Abstract
DNA vaccination is a promising strategy to induce effector T cells but also regulatory Foxp3+ CD25+ CD4+ Treg cells and inhibit autoimmune disorders such as type 1 diabetes. Little is known about the antigen requirements that facilitate priming of Treg cells but not autoreactive effector CD8+ T cells. We have shown that the injection of preproinsulin (ppins)-expressing pCI/ppins vector into PD-1- or PD-L1-deficient mice induced Kb/A12-21-monospecific CD8+ T cells and autoimmune diabetes. A pCI/ppinsΔA12-21 vector (lacking the critical Kb/A12-21 epitope) did not induce autoimmune diabetes but elicited a systemic Foxp3+ CD25+ Treg cell immunity that suppressed diabetes induction by a subsequent injection of the diabetogenic pCI/ppins. TGF-β expression was significantly enhanced in the Foxp3+ CD25+ Treg cell population of vaccinated/ppins-primed mice. Ablation of Treg cells in vaccinated/ppins-primed mice by anti-CD25 antibody treatment abolished the protective effect of the vaccine and enabled diabetes induction by pCI/ppins. Adoptive transfer of Treg cells from vaccinated/ppins-primed mice into PD-L1−/− hosts efficiently suppressed diabetes induction by pCI/ppins. We narrowed down the Treg-stimulating domain to a 15-residue ppins76–90 peptide. Vaccine-induced Treg cells thus play a crucial role in the control of de novo primed autoreactive effector CD8+ T cells in this diabetes model.
Type 1 diabetes mellitus (T1D) is an autoimmune disorder, in which insulin-producing beta cells are destroyed by the cellular immune system1. Diabetes development is characterized by progressive infiltration of T cells into the pancreatic islets and consecutive beta cell destruction. Disease in man is triggered by poorly defined antigens and factors that finally result in the breakdown of central and/or peripheral tolerance and activation of autoreactive T cells2. There is increasing evidence from patients with T1D that autoreactive CD8+ T cells play a crucial role in the development of the disease3,4,5,6,7. Facing a rise in the incidence of T1D there is thus a clear need for the development of immunotherapies that induce or restore peripheral tolerance and prevent T1D in a controlled and antigen-specific manner8,9,10.
Immune tolerance is regulated by a variety of mechanisms and checkpoints that affect the differentiation of lymphocytes in central lymphoid organs as well as mature lymphocytes in the periphery. Tolerance in the periphery is maintained by modulatory interactions through co-inhibitory ‘programmed death-1′ (PD-1)/‘programmed death-ligand-1′ (PD-L1 or B7-H1) signals11,12 and/or regulatory Foxp3+ CD25+ CD4+ T cells (Tregs) expressing the transcription factor forkhead box p3 (Foxp3) and the alpha chain of IL-2 receptor (CD25)13. Treg cells can be divided into naturally occurring Foxp3+ CD25+ CD4+ Treg cells (nTregs) and induced Treg cells (iTregs) which, upon antigen stimulation, specifically arise from conventional CD4+ T cells acquiring CD25 and Foxp3 expression outside of the thymus. Both, nTreg and iTreg cells suppress effector T cell responses through a variety of mechanisms. Treg cells can produce anti-inflammatory cytokines and/or impair antigen presenting cell- (APC) or effector T cell- functions by direct cell-to-cell interactions13. Furthermore, the co-inhibitory PD-1/PD-L1 pathway plays a crucial role in the regulation of autoimmune diabetes in non-obese diabetic NOD mice14,15,16, diabetes development in man17,18,19,20 and, in particular may affect the induction and function of autoantigen-specific Foxp3+ CD25+ CD4+ Treg cells20,21,22.
Animal models have been informative to study autoreactive T cell responses as well as immunotherapies to prevent diabetes development23,24. DNA vaccination is a promising strategy to induce CD4+ Treg cells and treat autoimmune disorders such as type 1 diabetes25,26. However, little is known about the antigen requirements that facilitate priming of CD4+ Treg cells (and inhibit autoimmune diabetes), but do not allow the priming of autoreactive effector CD8+ T cells by DNA vaccination. Injection of antigen-expressing vectors preferentially stimulates CD8+ T cell responses, because they allow direct antigen expression and MHC class I-restricted epitope presentation by in vivo transfected APCs. Furthermore, ‘cross-presentation’ of antigenic material, released from non-professional antigen-expressing APCs (e.g., myocytes) to professional APCs (e.g. DCs) facilitated priming of CD8+ T-cell responses27. Vector-encoded antigens also stimulate CD4+ T cells, indicating that endogenously expressed antigens are efficiently processed for MHC class II presentation28. It has been shown that a proinsulin (pins)-expressing DNA vaccine reduced the incidence of diabetes in NOD mice29 and the frequency of autoreactive CD8+ T cells in patients with T1D30. Conditions that promote Th1 to Th2 immunodeviation (e.g. co-expression of the insulin B chain and IL-4) or enhance apoptosis (e.g. by co-expression of glutamic acid decarboxylase and the proapoptotic factor Bax) favor the induction of a protective immunity in NOD mice31,32. However, there is a narrow ridge between the suppression and/or stimulation of T cell-mediated diabetes by ’self’-antigen expressing DNA vaccines. Diabetes development was accelerated in female and male NOD mice after preproinsulin (ppins)-specific DNA immunization, whereas glutamic acid decarboxylase-specific vector DNA conferred partial protection33,34. It is largely unknown why certain antigens or antigen domains either stimulate diabetogenic effector T cells or induce immunosuppressive Treg cells. Therefore, strategies that selectively induce antigen-specific Treg cells and suppress autoreactive T cell responses would significantly improve the functionality and safety of T1D vaccines.
The major advantage of DNA-based immunization is the flexibility in the design of vectors and the manipulation of endogenous antigen expression and/or antigen processing/presentation in distinct cellular compartments by molecular engineering. We have established a novel diabetes model in coinhibition-deficient PD-L1−/− (B7-H1−/−)35 and PD-1−/− mice36 to characterize the ppins-specific induction (or prevention) of autoreactive CD8+ T cells37,38. A single injection of ppins-encoding (pCI/ppins) DNA into PD-1- or PD-L1-deficient mice efficiently induced severe autoimmune diabetes37. Diabetes developed gender-independent in male and female mice with a median onset of 2–3 weeks post-immunization and a cumulative diabetes incidence of >95% by week 3–537. Epitope recognition of pancreas-infiltrating CD8+ T cells isolated from pCI/ppins-immune, diabetic PD-L1−/− and PD-1−/− mice was confined to the Kb-restricted A12-21 epitope at the COOH-terminus of ppins (i.e., the insulin A-chain; Fig. 1a)37. A vector-encoded, mutant ppinsΔA12-21 antigen (lacking the critical Kb/A12-21 epitope) did not induce autoimmune diabetes in PD-L1−/− or PD-1−/− mice38. We thus hypothesized that the mutant pCI/ppinsΔA12-21 vector could elicit a prophylactic, Treg cell-mediated immunity in PD-L1−/− and PD-1−/− mice and protect them from autoimmune diabetes induced by a subsequent injection of the diabetogenic pCI/ppins vector. This model allowed us to explore systematically under well-controlled experimental conditions the induction of ppins-specific Treg cells and the suppression of Kb/A12-21-monospecific effector CD8+ T cells and autoimmune diabetes.
Figure 1. Vaccination of PD-L1−/− mice with pCI/ppins∆A12-21 prevents diabetes induction by a subsequent injection of pCI/ppins.
