Abstract
Background The emergence of zoonotic viruses in domestic animals is a significant public health concern. Canine influenza virus (CIV) H3N2 is a virus that can infect companion animals and is, therefore, a potential public health concern.
Objective This study investigated the inter‐ and intraspecies transmission of CIV among dogs, cats, and ferrets, under laboratory conditions, to determine whether transmission of the virus was possible between as well as within these domestic animal species.
Method The transmission routes for inter‐ and intraspecies transmission were airborne and direct contact, respectively. Transmission was conducted through intranasal infection of dogs followed by exposure to either cats or ferrets and by comingling infected and naïve animals of the same species.
Results The interspecies transmission of CIV H3N2 via airborne was only observed from dogs to cats and not from dogs to ferrets. However, direct intranasal infection of either cats or ferrets with CIV could induce influenza‐like clinical signs, viral shedding, and serological responses. Additionally, naïve cats and ferrets could be infected by CIV via direct contact with infected animals of the same species.
Conclusion Cats appear to be another susceptible host of CIV H3N2, whereas ferrets are not likely natural hosts. The molecular‐based mechanism of interspecies and intraspecies transmission of CIV H3N2 should be further studied.
Keywords: Cats, CIV H3N2, ferrets, interspecies, intraspecies, transmission
Introduction
Canine influenza virus (CIV) H3N2 is a virus of avian origin and was first reported in 2007. 1 Dogs were demonstrated to have avian influenza virus receptors in their lower respiratory tract, and all gene segments of CIV H3N2 were shown to be homologous to avian influenza viruses; therefore, interspecies transmission of influenza virus from birds to dogs was suggested to be possible. 1 Furthermore, a recent study reported the susceptibility of cats to CIV H3N2 infection, suggesting the possibility of the emergence of recombinant, potentially zoonotic, feline, or CIVs in companion animals. 2 The emergence of novel, recombinant influenza viruses with zoonotic potential is a significant public health concern. 3 The concern is elevated further when the viruses can be transmitted between different species of animals that are in frequent contact with humans. For example, a triple reassortant H3N2 influenza virus isolated from pigs was shown to replicate and be transmissible among turkeys. 4 Such evidence amplifies the concerns about interspecies transmission of CIV H3N2 between different domestic animal species and underscores the concern that these same viruses might also be able to infect humans who come in contact with the animals.
The possibility of intraspecies transmission (dog‐to‐dog) of CIV H3N2 was demonstrated by direct contact between infected and naïve dogs. 5 Surveillance data show that the percentage of pet dogs that are seropositive for CIV H3N2 has dramatically increased from 0·48% (2007) to 5·2–6·5% (2008), 6 , 7 indicating continuous transmission among pet dogs. As cats, another popular domestic animal, have already been demonstrated to host CIV H3N2, intraspecies transmission among cat populations is also possible but not yet demonstrated. Ferrets are also known to be susceptible to influenza A infection and display many clinical signs of human influenza infection with a similar complexity of signs. 8 Therefore, ferrets can be considered to predict the possibility for interspecies transmission of CIV H3N2 to humans.
The influenza virus demonstrates high infectivity via both aerosol and respiratory droplet transmission, 9 indicating that the transmission of CIV H3N2 among animals may occur by similar routes, as well as by direct contact. Therefore, in this study, we investigated the inter‐ and intraspecies transmission of CIV H3N2 by these routes among dogs, cats, and ferrets under controlled conditions.
Materials and methods
Animals
Dogs (beagle, 9‐week‐old), cats (domestic short hair, 10‐week‐old), and ferrets (12‐week‐old) were housed in each cage of the isolation facility at Green Cross Veterinary Products (Yong‐in, South Korea) for the study. All animal experiments complied with the current laws of South Korea. Animal care and treatment were conducted in accordance with guidelines establishment by the Green Cross Veterinary Products Institutional Animal Care and Use Committee.
