesis of DA) were incubated on naïve mouse brain slices containing the VTA and detected using fluorescent secondary antibodies and confocal microscopy. ALK immunoreactivity was evident in the soma and processes of several cell types in the VTA (Fig. 3), including cells expressing TH, indicating that ALK is expressed in DA neurons in the VTA. However, ALK staining was not restricted to DA neurons. The presence of ALK in DA neurons and non-DA cells in the VTA supports a role for ALK in modulating VTA activity.
Figure 3.
ALK is expressed in DA neurons and in non-DA cells in the VTA. Confocal microscope images showing (a) nuclear (DAPI) staining in blue, (b) immunofluorescence to tyrosine hydroxylase (TH) in green, (c) immunofluorescence to ALK in red, and (d) a merged image of DAPI, TH and ALK immunofluorescence. Yellow arrows point to TH neurons expressing ALK and white arrowheads indicate expression of ALK in non-TH expressing cells. Scalebar, 50 µm.
ALK expression in the VTA regulates binge-like ethanol consumption
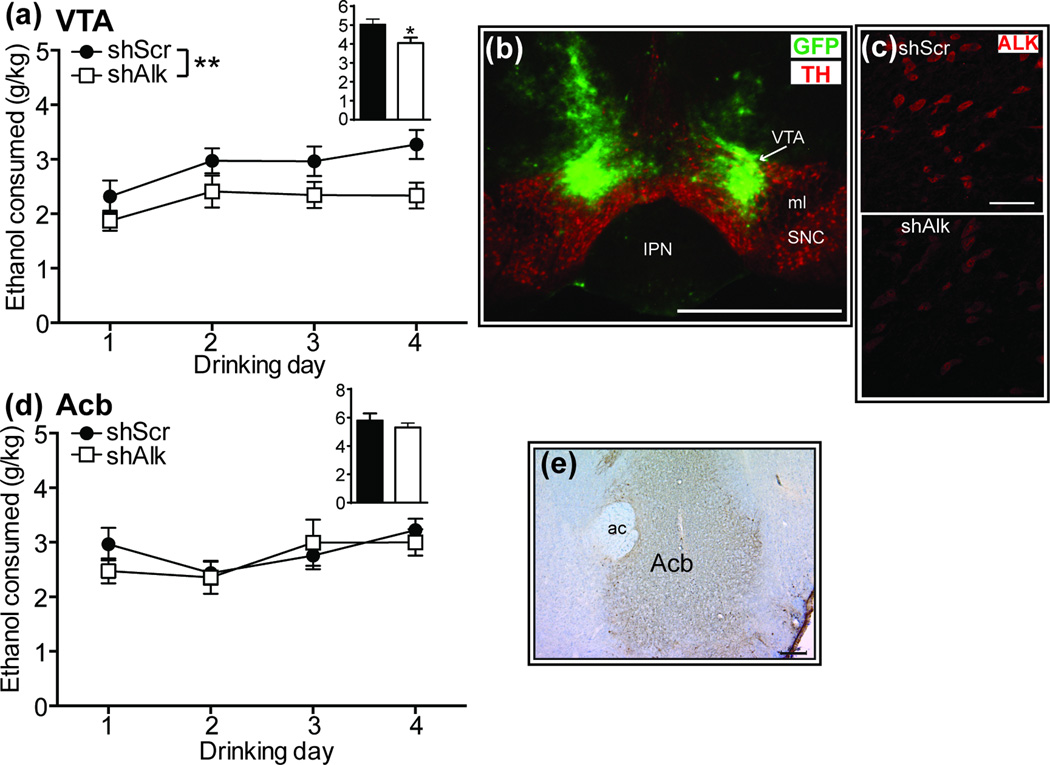
To determine if ALK functions in the VTA to affect alcohol drinking, we employed a lentiviral-delivered shRNA targeting Alk (shAlk) to site-specifically knockdown Alk expression using an shRNA that we previously validated to reduce Alk mRNA in the Acb by 30% (Lasek et al., 2011a). Mice underwent stereotaxic surgery and were injected in the VTA with lentivirus expressing shAlk or a control shRNA (shScr). Mice expressing shAlk in the VTA consumed less ethanol during the 2 h drinking sessions over the 4 day DID test compared to mice expressing shScr (Fig. 4a; 2 way RM ANOVA, shRNA [F(1, 20) = 8.68, P = 0.008], time [F(3, 60) = 3.71, P = 0.016]). Ethanol consumption was also significantly reduced during the 4 h drinking session on day 4 [t(20) = 2.51, P = 0.021]. Consistent with reduced ethanol consumption on day 4, BECs were decreased in mice expressing shAlk compared to shScr (shScr, 178.8 ± 22.8 mg%; shAlk, 82.3 ± 26.9 mg% [t(20) = 2.74, P = 0.013]). After the completion of the DID test, targeting of lentivirus to the VTA was verified by immunohistochemistry using antibodies to TH and green fluorescent protein (GFP), which is expressed by the lentivirus (Fig. 4b). In a separate group of animals, we analyzed ALK expression by immunohistochemistry in infected VTA to determine the effectiveness of knockdown by shAlk (Fig. 4c). ALK expression was quantified from confocal images using ImageJ. The intensity of ALK staining was reduced by 55% in mice expressing shAlk compared to shScr (shScr, 100 ± 14; shAlk, 45 ± 7.4 [t(39) = 3.41, P = 0.0015]). These results indicate that ALK acts in the VTA to regulate binge-like ethanol consumption.
Figure 4.
ALK expression in the VTA regulates binge-like ethanol consumption. (a) Ethanol consumed (g/kg) during 2 h drinking sessions on days 1–4 in mice infected with lentivirus expressing shScr (control, n=11) or shAlk (n=11) in the VTA. Inset bar graph shows the amount of ethanol consumed during the 4 h drinking session on day 4. Mice expressing shAlk consumed approximately 22% less ethanol than controls on average over 4 days. *p < 0.05 and **p < 0.01 by 2 way ANOVA. (b) Fluorescent image demonstrating lentiviral infection in the VTA. Mouse brain sections containing infected VTA were incubated with fluorescent antibodies to tyrosine hydroxylase (TH, red) to indicate DA neurons and green fluorescent protein (GFP, green), expressed by the lentivirus. Abbreviations: IPN, interpeduncular nucleus; ml, medial lemniscus; SNC, substantia nigra pars compacta. Scalebar, 1 mm. (c) Fluorescent images showing ALK expression in VTA sections infected with shScr (top panel) or shAlk lentiviruses (bottom panel). Confocal images were taken with identical laser settings. Note the reduced intensity of ALK immunostaining in VTA infected with shAlk compared to shScr. Scalebar, 50 µm. (d) Ethanol consumed during 2 h drinking sessions on days 1–4 in mice infected with lentivirus expressing shScr (n=12) or shAlk (n=12) in the nucleus accumbens (Acb). Inset bar graph shows the amount of ethanol consumed during the 4 h drinking session on day 4. (e) Lentiviral infection in the Acb. GFP staining is shown in brown. Sections were counterstained with Cresyl violet. ac, anterior commissure.