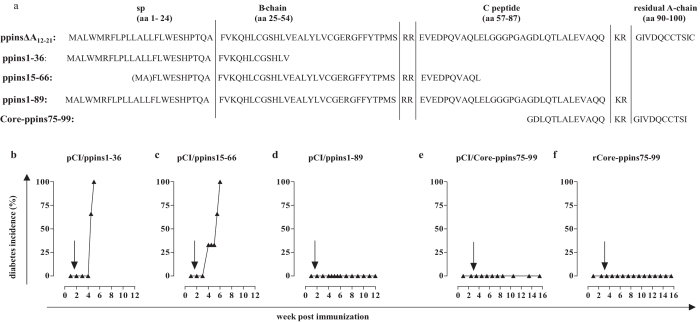
(a) Map of ppins and pCI/ppinsΔA12-21 (lacking the COOH-terminal Kb-binding epitope A12-21) antigens. The signal peptide (SP), the insulin B- and A-chains, the C-peptide as well as the position and sequence of the Kb/A12-21 epitope are indicated. (b–f) PD-L1−/− mice were either immunized with pCI/ppins (b; n = 7) or pCI/ppins∆12-21 (c; n = 10). Furthermore, groups of mice were either vaccinated with pCI/ppins∆A12-21 (d; n = 7) or a HBV core expressing pCI/C vector (e; n = 3) followed by an injection of the diabetogenic pCI/ppins at day 12 post vaccination. (f) PD-L1−/− mice (n = 4 per group) were vaccinated with pCI/ppins∆A12-21 followed by an injection of the diabetogenic pCI/ppins at d24 or d48, or by two injections at day 24 and 38 post vaccination. Arrows indicate the injection of the diabetogenic pCI/ppins. Blood glucose values were measured and cumulative diabetes incidences (%) were determined.
Results
A pCI/ppinsΔA12-21 vaccine (lacking the Kb/A12-21 epitope) efficiently suppressed de novo induction of autoimmune diabetes by the pCI/ppins vector
Injection of pCI/ppins but not pCI/ppinsΔA12-21 DNA (lacking the critical Kb/A12-21 epitope; Fig. 1a) induced autoimmune diabetes in PD-L1−/− and PD-1−/− mice (Fig. 1b,c; Supplementary Fig. S1a, b)37,38. To investigate whether the modified ppinsΔA12-21 antigen induces a prophylactic immunity, we immunized PD-L1−/− or PD-1−/− mice with pCI/ppinsΔA12-21 followed by an injection with the diabetogenic pCI/ppins vector at day 12 post vaccination. Interestingly, none of the mice developed autoimmune diabetes (Fig. 1d; Supplementary Fig. S1c). Vaccination of mice with a Hepatitis B Virus (HBV) core antigen-expressing pCI/C vector did not suppress diabetes induction by a subsequent injection of pCI/ppins (Fig. 1e) and, vice versa, the injection of pCI/ppinsΔA12-21 did not affect the priming of HBV core (Kb/C93-100)-specific CD8+ T cells by a subsequent injection (after 12 days) of the pCI/C vector (Supplementary Fig. S2). The immune response induced by the pCI/ppinsΔA12-21 vaccine was thus specific for the ppins antigen and suppressed diabetes development by de novo activated Kb/A12-21-specific effector CD8+ T cells.
The pCI/ppinsΔA12-21 vaccine efficiently suppressed diabetes development when the diabetogenic pCI/ppins vector was injected after 12, 24 but not 48 days (Fig. 1d,f). Repeated injections of pCI/ppins at d24 and d38 could not override the protective immunity induced by the initial injection of pCI/ppinsΔA12-21 (Fig. 1f). A single injection of the pCI/ppinsΔA12-21 vaccine thus induced a potent but temporary immunosuppressive immunity in co-inhibition deficient PD-L1−/− mice.
PD-L1-independent induction of immunosuppressive responses in wild-type C57BL/6 mice by pCI/ppinsΔA12-21
Little is known whether the missing co-inhibitory PD-1/PD-L1 signals in PD-1−/− and PD-L1−/− mice affect the priming of tolerogenic immune responses21,22. We previously showed that a single injection of pCI/ppins into PD-1/PD-L1-competent C57BL/6 (B6) mice induced IFNγ+ Kb/A12-21-monospecific CD8+ T cells, but these cells destroyed insulin-producing beta cells only after the injection of anti-PD-L1 antibody37. This showed that the priming of autoreactive IFNγ+ Kb/A12-21-specific CD8+ T cells by pCI/ppins is independent from co-inhibitory PD-1/PD-L1 signals37. In contrast, ppinsΔA12-21-vaccinated/ppins-primed B6 mice did not develop autoimmune diabetes and anti-PD-L1 antibody treatment failed to induce autoimmune diabetes (Supplementary Fig. S3) providing evidence that the pCI/ppinsΔA12-21 vaccine induced a prophylactic immunity in both, co-inhibition-competent B6 and co-inhibition-deficient PD-L1−/− and PD-1−/− mice.
The pCI/ppinsΔA12-21 vaccine induced regulatory Foxp3+ CD25+ CD4+ Treg cells
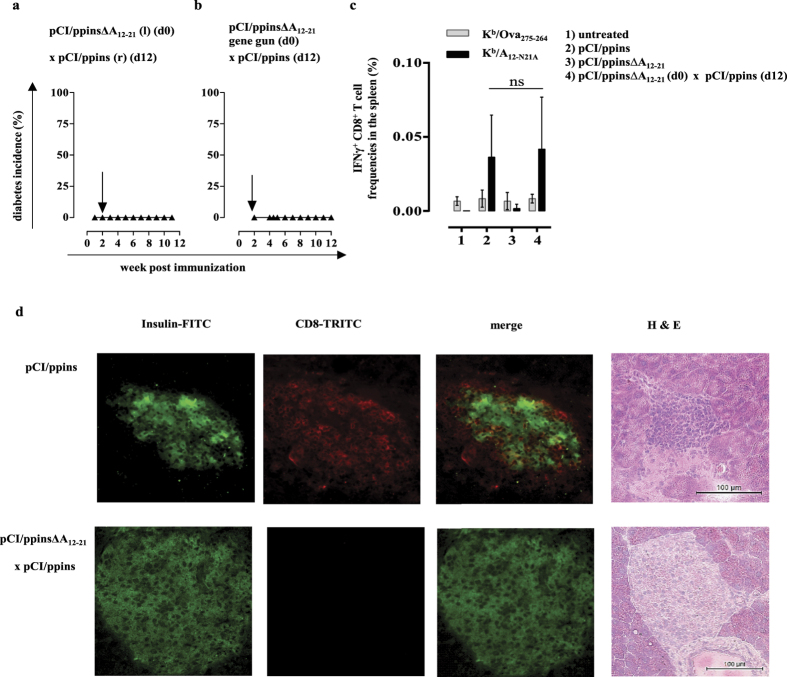
In the experiments described above, we injected both, the pCI/ppinsΔA12-21 vaccine and the diabetes-inducing pCI/ppins vector into the same tibialis anterior muscles. To exclude that the vaccine affected de novo priming of Kb/A12-21-specific CD8+ T cells at the level of the intramuscular injection site (or in the regional draining lymph nodes), we injected the pCI/ppinsΔA12-21 vaccine into the left muscles followed (after 12 days) by an injection of the diabetogenic pCI/ppins vector into the right muscles (Fig. 2a). Alternatively, we administered the pCI/ppinsΔA12-21 vaccine intradermally into the shaved abdominal skin using a helium-driven gene gun (see Supplementary Protocols), followed (after 12 days) by an i.m. injection of the diabetogenic pCI/ppins DNA (Fig. 2b). Again, none of these mice developed autoimmune diabetes (Fig. 2a,b) indicating that systemic but not local vaccine-induced responses suppressed de novo primed autoreactive effector CD8+ T cells.
Figure 2. The pCI/ppins∆A12-21-induced immune response did not prevent priming of Kb/A12-21-specific CD8+ T cells by pCI/ppins.