Interspecies transmission
Four dogs were infected with 106·9 of 50% egg infectious dose (EID50)/100 μl of CIV H3N2 [A/canine/Korea/01/2007 (H3N2)] via the intranasal route on day 0. Six hours after infection, interspecies aerosol transmission of the virus was monitored by housing two cats or four ferrets, in separate cages, in close proximity (10 cm) to the infected dogs. Under these conditions, the cats and ferrets were allowed to be exposed to airborne CIV H3N2 shed from the infected dogs without direct contact each other.
Following placing the sentinel animals in close proximity to the infected animals, clinical signs (tachypnea, dyspnea, and lethargy), as well as nasal shedding of the virus, were monitored for 2 weeks. Nasal swabs were collected from each animal on days 2, 3, 4, 5, 7, 8, 9, 10, 11, 12, 13, and 14 post‐inoculation. At the end of the study, blood samples were collected from each animal and the titer of antibodies against CIV H3N2 was determined using a commercial ELISA kit (Bionote, Suwon, South Korea).
Intraspecies transmission
Two cats and three ferrets were intranasally infected with CIV [(106·9 EID50/100 μl), A/canine/Korea/01/2007 (H3N2)] on day 0. Six hours after infection, the two infected cats were comingled with two naïve cats in the same cage. The three infected ferrets were similarly comingled with three naïve ferrets. Under these experimental conditions, the naïve animals were exposed to the CIV H3N2 shed from their infected cage‐mates by direct contact. The post‐infection sampling scheme was the same as described for the interspecies experiment.
Viral quantification from nasal swabs
The nasal swab was performed using PBS‐moisturized cotton swab from each nostrils. Two nasal swabs from each animal were suspended in 1 ml of Dulbecco’s modified Eagle’s medium (DMEM) and used in the RNA extraction protocol with Trizol LS (Invitrogen, Carlsbad, CA, USA). Viral shedding was quantified using real‐time polymer chain reaction, as described previously. 1 , 5
Serology
Serum antibodies were assessed by a commercially available competitive ELISA kit, which targets the nucleocapsid protein of Influenza A viruses (Bionote, South Korea). The competitive ELISA kit was previously validated for detecting CIV in both mammalian and avian species. 10 The results, expressed as percent inhibition (PI), were calculated according to the following formula: PI value = [1 − (ODsample/mean ODcontrol)] × 100. Samples were classified as positive if the PI value was ≥50 and negative if the PI value was ≤49.
Results
Clinical signs of infected or contact animals
Respiratory signs were observed in all animals infected by CIV H3N2 via the intranasal route (Table 1). The infected cats and dogs showed sneezing, coughing, abdominal breathing, and nasal discharges, while the infected ferrets showed only mild sneezing. Cats exposed to infected dogs (via airborne exposure) and to other infected cats (via direct contact) also showed respiratory signs such as sneezing, coughing, and nasal discharge. Ferrets exposed to either infected ferrets (via direct contact) or infected dogs (via airborne exposure) did not present with respiratory signs.
Table 1.
Infected animals’ clinical symptoms and seroconversion
| Group | Transmission route | No. of positive animals/no. tested | |||
|---|---|---|---|---|---|
| Clinical signs* | Seroconversion | ||||
| Infected | Contacted | Infected | Contacted | ||
| Dog** to cat | AE | 4/4 | 2/2 | 4/4 | 2/2 |
| Dog to ferret | AE | 0/2 | 0/2 | ||
| Cat** to cat | DC | 2/2 | 2/2 | 2/2 | 2/2 |
| Ferret*** to ferret | DC | 3/3 | 0/3 | 3/3 | 2/3 |
AE, airborne exposure; DC, direct contact.
*Only cats exposed to the infected dogs and cats with nasal challenge and direct contact, respectively, showed sneezing, coughing, and nasal discharge, while the exposed ferrets did not.