The Acb receives DA inputs from the VTA and also mediates binge-like ethanol consumption in mice (Cozzoli et al., 2015; Kasten and Boehm, 2014). We previously found that knockdown of Alk in the Acb decreases cocaine sensitization (Lasek et al., 2011a), so it seemed feasible that ALK might function in the Acb to regulate ethanol consumption. To test this, mice were injected in the Acb with lentivirus expressing shAlk or shScr and tested in the DID procedure. Mice that received shAlk and shScr lentiviruses consumed the same amount of ethanol, despite robust viral infection in the Acb (Fig. 4d, e). Together, these results indicate that ALK functions in the VTA, but not the Acb, to regulate binge-like ethanol consumption.
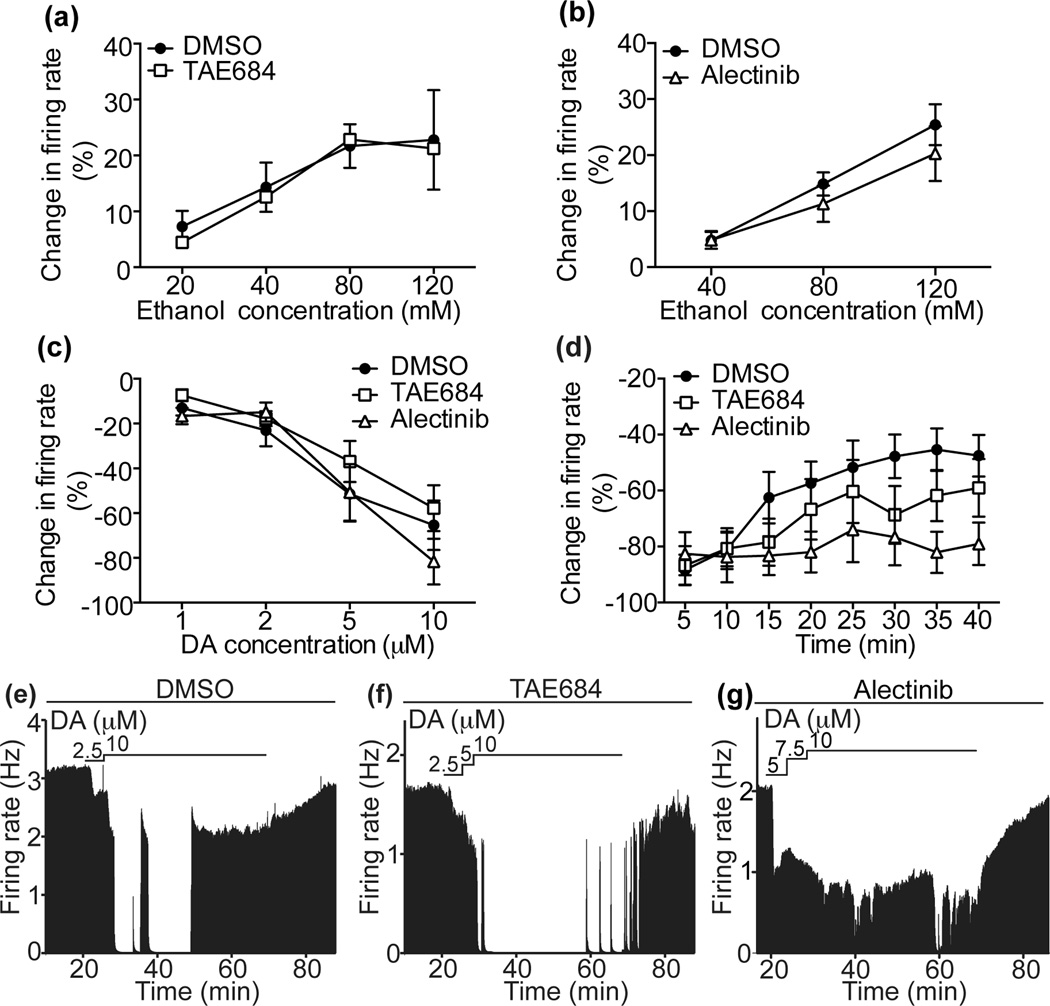
ALK inhibitors attenuate DIR in DA neurons
Since downregulation of ALK in the VTA decreases binge-like drinking and ALK is expressed in DA neurons, we hypothesized that pharmacological inhibition of ALK alters the physiology of VTA DA neurons. Neurons within the VTA were identified as DA according to electrophysiological criteria and sensitivity to DA, as we have reported (Nimitvilai et al., 2012a). We first examined the response of VTA DA neurons to ethanol in the presence of TAE684 or alectinib, since we previously showed that ethanol administered acutely to slices increases the firing rate of DA neurons in the VTA (Brodie et al., 1990). Treatment with TAE684 or alectinib did not change the ethanol-induced excitation of VTA DA neurons compared to controls (Fig. 5a, b). We next examined the effect of ALK inhibitors on the inhibitory response to DA. DA inhibits the firing of VTA DA neurons due to the action of DA at the D2R autoreceptor (Lacey et al., 1987). Acute inhibition of VTA DA neurons by DA was not altered in the presence of TAE684 or alectinib (Fig. 5c). These data indicate that inhibition of ALK does not change the acute response of VTA DA neurons to stimulation by ethanol or inhibition by DA.
Figure 5.
ALK inhibitors attenuate DIR but not the response of DA neurons to acutely administered ethanol or DA. Extracellular recordings of VTA DA neurons were performed in the presence or absence of ALK inhibitors or the DMSO control. (a) Ethanol dose-response curves, showing that ethanol-induced stimulation of DA neuron firing is unaffected by incubation of slices with 100 nM TAE684 (n=10) compared to DMSO controls (n=10). (b) Ethanol dose-response curves, showing that ethanol-induced stimulation of DA neuron firing is unaffected by incubation of slices with 100 nM alectinib (n=11) compared to DMSO controls (n=11). (c) DA dose-response curves, showing that inhibition of DA neurons by DA is unaffected by incubation of slices with TAE684 (open squares, n=9) or alectinib (open triangles, n=8) compared to DMSO controls (solid circles, n=9). (d) Percent change in firing rate over 40 min of exposure to DA in cells treated with DMSO (filled circles, n=9), TAE684 (open squares, n=10), or alectinib (open triangles, n=6). Change in firing rate at 5 min intervals is plotted as a function of time after the initiation of DA administration. DIR was delayed in TAE684-treated and abolished in alectinib-treated slices compared to DMSO-treated controls. (e–g) Representative ratemeter graphs from single neurons showing the effect of DMSO (e), TAE684 (f), or alectinib (g) treatment on DA inhibition. Vertical bars indicate the firing rate over 5 second intervals. Horizontal bars indicate the duration of drug application.