(a) The pCI/ppins∆A12-21 vaccine was injected into the left (l) and the diabetogenic pCI/ppins (after 12 days) into the right (r) tibialis anterior muscles of PD-L1−/− mice (n = 3). (b) Alternatively, the pCI/ppins∆A12-21 vaccine was administered intradermally into the abdominal skin with a gene gun followed by an injection of the diabetogenic pCI/ppins vector into both tibialis anterior muscles after 12 days (n = 4). Diabetes development was monitored by regular blood glucose measurements. (c) PD-L1−/− mice (n = 3/group) were either left untreated (group 1), or immunized with pCI/ppins (group 2), pCI/ppins∆A12-21 (group 3) or pCI/ppins∆A12-21 and (after 12 days) pCI/ppins (group 4). Spleen cells were prepared at d12 after the final DNA injection, respectively, and restimulated ex vivo with the ppins-specific Kb/A12-N21A (SLYQLENYCA) peptide40. Furthermore, we stimulated spleen cells with a Kb-binding control peptide (Kb/Ova275-264; SIINFEKL) to ensure that lymphocyte preparations specifically express IFNγ upon ppins-specific stimulation in vitro40. Frequencies of IFNγ+ CD8+ T cells were determined by flow cytometry (FCM). The mean % of IFNγ+ CD8+ T cells in the splenic CD8+ T cell population (+SD) of a representative experiment (out of two experiments performed) is shown. Statistically significant differences between the groups 2 and 4 were determined using the unpaired student’s t-test (ns, not significant). (d) Insulin expression and CD8+ T cell influx into pancreata of representative healthy, pCI/ppinsΔA12-21 vaccinated/ppins-primed (lower panel) and diabetic, pCI/ppins-primed PD-L1−/− mice (upper panel) were analysed by histology and hematoxylin/eosin (H&E) staining (see Supplementary Protocols).
We detected comparable frequencies of IFNγ+ Kb/A12-21-specific CD8+ T cells in the spleens of ppins-primed and ppinsΔA12-21-vaccinated/ppins-primed PD-L1−/− mice (Fig. 2c; groups 2 and 4) (see Supplementary Protocols). IFNγ+ Kb/A12-21-specific CD8+ T cells were not detectable in non-immunized or pCI/ppinsΔA12-21-vaccinated mice (Fig. 2c; groups 1 and 3). None of the lymphocyte preparations were stimulated by the control Kb/Ova257-264 (SIINFEKL) peptide (Fig. 2c; groups 1–4), confirming the ppins-specificity of the IFNγ response. The pCI/ppinsΔA12-21-induced systemic immunity thus did not affect de novo priming of autoreactive IFNγ+ Kb/A12-21-specific CD8+ T cells by pCI/ppins. In contrast, we detected a progressive influx of CD8+ T cells into the pancreatic target tissue and beta cell destruction in diabetic, ppins-primed PD-L1−/− mice38, but not in healthy, ppinsΔA12-21-vaccinated/ppins-primed PD-L1−/− mice (Fig. 2d). These findings suggested that vaccine-induced immune responses impaired trafficking of de novo primed Kb/A12-21-specific effector CD8+ T cells to the pancreatic target tissue and/or eliminated (or silenced) them in the pancreas39. The low affinity A12-21 epitope or presentation-optimized peptide variants (i.e., the A12-N21A peptide) inefficiently bound Kb-molecules and we could not generate epitope-specific Kb-dimers or tetramers40. Therefore, we were not able to track and characterize this critical CD8+ T cell population in vaccinated mice.
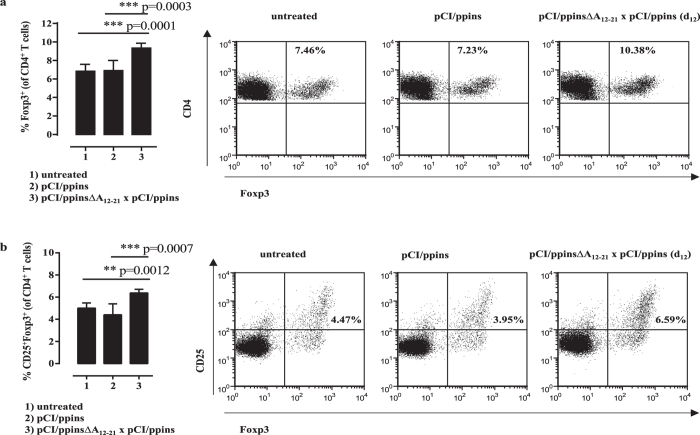
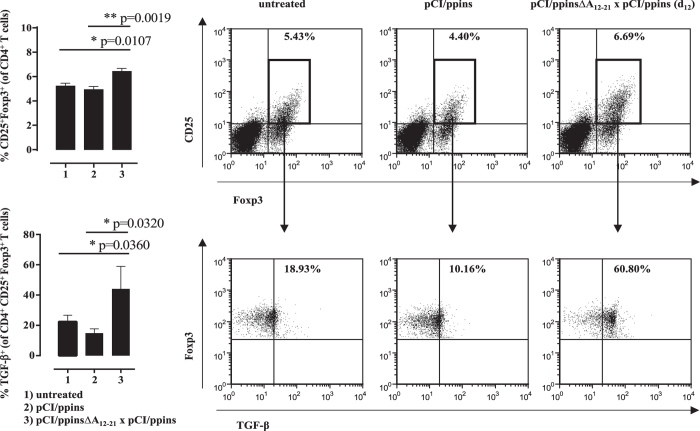
Key drivers of the pCI/ppinsΔA12-21-induced protective immunity could be regulatory Foxp3+ CD25+ CD4+ Treg cells that eliminate or reprogram autoreactive CD8+ T cells13. We detected significant higher numbers of Foxp3+ and Foxp3+ CD25+ Treg cells in the splenic CD4+ T cell population of ppinsΔA12-21-vaccinated/ppins-primed PD-L1−/− mice, than in pCI/ppins-immune or untreated mice (Fig. 3a,b). Furthermore, transforming growth factor beta (TGF-β) expression was augmented selectively in the Foxp3+ CD25+ Treg cell population of vaccinated/ppins-primed mice after non-specific stimulation with PMA/ionomycin (see Supplementary Protocols; Fig. 4). This indicated that TGF-β producing Treg cells could play a crucial role in the control of pCI/ppins-primed autoreactive effector CD8+ T cells and autoimmune diabetes13,41,42.
Figure 3. The ppins∆A12-21 vaccine induced Foxp3+ CD25+ Treg cells.
Splenic lymphocytes of untreated (group 1, n = 3), pCI/ppins-primed (group 2, n = 3) or ppins∆A12-21-vaccinated/ppins-primed (group 3, n = 3) PD-L1−/− mice were isolated 20 days after the injection of pCI/ppins, stained for surface CD4 and CD25 and intracellular Foxp3 expression and analysed by FCM. The actual percentage of Foxp3+ (a) and Foxp3+ CD25+ (b) Treg cells in gated CD4+ T cell populations ± SD of a representative experiment (out of two experiments performed) is shown (left panels). Statistically significant differences between the group 1 and group 3 or group 2 and group 3 were determined using the unpaired student’s t-test. P values of <0.05 (*), <0.01 (**) and <0.001 (***) were considered statistically significant. As example for the underlying flow cytometric analysis, Dot Plots from a representative mouse per group are shown (right panels).
Figure 4. The ppins∆A12-21 vaccine specifically induced TGF-β expressing Foxp3+ CD25+ Treg cells.
Splenic lymphocytes of untreated (group 1, n = 3), pCI/ppins-primed (group 2, n = 3) or ppins∆A12-21-vaccinated/ppins-primed (group 3, n = 3) PD-L1−/− mice were isolated 21 days after the injection of pCI/ppins. Spleen cells were treated with PMA/ionomycin (see Supplementary Protocols), followed by surface CD4 and CD25 staining and intracellular Foxp3 and TGF-β staining. The actual percentage of Foxp3+ CD25+ Treg cells in gated CD4+ T cell populations ± SD is shown (upper panels). Dot Plots from a representative mouse per group are shown. Boxed areas represent the gates set for analyzing TGF-β expression in Foxp3+ CD25+ Treg cells (lower panels). Statistically significant differences between the group 1 and group 3 or group 2 and group 3 were determined using the unpaired student’s t-test. P values of <0.05 (*) and <0.01 (**) were considered statistically significant.