**The CIV H3N2 infected dogs and cats via intranasal route showed sneezing, coughing, and nasal discharge.
***The CIV H3N2 infected ferrets via intranasal route showed only sneezing.
Viral shedding from infected or contact animals
Nasal shedding of CIV H3N2 was monitored, using the mean logEID50 over 14 days, from the infected dogs as well as from the cats and ferrets that were exposed to airborne virus (Figure 1). The CIV H3N2 titer increased from day 2 post‐infection in the infected dogs but was not detectable by day 8. The respiratory droplet‐exposed cats were shedding detectable quantities of virus (>5 logEID50/ml) by day 9. However, no viral shedding was detected by nasal swab from the respiratory droplet‐exposed ferrets at any point during the study.
Figure 1.
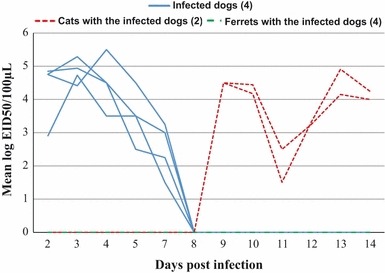
Quantitation of the viral shedding from infected dogs and from the naïve cats and ferrets, which were exposed to the infected dogs via respiratory droplets. Error bars indicate standard deviation.
Intranasally infected cats and ferrets were observed (by nasal swab) to shed virus between days 2 and 7 post‐infection (Figure 2). However, the quantity of virus shed was higher from the infected cats (5·9, 6·0, 6·0, 5·5, and 3·5 mean logEID50/ml on days 2, 3, 4, 5 and 7, respectively) than from the infected ferrets (5·6, 3·7, 3·0, 2·9, and 2·8 mean logEID50/ml at the same time points). Naïve cats exposed to CIV H3N2‐infected cats by direct contact began to shed detectable virus beginning on day 8 and continuing through day 13 (4·5, 4·1, 4·2, 3·4, 3·2, and 2·6 mean logEID50/ml on days 8, 9, 10, 11, 12, and 13, respectively). The naïve ferrets in direct contact with intranasally infected ferrets only shed detectable virus on days 10 and 11 post‐infection and only at a low level (2·8 and 2·5 mean logEID50/ml, respectively).
Figure 2.
Quantitation of viral shedding from intranasally infected cats and ferrets and from the naïve cats and ferrets exposed to the infected animals, via direct contact. (A) Virus shed from infected cats and exposed naïve cats; (B) Virus shed from infected ferrets and exposed naïve ferrets. Error bars indicate standard deviation.
Seroconversion in infected or contact animals
Serum samples were collected from the animals at the end of the study; only animals with a PI value of ≥50 (as described in the Methods section) were considered seropositive. At the end of the study, all of the animals that had been infected with CIV H3N2 intranasally were seropositive (Table 1). Cats exposed to infected dogs (by respiratory droplet) or to infected cats (by direct contact) had seroconverted by the end of the study. Two of the three ferrets exposed to the intranasally infected ferrets were also seropositive, but those exposed (via respiratory droplet) to infected dogs were not.
Discussion
Previous studies examining the intranasal infection of CIV H3N2 in dogs and cats revealed that the virus could induce severe respiratory disease. 1 , 2 However, it remained unclear as to whether CIV H3N2 was capable of interspecies transmission among pet animals via either direct contact or through respiratory droplet transmission. Therefore, in this study, the interspecies transmission of CIV H3N2 was investigated by aerosol exposure of naïve cats and ferrets with CIV H3N2‐infected dogs.
The results indicated that naïve cats exposed to infected dogs developed respiratory signs and nasal shedding of CIV H3N2 after 9 days of exposure to the infected animals. As a result, these cats were also seropositive for the virus by the end of the study. However, similarly exposed, naïve ferrets did not demonstrate clinical signs or viral shedding and did not seroconvert. These data suggest that CIV H3N2 is transmissible from infected dogs to naïve cats but not to naïve ferrets, via respiratory droplets. These results further indicate that cats are similarly susceptible to signatic CIV H3N2 infection as are dogs. In addition, infected cats shed the virus in sufficient quantities to result in transmission of the virus to other cats.