Inhibition of DA neurons by DA subsides over time in the continued presence of DA due to desensitization of the D2R, a process we have termed DA inhibition reversal (DIR) (Nimitvilai et al., 2012a; Nimitvilai and Brodie, 2010; Nimitvilai et al., 2012c). We assessed DIR in VTA slices treated with TAE684, alectinib, or vehicle by examining the DA-induced reduction in spontaneous firing rate at 5 min intervals over a 40 min period (Fig. 5d–g). The concentration of DA administered to control neurons was 6.5 ± 1.4 µM, the concentration of DA administered to neurons in the presence of TAE684 was 7.9 ± 1.5 µM, and the concentration of DA administered to neurons in the presence of alectinib was 6.75 ± 1.3 µM. After 5 min of exposure, DA produced an initial inhibition of −88.4 ± 5.4% in controls, −86.8 ± 6.9% in TAE684-treated, and −82.5 ± 7.7% in alectinib-treated slices. In control slices, DA neuron firing rate recovered to −47.5 ± 7.4% by 40 min, indicating DIR. In contrast, in TAE684-treated slices, the firing rate at 40 min was −59.0 ± 10.3%, and in alectinib-treated slices, the firing rate at 40 min was −79.0 ± 7.6% (2 way ANOVA, treatment [F(2, 22) = 1.90, P = 0.173], time [F(7, 154) = 7.0, P < 0.0001], treatment×time interaction [F(14, 154) = 1.42, p = 0.149]). Closer examination of the data using Tukey’s multiple comparisons test showed that the effect of time was driven primarily by DIR in the control slices. There were significant differences between the 5 min time point and the 15 (P = 0.041), 20 (P = 0.005), 25 (P = 0.0004), 30, 35, and 40 min time points (P < 0.0001) in the controls. In the TAE684-treated slices, there were significant differences between the 5 and 25 (P = 0.02), 35 (P = 0.035), and 40 (P = 0.012) min time points, suggesting a delayed DIR. In contrast, there were no significant differences over time in the alectinib-treated slices, indicating a complete antagonism of DIR. These results indicate that ALK inhibitors can delay or disrupt D2R desensitization in VTA DA neurons.
DISCUSSION
Our results indicate that ALK regulates binge-like drinking and the rewarding properties of ethanol in mice. Polymorphisms in the human ALK gene are associated with alcohol dependence and measures of subjective high and motor-incoordination following alcohol intake (Lasek et al., 2011b; Wang et al., 2011), suggesting that ALK may be a viable therapeutic target for the treatment of AUDs. Here, we tested two ALK inhibitors, TAE684 and alectinib. We found that both inhibitors decrease binge-like drinking in the DID test. Alectinib appears to be more effective than TAE684 in reducing ethanol consumption (Fig. 1). This could be due to differences in the doses we used (10 mg/kg TAE684 versus 60 mg/kg alectinib). We chose these doses based on published reports that they cause tumor regression in mouse models, including brain tumors in the case of alectinib (Galkin et al., 2007; Kodama et al., 2014; Sakamoto et al., 2011). We also previously showed that 10 mg/kg TAE684 attenuated cocaine sensitization and CPP in mice (Lasek et al., 2011a). We have not tried higher doses of TAE684 due to concerns about toxicity, but it would be interesting to know if higher doses of TAE684 are as effective as alectinib in reducing ethanol consumption. Alectinib and TAE684 have similar oral bioavailability (approximately 71% for alectinib and 60–70% for TAE684). However, alectinib may be more selective for ALK than TAE684 (Davis et al., 2011; Galkin et al., 2007; Sakamoto et al., 2011; Zhang et al., 2012) and is currently approved in Japan for the treatment of non-small cell lung cancer and is being tested in clinical trials in the United States. Although all kinase inhibitors potentially have off-target effects by inhibiting multiple kinases, we believe that the reduction in ethanol consumption is due to inhibition of ALK, since alectinib and TAE684 are in different chemical classes. In addition, we have demonstrated that an shRNA targeting ALK also reduces ethanol consumption. Due to its selectivity, potency, ability to penetrate the blood-brain barrier, and positive clinical development, alectinib is a potential new compound that may be useful for the treatment of alcohol use disorders.
In our DID experiments, there was a discrepancy between the amount of ethanol consumed and BECs in the DID test between mice that were treated by gavage versus infected with lentiviral-delivered shRNAs in the VTA. Control mice treated with vehicle in the inhibitor studies (by oral gavage) consumed 20% less ethanol compared to mice treated with the control shRNA in the VTA. This is probably due to the acute stress of the gavage procedure. However, the BECs in the control gavage group were half that of the shRNA control group. This may be due to the episodic nature of drinking in the DID test. Wilcox et al have shown that mice consume nearly half of their ethanol in the DID test in the first 30 minutes of the drinking session and that BECs correlate better with the rate of drinking during the early part of the drinking session (Wilcox et al., 2014). It is possible that the kinetics of drinking in the DID test are altered in mice treated by gavage. Moreover, it must be taken into consideration that alcohol is metabolized during the drinking session and can affect BECs, which are obtained at the end of the 4 h session. Others have also found a large discrepancy between the magnitude of changes in ethanol consumption and BECs (Lowery et al., 2010; Melon and Boehm, 2011).
One brain region in which ALK appears to regulate alcohol consumption is the VTA. We found that knockdown of Alk in the VTA reduces binge-like drinking in the DID test. In contrast, we previously demonstrated that AlkKO mice drink more ethanol in a limited, intermittent-access binge drinking test (Lasek et al., 2011b). Increased drinking in AlkKO mice may be due to compensatory effects of eliminating ALK during development. In support of this, we found that phosphorylation of MEK (an upstream regulator of ERK signaling) is increased in the brains of AlkKO mice (Lasek et al., 2011b). Since ALK activates ERK (Hallberg and Palmer, 2013), increased MEK phosphorylation in AlkKO mice suggests that compensatory changes have occurred. Over-activation of ERK is associated with increased ethanol consumption. Selectively bred mice with high ethanol preference over-express several genes in the ERK pathway (Mulligan et al., 2006). The difference between phenotypes observed with ALK suppression in adults and in AlkKO mice indicates significant compensatory developmental mechanisms. Interestingly, we found anatomical specificity with regard to the regulation of ethanol consumption by ALK. Knockdown of Alk in the Acb was ineffective in reducing ethanol intake, despite robust infection in the Acb and our demonstration previously that this same lentiviral construct injected into the Acb reduces cocaine sensitization (Lasek et al., 2011a). ALK therefore appears to be important for ethanol consumption through its activity in the VTA and for behavioral responses to cocaine through its activity in the Acb. Although ALK inhibition attenuates cocaine sensitization, it does not appear to affect ethanol sensitization (Fig. 2), indicating a molecular difference between these two drugs with regard to sensitization.