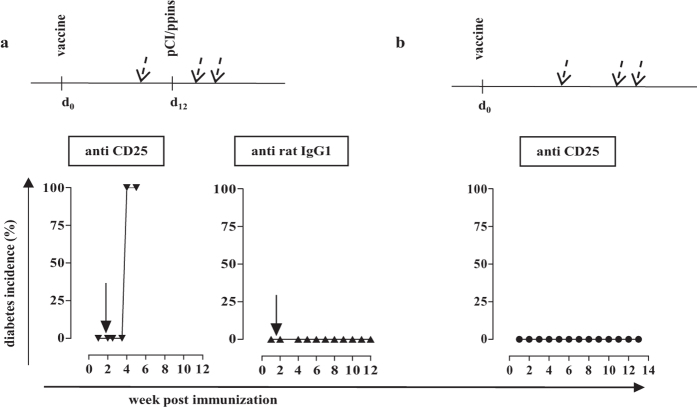
Most interestingly, acute depletion of Foxp3+ CD25+ Treg cells by anti-CD25 (PC61) antibody injections (Supplementary Fig. S4), but not isotype antibody injections, into vaccinated/ppins-primed PD-L1−/− mice resulted in diabetes development (Fig. 5a). Similarly, anti-CD25 antibody treatment of vaccinated/ppins-primed PD-1−/− mice resulted in diabetes development (Supplementary Fig. S1d). These findings showed that ablation of Treg cells specifically abolished the protective effect of the pCI/ppinsΔA12-21-vaccine and enabled diabetes induction by the diabetogenic pCI/ppins vector. Control experiments showed that depletion of Treg cells by anti-CD25 antibody treatment per se did not induce autoimmune diabetes in ppinsΔA12-21-immune PD-L1−/− mice (Fig. 5b). Furthermore, anti-CD25 treatment of mice had no measurable impact on the priming of CD8+ T cells by DNA-based immunization. Comparable frequencies of IFNγ+ Kb/A12-21-specific CD8+ T cells were induced in non-treated or anti-CD25 treated vaccinated/ppins-primed mice (Supplementary Fig. S5a,b). Similarly, anti-CD25 antibody treatment did not affect the priming of HBV core/(Kb/C93-100)-specific CD8+ T cells by pCI/C (Supplementary Fig. S5c,d).
Figure 5. Ablation of Treg cells by anti-CD25 antibody abolished the protective effect of the pCI/ppinsΔA12-21-vaccine.
(a) PD-L1−/− mice (n = 4) were immunized with pCI/ppinsΔA12-21 (d0) followed by an injection of pCI/ppins after 12 days. Furthermore, vaccinated/ppins-primed mice were treated three times (at day 3 before and days 3 and 6 after the injection of pCI/ppins; dashed arrows) with 120 μg anti-CD25 mAb PC61 (left panel) or 120 μg rat IgG1 κ isotype control (right panel). (b) PD-L1−/− mice (n = 4) were vaccinated with pCI/ppinsΔA12-21 and treated at the same days with anti CD25 mAb as described above, but without the injection of the pCI/ppins vector. Arrows indicate the injection of the diabetogenic pCI/ppins DNA at day 12 post vaccination. Blood glucose levels were measured and cumulative diabetes incidences (%) were determined by regular blood glucose measurements.
Vaccine-induced CD4+ Treg cells suppressed diabetes induction in adoptively transferred hosts
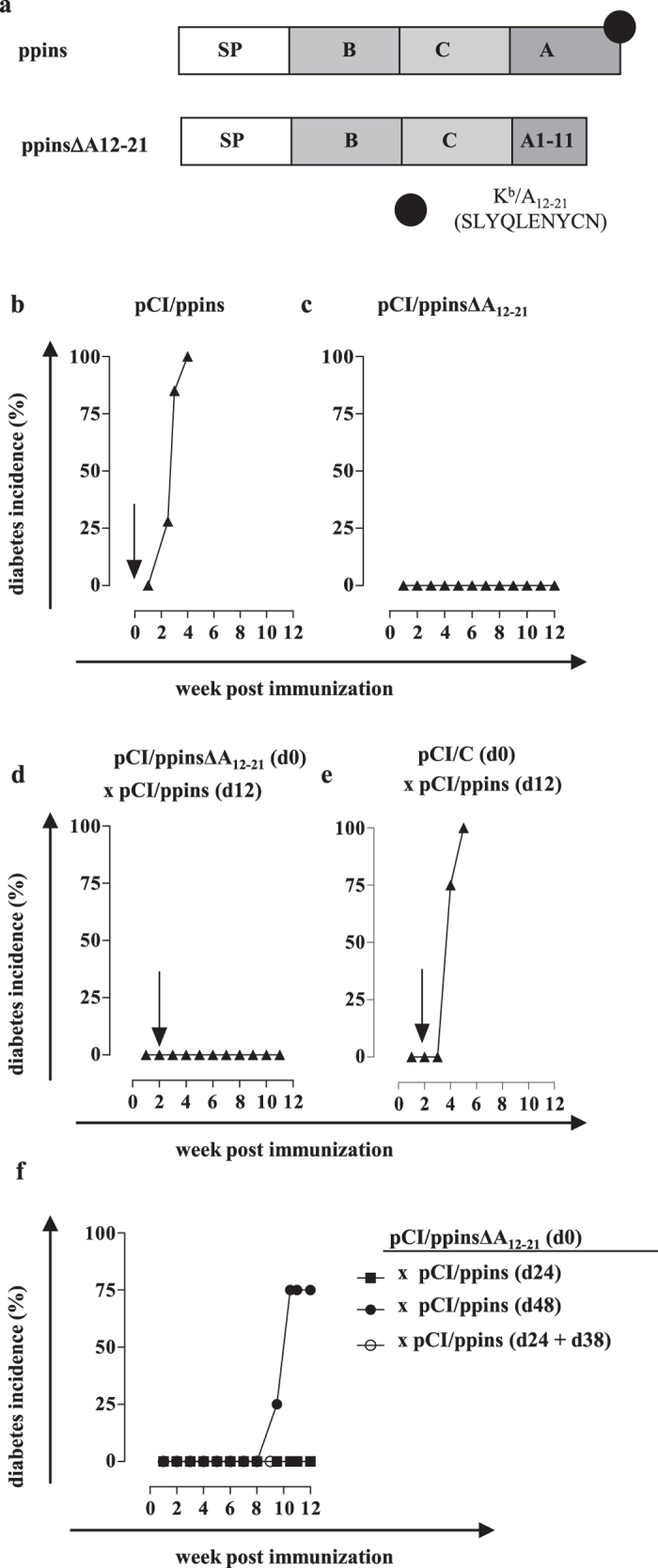
To confirm that vaccine-induced Treg cells are functional in vivo, we performed adoptive T cell transfer experiments. CD4+ or CD4+ CD25+ T cells were purified from spleen cell preparations using specific magnetic assisted cell sorting (MACS) kits, respectively (see Supplementary Protocols). Purified CD4+ T cells (3 × 106 cells) isolated from vaccinated/ppins-primed PD-L1−/− mice (containing the total pool of CD4+ T- and Treg cells) were injected intravenously into PD-L1−/− hosts followed by the injection of the diabetogenic pCI/ppins vector after 1 day. None of these mice developed diabetes (Fig. 6a). Similarly, the transfer of 3.5 × 105 purified CD25+ CD4+ T cells (containing largely Treg cells, see Fig. 3b) from vaccinated/ppins-primed, but not from non-immunized donor mice into PD-L1−/− hosts one week after the injection of the diabetogenic pCI/ppins DNA efficiently suppressed diabetes induction (Fig. 6b). Vaccine-induced Treg cells thus efficiently suppressed diabetes induction by an ongoing insulin-reactive CD8+ T cell response.
Figure 6. Adoptive transfer of vaccine-induced CD4+ T cells into ppins-immune hosts.