For an avian–human H9N2 influenza A virus to be transmitted by respiratory droplets to ferrets, it has been demonstrated that it is essential to have minimal molecular constraints (adaptation by minimal changes at the protein level) to infection in the host animal. 11 The necessity of adaptation to new host for airborne transmission was also demonstrated in a study showing that human influenza H3N2 virus, demonstrating preferential binding to α2,3‐linked sialic acid receptors, were not transmissible to ferrets (expressing α2,6‐linked sialic acid receptors), although the virus could be shed from directly inoculated ferrets. 12 Also, CIV H3N2 causes histopathological lesions especially in lower respiratory tract where α2,3‐linked sialic acid receptors were found to be abundant. 1 , 13 These observations, combined with those of the present study, help explain why CIV H3N2, a virus of avian origin with possible recognition of α2,3‐linked sialic acid receptors, could not be easily transmitted between CIV‐infected dogs and naïve ferrets.
In this study, cat‐to‐cat transmission of CIV H3N2 was demonstrated, based on clinical signs, active viral shedding, and seroconversion. An earlier study examining the seroprevalence of seasonal and pandemic H1N1 influenza virus in cats was conducted between 2009 and 2010. 14 The results of that study showed that 21·8%, 41·9%, and 25·6% of the examined cats were seropositive for pandemic H1N1 and seasonal human H1N1 and H3N2 influenza viruses, respectively. These results, combined with the findings from the present study, suggest that cats might play a role in the transmission of CIV H3N2, as well as human influenza viruses.
Although CIV H3N2 was not transmitted from the infected dogs to naïve ferrets, naïve ferrets that were exposed to infected ferrets did shed detectable quantities of CIV H3N2 (2·8 and 2·5 mean logEID50/ml on days 10 and 11), and 2 of the 3 exposed ferrets to the infected ferrets by direct contact seroconverted. This observation implies that interspecies transmission of CIV H3N2 to ferrets is not fully established and that further adaptation might be needed. Such additional adaptation may occur when ferrets are intranasally inoculated with the virus. A previous report has indicated that viral deposition was primarily restricted to the upper trachea in ferrets exposed to airborne virus, whereas the deposition occurred in the peripheral lung tissue of animals infected by intranasal inoculation. 15 Therefore, the ferrets with intranasal infection had higher chance to allow CIV H3N2 to be adapted and the adapted viruses could be transmitted from the infected ferrets to the naïve ferrets. Also, the direct contact by caging the ferrets together provided a much higher and prolonged exposure, even if the shed titers were lower, so that the ferrets to ferrets transmission of CIV H3N2 could be possible compared to dogs to ferrets transmission, which was restricted by aerosol transmission. Nevertheless, interspecies transmission of CIV H3N2 from dogs to ferrets was not as efficient as it was between dogs and cats, which implies further biological and epidemiological adaptation would be needed in the case of dogs to ferrets transmission.
In conclusion, this study investigated the interspecies and intraspecies transmissions of CIV H3N2 among 3 species (Figure 3). Interspecies transmission, via respiratory droplets, was demonstrated from dogs to cats but not directly from dogs to ferrets. The virus was also demonstrated to be effectively transmitted from intranasally infected cats and ferrets to naïve animals of the same species via direct contact. These data suggest that cats, in addition to dogs, can be another susceptible host of CIV H3N2; ferrets may not be susceptible host but may be susceptible after viral adaptation. The molecular‐based mechanism study should be followed to reveal the factors of inter‐ and intraspecies transmission of CIV H3N2.
Figure 3.
Overview of intra‐ and interspecies transmission of CIV H3N2.
Author contributions
Hyekwon Kim has involved in organizing data and writing the manuscript; Daesub Song, as the lead investigator, is responsible for whole study including conception, experimental design and performance; Hyoungjoon Moon and Woonseong Na have carried out clinical and laboratory works; Minjoo Yeom has performed viral culture and preparation of inoculums; Seongjoon Park has carried out papers and database analyses; Minki Hong has carried out laboratory works and data analysis; Richard J. Webby and Robert G. Webster have involved in revising the intellectual contents; Bongkyun Park has prepared the manuscript; Jeong‐Ki Kim has performed conception of study and data analyses and revising the manuscript; Bokyu Kang, as a chief investigator, has organized and supervised animal experiments and revised the manuscript.