Two potential ligands for ALK that may be relevant for ALK in promoting ethanol consumption are the growth factors pleiotrophin and midkine (Fig. 6) (Stoica et al., 2001; Stoica et al., 2002). Midkine expression is higher in the prefrontal cortex of human alcoholics and mice selectively bred for high alcohol consumption (Flatscher-Bader and Wilce, 2008; Mulligan et al., 2006) and pleiotrophin expression is regulated by ethanol (Vicente-Rodriguez et al., 2014a). In addition, both factors regulate behavioral responses to ethanol, including CPP (Vicente-Rodriguez et al., 2014a; Vicente-Rodriguez et al., 2014b). We found that ethanol activates ALK through midkine (He et al., 2015), suggesting that midkine might be a viable ligand for ALK. However, there is controversy as to whether midkine and pleiotrophin are true in vivo ligands for ALK. Pleiotrophin and midkine are also ligands for a phosphatase, RPTPβ/ζ, that regulates ALK activity (Maeda et al., 1999; Meng et al., 2000; Perez-Pinera et al., 2007). In addition, other ligands for ALK have been described, including FAM150A, FAM150B, and heparin (Guan et al., 2015; Murray et al., 2015).
Figure 6.
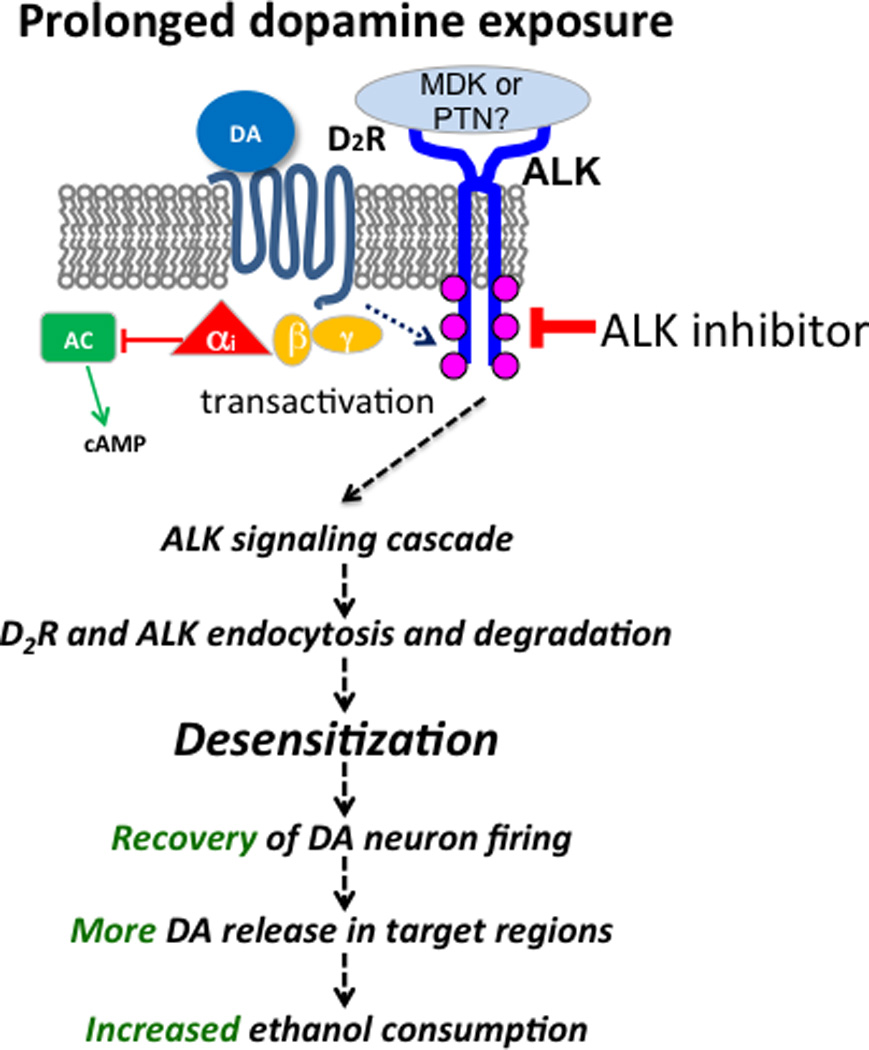
Model for regulation of D2R desensitization by ALK and predicted effect on ethanol consumption. Prolonged DA exposure leads to desensitization of the D2R through transactivation of ALK signaling, presumably resulting in endocytosis and degradation of ALK and D2R. This allows for the recovery of DA neuron firing. The prediction is increased DA release in target regions, leading to an increase in ethanol consumption. ALK inhibitors such as TAE684 and alectinib are predicted to block the process of ALK and D2R endocytosis and degradation, resulting in attenuated DA release and ethanol consumption. ALK ligands such as midkine (MDK) or pleiotrophin (PTN) may participate in this process.
Our results provide one potential mechanism by which ALK regulates binge drinking and ethanol reward. ALK alters the physiology of VTA DA neurons by promoting desensitization of D2R. We found that inhibition of ALK decreased desensitization but did not alter ethanol-stimulated firing or the initial inhibition of firing by DA in VTA DA neurons. Regulation of D2R desensitization is a completely novel function for ALK. A proposed model for this is illustrated in Fig. 6. ALK may facilitate the endocytosis and/or trafficking of D2R after prolonged DA exposure, but does not change the acute inhibitory response to DA. RTKs (such as ALK) are trans-activated by stimulation of GPCRs such as D2R (Cattaneo et al., 2014; Delcourt et al., 2007). For instance, stimulation of D2R by quinpirole activates the RTKs EGFR and PDGFR (Kotecha et al., 2002; Wang et al., 2005). We hypothesize that long-term DA stimulation of D2R may trans-activate ALK signaling, leading to internalization and degradation of complexes containing both D2R and ALK. Activation of ALK by an agonist antibody causes ALK internalization and degradation, while inhibition of ALK activity by TAE684 blocks this process (Mazot et al., 2012). Similarly, D2R is internalized and degraded after agonist stimulation (Bartlett et al., 2005). Cross talk between GPCRs and RTKs can promote receptor internalization (Gavi et al., 2007). We have shown that D2R desensitization in VTA DA neurons is mediated by the phosphatidylinositol system, protein kinase C and the endocytic protein dynamin (Nimitvilai et al., 2012a; Nimitvilai et al., 2012c; Nimitvilai et al., 2013). ALK can activate phospholipase C-γ, an upstream activator of PKC (Bai et al., 1998) and therefore may promote D2R trafficking via PKC signaling. Investigation of ALK-activated signaling pathways that promote D2R trafficking is an exciting area for future research.