(a) PD-L1−/− mice were immunized with pCI/ppinsΔA12-21 (d0) followed by an injection of pCI/ppins after 12 days. After three weeks, splenic CD4+ T cells were isolated, magnetically sorted (see Supplementary Protocols) and injected (3 × 106 cells/mouse) intravenously into PD-L1−/− hosts (n = 3) one day prior to the injection of the diabetogenic pCI/ppins. (b) CD25+ CD4+ T cells were sorted from ppinsΔA12-21-vaccinated/ppins-primed (see above) or untreated PD-L1−/− mice (see Supplementary Protocols) and injected (3.5 × 105 cells) into PD-L1−/− hosts (n = 3) seven days after the immunization with the diabetogenic pCI/ppins vector. Arrows and dashed arrows indicate the injections of pCI/ppins and purified T cells, respectively. Blood glucose levels were measured and cumulative diabetes incidences (%) were determined by regular blood glucose measurements.
Narrowing down the Treg cell-stimulating ppins domain
In H-2b mice, antigen-specific CD4+ Treg cells recognize and respond to epitopes presented on the cell surface of APCs by I-Ab MHC class II molecules. To narrow down the Treg-stimulating domain(s) on the ppinsΔA12-21 antigen, we generated vectors expressing overlapping sequences of this protein (Fig. 7a): a pCI/ppins1–36 vector (encoding the ER-targeting signal peptide up to aa 12 of the B-chain), a pCI/ppins15–66 vector (encoding a sequence from aa 15 of the SP up to aa 10 of the C-chain) and a pCI/ppins1–89 vector (encoding the ER-targeting SP up to the NH2-terminus of the A-chain). Injection of pCI/ppins1–89, but not pCI/ppins1–36 or pCI/ppins15–66 vaccines suppressed diabetes development after an injection (at d12 post vaccination) of the diabetogenic pCI/ppins into PD-L1−/− mice (Fig. 7b–d), indicating that the Treg-stimulating domain is localized around ppins67–89. In line with this finding, a 25-residue ppins75–99 fragment (encoding aa 19 of the C-peptide up to aa 10 of the A-chain; C19-A10), expressed as a chimeric fusion antigen with the HBV core antigen (pCI/Core-ppins75–99) (Fig. 7a; Supplementary Fig. S6), induced a prophylactic immunity in vaccinated PD-L1−/− mice (Fig. 7e). Vaccination of PD-L1−/− mice with the pCI/Core-ppins75–99 but not the pCI/C vector (expressing the HBV core without the ppins75–99 insert) efficiently suppressed CD8+ T cell-mediated diabetes induction by pCI/ppins (Figs 1e and 7e). Using the particle-forming HBV core antigen as delivery vehicle for the ppins75–99 fragment allowed us to produce recombinant chimeric protein particles43 and target directly the exogenous processing pathway for MHC class II presentation and CD4+ T cell activation28. We produced recombinant rCore-ppins75–99 particles in transiently transfected HEK-293 cells (Supplementary Fig. S6). Vaccination of PD-L1−/− mice with rCore-ppins75–99 particles protected them from pCI/ppins-induced diabetes development (Fig. 7f). Endogenous (DNA) and exogenous (protein) ppins75–99 vaccines thus induced a protective immunity in PD-L1−/− mice.
Figure 7. Narrowing down a Treg cell-stimulating domain on the ppinsΔA12-21 antigen.
(a) Aminoacid sequences of ppinsΔA12-21, ppins1–36, ppins15–66, ppins1–89 and ppins75–99 are shown. PD-L1−/− mice were vaccinated with pCI/ppins1–36 (b) (n = 3), pCI/ppins15–66 (c) (n = 3) or pCI/ppins1–89 (d) (n = 4) followed by an injection of the diabetogenic pCI/ppins vector after 12 days. (e,f) PD-1−/− mice (n = 4) were vaccinated (at day 0 and day 17) with pCI/Core-ppins75–99 encoding a chimeric fusion protein of the HBV Core antigen and ppins77–99 (e) or recombinant rCore-ppins75–99 particles adsorbed to alum (f) followed by an injection of the diabetogenic pCI/ppins DNA. Arrows indicate the injection of the diabetogenic pCI/ppins DNA. Cumulative diabetes incidences (%) were determined by regular blood glucose measurements.
Induced iTreg cells specifically arise from conventional CD4+ T cells acquiring Foxp3 and CD25 expression upon antigen stimulation13. We used B6-Foxp3eGFP mice that co-express the regulatory T cell-specific transcription factor Foxp3 and the enhanced green fluorescent reporter protein (eGFP) primarily in CD4+ T cells44 to map the Treg-stimulating motif on the ppins75–99 fragment in vitro. We isolated conventional Foxp3negative/eGFPnegative CD4+ T cells by magnetic assisted (MACS) and fluorescence assisted (FACS) cell sorting, stimulated them with CD3-depleted autologous splenocytes pulsed with the overlapping 15-residue ppins peptides (Fig. 8a) and determined the actual conversion into Foxp3+/eGFP+ CD25+ CD4+ Treg cells (see Supplementary Protocols) (Fig. 8b). The ppins76–90, but not the ppins78–92, ppins80–94, ppins82–96, ppins84–98 and I-Ab-binding control peptides induced Foxp3+/eGFP+ CD25+ CD4+ Treg cells (Fig. 8b). This showed that the ppins76–90 (C19-A1) peptide contains the Treg cell-stimulating motif.
Figure 8. Identification of a Treg-stimulating ppins76–90 peptide.
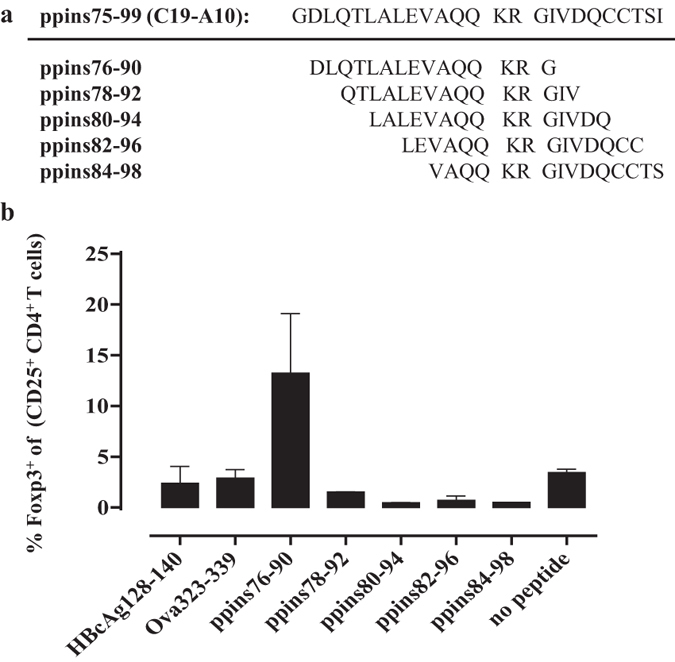
(a) Aminoacid sequences of the ppins75–99 (C19-A1) fragment and overlapping 15-residue peptides ppins76–90, ppins78–92, ppins80–94, ppins82–96 and ppins84–98. (b) Conventional Foxp3negative/eGFPnegative CD4+ T cells were isolated from spleens of B6-Foxp3eGFP mice using magnetic assisted (MACS) and fluorescence assisted (FACS) cell sorting and stimulated for three days with CD3-depleted autologous splenocytes pulsed with the respective ppins-derived peptides as well as two I-Ab-binding control peptides (HBcAg128-140 and Ova323-339). In vitro conversion of Foxp3negative/eGFPnegative CD4+ T cells into Foxp3+/eGFP+ CD25+ CD4+ T cells was determined by FCM (see Supplementary Protocols). The actual percentages of newly arising Foxp3 expressing CD25+ CD4+ Treg cells ± SD of a representative experiment (out of two experiments performed) are shown.
Discussion
Clinical manifestation of T1D is preceded by the development of autoantibodies to different islet antigens, marking the loss of immunological tolerance to beta cell antigens and an initial destruction of beta cells45,46. To date attempts to prevent T1D in individuals with HLA genotypes and autoantibodies conferring increased risk of disease were unsuccessful9. Therefore, specifically combating the initial autoreactive immune responses before seroconversion by antigen-specific immunotherapies is an attractive approach for preventing or redirecting pathogenic autoimmune reponses9,47. An optimal approach for the prevention of T1D could include an antigen-specific strategy to induce Foxp3+ CD25+ CD4+ Treg cells8. In this study, we used the PD-1/PD-L1 mouse model to explore systematically the induction of ppins-specific Treg cells by DNA vaccination. We showed that a ppinsΔA12-21 antigen (lacking the critical Kb/A12-21 epitope) primed regulatory T cells with a TGF-β+ Foxp3+ CD25+ CD4+ signature and efficiently suppressed CD8+ T cell-mediated diabetes development by a subsequent injection of the diabetogenic pCI/ppins vector.