Conflict of interests
There are no conflicts of interests.
Acknowledgement
This study was supported by a grant of the Korea Health Technology R&D Project, Ministry of Health & Welfare, Republic of Korea (Grant No. A103001). This study was also supported in part by Contract No. HHSN266200700005C (to RGW and RJW) from the National Institute of Allergy and Infectious Diseases and by the American Lebanese Syrian Associated Charities (ALSAC).
References
- 1. Song D, Kang B, Lee C et al. Transmission of avian influenza virus (H3N2) to dogs. Emerg Infect Dis 2008; 14:741–746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Song DS, An DJ, Moon HJ et al. Interspecies transmission of the canine influenza H3N2 virus to domestic cats in South Korea. 2010. J Gen Virol 2011; 92:2350–2355. [DOI] [PubMed] [Google Scholar]
- 3. Sponseller BA, Strait E, Jergens A et al. Influenza A pandemic (H1N1) 2009 virus infection in domestic cat. Emerg Infect Dis 2010; 16:534–537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Yassine H, Al‐Natour M, Lee C‐W, Saif Y. Interspecies and intraspecies transmission of triple reassortant H3N2 influenza A viruses. Virol J 2007; 4:129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Song D, Lee C, Kang B et al. Experimental infection of dogs with avian‐origin canine Influenza A virus (H3N2). Emerg Infect Dis 2009; 15:56–58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Lee C, Song D, Kang B et al. A serological survey of avian origin canine H3N2 influenza virus in dogs in Korea. Vet Microbiol 2009; 137:359–362. [DOI] [PubMed] [Google Scholar]
- 7. An DJ, Jeoung HY, Jeong W et al. A serological survey of canine respiratory coronavirus and canine influenza virus in Korean dogs. J Vet Med Sci 2010; 72:1217–1219. [DOI] [PubMed] [Google Scholar]
- 8. Barnard DL. Animal models for the study of influenza pathogenesis and therapy. Antiviral Res 2009; 82:A110–A122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Teunis PFM, Brienen N, Kretzschmar MEE. High infectivity and pathogenicity of influenza A virus via aerosol and droplet transmission. Epidemics 2010; 2:215–222. [DOI] [PubMed] [Google Scholar]
- 10. Song DS, Lee YJ, Jeong OM et al. Evaluation of a competitive ELISA for antibody detection against avian influenza virus. J Vet Sci 2009; 10:323–329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Sorrell EM, Wan H, Araya Y, Song H, Perez DR. Minimal molecular constraints for respiratory droplet transmission of an avian–human H9N2 influenza A virus. Proc Natl Acad Sci U S A 2009; 106:7565–7570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Roberts KL, Shelton H, Scull M, Pickles R, Barclay WS. Lack of transmission of a human influenza virus with avian receptor specificity between ferrets is not due to decreased virus shedding but rather a lower infectivity in vivo. J Gen Virol 2011; 92:1822–1831. [DOI] [PubMed] [Google Scholar]
- 13. Jung K, Lee CS, Kang BK et al. Pathology in dogs with experimental canine H3N2 influenza virus infection. Res Vet Sci 2010; 88:523–527. [DOI] [PubMed] [Google Scholar]
- 14. McCullers J, Van De Velde L‐A, Schultz R et al. Seroprevalence of seasonal and pandemic influenza A viruses in domestic cats. Arch Virol 2011; 156:117–120. [DOI] [PubMed] [Google Scholar]
- 15. Gustin KM, Belser JA, Wadford DA et al. Influenza virus aerosol exposure and analytical system for ferrets. Proc Natl Acad Sci USA 2011; 108:8432–8437. [DOI] [PMC free article] [PubMed] [Google Scholar]