It is currently not clear how altered desensitization of D2R in VTA DA neurons affects ethanol binge drinking and reward, since there are no studies that have specifically addressed this issue. However, we predict that attenuating desensitization of D2R by ALK inhibitors would sustain inhibitory signaling by D2R, decreasing DA neuronal activity and thereby reduce DA release in VTA projection targets such as the Acb, amygdala and prefrontal cortex. We hypothesize that this would decrease ethanol consumption, since DA is released upon ethanol exposure and contributes to ethanol reinforcement and reward. A model for how ALK inhibition would affect the firing of DA neurons and subsequent DA release and ethanol consumption is illustrated in Fig. 6. Control of DA output plays an important role in ethanol consumption, as blocking DA neurotransmission in the Acb reduces ethanol consumption (Gonzales et al., 2004). Likewise, inhibition of VTA DA neurons by the D2R agonist quinpirole decreases operant ethanol administration and intra-VTA ethanol self-administration (Hodge et al., 1993; Rodd et al., 2004), and D2R knockout mice show altered ethanol consumption and reward (Bulwa et al., 2011; Cunningham et al., 2000; Delis et al., 2013; Risinger et al., 2000).
We previously demonstrated that acute application of ethanol to VTA slices blocks DIR (Nimitvilai et al., 2012b), similar to the effect of the ALK inhibitors on DIR. This result was counterintuitive, since ALK inhibitors also decrease binge-like ethanol consumption. Perhaps the inhibition of D2R desensitization by acute ethanol is a homeostatic, negative regulatory response of DA neurons that maintains autoreceptor inhibition of neuronal activity in the face of excessive activation of DA neurons. Decreased DA release would be expected to curb excessive alcohol drinking. Inhibition of ALK has the same effect, effectively “substituting” for the pharmacological effect of alcohol, engaging this negative regulatory mechanism on DA neurons, and consequently making continued alcohol consumption undesirable. Alternatively, ALK inhibition may make alcohol aversive. Interestingly, we found that TAE684 blocked ethanol CPP but was not rewarding or aversive in the absence of ethanol, indicating a specific interaction between ALK inhibition and the rewarding properties of ethanol.
The prolonged effects of ethanol on ALK signaling and D2R desensitization are not known. This is important to investigate, since chronic ethanol exposure likely results in neuroadaptations in DA neurons and a dysregulation of D2R signaling. Examining the role of D2R desensitization after chronic ethanol exposure will be important to understand mechanisms that promote chronic alcohol abuse. However, our findings suggest a novel and potentially critical link between ALK signaling, D2R desensitization and binge drinking and provide an exciting new area of research aimed at preventing harmful drinking.
Supplementary Material
Acknowledgments
This study was supported by the National Institute on Alcohol Abuse and Alcoholism (AA016654, AA020912, and AA022538 to AWL; AA09125 and AA05846 to MSB) and the National Institutes of Health Office of the Director (OD010914). We thank Drs. Liquan Xue and Stephan Morris for the gift of the anti-ALK antiserum, Donghong He for producing lentivirus, and Maureen A. McElvain for assistance with electrophysiology. We also thank the Confocal Microscopy Facility at the Research Resources Center at UIC.
Footnotes
Disclosure
The authors declare no competing financial interests.
Authors Contribution
AWL and MSB conceived of and designed the research. AWL, JWD, HC, and CY performed the experiments. All authors analyzed the data and assisted in interpretation. AWL and MSB wrote the manuscript. All authors critically reviewed content and approved the final version for publication.
References
- Bai RY, Dieter P, Peschel C, Morris SW, Duyster J. Nucleophosmin-anaplastic lymphoma kinase of large-cell anaplastic lymphoma is a constitutively active tyrosine kinase that utilizes phospholipase C-gamma to mediate its mitogenicity. Molecular and cellular biology. 1998;18:6951–6961. doi: 10.1128/mcb.18.12.6951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bartlett SE, Enquist J, Hopf FW, Lee JH, Gladher F, Kharazia V, Waldhoer M, Mailliard WS, Armstrong R, Bonci A, Whistler JL. Dopamine responsiveness is regulated by targeted sorting of D2 receptors. Proceedings of the National Academy of Sciences of the United States of America. 2005;102:11521–11526. doi: 10.1073/pnas.0502418102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bilsland JG, Wheeldon A, Mead A, Znamenskiy P, Almond S, Waters KA, Thakur M, Beaumont V, Bonnert TP, Heavens R, Whiting P, McAllister G, Munoz-Sanjuan I. Behavioral and neurochemical alterations in mice deficient in anaplastic lymphoma kinase suggest therapeutic potential for psychiatric indications. Neuropsychopharmacology : official publication of the American College of Neuropsychopharmacology. 2008;33:685–700. doi: 10.1038/sj.npp.1301446. [DOI] [PubMed] [Google Scholar]
- Bonomo YA, Bowes G, Coffey C, Carlin JB, Patton GC. Teenage drinking and the onset of alcohol dependence: a cohort study over seven years. Addiction. 2004;99:1520–1528. doi: 10.1111/j.1360-0443.2004.00846.x. [DOI] [PubMed] [Google Scholar]
- Brodie MS, McElvain MA, Bunney EB, Appel SB. Pharmacological reduction of small conductance calcium-activated potassium current (SK) potentiates the excitatory effect of ethanol on ventral tegmental area dopamine neurons. The Journal of pharmacology and experimental therapeutics. 1999;290:325–333. [PubMed] [Google Scholar]
- Brodie MS, Shefner SA, Dunwiddie TV. Ethanol increases the firing rate of dopamine neurons of the rat ventral tegmental area in vitro. Brain research. 1990;508:65–69. doi: 10.1016/0006-8993(90)91118-z. [DOI] [PubMed] [Google Scholar]