Ablation of Treg cells in PD-1−/− and PD-L1−/− mice by anti-CD25 (PC61) antibody injections specifically abolished the protective effect of the vaccines and enabled diabetes induction by the diabetogenic pCI/ppins vector. The clone PC61 has been widely used to examine the function of Foxp3+ Treg cells in different murine model systems48. The abrogation of the Treg-specific suppressor activity with PC61 mAb could be explained by the actual deletion of CD25+ Tregs (Supplementary Fig. S5) and/or by the functional inactivation of these cells48. CD25 is expressed on Foxp3+ Treg cells but also on activated effector T cells. It is unlikely that anti-CD25 treatment (at the indicated intervals and dose used in this study) has an impact on the priming of CD8+ T cell responses by DNA-based immunization. We showed that the treatment of ppinsΔA12-21-vaccinated/ppins-primed mice or pCI/C-primed mice with PC61 did not influence de novo priming of ppins (Kb/A12-21)- or HBV core (Kb/C93-100)-specific CD8+ T cell responses, respectively. Furthermore, the time-course as well as diabetes incidence in anti CD25 treated vaccinated/ppins-primed mice resembled pCI/ppins-induced diabetes in both, PD-L1−/− (Figs 1b and 5a) and PD-1−/− mice (Supplementary Fig. S1a,d). This suggested that vaccine-induced Foxp3+ CD25+ Treg cells play a major role in silencing the pathogenic effector functions of de novo primed IFNγ+ Kb/A12-21-specific CD8+ T cells in PD-1−/− and PD-L1−/− mice. The mechanism(s) of Treg-mediated suppression of autoreactive Kb/A12-21-specific effector CD8+ T cells and diabetes development are unknown. Treg cells suppress effector T cell responses through a variety of mechanisms13. Recent studies have defined the cytokine TGF-β as a crucial player in peripheral T cell homeostasis, immune tolerance to self antigens and T cell differentiation during immune responses41. In particular, Foxp3+ Treg cells utilize TGF-β to suppress immune responses but also to facilitate the conversion of naïve T cells into Treg cells and/or to protect themselves against apoptosis41,42. We here showed that TGF-β expression was augmented selectively in the Foxp3+ CD25+ Treg cell population of vaccinated/ppins-primed mice (Fig. 4), providing evidence that TGF-β producing Treg cells could play a crucial role in the suppression of de novo primed autoreactive effector CD8+ T cells and autoimmune diabetes. Vaccine-induced Treg cells may exert their suppressor activities in the pancreas. It has been shown that DNA-based vaccination with an insulin B-chain-expressing vector significantly reduced the incidence of diabetes in transgenic mice that express the nucleoprotein of LCMV (LCMV-NP) in beta cells and are infected with LCMV. The insulin B-chain vaccination was effective through induction of Treg cells that react with the insulin B chain, secrete IL-4, and locally reduce the activity of LCMV-NP-specific autoreactive CD8+ T cells in the pancreatic draining lymph nodes49. Furthermore, Treg cells primarily impinge on autoimmune diabetes by eliminating pathogenic T cells inside the islets39.
Co-inhibitory PD-1/PD-L1 signals may directly affect induction and function of autoantigen-specific Foxp3+ CD25+ CD4+ Treg cells20,21,22. PD-L1-deficient APCs inefficiently convert conventional CD4+ T cells into Foxp3+ CD25+ iTreg cells, indicating that PD-L1 itself has an impact on iTreg development. Furthermore, culturing iTregs with PD-L1-coated beads enhanced and sustained Foxp3 expression as well as the suppressive function of iTreg cells21. We here showed that ppins-specific vaccines elicited functional Treg cell responses that suppressed CD8+ T cell-mediated diabetes development in PD-1- or PD-L1-deficient mice. This indicated that ppins-specific Treg cells were efficiently induced in the absence of the PD-1/PD-L1 signaling pathway.
DNA- (endogenous) and protein-based (exogenous) vaccines efficiently induced ppins-specific Treg cells and suppressed diabetes development by a subsequent injection of the diabetogenic pCI/ppins vector (see Fig. 7e,f). Exogenous antigens preferentially stimulate CD4+ T cell responses, because they are specifically processed and loaded on MHC class II molecules in the exogenous processing pathway28. However, little is known how endogenously expressed antigens stimulate CD4+ T cells. Endogenously expressed antigens could be directly processed for MHC class II presentation in alternative intracellular pathways28. Furthermore, ‘cross-presentation’ of antigenic material that is released from non-professional antigen-expressing cells (e.g., myocytes) to professional APCs (DCs) may facilitate priming of CD4+ Treg cell responses by DNA vaccines. MHC class II-binding peptides are usually 10–20 residues long. We narrowed down the Treg cell stimulating ppins-domain in vivo to a 25-residue ppins75–99 (C18-A10) fragment by vectors expressing ppins fragments (Fig. 7) and in vitro to a 15-residue ppins76–90 (C19-A1) fragment that specifically stimulated the conversion of conventional Foxp3negative/eGFPnegative CD4+ T cells into Foxp3+/eGFP+ CD25+ CD4+ Treg cells (Fig. 8). Interestingly, the ppins75–99 (C18-A10) domain also contained overlapping (nested) immunodominant HLA-DRB*0401-restricted epitopes (i.e., C13-C32, C19-A3 and C22-A5)50, indicating that this domain is efficiently processed for MHC class II epitope presentation. Structural features and/or intrinsic expression of ppins designer antigens could affect processing and MHC class I- and class II- epitope presentation40,51. We previously showed that the expression of mutant ppinsΔA12-21 and ppins differed substantially in transiently transfected HEK-293 cells38. Both, ppins and ppinsΔA12-21 contain the ppins signal peptide (SP) (Fig. 1a) that targets the proteins into the Endoplasmic Reticulum (ER), where the SP is removed by ER-resident signal peptidases. However, the expression levels ppinsΔA12-21 were weaker than that of ppins and treatment of transfectants with the proteasome inhibitors epoxomicin or lactacystin efficiently restored ppinsΔA12-21 levels38. In contrast, the expression of ppins in transiently transfected HEK-293 cells was not changed by proteasome inhibitors40. This indicated that ppinsΔA12-21, but not ppins was efficiently processed by proteasomal degradation. The altered endogenous antigen expression and processing of mutant ppins proteins may thus facilitate MHC class II epitope presentation and Treg cell priming by DNA vaccination.
Vaccines against self-proteins contain a non-predictable risk to induce or stimulate autoreactive T cell responses rather than a protective immunity in individual recipients. Factors like MHC I and II composition or genetic factors, but also antigen expression and processing could influence the priming of immune responses40,51. We here showed that the ppinsΔA12-21 antigen (lacking the dominant Kb/A12-21 epitope) induced Treg cells in PD-L1−/− and PD-1−/− mice. In contrast, the pCI/ppinsΔA12-21 vaccine elicited insulin B-chain-specific Kb/B22-29-specific CD8+ T cells and autoimmune diabetes in RIP-B7.1 tg mice expressing the co-stimulator molecule B7.1 (CD80) in beta cells38. This was unexpected, because the pCI/ppins vector did not induce Kb/B22-29-specific CD8+ T cells in RIP-B7.1 tg mice and the pCI/ppinsΔA12-21 vector did not induce Kb/B22-29-specific CD8+ T cells in PD-L1−/− or PD-1−/− mice37,38. Deletion of the A12-21 sequence may generate a secondary antigen that, in contast to ppins, is efficiently processed for Kb/B22-29-specific epitope presentation. Priming and/or expansion of CD8+ T cells specific for this epitope required co-stimulatory ‘help’ from transgenic B7.1-expressing beta cells. Similarly, a proinsulin-expressing pCI/pins vector inefficiently induced late autoimmune diabetes in RIP-B7.1 tg mice40. Therefore, RIP-B7.1 tg mice can be used to test ppins antigens if they contain a residual risk to induce autoreactive T cell responses and diabetes. Using this mouse model, we previously showed that the expression of ppins designer antigens in the ER by homologous (SP; Fig. 1a) or heterologous ER targeting signal peptides (e.g., derived from the murine Igκ chain) was crucial to induce Kb/A12-21-specific effector CD8+ T cells and autoimmune diabetes, indicating that direct expression and processing of ppins antigens in the ER favors MHC class I presentation of the ‘weak’ Kb/A12-21 epitope40. Designer antigens without these signal sequences that are stable expressed in the cytosol and/or the nucleus, for example, by fusing the ppins sequence COOH-terminally to the green fluorescent protein, did not induce Kb/A12-21-specific effector CD8+ T cells and autoimmune diabetes in RIP-B7.1 tg mice40. In ongoing experiments, we analyse whether ppins designer antigens that prevent antigen expression, processing and/or presentation in the ER could be a general strategy to induce a prophylactic Treg cell-mediated immunity in mice expressing different MHC haplotypes.
Methods
Mice
C57BL/6 (B6) mice (Janvier; Le Genets-St-Isle; France), B6-Foxp3eGFP mice (B6.Cg-Foxp3tm2(EGFP)Tch/J; Jackson # 006772), PD-1−/− mice36 and PD-L1−/− (B7-H1−/−) mice35 and were bred and kept under standard pathogen-free conditions in the animal colony of Ulm University (Ulm, Germany). All mouse immunization studies were carried out in strict accordance with the recommendations in the Guide for the Care and Use of Laboratory Animals of the German Federal Animal Protection Law. The protocols were approved by the Committee on the Ethics of Animal Experiments of the University of Ulm (Tierforschungszentrum Ulm, Oberberghof) and the Regierungspräsidium Tübingen (Permit Numbers: 1105 and 1199 to RS). All studies were carried out in accordance with the approved guidelines. Immunizations were performed under short time Isofluran anesthesia, and all efforts were made to minimize suffering. Development of autoimmune diabetes was analysed by regular blood glucose measurements and diagnosed if two consecutive blood glucose values (within 2 days) exceeded 250 mg/dl, i.e. 13.8 mmol/l (Disetronic Freestyle, Sulzbach, Germany).
Construction of expression vectors
The antigenic sequences of the different ppins antigens were codon-optimized and synthesized by GeneArt (Regensburg, Germany). All constructs were cloned into the pCI vector (cat. no. E1731, Promega, Mannheim, Germany) using the NheI and NotI restriction sites. Batches of DNA were produced in E. coli using the Qiagen Plasmid Mega Kit (cat. no. 12183; Qiagen, Hilden, Germany). Where indicated, antigens were modified with a NH2-terminal Strep-tag (st) sequence to purify recombinant proteins from lysates of transiently transfected HEK-293 cells as described previously52. HEK-293 cells were used, because they can be transfected with high efficacy (≥90%) using the calcium phosphate method and express high levels of vector-encoded antigens52.
Immunization of mice
Mice were immunized into the tibialis anterior muscles with 100 μg/mouse of plasmid DNA or 10 μg/mouse of recombinant antigens adsorbed to alum (Alhydrogel “85”, Brenntag Biosector, Frederikssund, Denmark). Where indicated, mice were treated with blocking PD-L1 (B7-H1) antibody (clone MIH5, cat. no. 16-5982-85, eBioscience, Frankfurt, Germany) or with anti-CD25 antibody PC61 (ImmunoTools GmbH, Friesoythe, Germany).
Statistical analysis
Data were analysed using PRISM software (GraphPad, San Diego, CA, USA). The statistical significance of differences in the mean T cell frequencies between groups was determined by the unpaired student’s t-test. A value of (*)p < 0.05 was considered significant (**significant at p < 0.01, ***significant at p < 0.001).
Additional Information
How to cite this article: Stifter, K. et al. Exploring the induction of preproinsulin-specific Foxp3+ CD4+ Treg cells that inhibit CD8+ T cell-mediated autoimmune diabetes by DNA vaccination. Sci. Rep. 6, 29419; doi: 10.1038/srep29419 (2016).
Supplementary Material
Acknowledgments
We thank Ellen Allmendinger and Andrea Wissmann for outstanding technical assistance. We thank Dr. T. Honjo (Department of Immunology and Genomic Medicine, Kyoto University, Kyoto, Japan) for PD-1−/− mice and Dr. Lieping Chen (Department of Oncology, Johns Hopkins University School of Medicine, Baltimore, Maryland, USA) for PD-L1 (B7-H1)−/− mice. We thank Dr. Erik Lindblad (Brenntag Biosector, Frederikssund, Denmark) for providing Alhydrogel. This work was supported by grants from the Deutsche Forschungsgemeinschaft: GRK-1789 ‘CEMMA’ and DFG SCHI-505/4-1 to R.S.
Footnotes
Author Contributions K.S., C.S., B.O.B. and R.S. designed the research K.S., C.S. and M.S. performed experiments K.S. and R.S. wrote the manuscript B.O.B. reviewed/edited the manuscript.
References
- Santamaria P. The long and winding road to understanding and conquering type 1 diabetes. Immunity. 32, 437–445 (2010). [DOI] [PubMed] [Google Scholar]
- Jeker L. T., Bour-Jordan H. & Bluestone J. A. Breakdown in peripheral tolerance in type 1 diabetes in mice and humans. Cold Spring Harb. Perspect. Med. 2, a007807 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pinkse G. G. et al. Autoreactive CD8 T cells associated with beta cell destruction in type 1 diabetes. Proc. Natl. Acad. Sci. USA 102, 18425–18430 (2005). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mallone R. et al. CD8+ T-cell responses identify beta-cell autoimmunity in human type 1 diabetes. Diabetes 56, 613–621 (2007). [DOI] [PubMed] [Google Scholar]
- Skowera A. et al. CTLs are targeted to kill beta cells in patients with type 1 diabetes through recognition of a glucose-regulated preproinsulin epitope. J. Clin. Invest 118, 3390–3402 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Abreu J. R. & Roep B. O. Targeting proinsulin-reactive CD8+ T cells: a new direction for type 1 diabetes treatment. Expert. Rev. Clin. Immunol. 9, 1001–1003 (2013). [DOI] [PubMed] [Google Scholar]
- Sachdeva N. et al. Preproinsulin specific CD8+ T cells in subjects with latent autoimmune diabetes show lower frequency and different pathophysiological characteristics than those with type 1 diabetes. Clin. Immunol. 157, 78–90 (2015). [DOI] [PubMed] [Google Scholar]
- Serr I., Weigmann B., Franke R. K. & Daniel C. Treg vaccination in autoimmune type 1 diabetes. BioDrugs. 28, 7–16 (2014). [DOI] [PubMed] [Google Scholar]
- Roep B. O. & Tree T. I. Immune modulation in humans: implications for type 1 diabetes mellitus. Nat. Rev. Endocrinol. 10, 229–242 (2014). [DOI] [PubMed] [Google Scholar]