- Bulwa ZB, Sharlin JA, Clark PJ, Bhattacharya TK, Kilby CN, Wang Y, Rhodes JS. Increased consumption of ethanol and sugar water in mice lacking the dopamine D2 long receptor. Alcohol. 2011;45:631–639. doi: 10.1016/j.alcohol.2011.06.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cattaneo F, Guerra G, Parisi M, De Marinis M, Tafuri D, Cinelli M, Ammendola R. Cell-surface receptors transactivation mediated by g protein-coupled receptors. International journal of molecular sciences. 2014;15:19700–19728. doi: 10.3390/ijms151119700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cozzoli DK, Courson J, Rostock C, Campbell RR, Wroten MG, McGregor H, Caruana AL, Miller BW, Hu JH, Wu Zhang P, Xiao B, Worley PF, Crabbe JC, Finn DA, Szumlinski KK. Protein Kinase C Epsilon Activity in the Nucleus Accumbens and Central Nucleus of the Amygdala Mediates Binge Alcohol Consumption. Biological psychiatry. 2015 doi: 10.1016/j.biopsych.2015.01.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cunningham CL, Howard MA, Gill SJ, Rubinstein M, Low MJ, Grandy DK. Ethanol-conditioned place preference is reduced in dopamine D2 receptor-deficient mice. Pharmacology, biochemistry, and behavior. 2000;67:693–699. doi: 10.1016/s0091-3057(00)00414-7. [DOI] [PubMed] [Google Scholar]
- Cunningham CL, Niehus DR, Malott DH, Prather LK. Genetic differences in the rewarding and activating effects of morphine and ethanol. Psychopharmacology. 1992;107:385–393. doi: 10.1007/BF02245166. [DOI] [PubMed] [Google Scholar]
- Davis MI, Hunt JP, Herrgard S, Ciceri P, Wodicka LM, Pallares G, Hocker M, Treiber DK, Zarrinkar PP. Comprehensive analysis of kinase inhibitor selectivity. Nature biotechnology. 2011;29:1046–1051. doi: 10.1038/nbt.1990. [DOI] [PubMed] [Google Scholar]
- Delcourt N, Bockaert J, Marin P. GPCR-jacking: from a new route in RTK signalling to a new concept in GPCR activation. Trends in pharmacological sciences. 2007;28:602–607. doi: 10.1016/j.tips.2007.09.007. [DOI] [PubMed] [Google Scholar]
- Delis F, Thanos PK, Rombola C, Rosko L, Grandy D, Wang GJ, Volkow ND. Chronic mild stress increases alcohol intake in mice with low dopamine D2 receptor levels. Behavioral neuroscience. 2013;127:95–105. doi: 10.1037/a0030750. [DOI] [PubMed] [Google Scholar]
- Flatscher-Bader T, Wilce PA. Impact of alcohol abuse on protein expression of midkine and excitatory amino acid transporter 1 in the human prefrontal cortex. Alcoholism, clinical and experimental research. 2008;32:1849–1858. doi: 10.1111/j.1530-0277.2008.00754.x. [DOI] [PubMed] [Google Scholar]
- Galkin AV, Melnick JS, Kim S, Hood TL, Li N, Li L, Xia G, Steensma R, Chopiuk G, Jiang J, Wan Y, Ding P, Liu Y, Sun F, Schultz PG, Gray NS, Warmuth M. Identification of NVP-TAE684, a potent, selective, and efficacious inhibitor of NPM-ALK. Proceedings of the National Academy of Sciences of the United States of America. 2007;104:270–275. doi: 10.1073/pnas.0609412103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gavi S, Yin D, Shumay E, Wang HY, Malbon CC. Insulin-like growth factor-I provokes functional antagonism and internalization of beta1-adrenergic receptors. Endocrinology. 2007;148:2653–2662. doi: 10.1210/en.2006-1569. [DOI] [PubMed] [Google Scholar]
- Gonzales RA, Job MO, Doyon WM. The role of mesolimbic dopamine in the development and maintenance of ethanol reinforcement. Pharmacology & therapeutics. 2004;103:121–146. doi: 10.1016/j.pharmthera.2004.06.002. [DOI] [PubMed] [Google Scholar]
- Green AS, Grahame NJ. Ethanol drinking in rodents: is free-choice drinking related to the reinforcing effects of ethanol? Alcohol. 2008;42:1–11. doi: 10.1016/j.alcohol.2007.10.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guan J, Umapathy G, Yamazaki Y, Wolfstetter G, Mendoza P, Pfeifer K, Mohammed A, Hugosson F, Zhang H, Hsu AW, Halenbeck R, Hallberg B, Palmer RH. FAM150A and FAM150B are activating ligands for Anaplastic Lymphoma Kinase. Elife. 2015;4 doi: 10.7554/eLife.09811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hallberg B, Palmer RH. Mechanistic insight into ALK receptor tyrosine kinase in human cancer biology. Nature reviews Cancer. 2013;13:685–700. doi: 10.1038/nrc3580. [DOI] [PubMed] [Google Scholar]
- He D, Chen H, Muramatsu H, Lasek AW. Ethanol activates midkine and anaplastic lymphoma kinase signaling in neuroblastoma cells and in the brain. Journal of neurochemistry. 2015 doi: 10.1111/jnc.13252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hilderbrand ER, Lasek AW. Sex differences in cocaine conditioned place preference in C57BL/6J mice. Neuroreport. 2014;25:105–109. doi: 10.1097/WNR.0000000000000053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hodge CW, Haraguchi M, Erickson H, Samson HH. Ventral tegmental microinjections of quinpirole decrease ethanol and sucrose-reinforced responding. Alcoholism, clinical and experimental research. 1993;17:370–375. doi: 10.1111/j.1530-0277.1993.tb00778.x. [DOI] [PubMed] [Google Scholar]
- Iwahara T, Fujimoto J, Wen D, Cupples R, Bucay N, Arakawa T, Mori S, Ratzkin B, Yamamoto T. Molecular characterization of ALK, a receptor tyrosine kinase expressed specifically in the nervous system. Oncogene. 1997;14:439–449. doi: 10.1038/sj.onc.1200849. [DOI] [PubMed] [Google Scholar]
- Kanny D, Liu Y, Brewer RD, Lu H Centers for Disease C, Prevention. Binge drinking - United States, 2011. Morbidity and mortality weekly report Surveillance summaries. 2013;62(Suppl 3):77–80. [Google Scholar]
- Kasten CR, Boehm SL., 2nd Intra-nucleus accumbens shell injections of R(+)- and S(−)-baclofen bidirectionally alter binge-like ethanol, but not saccharin, intake in C57Bl/6J mice. Behavioural brain research. 2014;272:238–247. doi: 10.1016/j.bbr.2014.07.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kharazia VN, Jacobs KM, Prince DA. Light microscopic study of GluR1 and calbindin expression in interneurons of neocortical microgyral malformations. Neuroscience. 2003;120:207–218. doi: 10.1016/s0306-4522(03)00282-3. [DOI] [PubMed] [Google Scholar]