- Bluestone J. A., Bour-Jordan H., Cheng M. & Anderson M. T cells in the control of organ-specific autoimmunity. J. Clin. Invest 125, 2250–2260 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Francisco L. M., Sage P. T. & Sharpe A. H. The PD-1 pathway in tolerance and autoimmunity. Immunol. Rev. 236, 219–242 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gianchecchi E., Delfino D. V. & Fierabracci A. Recent insights into the role of the PD-1/PD-L1 pathway in immunological tolerance and autoimmunity. Autoimmun. Rev. 12, 1091–1100 (2013). [DOI] [PubMed] [Google Scholar]
- Shevach E. M. & Thornton A. M. tTregs, pTregs, and iTregs: similarities and differences. Immunol. Rev. 259, 88–102 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ansari M. J. et al. The programmed death-1 (PD-1) pathway regulates autoimmune diabetes in nonobese diabetic (NOD) mice. J. Exp. Med. 198, 63–69 (2003). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fife B. T. et al. Insulin-induced remission in new-onset NOD mice is maintained by the PD-1-PD-L1 pathway. J. Exp. Med. 203, 2737–2747 (2006). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pauken K. E., Jenkins M. K., Azuma M. & Fife B. T. PD-1, but not PD-L1, expressed by islet-reactive CD4+ T cells suppresses infiltration of the pancreas during type 1 diabetes. Diabetes 62, 2859–2869 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ni R. et al. PD-1 gene haplotype is associated with the development of type 1 diabetes mellitus in Japanese children. Hum. Genet. 121, 223–232 (2007). [DOI] [PubMed] [Google Scholar]
- Fujisawa R. et al. Low programmed cell death-1 (PD-1) expression in peripheral CD4(+) T cells in Japanese patients with autoimmune type 1 diabetes. Clin. Exp. Immunol. 180, 452–457 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mellati M. et al. Anti-PD-1 and Anti-PDL-1 Monoclonal Antibodies Causing Type 1 Diabetes. Diabetes Care 38, e137–e138 (2015). [DOI] [PubMed] [Google Scholar]
- Perri V. et al. Expression of PD-1 Molecule on Regulatory T Lymphocytes in Patients with Insulin-Dependent Diabetes Mellitus. Int. J. Mol. Sci. 16, 22584–22605 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Francisco L. M. et al. PD-L1 regulates the development, maintenance, and function of induced regulatory T cells. J. Exp. Med. 206, 3015–3029 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang L. et al. Programmed death 1 ligand signaling regulates the generation of adaptive Foxp3+CD4+ regulatory T cells. Proc. Natl. Acad. Sci. USA 105, 9331–9336 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Belle T. L., Taylor P. & von Herrath M. G. Mouse Models for Type 1 Diabetes. Drug Discov. Today Dis. Models. 6, 41–45 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reed J. C. & Herold K. C. Thinking bedside at the bench: the NOD mouse model of T1DM. Nat. Rev. Endocrinol. 11, 308–314 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garren H. DNA vaccines for autoimmune diseases. Expert. Rev. Vaccines. 8, 1195–1203 (2009). [DOI] [PubMed] [Google Scholar]
- Johnson M. C., Wang B. & Tisch R. Genetic vaccination for re-establishing T-cell tolerance in type 1 diabetes. Hum. Vaccin. 7, 27–36 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neefjes J. & Sadaka C. Into the intracellular logistics of cross-presentation. Front Immunol. 3, 31 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roche P. A. & Furuta K. The ins and outs of MHC class II-mediated antigen processing and presentation. Nat. Rev. Immunol. 15, 203–216 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Solvason N. et al. Improved efficacy of a tolerizing DNA vaccine for reversal of hyperglycemia through enhancement of gene expression and localization to intracellular sites. J. Immunol. 181, 8298–8307 (2008). [DOI] [PubMed] [Google Scholar]
- Roep B. O. et al. Plasmid-encoded proinsulin preserves C-peptide while specifically reducing proinsulin-specific CD8(+) T cells in type 1 diabetes. Sci. Transl. Med. 5, 191ra82 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bot A. et al. Plasmid vaccination with insulin B chain prevents autoimmune diabetes in nonobese diabetic mice. J. Immunol. 167, 2950–2955 (2001). [DOI] [PubMed] [Google Scholar]
- Li A. F., Hough J., Henderson D. & Escher A. Co-delivery of pro-apoptotic BAX with a DNA vaccine recruits dendritic cells and promotes efficacy of autoimmune diabetes prevention in mice. Vaccine 22, 1751–1763 (2004). [DOI] [PubMed] [Google Scholar]
- Balasa B. et al. Vaccination with glutamic acid decarboxylase plasmid DNA protects mice from spontaneous autoimmune diabetes and B7/CD28 costimulation circumvents that protection. Clin. Immunol. 99, 241–252 (2001). [DOI] [PubMed] [Google Scholar]
- Karges W. et al. Induction of autoimmune diabetes through insulin (but not GAD65) DNA vaccination in nonobese diabetic and in RIP-B7.1 mice. Diabetes 51, 3237–3244 (2002). [DOI] [PubMed] [Google Scholar]
- Dong H. et al. B7-H1 determines accumulation and deletion of intrahepatic CD8(+) T lymphocytes. Immunity. 20, 327–336 (2004). [DOI] [PubMed] [Google Scholar]
- Nishimura H., Minato N., Nakano T. & Honjo T. Immunological studies on PD-1 deficient mice: implication of PD-1 as a negative regulator for B cell responses. Int. Immunol. 10, 1563–1572 (1998). [DOI] [PubMed] [Google Scholar]
- Rajasalu T. et al. Deficiency in B7-H1 (PD-L1)/PD-1 coinhibition triggers pancreatic beta cell-destruction by insulin-specific, murine CD8 T cells. Diabetes 59, 1966–1973 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schuster C., Brosi H., Stifter K. & Boehm B. O. & Schirmbeck,R. A missing PD-L1/PD-1 coinhibition regulates diabetes induction by preproinsulin-specific CD8 T-cells in an epitope-specific manner. PLoS. ONE. 8, e71746 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen Z., Herman A. E., Matos M., Mathis D. & Benoist C. Where CD4+CD25+ T reg cells impinge on autoimmune diabetes. J. Exp. Med. 202, 1387–1397 (2005). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brosi H. et al. Processing in the endoplasmic reticulum generates an epitope on the insulin A chain that stimulates diabetogenic CD8 T cell responses. J. Immunol. 183, 7187–7195 (2009). [DOI] [PubMed] [Google Scholar]
- Li M. O. & Flavell R. A. TGF-beta: a master of all T cell trades. Cell 134, 392–404 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tran D. Q. TGF-beta: the sword, the wand, and the shield of FOXP3(+) regulatory T cells. J. Mol. Cell Biol. 4, 29–37 (2012). [DOI] [PubMed] [Google Scholar]
- Whitacre D. C., Lee B. O. & Milich D. R. Use of hepadnavirus core proteins as vaccine platforms. Expert. Rev. Vaccines. 8, 1565–1573 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haribhai D. et al. Regulatory T cells dynamically control the primary immune response to foreign antigen. J. Immunol. 178, 2961–2972 (2007). [DOI] [PubMed] [Google Scholar]
- Mallone R. & Roep B. O. Biomarkers for immune intervention trials in type 1 diabetes. Clin. Immunol. 149, 286–296 (2013). [DOI] [PubMed] [Google Scholar]
- Bonifacio E. Predicting type 1 diabetes using biomarkers. Diabetes Care 38, 989–996 (2015). [DOI] [PubMed] [Google Scholar]
- Bonifacio E. et al. Effects of high-dose oral insulin on immune responses in children at high risk for type 1 diabetes: the Pre-POINT randomized clinical trial. JAMA 313, 1541–1549 (2015). [DOI] [PubMed] [Google Scholar]
- Petzold C. et al. Foxp3(+) regulatory T cells in mouse models of type 1 diabetes. J. Diabetes Res. 2013, 940710 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coon B., An L. L., Whitton J. L. & von Herrath M. G. DNA immunization to prevent autoimmune diabetes. J. Clin. Invest 104, 189–194 (1999). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gibson V. B. et al. Proinsulin multi-peptide immunotherapy induces antigen-specific regulatory T cells and limits autoimmunity in a humanized model. Clin. Exp. Immunol. 182, 251–260 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hsu H. T. et al. Endoplasmic reticulum targeting alters regulation of expression and antigen presentation of proinsulin. J. Immunol. 192, 4957–4966 (2014). [DOI] [PubMed] [Google Scholar]
- Wieland A. et al. Recombinant complexes of antigen with stress proteins are potent CD8 T-cell-stimulating immunogens. J. Mol. Med. 86, 1067–1079 (2008). [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.