- King AC, de Wit H, McNamara PJ, Cao D. Rewarding, stimulant, and sedative alcohol responses and relationship to future binge drinking. Archives of general psychiatry. 2011;68:389–399. doi: 10.1001/archgenpsychiatry.2011.26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- King AC, McNamara PJ, Hasin DS, Cao D. Alcohol challenge responses predict future alcohol use disorder symptoms: a 6-year prospective study. Biological psychiatry. 2014;75:798–806. doi: 10.1016/j.biopsych.2013.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kodama T, Hasegawa M, Takanashi K, Sakurai Y, Kondoh O, Sakamoto H. Antitumor activity of the selective ALK inhibitor alectinib in models of intracranial metastases. Cancer chemotherapy and pharmacology. 2014;74:1023–1028. doi: 10.1007/s00280-014-2578-6. [DOI] [PubMed] [Google Scholar]
- Kotecha SA, Oak JN, Jackson MF, Perez Y, Orser BA, Van Tol HH, MacDonald JF. A D2 class dopamine receptor transactivates a receptor tyrosine kinase to inhibit NMDA receptor transmission. Neuron. 2002;35:1111–1122. doi: 10.1016/s0896-6273(02)00859-0. [DOI] [PubMed] [Google Scholar]
- Lacey MG, Mercuri NB, North RA. Dopamine acts on D2 receptors to increase potassium conductance in neurones of the rat substantia nigra zona compacta. The Journal of physiology. 1987;392:397–416. doi: 10.1113/jphysiol.1987.sp016787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lasek AW, Gesch J, Giorgetti F, Kharazia V, Heberlein U. Alk is a transcriptional target of LMO4 and ERalpha that promotes cocaine sensitization and reward. The Journal of neuroscience : the official journal of the Society for Neuroscience. 2011a;31:14134–14141. doi: 10.1523/JNEUROSCI.3415-11.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lasek AW, Lim J, Kliethermes CL, Berger KH, Joslyn G, Brush G, Xue L, Robertson M, Moore MS, Vranizan K, Morris SW, Schuckit MA, White RL, Heberlein U. An evolutionary conserved role for anaplastic lymphoma kinase in behavioral responses to ethanol. PloS one. 2011b;6:e22636. doi: 10.1371/journal.pone.0022636. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lowery EG, Spanos M, Navarro M, Lyons AM, Hodge CW, Thiele TE. CRF-1 antagonist and CRF-2 agonist decrease binge-like ethanol drinking in C57BL/6J mice independent of the HPA axis. Neuropsychopharmacology : official publication of the American College of Neuropsychopharmacology. 2010;35:1241–1252. doi: 10.1038/npp.2009.209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maeda N, Ichihara-Tanaka K, Kimura T, Kadomatsu K, Muramatsu T, Noda M. A receptor-like protein-tyrosine phosphatase PTPzeta/RPTPbeta binds a heparin-binding growth factor midkine. Involvement of arginine 78 of midkine in the high affinity binding to PTPzeta. The Journal of biological chemistry. 1999;274:12474–12479. doi: 10.1074/jbc.274.18.12474. [DOI] [PubMed] [Google Scholar]
- Mazot P, Cazes A, Dingli F, Degoutin J, Irinopoulou T, Boutterin MC, Lombard B, Loew D, Hallberg B, Palmer RH, Delattre O, Janoueix-Lerosey I, Vigny M. Internalization and down-regulation of the ALK receptor in neuroblastoma cell lines upon monoclonal antibodies treatment. PloS one. 2012;7:e33581. doi: 10.1371/journal.pone.0033581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Melon LC, Boehm SL., 2nd GABAA receptors in the posterior, but not anterior, ventral tegmental area mediate Ro15-4513-induced attenuation of binge-like ethanol consumption in C57BL/6J female mice. Behavioural brain research. 2011;220:230–237. doi: 10.1016/j.bbr.2011.02.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meng K, Rodriguez-Pena A, Dimitrov T, Chen W, Yamin M, Noda M, Deuel TF. Pleiotrophin signals increased tyrosine phosphorylation of beta beta-catenin through inactivation of the intrinsic catalytic activity of the receptor-type protein tyrosine phosphatase beta/zeta. Proceedings of the National Academy of Sciences of the United States of America. 2000;97:2603–2608. doi: 10.1073/pnas.020487997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mulligan MK, Ponomarev I, Hitzemann RJ, Belknap JK, Tabakoff B, Harris RA, Crabbe JC, Blednov YA, Grahame NJ, Phillips TJ, Finn DA, Hoffman PL, Iyer VR, Koob GF, Bergeson SE. Toward understanding the genetics of alcohol drinking through transcriptome meta-analysis. Proceedings of the National Academy of Sciences of the United States of America. 2006;103:6368–6373. doi: 10.1073/pnas.0510188103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murray PB, Lax I, Reshetnyak A, Ligon GF, Lillquist JS, Natoli EJ, Jr, Shi X, Folta-Stogniew E, Gunel M, Alvarado D, Schlessinger J. Heparin is an activating ligand of the orphan receptor tyrosine kinase ALK. Sci Signal. 2015;8:ra6. doi: 10.1126/scisignal.2005916. [DOI] [PubMed] [Google Scholar]
- Nimitvilai S, Arora DS, Brodie MS. Reversal of dopamine inhibition of dopaminergic neurons of the ventral tegmental area is mediated by protein kinase C. Neuropsychopharmacology : official publication of the American College of Neuropsychopharmacology. 2012a;37:543–556. doi: 10.1038/npp.2011.222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nimitvilai S, Arora DS, McElvain MA, Brodie MS. Ethanol blocks the reversal of prolonged dopamine inhibition of dopaminergic neurons of the ventral tegmental area. Alcoholism, clinical and experimental research. 2012b;36:1913–1921. doi: 10.1111/j.1530-0277.2012.01814.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nimitvilai S, Brodie MS. Reversal of prolonged dopamine inhibition of dopaminergic neurons of the ventral tegmental area. The Journal of pharmacology and experimental therapeutics. 2010;333:555–563. doi: 10.1124/jpet.109.163931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nimitvilai S, McElvain MA, Arora DS, Brodie MS. Reversal of quinpirole inhibition of ventral tegmental area neurons is linked to the phosphatidylinositol system and is induced by agonists linked to G(q) Journal of neurophysiology. 2012c;108:263–274. doi: 10.1152/jn.01137.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nimitvilai S, McElvain MA, Brodie MS. Reversal of dopamine D2 agonist-induced inhibition of ventral tegmental area neurons by Gq-linked neurotransmitters is dependent on protein kinase C, G protein-coupled receptor kinase, and dynamin. The Journal of pharmacology and experimental therapeutics. 2013;344:253–263. doi: 10.1124/jpet.112.199844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O'Keefe JH, Bybee KA, Lavie CJ. Alcohol and cardiovascular health: the razor-sharp double-edged sword. Journal of the American College of Cardiology. 2007;50:1009–1014. doi: 10.1016/j.jacc.2007.04.089. [DOI] [PubMed] [Google Scholar]
- Perez-Pinera P, Zhang W, Chang Y, Vega JA, Deuel TF. Anaplastic lymphoma kinase is activated through the pleiotrophin/receptor protein-tyrosine phosphatase beta/zeta signaling pathway: an alternative mechanism of receptor tyrosine kinase activation. The Journal of biological chemistry. 2007;282:28683–28690. doi: 10.1074/jbc.M704505200. [DOI] [PubMed] [Google Scholar]
- Petit G, Maurage P, Kornreich C, Verbanck P, Campanella S. Binge drinking in adolescents: a review of neurophysiological and neuroimaging research. Alcohol and alcoholism. 2014;49:198–206. doi: 10.1093/alcalc/agt172. [DOI] [PubMed] [Google Scholar]
- Phillips TJ, Dickinson S, Burkhart-Kasch S. Behavioral sensitization to drug stimulant effects in C57BL/6J and DBA/2J inbred mice. Behavioral neuroscience. 1994;108:789–803. doi: 10.1037//0735-7044.108.4.789. [DOI] [PubMed] [Google Scholar]
- Rhodes JS, Best K, Belknap JK, Finn DA, Crabbe JC. Evaluation of a simple model of ethanol drinking to intoxication in C57BL/6J mice. Physiology & behavior. 2005;84:53–63. doi: 10.1016/j.physbeh.2004.10.007. [DOI] [PubMed] [Google Scholar]
- Risinger FO, Freeman PA, Rubinstein M, Low MJ, Grandy DK. Lack of operant ethanol self-administration in dopamine D2 receptor knockout mice. Psychopharmacology. 2000;152:343–350. doi: 10.1007/s002130000548. [DOI] [PubMed] [Google Scholar]
- Rodd ZA, Melendez RI, Bell RL, Kuc KA, Zhang Y, Murphy JM, McBride WJ. Intracranial self-administration of ethanol within the ventral tegmental area of male Wistar rats: evidence for involvement of dopamine neurons. The Journal of neuroscience : the official journal of the Society for Neuroscience. 2004;24:1050–1057. doi: 10.1523/JNEUROSCI.1319-03.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sakamoto H, Tsukaguchi T, Hiroshima S, Kodama T, Kobayashi T, Fukami TA, Oikawa N, Tsukuda T, Ishii N, Aoki Y. CH5424802, a selective ALK inhibitor capable of blocking the resistant gatekeeper mutant. Cancer cell. 2011;19:679–690. doi: 10.1016/j.ccr.2011.04.004. [DOI] [PubMed] [Google Scholar]
- Stoica GE, Kuo A, Aigner A, Sunitha I, Souttou B, Malerczyk C, Caughey DJ, Wen D, Karavanov A, Riegel AT, Wellstein A. Identification of anaplastic lymphoma kinase as a receptor for the growth factor pleiotrophin. The Journal of biological chemistry. 2001;276:16772–16779. doi: 10.1074/jbc.M010660200. [DOI] [PubMed] [Google Scholar]
- Stoica GE, Kuo A, Powers C, Bowden ET, Sale EB, Riegel AT, Wellstein A. Midkine binds to anaplastic lymphoma kinase (ALK) and acts as a growth factor for different cell types. The Journal of biological chemistry. 2002;277:35990–35998. doi: 10.1074/jbc.M205749200. [DOI] [PubMed] [Google Scholar]
- Vernersson E, Khoo NK, Henriksson ML, Roos G, Palmer RH, Hallberg B. Characterization of the expression of the ALK receptor tyrosine kinase in mice. Gene Expr Patterns. 2006;6:448–461. doi: 10.1016/j.modgep.2005.11.006. [DOI] [PubMed] [Google Scholar]
- Vicente-Rodriguez M, Perez-Garcia C, Ferrer-Alcon M, Uribarri M, Sanchez-Alonso MG, Ramos MP, Herradon G. Pleiotrophin differentially regulates the rewarding and sedative effects of ethanol. Journal of neurochemistry. 2014a;131:688–695. doi: 10.1111/jnc.12841. [DOI] [PubMed] [Google Scholar]
- Vicente-Rodriguez M, Perez-Garcia C, Haro M, Ramos MP, Herradon G. Genetic inactivation of midkine modulates behavioural responses to ethanol possibly by enhancing GABA(A) receptor sensitivity to GABA(A) acting drugs. Behavioural brain research. 2014b;274:258–263. doi: 10.1016/j.bbr.2014.08.023. [DOI] [PubMed] [Google Scholar]
- Wang C, Buck DC, Yang R, Macey TA, Neve KA. Dopamine D2 receptor stimulation of mitogen-activated protein kinases mediated by cell type-dependent transactivation of receptor tyrosine kinases. Journal of neurochemistry. 2005;93:899–909. doi: 10.1111/j.1471-4159.2005.03055.x. [DOI] [PubMed] [Google Scholar]
- Wang KS, Liu X, Zhang Q, Pan Y, Aragam N, Zeng M. A meta-analysis of two genome-wide association studies identifies 3 new loci for alcohol dependence. Journal of psychiatric research. 2011;45:1419–1425. doi: 10.1016/j.jpsychires.2011.06.005. [DOI] [PubMed] [Google Scholar]
- Weiss JB, Xue C, Benice T, Xue L, Morris SW, Raber J. Anaplastic lymphoma kinase and leukocyte tyrosine kinase: functions and genetic interactions in learning, memory and adult neurogenesis. Pharmacology, biochemistry, and behavior. 2012;100:566–574. doi: 10.1016/j.pbb.2011.10.024. [DOI] [PubMed] [Google Scholar]
- Wilcox MV, Cuzon Carlson VC, Sherazee N, Sprow GM, Bock R, Thiele TE, Lovinger DM, Alvarez VA. Repeated binge-like ethanol drinking alters ethanol drinking patterns and depresses striatal GABAergic transmission. Neuropsychopharmacology : official publication of the American College of Neuropsychopharmacology. 2014;39:579–594. doi: 10.1038/npp.2013.230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zakhari S, Li TK. Determinants of alcohol use and abuse: Impact of quantity and frequency patterns on liver disease. Hepatology. 2007;46:2032–2039. doi: 10.1002/hep.22010. [DOI] [PubMed] [Google Scholar]
- Zapata A, Gonzales RA, Shippenberg TS. Repeated ethanol intoxication induces behavioral sensitization in the absence of a sensitized accumbens dopamine response in C57BL/6J and DBA/2J mice. Neuropsychopharmacology : official publication of the American College of Neuropsychopharmacology. 2006;31:396–405. doi: 10.1038/sj.npp.1300833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang J, Deng X, Choi HG, Alessi DR, Gray NS. Characterization of TAE684 as a potent LRRK2 kinase inhibitor. Bioorganic & medicinal chemistry letters. 2012;22:1864–1869. doi: 10.1016/j.bmcl.2012.01.084. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.